Spatial and Temporal Controls on the Distribution of Indium in Xenothermal Vein-Deposits: The Huari Huari District, Potosí, Bolivia
Abstract
:1. Introduction
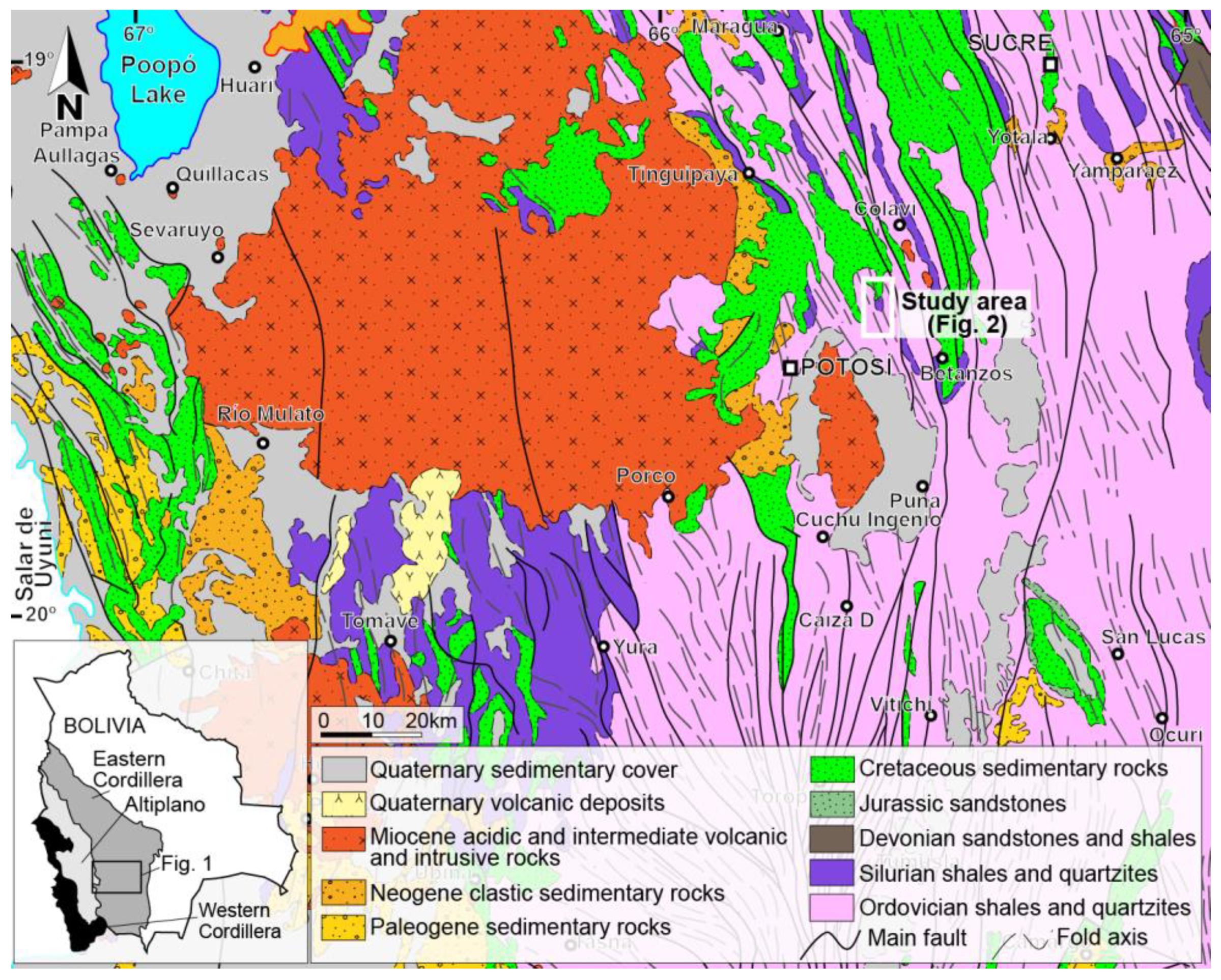
2. Geologic Setting
2.1. Geodynamic Setting
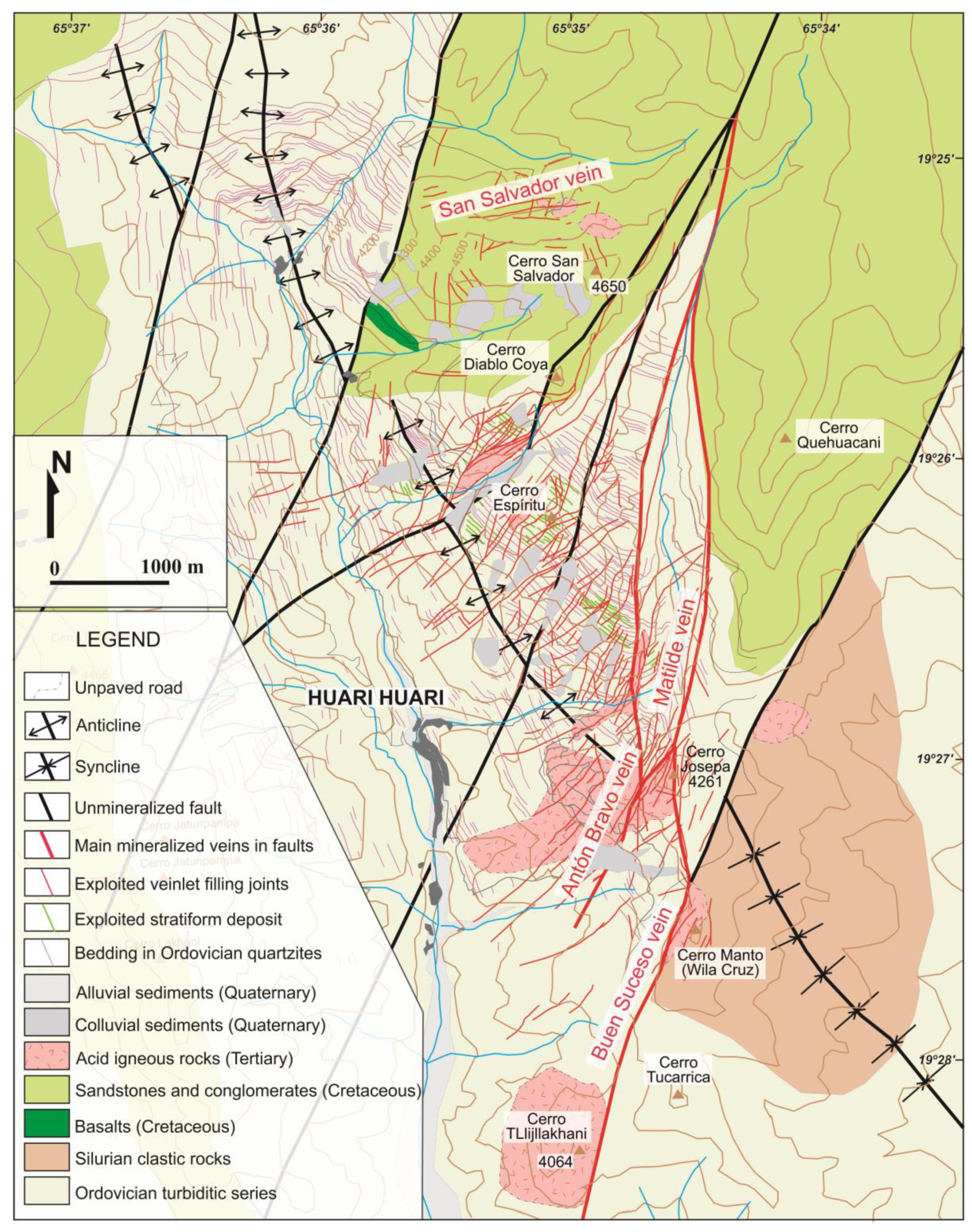
2.2. Geology of the Deposit
3. Materials and Methods
4. Mineralogy and Textures
4.1. Stratiform Mineralization
4.1.1. Pyrite
4.1.2. Sphalerite
4.1.3. Cassiterite
4.1.4. Galena
4.2. Polymetallic Vein Mineralization
4.2.1. Quartz
4.2.2. Cassiterite
4.2.3. Arsenopyrite
4.2.4. Pyrrhotite
4.2.5. Sphalerite
4.2.6. Pyrite
4.2.7. Chalcopyrite
4.2.8. Marcasite
4.2.9. Galena
4.2.10. Stannite
4.2.11. Tetrahedrite Group Minerals
4.2.12. Other Sulfosalts
4.2.13. Siderite
4.2.14. Other
4.3. Paragenetic Sequence
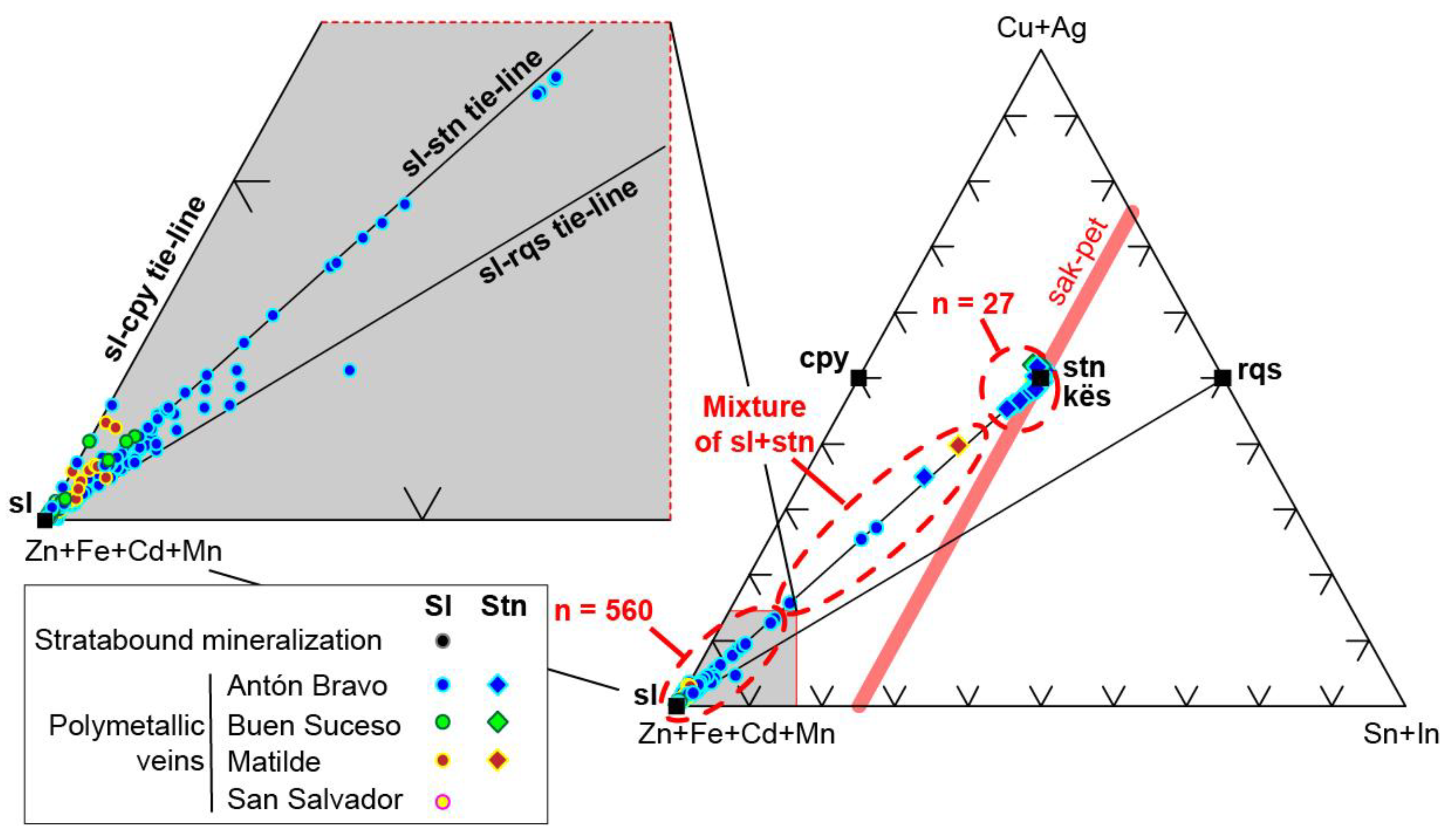
5. Ore Mineral Geochemistry
5.1. Arsenopyrite
5.2. Pyrrhotite
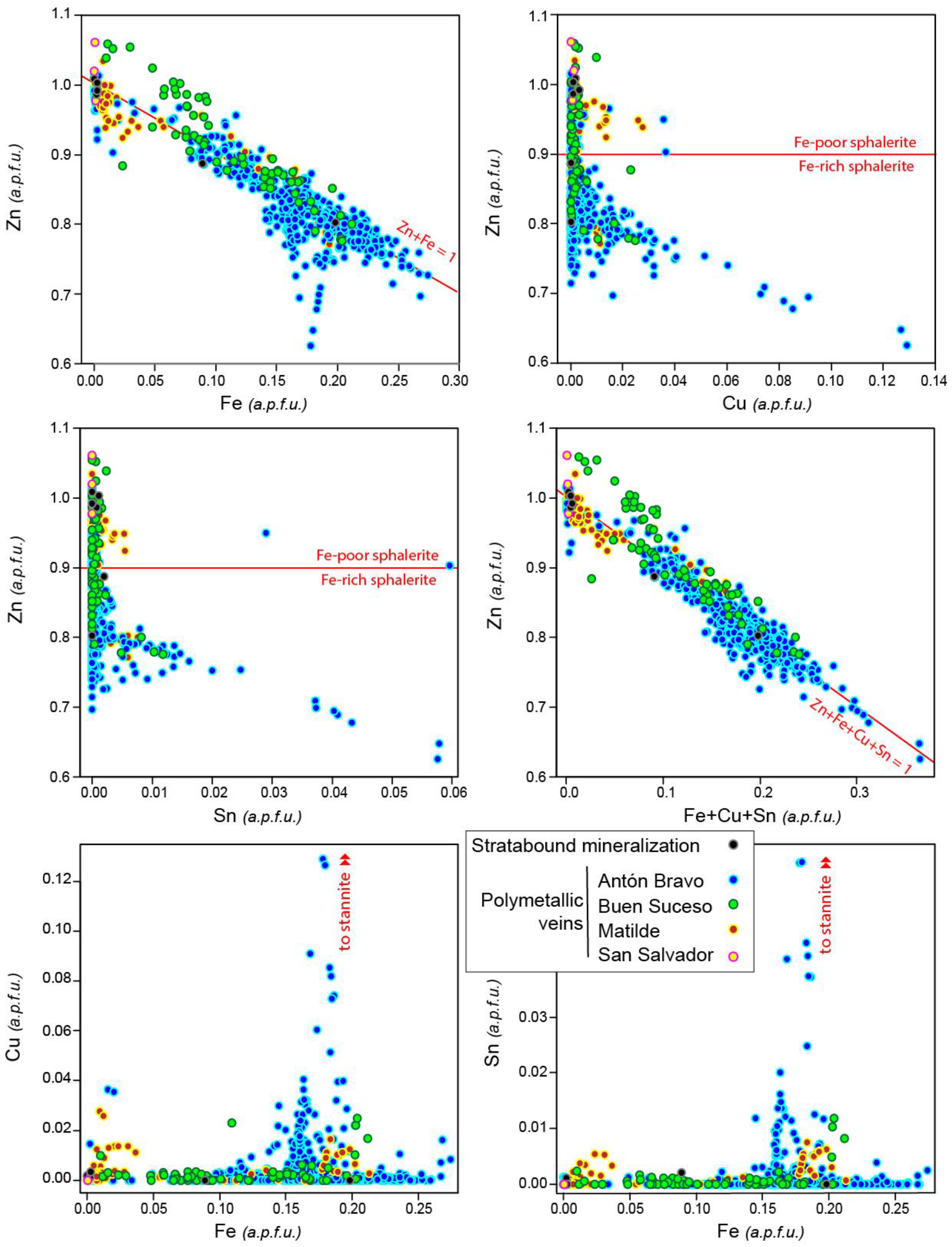
5.3. Sphalerite
5.4. Stannite Group Minerals
5.5. Galena
5.6. Tetrahedrite Group Minerals
5.7. Other Sulfosalts
6. Discussion
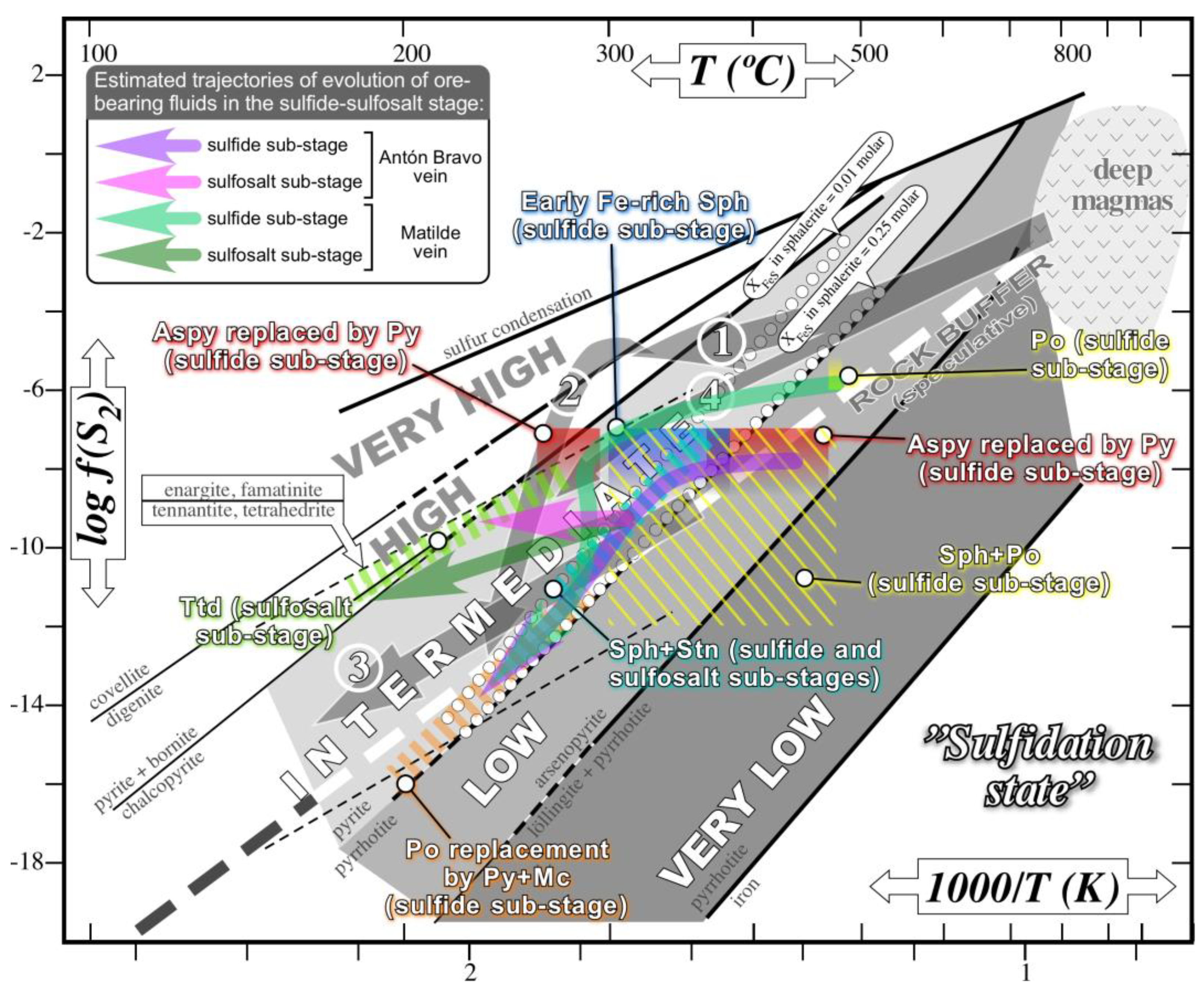
6.1. Ore-Forming Conditions
6.2. Mineralogical Expression of Indium
6.3. Temporal and Spatial Controls of Indium Distribution
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- European Commission, Critical Raw Materials. Available online: https://ec.europa.eu/growth/sectors/raw-materials/specific-interest/critical_en (accessed on 1 April 2019).
- Schulz, K.J.; DeYoung, J.H., Jr.; Seal, R.R., II; Bradley, D.C. Critical Mineral Resources of the United States—Economic and Environmental Geology and Prospects for Future Supply; U.S. Geological Survey Professional Paper; U.S. Geological Survey: Reston, VI, USA, 2017.
- Kesler, S.E.; Simon, A.C. Mineral Resources, Economics and the Environment, 2nd ed.; Cambridge University Press: Cambridge, UK, 2015; pp. 243–244. [Google Scholar]
- Bleiwas, D.I. Byproduct Mineral Commodities Used for the Production Of Photovoltaic Cells; U.S. Geological Survey Circular; US Geological Survey: Reston, VI, USA, 2010; Volume 1365, pp. 1–10.
- Jorgenson, J.D.; George, M.W. Mineral Commodity Profile: Indium. In U.S. Geological Survey Open-File Report 2004-1300. Available online: https://pubs.usgs.gov/of/2004/1300/2004-1300.pdf (accessed on 3 December 2018).
- Werner, T.T.; Mudd, G.M.; Jowitt, S.M. The world’s by-product and critical metal resources part III: A global assessment of indium. Ore Geol. Rev. 2017, 86, 939–956. [Google Scholar] [CrossRef]
- Pavlova, G.G.; Palessky, S.V.; Borisenko, A.S.; Vladimirov, A.G.; Seifert, T.; Phan, L.A. Indium in cassiterite and ores of tin deposits. Ore Geol. Rev. 2015, 66, 99–113. [Google Scholar] [CrossRef]
