Osteoclasts and Microgravity
Abstract
1. Introduction
2. Materials and Methods
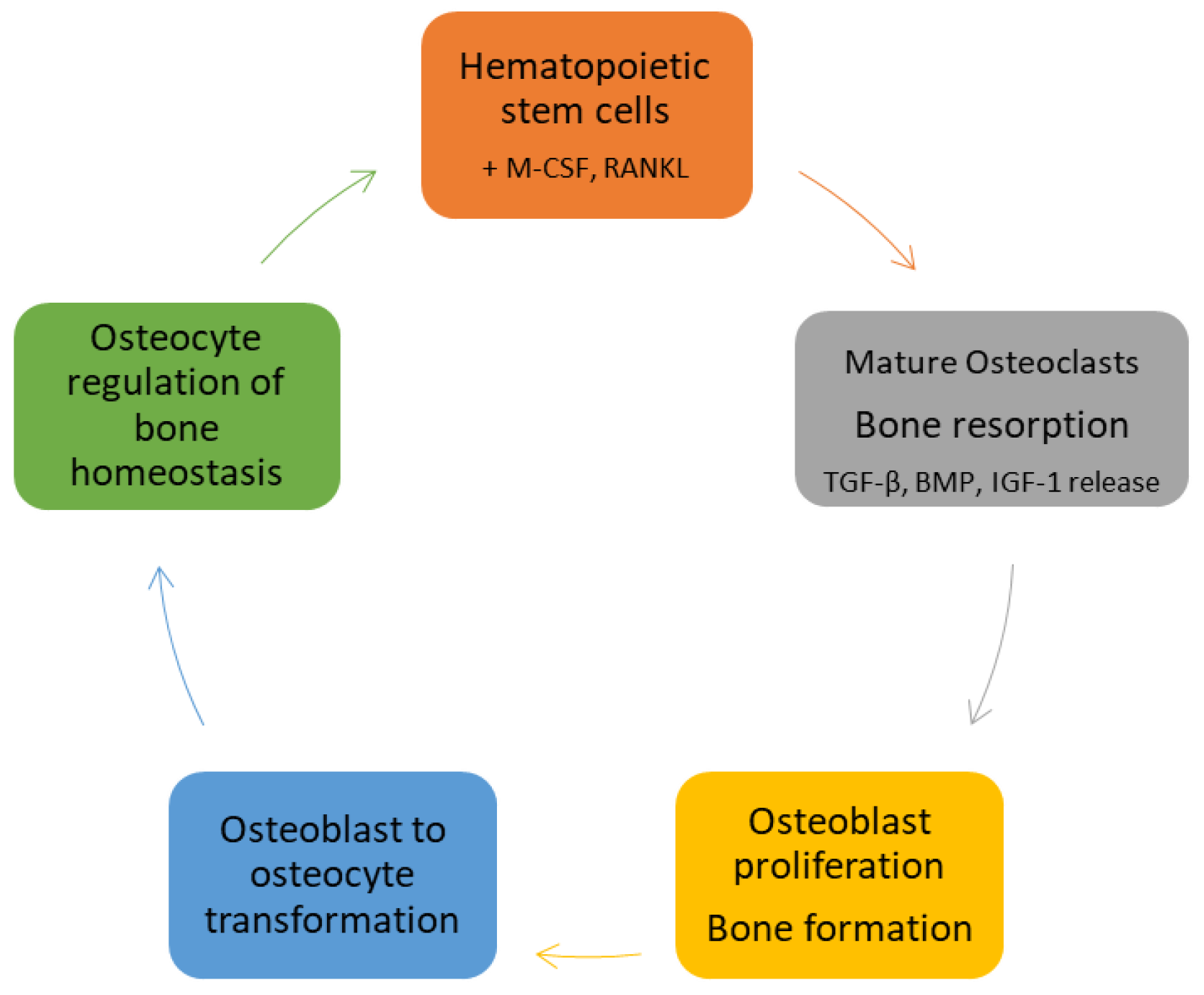
3. Bone Homeostasis
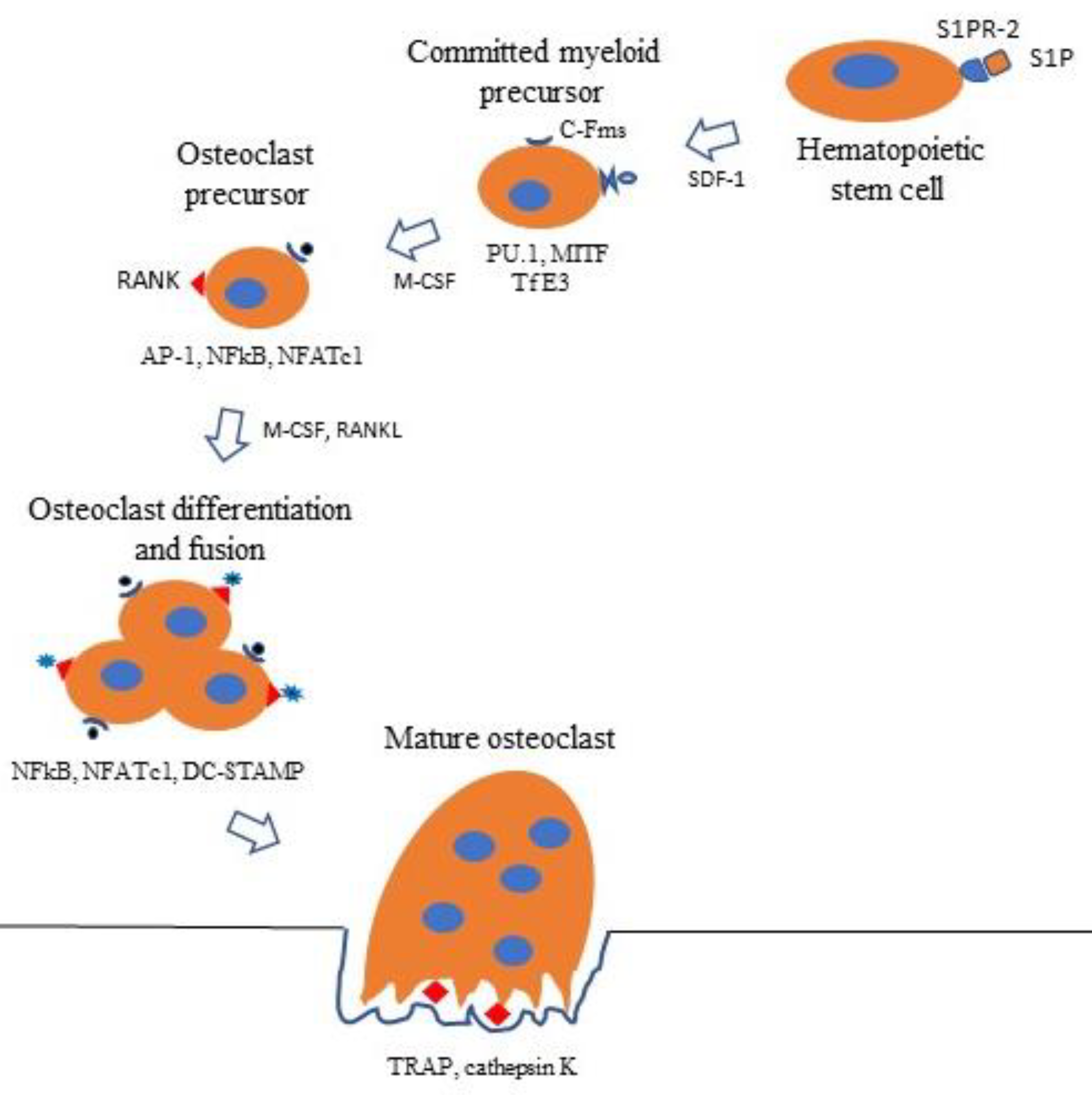
4. Osteoclast Stem Cells
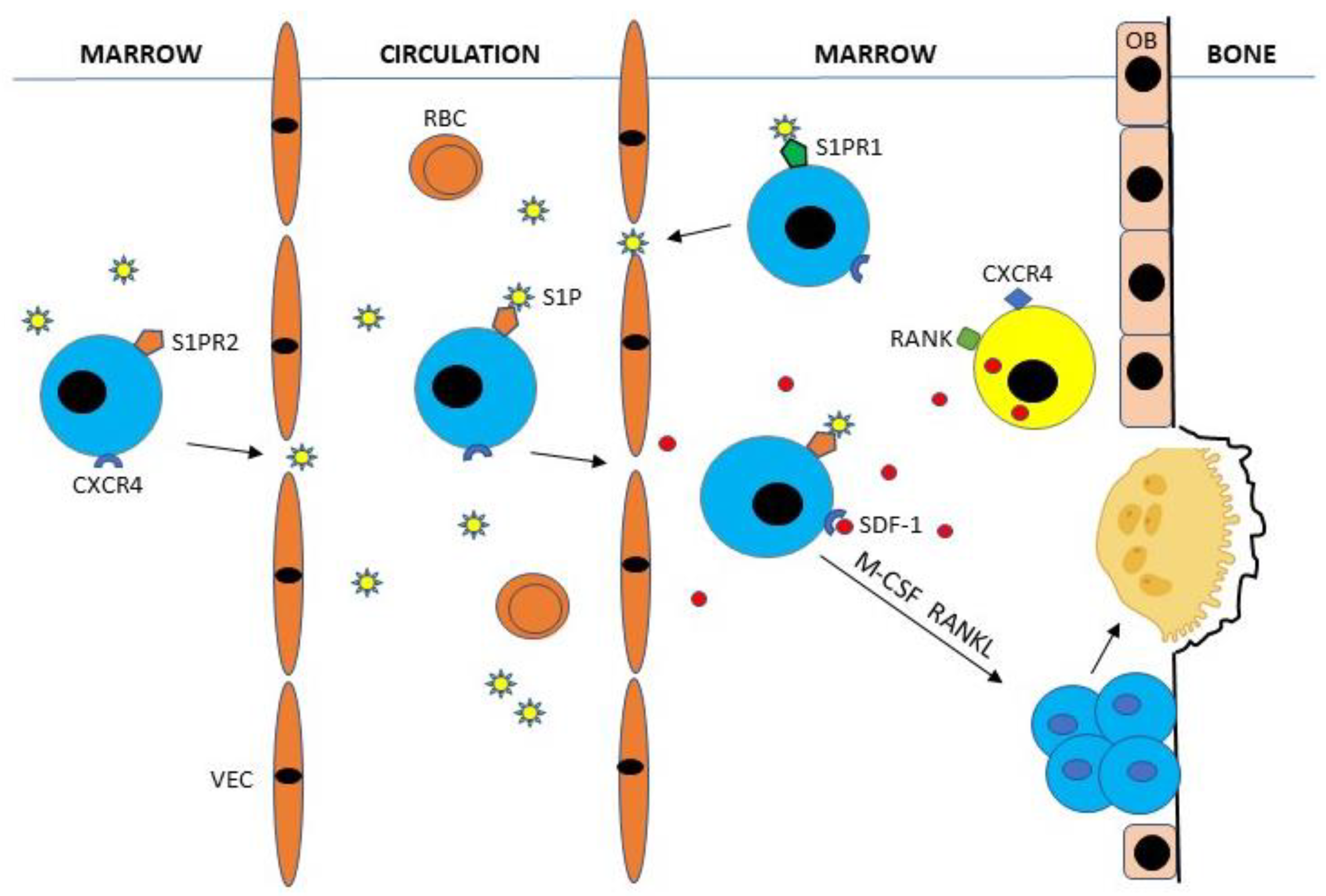
4.1. Osteoclast Stem Cell Niche
4.2. Osteoclast Stem Cell Circulation
5. Osteoclast Differentiation
5.1. M-CSF Pathway
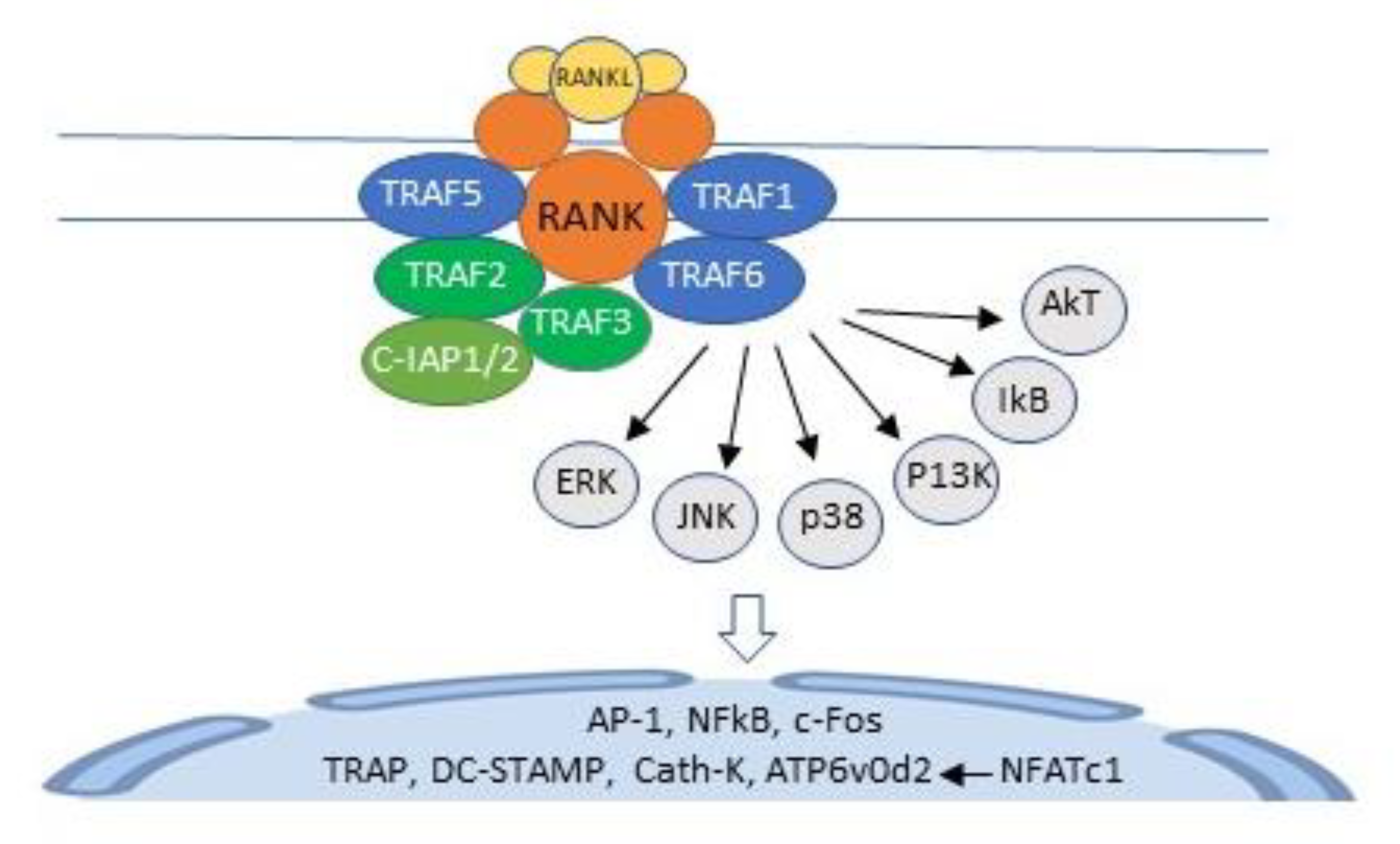
5.2. RANKL Pathway
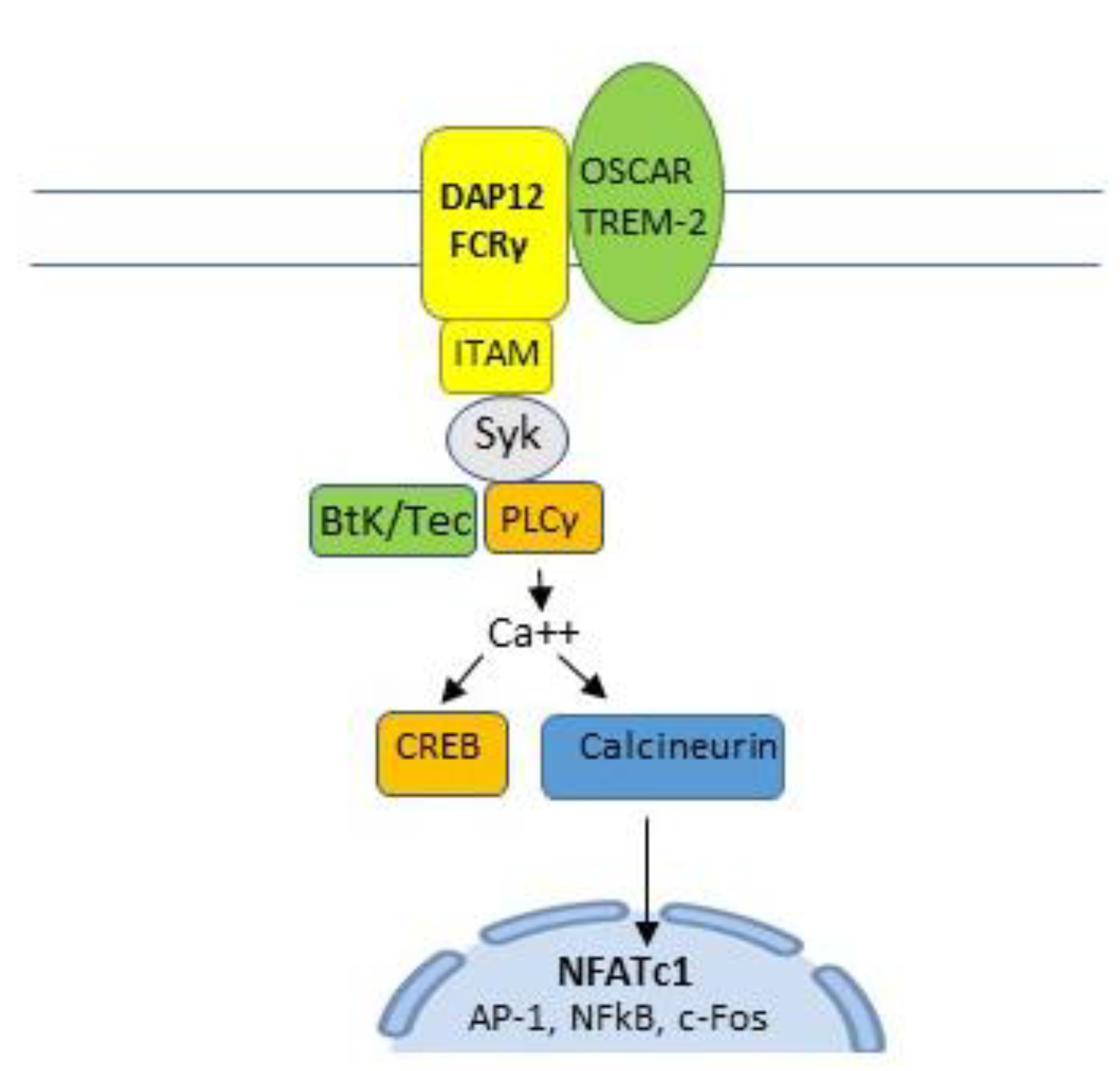
5.3. Calcineurin Pathway
6. Osteoclast Cytoskeleton
6.1. Cytoskeleton Elements
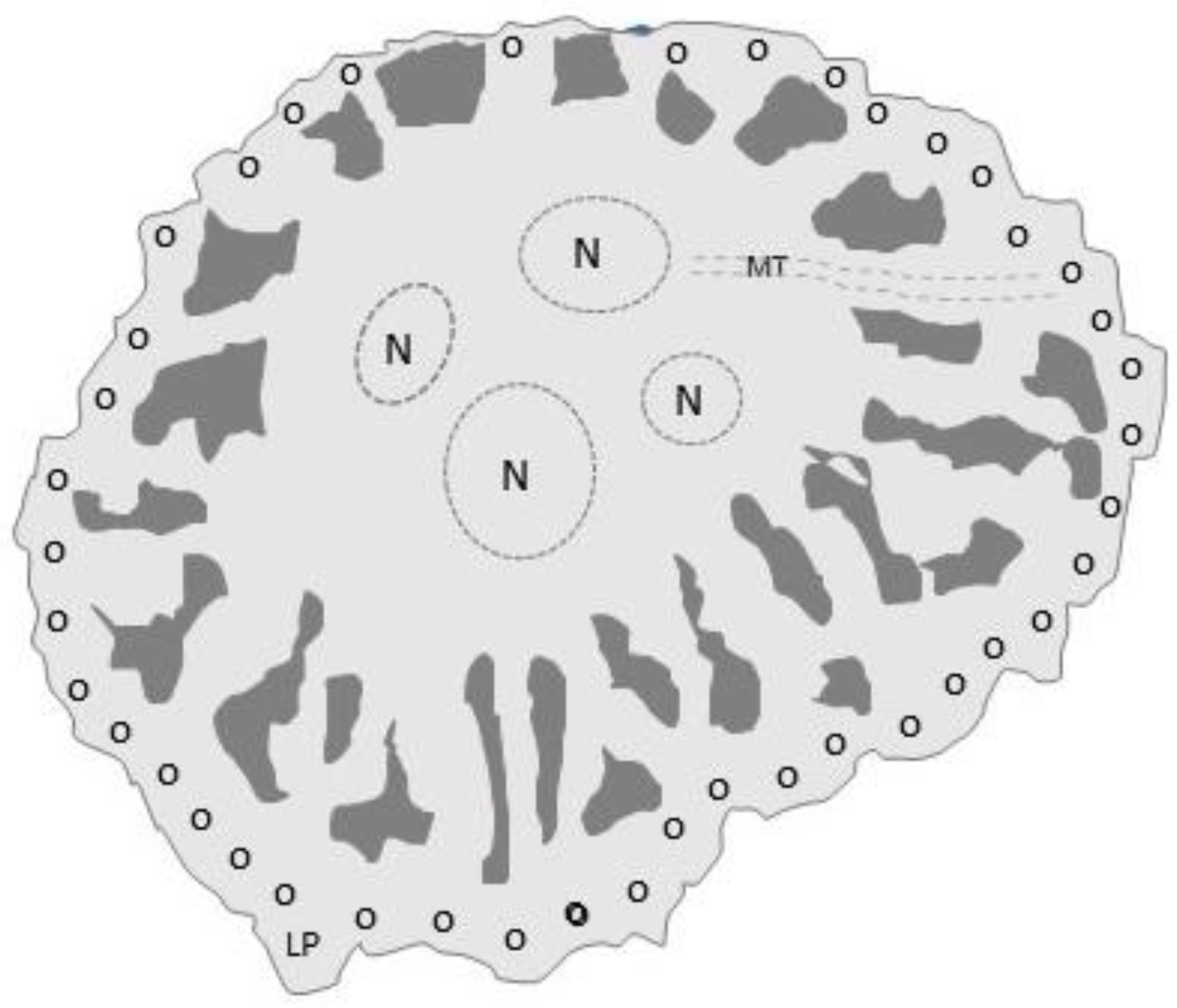
6.1.1. The Sealing Zone
6.1.2. The Actin Cytoskeleton
6.1.3. Crosstalk between Actin and Microtubular Networks
6.1.4. Intermediate and Septin Filaments
7. Immunoregulation
7.1. Cytokines
7.1.1. Osteoclastogenic Cytokines
