UPLC-MS/MS Phytochemical Analysis of Two Croatian Cistus Species and Their Biological Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. UPLC-MS/MS Phytochemical Analysis of Plant Extracts
2.3. DPPH Test
2.4. FRAP Assay
2.5. Total Phenolic Content
2.6. MTT Assay
2.7. Determination of the Antimicrobial Activity
2.8. Microbial Strains
3. Results and Discussion
3.1. UPLC-MS/MS Phytochemical Analysis of Plant Extracts
3.2. Antioxidant Activity of Cistus Extracts
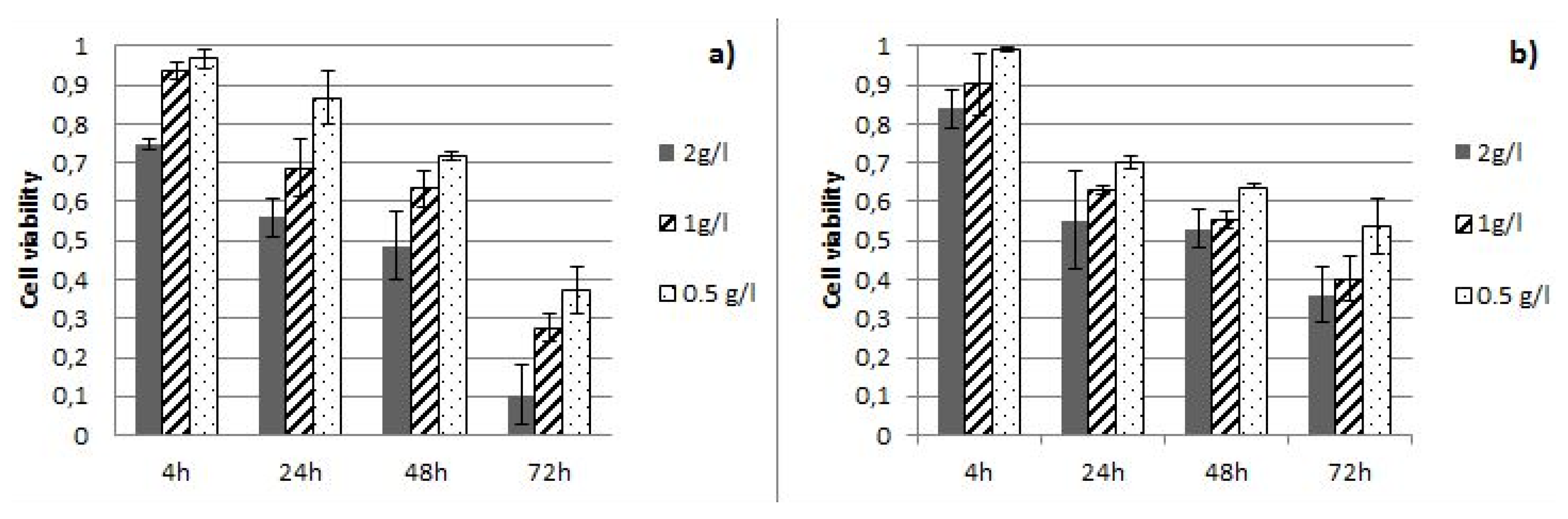
3.3. Antiproliferative Activity of Cistus Extracts
3.4. Antimicrobial Potential of Cistus Extracts
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mabberley, D.J. The Plant, 2nd ed.; Cambridge University Press: Cambridge, UK, 1997; p. 680. [Google Scholar]
- Petropoulos, S.A.; Karkanis, A.; Martins, N.; Ferreira, I.C. Halophytic herbs of the Mediterranean basin: An alternative approach to health. Food Chem. Toxicol. 2018, 114, 155–169. [Google Scholar] [CrossRef]
- Papaefthimiou, D.; Papanikolaou, A.; Falara, V.; Givanoudi, S.; Kostas, S.; Kanellis, A. Genus Cistus: A model for exploring labdane-type diterpenes’ biosynthesis and a natural source of high value products with biological, aromatic, and pharmacological properties. Front. Chem. 2014, 2, 35. [Google Scholar] [CrossRef]
- Alkofahi, A.; Al-Khalil, S. Mutagenic and Toxic Activity of some Jordanian Medicinal Plants. Int. J. Pharmacogn. 1995, 33, 61–64. [Google Scholar] [CrossRef]
- Bouamama, H.; Villard, J.; Benharref, A.; Jana, M. Antibacterial and antifungal activities of Cistus incanus and C. monspeliensis leaf extracts. Therapie 2000, 54, 731–733. [Google Scholar]
- Attaguile, G.; Russo, A.; Campisi, A.; Savoca, F.; Acquaviva, R.; Ragusa, N.; Vanella, A. Antioxidant activity and protective effect on DNA cleavage of extracts from Cistus incanus L. and Cistus monspeliensis L. Cell Biol. Toxicol. 2000, 16, 83–90. [Google Scholar] [CrossRef]
- Rebensburg, S.; Helfer, M.; Schneider, M.; Koppensteiner, H.; Eberle, J.; Schindler, M.; Gürtler, L.G.; Brack-Werner, R. Potent in vitro antiviral activity of Cistus incanus extract against HIV and Filoviruses targets viral envelope proteins. Sci. Rep. 2016, 6, 20394. [Google Scholar] [CrossRef]
- Sayah, K.; Chemlal, L.; Marmouzi, I.; El Jemli, M.; Cherrah, Y.; Faouzi, M.E.A. In vivo anti-inflammatory and analgesic activities of Cistus salviifolius (L.) and Cistus monspeliensis (L.) aqueous extracts. S. Afr. J. Bot. 2017, 113, 160–163. [Google Scholar] [CrossRef]
- Wannes, W.A.; Tounsi, M.; Marzouk, B. A review of Tunisian medicinal plants with anticancer activity. J. Complement. Integr. Med. 2017, 15, 1–35. [Google Scholar] [CrossRef]
- Bereksi, M.S.; Hassaine, H.; Bekhechi, C.; Abdelouahid, D.E. Evaluation of Antibacterial Activity of some Medicinal Plants Extracts Commonly Used in Algerian Traditional Medicine against some Pathogenic Bacteria. Pharmacogn. J. 2018, 10, 507–512. [Google Scholar] [CrossRef]
- Jeszka-Skowron, M.; Zgoła-Grześkowiak, A.; Frankowski, R. Cistus incanus a promising herbal tea rich in bioactive compounds: LC–MS/MS determination of catechins, flavonols, phenolic acids and alkaloids—A comparison with Camellia sinensis, Rooibos and Hoan Ngoc herbal tea. J. Food Compos. Anal. 2018, 74, 71–81. [Google Scholar] [CrossRef]