- Lerouge, C.; Gloaguen, E.; Wille, G.; Bailly, L. Distribution of In and other trace metals in cassiterite and associated minerals in Sn ± W ore deposits of western Variscan Belt. Eur. J. Mineral. 2017, 29, 739–753. [Google Scholar] [CrossRef]
- Ohta, E. Occurrence and chemistry of indium-containing minerals from the Toyoha Mina, Hokkaido, Japan. Min. Geol. 1989, 39, 355–372. [Google Scholar]
- Schwarz-Schampera, U.; Herzig, P.M. Indium: Geology, Mineralogy and Economics; Springer: Heidelberg, Germany, 2002. [Google Scholar]
- Moura, M.A.; Botelho, N.F.; Carvalho de Mendonça, F. The indium-rich sulfides and rare arsenates of the Sn-In-mineralized Mangabeira A-type granite, central Brazil. Can. Mineral. 2007, 45, 485–496. [Google Scholar] [CrossRef]
- Cook, N.J.; Sundblad, K.; Valkama, M.; Nygård, R.; Ciobanu, C.L.; Danyushevsky, L. Indium mineralization in A-type granites in southeastern Finland: Insights into mineralogy and partitioning between co-existing minerals. Chem. Geol. 2011, 284, 62–73. [Google Scholar] [CrossRef]
- Jovic, S.M.; Guido, D.M.; Melgarejo, J.C.; Páez, G.N.; Ruiz, R.; Schalamuk, I.B. The indium-bearing minerals of the Pingüino polymetallic vein system, Deseado massif, Patagonia, Argentina. Can. Mineral. 2011, 49, 931–946. [Google Scholar] [CrossRef]
- Shimizu, T.; Morishita, Y. Petrographic, chemistry and near-infrared microthermometry of indium-bearing sphalerite from the Toyoha polymetallic deposit, Japan. Econ. Geol. 2012, 107, 723–735. [Google Scholar] [CrossRef]
- Jonsson, E.; Högdahl, K.; Majka, J.; Lindeberg, T. Roquesite and associated indium-bearing sulfides from a Paleoproterozoic carbonate-hosted mineralization: Lindbom’s prospect, Bergslagen, Sweden. Can. Mineral. 2013, 51, 629–641. [Google Scholar] [CrossRef]
- Murakami, H.; Ishihara, S. Trace elements of indium-bearing sphalerite from tin-polymetallic deposits in Bolivia, China and Japan: A femto-second LA-ICPMS study. Ore Geol. Rev. 2013, 53, 223–243. [Google Scholar] [CrossRef]
- Belissont, R.; Boiron, M.C.; Luais, B.; Cathelineau, M. LA-ICP-MS analyses of minor and trace elements and bulk Ge isotopes in zoned Ge-rich sphalerites from the Noailhac—Saint Salvy deposit (France): Insights into incorporation mechanisms and ore deposition processes. Geochim. Cosmochim. Acta 2014, 126, 518–540. [Google Scholar] [CrossRef]
- Jiménez-Franco, A.; Alfonso, P.; Canet, C.; Trujillo, J.E. Mineral chemistry of In-bearing minerals in the Santa Fe mining district, Bolivia. Andean Geol. 2018, 45, 410–432. [Google Scholar] [CrossRef]
- Bauer, M.E.; Seifert, T.; Burisch, M.; Krause, J.; Richter, N.; Gutzmer, J. Indium-bearing sulfides from the Hämmerlein skarn deposit, Erzgebirge, Germany: Evidence for late-stage diffusion of indium into sphalerite. Miner. Deposita 2019, 54, 175–192. [Google Scholar] [CrossRef]
