Risk Factors for Infections, Antibiotic Therapy, and Its Impact on Cancer Therapy Outcomes for Patients with Solid Tumors
Abstract
1. Introduction
2. Immunosuppression in Cancer Patients
3. Immunosuppression Induced by Chemotherapy
4. Immunosuppression Induced by Targeted Therapy
5. Risk Scores for Febrile Neutropenia
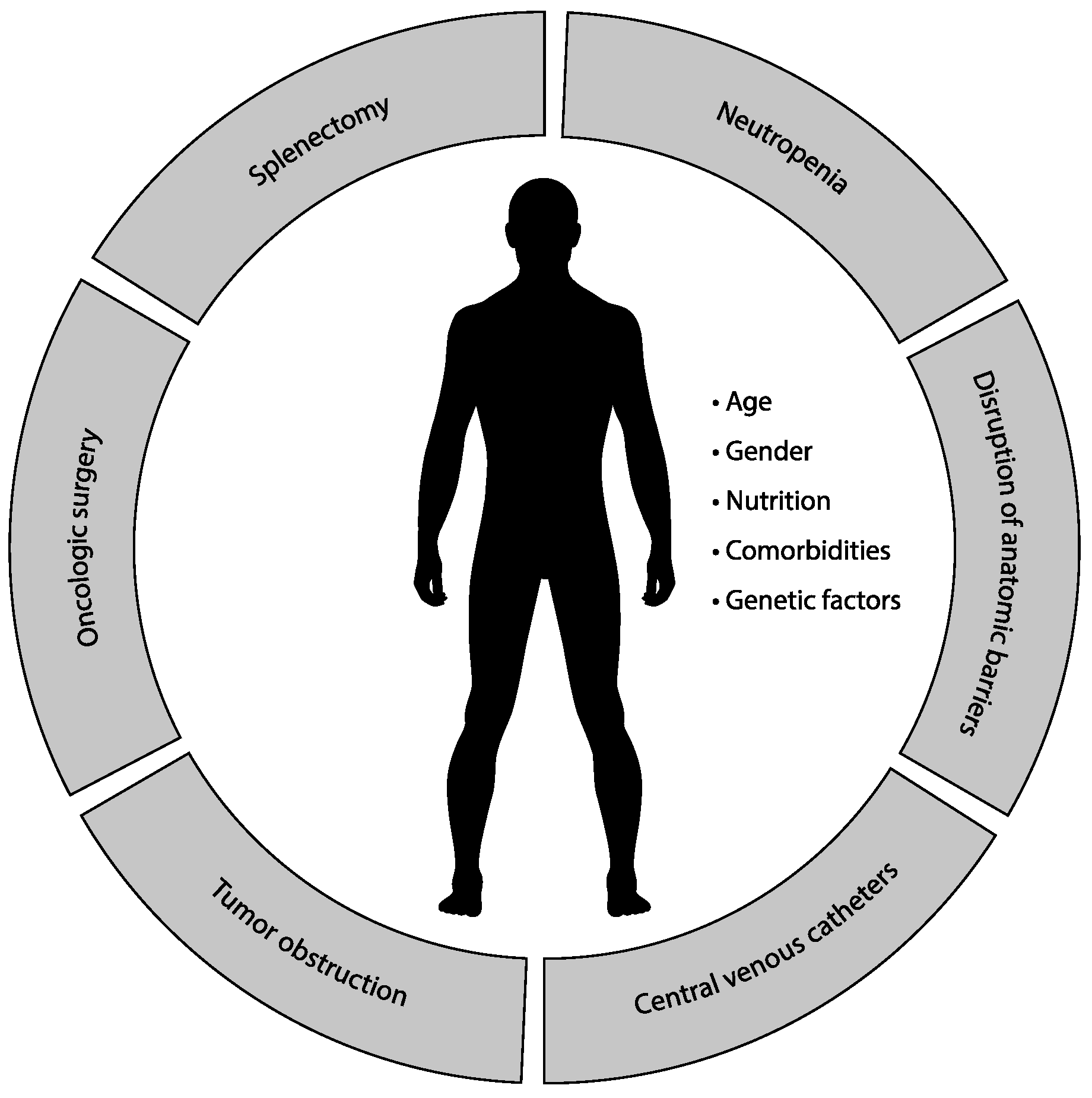
6. Risk Factors for Infections in Patients with Solid Tumors
6.1. Neutropenia
6.2. Disruption of Anatomic Barriers
6.3. Central Venous Catheters
6.4. Tumor Obstruction
6.5. Oncologic Surgery
6.6. Splenectomy and Function Asplenia
6.7. Patient-Related Factors
6.7.1. Age
6.7.2. Gender
6.7.3. Nutrition
6.7.4. Comorbidities
6.7.5. Genetic Factors
7. Antibiotic Therapy in Patients with Solid Tumors
8. Antibiotic Therapy of Specific Infections
8.1. Febrile Neutropenia
| Risk of Serious Complications | Low | High |
|---|---|---|
| Initial antibiotic | Oral or parenteral | Parenteral |
| Inpatient or outpatient | Inpatient | |
| Amoxicillin-clavulanate + fluoroquinolone (ciprofloxacin or levofloxacin) | Antipseudomonal beta-lactam * (cefepime or meropenem or imipenem or piperacillin-tazobactam) | |
| Suspicion of catheter-related infection, severe skin and soft tissue infection, pneumonia, or risk of MRSA infection | Shift to high-risk group | Add gram-positive bacteria targeted antibiotic (vancomycin or linezolid or daptomycin †), in case of VRE add linezolid or daptomycin † |
| Suspicion of abdominal infection | Shift to high-risk group | Add metronidazole |
| Risk of multiresistant strain infection | Shift to high-risk group | Choose carbapenem (in case of ESBL), add polymyxin-colistin or tigecycline (in cases of KPC) |
8.2. Central Venous Catheter-Related Infections
| Antibiotic | Spectrum of Bacteria | Concentration * (mg/mL) | Heparin Content (IU/mL) | Stability (Hours) | References |
|---|---|---|---|---|---|
| Vancomycin | gram-positive | 2.0–5.0 | 2500 or 5000 | 72 | [161,162] |
| Teicoplanin | gram-positive | 5.0–10.0 | 0 or 100 | 96 | [163,164] |
| Daptomycin | gram-positive | 5.0 | 0 or 5000 | 72 | [165] |
| Gentamicin | gram-positive, gram-negative | 1.0–5.0 | 0, 2500 or 5000 | 72 | [166,167] |
| Amikacin | gram-positive, gram-negative | 1.0–40.0 † | 0 or 5000 | 72 | [168] |
| Ceftazidime | gram-negative | 0.5–10.0 | 0 or 5000 | 48 | [167,169,170] |
| Cefazolin | Methicillin-sensitive staphylococci | 5.0–10.0 | 2500 or 5000 | 72 | [168] |
| Ciprofloxacin | gram-negative | 0.2–5.0 | 0 or 5000 | 48 | [171,172] |
| Ampicillin | Ampicillin-sensitive enterococci | 10.0 | 10 or 5000 | 8 ‡ | [161] |
| Ethanol | gram-positive, gram-negative | 70% | 0 | 24 | [173] |
8.3. Pneumonia
8.4. Intra-Abdominal Infections
8.5. Urinary Tract Infections
9. The Impact of Antibiotic Therapy on Cancer Therapy Outcomes
9.1. Impact on Cancer Therapy Efficacy
9.2. Impact on Cancer Therapy Toxicity
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gudiol, C.; Aguado, J.M.; Carratalà, J. Bloodstream infections in patients with solid tumors. Virulence 2016, 7, 298–308. [Google Scholar] [CrossRef]
- Safdar, A.; Armstrong, D. Infectious morbidity in critically ill patients with cancer. Crit. Care Clin. 2001, 17, 531–570. [Google Scholar] [CrossRef]
- Rolston, K.V.I. Infections in Cancer Patients with Solid Tumors: A Review. Infect. Dis. Ther. 2017, 6, 69–83. [Google Scholar] [CrossRef]
- Williams, M.D.; Braun, L.A.; Cooper, L.M.; Johnston, J.; Weiss, R.V.; Qualy, R.L.; Linde-Zwirble, W. Hospitalized cancer patients with severe sepsis: Analysis of incidence, mortality, and associated costs of care. Crit. Care 2004, 8, R291–R298. [Google Scholar] [CrossRef] [PubMed]
- Schelenz, S.; Nwaka, D.; Hunter, P.R. Longitudinal surveillance of bacteraemia in haematology and oncology patients at a UK cancer centre and the impact of ciprofloxacin use on antimicrobial resistance. J. Antimicrob. Chemother. 2013, 68, 1431–1438. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, Y.; Yu, K.; Yang, Y.; Wang, X.; Yang, X.; Qian, J.; Liu, Z.-X.; Wu, B. Fatal Infections among Cancer Patients: A Population-Based Study in the United States. Infect. Dis. Ther. 2021, 10, 871–895. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Prevention and Treatment of Cancer-Related Infections 1. 2021. Available online: https://www.nccn.org/professionals/physician_gls/pdf/infections.pdf (accessed on 21 September 2021).
