Comparison of Treatment Effects of Different Iron Chelators in Experimental Models of Sepsis
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals
2.3. Endotoxemia Model
2.3.1. Anesthesia and Surgery
2.3.2. Experimental Timeline
2.3.3. Experimental Groups
2.4. CASP-Induced Sepsis
2.4.1. Anesthesia and Surgery
2.4.2. Experimental Timeline
2.4.3. Experimental Groups
2.5. Intravital Microscopy
2.5.1. Preparation before Microscopy
2.5.2. Microscopy
2.5.3. Offline Video Analysis
2.6. Bacterial Counts
2.7. Microbiome Sequencing
2.8. Plasma Inflammatory Mediators
2.9. Statistical Analysis
3. Results
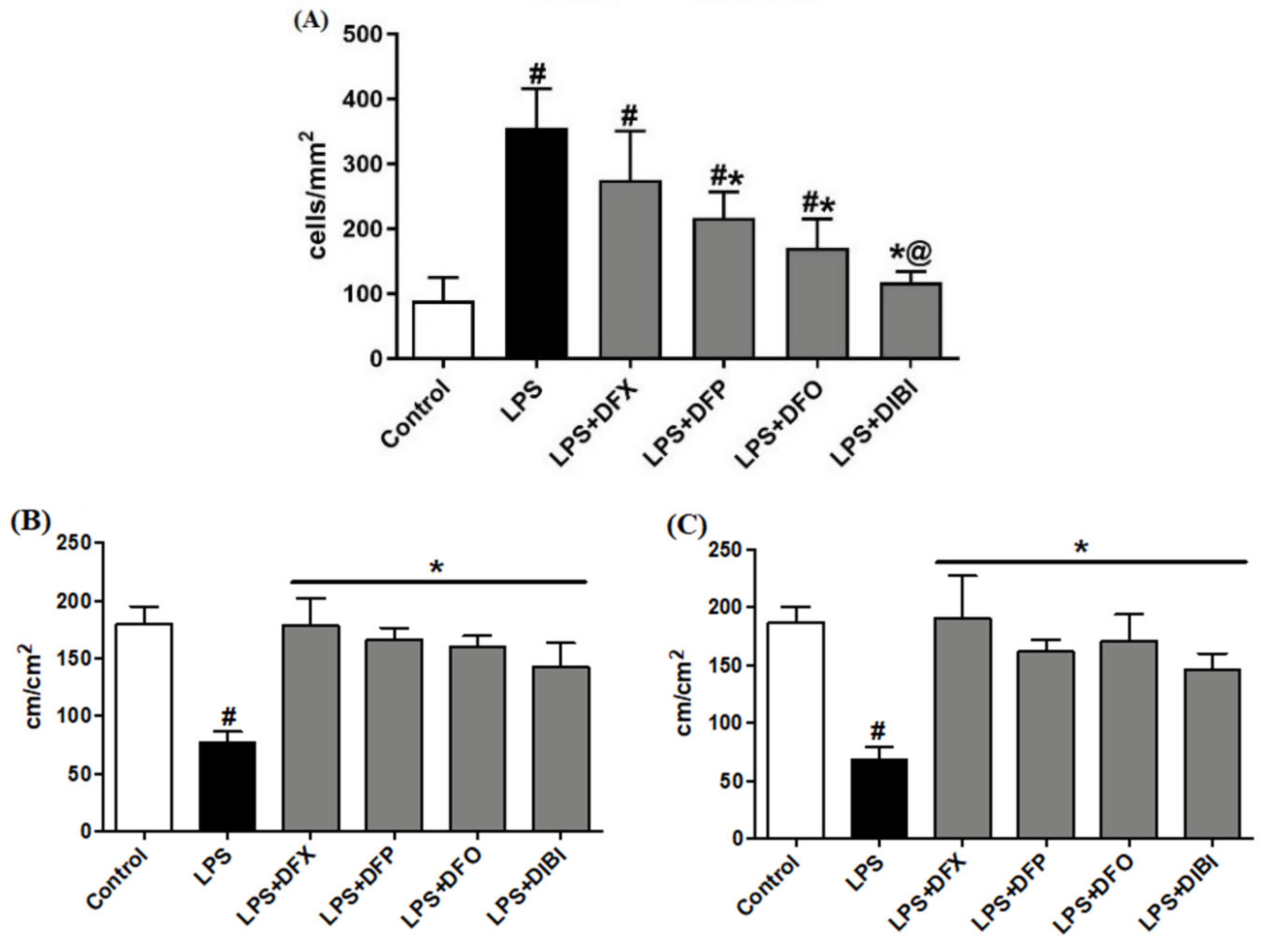
3.1. Endotoxemia Model
3.1.1. Leukocyte–Endothelial Interactions
3.1.2. Capillary Perfusion
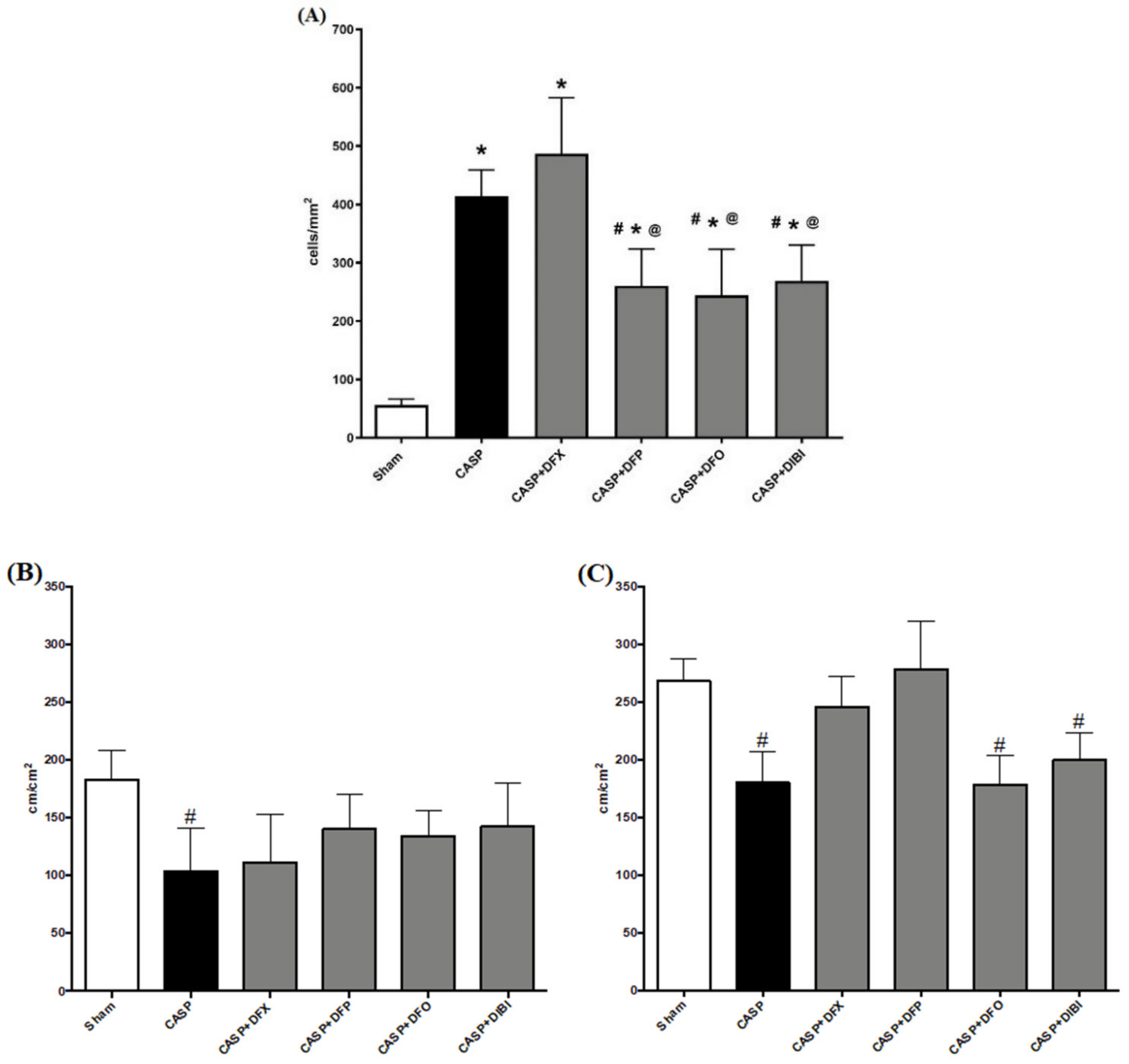
3.2. CASP-Induced Sepsis
3.2.1. Leukocyte–Endothelial Interaction
3.2.2. Capillary Perfusion
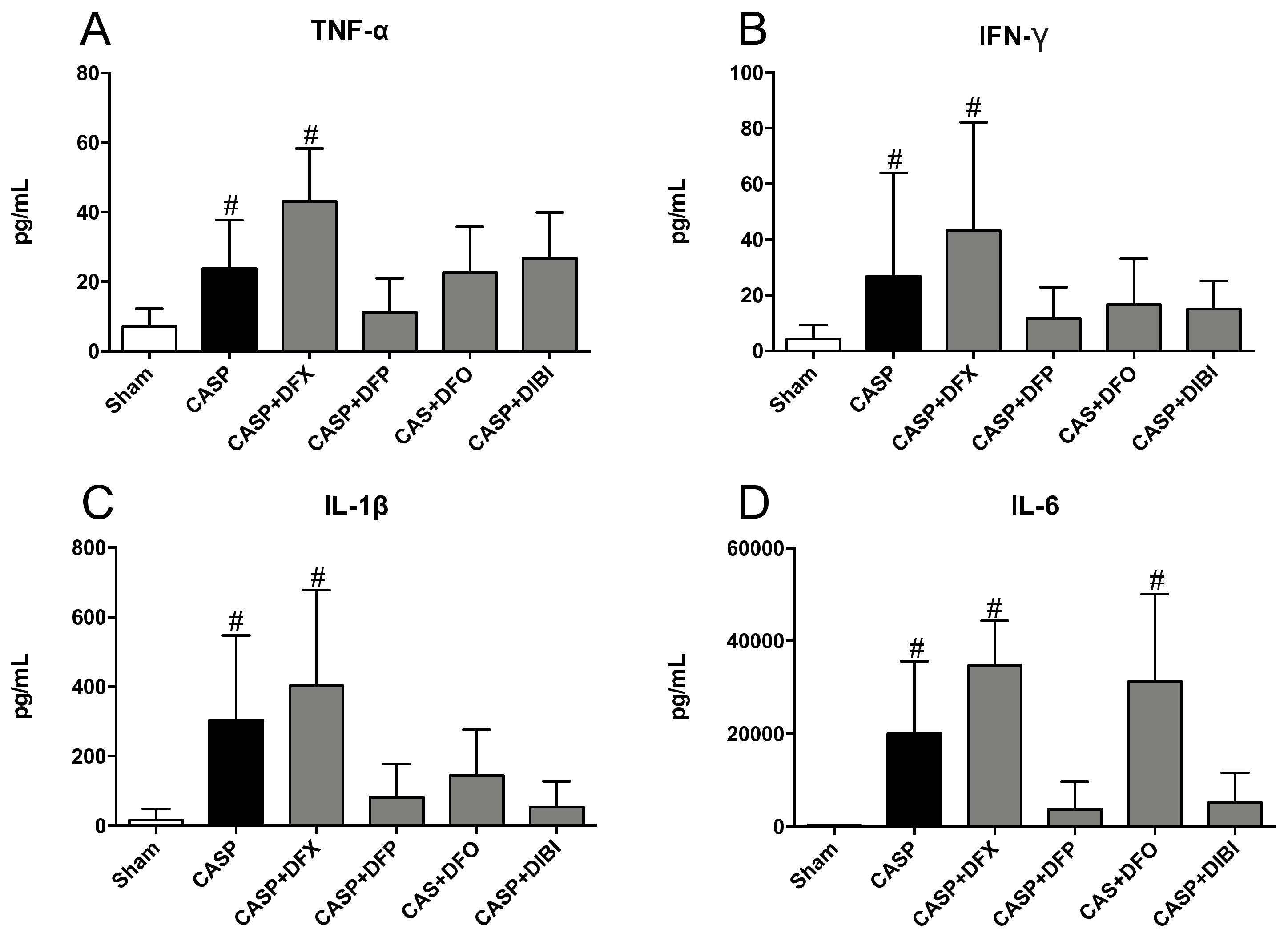
3.2.3. Plasma Inflammatory Mediators
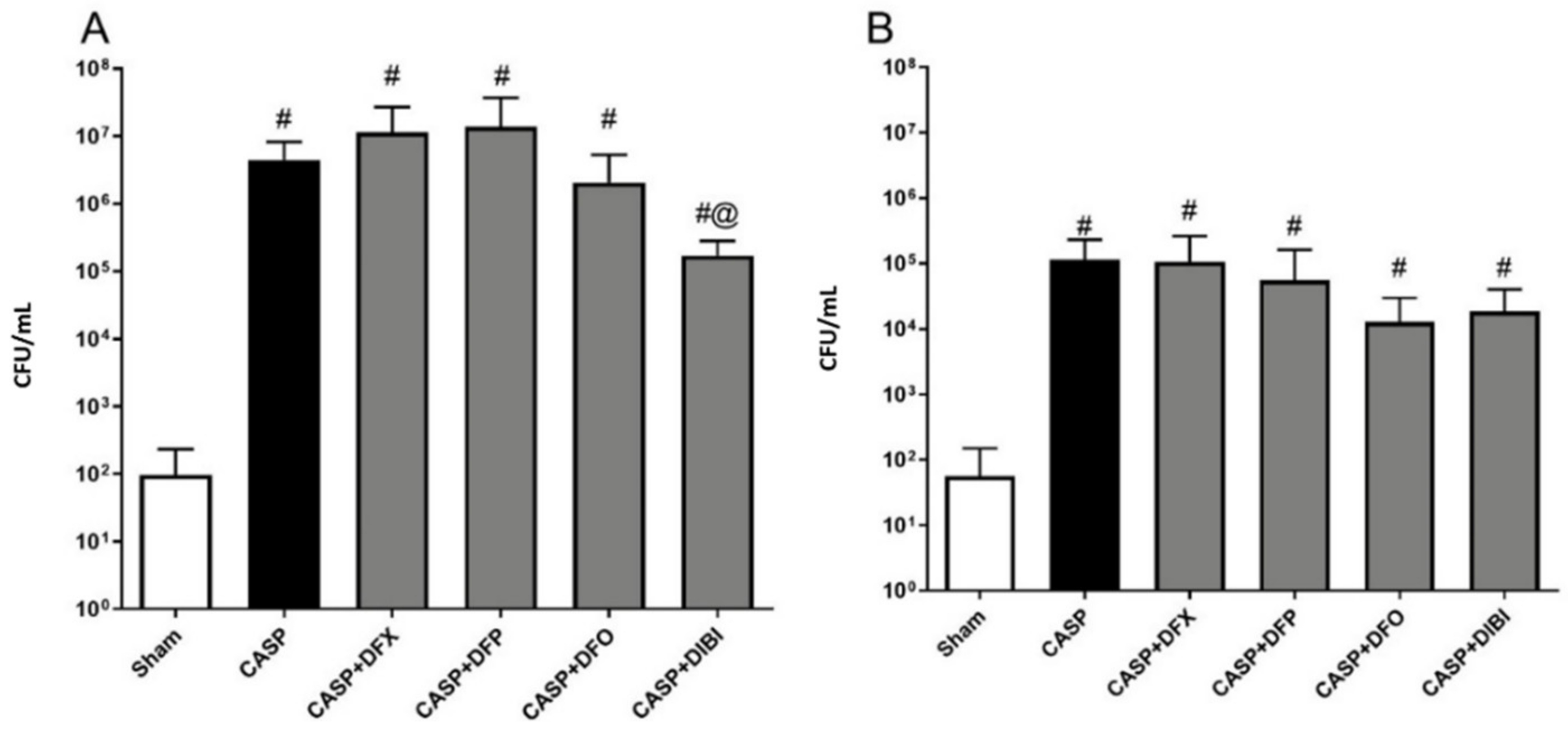
3.2.4. Bacterial Counts
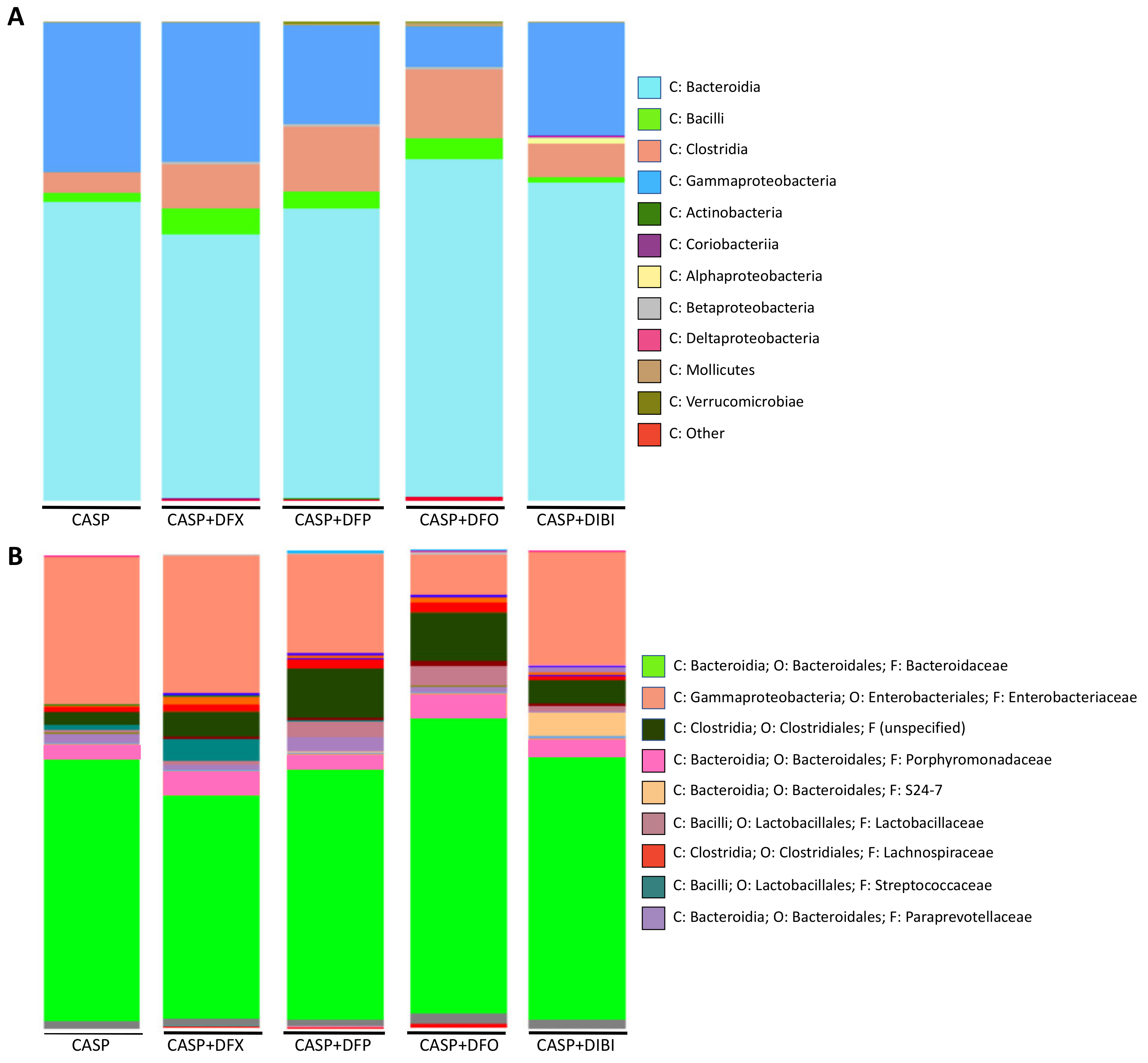
3.2.5. Microbiome Sequencing
4. Discussion
4.1. Iron Chelation in Endotoxemia
4.2. Iron Chelation in CASP-Induced Sepsis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dev, S.; Babitt, J.L. Overview of iron metabolism in health and disease. Hemodial. Int. 2017, 21, S6–S20. [Google Scholar] [CrossRef]
- Dixon, S.J.; Stockwell, B.R. The role of iron and reactive oxygen species in cell death. Nat. Chem. Biol. 2014, 10, 9–17. [Google Scholar] [CrossRef]
- Rocha, M.; Herance, R.; Rovira, S.; Hernandez-Mijares, A.; Victor, V.M. Mitochondrial Dysfunction and Antioxidant Therapy in Sepsis. Infect. Disord. Drug Targets 2012, 12, 161–178. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Matsoso, P.; Pant, S.; Brower, C.; Røttingen, J.A.; Klugman, K.; Davies, S. Access to effective antimicrobials: A worldwide challenge. Lancet 2016, 387, 168–175. [Google Scholar] [CrossRef]
- Neufeld, E.J. Oral chelators deferasirox and deferiprone for transfusional iron overload in thalassemia major: New data, new questions. Blood 2006, 107, 3436–3441. [Google Scholar] [CrossRef]