- Attaguile, G.; Perticone, G.; Mania, G.; Savoca, F.; Pennisi, G.; Salomone, S. Cistus incanus and Cistus monspeliensis inhibit the contractile response in isolated rat smooth muscle. J. Ethnopharmacol. 2004, 92, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Bouamama, H.; Noel, T.; Villard, J.; Benharref, A.; Jana, M. Antimicrobial activities of the leaf extracts of two Moroccan cistus L. species. J. Ethnopharmacol. 2006, 104, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Menor, L.; Morales-Soto, A.; Barrajón-Catalán, E.; Roldan-Segura, C.M.; Segura-Carretero, A.; Micol, V. Correlation between the antibacterial activity and the composition of extracts derived from various Spanish Cistus species. Food Chem. Toxicol. 2013, 55, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Vitali, F.; Pennisi, G.; Attaguile, G.; Savoca, F.; Tita, B. Antiproliferative and cytotoxic activity of extracts from Cistus incanus L. and Cistus monspeliensis L. on human prostate cell lines. Nat. Prod. Res. 2011, 25, 188–202. [Google Scholar] [CrossRef]
- Ustun, O.; Berrin-Ozcelik, T.B. Bioactivities of ethanolic extract and its fractions of Cistus laurifolius L. (Cistaceae) and Salvia wiedemannii Boiss. (Lamiaceae) species. Pharmacogn. Mag. 2016, 12, S82–S85. [Google Scholar] [CrossRef]
- Attaguile, G. Gastroprotective effect of aqueous extract of Cistus incanus L. in rats. Pharmacol. Res. 1995, 31, 29–32. [Google Scholar] [CrossRef]
- Venditti, A.; Bianco, A.; Bruno, M.; Ben Jemia, M.; Nicoletti, M. Phytochemical study of Cistus libanotis L. Nat. Prod. Res. 2014, 29, 189–192. [Google Scholar] [CrossRef]
- Politeo, O.; Maravić, A.; Burčul, F.; Carev, I.; Kamenjarin, J. Phytochemical Composition and Antimicrobial Activity of Essential Oils of Wild Growing Cistus species in Croatia. Nat. Prod. Commun. 2018, 13, 771–774. [Google Scholar] [CrossRef]
- Fernández-Calvet, A.; Euba, B.; Caballero, L.; Díez-Martínez, R.; Menendez, M.; Ortiz-De-Solorzano, C.; Leiva, J.; Micol, V.; Barrajón-Catalán, E.; Garmendia, J. Preclinical Evaluation of the Antimicrobial-Immunomodulatory Dual Action of Xenohormetic Molecules against Haemophilus influenzae Respiratory Infection. Biomolecules 2019, 9, 891. [Google Scholar] [CrossRef]
- Umarusman, M.A.; Aysan, Y.; Özgüven, M. Investigation of the antibacterial effects of different plant extracts against pea bacterial leaf blight disease caused by pseudomonas syringae pv. pisi|Farkli Bitki Ekstraktlarinin Bezelye Bakteriyel Yaprak Yanikligina (Pseudomonas syringae pv. pisi) A. J. Tekirdag Agric. Fac. 2019, 16, 297–314. [Google Scholar]
- Vasiliki, G.; Charalampia, D.; Haralabos, K.C. In Vitro Antioxidant, Antithrombotic, Antiatherogenic and Antidiabetic Activities of Urtica dioica, Sideritis euboea and Cistus creticus Water Extracts and Investigation of Pasta Fortification with the Most Bioactive One. Curr. Pharm. Biotechnol. 2019, 20, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Lahcen, S.A.; El Hattabi, L.; Benkaddour, R.; Chahboun, N.; Ghanmi, M.; Satrani, B.; Tabyaoui, M.; Zarrouk, A. Chemical composition, antioxidant, antimicrobial and antifungal activity of Moroccan Cistus creticus leaves. Chem. Data Collect. 2020, 26, 100346. [Google Scholar] [CrossRef]
- Ammendola, M.; Haponska, M.; Balik, K.; Modrakowska, P.; Matulewicz, K.; Kazmierski, L.; Lis, A.; Kozlowska, J.; Garcia-Valls, R.; Giamberini, M.; et al. Stability and anti-proliferative properties of biologically active compounds extracted from Cistus L. after sterilization treatments. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Jerónimo, E.; Soldado, D.; Sengo, S.; Francisco, A.; Fernandes, F.; Portugal, A.P.; Alves, S.P.; Santos-Silva, J.; Bessa, R.J. Increasing the α-tocopherol content and lipid oxidative stability of meat through dietary Cistus ladanifer L. in lamb fed increasing levels of polyunsaturated fatty acid rich vegetable oils. Meat Sci. 2020, 164, 108092. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, H.; Aouadhi, C.; Kaddour, R.; Gruber, M.; Zargouni, H.; Zaouali, W.; Hamida, N.B.; Nasri, M.B.; Ouerghi, Z.; Hosni, K. Comparison of antioxidant and antimicrobial activities of two cultivated cistus species from Tunisia|Comparação das atividades antioxidante e antimicrobiana de duas espécies cultivada de cistus da Tunísia. Biosci. J. 2016, 32, 226–237. [Google Scholar] [CrossRef]