- Marin, M.; Gudiol, C.; Ardanuy, C.; Garcia-Vidal, C.; Calvo, M.; Arnan, M.; Carratalà, J. Bloodstream infections in neutropenic patients with cancer: Differences between patients with haematological malignancies and solid tumours. J. Infect. 2014, 69, 417–423. [Google Scholar] [CrossRef]
- Fillatre, P.; Decaux, O.; Jouneau, S.; Revest, M.; Gacouin, A.; Robert-Gangneux, F.; Fresnel, A.; Guiguen, C.; Le Tulzo, Y.; Jégo, P.; et al. Incidence of Pneumocystis jiroveci Pneumonia among Groups at Risk in HIV-negative Patients. Am. J. Med. 2014, 127, 1242.e11–1242.e17. [Google Scholar] [CrossRef] [PubMed]
- Klastersky, J.; de Naurois, J.; Rolston, K.; Rapoport, B.; Maschmeyer, G.; Aapro, M.; Herrstedt, J. Management of febrile neutropaenia: ESMO Clinical Practice Guidelines. Ann. Oncol. 2016, 27, v111–v118. [Google Scholar] [CrossRef] [PubMed]
- Aapro, M.; Bohlius, J.; Cameron, D.; Lago, L.D.; Donnelly, J.P.; Kearney, N.; Lyman, G.; Pettengell, R.; Tjan-Heijnen, V.; Walewski, J.; et al. 2010 update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumours. Eur. J. Cancer 2011, 47, 8–32. [Google Scholar] [CrossRef]
- De Miguel, S.C.; Calleja-Hernández, M.; Menjón-Beltrán, S.; Vallejo-Rodríguez, I. Granulocyte colony-stimulating factors as prophylaxis against febrile neutropenia. Support. Care Cancer 2015, 23, 547–559. [Google Scholar] [CrossRef] [PubMed]
- Truong, L.D.; Shen, S.S. Immunohistochemical diagnosis of renal neoplasms. Arch. Pathol. Lab. Med. 2011, 135, 92–109. [Google Scholar] [CrossRef] [PubMed]
- Perez, E.A.; Geeraerts, L.; Suman, V.J.; Adjei, A.A.; Baron, A.T.; Hatfield, A.K.; Maihle, N.; Michalak, J.C.; Kuross, S.A.; Kugler, J.W.; et al. A randomized phase II study of sequential docetaxel and doxorubicin/cyclophosphamide in patients with metastatic breast cancer. Ann. Oncol. 2002, 13, 1225–1235. [Google Scholar] [CrossRef] [PubMed]
- von Minckwitz, G.; Schneeweiss, A.; Loibl, S.; Salat, C.; Denkert, C.; Rezai, M.; Blohmer, J.U.; Jackisch, C.; Paepke, S.; Gerber, B.; et al. Neoadjuvant carboplatin in patients with triple-negative and HER2-positive early breast cancer (GeparSixto; GBG 66): A randomised phase 2 trial. Lancet Oncol. 2014, 15, 747–756. [Google Scholar] [CrossRef]
- Kosaka, Y.; Rai, Y.; Masuda, N.; Takano, T.; Saeki, T.; Nakamura, S.; Shimazaki, R.; Ito, Y.; Tokuda, Y.; Tamura, K. Phase III placebo-controlled, double-blind, randomized trial of pegfilgrastim to reduce the risk of febrile neutropenia in breast cancer patients receiving docetaxel/cyclophosphamide chemotherapy. Support. Care Cancer 2015, 23, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Gilbar, P.; McPherson, I.; Sorour, N.; Sanmugarajah, J. High incidence of febrile neutropenia following adjuvant breast chemotherapy with docetaxel, carboplatin and trastuzumab. Breast Cancer Manag. 2014, 3, 327–333. [Google Scholar] [CrossRef]
- Marty, M.; Cognetti, F.; Maraninchi, D.; Snyder, R.; Mauriac, L.; Tubiana-Hulin, M.; Chan, S.; Grimes, D.; Antón, A.; Lluch, A.; et al. Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2–Positive metastatic breast cancer administered as first-line treatment: The M77001 study group. J. Clin. Oncol. 2005, 23, 4265–4274. [Google Scholar] [CrossRef]
- Sternberg, C.N.; De Mulder, P.H.; Schornagel, J.H.; Théodore, C.; Fossa, S.D.; Van Oosterom, A.T.; Witjes, F.; Spina, M.; Van Groeningen, C.J.; De Balincourt, C.; et al. Randomized phase III trial of high-dose-intensity methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) chemo-therapy and recombinant human granulocyte colony-stimulating factor versus classic MVAC in advanced urothelial tract tumors: European Organization for Research and Treatment of Cancer Protocol no. 30924. J. Clin. Oncol. 2001, 19, 2638–2646. [Google Scholar] [CrossRef]
- Rose, P.G.; Blessing, J.A.; Gershenson, D.M.; McGehee, R. Paclitaxel and cisplatin as first-line therapy in recurrent or advanced squamous cell carcinoma of the cervix: A gynecologic oncology group study. J. Clin. Oncol. 1999, 17, 2676–2680. [Google Scholar] [CrossRef]
- Long, H.J., III; Bundy, B.N.; Grendys, E.C., Jr.; Benda, J.A.; McMeekin, D.S.; Sorosky, J.; Miller, D.; Eaton, L.A.; Fiorica, J.V. Randomized Phase III Trial of Cisplatin with or without Topotecan in Carcinoma of the Uterine Cervix: A Gynecologic Oncology Group Study. J. Clin. Oncol. 2005, 23, 4626–4633. [Google Scholar] [CrossRef]
- Van Cutsem, E.; Moiseyenko, V.; Tjulandin, S.; Majlis, A.; Constenla, M.; Boni, C.; Rodrigues, A.; Fodor, M.; Chao, Y.; Voznyi, E.; et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: A report of the v325 study group. J. Clin. Oncol. 2006, 24, 4991–4997. [Google Scholar] [CrossRef]
- Roth, A.D.; Fazio, N.; Stupp, R.; Falk, S.; Bernhard, J.; Saletti, P.; Köberle, D.; Borner, M.M.; Rufibach, K.; Maibach, R.; et al. Docetaxel, cisplatin, and fluorouracil; Docetaxel and cisplatin; and epirubicin, cisplatin, and fluorouracil as systemic treatment for advanced gastric carcinoma: A randomized phase II trial of the swiss group for clinical cancer research. J. Clin. Oncol. 2007, 25, 3217–3223. [Google Scholar] [CrossRef]
- Cunningham, D.; Starling, N.; Rao, S.; Iveson, T.; Nicolson, M.; Coxon, F.; Middleton, G.; Daniel, F.; Oates, J.; Norman, A.R. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N. Engl. J. Med. 2008, 358, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Fossa, S.D.; Kaye, S.B.; Mead, G.M.; Cullen, M.; De Wit, R.; Bodrogi, I.; Van Groeningen, C.J.; De Mulder, P.H.; Stenning, S.; Lallemand, E.; et al. Filgrastim during combination chemotherapy of patients with poor-prognosis metastatic germ cell malignancy. J. Clin. Oncol. 1998, 16, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Sheinfeld, J.; Mazumdar, M.; Bajorin, D.F.; Bosl, G.J.; Herr, H.; Lyn, P.; Vlamis, V. Etoposide and cisplatin adjuvant therapy for patients with pathologic stage II germ cell tumors. J. Clin. Oncol. 1995, 13, 2700–2704. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, M.; Tanaka, H.; Yuasa, T.; Komai, Y.; Oguchi, T.; Fujiwara, R.; Numao, N.; Yamamoto, S.; Fujii, Y.; Fukui, I.; et al. First-Line combination chemotherapy with etoposide, ifosfamide and cisplatin for the treatment of disseminated germ cell cancer: Efficacy and feasibility in current clinical practice. Int. J. Urol. 2021, 28, 920–926. [Google Scholar] [CrossRef]
- Miller, K.D.; Loehrer, P.J.; Gonin, R.; Einhorn, L.H. Salvage chemotherapy with vinblastine, ifosfamide, and cisplatin in recurrent seminoma. J. Clin. Oncol. 1997, 15, 1427–1431. [Google Scholar] [CrossRef]
- Kondagunta, G.V.; Bacik, J.; Donadio, A.; Bajorin, D.; Marion, S.; Sheinfeld, J.; Bosl, G.J.; Motzer, R.J. Combination of paclitaxel, ifosfamide, and cisplatin is an effective second-line therapy for patients with relapsed testicular germ cell tumors. J. Clin. Oncol. 2005, 23, 6549–6555. [Google Scholar] [CrossRef]
- Pointreau, Y.; Garaud, P.; Chapet, S.; Sire, C.; Tuchais, C.; Tortochaux, J.; Faivre, S.; Guerrif, S.; Alfonsi, M.; Calais, G. Randomized trial of induction chemotherapy with cisplatin and 5-fluorouracil with or without docetaxel for larynx preservation. J. Natl. Cancer Inst. 2009, 101, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Schiller, J.H.; Harrington, D.; Belani, C.; Langer, C.; Sandler, A.; Krook, J.; Zhu, J.; Johnson, D.H. Comparison of four chemotherapy regimens for advanced non–small-cell lung cancer. N. Engl. J. Med. 2002, 346, 92–98. [Google Scholar] [CrossRef]
- Pujol, J.-L.; Breton, J.-L.; Gervais, R.; Rebattu, P.; Depierre, A.; Morère, J.-F.; Milleron, B.; Debieuvre, D.; Castéra, D.; Souquet, P.-J.; et al. Gemcitabine–Docetaxel versus cisplatin–vinorelbine in advanced or metastatic non-small-cell lung cancer: A phase III study addressing the case for cisplatin. Ann. Oncol. 2005, 16, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Fossella, F.; Pereira, J.R.; Von Pawel, J.; Pluzanska, A.; Gorbounova, V.; Kaukel, E.; Mattson, K.V.; Ramlau, R.; Szczęsna, A.; Fidias, P.; et al. Randomized, Multinational, Phase III Study of Docetaxel Plus Platinum Combinations Versus Vinorelbine Plus Cisplatin for Advanced Non–Small-Cell Lung Cancer: The TAX 326 Study Group. J. Clin. Oncol. 2003, 21, 3016–3024. [Google Scholar] [CrossRef]