- Jordan, L.B.; Vekeman, F.; Sengupta, A.; Corral, M.; Guo, A.; Duh, M.S. Persistence and compliance of deferoxamine versus deferasirox in Medicaid patients with sickle-cell disease. J. Clin. Pharm. Ther. 2012, 37, 173–181. [Google Scholar] [CrossRef]
- Desferal® Deferoxamine Mesylate for Injection USP Vials Rx Only Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2007/016267s044lbl.pdf (accessed on 28 December 2020).
- Molitoris, B.A.; Alfrey, P.S.; Miller, N.L.; Hasbargen, J.A.; Kaehney, W.D.; Smith, B.J. Efficacy of intramuscular and intraperitoneal deferoxamine for aluminium chelation. Kidney Int. 1987, 31, 986–991. [Google Scholar] [CrossRef]
- Gumbau-Brisa, R.; Ang, M.T.C.; Holbein, B.E.; Bierenstiel, M. Enhanced Fe3+ binding through cooperativity of 3-hydroxypyridin-4-one groups within a linear co-polymer: Wrapping effect leading to superior antimicrobial activity. BioMetals 2020, 33, 339–351. [Google Scholar] [CrossRef]
- Comeau, A.M.; Douglas, G.M.; Langille, M.G.I. Microbiome Helper: A Custom and Streamlined Workflow for Microbiome Research. Am. Soc. Microbiol. 2017, 2. [Google Scholar] [CrossRef]
- Dickson, K.; Lehmann, C. Inflammatory response to different toxins in experimental sepsis models. Int. J. Mol. Sci. 2019, 20, 4341. [Google Scholar] [CrossRef]
- Thorburn, T.V. Iron-Related Immune Cell Function in Sepsis; Dalhousie University: Halifax, NS, Canada, 2018. [Google Scholar]
- Margraf, A.; Ley, K.; Zarbock, A. Neutrophil Recruitment: From Model Systems to Tissue-Specific Patterns. Trends Immunol. 2019, 40, 613–634. [Google Scholar] [CrossRef] [PubMed]
- Sônego, F.; Alves-Filho, J.C.; Cunha, F.Q. Targeting neutrophils in sepsis. Expert Rev. Clin. Immunol. 2014, 10, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Mittal, R.; Coopersmith, C.M. Redefining the gut as the motor of critical illness. Trends Mol. Med. 2014, 20, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Fokam, D.; Dickson, K.; Kamali, K.; Holbein, B.; Colp, P.; Stueck, A.; Zhou, J.; Lehmann, C. Iron chelation in murine models of systemic inflammation induced by gram-positive and gram-negative toxins. Antibiotics 2020, 9, 283. [Google Scholar] [CrossRef]