- Mikulec, A.; Kowalski, S.; Makarewicz, M.; Skoczylas, Ł.; Tabaszewska, M. Cistus extract as a valuable component for enriching wheat bread. LWT 2020, 118, 108713. [Google Scholar] [CrossRef]
- Lisiecka, K.; Wójtowicz, A.; Dziki, D.; Gawlik-Dziki, U. The influence of Cistus incanus L. leaves on wheat pasta quality. J. Food Sci. Technol. 2019, 56, 4311–4322. [Google Scholar] [CrossRef]
- Stępień, A.E.; Gorzelany, J.; Matłok, N.; Lech, K.; Figiel, A. The effect of drying methods on the energy consumption, bioactive potential and colour of dried leaves of Pink Rock Rose (Cistus creticus). J. Food Sci. Technol. 2019, 56, 2386–2394. [Google Scholar] [CrossRef]
- Viapiana, A.; Konopacka, A.; Waleron, K.; Wesolowski, M. Cistus incanus L. commercial products as a good source of polyphenols in human diet. Ind. Crop. Prod. 2017, 107, 297–304. [Google Scholar] [CrossRef]
- Chebli, Y.; El Otmani, S.; Chentouf, M.; Hornick, J.-L.; Bindelle, J.; Cabaraux, J.F.; Chebli, Y. Foraging Behavior of Goats Browsing in Southern Mediterranean Forest Rangeland. Animals 2020, 10, 196. [Google Scholar] [CrossRef]
- Guerreiro, O.; Alves, S.P.; Soldado, D.; Cachucho, L.; Almeida, J.M.; Francisco, A.E.; Santos-Silva, J.; Bessa, R.J.; Jerónimo, E. Inclusion of the aerial part and condensed tannin extract from Cistus ladanifer L. in lamb diets—Effects on growth performance, carcass and meat quality and fatty acid composition of intramuscular and subcutaneous fat. Meat Sci. 2020, 160, 107945. [Google Scholar] [CrossRef] [PubMed]
- Mignacca, S.A.; Mucciarelli, M.; Colombino, E.; Biasibetti, E.; Muscia, S.; Amato, B.; Presti, V.D.M.L.; Vazzana, I.; Galbo, A.; Capucchio, M.T. Cistus salviifolius Toxicity in Cattle. Veter. Pathol. 2019, 57, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Wittpahl, G.; Kölling-Speer, I.; Basche, S.; Herrmann, E.; Hannig, M.; Speer, K.; Hannig, C. The Polyphenolic Composition of Cistus incanus Herbal Tea and Its Antibacterial and Anti-adherent Activity against Streptococcus mutans. Planta Med. 2015, 81, 1727–1735. [Google Scholar] [CrossRef] [PubMed]
- Dimas, K.; Demetzos, C.; Marsellos, M.; Sotiriadou, R.; Malamas, M.; Kokkinopoulos, D. Cytotoxic Activity of Labdane Type Diterpenes Against Human Leukemic Cell Lines in vitro. Planta Med. 1998, 64, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Raimundo, J.R.; Frazão, D.F.; Domingues, J.L.; Quintela-Sabarís, C.; Dentinho, T.P.; Anjos, O.; Alves, M.N.; Delgado, F. Neglected Mediterranean plant species are valuable resources: The example of Cistus ladanifer. Planta 2018, 248, 1351–1364. [Google Scholar] [CrossRef] [PubMed]
- El Euch, S.K.; Bouajila, J.; Bouzouita, N. Chemical composition, biological and cytotoxic activities of Cistus salviifolius flower buds and leaves extracts. Ind. Crop. Prod. 2015, 76, 1100–1105. [Google Scholar] [CrossRef]
- Riehle, P.; Vollmer, M.; Rohn, S. Phenolic compounds in Cistus incanus herbal infusions—Antioxidant capacity and thermal stability during the brewing process. Food Res. Int. 2013, 53, 891–899. [Google Scholar] [CrossRef]
- Rebaya, A.; Belghith, S.I.; Cherif, J.K.; Trabelsi-Ayadi, M. Total phenolic compounds and antioxidant potential of rokrose (Cistus salviifolius) leaves and flowers grown in Tunisia. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 327–331. [Google Scholar]
- El Menyiy, N.; Al-Waili, N.; El-Haskoury, R.; Bakour, M.; Zizi, S.; Al-Waili, T.; Lyoussi, B. Potential effect of Silybum marianum L. and Cistus ladaniferus L. extracts on urine volume, creatinine clearance and renal function. Asian Pac. J. Trop. Med. 2018, 11, 393. [Google Scholar] [CrossRef]
