Antioxidant Intervention Attenuates Aging-Related Changes in the Murine Ovary and Oocyte
Abstract
1. Introduction
2. Results
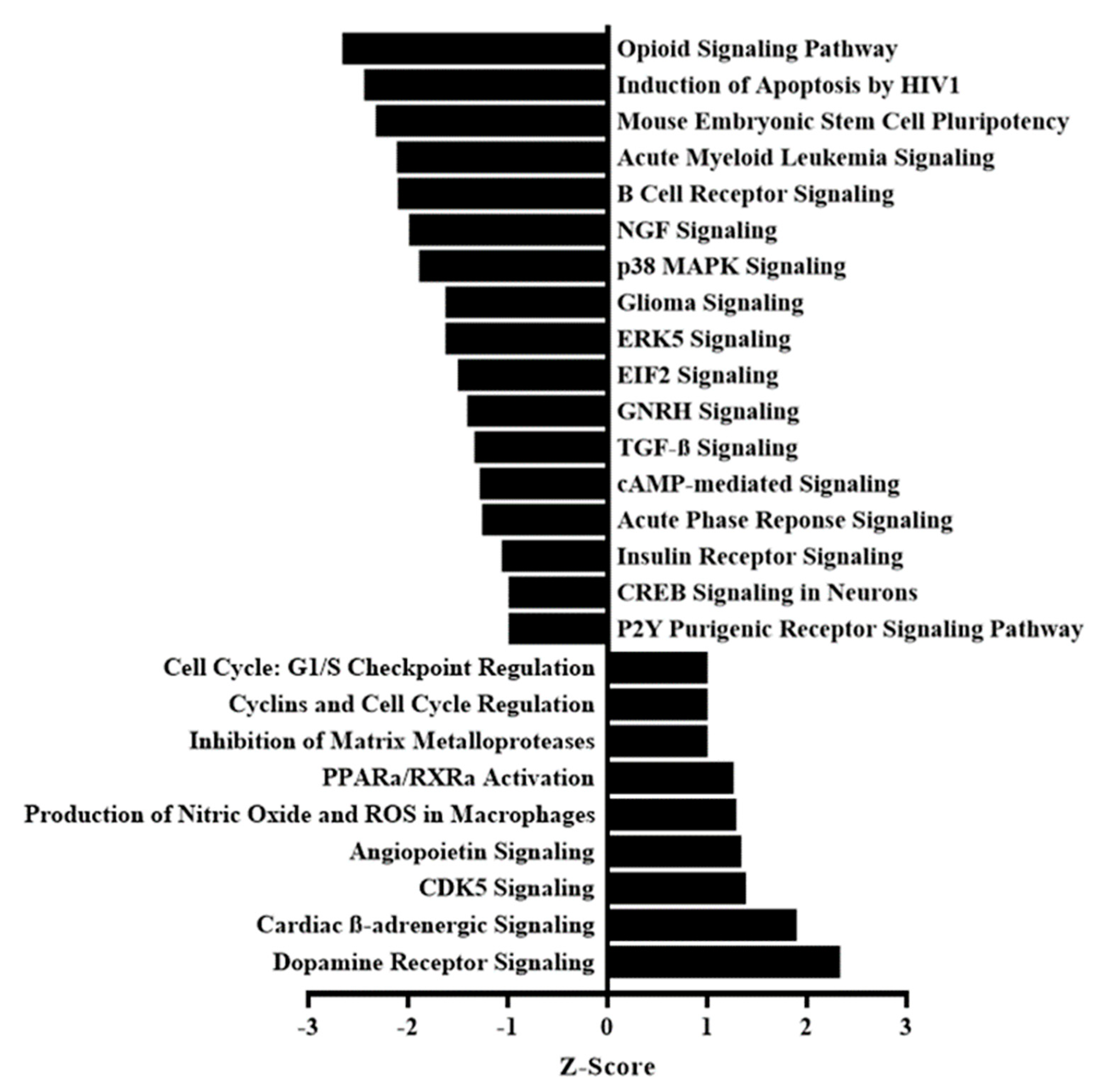
2.1. Pathway Analysis of the Aged Murine Ovarian Transcriptome Following Antioxidant Intervention
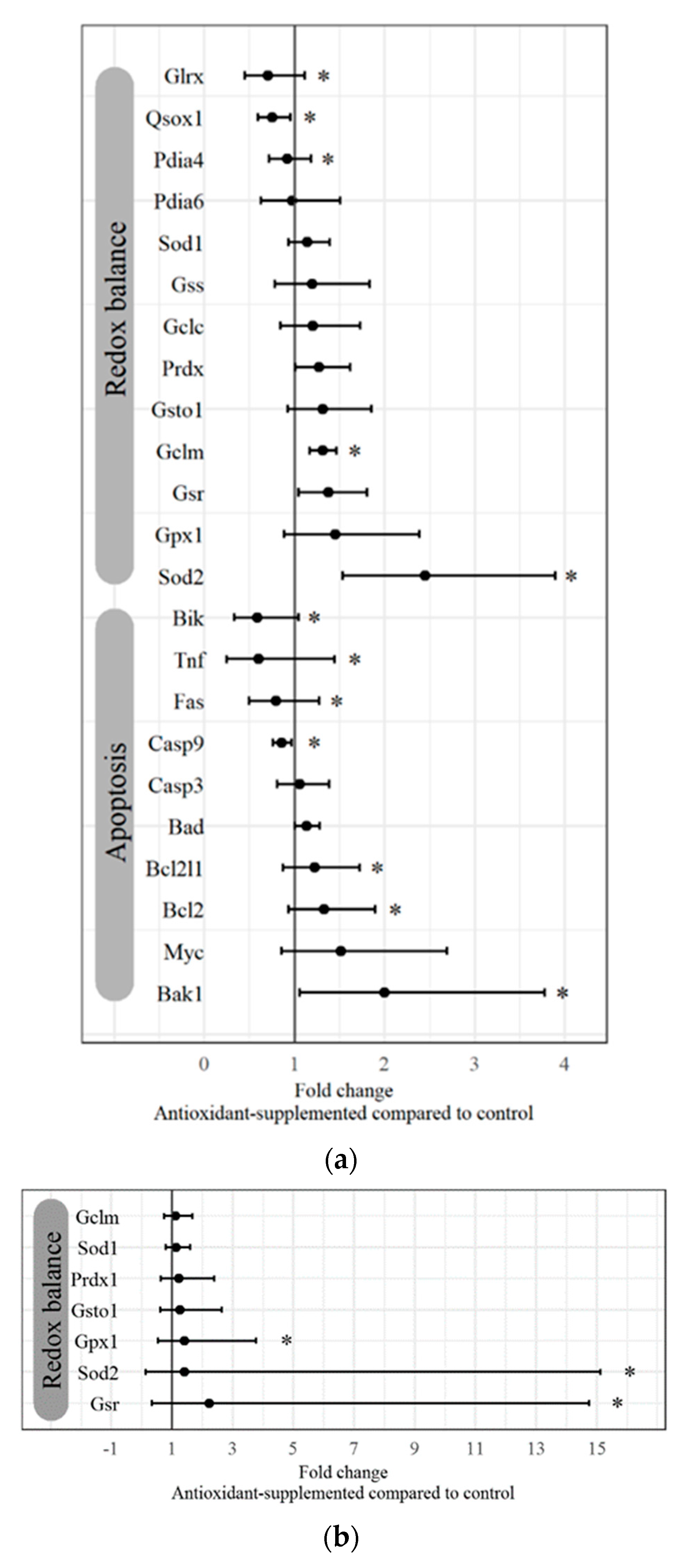
2.2. Differential Gene Expression in the Murine Ovary Following Antioxidant Intervention
2.3. Effects of Antioxidant Administration on Murine Oocyte Gene Expression
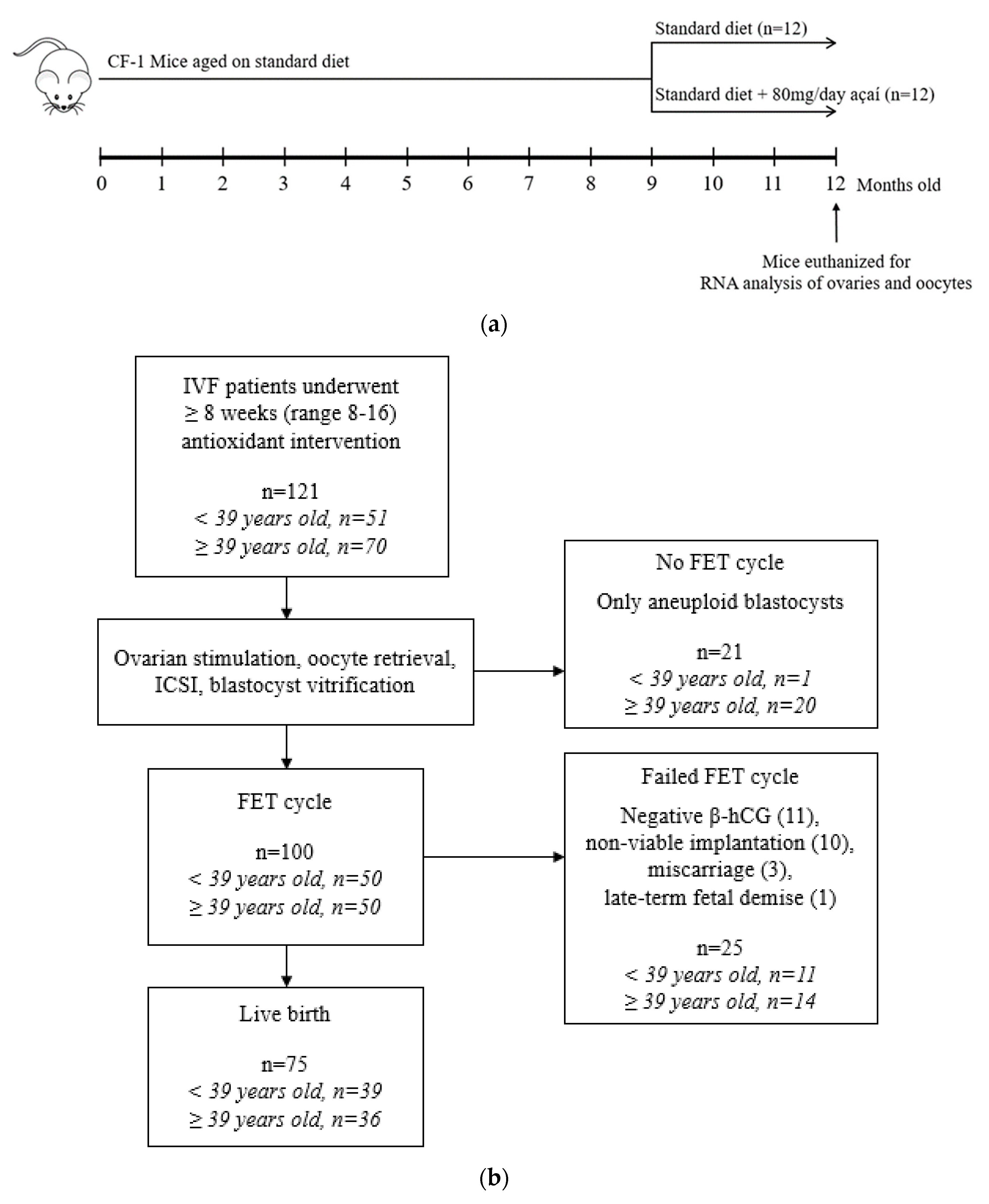
2.4. Feasibility Analysis after Antioxidant Intervention
2.5. Outcomes of FET Following Antioxidant Supplementation
3. Discussion
4. Materials and Methods
4.1. Murine Antioxidant Intervention Protocol
4.2. Murine RNA Isolation, Transcriptome Analysis, and Quantitative Real-Time PCR
4.3. Feasibility Analysis of Antioxidant Intervention Protocol and Outcomes
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AMA | Advanced maternal age |
| ROS | Reactive oxygen species |
| IVF | In vitro fertilization |
| CASP9 | Caspase 9 |
| CDK5 | Cyclin-dependent kinase 5 |
| HIV1 | Human immunodeficiency virus 1 |
| TNFRSF1B | TNF receptor superfamily member 1B |
| TGFβ | Transforming growth factor beta |
| MAPK | Mitogen-activated protein kinase |
| NF-κB | Nuclear factor kappa light chain enhancer of activated B cells |
| PAX4 | Paired box gene 4 |
| TNF | Tumor necrosis factor |
| FAS | Fas cell surface death receptor |
| BIK | Bcl-2-interacting killer |
| BCL2L1 | Bcl-2-like 1 |
| BCL2 | B-cell lymphoma 2 apoptosis regulator |
| BAK1 | Bcl-2 homologous antagonist killer |
| GLRX | Glutaredoxin |
| QSOX1 | Quiescin sulfhydryl oxidase 1 |
| PDIA4 | Protein disulfide isomerase family A member 4 |
| GCLM | Glutamate cysteine ligase modifier subunit |
| SOD2 | Superoxide dismutase 2 |
| GPX1 | Glutathione peroxidase |
| GSR | Glutathione reductase |
| FSH | Follicle stimulating hormone |
| FET | Frozen embryo transfer |
| PGT-A | Preimplantation genetic testing for aneuploidy |
| aCGH | Array comparative genomic hybridization |
| NGS | Next generation sequencing |
| β-hCG | Beta-human chorionic gonadotropin |
| GAE | Gallic acid equivalent |