- Lehmann, C.; Konig, J.P.; Dettmann, J.; Birnbaum, J.; Kox, W.J. Effects of iloprost, a stable prostacyclin analog, on intestinal leukocyte adherence and microvascular blood flow in rat experimental endotoxemia. Crit. Care Med. 2001, 29, 1412–1416. [Google Scholar] [CrossRef]
- Kupper, S.; Mees, S.T.; Gassmann, P.; Brodde, M.F.; Kehrel, B.; Haier, J. Hydroxyethyl starch normalizes platelet and leukocyte adhesion within pulmonary microcirculation during LPS-induced endotoxemia. Shock 2007, 28, 300–308. [Google Scholar] [CrossRef]
- Brazil, J.C.; Parkos, C.A. Pathobiology of neutrophil-epithelial interactions. Immunol. Rev. 2016, 273, 94–111. [Google Scholar] [CrossRef]
- Lehmann, C.; Islam, S.; Jarosch, S.; Zhou, J.; Hoskin, D.; Greenshields, A.; Sharawy, N.; Sczcesniak, A.; Kelly, M.; Wafa, K.; et al. The Utility of Iron Chelators in the Management of Inflammatory Disorders. Mediat. Inflamm. 2015, 2015, 516740. [Google Scholar] [CrossRef]
- Heli, H.; Mirtorabi, S.; Karimian, K. Advances in iron chelation: An update. Expert Opin. Ther. Pat. 2011, 21, 819–856. [Google Scholar] [CrossRef]
- Badeli, H.; Baghersalimi, A.; Eslami, S.; Saadat, F.; Rad, A.H.; Basavand, R.; Papkiadeh, S.R.; Darbandi, B.; Kooti, W.; Peluso, I. Early Kidney Damage Markers after Deferasirox Treatment in Patients with Thalassemia Major: A Case-Control Study. Oxidative Med. Cell. Longev. 2019, 2019, 5461617. [Google Scholar] [CrossRef]
- Matharu, N.M.; Butler, L.M.; Rainger, G.E.; Gosling, P.; Vohra, R.K.; Nash, G.B. Mechanisms of the anti-inflammatory effects of hydroxyethyl starch demonstrated in a flow-based model of neutrophil recruitment by endothelial cells. Crit. Care Med. 2008, 36, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Nohé, B.; Johannes, T.; Reutershan, J.; Rothmund, A.; Haeberle, H.A.; Ploppa, A.; Schroeder, T.H.; Dieterich, H.