- Gürbüz, P.; Demirezer, L.Ö.; Güvenalp, Z.; Kuruüzüm-Uz, A.; Kazaz, C. Isolation and Structure Elucidation of Uncommon Secondary Metabolites from Cistus salviifolius L. Rec. Nat. Prod. 2015, 9, 175–183. [Google Scholar]
- Gori, A.; Ferrini, F.; Marzano, M.C.; Tattini, M.; Centritto, M.; Baratto, M.C.; Pogni, R.; Brunetti, C. Characterisation and Antioxidant Activity of Crude Extract and Polyphenolic Rich Fractions from C. incanus Leaves. Int. J. Mol. Sci. 2016, 17, 1344. [Google Scholar] [CrossRef] [PubMed]
- Kalpoutzaki, E.; Aligiannis, N.; Mitaku, S.; Harvala, C.; Skaltsounis, L. New Semisynthetic Antimicrobial Labdane-Type Diterpenoids Derived from the Resin “Ladano” of Cistus creticus. Z. Naturforschung C 2001, 56, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Güvenç, A.; Yıldız, S.; Özkan, A.M.; Erdurak, C.S.; Coskun, M.; Yılmaz, G.; Okuyama, T.; Okada, Y.; Yildiz, S.; Yilmaz, G. Antimicrobiological Studies on Turkish Cistus. Species. Pharm. Biol. 2005, 43, 178–183. [Google Scholar] [CrossRef]
- Garofulić, I.E.; Zorić, Z.; Pedisić, S.; Brnčić, M.; Dragović-Uzelac, V. UPLC-MS2 Profiling of Blackthorn Flower Polyphenols Isolated by Ultrasound-Assisted Extraction. J. Food Sci. 2018, 83, 2782–2789. [Google Scholar] [CrossRef]
- Abu-Reidah, I.M.; Ali-Shtayeh, M.S.; Jamous, R.M.; Arráez-Román, D.; Segura-Carretero, A. HPLC-DAD-ESI-MS/MS screening of bioactive components from Rhus coriaria L. (Sumac) fruits. Food Chem. 2015, 166, 179–191. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.; Strain, J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Singleton, S.; Rossi, J.A.; Singleton, V.L.; Singleton, V.; Rossi, J.; Singleton, V.I. Colorimetry of total phenolic with phosphomolybdic—Photsphotunstic acid reagents. Am. J. Vitic. 1965, 16, 144–158. [Google Scholar]
- Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi: Approved Standard; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- Clinical and Laboratory Standards Institute. Performance Standard for Antimicrobial Susceptibility Testing—Approved Standard; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Maravić, A.; Skocibusic, M.; Šamanić, I.; Fredotović, Ž.; Cvjetan, S.; Jutronić, M.; Puizina, J. Aeromonas spp. simultaneously harbouring blaCTX-M-15, blaSHV-12, blaPER-1 and blaFOX-2, in wild-growing Mediterranean mussel (Mytilus galloprovincialis) from Adriatic Sea, Croatia. Int. J. Food Microbiol. 2013, 166, 301–308. [Google Scholar] [CrossRef]
- Gürbüz, P.; Koşar, M.; Güvenalp, Z.; Kuruuzum-Uz, A.; Demirezer, L. Ömür Simultaneous determination of selected flavonoids from different Cistus species by HPLC-PDA. J. Res. Pharm. 2018, 22, 78–83. [Google Scholar] [CrossRef]
- Barrajón-Catalán, E.; Fernández-Arroyo, S.; Roldán, C.; Guillén, E.; Saura, D.; Segura-Carretero, A.; Micol, V. A systematic study of the polyphenolic composition of aqueous extracts deriving from several Cistus genus species: Evolutionary relationship. Phytochem. Anal. 2011, 22, 303–312. [Google Scholar] [CrossRef]
- Matłok, N.; Lachowicz, S.; Gorzelany, J.; Balawejder, M. Influence of Drying Method on Some Bioactive Compounds and the Composition of Volatile Components in Dried Pink Rock Rose (Cistus creticus L.). Molecules 2020, 25, 2596. [Google Scholar] [CrossRef] [PubMed]
- Gaweł-Bęben, K.; Kukula-Koch, W.; Hoian, U.; Czop, M.; Strzępek-Gomółka, M.; Antosiewicz, B. Characterization of Cistus × incanus L. and Cistus ladanifer L. Extracts as Potential Multifunctional Antioxidant Ingredients for Skin Protecting Cosmetics. Antioxidants 2020, 9, 202. [Google Scholar] [CrossRef]