- Bauer, M.E.; Burisch, M.; Ostendorf, J.; Krause, J.; Frenzel, M.; Seifert, T.; Gutzmer, J. Trace element geochemistry of sphalerite in contrasting hydrothermal fluid systems of the Freiberg district, Germany: Insights from LA-ICP-MS analysis, near-infrared light microthermometry of sphalerite-hosted fluid inclusions, and sulfur isotope geochemistry. Miner. Deposita 2019, 54, 237–262. [Google Scholar]
- Heuschmidt, B.; Bellot de la Torre, J.; Miranda Angles, V.; Claure Zapata, M. Las Areas Prospectivas de Bolivia para yacimientos metalíferos. Boletín del Servicio Nacional de Geología y Minería 2002, 30, 1–154. [Google Scholar]
- Roskill Information Services. The Economics of Indium, 6th ed.; Roskill Information Services Ltd.: London, UK, 1996. [Google Scholar]
- Ishihara, S.; Murakami, H.; Marquez-Zavalia, M.F. Inferred indium resources of the Bolivian tin-polymetallic deposits. Resour. Geol. 2011, 61, 174–191. [Google Scholar] [CrossRef]
- Ahlfeld, F.; Schneider-Scherbina, A. Los yacimientos minerales y de hidrocarburos de Bolivia; Ministerio de Minas y Petróleo: La Paz, Bolivia, 1964; pp. 1–388. [Google Scholar]
- Kelly, W.C.; Turneaure, F.S. Mineralongy, paragenesis and geothermometry of the tin and tungsten deposits of the Eastern Andes, Bolivian. Econ. Geol. 1970, 65, 609–680. [Google Scholar] [CrossRef]
- Arce-Burgoa, O. Metalliferous Ore Deposits of Bolivia, 2nd ed.; SPC Impresores: La Paz, Bolivia, 2009; pp. 1–233. [Google Scholar]
- Suarez-Soruco, R. Compendio de Geología de Bolivia; Servicio Nacional de Geología y Minería, Yacimientos Petrolíferos Fiscales Bolivianos: Cochabamba, Bolivia, 2000; pp. 39–76. [Google Scholar]
- Jiménez, N.; López-Velásquez, S. Magmatism in the Huarina belt, Bolivia, and its geotectonic implications. Tectonophysics 2008, 459, 85–106. [Google Scholar] [CrossRef]
- Wörner, G.; Schildgen, T.F.; Reich, M. The Central Andes: Elements of an extreme land. Elements 2018, 14, 225–230. [Google Scholar] [CrossRef]
- Wörner, G.; Mamani, M.; Blum-Oeste, M. Magmatism in the Central Andes. Elements 2018, 14, 237–244. [Google Scholar] [CrossRef]
- Sempere, T. Phanerozoic Evolution of Bolivia and Adjacent Regions; American Association of Petroleum Geologists (AAPG) Memoir: Tulsa, OK, USA, 1995; Volume 62, pp. 207–230. [Google Scholar]
- Sempere, T.; Hérail, G.; Oller, J.; Bonhomme, M.G. Late Oligocene-Early Miocene major tectonic crisis and related basins in Bolivia. Geology 1990, 18, 946–949. [Google Scholar] [CrossRef]
- Lehmann, B.; Ishihara, S.; Michel, H.; Miller, J.; Rapela, C.; Sanchez, A.; Tistl, M.; Winkelmann, L. The Bolivian tin province and regional tin distribution in the Central Andes: A reassessment. Econ. Geol. 1990, 85, 1044–1058. [Google Scholar] [CrossRef]
- Lehmann, B.; Dietrich, A.; Heinhorst, J.; Metrich, N.; Mosbah, M.; Palacios, C.; Schneider, H.J.; Wallianos, A.; Webster, J.; Winkelmann, L. Boron in the Bolivian tin belt. Miner. Depos. 2000, 35, 223–232. [Google Scholar] [CrossRef]