-J. Synthetic colloids attenuate leukocyte-endothelial interactions by inhibition of integrin function. Anesthesiology 2005, 103, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Handrigan, M.T.; Burns, A.R.; Donnachie, E.M.; Bowden, R.A. Hydroxyethyl starch inhibits neutrophil adhesion and transendothelial migration. Shock 2005, 24, 434–439. [Google Scholar] [CrossRef]
- Pascual, J.L.; Ferri, L.E.; Chaudhury, P.; Seely, A.J.E.; Campisi, G.; Giannias, B.; Evans, D.C.; Christou, N.V. Hemorrhagic shock resuscitation with a low molecular weight starch reduces neutrophil-endothelial interactions and vessel leakage in vivo. Surg. Infect. 2001, 2, 275–288. [Google Scholar] [CrossRef]
- Islam, S.; Jarosch, S.; Zhou, J.; Parquet, M.D.C.; Toguri, J.T.; Colp, P.; Holbein, B.E.; Lehmann, C. Anti-inflammatory and anti-bacterial effects of iron chelation in experimental sepsis. J. Surg. Res. 2016, 200, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Fokam, D.; Aali, M.; Dickson, K.; Scott, C.; Holbein, B.; Zhou, J.; Lehmann, C. The novel iron chelator, DIBI, attenuates inflammation and improves outcome in colon ascendens stent peritonitis-induced experimental sepsis. Clin. Hemorheol. Microcirc. 2020, 76, 241–261. [Google Scholar] [CrossRef]
- Ritter, C.; Andrades, M.E.; Reinke, A.; Menna-Barreto, S.; Moreira, J.C.F.; Dal-Pizzol, F. Treatment with N-acetylcysteine plus deferoxamine protects rats against oxidative stress and improves survival in sepsis. Crit. Care Med. 2004, 32, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Messaris, E.; Antonakis, P.T.; Memos, N.; Chatzigianni, E.; Leandros, E.; Konstadoulakis, M.M. Deferoxamine administration in septic animals: Improved survival and altered apoptotic gene expression. Int. Immunopharmacol. 2004, 4, 455–459. [Google Scholar] [CrossRef]
- Schulte, W.; Bernhagen, J.; Bucala, R. Cytokines in Sepsis: Potent Immunoregulators and Potential Therapeutic Targets—An Updated View. Mediat. Inflamm. 2013, 2013, 165974. [Google Scholar] [CrossRef]