- Ghalia, S.; Adawia, K.; Waed, A. Evaluation of radical scavenging activity, total phenolics and total flavonoids contents of Cistus species in Syria. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 1071–1077. [Google Scholar]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, M.; Toniolo, C.; Venditti, A.; Bruno, M.; Ben-Jemia, M. Antioxidant activity and chemical composition of three Tunisian Cistus: Cistus monspeliensis Cistus villosus and Cistus libanotis? Nat. Prod. Res. 2014, 29, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Dudonné, S.; Vitrac, X.; Coutière, P.; Woillez, M.; Mérillon, J.-M. Comparative Study of Antioxidant Properties and Total Phenolic Content of 30 Plant Extracts of Industrial Interest Using DPPH, ABTS, FRAP, SOD, and ORAC Assays. J. Agric. Food Chem. 2009, 57, 1768–1774. [Google Scholar] [CrossRef]
- Piluzza, G.; Bullitta, S. Correlations between phenolic content and antioxidant properties in twenty-four plant species of traditional ethnoveterinary use in the Mediterranean area. Pharm. Biol. 2011, 49, 240–247. [Google Scholar] [CrossRef]
- Brunetti, C.; Di Ferdinando, M.; Fini, A.; Pollastri, S.; Tattini, M. Flavonoids as Antioxidants and Developmental Regulators: Relative Significance in Plants and Humans. Int. J. Mol. Sci. 2013, 14, 3540–3555. [Google Scholar] [CrossRef] [PubMed]
- Agati, G.; Brunetti, C.; Di Ferdinando, M.; Ferrini, F.; Pollastri, S.; Tattini, M. Functional roles of flavonoids in photoprotection: New evidence, lessons from the past. Plant Physiol. Biochem. 2013, 72, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Tsatsakis, A.; Mamoulakis, C.; Teodoro, M.; Briguglio, G.; Caruso, E.; Sarandi, E.; Margină, D.; Dardiotis, E.; Kouretas, D.; et al. Current evidence on the effect of dietary polyphenols intake on chronic diseases. Food Chem. Toxicol. 2017, 110, 286–299. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Mackey, B.E.; Kim, H.-J.; Lee, I.-S.; Lee, K.-R.; Lee, S.-U.; Kozukue, E.; Kozukue, N. Structure−Activity Relationships of Tea Compounds against Human Cancer Cells. J. Agric. Food Chem. 2007, 55, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Barrajón-Catalán, E.; Fernández-Arroyo, S.; Saura, D.; Guillén, E.; Gutierrez, A.F.; Segura-Carretero, A.; Micol, V. Cistaceae aqueous extracts containing ellagitannins show antioxidant and antimicrobial capacity, and cytotoxic activity against human cancer cells. Food Chem. Toxicol. 2010, 48, 2273–2282. [Google Scholar] [CrossRef]
- Skorić, M.; Todorović, S.I.; Gligorijević, N.; Janković, R.; Živković, S.; Ristić, M.; Radulović, S. Cytotoxic activity of ethanol extracts of in vitro grown Cistus creticus subsp. Creticus L. on human cancer cell lines. Ind. Crop. Prod. 2012, 38, 153–159. [Google Scholar] [CrossRef]
- Ismail, T.; Calcabrini, C.; Diaz, A.R.; Fimognari, C.; Turrini, E.; Catanzaro, E.; Akhtar, S.; Sestili, P. Ellagitannins in Cancer Chemoprevention and Therapy. Toxins 2016, 8, 151. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Lazaro, M.; Calderón-Montaño, J.M.; Morón, E.B.; Austin, C.A. Green tea constituents (−)-epigallocatechin-3-gallate (EGCG) and gallic acid induce topoisomerase I- and topoisomerase II-DNA complexes in cells mediated by pyrogallol-induced hydrogen peroxide. Mutagenesis 2011, 26, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Calderón-Montaño, J.M.; Morón, E.B.; Pérez-Guerrero, C.; Lopez-Lazaro, M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Semwal, D.K.; Semwal, R.B.; Combrinck, S.; Viljoen, A.M. Myricetin: A Dietary Molecule with Diverse Biological Activities. Nutrients 2016, 8, 90. [Google Scholar] [CrossRef]
- Abad, M.J.; Bedoya, L.M.; Bermejo, P. Chapter 14—Essential Oils from the Asteraceae Family Active against Multidrug-Resistant Bacteria A2—Kon, Mahendra Kumar RaiKateryna Volodymyrivna. In Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components; Academic Press: Cambridge, MA, USA, 2013; pp. 205–221. Available online: http://www.sciencedirect.com/science/article/pii/B9780123985392000148 (accessed on 9 July 2020).