- Font, A.; Moyano, A.J.; Puerto, J.M.; Tres, A.; Garcia-Giron, C.; Barneto, I.; Anton, A.; Sanchez, J.J.; Salvador, A.; Rosell, R. Increasing dose intensity of cisplatin-etoposide in advanced nonsmall cell lung carcinoma. A phase III randomized trial of the spanish lung cancer group. Cancer 1999, 85, 855–863. [Google Scholar] [CrossRef]
- Cardenal, F.; López-Cabrerizo, M.P.; Antón, A.; Alberola, V.; Massuti, B.; Carrato, A.; Barneto, I.; Lomas, M.; García, M.; Lianes, P.; et al. Randomized phase III study of gemcitabine-cisplatin versus etoposide-cisplatin in the treatment of locally advanced or metastatic non-small-cell lung cancer. J. Clin. Oncol. 1999, 17, 12. [Google Scholar] [CrossRef]
- Millward, M.J.; Boyer, M.J.; Lehnert, M.; Clarke, S.; Rischin, D.; Goh, B.-C.; Wong, J.; McNeil, E.; Bishop, J.F. Docetaxel and carboplatin is an active regimen in advancednon-small-cell lung cancer: A phase II study in Caucasian and Asian patients. Ann. Oncol. 2003, 14, 449–454. [Google Scholar] [CrossRef]
- Swisher, E.M.; Mutch, D.G.; Rader, J.S.; Elbendary, A.; Herzog, T.J. Topotecan in platinum- and paclitaxel-resistant ovarian cancer. Gynecol. Oncol. 1997, 66, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Verschraegen, C.F.; Sittisomwong, T.; Kudelka, A.P.; Guedes, E.D.P.; Steger, M.; Nelson-Taylor, T.; Vincent, M.; Rogers, R.; Atkinson, E.N.; Kavanagh, J.J. Docetaxel for Patients With Paclitaxel-Resistant Müllerian Carcinoma. J. Clin. Oncol. 2000, 18, 2733–2739. [Google Scholar] [CrossRef]
- Omura, G.A.; Brady, M.F.; Look, K.Y.; Averette, H.E.; Delmore, J.E.; Long, H.J.; Wadler, S.; Spiegel, G.; Arbuck, S.G. Phase III trial of paclitaxel at two dose levels, the higher dose accompanied by filgrastim at two dose levels in platinum-pretreated epithelial ovarian cancer: An intergroup study. J. Clin. Oncol. 2003, 21, 2843–2848. [Google Scholar] [CrossRef]
- Hosein, P.J.; MacIntyre, J.; Kawamura, C.; Maldonado, J.C.; Ernani, V.; Loaiza-Bonilla, A.; Narayanan, G.; Ribeiro, A.; Portelance, L.; Merchan, J.R.; et al. A retrospective study of neoadjuvant FOLFIRINOX in unresectable or borderline-resectable locally advanced pancreatic adenocarcinoma. BMC Cancer 2012, 12, 199. [Google Scholar] [CrossRef]
- Yilmaz, U.; Anar, C.; Polat, G.; Halilcolar, H. Carboplatin plus etoposide for extensive stage small-cell lung cancer: An experience with AUC 6 doses of carboplatin. Indian J. Cancer 2011, 48, 454–459. [Google Scholar] [CrossRef]
- Von Pawel, J.; Schiller, J.H.; Shepherd, F.A.; Fields, S.Z.; Kleisbauer, J.; Chrysson, N.G.; Stewart, D.J.; Clark, P.I.; Palmer, M.C.; De Pierre, A.; et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J. Clin. Oncol. 1999, 17, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Lorigan, P.; Woll, P.; O’Brien, M.E.R.; Ashcroft, L.F.; Sampson, M.R.; Thatcher, N. Randomized phase III trial of dose-dense chemotherapy supported by whole-blood hematopoietic progenitors in better-prognosis small-cell lung cancer. J. Natl. Cancer Inst. 2005, 97, 666–674. [Google Scholar] [CrossRef] [PubMed]
- White, S.C.; Lorigan, P.; Middleton, M.R.; Anderson, H.; Valle, J.; Summers, Y.; Burt, P.A.; Arance, A.; Stout, R.; Thatcher, N. Randomized phase II study of cyclophosphamide, doxorubicin, and vincristine compared with single-agent carboplatin in patients with poor prognosis small cell lung carcinoma. Cancer 2001, 92, 601–608. [Google Scholar] [CrossRef]
- Bui, B.N.; Chevallier, B.; Chevreau, C.; Krakowski, I.; Peny, A.M.; Thyss, A.; Maugard-Louboutin, C.; Cupissol, D.; Fargeot, P.; Bonichon, F. Efficacy of lenograstim on hematologic tolerance to MAID chemotherapy in patients with advanced soft tissue sarcoma and consequences on treatment dose-intensity. J. Clin. Oncol. 1995, 13, 2629–2636. [Google Scholar] [CrossRef]
- Lorigan, P.; Verweij, J.; Papai, Z.; Rodenhuis, S.; Le Cesne, A.; Leahy, M.; Radford, J.; Van Glabbeke, M.M.; Kirkpatrick, A.; Hogendoorn, P.; et al. Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: A european organisation for research and treatment of cancer soft tissue and bone sarcoma group study. J. Clin. Oncol. 2007, 25, 3144–3150. [Google Scholar] [CrossRef] [PubMed]
- Thomson, A.W.; Turnquist, H.R.; Raimondi, G. Immunoregulatory functions of mTOR inhibition. Nat. Rev. Immunol. 2009, 9, 324–337. [Google Scholar] [CrossRef]
- Kaymakcalan, M.; Je, Y.; Sonpavde, G.; Galsky, M.; Nguyen, P.L.; Heng, D.Y.C.; Richards, C.J.; Choueiri, T.K. Risk of infections in renal cell carcinoma (RCC) and non-RCC patients treated with mammalian target of rapamycin inhibitors. Br. J. Cancer 2013, 108, 2478–2484. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, R.H.; Bechara, R.I.; Naughton, M.J.; Adachi, J.A.; Reuben, J.M. Emerging perspectives on mtor inhibitor-associated pneumonitis in breast cancer. Oncologist 2018, 23, 660–669. [Google Scholar] [CrossRef]
- Maschmeyer, G.; De Greef, J.; Mellinghoff, S.C.; Nosari, A.; Thiebaut-Bertrand, A.; Bergeron, A.; Franquet, T.; Blijlevens, N.M.A.; Maertens, J.A.; on behalf of the European Conference on Infections in Leukemia. Infections associated with immunotherapeutic and molecular targeted agents in hematology and oncology. A position paper by the European Conference on Infections in Leukemia (ECIL). Leukemia 2019, 33, 844–862. [Google Scholar] [CrossRef]
- Finn, R.S.; Aleshin, A.; Slamon, D.J. Targeting the cyclin-dependent kinases (CDK) 4/6 in estrogen receptor-positive breast cancers. Breast Cancer Res. 2016, 18, 17. [Google Scholar] [CrossRef]
- Hu, W.; Sung, T.; Jessen, B.A.; Thibault, S.; Finkelstein, M.B.; Khan, N.K.; Sacaan, A.I. Mechanistic Investigation of Bone Marrow Suppression Associated with Palbociclib and its Differentiation from Cytotoxic Chemotherapies. Clin. Cancer Res. 2016, 22, 2000–2008. [Google Scholar] [CrossRef] [PubMed]
- Gelbert, L.M.; Cai, S.; Lin, X.; Sanchez-Martinez, C.; Del Prado, M.; Lallena, M.J.; Torres, R.; Ajamie, R.T.; Wishart, G.N.; Flack, R.S.; et al. Preclinical characterization of the CDK4/6 inhibitor LY2835219: In-Vivo cell cycle-dependent/independent anti-tumor activities alone/in combination with gemcitabine. Investig. N. Drugs 2014, 32, 825–837. [Google Scholar] [CrossRef]
- Som, A.; Mandaliya, R.; Alsaadi, D.; Farshidpour, M.; Charabaty, A.; Malhotra, N.; Mattar, M.C. Immune checkpoint inhibitor-induced colitis: A comprehensive review. World J. Clin. Cases 2019, 7, 405–418. [Google Scholar] [CrossRef]
- Hosmer, W.; Malin, J.; Wong, M. Development and validation of a prediction model for the risk of developing febrile neutropenia in the first cycle of chemotherapy among elderly patients with breast, lung, colorectal, and prostate cancer. Support. Care Cancer 2011, 19, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Lyman, G.H.; Kuderer, N.M.; Crawford, J.; Wolff, D.A.; Culakova, E.; Poniewierski, M.S.; Dale, D.C. Predicting individual risk of neutropenic complications in patients receiving cancer chemotherapy. Cancer 2011, 117, 1917–1927. [Google Scholar] [CrossRef]
- Aagaard, T.; Roen, A.; Reekie, J.; Daugaard, G.; Brown, P.D.N.; Specht, L.; Sengeløv, H.; Mocroft, A.; Lundgren, J.; Helleberg, M. Development and validation of a risk score for febrile neutropenia after chemotherapy in patients with cancer: The FENCE score. JNCI Cancer Spectr. 2018, 2, pky053. [Google Scholar] [CrossRef]
- Razzaghdoust, A.; Mofid, B.; Moghadam, M. Development of a simplified multivariable model to predict neutropenic complications in cancer patients undergoing chemotherapy. Support. Care Cancer 2018, 26, 3691–3699. [Google Scholar] [CrossRef]
- Aagaard, T.; Reekie, J.; Roen, A.; Daugaard, G.; Specht, L.; Sengeløv, H.; Mocroft, A.; Lundgren, J.; Helleberg, M. Development and validation of a cycle-specific risk score for febrile neutropenia during chemotherapy cycles 2–6 in patients with solid cancers: The CSR FENCE score. Int. J. Cancer 2020, 146, 321–328. [Google Scholar] [CrossRef] [PubMed]
- NCI. Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. Available online: https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/CTCAE_4.03_2010-06-14_QuickReference_8.5x11.pdf (accessed on 22 May 2021).