- Traeger, T.; Koerner, P.; Kessler, W.; Cziupka, K.; Diedrich, S.; Busemann, A.; Heidecke, C.-D.; Maier, S. Colon ascendens stent peritonitis (CASP)—A standardized model for polymicrobial abdominal sepsis. J. Vis. Exp. 2010. [Google Scholar] [CrossRef]
- Emmanuilidis, K.; Weighardt, H.; Maier, S.; Gerauer, K.; Fleischmann, T.; Zheng, X.X.; Hancock, W.W.; Holzmann, B.; Heidecke, C.-D. Critical role of kupffer cell-derived IL-10 for host defense in septic peritonitis. J. Immunol. 2001, 167, 3919–3927. [Google Scholar] [CrossRef] [PubMed]
- Jusek, G.; Reim, D.; Tsujikawa, K.; Holzmann, B. Deficiency of the CGRP receptor component RAMP1 attenuates immunosuppression during the early phase of septic peritonitis. Immunobiology 2012, 217, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Holbein, B.E.; Mira De Orduña, R. Effect of trace iron levels and iron withdrawal by chelation on the growth of Candida albicans and Candida vini. FEMS Microbiol. Lett. 2010, 307, 19–24. [Google Scholar] [CrossRef]
- Savage, K.A.; del Carmen Parquet, M.; Allan, D.S.; Davidson, R.J.; Holbein, B.E.; Lilly, E.A.; Fidel, P.L. Iron restriction to clinical isolates of candida albicans by the novel chelator dibi inhibits growth and increases sensitivity to azoles in vitro and in vivo in a murine model of experimental vaginitis. Antimicrob. Agents Chemother. 2018, 62. [Google Scholar] [CrossRef] [PubMed]
- Wexler, H.M. Bacteroides: The good, the bad, and the nitty-gritty. Clin. Microbiol. Rev. 2007, 20, 593–621. [Google Scholar] [CrossRef] [PubMed]
- Choi, V.M.; Herrou, J.; Hecht, A.L.; Teoh, W.P.; Turner, J.R.; Crosson, S.; Wardenburg, J.B. Activation of Bacteroides fragilis toxin by a novel bacterial protease contributes to anaerobic sepsis in mice. Nat. Med. 2016, 22, 563–567. [Google Scholar] [CrossRef]
- Rocha, E.R.; Bergonia, H.A.; Gerdes, S.; Jeffrey Smith, C. Bacteroides fragilis requires the ferrous-iron transporter FeoAB and the CobN-like proteins BtuS1 and BtuS2 for assimilation of iron released from heme. Microbiologyopen 2019, 8, 669. [Google Scholar] [CrossRef]