| Compound | Rt, min | Collision Energy (V) | Ionization Mode | Precursor Ion (m/z) | Fragment Ions (m/z) | Identification | C. C. ** (mg/L) | C. S. ** (mg/L) |
|---|---|---|---|---|---|---|---|---|
| 1 | 1.131 | 10 | − | 367 | 193 | Feruloylquinic acid | 0.31 ± 0.02 | 0.07 ± 0.02 |
| 2 | 1.135 | 10 | − | 353 | 119 | Caffeoylqunic acid derivative 1 | 0.16 ± 0.04 | 0.18 ± 0.28 |
| 3 | 1.169 | 10 | − | 324 | 173 | Caffeoylquinic acid derivative 2 | 0.15 ± 0.06 | 0.05 ± 0.04 |
| 4 | 1.686 | 10 | − | 343 | 191 | Galloylquinic acid | 1.16 ± 0.05 | 5.47 ± 0.49 |
| 5 | 1.693 | 15 | + | 442.9 | 139 | Epicatechin gallate * | 0.52 ± 0.03 | 0.23 ± 0.10 |
| 6 | 1.71 | 10 | − | 331 | 169 | Monogalloyl glucose [42] | 0.66 ± 0.49 | 2.84 ± 0.22 |
| 7 | 1.758 | 10 | − | 169 | 125 | Gallic acid * | 2.83 ± 0.11 | 24.66 ± 0.99 |
| 8 | 2.508 | 10 | − | 495 | 169 | Digalloylquinic acid | 0.37 ± 0.04 | 0.33 ± 0.11 |
| 9 | 2.973 | 15 | + | 653 | 303 | Quercetinacetylrutinoside | 0.41 ± 0.50 | 0.08 ± 0.01 |
| 10 | 3.993 | 10 | 337 | 163 | Coumaroilquinic acid derivative 1 | 2.48 ± 1.35 | 0.34 ± 0.47 | |
| 11 | 4.014 | 10 | + | 579 | 427 | Procyanidin B2 * | 11.65 ± 0.31 | 3.83 ± 0.29 |
| 12 | 4.305 | 10 | + | 291 | 139 | Catechin * | 5.49 ± 0.26 | 0.57 ± 0.06 |
| 13 | 4.311 | 10 | + | 291 | 139 | Epicatechin * | 5.46 ± 0.05 | 0.52 ± 0.13 |
| 14 | 4.459 | 10 | − | 353 | 119 | Chlorogenic acid * | 0.06 ± 0.01 | 0.02 ± 0.01 |
| 15 | 4.974 | 15 | + | 597 | 303 | Quercetin-pentosyl-hexoside | 0.04 ± 0.01 | 0.97 ± 0.15 |
| 16 | 5.016 | 10 | − | 179 | 135 | Caffeic acid * | 0.36 ± 0.55 | 0.05 ± 0.02 |
| 17 | 5.541 | 10 | − | 337 | 163 | Coumaroilquinic acid derivative 2 | 0.41 ± 0.01 | 0.39 ± 0.53 |
| 18 | 6.025 | 15 | + | 459 | 139 | Epigallocatechin gallate * | 0.23 ± 0.06 | 4.77 ± 0.70 |
| 19 | 6.037 | 5 | + | 403 | 271 | Apigenin-pentoside | 0.07 ± 0.04 | 0.03 ± 0.01 |
| 20 | 6.362 | 5 | + | 627 | 315 | Myricetin-rutinoside [53] | 31.23 ± 0.10 | 3.09 ± 0.19 |
| 21 | 6.474 | 15 | + | 481 | 319 | Myricetin-hexoside [42] | 937.44 ± 0.49 | 564.13 ± 3.78 |
| 22 | 6.571 | 10 | − | 163 | 119 | p-coumaric acid * | 0.25 ± 0.03 | 0.23 ± 0.05 |
| 23 | 7.19 | 15 | + | 611 | 303 | Ruthin * | 3.46 ± 0.06 | 0.99 ± 0.08 |
| 24 | 7.237 | 5 | + | 465 | 319 | Myricetin-rhamnoside [53] | 874.51 ± 0.46 | 3.13 ± 0.71 |
| 25 | 7.254 | 25 | + | 319 | 273 | Myricetin * | 87.14 ± 0.42 | 28.27 ± 1.11 |
| 26 | 7.315 | 10 | − | 193 | 134 | Ferulic acid * | 0.55 ± 0.39 | 0.07 ± 0.02 |
| 27 | 7.452 | 5 | + | 465 | 303.1 | Quercetin-3-glucoside * | 26.08 ± 0.20 | 76.49 ± 1.50 |