| ORAC | Oxygen radical absorbance capacity |
| TE | Trolox equivalent |
| RPL19 | Ribosomal protein L19 |
References
- Eichenlaub-Ritter, U. Oocyte ageing and its cellular basis. Int. J. Dev. Biol. 2012, 56, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Crawford, N.M.; Steiner, A.Z. Age-related Infertility. Obs. Gynecol. Clin. N. Am. 2015, 42, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Llarena, N.; Hine, C. Reproductive Longevity and Aging: Geroscience Approaches to Maintain Long-Term Ovarian Fitness. J. Gerontol. A Biol. Sci. Med. Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tarin, J.J. Potential effects of age-associated oxidative stress on mammalian oocytes/embryos. Mol. Hum. Reprod. 1996, 2, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Ruder, E.H.; Hartman, T.J.; Blumberg, J.; Goldman, M.B. Oxidative stress and antioxidants: Exposure and impact on female fertility. Hum. Reprod. Update 2008, 14, 345–357. [Google Scholar] [CrossRef]
- Carbone, M.C.; Tatone, C.; Delle Monache, S.; Marci, R.; Caserta, D.; Colonna, R.; Amicarelli, F. Antioxidant enzymatic defences in human follicular fluid: Characterization and age-dependent changes. Mol. Hum. Reprod. 2003, 9, 639–643. [Google Scholar] [CrossRef]
- Lim, J.; Luderer, U. Oxidative damage increases and antioxidant gene expression decreases with aging in the mouse ovary. Biol. Reprod. 2011, 84, 775–782. [Google Scholar] [CrossRef]
- Steuerwald, N.M.; Bermudez, M.G.; Wells, D.; Munne, S.; Cohen, J. Maternal age-related differential global expression profiles observed in human oocytes. Reprod. BioMed. Online 2007, 14, 700–708. [Google Scholar] [CrossRef]
- Hamatani, T.; Falco, G.; Carter, M.G.; Akutsu, H.; Stagg, C.A.; Sharov, A.A.; Dudekula, D.B.; VanBuren, V.; Ko, M.S.H. Age-associated alteration of gene expression patterns in mouse oocytes. Hum. Mol. Genet. 2004, 13, 2263–2278. [Google Scholar] [CrossRef]
- McCallie, B.R.; Parks, J.C.; Trahan, G.D.; Jones, K.L.; Coate, B.D.; Griffin, D.K.; Schoolcraft, W.B.; Katz-Jaffe, M.G. Compromised global embryonic transcriptome associated with advanced maternal age. J. Assist. Reprod. Genet. 2019, 36, 915–924. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, Y.; Li, J.; Yu, Y.; Zhang, W.; Song, M.; Liu, Z.; Min, Z.; Hu, H.; Jing, Y.; et al. Single-Cell Transcriptomic Atlas of Primate Ovarian Aging. Cell 2020, 180, 585–600. [Google Scholar] [CrossRef] [PubMed]