- Ishihara, S. The granitoid series and mineralization. Econ. Geol. 1981, 75th Anniv. Volume. 458–484. [Google Scholar] [CrossRef]
- Clark, A.H.; Farrar, E.; Caelles, J.C.; Haynes, S.J.; Lortie, R.B.; McBride, S.L.; Quirt, G.S.; Robertson, R.C.R.; Zentilli, M. Longitudinal variations in the metallogenetic evolution of the Central Andes: A progress report. Geol. Assoc. Can. Spec. Pap. 1976, 14, 23–58. [Google Scholar]
- Sillitoe, R.H. Andean mineralization: A model for the metal-logeny of convergent plate margins. Geol. Assoc. Can. Spec. Pap. 1976, 14, 59–100. [Google Scholar]
- Sillitoe, R.H. Musings on future exploration targets and strategies in the Andes. Econ. Geol. Sp. Publ. 2004, 11, 1–14. [Google Scholar]
- Mlynarczyk, M.S.J.; Williams-Jones, A.E. The role of collisional tectonics in the metallogeny of the Central Andean tin belt. Earth Planet. Sci. Lett. 2005, 240, 656–667. [Google Scholar] [CrossRef]
- Fontboté, L. Ore Deposits in the Central Andes. Elements 2018, 14, 257–261. [Google Scholar] [CrossRef]
- Sillitoe, R.H.; Halls, C.; Grant, J.N. Porphyry tin deposits in Bolivia. Econ. Geol. 1975, 70, 913–927. [Google Scholar] [CrossRef]
- Turneaure, F.S. The Bolivian tin-silver province. Econ. Geol. 1971, 66, 215–225. [Google Scholar] [CrossRef]
- Cunningham, C.G.; Zartman, R.E.; McKee, E.H.; Rye, R.O.; Naeser, C.W.; Sanjinés, O.; Ericksen, G.E.; Tavera, F. The age and thermal history of Cerro Rico de Potosi, Bolivia. Miner. Deposita 1996, 31, 374–385. [Google Scholar] [CrossRef]
- Kempe, U.; Lehmann, B.; Wolf, D.; Rodionov, N.; Bombach, K.; Schwengfelder, U.; Dietrich, A. U-Pb SHRIMP geochronology of Th-poor, hydrothermal monazite: An example from the Llallagua tin-porphyry deposit. Geochim. Cosmochim. Ac. 2008, 72, 4352–4366. [Google Scholar] [CrossRef]
- Putzer, H. Metallogenetische Provinzen in Stidamerika. Schweizerbart; E. Schweizerbart’sche Verlagsbuchhandlung: Stuttgart, Germany, 1976; p. 316. [Google Scholar]
- Sugaki, A.; Hueno, H.; Shimada, N.; Kusachi, I.; Kitakaze, A.; Hayashi, K.; Kojima, S.Y.; Sanjines, O. Geological study on the polymetallic ore deposits in the Potosi District, Bolivia. Sci. Rep. Tohoku Univ. 1983, Series III. 15, 409–460. [Google Scholar]
- Ludington, S.; Orris, G.J.; Cox, D.P.; Long, K.R.; Asher-Bolinder, S. Mineral deposit models. In Geology and Mineral Resources of the Altiplano and Cordillera Occidental, Bolivia; U.S. Geological Survey and Servicio Geologico de Bolivia, Ed.; U.S. Geological Survey: Denver, CO, USA, 1975; pp. 63–89. [Google Scholar]
- SERGEOMIN, Mapa Geológico de Bolivia, Scale 1:1,000,000; Servicio Nacional de Geología y Minería and Yacimientos Petrolíferos Fiscales Bolivianos: La Paz, Bolivia, 2001.