| 28 | 7.561 | 15 | + | 581 | 287 | Kaempferol-pentosylhexoside | 0.03 ± 0.01 | 0.51 ± 0.11 |
| 29 | 7.65 | 10 | + | 647 | 169 | Trigalloylquinic acid | 0.24 ± 0.04 | 0.04 ± 0.02 |
| 30 | 7.971 | 5 | + | 495 | 319 | Myricetin-glucuronide | 0.76 ± 0.07 | 0.22 ± 0.08 |
| 31 | 8.044 | 5 | + | 435 | 303 | Quercetin-pentoside [54] | 6.02 ± 0.02 | 54.88 ± 1.82 |
| 32 | 8.119 | 5 | + | 449 | 287 | Kaempferol-3-glucoside [54] | 15.05 ± 0.41 | 20.56 ± 1.25 |
| 33 | 8.164 | 15 | + | 625 | 317 | Isorhamnetin-rutinoside | 0.33 ± 0.02 | 0.03 ± 0.02 |
| 34 | 8.203 | 35 | + | 287 | 153 | Luteolin * | 4.28 ± 0.23 | 5.46 ± 1.40 |
| 35 | 8.422 | 5 | + | 449 | 303 | Quercetin-rhamnoside [53] | 23.02 ± 1.72 | 0.56 ± 0.29 |
| 36 | 8.651 | 5 | + | 419 | 287 | Kaempferol-pentoside | 1.14 ± 0.12 | 5.81 ± 0.79 |
| 37 | 8.723 | 15 | + | 637 | 287 | Kaempferol-acetylrutinoside | 0.03 ± 0.01 | 0.05 ± 0.03 |
| 38 | 8.78 | 10 | + | 507 | 303 | Quercetin-acetylhexoside | 0.13 ± 0.05 | 0.36 ± 0.56 |
| 39 | 9.541 | 5 | + | 491 | 287 | Kaempferol-acetylhexoside | 0.05 ± 0.03 | 0.24 ± 0.15 |
| 40 | 10.610 | 5 | + | 433 | 287 | Kaempferol-rhamnoside | 1.28 ± 0.07 | 0.44 ± 0.30 |
| 41 | 10.843 | 15 | + | 595 | 287 | Kaempferol-3-rutinoside * | 4.56 ± 0.38 | 12.58 ± 0.80 |
| 42 | 10.846 | 15 | + | 595 | 287 | Kaempferol-rhamnosyl-hexoside | 7.07 ± 2.89 | 13.12 ± 1.64 |
| 43 | 11.475 | 30 | + | 271 | 153 | Apigenin * | 0.35 ± 0.39 | 0.05 ± 0.03 |
| Antioxidant Assay (units) | C. creticus | C. salvifoliius | Ascorbic Acid |
|---|---|---|---|
| Folin-Ciocalteu (mg GAE a) | 209.27 ± 18.5 | 161.09 ± 7.2 | nd |
| DPPH b (mg/mL) | 0.52 ± 0.03 | 0.62 ± 0.04 | 0.02 ± 0.01 |
| FRAP (equivalents of Fe2+ mM c) | 0.78 ± 0.02 | 0.78 ± 0.06 | 5.57 ± 0.13 |
| Strain No. | Species | β-lactam Resistance Phenotype | MIC (µg/mL) of Selected Antimicrobial Agents a | β-Lactamase(s) Produced | Non β-Lactam Resistance Phenotype | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AMP | CAZ | IPM | GEN | TET | OX | ESBLs | MBLs | Others | ||||
| FSST 02 | Klebsiella pneumoniae | AMP, SAM, PIP, TZP, TIC, CAZ, CTX, ATM | >1024 (R) | 512 (R) | 0.5 (S) | 8 (R) | 128 (R) | NA | SHV-12, CTX-M-15 | - | ACC-1, TEM-1 | CIP, GEN, TET, SXT |
| FSST 20 | Acinetobacter baumannii | AMP, SAM, PIP, TZP, TIC, CTX, CAZ, FEP, ATM, IMP, MEM | >1024 (R) | >256 (R) | 16 (R) | 16 (R) | 64 (R) | NA | - | SIM-1 | - | GEN, TOB, AMK |
| FSST 21 | Pseudomonas aeruginosa | AMP, SAM, PIP, TZP, TIC, CTX, CAZ, FEP, ATM, IMP, MEM | >1024 (R) | >16 (R) | >8 (R) | >8 (R) | >8 (R) | NA | - | GIM-1 | - | CIP, GAT, LVX, AMK, GEN, TOB, NET, TET, SXT |