- McCallie, B.R.; Parks, J.C.; Strieby, A.L.; Schoolcraft, W.B.; Katz-Jaffe, M.G. Human blastocysts exhibit unique microrna profiles in relation to maternal age and chromosome constitution. J. Assist. Reprod. Genet. 2014, 31, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Dumesic, D.A.; Meldrum, D.R.; Katz-Jaffe, M.G.; Krisher, R.L.; Schoolcraft, W.B. Oocyte environment: Follicular fluid and cumulus cells are critical for oocyte health. Fertil. Steril. 2015, 103, 303–316. [Google Scholar] [CrossRef] [PubMed]
- McReynolds, S.; Dzieciatkowska, M.; McCallie, B.R.; Mitchell, S.D.; Stevens, J.; Hansen, K.; Schoolcraft, W.B.; Katz-Jaffe, M.G. Impact of maternal aging on the molecular signature of human cumulus cells. Fertil. Steril. 2012, 98, 1574–1580. [Google Scholar] [CrossRef]
- Mihalas, B.P.; Redgrove, K.A.; McLaughlin, E.A.; Nixon, B. Molecular Mechanisms Responsible for Increased Vulnerability of the Ageing Oocyte to Oxidative Damage. Oxid. Med. Cell. Longev. 2017, 2017, 4015874. [Google Scholar] [CrossRef]
- Fujii, J.; Iuchi, Y.; Okada, F. Fundamental roles of reactive oxygen species and protective mechanisms in the female reproductive system. Reprod. Biol. Endocrinol. 2005, 3, 43. [Google Scholar] [CrossRef]
- Behrman, H.R.; Kodaman, P.H.; Preston, S.L.; Gao, S. Oxidative Stress and the Ovary. J. Soc. Gynecol. Investig. 2001, 8, S40–S42. [Google Scholar]
- Kodaman, P.H.; Behrman, H.R. Endocrine-Regulated and Protein Kinase C-Dependent Generation of Superoxide by Rat Preovulatory Follicles. Endocrinology 2001, 142, 687–693. [Google Scholar] [CrossRef]
- Freitas, C.; Neto, A.C.; Matos, L.; Silva, E.; Ribeiro, A.; Silva-Carvalho, J.L.; Almeida, H. Follicular Fluid redox involvement for ovarian follicle growth. J. Ovarian Res. 2017, 10, 44. [Google Scholar] [CrossRef]
- Tsai-Turton, M.; Luderer, U. Opposing Effects of Glutathione Depletion and Follicle-Stimulating Hormone on Reactive Oxygen Species and Apoptosis in Cultured Preovulatory Rat Follicles. Endocrinology 2006, 147, 1224–1236. [Google Scholar] [CrossRef]