7.1.2. Anti-Osteoclastogenic Cytokines
8. Mechanotransduction
8.1. Mechanotransduction in Space (10−6 g)
8.1.1. Studies Using Live Animals
8.1.2. Studies Using Cell Cultures
8.1.3. Summary
8.2. Mechanotransduction in Simulated Microgravity (g ≥ 10−3)
8.2.1. Studies Using Live Animals
8.2.2. Studies Using Cell Cultures
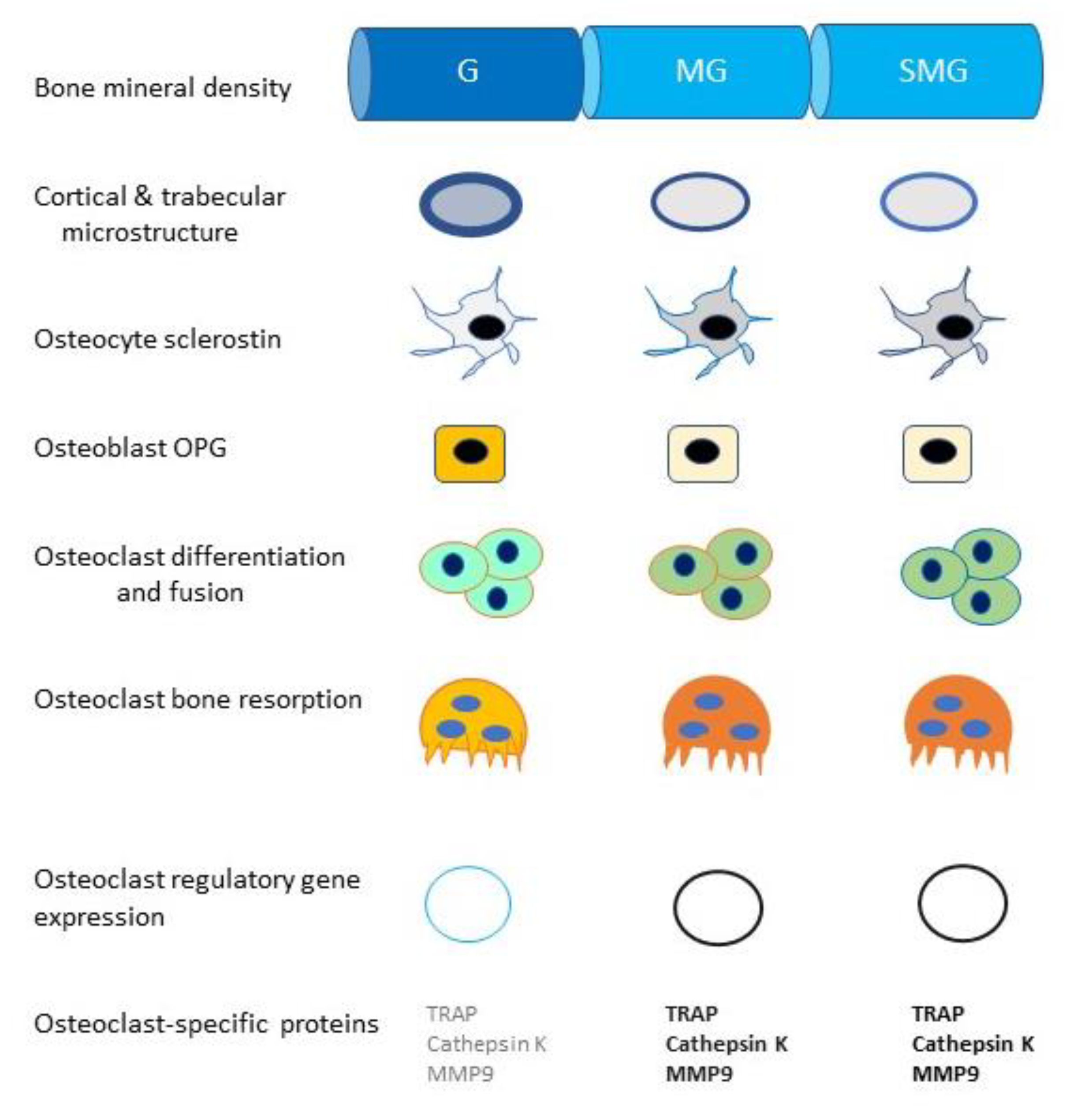
9. Discussion
9.1. Biphosphonates
9.2. Anti-RANKL Antibody
9.3. Cathepsin K Inhibitor
9.4. Anti-Sclerostin Antibody
9.5. Osteoprotegerin
9.6. Melatonin
9.7. Insulin-Like Growth Factor-1
10. Future Prospects
11. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Okamoto, K.; Nakashima, T.; Shinohara, M.; Negishi-Koga, T.; Komatsu, N.; Terashima, A.; Sawa, S.; Nitta, T.; Takayanagi, H. Osteoimmunology: The conceptual framework unifying the immune and skeletal systems. Physiol. Rev. 2017, 97, 1295–1349. [Google Scholar] [CrossRef] [PubMed]