- Mayr, F.B.; Yende, S.; Angus, D.C. Epidemiology of severe sepsis. Virulence 2014, 5, 4–11. [Google Scholar] [CrossRef]
- Lesic, B.; Foulon, J.; Carniel, E. Comparison of the effects of deferiprone versus deferoxamine on growth and virulence of Yersinia enterocolitica. Antimicrob. Agents Chemother. 2002, 46, 1741–1745. [Google Scholar] [CrossRef]
- Thompson, M.G.; Corey, B.W.; Si, Y.; Craft, D.W.; Zurawski, D.V. Antibacterial activities of iron chelators against common nosocomial pathogens. Antimicrob. Agents Chemother. 2012, 56, 5419–5421. [Google Scholar] [CrossRef]
- Del Carmen Parquet, M.; Savage, K.A.; Allan, D.S.; Ang, M.T.C.; Chen, W.; Logan, S.M.; Holbein, B.E. Antibiotic-Resistant Acinetobacter baumannii Is Susceptible to the Novel Iron-Sequestering Anti-infective DIBI in Vitro and in Experimental Pneumonia in Mice. Antimicrob. Agents Chemother. 2019, 63, e00855-19. [Google Scholar]
- Peng, Z.; Jin, D.; Kim, H.B.; Stratton, C.W.; Wu, B.; Tang, Y.W.; Suna, X. Update on antimicrobial resistance in Clostridium difficile: Resistance mechanisms and antimicrobial susceptibility testing. J. Clin. Microbiol. 2017, 55, 1998–2008. [Google Scholar] [CrossRef] [PubMed]
- Hirabayashi, K.; Shiohara, M.; Suzuki, T.; Saito, S.; Tanaka, M.; Yanagisawa, R.; Tsuruta, G.; Fukuyama, T.; Hidaka, Y.; Nakazawa, Y.; et al. Critical illness polyneuropathy and myopathy caused by Bacillus cereus sepsis in acute lymphoblastic leukemia. J. Pediatr. Hematol. Oncol. 2012, 34, e110–e113. [Google Scholar] [CrossRef]
- Luyt, C.E.; Hékimian, G.; Koulenti, D.; Chastre, J. Microbial cause of ICU-acquired pneumonia: Hospital-acquired pneumonia versus ventilator-associated pneumonia. Curr. Opin. Crit. Care 2018, 24, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, H.; Becker, K.; Dohmen, P.M.; Petrosillo, N.; Spencer, M.; van Rijen, M.; Wechsler-Fördös, A.; Pujol, M.; Dubouix, A.; Garau, J. Staphylococcus aureus and surgical site infections: Benefits of screening and decolonization before surgery. J. Hosp. Infect. 