- Bodey, G.P.; Buckley, M.; Sathe, Y.S.; Freireich, E.J. Quantitative relationships between circulating leukocytes and infection in patients with acute leukemia. Ann. Intern. Med. 1966, 64, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Castagnola, E.; Mikulska, M.; Viscoli, C. Prophylaxis and Empirical Therapy of Infection in Cancer Patients. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 8th ed.; Bennett, J.E., Dolin, R., Blaser, M.J., Eds.; Elsevier: Philadelphia, PA, USA, 2015; pp. 3395–3413. [Google Scholar]
- Nishimura, N.; Yamada, S.; Ueda, K.; Mishima, Y.; Yokoyama, M.; Saotome, T.; Terui, Y.; Takahashi, S.; Hatake, K.; Nishimura, M. Incidence and severity of oral mucositis induced by conventional chemotherapy: A comprehensive prospective analysis of 227 cancer patients. J. Clin. Oncol. 2010, 28, e19634. [Google Scholar] [CrossRef]
- Elting, L.S.; Chang, Y.-C.; Parelkar, P.; Boers-Doets, C.B.; Michelet, M.; Hita, G.; Rouleau, T.; Cooksley, C.; Halm, J.; Vithala, M.; et al. Risk of oral and gastrointestinal mucosal injury among patients receiving selected targeted agents: A meta-analysis. Support. Care Cancer 2013, 21, 3243–3254. [Google Scholar] [CrossRef]
- Kwitkowski, V.E.; Prowell, T.M.; Ibrahim, A.; Farrell, A.T.; Justice, R.; Mitchell, S.S.; Sridhara, R.; Pazdur, R. FDA Approval Summary: Temsirolimus as Treatment for Advanced Renal Cell Carcinoma. Oncologist 2010, 15, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Peterson, D.E.; Boers-Doets, C.; Bensadoun, R.J.; Herrstedt, J. Management of oral and gastrointestinal mucosal injury: ESMO Clinical Practice Guidelines for diagnosis, treatment, and follow-up. Ann. Oncol. 2015, 26, v139–v151. [Google Scholar] [CrossRef]
- Böll, B.; Schalk, E.; Buchheidt, D.; Hasenkamp, J.; Kiehl, M.; Kiderlen, T.R.; Kochanek, M.; Koldehoff, M.; Kostrewa, P.; Claßen, A.Y.; et al. Central venous catheter–related infections in hematology and oncology: 2020 updated guidelines on diagnosis, management, and prevention by the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Medical Oncology (DGHO). Ann. Hematol. 2021, 100, 239–259. [Google Scholar] [CrossRef]
- Taxbro, K.; Hammarskjöld, F.; Thelin, B.; Lewin, F.; Hagman, H.; Hanberger, H.; Berg, S. Clinical impact of peripherally inserted central catheters vs implanted port catheters in patients with cancer: An open-label, randomised, two-centre trial. Br. J. Anaesth. 2019, 122, 734–741. [Google Scholar] [CrossRef]
- Pu, Y.-L.; Li, Z.-S.; Zhi, X.-X.; Shi, Y.-A.; Meng, A.-F.; Cheng, F.; Ali, A.; Li, C.; Fang, H.; Wang, C. Complications and costs of peripherally inserted central venous catheters compared with implantable port catheters for cancer patients. Cancer Nurs. 2020, 43, 455–467. [Google Scholar] [CrossRef]
- Corti, F.; Brambilla, M.; Manglaviti, S.; Di Vico, L.; Pisanu, M.N.; Facchinetti, C.; Dotti, K.F.; Lanocita, R.; Marchianò, A.; De Braud, F.; et al. Comparison of outcomes of central venous catheters in patients with solid and hematologic neoplasms: An Italian real-world analysis. Tumori J. 2021, 107, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Dezfulian, C.; Lavelle, J.; Nallamothu, B.K.; Kaufman, S.R.; Saint, S. Rates of infection for single-lumen versus multilumen central venous catheters: A meta-analysis. Crit. Care Med. 2003, 31, 2385–2390. [Google Scholar] [CrossRef] [PubMed]
- Bouza, E.; Burillo, A.; Muñoz, P. Catheter-related infections: Diagnosis and intravascular treatment. Clin. Microbiol. Infect. 2002, 8, 265–274. [Google Scholar] [CrossRef]
- Wisplinghoff, H.; Seifert, H.; Wenzel, R.P.; Edmond, M. Current trends in the epidemiology of nosocomial bloodstream infections in patients with hematological malignancies and solid neoplasms in hospitals in the united states. Clin. Infect. Dis. 2003, 36, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Marcos, M.; Soriano, A.; Iñurrieta, A.; Martínez, J.A.; Romero, A.; Cobos, N.; Hernández, C.; Almela, M.; Marco, F.; Mensa, J. Changing epidemiology of central venous catheter-related bloodstream infections: Increasing prevalence of Gram-negative pathogens. J. Antimicrob. Chemother. 2011, 66, 2119–2125. [Google Scholar] [CrossRef]
- Chaftari, A.M.; Hachem, R.; Jiang, Y.; Shah, P.; Hussain, A.; Al Hamal, Z.; Yousif, A.; Jordan, M.; Michael, M.; Raad, I. Changing Epidemiology of Catheter-Related Bloodstream Infections in Cancer Patients. Infect. Control. Hosp. Epidemiol. 2018, 39, 727–729. [Google Scholar] [CrossRef]
- Abers, M.S.; Sandvall, B.P.; Sampath, R.; Zuno, C.; Uy, N.; Yu, V.L.; Stager, C.E.; Musher, D.M. Postobstructive pneumonia: An underdescribed syndrome. Clin. Infect. Dis. 2016, 62, 957–961. [Google Scholar] [CrossRef]
- Rolston, K.V. Postobstructive pneumonia in cancer patients. Clin. Infect. Dis. 2016, 63, 707–708. [Google Scholar] [CrossRef] [PubMed]
- Kalkat, M.S.; Bonser, R.S. Obstructive pneumonia: An indication for surgery in mega aorta syndrome. Ann. Thorac. Surg. 2003, 75, 1313–1315. [Google Scholar] [CrossRef]
- Rolston, K.V.I.; Nesher, L. Post-Obstructive pneumonia in patients with cancer: A review. Infect. Dis. Ther. 2018, 7, 29–38. [Google Scholar] [CrossRef]
- Seo, S.K.; Liu, C.; Dadwal, S.S. Infectious disease complications in patients with cancer. Crit. Care Clin. 2021, 37, 69–84. [Google Scholar] [CrossRef]
- Battaglia, C.C.; Hale, K. Hospital-Acquired infections in critically III patients with cancer. J. Intensive Care Med. 2018, 34, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Bahu, R.; Chaftari, A.-M.; Hachem, R.Y.; Ahrar, K.; Shomali, W.; El Zakhem, A.; Jiang, Y.; AlShuaibi, M.; Raad, I.I. Nephrostomy tube related pyelonephritis in patients with cancer: Epidemiology, infection rate and risk factors. J. Urol. 2013, 189, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Pu, L.Z.C.T.; Singh, R.; Loong, C.K.; de Moura, E.G.H. Malignant Biliary Obstruction: Evidence for Best Practice. Gastroenterol. Res. Pract. 2016, 2016, 3296801–3296807. [Google Scholar] [CrossRef] [PubMed]
- Cassani, L.; Lee, J.H. Management of malignant distal biliary obstruction. Gastrointest. Interv. 2015, 4, 15–20. [Google Scholar] [CrossRef]
- Shi, S.; Xia, W.; Guo, H.; Kong, H.; Zheng, S. Unique characteristics of pyogenic liver abscesses of biliary origin. Surgery 2016, 159, 1316–1324. [Google Scholar] [CrossRef] [PubMed]
- Rolston, K.V.I.; Dholakia, N.; Rodriguez, S.; Rubenstein, E.B. Nature and outcome of febrile episodes in patients with pancreatic and hepatobiliary cancer. Support. Care Cancer 1995, 3, 414–417. [Google Scholar] [CrossRef]
- Xu, C.; Lv, P.-H.; Huang, X.-E.; Wang, S.-X.; Sun, L.; Wang, F.-A. Analysis of different ways of drainage for obstructive jaundice caused by hilar cholangiocarcinoma. Asian Pac. J. Cancer Prev. 2014, 15, 5617–5620. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aljahdli, E.S. Management of distal malignant biliary obstruction. Saudi J. Gastroenterol. 2018, 24, 71–72. [Google Scholar] [CrossRef]
- Avritscher, E.B.C.; Cooksley, C.D.; Rolston, K.V.; Swint, J.M.; Delclos, G.L.; Franzini, L.; Swisher, S.G.; Walsh, G.L.; Mansfield, P.F.; Elting, L.S. Serious postoperative infections following resection of common solid tumors: Outcomes, costs, and impact of hospital surgical volume. Support. Care Cancer 2014, 22, 527–535. [Google Scholar] [CrossRef]
- Yang, K.; Zang, Z.-Y.; Niu, K.-F.; Sun, L.-F.; Zhang, W.-H.; Zhang, Y.-X.; Chen, X.-L.; Zhou, Z.-G.; Hu, J.-K. The survival benefit and safety of splenectomy for gastric cancer with total gastrectomy: Updated results. Front. Oncol. 2021, 10, 2786. [Google Scholar] [CrossRef]
- Lee, S.S.; Morgenstern, L.; Phillips, E.H.; Hiatt, J.R.; Margulies, D.R. Splenectomy for splenic metastases: A changing clinical spectrum. Am. Surg. 2000, 66, 837–840. [Google Scholar] [PubMed]
- Feola, A.; Niola, M.; Conti, A.; Delbon, P.; Graziano, V.; Paternoster, M.; Della Pietra, B. Iatrogenic splenic injury: Review of the literature and medico-legal issues. Open Med. 2016, 11, 307–315. [Google Scholar] [CrossRef]
- Di Sabatino, A.; Carsetti, R.; Corazza, G.R. Post-Splenectomy and hyposplenic states. Lancet 2011, 378, 86–97. [Google Scholar] [CrossRef]
- Buzelé, R.; Barbier, L.; Sauvanet, A.; Fantin, B. Medical complications following splenectomy. J. Visc. Surg. 2016, 153, 277–286. [Google Scholar] [CrossRef]
- Pawelec, G. Immunosenescence: Impact in the young as well as the old? Mech. Ageing Dev. 1999, 108, 1–7. [Google Scholar] [CrossRef]
- Eşme, M.; Topeli, A.; Yavuz, B.B.D.; Akova, M. Infections in the elderly Critically-III patients. Front. Med. 2019, 6, 118. [Google Scholar] [CrossRef]
- Tannou, T.; Koeberle, S.; Manckoundia, P.; Aubry, R. Multifactorial immunodeficiency in frail elderly patients: Contributing factors and management. Med. Mal. Infect. 2019, 49, 167–172. [Google Scholar] [CrossRef]
- Lyman, G.H.; Abella, E.; Pettengell, R. Risk factors for febrile neutropenia among patients with cancer receiving chemotherapy: A systematic review. Crit. Rev. Oncol. Hematol. 2014, 90, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Balducci, L.; Hardy, C.L.; Lyman, G.H. Hemopoiesis and aging. Cancer Treat. Res. 2005, 124, 109–134. [Google Scholar] [CrossRef]
- Gay, L.; Melenotte, C.; Lakbar, I.; Mezouar, S.; Devaux, C.; Raoult, D.; Bendiane, M.-K.; Leone, M.; Mège, J.-L. Sexual dimorphism and gender in infectious diseases. Front. Immunol. 2021, 12, 698121. [Google Scholar] [CrossRef] [PubMed]
- García-Gómez, E.; González-Pedrajo, B.; Camacho-Arroyo, I. Role of Sex Steroid Hormones in Bacterial-Host Interactions. BioMed Res. Int. 2013, 2013, 928290. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.A.; Karpuzoglu, E.; Khan, D. Effects of sex steroids on innate and adaptive immunity. In Sex Hormones and Immunity to Infection; Klein, S.L., Roberts, C.W., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 19–51. [Google Scholar]
- Fish, E.N. The X-files in immunity: Sex-Based differences predispose immune responses. Nat. Rev. Immunol. 2008, 8, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Harrington, R.D.; Hooton, T.M. Urinary tract infection risk factors and gender. J. Gend. Specif. Med. 2000, 3, 27–34. [Google Scholar]
- Abdel-Rahman, O. Impact of sex on chemotherapy toxicity and efficacy among patients with metastatic colorectal cancer: Pooled analysis of 5 randomized trials. Clin. Color. Cancer 2019, 18, 110–115.e2. [Google Scholar] [CrossRef] [PubMed]
- Fontanella, C.; Bolzonello, S.; Lederer, B.; Aprile, G. Management of breast cancer patients with chemotherapy-induced neutropenia or febrile neutropenia. Breast Care 2014, 9, 239–245. [Google Scholar] [CrossRef]
- Özdemir, B.C.; Csajka, C.; Dotto, G.-P.; Wagner, A.D. Sex differences in efficacy and toxicity of systemic treatments: An undervalued issue in the era of precision oncology. J. Clin. Oncol. 2018, 36, 2680–2683. [Google Scholar] [CrossRef] [PubMed]