- Jana, S.K.; K, N.B.; Chattapadhyay, R.; Chakravarty, B.; Chaudhury, K. Upper control limit of reactive oxygen species in follicular fluid beyond which viable embryo formation is not favorable. Reprod. Toxicol. 2010, 29, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Wheeler, M.B.; Krisher, R.L. Disrupted Redox Homeostasis and Aberrant Redox Gene Expression in Porcine Oocytes Contribute to Decreased Developmental Competence. Biol. Reprod. 2012, 87, 78. [Google Scholar] [CrossRef] [PubMed]
- Yeh, J.; Bowman, M.J.; Browne, R.W.; Chen, N. Reproductive aging results in a reconfigured ovarian antioxidant defense profile in rats. Fertil. Steril. 2005, 84, 1109–1113. [Google Scholar] [CrossRef] [PubMed]
- Lord, T.; Martin, J.H.; Aitken, R.J. Accumulation of Electrophilic Aldehydes During Postovulatory Aging of Mouse Oocytes Causes Reduced Fertility, Oxidative Stress, and Apoptosis. Biol. Reprod. 2015, 92, 33. [Google Scholar] [CrossRef]
- Xian, Y.; Liang, L.; Qi, S.; Xie, Y.; Song, B.; Ouyang, S.; Xie, Y.; Sun, X.; Wang, W. Antioxidants retard the ageing of mouse oocytes. Mol. Med. Rep. 2018, 18, 1981–1986. [Google Scholar] [CrossRef]
- Lian, H.-Y.; Gao, Y.; Jiao, G.-Z.; Sun, M.-J.; Wu, W.-F.; Wang, T.-Y.; Li, H.; Tan, J.-H. Antioxidant supplementation overcomes the deleterious effects of maternal restraint stress-induced oxidative stress on mouse oocytes. Reproduction 2013, 146, 559–568. [Google Scholar] [CrossRef]
- Song, C.; Peng, W.; Yin, S.; Zhao, J.; Fu, B.; Zhang, J.; Mao, T.; Wu, H.; Zhang, Y. Melatonin improves age-induced fertility decline and attenuates ovarian mitochondrial oxidative stress in mice. Sci. Rep. 2016, 6, 35165. [Google Scholar] [CrossRef]
- Kandemir, Y.B.; Aydin, C.; Gorgisen, G. The effects of melatonin on oxidative stress and prevention of primordial follicle loss via activation of mTOR pathway in the rat ovary. Cell. Mol. Biol. 2017, 63, 100–106. [Google Scholar] [CrossRef]
- Liu, X.; Lin, X.; Mi, Y.; Li, J.; Zhang, C. Grape Seed Proanthocyanidin Extract Prevents Ovarian Aging by Inhibiting Oxidative Stress in the Hens. Oxid. Med. Cell. Longev. 2018, 2018, 9390810. [Google Scholar] [CrossRef]