- Díaz-Martínez, E.; Grahn, Y. Early Silurian glaciation along the western margin of Gondwana (Peru, Bolivia and northern Argentina): Palaeogeographic and geodynamic setting. Palaeogeogr. Palaeocl. 2007, 245, 62–81. [Google Scholar] [CrossRef]
- Fink, R.J. Sedimentology and stratigraphy of the Upper Cretaceous-Paleocene El Molino Formation, Eastern Cordillera and Altiplano, Central Andes, Bolivia: Implications for the tectonic development of the Central Andes. Master’s Thesis, LSU. 3925, Louisiana State University and Agricultural and Mechanical College, LA, USA, August 2002. [Google Scholar]
- Oen, I.S.; Kager, P.; Kieft, C. Oscillatory zoning of a discontinuous solid-solution series: Sphalerite-stannite. Am. Mineral. 1980, 65, 1220–1232. [Google Scholar]
- Sugaki, A.; Kojima, S.; Shimada, N. Fluid inclusion studies of the polymetallic hydrothermal ore deposits in Bolivia. Miner. Deposita 1988, 23, 9–15. [Google Scholar] [CrossRef]
- Müller, B.; Frischknechtm, R.; Seward, T.M.; Heinrich, C.A.; Camargo Gallegos, W. A fluid inclusion reconnaissance study of the Huanuni tin deposit (Bolivia), using LA-ICP-MS micro-analysis. Miner. Deposita 2001, 36, 680–688. [Google Scholar] [CrossRef]
- Kretschmar, U.; Scott, S.D. Phase relations involving arsenopyrite in the system Fe-As-S and their application. Can. Mineral. 1976, 14, 364–386. [Google Scholar]
- Sharp, Z.D.; Essene, E.J.; Kelly, W.C. A re-examination of the arsenopyrite geothermometer: Pressure considerations and applications to natural assemblages. Can. Mineral. 1985, 23, 517–534. [Google Scholar]
- Scott, S.D.; Barnes, H.L. Sphalerite geothermometry and geobarometry. Econ. Geol. 1971, 66, 653–669. [Google Scholar] [CrossRef]
- Hutchison, M.N.; Scott, S.D. Sphalerite geobarometry in the Cu-Fe-Zn-S system. Econ. Geol. 1981, 76, 143–153. [Google Scholar]
- Arnold, R.G. Equilibrium relations between pyrrhotite and pyrite from 325 and 743 ºC. Econ. Geol. 1962, 75, 72–90. [Google Scholar] [CrossRef]
- Toulmin, P.; Barton, P.B. A thermodynamic study of pyrite and pyrrhotite. Geochim. Cosmochim. Acta 1964, 28, 641–671. [Google Scholar] [CrossRef]
- Osadchii, E.G.; Chareev, D.A. Thermodynamic studies of pyrrhotite-pyrite equilibria in the Ag-Fe-S system by solid-state galvanic cell technique at 518–723 K and total pressure of 1 atm. Geochim. Cosmochim. Acta 2006, 70, 5617–5633. [Google Scholar] [CrossRef]
- Nakamura, Y.; Shima, H. Fe and Zn partitioning between sphalerite and stannite. In Proceedings of the Joint Meeting of Society of Mining Geologists of Japan; The Japanese Association of Mineralogists, Petrologists and Economic Geologists and the Mineralogical Society of Japan: Sendai, Japan, 1982. (In Japanese) [Google Scholar]
- Watanabe, M.; Hoshino, K.; Myint, K.K.; Miyazaki, K.; Nishido, H. Stannite from the Otoge kaolin-pyrophyllite deposits, Yamagata Prefecture, NE Japan and its genetical significance. Resour. Geol. 1994, 44, 439–444. [Google Scholar]