- Ruzzo, A.; Graziano, F.; Galli, F.; Galli, F.; Rulli, E.; Lonardi, S.; Ronzoni, M.; Massidda, B.; Zagonel, V.; Pella, N.; et al. Sex-Related Differences in impact on safety of pharmacogenetic profile for colon cancer patients treated with FOLFOX-4 or XELOX adjuvant chemotherapy. Sci. Rep. 2019, 9, 11527. [Google Scholar] [CrossRef]
- Bossi, P.; Delrio, P.; Mascheroni, A.; Zanetti, M. The spectrum of malnutrition/cachexia/sarcopenia in oncology according to different cancer types and settings: A narrative review. Nutrients 2021, 13, 1980. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.K. Nutrition, immunity and infection: From basic knowledge of dietary manipulation of immune responses to practical application of ameliorating suffering and improving survival. Proc. Natl. Acad. Sci. USA 1996, 93, 14304–14307. [Google Scholar] [CrossRef]
- Triarico, S.; Rinninella, E.; Cintoni, M.; Capozza, M.A.; Mastrangelo, S.; Mele, M.C.; Ruggiero, A. Impact of malnutrition on survival and infections among pediatric patients with cancer: A retrospective study. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 1165–1175. [Google Scholar]
- Falagas, M.E.; Kompoti, M. Obesity and infection. Lancet Infect. Dis. 2006, 6, 438–446. [Google Scholar] [CrossRef]
- Ghilotti, F.; Bellocco, R.; Ye, W.; Adami, H.-O.; Lagerros, Y.T. Obesity and risk of infections: Results from men and women in the Swedish National March Cohort. Int. J. Epidemiol. 2019, 48, 1783–1794. [Google Scholar] [CrossRef]
- Huttunen, R.; Syrjänen, J. Obesity and the risk and outcome of infection. Int. J. Obes. 2013, 37, 333–340. [Google Scholar] [CrossRef]
- Carey, I.M.; Critchley, J.A.; DeWilde, S.; Harris, T.; Hosking, F.J.; Cook, D.G. Risk of infection in type 1 and type 2 diabetes compared with the general population: A matched cohort study. Diabetes Care 2018, 41, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Berman, S.J.; Johnson, E.W.; Nakatsu, C.; Alkan, M.; Chen, R.; LeDuc, J. Burden of infection in patients with end-stage renal disease requiring long-term dialysis. Clin. Infect. Dis. 2004, 39, 1747–1753. [Google Scholar] [CrossRef] [PubMed]
- Cohen, G.; Hörl, W.H. Immune dysfunction in Uremia—An update. Toxins 2012, 4, 962–990. [Google Scholar] [CrossRef] [PubMed]
- Lange, P. Chronic obstructive pulmonary disease and risk of infection. Pneumonol. Alergol. Polska 2009, 77, 284–288. [Google Scholar]
- Fragoulis, G.E.; Sipsas, N.V. When rheumatology and infectious disease come together. Ther. Adv. Musculoskelet. Dis. 2019, 11, 1–3. [Google Scholar] [CrossRef]
- Hsu, C.-Y.; Ko, C.-H.; Wang, J.-L.; Hsu, T.-C.; Lin, C.-Y. Comparing the burdens of opportunistic infections among patients with systemic rheumatic diseases: A nationally representative cohort study. Arthritis Res. 2019, 21, 211. [Google Scholar] [CrossRef] [PubMed]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.; Gustot, T. Management of bacterial infections in cirrhosis. J. Hepatol. 2012, 56 (Suppl. 1), S1–S12. [Google Scholar] [CrossRef]
- McCusker, C.; Warrington, R. Primary immunodeficiency. Allergy Asthma Clin. Immunol. 2011, 7 (Suppl. 1), S11. [Google Scholar] [CrossRef]
- Okishiro, M.; Kim, S.J.; Tsunashima, R.; Nakayama, T.; Shimazu, K.; Shimomura, A.; Maruyama, N.; Tamaki, Y.; Noguchi, S. MDM2 SNP309 and TP53 R72P associated with severe and febrile neutropenia in breast cancer patients treated with 5-FU/epirubicin/cyclophosphamide. Breast Cancer Res. Treat. 2012, 132, 947–953. [Google Scholar] [CrossRef]
- Vulsteke, C.; Lambrechts, D.; Dieudonné, A.; Hatse, S.; Brouwers, B.; van Brussel, T.; Neven, P.; Belmans, A.; Schöffski, P.; Paridaens, R.; et al. Genetic variability in the multidrug resistance associated protein-1 (ABCC1/MRP1) predicts hematological toxicity in breast cancer patients receiving (neo-)adjuvant chemotherapy with 5-fluorouracil, epirubicin and cyclophosphamide (FEC). Ann. Oncol. 2013, 24, 1513–1525. [Google Scholar] [CrossRef]
- McLeod, H.L.; Sargent, D.; Marsh, S.; Green, E.M.; King, C.R.; Fuchs, C.S.; Ramanathan, R.K.; Williamson, S.K.; Findlay, B.P.; Thibodeau, S.N.; et al. Pharmacogenetic predictors of adverse events and response to chemotherapy in metastatic colorectal cancer: Results from north american gastrointestinal intergroup trial N9741. J. Clin. Oncol. 2010, 28, 3227–3233. [Google Scholar] [CrossRef] [PubMed]
- Cremolini, C.; Del Re, M.; Antoniotti, C.; Lonardi, S.; Bergamo, F.; Loupakis, F.; Borelli, B.; Marmorino, F.; Citi, V.; Cortesi, E.; et al. DPYD and UGT1A1 genotyping to predict adverse events during first-line FOLFIRI or FOLFOXIRI plus bevacizumab in metastatic colorectal cancer. Oncotarget 2017, 9, 7859–7866. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Iwasa, S.; Shoji, H.; Honma, Y.; Takashima, A.; Kato, K.; Hamaguchi, T.; Higuchi, K.; Boku, N. Association between UGT1A1 gene polymorphism and safety and efficacy of irinotecan monotherapy as the third-line treatment for advanced gastric cancer. Gastric Cancer 2019, 22, 778–784. [Google Scholar] [CrossRef] [PubMed]
- Wood, A.J.; Pizzo, P.A. Management of fever in patients with cancer and treatment-induced neutropenia. N. Engl. J. Med. 1993, 328, 1323–1332. [Google Scholar] [CrossRef]
- DiNubile, M.J. Fever and neutropenia: Still a challenge. Contemp. Intern. Med. 1995, 7, 35–41. [Google Scholar] [PubMed]
- Zell, J.A.; Chang, J.C. Neoplastic fever: A neglected paraneoplastic syndrome. Support. Care Cancer 2005, 13, 870–877. [Google Scholar] [CrossRef] [PubMed]
- Kasuga, I.; Makino, S.; Kiyokawa, H.; Katoh, H.; Ebihara, Y.; Ohyashiki, K. Tumor-Related leukocytosis is linked with poor prognosis in patients with lung carcinoma. Cancer 2001, 92, 2399–2405. [Google Scholar] [CrossRef]
- Hart, P.C.; Rajab, I.M.; Alebraheem, M.; Potempa, L.A. C-Reactive protein and cancer—Diagnostic and therapeutic insights. Front. Immunol. 2020, 11, 595835. [Google Scholar] [CrossRef]
- Vincenzi, B.; Fioroni, I.; Pantano, F.; Angeletti, S.; Dicuonzo, G.; Zoccoli, A.; Santini, D.; Tonini, G. Procalcitonin as diagnostic marker of infection in solid tumors patients with fever. Sci. Rep. 2016, 6, 28090. [Google Scholar] [CrossRef]
- Palmore, T.N.; Parta, M.; Cuellar-Rodriguez, J.; Gea-Banacloche, J.C. Infections in the Cancer Patient. In DeVita, Hellman, and Rosenberg’s Cancer: Principles & Practice of Oncology, 10th ed.; Vincent, T.D., Jr., Theodore, S.L., Steven, A.R., Eds.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2011; pp. 1931–1959. [Google Scholar]
- Gao, Y.; Shang, Q.; Li, W.; Guo, W.; Stojadinovic, A.; Mannion, C.; Man, Y.-G.; Chen, T. Antibiotics for cancer treatment: A double-edged sword. J. Cancer 2020, 11, 5135–5149. [Google Scholar] [CrossRef]
- Hecker, M.T.; Aron, D.C.; Patel, N.P.; Lehmann, M.K.; Donskey, C.J. Unnecessary use of antimicrobials in hospitalized patients: Current patterns of misuse with an emphasis on the antianaerobic spectrum of activity. Arch. Intern. Med. 2003, 163, 972–978. [Google Scholar] [CrossRef]
- Fridkin, S.; Baggs, J.; Fagan, R.; Magill, S.; Pollack, L.A.; Malpiedi, P.; Slayton, R.; Khader, K.; Rubin, M.A.; Jones, M.; et al. Vital signs: Improving antibiotic use among hospitalized patients. MMWR. Morb. Mortal. Wkly. Rep. 2014, 63, 194–200. [Google Scholar] [PubMed]
- Dellit, T.H.; Owens, R.C.; McGowan, J.E., Jr.; Gerding, D.N.; Weinstein, R.A.; Burke, J.P.; Huskins, W.C.; Paterson, D.L.; Fishman, N.O.; Carpenter, C.F.; et al. Infectious diseases society of america and the society for healthcare epidemiology of america guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin. Infect. Dis. 2007, 44, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Islas-Muñoz, B.; Volkow-Fernández, P.; Ibanes-Gutiérrez, C.; Villamar-Ramírez, A.; Vilar-Compte, D.; Cornejo-Juárez, P. Bloodstream infections in cancer patients. Risk factors associated with mortality. Int. J. Infect. Dis. 2018, 71, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Baur, D.; Gladstone, B.P.; Burkert, F.; Carrara, E.; Foschi, F.; Döbele, S.; Tacconelli, E. Effect of antibiotic stewardship on the incidence of infection and colonisation with antibiotic-resistant bacteria and Clostridium difficile infection: A systematic review and meta-analysis. Lancet Infect. Dis. 2017, 17, 990–1001. [Google Scholar] [CrossRef]
- Nathwani, D.; Varghese, D.; Stephens, J.; Ansari, W.; Martin, S.; Charbonneau, C. Value of hospital antimicrobial stewardship programs [ASPs]: A systematic review. Antimicrob. Resist. Infect. Control. 2019, 8, 35. [Google Scholar] [CrossRef]
- Ramos-Casals, M.; Brahmer, J.R.; Callahan, M.K.; Flores-Chávez, A.; Keegan, N.; Khamashta, M.A.; Lambotte, O.; Mariette, X.; Prat, A.; Suárez-Almazor, M.E. Immune-Related adverse events of checkpoint inhibitors. Nat. Rev. Dis. Prim. 2020, 6, 38. [Google Scholar] [CrossRef]
- Del Castillo, M.; Romero, F.A.; Argüello, E.; Kyi, C.; Postow, M.A.; Redelman-Sidi, G. The spectrum of serious infections among patients receiving immune checkpoint blockade for the treatment of melanoma. Clin. Infect. Dis. 2016, 63, 1490–1493. [Google Scholar] [CrossRef]
- Elkrief, A.; DeRosa, L.; Kroemer, G.; Zitvogel, L.; Routy, B. The negative impact of antibiotics on outcomes in cancer patients treated with immunotherapy: A new independent prognostic factor? Ann. Oncol. 2019, 30, 1572–1579. [Google Scholar] [CrossRef]
- Freifeld, A.G.; Bow, E.J.; Sepkowitz, K.A.; Boeckh, M.J.; Ito, J.I.; Mullen, C.A.; Raad, I.I.; Rolston, K.V.; Young, J.-A.H.; Wingard, J.R. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america. Clin. Infect. Dis. 2011, 52, e56–e93. [Google Scholar] [CrossRef]
- Carmona-Bayonas, A.; Jiménez-Fonseca, P.; Echaburu, J.V.; Cánovas, M.S.; De La Peña, F.A. The time has come for new models in febrile neutropenia: A practical demonstration of the inadequacy of the MASCC score. Clin. Transl. Oncol. 2017, 19, 1084–1090. [Google Scholar] [CrossRef]
- Peyrony, O.; Gerlier, C.; Barla, I.; Ellouze, S.; Legay, L.; Azoulay, E.; Chevret, S.; Fontaine, J.-P. Antibiotic prescribing and outcomes in cancer patients with febrile neutropenia in the emergency department. PLoS ONE 2020, 15, e0229828. [Google Scholar] [CrossRef]
- Elting, L.S.; Lu, C.; Escalante, C.P.; Giordano, S.H.; Trent, J.C.; Cooksley, C.; Avritscher, E.B.; Shih, Y.-C.T.; Ensor, J.; Bekele, B.N.; et al. Outcomes and cost of outpatient or inpatient management of 712 patients with febrile neutropenia. J. Clin. Oncol. 2008, 26, 606–611. [Google Scholar] [CrossRef] [PubMed]
- AJMC. Guidelines in the Management of Febrile Neutropenia for Clinical Practice. Available online: https://www.ajmc.com/view/guidelines-in-the-management-of-febrile-neutropenia-for-clinical-practice (accessed on 20 November 2021).