- Liu, X.; Lin, X.; Zhang, S.; Guo, C.; Li, J.; Mi, Y.; Zhang, C. Lycopene ameliorates oxidative stress in the aging chicken ovary via activation of Nrf2/HO-1 pathway. Aging 2018, 10, 2016–2036. [Google Scholar] [CrossRef]
- Ruder, E.H.; Hartman, T.J.; Reindollar, R.H.; Goldman, M.B. Female dietary antioxidant intake and time to pregnancy among couples treated for unexplained infertility. Fertil. Steril. 2014, 101, 759–766. [Google Scholar] [CrossRef][Green Version]
- Velthut, A.; Zilmer, M.; Zilmer, K.; Kaart, T.; Karro, H.; Salumets, A. Elevated blood plasma antioxidant status is favourable for achieving IVF/ICSI pregnancy. Reprod. Biomed. Online 2013, 26, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sharma, R.K.; Falcone, T.; Goldberg, J.; Agarwal, A. Importance portance of reactive oxygen species in the peritoneal fluid of women with endometriosis or idiopathic infertility. Fertil. Steril. 1997, 68, 826–830. [Google Scholar] [CrossRef]
- Bedaiwy, M.A.; Falcone, T.; Sharma, R.K.; Goldberg, J.M.; Attaran, M.; Nelson, D.R.; Agarwal, A. Prediction of endometriosis with serum and peritoneal fluid markers: A prospective controlled trial. Hum. Reprod. 2002, 17, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Schauss, A.G.; Wu, X.; Prior, R.L.; Ou, B.; Huang, D.; Owens, J.; Agarwal, A.; Jensen, G.S.; Hart, A.N.; Shanbrom, E. Antioxidant capacity and other bioactivities of the freeze-dried Amazonian palm berry, Euterpe oleraceae mart. (acai). J. Agric. Food Chem. 2006, 54, 8604–8610. [Google Scholar] [CrossRef]
- Kang, J.; Xie, C.; Li, Z.; Nagarajan, S.; Schauss, A.G.; Wu, T.; Wu, X. Flavonoids from acai (Euterpe oleracea Mart.) pulp and their antioxidant and anti-inflammatory activities. Food Chem. 2011, 128, 152–157. [Google Scholar] [CrossRef]
- Merz, C.; Saller, S.; Kunz, L.; Xu, J.; Yeoman, R.R.; Ting, A.Y.; Lawson, M.S.; Stouffer, R.L.; Hennebold, J.D.; Pau, F.; et al. Expression of the beta-2 adrenergic receptor (ADRB-2) in human and monkey ovarian follicles: A marker of growing follicles? J. Ovarian Res. 2015, 8, 8. [Google Scholar] [CrossRef]