2016, 94, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Magill, S.S.; Edwards, J.R.; Bamberg, W.; Beldavs, Z.G.; Dumyati, G.; Kainer, M.A.; Lynfield, R.; Maloney, M.; McAllister-Hollod, L.; Nadle, J.; et al. Multistate point-prevalence survey of health care-associated infections. N. Engl. J. Med. 2014, 370, 1198–1208. [Google Scholar] [CrossRef] [PubMed]
- Allan, D.S.; Del Carmen Parquet, M.; Savage, K.A.; Holbein, B.E. Iron sequestrant DIBI, a potential alternative for nares decolonization of methicillin-resistant Staphylococcus aureus, is anti-infective and inhibitory for mupirocin-resistant isolates. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef]
- Del Parquet, M.C.; Savage, K.A.; Allan, D.S.; Davidson, R.J.; Holbein, B.E. Novel Iron-Chelator DIBI Inhibits Staphylococcus aureus Growth, Suppresses Experimental MRSA Infection in Mice and Enhances the Activities of Diverse Antibiotics in vitro. Front. Microbiol. 2018, 9, 1811. [Google Scholar] [CrossRef]
- Gentile, L.F.; Nacionales, D.C.; Lopez, M.C.; Vanzant, E.; Cuenca, A.; Cuenca, A.G.; Ungaro, R.; Szpila, B.E.; Larson, S.; Joseph, A.; et al. Protective immunity and defects in the neonatal and elderly immune response to sepsis. J. Immunol. 2014, 192, 3156–3165. [Google Scholar] [CrossRef]
- Gentile, L.F.; Nacionales, D.C.; Lopez, M.C.; Vanzant, E.; Cuenca, A.; Szpila, B.E.; Cuenca, A.G.; Joseph, A.; Moore, F.A.; Leeuwenburgh, C.; et al. Host responses to sepsis vary in different low-lethality murine models. PLoS ONE 2014, 9, e94404. [Google Scholar] [CrossRef] [PubMed]
- Whiles, B.; Deis, A.; Miller, P.; Simpson, S. Comorbid Conditions Predict Outcomes in Patients With Severe Sepsis. Chest 2016, 149, A170. [Google Scholar] [CrossRef]