- Shimizu, M.; Shikazono, N. Iron and zinc partitioning between coexisting stannite and sphalerite: A possible indicator of temperature and sulfur fugacity. Mineral. Deposita 1985, 20, 314–320. [Google Scholar] [CrossRef]
- Sack, R.O.; Lynch, J.V.G.; Foit, F. Fahlore as a petrogenetic indicator: Keno Hill Ag-Pb-Zn district, Yukon, Canada. Mineral. Mag. 2003, 67, 1023–1038. [Google Scholar] [CrossRef]
- Ghosal, S.; Sack, R.O. As-Sb energetics in argentian sulfosalts. Geochim. Cosmochim. Acta 1995, 17, 3573–3579. [Google Scholar] [CrossRef]
- Einaudi, M.T.; Hedenquist, J.W.; Inan, E.E. Sulfidation state of fluids in active and extinct hydrothermal systems: Transitions from porphyry to epithermal environments. Soc. Econ. Geol. Spec. P. 2003, 10, 285–313. [Google Scholar]
- Camprubí, A.; Albinson, T. Epithermal deposits in México—An update of current knowledge, and an empirical reclassification. In Geology of México: Celebrating the Centenary of the Geological Society of México; Alaniz-Álvarez, S.A., Nieto-Samaniego, A.F., Eds.; Geological Society of America: Boulder, CO, USA, 2007; Volume 422, pp. 377–415. [Google Scholar]
- Sillitoe, R.H.; Hedenquist, J.W. Linkages between volcanotectonic settings, ore-fluid compositions, and epithermal precious metal deposits. Geol. Soc. Am. 2003, 10, 314–343. [Google Scholar]
- Cook, N.J.; Ciobanu, C.L.; Pring, A.; Skinner, W.; Shimizu, M.; Danyushevsky, L.; Saini-Eidukat, B.; Melcher, F. Trace and minor elements sphalerite: A LA-ICPMS study. Geochim. Cosmochim. Acta 2009, 73, 4761–4791. [Google Scholar] [CrossRef]
- Cook, N.J.; Ciobanu, C.; Williams, T. The mineralogy and mineral chemistry of indium in sulphide deposits and implications for mineral processing. Hydrometallurgy 2011, 108, 226–228. [Google Scholar] [CrossRef]
- Gaspar, O.C. Mineralogy and sulfide mineral chemistry of the Neves Corvo ores, Portugal: Insight into their genesis. Can. Mineral. 2002, 40, 611–636. [Google Scholar] [CrossRef]
- Serranti, S.; Ferrini, V.; Masi, U.; Cabri, L.J. Trace-element distribution in cassiterite and sulfides from rubané and massive ores of the Corvo deposit, Portugal. Can. Mineral. 2002, 40, 815–835. [Google Scholar] [CrossRef]
- Sinclair, W.D.; Kooiman, G.J.A.; Martin, D.A.; Kjarsgaard, I.M. Geology, geochemistry and mineralogy of indium resources at Mount Pleasant, New Brunswick, Canada. Ore Geol. Rev. 2006, 28, 123–145. [Google Scholar] [CrossRef]
- Ivanov, V.V.; Rodionov, D.A.; Tarkhov, Y.A. Character of the distribution and the average content of indium in some mineral from deposits of various genetic types. Gechemistry 1963, 11, 1056–1067. [Google Scholar]