- Taplitz, R.A.; Kennedy, E.B.; Bow, E.J.; Crews, J.; Gleason, C.; Hawley, D.K.; Langston, A.A.; Nastoupil, L.J.; Rajotte, M.; Rolston, K.; et al. Outpatient management of fever and neutropenia in adults treated for malignancy: American society of clinical oncology and infectious diseases society of america clinical practice guideline update. J. Clin. Oncol. 2018, 36, 1443–1453. [Google Scholar] [CrossRef] [PubMed]
- Anatoliotaki, M.; Valatas, V.; Mantadakis, E.; Apostolakou, H.; Mavroudis, D.; Georgoulias, V.; Rolston, K.V.; Kontoyiannis, D.P.; Galanakis, E.; Samonis, G. Bloodstream infections in patients with solid tumors: Associated factors, microbial spectrum and outcome. Infection 2004, 32, 65–71. [Google Scholar] [CrossRef]
- Marín, M.; Gudiol, C.; Garcia-Vidal, C.; Ardanuy, C.; Carratala, J. Bloodstream Infections in patients with solid tumors: Epidemiology, antibiotic therapy, and outcomes in 528 episodes in a single cancer center. Medicine 2014, 93, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Seifert, H.; Cornely, O.; Seggewiss, K.; Decker, M.; Stefanik, D.; Wisplinghoff, H.; Fätkenheuer, G. Bloodstream infection in neutropenic cancer patients related to short-term nontunnelled catheters determined by quantitative blood cultures, differential time to positivity, and molecular epidemiological typing with pulsed-field gel electrophoresis. J. Clin. Microbiol. 2003, 41, 118–123. [Google Scholar] [CrossRef]
- Mermel, L.A.; Allon, M.; Bouza, E.; Craven, D.E.; Flynn, P.; O’Grady, N.P.; Raad, I.I.; Rijnders, B.J.A.; Sherertz, R.J.; Warren, D.K. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 update by the infectious diseases society of america. Clin. Infect. Dis. 2009, 49, 1–45. [Google Scholar] [CrossRef]
- Cantón-Bulnes, M.L.; Garnacho-Montero, J. Practical approach to the management of catheter-related bloodstream infection. Rev. Esp. Quimioter 2019, 32, 38–41. [Google Scholar]
- Muff, S.; Tabah, A.; Que, Y.-A.; Timsit, J.-F.; Mermel, L.; Harbarth, S.; Buetti, N. Short-Course versus long-course systemic antibiotic treatment for uncomplicated intravascular catheter-related bloodstream infections due to gram-negative bacteria, enterococci or coagulase-negative staphylococci: A systematic review. Infect. Dis. Ther. 2021, 10, 1591–1605. [Google Scholar] [CrossRef] [PubMed]
- Chaves, F.; Garnacho-Montero, J.; Del Pozo, J.L.; Bouza, E.; Capdevila, J.; de Cueto, M.; Domínguez, M.; Esteban, J.; Fernández-Hidalgo, N.; Sampedro, M.F.; et al. Diagnosis and treatment of catheter-related bloodstream infection: Clinical guidelines of the Spanish Society of Infectious Diseases and Clinical Microbiology and (SEIMC) and the Spanish Society of Spanish Society of Intensive and Critical Care Medicine and Coronary Units (SEMICYUC). Med. Intensiv. 2018, 42, 5–36. [Google Scholar] [CrossRef]
- Norris, L.B.; Kablaoui, F.; Brilhart, M.K.; Bookstaver, P.B. Systematic review of antimicrobial lock therapy for prevention of central-line-associated bloodstream infections in adult and pediatric cancer patients. Int. J. Antimicrob. Agents 2017, 50, 308–317. [Google Scholar] [CrossRef]
- Pliakos, E.E.; Andreatos, N.; Ziakas, P.; Mylonakis, E. The cost-effectiveness of antimicrobial lock solutions for the prevention of central line–associated bloodstream infections. Clin. Infect. Dis. 2019, 68, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.L.; Tawfik, G.; Saxinger, L.; Stang, L.; Etches, W.; Lee, B. Stability of heparin and physical compatibility of heparin/antibiotic solutions in concentrations appropriate for antibiotic lock therapy. J. Antimicrob. Chemother. 2005, 56, 951–953. [Google Scholar] [CrossRef]
- Luther, M.K.; Mermel, L.A.; Laplante, K.L. Comparison of linezolid and vancomycin lock solutions with and without heparin against biofilm-producing bacteria. Am. J. Health Pharm. 2017, 74, e193–e201. [Google Scholar] [CrossRef] [PubMed]
- Del Pozo, J.L. Role of antibiotic lock therapy for the treatment of catheter-related bloodstream infections. Int. J. Artif. Organs 2009, 32, 678–688. [Google Scholar] [CrossRef] [PubMed]
- Bookstaver, P.B.; Rokas, K.E.E.; Norris, L.B.; Edwards, J.M.; Sherertz, R.J. Stability and compatibility of antimicrobial lock solutions. Am. J. Health Syst. Pharm. 2013, 70, 2185–2198. [Google Scholar] [CrossRef]
- LaPlante, K.L.; Mermel, L.A. In Vitro activity of daptomycin and vancomycin lock solutions on staphylococcal biofilms in a central venous catheter model. Nephrol. Dial. Transpl. 2007, 22, 2239–2246. [Google Scholar] [CrossRef] [PubMed]
- Krishnasami, Z.; Carlton, D.; Bimbo, L.; Taylor, M.E.; Balkovetz, D.F.; Barker, J.; Allon, M. Management of hemodialysis catheter-related bacteremia with an adjunctive antibiotic lock solution. Kidney Int. 2002, 61, 1136–1142. [Google Scholar] [CrossRef]
- Vercaigne, L.M.; Sitar, D.S.; Penner, S.B.; Bernstein, K.; Wang, G.Q.; Burczynski, F. Antibiotic-Heparin lock: In Vitro antibiotic stability combined with heparin in a central venous catheter. Pharmacotherapy 2000, 20, 394–399. [Google Scholar] [CrossRef]
- Justo, J.A.; Bookstaver, P.B. Antibiotic lock therapy: Review of technique and logistical challenges. Infect. Drug Resist. 2014, 7, 343–363. [Google Scholar] [CrossRef]
- Rijnders, B.J.; Van Wijngaerden, E.; Vandecasteele, S.J.; Stas, M.; Peetermans, W.E. Treatment of long-term intravascular catheter-related bacteraemia with antibiotic lock: Randomized, placebo-controlled trial. J. Antimicrob. Chemother. 2005, 55, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Ko, K.S.; Peck, K.R.; Oh, W.S.; Song, J.-H. In Vitro evaluation of the antibiotic lock technique (ALT) for the treatment of catheter-related infections caused by staphylococci. J. Antimicrob. Chemother. 2006, 57, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Droste, J.C.; Jeraj, H.A.; Macdonald, A.; Farrington, K. Stability and in vitro efficacy of antibiotic-heparin lock solutions potentially useful for treatment of central venous catheter-related sepsis. J. Antimicrob. Chemother. 2003, 51, 849–855. [Google Scholar] [CrossRef]
- Lee, M.Y.; Ko, K.S.; Song, J.-H.; Peck, K.R. In Vitro effectiveness of the antibiotic lock technique (ALT) for the treatment of catheter-related infections by Pseudomonas aeruginosa and Klebsiella pneumoniae. J. Antimicrob. Chemother. 2007, 60, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Onland, W.; Shin, C.E.; Fustar, S.; Rushing, T.; Wong, W.-Y. Ethanol-Lock technique for persistent bacteremia of long-term intravascular devices in pediatric patients. Arch. Pediatr. Adolesc. Med. 2006, 160, 1049–1053. [Google Scholar] [CrossRef]
- EMC. Ampicillin 500 mg powder for solution for injection—Summary of Product Characteristics (SPC). Available online: https://www.medicines.org.uk/emc/product/12892/smpc#gref (accessed on 12 November 2021).