- Huang, W.; Xie, R.; Hong, Y.; Chen, Q. Association between comorbid chronic obstructive pulmonary disease and prognosis of patients admitted to the intensive care unit for non-COPD reasons: A retrospective cohort study. Int. J. COPD 2020, 15, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-H.; Lai, C.-C.; Wang, Y.-H.; Wang, C.-Y.; Wang, H.-C.; Yu, C.-J.; Chen, L. The impact of sepsis on the outcomes of COPD patients: A population-based cohort study. J. Clin. Med. 2018, 7, 393. [Google Scholar] [CrossRef] [PubMed]
- Sinapidis, D.; Kosmas, V.; Vittoros, V.; Koutelidakis, I.M.; Pantazi, A.; Stefos, A.; Katsaros, K.E.; Akinosoglou, K.; Bristianou, M.; Toutouzas, K.; et al. Progression into sepsis: An individualized process varying by the interaction of comorbidities with the underlying infection. BMC Infect. Dis. 2018, 18, 242. [Google Scholar] [CrossRef] [PubMed]
- Perlman, R.L. Mouse models of human disease: An evolutionary perspective. Evol. Med. Public Health 2016, 2016, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Vlahakos, D.; Arkadopoulos, N.; Kostopanagiotou, G.; Siasiakou, S.; Kaklamanis, L.; Degiannis, D.; Demonakou, M.; Smyrniotis, V. Deferoxamine attenuates lipid peroxidation, blocks interleukin-6 production, ameliorates sepsis inflammatory response syndrome, and confers renoprotection after acute hepatic ischemia in pigs. Artif. Organs. 2012, 36, 400–408. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lehmann, C.; Aali, M.; Zhou, J.; Holbein, B. Comparison of Treatment Effects of Different Iron Chelators in Experimental Models of Sepsis. Life 2021, 11, 57. https://doi.org/10.3390/life11010057
Lehmann C, Aali M, Zhou J, Holbein B. Comparison of Treatment Effects of Different Iron Chelators in Experimental Models of Sepsis. Life. 2021; 11(1):57. https://doi.org/10.3390/life11010057
Chicago/Turabian StyleLehmann, Christian, Maral Aali, Juan Zhou, and Bruce Holbein. 2021. "Comparison of Treatment Effects of Different Iron Chelators in Experimental Models of Sepsis" Life 11, no. 1: 57. https://doi.org/10.3390/life11010057
APA StyleLehmann, C., Aali, M., Zhou, J., & Holbein, B. (2021). Comparison of Treatment Effects of Different Iron Chelators in Experimental Models of Sepsis. Life, 11(1), 57. https://doi.org/10.3390/life11010057