- Parasyuk, O.V.; Voronyuk, S.V.; Gulay, L.D.; Davidyuk, G.Y.; Halka, V.O. Phase diagram in the of the CuInS-ZnS system and some physical properties of solid solutions phases. J. Alloy. Compd. 2003, 348, 57–64. [Google Scholar] [CrossRef]
- Schorr, S.; Wagner, G. Structure and phase relations of the Zn[2x](CuIn)[1 − x]S [2] solid solution series. J. Alloy. Compd. 2005, 396, 202–207. [Google Scholar] [CrossRef]
- Johan, Z. Indium and germanium in the structure of sphalerite: An example of coupled substitution with copper. Miner. Petrol. 1988, 39, 211–229. [Google Scholar] [CrossRef]
- Cook, N.J.; Ciobanu, C.L.; Brugger, J.; Etschmann, B.; Howard, D.L.; de Jonge, M.D.; Ryan, C.; Paterson, D. Determination of the oxidation state of Cu in substituted Cu-In-Fe-bearing sphalerite via μ- XANES spectroscopy. Am. Mineral. 2012, 97, 476–479. [Google Scholar] [CrossRef]
- Bente, K.; Doering, T. Solid-state diffusion in sphalerites: An experimental verification of the “chalcopyrite-disease”. Eur. J. Mineral. 1993, 5, 465–478. [Google Scholar] [CrossRef]
- Bente, K.; Doering, T. Experimental studies on the solid state diffusion of Cu + In in ZnS and on “Disease”, DIS (Diffusion Induced Degregations), in sphalerite and their geological applications. Miner. Petrol. 1995, 53, 285–305. [Google Scholar] [CrossRef]
- Seward, T.M.; Henderson, C.M.B.; Charnock, J.M. Indium(III) chloride complexing and solvatation in hydrothermal solutions to 350 ºC: An EXAFS study. Chem. Geol. 2000, 167, 117–127. [Google Scholar] [CrossRef]





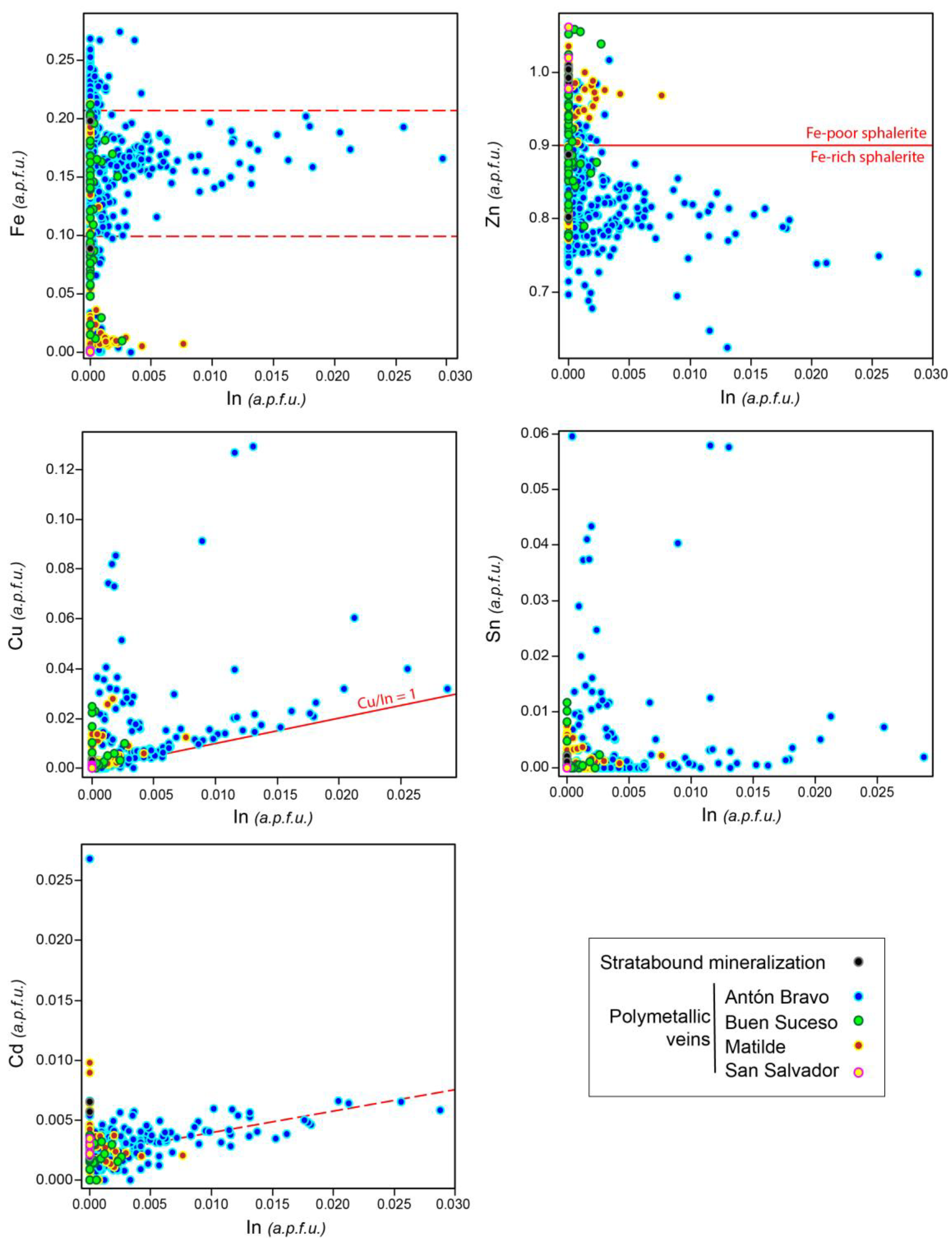

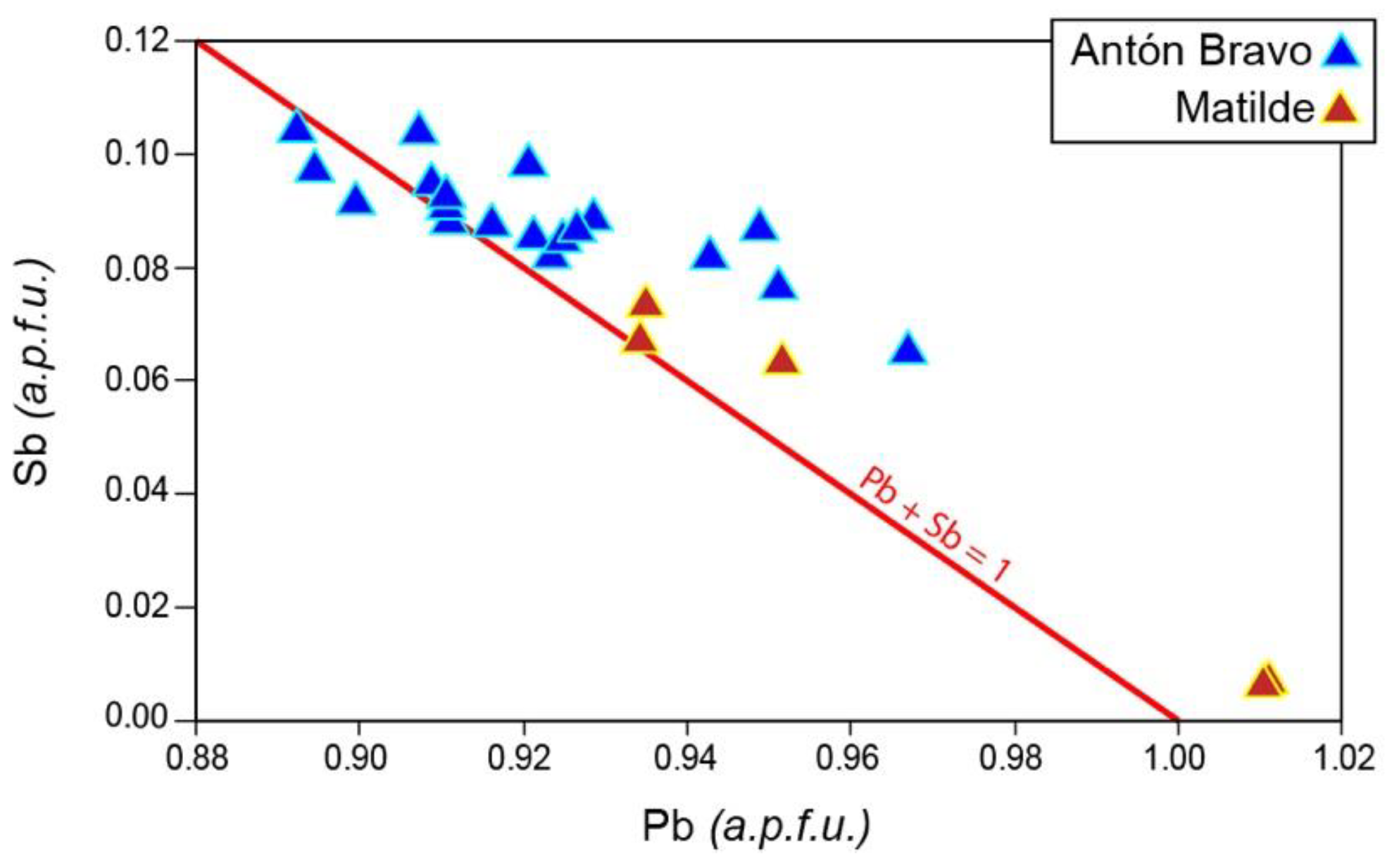
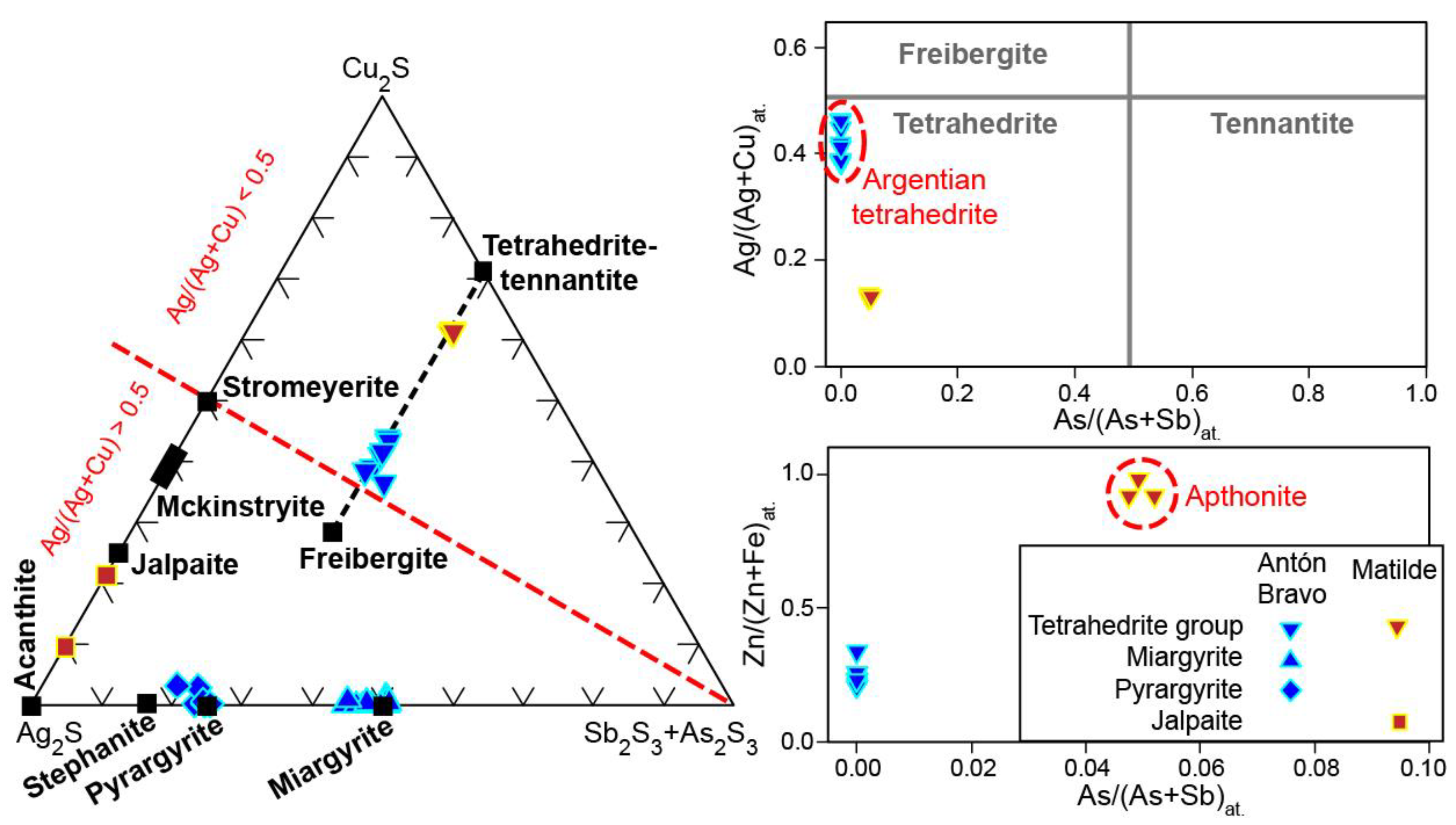
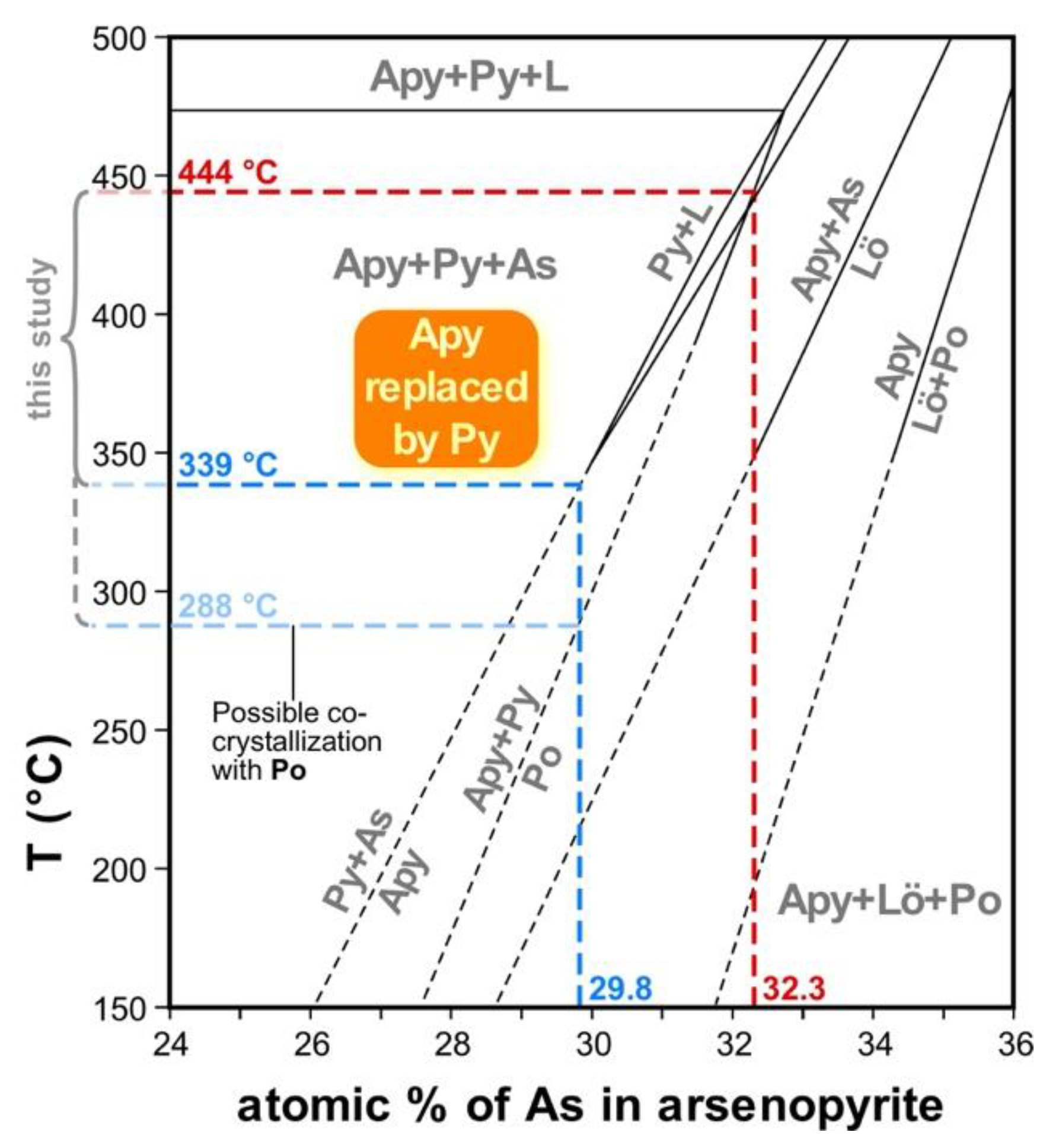
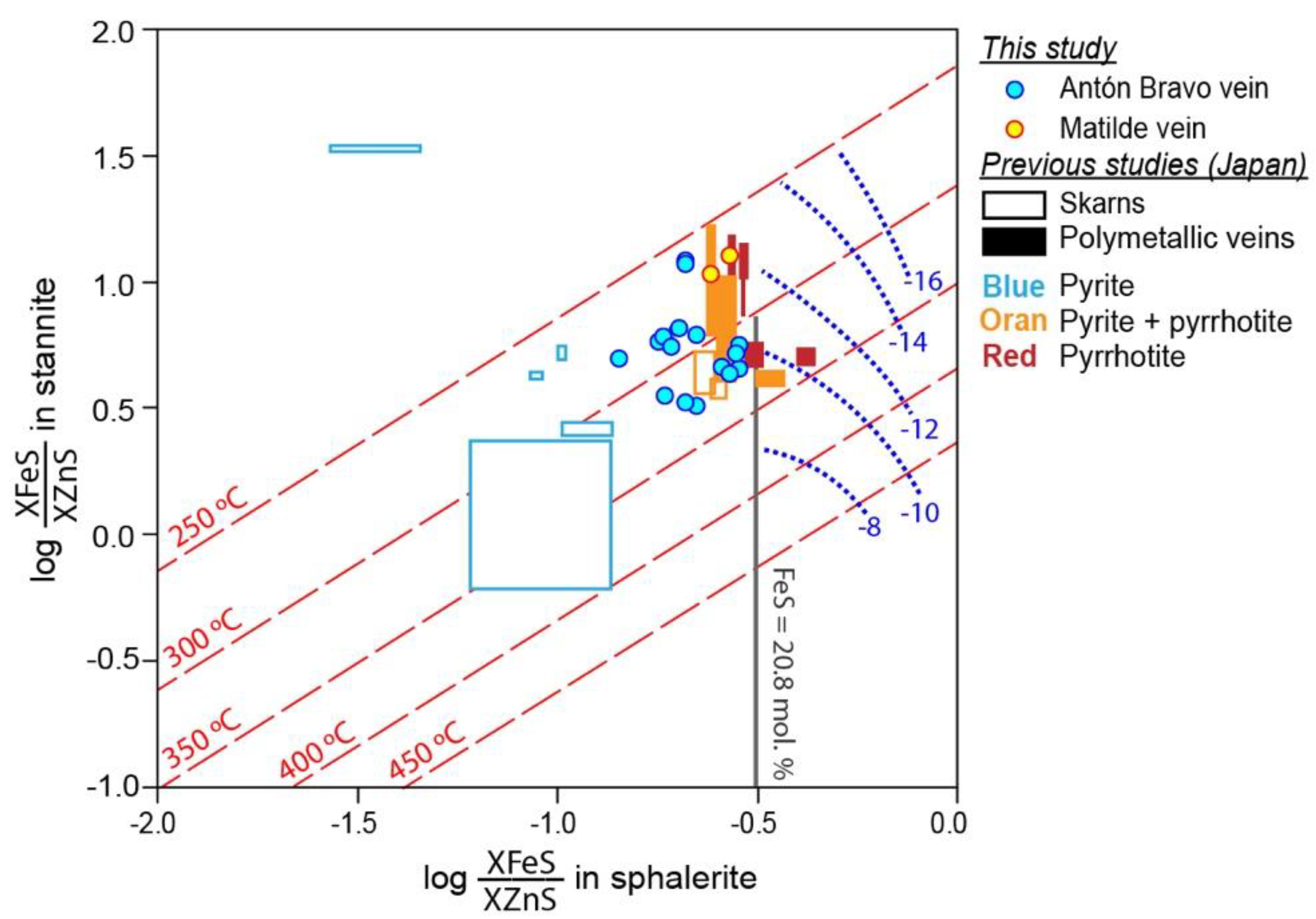
| Mineral | Arsenopyrite | Pyrrhotite | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Vein Group | Antón Bravo | Matilde | Antón Bravo | ||||||
| Sample | 1315 | 1315 | 1272 | 1275 | 1275 | 1328 | 1330 | 2018hh11 | 2018hh11 |
| Analysis | 19 | 20 | 114 | 182 | 189 | 118 | 122 | B-06 | B-07 |
| As (wt. %) | 42.05 | 43.31 | 44.35 | 42.80 | 42.69 | 0.14 | b.d.l. | b.d.l. | b.d.l. |
| S | 20.74 | 21.13 | 19.90 | 21.28 | 19.75 | 39.29 | 39.11 | 39.62 | 38.90 |
| Fe | 34.45 | 34.41 | 35.24 | 35.07 | 33.88 | 59.59 | 59.64 | 60.65 | 60.89 |
| Sb | 2.07 | 0.32 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Se | b.d.l. | b.d.l. | 0.30 | 0.21 | 0.88 | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Zn | b.d.l. | 0.10 | 0.04 | 1.50 | 1.71 | b.d.l. | b.d.l. | 0.43 | 0.10 |
| Cu | b.d.l. | b.d.l. | 0.02 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Pb | 0.09 | 0.05 | b.d.l. | 0.09 | 0.12 | b.d.l. | 0.11 | 0.11 | 0.13 |
| Bi | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.05 | 0.09 | b.d.l. | b.d.l. |
| In | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Total | 99.40 | 99.33 | 99.94 | 100.96 | 99.11 | 99.06 | 98.95 | 100.81 | 100.02 |
| As (at. %) | 30.46 | 31.12 | 32.01 | 30.24 | 31.11 | 0.08 | - | - | - |