- Vila-Corcoles, A.; Ochoa-Gondar, O.; Rodriguez-Blanco, T.; Raga-Luria, X.; Gomez-Bertomeu, F. Epidemiology of community-acquired pneumonia in older adults: A population-based study. Respir. Med. 2009, 103, 309–316. [Google Scholar] [CrossRef]
- Parakh, A.; Krishnamurthy, S.; Bhattacharya, M. Ertapenem. Kathmandu Univ. Med. J. (KUMJ) 2009, 7, 454–460. [Google Scholar] [CrossRef][Green Version]
- Kalil, A.C.; Metersky, M.L.; Klompas, M.; Muscedere, J.; Sweeney, D.A.; Palmer, L.B.; Napolitano, L.M.; O’Grady, N.P.; Bartlett, J.G.; Carratala, J.; et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the infectious diseases society of america and the american thoracic society. Clin. Infect. Dis. 2016, 63, e61–e111. [Google Scholar] [CrossRef]
- Metlay, J.P.; Waterer, G.W.; Long, A.C.; Anzueto, A.; Brozek, J.; Crothers, K.; Cooley, L.A.; Dean, N.C.; Fine, M.J.; Flanders, S.A.; et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the american thoracic society and infectious diseases society of America. Am. J. Respir. Crit. Care Med. 2019, 200, e45–e67. [Google Scholar] [CrossRef] [PubMed]
- Hanson, K.E.; Azar, M.M.; Banerjee, R.; Chou, A.; Colgrove, R.C.; Ginocchio, C.C.; Hayden, M.K.; Holodiny, M.; Jain, S.; Koo, S.; et al. Molecular testing for acute respiratory tract infections: Clinical and diagnostic recommendations from the IDSA’s diagnostics committee. Clin. Infect. Dis. 2020, 71, 2744–2751. [Google Scholar] [CrossRef]
- Sartelli, M.; Chichom-Mefire, A.; Labricciosa, F.M.; Hardcastle, T.; Abu-Zidan, F.M.; Adesunkanmi, A.K.; Ansaloni, L.; Bala, M.; Balogh, Z.J.; Beltrán, M.A.; et al. The management of intra-abdominal infections from a global perspective: 2017 WSES guidelines for management of intra-abdominal infections. World J. Emerg. Surg. 2017, 12, 29. [Google Scholar] [CrossRef]
- Solomkin, J.S.; Mazuski, J.E.; Bradley, J.S.; Rodvold, K.A.; Goldstein, E.J.; Baron, E.J.; O’Neill, P.J.; Chow, A.W.; Dellinger, E.P.; Eachempati, S.R.; et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: Guidelines by the surgical infection society and the infectious diseases society of america. Clin. Infect. Dis. 2010, 50, 133–164. [Google Scholar] [CrossRef]
- Guevara, E.A.Y.; Aitken, S.L.; Olvera, A.V.; Carlin, L.; Fernandes, K.E.; Bhatti, M.M.; Garey, K.W.; Adachi, J.; Okhuysen, P.C. Clostridioides difficile infection in cancer and immunocompromised patients: Relevance of a two-step diagnostic algorithm and infecting ribotypes on clinical outcomes. Clin. Infect. Dis. 2020, 72, e460–e465. [Google Scholar] [CrossRef]
- Abughanimeh, O.; Qasrawi, A.; Kaddourah, O.; Al Momani, L.; Abu Ghanimeh, M. Clostridium difficileinfection in oncology patients: Epidemiology, pathophysiology, risk factors, diagnosis, and treatment. Hosp. Pract. 2018, 46, 266–277. [Google Scholar] [CrossRef]
- Johnson, S.; Lavergne, V.; Skinner, A.M.; Gonzales-Luna, A.J.; Garey, K.W.; Kelly, C.P.; Wilcox, M.H. Clinical practice guideline by the infectious diseases society of america (idsa) and society for Healthcare Epidemiology of America (SHEA): 2021 focused update guidelines on management of clostridioides difficile infection in adults. Clin. Infect. Dis. 2021, 73, e1029–e1044. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, I.D.C.; Greenberg, H.M. Gastrointestinal complications in the neutropenic patient: Characterization and differentiation with abdominal CT. Radiology 2003, 226, 668–674. [Google Scholar] [CrossRef]
- Song, H.K.; Kreisel, D.; Canter, R.; Krupnick, A.S.; Stadtmauer, E.A.; Buzby, G. Changing presentation and management of neutropenic enterocolitis. Arch. Surg. 1998, 133, 979–982. [Google Scholar] [CrossRef] [PubMed]
- Tigabu, A.; Ferede, W.; Belay, G.; Gelaw, B. Prevalence of asymptomatic bacteriuria and antibiotic susceptibility patterns of bacterial isolates among cancer patients and healthy blood donors at the University of Gondar Specialized Hospital. Int. J. Microbiol. 2020, 2020, 3091564. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, G.; Wei, X.; Hann, K.; Soe, K.; Satyanarayana, S.; Siwakoti, B.; Bastakoti, S.; Mulmi, R.; Rana, K.; Lamichhane, N. Bacterial profile and antibiotic resistance among cancer patients with urinary tract infection in a national tertiary cancer hospital of Nepal. Trop. Med. Infect. Dis. 2021, 6, 49. [Google Scholar] [CrossRef] [PubMed]
- Parikh, P.; Bhat, V. Urinary tract infection in cancer patients in a tertiary cancer setting in India: Microbial spectrum and antibiotic susceptibility pattern. Antimicrob. Resist. Infect. Control 2015, 4, 221. [Google Scholar] [CrossRef]
- Khaparkuntikar, M.; Siddiqui, N.; Bhirud, P. Urinary tract infection in cancer patients at Government Cancer Hospital Aurangabad, India. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 2259–2263. [Google Scholar] [CrossRef]
- Bonkat, G.; Bartoletti, R.; Bruyère, F.; Cai, T.; Geerlings, S.E.; Köves, B.; Schubert, S.; Wagenlehner, F. EAU Guidelines on Urological Infections; EAU Guidelines Office: Arnhem, The Netherlands, 2020. [Google Scholar]
- Nicolle, L. Complicated urinary tract infection in adults. Can. J. Infect. Dis. Med. Microbiol. 2005, 16, 349–360. [Google Scholar] [CrossRef]
- Lopes, M.S.M.; Machado, L.M.; Silva, P.A.I.A.; Uchiyama, A.A.T.; Yen, C.T.; Ricardo, E.D.; Mutao, T.S.; Pimenta, J.R.; Shimba, D.S.; Hanriot, R.M.; et al. Antibiotics, cancer risk and oncologic treatment efficacy: A practical review of the literature. Ecancermedicalscience 2020, 14, 1106. [Google Scholar]
- Shui, L.; Yang, X.; Li, J.; Yi, C.; Sun, Q.; Zhu, H. Gut microbiome as a potential factor for modulating resistance to cancer immunotherapy. Front. Immunol. 2020, 10, 2989. [Google Scholar] [CrossRef]
- Ma, W.; Mao, Q.; Xia, W.; Dong, G.; Yu, C.; Jiang, F. Gut microbiota shapes the efficiency of cancer therapy. Front. Microbiol. 2019, 10, 1050. [Google Scholar] [CrossRef]
- Reed, J.P.; Devkota, S.; Figlin, R.A. Gut microbiome, antibiotic use, and immunotherapy responsiveness in cancer. Ann. Transl. Med. 2019, 7, S309. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, V.; Spencer, C.N.; Nezi, L.; Reuben, A.; Andrews, M.C.; Karpinets, T.V.; Prieto, P.A.; Vicente, D.; Hoffman, K.; Wei, S.C.; et al. Gut microbiome modulates response to anti–PD-1 immunotherapy in melanoma patients. Science 2018, 359, 97–103. [Google Scholar] [CrossRef]
- Matson, V.; Fessler, J.; Bao, R.; Chongsuwat, T.; Zha, Y.; Alegre, M.-L.; Luke, J.J.; Gajewski, T.F. The commensal microbiome is associated with anti–PD-1 efficacy in metastatic melanoma patients. Science 2018, 359, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Routy, B.; Le Chatelier, E.; DeRosa, L.; Duong, C.P.M.; Alou, M.T.; Daillère, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science 2018, 359, 91–97. [Google Scholar] [CrossRef]
- Vétizou, M.; Pitt, J.M.; Daillère, R.; Lepage, P.; Waldschmitt, N.; Flament, C.; Rusakiewicz, S.; Routy, B.; Roberti, M.P.; Duong, C.P.M.; et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 2015, 350, 1079–1084. [Google Scholar] [CrossRef] [PubMed]
- Sivan, A.; Corrales, L.; Hubert, N.; Williams, J.B.; Aquino-Michaels, K.; Earley, Z.M.; Benyamin, F.W.; Lei, Y.M.; Jabri, B.; Alegre, M.-L.; et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science 2015, 350, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Chaput, N.; Lepage, P.; Coutzac, C.; Soularue, E.; Le Roux, K.; Monot, C.; Boselli, L.; Routier, E.; Cassard, L.; Collins, M.; et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann. Oncol. 2017, 28, 1368–1379. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, N.; Zhou, C.; Tan, G.; Rack, S.; Lorigan, P.; Blackhall, F.; Krebs, M.; Carter, L.; Thistlethwaite, F.; Graham, D.; et al. Cumulative antibiotic use significantly decreases efficacy of checkpoint inhibitors in patients with advanced cancer. Oncologist 2020, 25, 55–63. [Google Scholar] [CrossRef]
- Mohiuddin, J.J.; Chu, B.; Facciabene, A.; Poirier, K.; Wang, X.; Doucette, A.; Zheng, C.; Xu, W.; Anstadt, E.J.; Amaravadi, R.K.; et al. Association of antibiotic exposure with survival and toxicity in patients with melanoma receiving immunotherapy. J. Natl. Cancer Inst. 2020, 113, 162–170. [Google Scholar] [CrossRef]
- Derosa, L.; Hellmann, M.; Spaziano, M.; Halpenny, D.; Fidelle, M.; Rizvi, H.; Long, N.; Plodkowski, A.; Arbour, K.; Chaft, J.; et al. Negative association of antibiotics on clinical activity of immune checkpoint inhibitors in patients with advanced renal cell and non-small-cell lung cancer. Ann. Oncol. 2018, 29, 1437–1444. [Google Scholar] [CrossRef]
- Rubio, X.M.; Chara, L.; Sotelo-Lezama, M.; Castro, R.L.; Rubio-Martínez, J.; Velastegui, A.; Olier-Garate, C.; Falagan, S.; Gómez-Barreda, I.; Bautista-Sanz, P.; et al. MA10.01 antibiotic use and PD-1 inhibitors: Shorter survival in lung cancer, especially when given intravenously. Type of infection also matters. J. Thorac. Oncol. 2018, 13, S389. [Google Scholar] [CrossRef]
- Galli, G.; Triulzi, T.; Proto, C.; Signorelli, D.; Imbimbo, M.; Poggi, M.; Fucà, G.; Ganzinelli, M.; Vitali, M.; Palmieri, D.; et al. Association between antibiotic-immunotherapy exposure ratio and outcome in metastatic non small cell lung cancer. Lung Cancer 2019, 132, 72–78. [Google Scholar] [CrossRef]