- Wheeler, A.G.; Lean, J.; Walker, M. Peripheral progesterone concentrations in the luteal-phase ewe: Effects of a beta-adrenergic receptor antagonist and two beta 2-adrenergic agonists. J. Endocrinol. 1988, 116, 137–142. [Google Scholar] [CrossRef]
- Parillo, F.; Maranesi, M.; Mignini, F.; Marinelli, L.; Di Stefano, A.; Boiti, C.; Zerani, M. Evidence for a dopamine intrinsic direct role in the regulation of the ovary reproductive function: In vitro study on rabbit corpora lutea. PLoS ONE 2014, 9, e104797. [Google Scholar] [CrossRef]
- Cruz, G.; Fernandois, D.; Paredes, A.H. Ovarian function and reproductive senescence in the rat: Role of ovarian sympathetic innervation. Reproduction 2017, 153, R59–R68. [Google Scholar] [CrossRef]
- Xie, C.; Kang, J.; Li, Z.; Schauss, A.G.; Badger, T.M.; Nagarajan, S.; Wu, T.; Wu, X. The açaí flavonoid velutin is a potent anti-inflammatory agent: Blockade of LPS-mediated TNF-α and IL-6 production through inhibiting NF-κB activation and MAPK pathway. J. Nutr. Biochem. 2012, 23, 1184–1191. [Google Scholar] [CrossRef]
- Lim, J.; Nakamura, B.N.; Mohar, I.; Kavanagh, T.J.; Luderer, U. Glutamate Cysteine Ligase Modifier Subunit (Gclm) Null Mice Have Increased Ovarian Oxidative Stress and Accelerated Age-Related Ovarian Failure. Endocrinology 2015, 156, 3329–3343. [Google Scholar] [CrossRef]
- Perkins, A.T.; Das, T.M.; Panzera, L.C.; Bickel, S.E. Oxidative stress in oocytes during midprophase induces premature loss of cohesion and chromosome segregation errors. Proc. Natl. Acad. Sci. USA 2016, 113, E6823–E6830. [Google Scholar] [CrossRef]
- Cheng, J.M.; Liu, Y.X. Age-Related Loss of Cohesion: Causes and Effects. Int. J. Mol. Sci. 2017, 18, 1578. [Google Scholar] [CrossRef]
- Fortier, M.E.; Audet, I.; Giguere, A.; Laforest, J.P.; Bilodeau, J.F.; Quesnel, H.; Matte, J.J. Effect of dietary organic and inorganic selenium on antioxidant status, embryo development, and reproductive performance in hyperovulatory first-parity gilts. J. Anim. Sci. 2012, 90, 231–240. [Google Scholar] [CrossRef]
- Xiong, X.; Lan, D.; Li, J.; Lin, Y.; Li, M. Selenium supplementation during in vitro maturation enhances meiosis and developmental capacity of yak oocytes. Anim. Sci. J. 2018, 89, 298–306. [Google Scholar] [CrossRef]