- Blangy, A.; Bompard, G.; Guerit, D.; Marie, P.; Maurin, J.; Morel, A.; Vives, V. The osteoclast—Current understanding and therapeutic perspectives for osteoporosis. J. Cell Sci. 2020, 133, jcs244798. [Google Scholar] [CrossRef] [PubMed]
- Svitkina, T. The actin cytoskeleton and actin-based motility. Cold Spring Harb. Perspect. Biol. 2018, 10, a018267. [Google Scholar] [CrossRef] [PubMed]
- LaFlamme, S.E.; Mathew-Steiner, S.; Singh, N.; Colello-Borges, D.; Nieves, B. Integrin and microtubule crosstalk in the regulation of cellular processes. Cell Mol. Life Sci. 2018, 75, 4177–4185. [Google Scholar] [CrossRef]
- Martínez, P.T.; Navajas, P.L.; Lietha, D. FAK Structure and regulation by membrane interactions and force in focal adhesions. Biomolecules 2020, 10, 179. [Google Scholar] [CrossRef]
- Burke, B. Chain reaction: LINC complexes and nuclear positioning. F1000Research 2019, 8, 136. [Google Scholar] [CrossRef]
- Howard, R.; Scheiner, A.; Cunningham, J.; Gatenby, R. Cytoplasmic convection currents and intracellular temperature gradients. PLoS Comput. Biol. 2019, 15, e1007372. [Google Scholar] [CrossRef]
- Smith, J.K. Microgravity, bone homeostasis, and insulin-like growth factor-1. Appl. Sci. 2020, 10, 4433. [Google Scholar] [CrossRef]
- Iwamoto, J.; Takeda, T.; Sato, Y. Interventions to prevent bone loss in astronauts during space flight. Keio J. Med. 2005, 54, 55–59. [Google Scholar] [CrossRef]
- Cavanagh, P.R.; Licata, A.A.; Rice, A.J. Exercise and pharmacological countermeasures for bone loss during long-duration space flight. Gravit. Space Biol. Bull. 2005, 18, 39–58. [Google Scholar]
- Smith, S.M.; McCoy, T.; Gazda, D.; Morgan, J.L.L.; Heer, M.; Zwart, S.R. Space flight calcium: Implications for astronaut health, spaceflight operations, and Earth. Nutrients 2012, 4, 2047–2068. [Google Scholar] [CrossRef] [PubMed]
- Leblanc, A.; Matsumoto, T.; Jones, J.; Shapiro, J.; Lang, T.; Shackelford, L.; Smith, S.M.; Evans, H.; Spector, E.; Ploutz-Snyder, R.; et al. Biphosphonates as a supplement to exercise to protect bone during long-duration spaceflight. Osteoporos. Int. 2013, 24, 2105–2114. [Google Scholar] [CrossRef] [PubMed]
- NASA—Food for Space Flight. Available online: https:/www.nasa.gov/specials/apollo50thy/index.html (accessed on 12 June 2020).