- Geum, M.; Kim, C.; Kang, J.; Choi, J.; Kim, J.; Son, E.; Lim, S.; Rhie, S. Broad-Spectrum antibiotic regimen affects survival in patients receiving nivolumab for non-small cell lung cancer. Pharmaceuticals 2021, 14, 445. [Google Scholar] [CrossRef]
- Lalani, A.-K.A.; Xie, W.; Braun, D.A.; Kaymakcalan, M.; Bossé, D.; Steinharter, J.A.; Martini, D.; Simantov, R.; Lin, X.; Wei, X.X.; et al. Effect of antibiotic use on outcomes with systemic therapies in metastatic renal cell carcinoma. Eur. Urol. Oncol. 2020, 3, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.-Z.; Gao, P.; Song, Y.-X.; Xu, Y.; Sun, J.-X.; Chen, X.-W.; Zhao, J.-H.; Wang, Z.-N. Antibiotic use and the efficacy of immune checkpoint inhibitors in cancer patients: A pooled analysis of 2740 cancer patients. OncoImmunology 2019, 8, e1665973. [Google Scholar] [CrossRef] [PubMed]
- Lurienne, L.; Cervesi, J.; Duhalde, L.; de Gunzburg, J.; Andremont, A.; Zalcman, G.; Buffet, R.; Bandinelli, P.-A. NSCLC immunotherapy efficacy and antibiotic use: A systematic review and meta-analysis. J. Thorac. Oncol. 2020, 15, 1147–1159. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.E.; Routy, B.; Nagrial, A.; Chin, V.T. The effect of antibiotics on clinical outcomes in immune-checkpoint blockade: A systematic review and meta-analysis of observational studies. Cancer Immunol. Immunother. 2020, 69, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Uribe-Herranz, M.; Rafail, S.; Beghi, S.; Gil-De-Gómez, L.; Verginadis, I.; Bittinger, K.; Pustylnikov, S.; Pierini, S.; Perales-Linares, R.; Blair, I.A.; et al. Gut microbiota modulate dendritic cell antigen presentation and radiotherapy-induced antitumor immune response. J. Clin. Investig. 2020, 130, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Hou, Y.; Zhang, Y.; Liang, H.; Sharma, A.; Zheng, W.; Wang, L.; Torres, R.; Tatebe, K.; Chmura, S.J.; et al. Suppression of local type I interferon by gut microbiota–derived butyrate impairs antitumor effects of ionizing radiation. J. Exp. Med. 2021, 218. [Google Scholar] [CrossRef] [PubMed]
- Nenclares, P.; Bhide, S.A.; Sandoval-Insausti, H.; Pialat, P.; Gunn, L.; Melcher, A.; Newbold, K.; Nutting, C.M.; Harrington, K.J. Impact of antibiotic use during curative treatment of locally advanced head and neck cancers with chemotherapy and radiotherapy. Eur. J. Cancer 2020, 131, 9–15. [Google Scholar] [CrossRef]
- Corty, R.W.; Langworthy, B.W.; Fine, J.P.; Buse, J.B.; Sanoff, H.K.; Lund, J.L. Antibacterial Use Is Associated with an Increased Risk of Hematologic and Gastrointestinal Adverse Events in Patients Treated with Gemcitabine for Stage IV Pancreatic Cancer. Oncologist 2020, 25, 579–584. [Google Scholar] [CrossRef]
- Lee, N.; Kim, W.-U. Microbiota in T-cell homeostasis and inflammatory diseases. Exp. Mol. Med. 2017, 49, e340. [Google Scholar] [CrossRef]
| Tumor Type | Chemotherapy Regimen | Risk of FN (%) | Reference |
|---|---|---|---|
| Breast cancer | AC (Doxorubicin/Cyclophosphamide) | 7–13 * | Truong et al. [13] |
| AC⟶D (Doxorubicin/Cyclophosphamide⟶Docetaxel) | 25 | Perez et al. [14] | |
| TAC (Docetaxel/Doxorubicin/Cyclophosphamide) | 22 | Von Minckwitz et al. [15] | |
| TC (Docetaxel/Cyclophosphamide) | 70 | Kosaka et al. [16] | |
| TCH (Docetaxel/Carboplatin/Trastuzumab) | 41 | Gilbar et al. [17] | |
| Docetaxel | 17 | Marty et al. [18] | |
| Bladder cancer | MVAC (Methotrexate/Vinblastine/Doxorubicin/Cisplatin) | 26 | Sternberg et al. [19] |
| Cervical cancer | Cisplatin/Paclitaxel | 28 | Rose et al. [20] |
| Cisplatin/Topotecan | 18 | Long et al. [21] | |
| Gastric cancer | DCF (Docetaxel/Cisplatin/5-FU) † | 29 | Van Cutsem et al. [22] |
| TCF (Docetaxel/Cisplatin/5-FU) ‡ | 41 | Roth et al. [23] | |
| ECF (Epirubicin/Cisplatin/5-FU) | 13–18 | Roth et al. [23], Cunningham et al. [24] | |
| ECX (Epirubicin/Cisplatin/Capecitabine) | 11 | Cunningham et al. [24] | |
| Germ cell tumors | BEP (Bleomycin/Etoposide/Cisplatin) | 13 | Fossa et al. [25] |
| EP (Etoposide/Cisplatin) | 10 | Motzer et al. [26] | |
| VIP (Etoposide/Ifosfamide/Cisplatin) | 15 | Fujiwara et al. [27] | |
| VeIP (Vinblastine/Etoposide/Cisplatin) | 67 | Miller et al. [28] | |
| TIP (Paclitaxel/Ifosfamide/Cisplatin) | 48 | Kondagunta et al. [29] | |
| HNSCC | TPF (Docetaxel/Cisplatin/5-FU) | 11 | Pointreau et al. [30] |
| NSCLC | Cisplatin/Paclitaxel | 16 | Schiller et al. [31] |
| Cisplatin/Vinorelbine | 22 | Pujol et al. [32] | |
| Cisplatin/Docetaxel | 5–11 | Fossella et al. [33], Schiller et al. [31] | |
| Cisplatin/Etoposide | 54 § 12 ¶ | Font et al. [34] Cardenal et al. [35] | |
| Docetaxel/Carboplatin | 26 | Millward et al. [36] | |
| Ovarian cancer | Topotecan | 42 | Swisher et al. [37] |
| Docetaxel | 33 | Verschraegen et al. [38] | |
| Paclitaxel | 22 | Omura et al. [39] | |
| Pancreatic cancer | FOLFIRINOX (5-FU/Leucovorin/Oxaliplatin/Irinotecan) | 17 | Hosein et al. [40] |
| SCLC | Etoposide/Carboplatin | 14 | Yilmaz et al. [41] |
| Topotecan | 28 | Von Pawel et al. [42] | |
| ICE (Ifosfamide/Carboplatin/Etoposide) | 24 | Lorigan et al. [43] | |
| CAV (Cyclophosphamide/Doxorubicin/Vincristine) | 14 | White et al. [44] | |
| Soft tissue sarcoma | MAID (Mesna/Doxorubicin/Ifosfamide/Dacarbazin) | 58 | Binh Nguyen et al. [45] |
| Ifosfamide | 18 #, 20 ## | Lorigan et al. [46] |
| Reference | Study Population | Risk Factors (Multivariate Analysis) |
|---|---|---|
| Aagaard et al. (2018) [57] | Patients with solid tumors and DLBCL treated with first-line chemotherapy (n = 9458) | Female sex, age > 65 years, cancer type, disease stage, low albumin, elevated bilirubin, low estimated glomerular filtration rate, infection before baseline, treatment with more than one chemotherapy drug (two to four), receiving taxane-based chemotherapy |
| Aagaard et al. (2020) [59] | Patients with solid tumors who initiated cycle 2 of standard first-line chemotherapy (n = 6885) | Higher predicted risk for FN in the first cycle, platinum- and taxane-containing regimens, concurrent radiotherapy, treatment in cycle 2 compared to later cycles, previous FN or neutropenia, not receiving G-CSF |
| Hosmer et al. (2011) [55] | Elderly patients with breast, lung, colorectal, and prostate cancer (n = 58,053) | Advanced age at diagnosis, number of associated comorbid conditions, receipt of immunosuppressive chemotherapy, receipt of chemotherapy within one month of diagnosis |
| Lyman et al. (2011) [56] * | Patients with breast, lung, colorectal, ovarian cancer, and lymphoma patients (n = 4458) | Prior chemotherapy, use of other immunosuppressive medications, abnormal hepatic and renal function, low white blood count, chemotherapy and planned delivery ≥ 85%, small cell lung cancer, specific classes of chemotherapy (anthracyclines, taxanes, certain alkylating agents [cyclophosphamide, ifosfamide], class I/II topoisomerase inhibitors, platinum derivates [cisplatin, carboplatin], gemcitabine, vinorelbine) |
| Razzaghdoust et al. (2018) [58] * | Patients with various solid tumors and lymphomas (n = 305) | High-risk chemotherapy regimen without G-CSF, intermediate-risk regimen without G-CSF, age > 65 years, elevated ferritin, BMI < 1.73 kg/m2, BSA < 2 m2, estimated glomerular filtration rate < 60 mL/min/1.73 m2, elevated C-reactive protein |
| Isolated Pathogen | Catheter Removal | Antibiotic Therapy | |
|---|---|---|---|
| Choice | Duration | ||
| Coagulase-negative staphylococci | Not necessary Risk factor for recurrence | Vancomycin Oxacillin * Flucloxacillin * Cefazoline * | Catheter removed: 5–7 days Retained catheter: 10–14 days + ALT 10–14 days |
| St. aureus, St. lugdunensis | Yes | Vancomycin Oxacillin * Flucloxacillin * Cefazoline * | ≥14 days. Necessary to rule out complications. Complications: 4–6 weeks |
| Enterococci | Yes Long-term CVC may retain | Vancomycin Ampicillin * | 5–14 days. Retained long-term CVC: 7–14 days + ALT 7–14 days |
| gram-negative bacilli | Yes, especially in case of multiresistant bacteria CVC retaining unsuitable for immunosuppressed patients | Based on severity of disease: Piperacillin/tazobactam *, 4th gen. Cephalosporin *, Carbapenem +/- Aminoglycoside * | 7–14 days |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubeček, O.; Paterová, P.; Novosadová, M. Risk Factors for Infections, Antibiotic Therapy, and Its Impact on Cancer Therapy Outcomes for Patients with Solid Tumors. Life 2021, 11, 1387. https://doi.org/10.3390/life11121387
Kubeček O, Paterová P, Novosadová M. Risk Factors for Infections, Antibiotic Therapy, and Its Impact on Cancer Therapy Outcomes for Patients with Solid Tumors. Life. 2021; 11(12):1387. https://doi.org/10.3390/life11121387
Chicago/Turabian StyleKubeček, Ondřej, Pavla Paterová, and Martina Novosadová. 2021. "Risk Factors for Infections, Antibiotic Therapy, and Its Impact on Cancer Therapy Outcomes for Patients with Solid Tumors" Life 11, no. 12: 1387. https://doi.org/10.3390/life11121387
APA StyleKubeček, O., Paterová, P., & Novosadová, M. (2021). Risk Factors for Infections, Antibiotic Therapy, and Its Impact on Cancer Therapy Outcomes for Patients with Solid Tumors. Life, 11(12), 1387. https://doi.org/10.3390/life11121387