- Dattilo, M.; D’Amato, G.; Caroppo, E.; Menezo, Y. Improvement of gamete quality by stimulating and feeding the endogenous antioxidant system: Mechanisms, clinical results, insights on gene-environment interactions and the role of diet. J. Assist. Reprod. Genet. 2016, 33, 1633–1648. [Google Scholar] [CrossRef]
- Mikwar, M.; MacFarlane, A.J.; Marchetti, F. Mechanisms of oocyte aneuploidy associated with advanced maternal age. Mutat. Res. 2020, 785, 108320. [Google Scholar] [CrossRef]
- Franasiak, J.M.; Forman, E.J.; Hong, K.H.; Werner, M.D.; Upham, K.M.; Treff, N.R.; Scott, R.T. The nature of aneuploidy with increasing age of the female partner: A review of 15,169 consecutive trophectoderm biopsies evaluated with comprehensive chromosomal screening. Fertil. Steril. 2014, 101, 656–663. [Google Scholar] [CrossRef]
- Katz-Jaffe, M.G.; McReynolds, S.; Henry, L.; McCubbin, N.; Mann, R.S.; Tucci, R.; McCormick, S.; Schoolcraft, W.B. Age-related pregnancy loss is largely overcome with single euploid embryo transfer. Fertil. Steril. 2020, 114, e428. [Google Scholar] [CrossRef]
- Riley, J.K.; Jungheim, E.S. Is there a role for diet in ameliorating the reproductive sequelae associated with chronic low-grade inflammation in polycystic ovary syndrome and obesity? Fertil. Steril. 2016, 106, 520–527. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Poston, L.; Igosheva, N.; Mistry, H.D.; Seed, P.T.; Shennan, A.H.; Rana, S.; Karumanchi, S.A.; Chappell, L.C. Role of oxidative stress and antioxidant supplementation in pregnancy disorders. Am. J. Clin. Nutr. 2011, 94, 1980S–1985S. [Google Scholar] [CrossRef]
- Young, D.; Klepacka, D.; McGarvey, M.; Schoolcraft, W.B.; Katz-Jaffe, M.G. Infertility patients with chromosome inversions are not susceptible to an inter-chromosomal effect. J. Assist. Reprod. Genet. 2019, 36, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Alessandra-Perini, J.; Rodrigues-Baptista, K.C.; Machado, D.E.; Nasciutti, L.E.; Perini, J.A. Anticancer potential, molecular mechanisms and toxicity of Euterpe oleracea extract (açaí): A systematic review. PLoS ONE 2018, 13, e0200101. [Google Scholar] [CrossRef] [PubMed]