- Goto, Y.; Aoyama, M.; Sekiya, T.; Katika, H.; Waguri-Nagaya, Y.; Miyazawa, K.; Asai, K.; Goto, S. CXCR4+ CD45− cells are niche forming for osteoclastogenesis via the SDF-1, CXCL7, and CX3CL1 signaling pathways in bone marrow. Stem Cells 2016, 34, 2733–2741. [Google Scholar] [CrossRef] [PubMed]
- Ishii, M.; Egen, J.G.; Klauschen, F.; Meier-Schellersheim, M.; Saeki, Y.; Vacher, J.; Proia, R.L.; Germain, R.N. Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature 2009, 458, 524–528. [Google Scholar] [CrossRef]
- Ishii, T.; Shimazu, Y.; Nishiyama, I.; Kikuta, J.; Ishii, M. The role of sphingosine 1-phosphjate in migration of osteoclast precursors; an application of intravital two-photon microscopy. Mol. Cells 2011, 31, 399–403. [Google Scholar] [CrossRef]
- Tamura, M.; Sato, M.M.; Nashimoto, M. Regulation of CXCL12 expression by canonical Wnt signaling in bone marrow stromal cells. Int. J. Biochem. Cell Biol. 2011, 43, 760–767. [Google Scholar] [CrossRef]
- Boyce, B.F. Advances in the regulation of osteoclasts and osteoclast functions. J. Dent. Res. 2013, 92, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Cappellen, D.; Luong-Nguyen, N.H.; Bongiovanni, S.; Grenet, O.; Wanke, C.; Šuša, M. Transcriptional program of mouse osteoclast differentiation governed by the macrophage colony-stimulating factor and the ligand for receptor activator of NFkB. J. Biol. Chem. 2002, 277, 21971–21982. [Google Scholar] [CrossRef]
- Kim, J.H.; Kin, N. Regulation of NFATc1 in osteoclast differentiation. J. Bone Metab. 2014, 21, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Zawawi, M.S.F.; Dharmapatni, A.A.S.S.K.; Cantley, M.D.; McHugh, K.P.; Haynes, D.R.; Crotti, T.N. Regulation of ITAM adapter molecules and their receptors by inhibition of calcineurin-NFAT signaling during late stage osteoclast differentiation. Biochem. Biophys. Res. Commun. 2012, 427, 404–409. [Google Scholar] [CrossRef]
- Lamothe, B.; Webster, W.K.; Gopinathan, A.; Besse, A.; Campos, A.D.; Darnay, B.G. TRAF-s ubiquitin ligase is essential for RANKL signaling and osteoclast differentiation. Biochem. Biophys. Res. Commun. 2007, 359, 1044–1049. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Udagawa, N.; Takahashi, N. Action of RANKL and OPG for osteoclastogenesis. Crit. Rev. Eukaryot. Gene Expr. 2009, 19, 61–72. [Google Scholar] [CrossRef]
- Negishi-Koga, T.; Takayanagi, H. Ca2+ NFATc1 signaling is an essential axis of osteoclast differentiation. Immunol. Rev. 2009, 231, 241–256. [Google Scholar] [CrossRef]
- Takayanagi, H. The role of NFAT in osteoclast formation. Ann. N. Y. Acad. Sci. 2007, 16, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Asagiri, M.; Takayanagi, H. The molecular understanding of osteoclast differentiation. Bone 2007, 40, 251–264. [Google Scholar] [CrossRef]
- Shinohara, M.; Takayanagi, H. Novel osteoclast signaling mechanism. Curr. Osteoporos. Rep. 2007, 5, 67–72. [Google Scholar] [CrossRef]
- Takayanagi, H. Mechanistic insight into osteoclast differentiation in osteoimmunology. J. Mol. Med. 2005, 83, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; Takeshita, S.; Barker, J.E.; Kanagawa, O.; Ross, F.P.; Teitelbaum, S.L. TNF-alpha induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J. Clin. Investig. 2000, 106, 1481–1488. [Google Scholar] [CrossRef]
- Kitaura, H.; Sands, M.S.; Aya, K.; Zhou, P.; Hirayama, T.; Uthgenannt, B.; Wei, S.; Takeshita, S.; Novack, D.V.; Silva, M.J.; et al. Marrow stromal cells and osteoclast precursors differentially contribute to TNF-alpha-induced osteoclastogenesis in vivo. J. Immunol. 2004, 173, 4838–4846. [Google Scholar] [CrossRef]
- Azuma, Y.; Kaji, K.; Katogi, R.; Takeshita, S.; Kudo, A. Tumor necrosis factor-alpha induces differentiation of and bone resorption by osteoclasts. J. Biol. Chem. 2000, 275, 4858–4864. [Google Scholar] [CrossRef]
- Gilbert, L.; He, X.; Farmer, P.; Boden, S.; Kozlowski, M.; Rubin, J.; Nanes, M.S. Inhibition of osteoblast differentiation by tumor necrosis factor-alpha. Endocrinology 2000, 141, 3956–3964. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, N.; Maeno, M.; Suzuki, N.; Fujisaki, K.; Tanaka, H.; Ogiso, B.; Ito, K. IL-alpha stimulates the formation of osteoclast-like cells by increasing M-CSF and PGE2 production and decreasing OPG production by osteoblasts. Life Sci. 2005, 77, 615–626. [Google Scholar] [CrossRef]
- Watanabe, Y.; Namba, A.; Aida, Y.; Honda, K.; Tanaka, H.; Suzuki, N.; Matsumura, H.; Maeno, M. IL-1 beta suppresses the formation of osteoclasts by increasing OPG production via an autocrine mechanism involving celecoxib-related prostaglandins in chondrocytes. Mediat. Inflamm. 2009, 2009, 308596. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.M.; Itoh, K.; Udagawa, N.; Hausler, K.; Yasuda, H.; Shima, N.; Mizuno, A.; Higashio, K.; Takahashi, N.; Suda, T.; et al. Transforming growth factor beta affects osteoclast differentiation via direct and indirect actions. J. Bone Miner. Res. 2001, 16, 1787–1794. [Google Scholar] [CrossRef] [PubMed]
- Itoh, K.; Udagawa, N.; Takagiri, T.; Iemura, S.; Ueno, N.; Yasuda, H.; Higashio, K.; Quinn, J.M.; Gillespie, M.T.; Martin, T.J.; et al. Bone morphogenic protein 2 stimulates osteoclast differentiation and survival supported by receptor activator of nuclear factor-kappaB ligand. Endocrinology 2001, 142, 3656–3662. [Google Scholar] [CrossRef] [PubMed]
- Salamanna, F.; Maglio, M.; Borsari, V.; Giavaresi, G.; Aldini, N.N.; Fini, M. Peripheral blood mononuclear cells spontaneous osteoclastogenesis: Mechanisms driving the process and clinical relevance in skeletal disease. J. Cell Physiol. 2016, 231, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Guillonneau, C.; Bézie, S.; Anegon, I. Immunoregulatory properties of the cytokine IL-34. Cell Mol. Life Sci. 2017, 74, 2569–2586. [Google Scholar] [CrossRef]