| MRSA-1 | Staphylococcus aureus | OX, P | NA | NA | NA | >16 (R) | <1 (S) | >4 (R) | Modification of PBP (mecA) | CC, CIP, E, GEN, MFX | ||
| Species | Strain No. | Inhibition Zone Diameter (mm) a | MIC (µg/mL) c | ||||
|---|---|---|---|---|---|---|---|
| C. creticus Extract | C. salviifolius Extract | TET b | C. creticus Extract | C. salviifolius Extract | TET | ||
| Gram-positive bacteria | |||||||
| Staphylococcus aureus | ATCC 29213 | 18.4 ± 1.1 | 15.2 ± 0.9 | 31.0 ± 0.5 | 500 | 1000 | 0.5 (S) |
| Staphylococcus aureus | MRSA-1 | 29.0 ± 0.8 | 25.0 ± 1.0 | NT | 500 | 500 | <1 (S) |
| Bacillus cereus | FSST-22 | 13.0 ± 0.5 | 13.0 ± 0.7 | 24.6 ± 0.6 | 1000 | 1000 | 1 (S) |
| Clostridiumperfringens | FSST-24 | 12.1 ± 0.5 | 12.4 ± 1.0 | NA | 1000 | 500 | NA |
| Gram-negative bacteria | |||||||
| Klebsiella pneumoniae | ATCC 13883 | 17.4 ± 0.5 | 13.5 ± 1.0 | 21.3 ± 0.2 | 1000 | 1000 | 1 (S) |
| Klebsiella pneumoniae | FSST-02 | 10.5 ± 0.0 | 10.0 ± 0.5 | 6.0 ± 0.0 | 2000 | 2000 | 128 (R) |
| Pseudomonas aeruginosa | ATCC 27853 | 12.6 ± 0.9 | 10.3 ± 0.8 | 11.5 ± 0.4 | 1000 | 1000 | 32 (R) |
| Pseudomonas aeruginosa | FSST-21 | 13.2 ± 0.4 | 11.0 ± 0.5 | 7.3 ± 0.3 | 500 | 1000 | >16 (R) |
| Acinetobacter baumannii | ATCC 19606 | 17.1 ± 0.5 | 20.5 ± 1.5 | 17.3 ± 0.6 | 500 | 500 | 2 (S) |
| Acinetobacter baumannii | FSST-20 | 23.0 ± 0.8 | 24.2 ± 0.0 | 6.0 ± 0.0 | 250 | 250 | 64 (R) |
| AMB | MIC90 | AMB | |||||
| Yeast | |||||||
| Candida albicans | FSST-29 | 19.3 ± 0.8 | 23.2 ± 1.2 | 21.6 ± 1.7 | 500 | 125 | 1 (S) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carev, I.; Maravić, A.; Ilić, N.; Čikeš Čulić, V.; Politeo, O.; Zorić, Z.; Radan, M. UPLC-MS/MS Phytochemical Analysis of Two Croatian Cistus Species and Their Biological Activity. Life 2020, 10, 112. https://doi.org/10.3390/life10070112
Carev I, Maravić A, Ilić N, Čikeš Čulić V, Politeo O, Zorić Z, Radan M. UPLC-MS/MS Phytochemical Analysis of Two Croatian Cistus Species and Their Biological Activity. Life. 2020; 10(7):112. https://doi.org/10.3390/life10070112
Chicago/Turabian StyleCarev, Ivana, Ana Maravić, Nada Ilić, Vedrana Čikeš Čulić, Olivera Politeo, Zoran Zorić, and Mila Radan. 2020. "UPLC-MS/MS Phytochemical Analysis of Two Croatian Cistus Species and Their Biological Activity" Life 10, no. 7: 112. https://doi.org/10.3390/life10070112
APA StyleCarev, I., Maravić, A., Ilić, N., Čikeš Čulić, V., Politeo, O., Zorić, Z., & Radan, M. (2020). UPLC-MS/MS Phytochemical Analysis of Two Croatian Cistus Species and Their Biological Activity. Life, 10(7), 112. https://doi.org/10.3390/life10070112