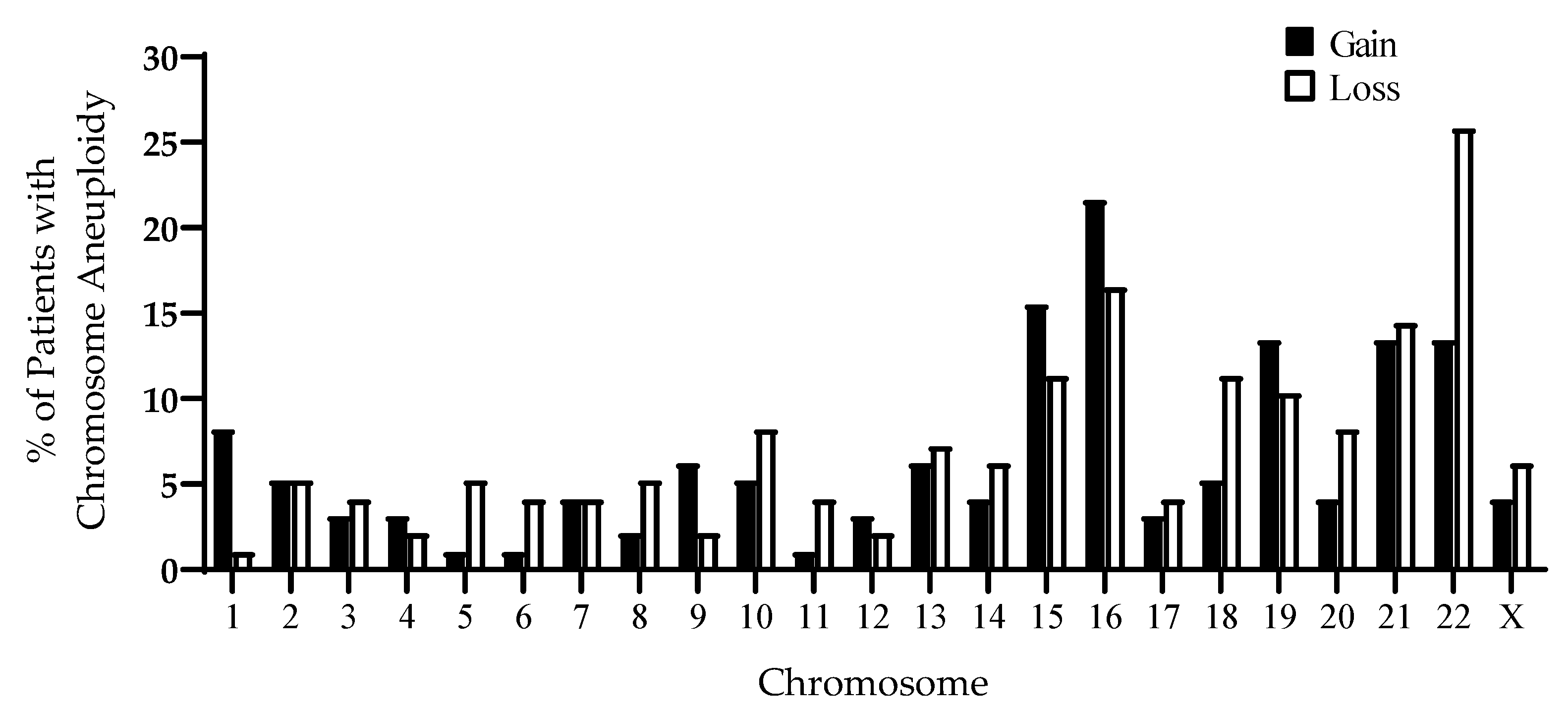
| FET Patients <39 Years, n = 50 | FET Patients ≥39 Years, n = 50 | |
|---|---|---|
| Age, years | 34.5 ± 2.4 | 40.3 ± 1.3 |
| Prior failed IVF cycles, n (range) | 2.1 ± 1.4 (1–8) | 1.9 ± 1.4 (1–7) |
| Baseline ovarian reserve parameters | ||
| AMH, ng/mL | 2.9 ± 2.9 | 2.8 ± 2.1 |
| D3 FSH, ng/mL | 8.0 ± 3.4 | 8.4 ± 3.1 |
| D3 AFC, n | 16.6 ± 8.3 | 15.7 ± 8.5 |
| IVF outcomes | ||
| Oocytes collected, n | 17.4 ± 8.3 | 17.4 ± 9.2 |
| Total blastocysts, n | 5.0 ± 2.9 | 5.2 ± 3.3 |
| Total blastocysts, % | 30.4 ± 14.1 | 33.7 ± 18.3 |
| Euploid blastocysts, n | 3.0 ± 1.9 | 2.1 ± 1.6 |
| Euploid blastocysts, % | 63.8 ± 24.8 | 43.4 ± 22.3 * |
| Embryos transferred | 1.5 ± 0.5 | 1.4 ± 0.5 |
| FET outcomes | ||
| Implantation rate (FHT) | 72.0% | 75.3% |
| Negative β-hCG | 4 (8%) | 7 (14%) |
| Nonviable implantation | 6 (12%) | 4 (8%) |
| Miscarriage | 1 (2%) | 2 (4%) |
| Fetal demise at 34 weeks | 0 (0%) | 1 (2%) |
| Live birth | 39 (78%) | 36 (72%) |
| Twin birth | 11 (28%) | 9 (24%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katz-Jaffe, M.G.; Lane, S.L.; Parks, J.C.; McCallie, B.R.; Makloski, R.; Schoolcraft, W.B. Antioxidant Intervention Attenuates Aging-Related Changes in the Murine Ovary and Oocyte. Life 2020, 10, 250. https://doi.org/10.3390/life10110250
Katz-Jaffe MG, Lane SL, Parks JC, McCallie BR, Makloski R, Schoolcraft WB. Antioxidant Intervention Attenuates Aging-Related Changes in the Murine Ovary and Oocyte. Life. 2020; 10(11):250. https://doi.org/10.3390/life10110250
Chicago/Turabian StyleKatz-Jaffe, Mandy G., Sydney L. Lane, Jason C. Parks, Blair R. McCallie, Rachel Makloski, and William B. Schoolcraft. 2020. "Antioxidant Intervention Attenuates Aging-Related Changes in the Murine Ovary and Oocyte" Life 10, no. 11: 250. https://doi.org/10.3390/life10110250
APA StyleKatz-Jaffe, M. G., Lane, S. L., Parks, J. C., McCallie, B. R., Makloski, R., & Schoolcraft, W. B. (2020). Antioxidant Intervention Attenuates Aging-Related Changes in the Murine Ovary and Oocyte. Life, 10(11), 250. https://doi.org/10.3390/life10110250