- Mohamed, S.G.K.; Suglyama, E.; Shinoda, K.; Hounoki, H.; Taki, H.; Maruyama, M.; Miyahara, T.; Kobayashi, M. Interleukin-4 inhibits RANKL-induced expression of NFATc1 and c-Fos: A possible mechanism for downregulation of osteoclastogenesis. Biochem. Biophys. Res. Commun. 2005, 329, 839–845. [Google Scholar] [CrossRef]
- Wei, S.; Wang, M.W.-H.; Teitelbaum, S.L.; Ross, F.P. Interleukin-4 reversibly inhibits osteoclastogenesis via inhibition of NF-kappa B and mitogen-activated protein kinase signaling. J. Biol. Chem. 2002, 277, 6622–6630. [Google Scholar] [CrossRef]
- Fujii, T.; Kitaura, H.; Kimura, K.; Hakami, Z.W.; Takano-Yamamoto, T. IL-4 inhibits TNF-α-mediated osteoclast formation by inhibiting RANKL expression in TNF-α-activated stromal cells and direct inhibition of TNF-α-activated osteoclast precursors via a T-cell-independent mechanism in vivo. Bone 2012, 51, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Ivashkiv, L.B. Negative regulation of osteoclastogenesis and bone resorption by cytokines and transcriptional repressors. Arthritis Res. Ther. 2011, 13, 234. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S.G.-K.; Sugiyama, E.; Shinoda, K.; Taki, H.; Hounoki, H.; Abdel-Aziz, H.O.; Maruyama, M.; Kobayashi, M.; Ogawa, H.; Miyahara, T. Interleukin-10 inhibits RANKL-mediated expression of NFATc1 in part via suppression of c-Fos and c-Jun in RAW264.7 cells and mouse bone marrow cells. Bone 2007, 41, 592–602. [Google Scholar] [CrossRef]
- Honda, K. Interleukin-6 and soluble interleukin-6 receptor suppress osteoclast differentiation by inducing PGE(2) production in chondrocytes. J. Oral Sci. 2011, 53, 87–96. [Google Scholar] [CrossRef][Green Version]
- Kitaura, H.; Nagata, N.; Fujimura, Y.; Hotokezaka, H.; Yoshida, N.; Nakayama, K. Effect of IL-12 on TNF-α -mediated osteoclast formation in bone marrow cells: Apoptosis mediated by Fas/Fas ligand interaction. J. Immunol. 2002, 169, 4732–4738. [Google Scholar] [CrossRef]
- Morita, Y.; Kitaura, H.; Yoshimatsu, M.; Fujimura, Y.; Kohara, H.; Eguchi, T.; Yoshida, N. IL-18 inhibits TNF-alpha-induced osteoclastogenesis possibly via a T cell-independent mechanism in synergy with IL-12 in vivo. Calcif. Tissue Int. 2010, 86, 242–248. [Google Scholar] [CrossRef]
- Horwood, N.J.; Elliott, J.; Martin, T.J.; Gillespie, M.T. IL-12 alone and in synergy with IL-18 inhibits osteoclast formation in vitro. J. Immunol. 2001, 166, 4915–4921. [Google Scholar] [CrossRef] [PubMed]
- Udagawa, N.; Horwood, N.J.; Elliott, J.; Mackay, A.; Owens, J.; Okamura, H.; Kurimoto, M.; Chambers, T.J.; Martin, T.J.; Gillespie, M.T. Interleukin-18 (interferon-gamma-inducing-factor) is produced by osteoblasts and acts via granulocyte/macrophage colony-stimulating factor and not via interferon-gamma to inhibit osteoclast formation. J. Exp. Med. 1997, 185, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Kohara, H.; Kitaura, H.; Fujimura, Y.; Yoshimatsu, M.; Morita, Y.; Eguchi, T.; Masuyama, R.; Yoshida, N. IFN-γ directly inhibits TNF-α-induced osteoclastogenesis in vitro and in vivo and induces apoptosis mediated by Fas/Fas ligand interactions. Immunol. Lett. 2011, 137, 53–61. [Google Scholar] [CrossRef]
- Hillsley, M.V.; Frangos, J.A. Bone tissue engineering: Role of interstitial fluid flow. Biotechnol. Bioeng. 1994, 43, 573–581. [Google Scholar] [CrossRef]
- Liu, C.; Zhao, Y.; Cheung, W.-Y.; Gandhi, R.; Wang, L.; Lidan, Y. Effects of cyclic hydraulic pressure on osteocytes. Bone 2010, 46, 1449–1456. [Google Scholar] [CrossRef]
- Smith, J.K. IL-6 and the dysregulation of immune, bone, muscle, and metabolic homeostasis during spaceflight. NPJ Microgravity 2018, 4, 24. [Google Scholar] [CrossRef]
- De Maré, A.S.; D’Haese, P.C.; Verhulst, A. The role of sclerostin in bone and ectopic calcification. Int. J. Mol. Sci. 2020, 21, 3199. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, N.; Khandani, A.; Camirand, A.; Harrison, R.E. Effects of microgravity on osteoclast bone resorption and osteoblast cytoskeletal organization and adhesion. Bone 2011, 49, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Hughs-Fulford, M. Function of the cytoskeleton in gravisensing during spaceflight. Adv. Space Res. 2003, 32, 1583–1593. [Google Scholar] [CrossRef]
- Gerbaix, M.; Gnyubkin, V.; Farlay, D.; Olivier, C.; Ammann, P.; Courbon, G.; Laroche, N.; Genthial, R.; Follet, H.; Peyrin, F.; et al. One-month spaceflight compromises the bone microstructure, tissue-level mechanical properties, osteocyte survival and lacunar volume in mature mice skeletons. Sci. Rep. 2017, 7, 2659. [Google Scholar] [CrossRef]
- Vico, L.; Chappard, D.; Palle, S.; Bakulin, A.V.; Novikov, V.E.; Alexandre, C. Trabecular bone remodeling after seven days of weightlessness exposure (BIOCOSMOS 1667). Am. J. Physiol. 1988, 255, R243–R247. [Google Scholar] [CrossRef]
- Vico, L.; Chappard, D.; Alexandre, C.; Palle, S.; Minaire, P.; Riffat, G.; Novikov, V.E.; Bakulin, A.V. Effects of weightlessness on bone mass and osteoclast number in pregnant rats after a five-day spaceflight (COSMOS 1514). Bone 1987, 8, 95–103. [Google Scholar] [CrossRef]
- Vico, L.; Bourrin, S.; Genty, C.; Palle, S.; Alexandre, C. Histomorphometric analysis of cancellous bone from COSMOS 2044 rats. J. Appl. Physiol. 1985, 75, 2203–2208. [Google Scholar] [CrossRef] [PubMed]
- Blaber, E.A.; Dvorochkin, N.; Lee, C.; Alwood, J.S.; Yousuf, R.; Pianetta, P.; Globus, R.K.; Burns, B.P.; Almeida, E.A. Microgravity induces pelvic bone loss through osteoclastic activity, osteocytic osteolysis, and osteoblastic cell cycle inhibition by CDKN1a/p21. PLoS ONE 2013, 8, e61372. [Google Scholar] [CrossRef]
- Chatani, M.; Morimoto, H.; Takeyama, K.; Mantoku, A.; Tanigawa, N.; Kubota, K.; Suzuki, H.; Uchida, S.; Tanigaki, F.; Shirakawa, M.; et al. Acute transcriptional up-regulation specific to osteoblasts/osteoclasts in medaka fish immediately after exposure to microgravity. Sci. Rep. 2016, 6, 39545. [Google Scholar] [CrossRef]
- Tamma, R.; Colaianni, G.; Camerino, C.; Di Benedetto, A.; Greco, G.; Strippoli, M.; Vergari, R.; Grano, A.; Mancini, L.; Mori, G.; et al. Microgravity during spaceflight directly affects in vitro osteoclastogenesis and bone resorption. FASEB J. 2009, 23, 2549–2554. [Google Scholar] [CrossRef] [PubMed]
- Saxena, R.; Pan, G.; Dohm, E.D.; McDonald, J.M. Modeled microgravity and hindlimb unloading sensitize osteoclast precursors to RANKL-mediated osteoclastogenesis. J. Bone Miner. Metab. 2011, 29, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, J.I.; Plotkin, L.I.; Stewart, S.A.; Weinstein, R.S.; Parfitt, A.M.; Manolagas, S.C.; Bellido, T. Osteocyte apoptosis is induced by weightlessness in mice and precedes osteoclast recruitment and bone loss. J. Bone Miner. Res. 2006, 21, 605–615. [Google Scholar] [CrossRef]
- Rucci, N.; Rufo, A.; Alamanou, M.; Teti, A. Modeled microgravity stimulates osteoclastogenesis and bone resorption by increasing osteoblast RANKL/OPG ratio. J. Cell. Biochem. 2007, 100, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Sambandam, Y.; Blanchard, J.J.; Daughtridge, G.; Kolb, R.J.; Shanmugarajan, S.; Pandruvada, S.N.M.; Bateman, T.A.; Reddy, S.V. Microarray profile of gene expression during osteoclast differentiation in modelled microgravity. J. Cell. Biochem. 2010, 111, 1179–1187. [Google Scholar] [CrossRef]
- Sambandam, Y.; Townsend, T.; Pierce, J.J.; Lipman, C.M.; Haque, A.; Bateman, T.A.; Reddy, S.V. Microgravity control of autophagy modulates osteoclastogenesis. Bone 2014, 61, 125–131. [Google Scholar] [CrossRef]
- Sambandam, Y.; Baird, K.L.; Stroebel, M.; Kowai, E.; Balasubramanian, S.; Reddy, S.V. Microgravity induction of TRAIL expression in preosteoclast cells enhances osteoclast differentiation. Sci. Rep. 2016, 6, 25143. [Google Scholar] [CrossRef]
- Ethiraj, P.; Link, J.R.; Sinkway, J.M.; Brown, C.D.; Parler, W.A.; Reddy, S.V. Microgravity modulation of syncytin-A expression enhance osteoclast formation. J. Cell. Biochem. 2018, 119, 5695–5703. [Google Scholar] [CrossRef]
- Vargas-Franco, J.W.; Castaneda, B.; Rédini, F.; Gómez, D.F.; Heymann, D.; Lézot, F. Paradoxical side effects of biphosphonates on the skeleton: What do we know and what can we do? J. Cell. Physiol. 2018, 233, 5696–5715. [Google Scholar] [CrossRef]
- Deeks, E.D. Denosumab: A review in postmenopausal osteoporosis. Drugs Aging 2018, 35, 163–173. [Google Scholar] [CrossRef]
- Stone, J.A.; McCrea, J.B.; Witter, R.; Zajic, S.; Stoch, S.A. Clinical and translational pharmacology of the cathepsin K inhibitor odanacatib studied for osteoporosis. Br. J. Clin. Pharmacol. 2019, 85, 1072–1083. [Google Scholar] [CrossRef] [PubMed]
- Bandeira, L.; Lewiecki, E.M.; Bilezikian, J.P. Romosozumab for treatment of osteoporosis. Expert Opin. Biol. Ther. 2017, 17, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Adanty, C. Romosozumab (sclerostin monoclonal antibody) for the treatment of osteoporosis in postmenopausal women: A review. J. Popul. Ther. Clin. Pharmacol. 2020, 27, e25–e31. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.C.; Christiansen, B.A.; Murugesh, D.K.; Sebastian, A.; Hum, N.R.; Collette, N.M.; Hatsell, S.; Economides, A.N.; Blanchette, C.D.; Loots, G.G. SOST/Sclerostin improves posttraumatic osteoarthritis and inhibits MMP2/3 expression after injury. J. Bone Miner. Res. 2018, 33, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.-P.; Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef]
- Lloyd, S.A.; Morony, S.E.; Ferguson, V.L.; Simske, S.J.; Stodieck, L.S.; Warmington, K.S.; Livingston, E.W.; Lacey, D.L.; Kostenuik, P.J.; Bateman, T.A. Osteoprotegerin is an effective countermeasure for spaceflight-induced bone loss in mice. Bone 2015, 81, 562–572. [Google Scholar] [CrossRef]
- Ikegame, M.; Hattori, A.; Tabata, M.J.; Kitamura, K.; Tabuchi, Y.; Furusawa, Y.; Maruyama, Y.; Yamamoto, T.; Sekiguchi, T.; Matsuoka, R.; et al. Melatonin is a potential drug for the prevention of bone loss during spaceflight. J. Pineal Res. 2019, 67, e1259w. [Google Scholar] [CrossRef]
| Cytokine | M-CSF/RANKL Pathways | Calcineurin Pathway | Apoptotic Pathway | RANK/RANKL/OPG Expression | References |
|---|---|---|---|---|---|
| IL-4 | Blocks JNK, P38, ERK protein kinase signaling. Inhibits NF-κB, c-Fos, NFATc1 transcription | ↓ calcium signaling | ↓ RANK, RANKL expression ↑ osteoblast OPG expression | [39,40,41,42] | |
| IL-6 sIL-6R | PGE2-mediated ↓ osteoclastogenesis | [44] | |||
| IL-10 | Inhibits c-Fos, c-Jun, NFATc1 transcription | Inhibits TREM2 expression | [43] | ||
| IL-12 | ↑ IL-18 inhibition of TNF-α-mediated osteoclastogenesis | ↑ Fas/Fas ligand expression | [45,47] | ||
| IL-18 | ↓ osteoblast M-CSF production. Inhibits TNF-α-mediated osteoclastogenesis | [46,47] | |||
| IFN-γ | Inhibits TNF-α-mediated osteoclastogenesis | ↑ Fas/Fas ligand binding | [49] |
| Cell | M-CSF/RANKL Pathways | Calcineurin Pathway | Sclerostin Pathway * |
|---|---|---|---|
| HSC | ↑ c-Jun, MITF, CREB [63], TRAIL, TRAF-6, OC-STAMP, DC-STAMP [64], syncytin-A [60] | ↑ S100AB protein and cytosolic calcium [63] | ↑ osteoclastogenesis secondary to ↓ OPG production [53] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, J.K. Osteoclasts and Microgravity. Life 2020, 10, 207. https://doi.org/10.3390/life10090207
Smith JK. Osteoclasts and Microgravity. Life. 2020; 10(9):207. https://doi.org/10.3390/life10090207
Chicago/Turabian StyleSmith, John Kelly. 2020. "Osteoclasts and Microgravity" Life 10, no. 9: 207. https://doi.org/10.3390/life10090207
APA StyleSmith, J. K. (2020). Osteoclasts and Microgravity. Life, 10(9), 207. https://doi.org/10.3390/life10090207