| S | 35.11 | 35.48 | 33.56 | 35.14 | 33.64 | 53.41 | 53.30 | 53.06 | 52.62 |
| Fe | 33.48 | 33.17 | 34.12 | 33.24 | 33.13 | 46.50 | 46.66 | 46.63 | 47.29 |
| Sb | 0.92 | 0.14 | - | - | - | - | - | - | - |
| Se | - | - | 0.20 | 0.14 | 0.61 | - | - | - | - |
| Zn | - | 0.09 | 0.04 | 1.21 | 1.43 | - | - | 0.28 | 0.07 |
| Cu | - | - | 0.02 | - | - | - | - | - | - |
| Pb | 0.02 | 0.01 | - | 0.02 | 0.03 | - | 0.02 | 0.02 | 0.03 |
| Bi | - | - | - | - | - | 0.01 | 0.02 | - | - |
| In | - | - | - | - | - | - | - | - | - |
| Wt. % | S | Zn | Fe | Cu | Sn | Cd | Mn | Ag | In | Ge | Ga | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| d.l. | 0.01 | 0.03 | 0.02 | 0.02 | 0.04 | 0.08 | 0.02 | 0.06 | 0.03 | 0.02 | 0.03 | |
| Entire district (n = 560) | MIN | 30.43 | 41.66 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| MAX | 34.77 | 68.30 | 16.12 | 8.37 | 6.98 | 2.91 | 0.39 | 2.04 | 3.49 | 0.08 | 0.64 | |
| Av. | 33.12 | 56.56 | 8.21 | 0.36 | 0.22 | 0.28 | 0.02 | 0.04 | 0.17 | 0.00 | 0.07 | |
| Stratiform mineralization (n = 6) | MIN | 32.55 | 53.26 | b.d.l. | b.d.l. | b.d.l. | 0.23 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| MAX | 33.67 | 68.22 | 11.24 | 0.21 | 0.25 | 0.77 | 0.10 | 0.54 | b.d.l. | b.d.l. | 0.20 | |
| Av. | 33.12 | 63.97 | 2.81 | 0.08 | 0.08 | 0.42 | 0.02 | - | - | - | 0.06 | |
| Polymetallic veins (n = 554) | MIN | 30.43 | 41.66 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| MAX | 34.77 | 68.30 | 16.12 | 8.37 | 6.98 | 2.91 | 0.39 | 2.04 | 3.49 | 0.08 | 0.64 | |
| Av. | 33.12 | 57.49 | 8.26 | 0.36 | 0.22 | 0.28 | 0.02 | 0.04 | 0.17 | 0.00 | 0.07 | |
| Antón Bravo (n = 429) | MIN | 30.43 | 41.66 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| MAX | 34.77 | 65.83 | 16.12 | 8.37 | 6.98 | 2.91 | 0.08 | 2.04 | 3.49 | 0.08 | 0.63 | |
| Av. | 33.27 | 56.46 | 9.19 | 0.38 | 0.24 | 0.29 | 0.02 | 0.04 | 0.21 | 0.00 | 0.07 | |
| Buen Suceso (n = 60) | MIN | 30.45 | 52.00 | 0.54 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| MAX | 33.53 | 66.98 | 11.82 | 1.61 | 1.43 | 0.41 | 0.07 | 0.38 | 0.29 | 0.04 | 0.64 | |
| Av. | 32.22 | 59.74 | 6.37 | 0.20 | 0.12 | 0.24 | 0.02 | 0.04 | 0.03 | 0.00 | 0.06 | |
| Matilde (n = 62) | MIN | 32.13 | 52.39 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| MAX | 33.85 | 68.29 | 12.36 | 1.83 | 0.92 | 1.11 | 0.39 | 0.28 | 0.90 | 0.08 | 0.43 | |
| Av. | 32.95 | 61.91 | 4.06 | 0.35 | 0.20 | 0.28 | 0.04 | 0.04 | 0.07 | 0.01 | 0.08 | |
| San Salvador (n = 3) | MIN | 31.28 | 67.51 | 0.03 | b.d.l. | b.d.l. | 0.26 | b.d.l. | n.a. | b.d.l. | b.d.l. | b.d.l. |
| MAX | 33.86 | 68.30 | 0.08 | 0.08 | 0.03 | 0.38 | 0.04 | n.a. | b.d.l. | b.d.l. | 0.34 | |
| Av. | 32.66 | 67.83 | 0.05 | 0.04 | 0.02 | 0.33 | 0.02 | n.a. | - | - | 0.11 |
| Mineralization Style | Stratiform | Polymetallic Veins | ||||||||
| Vein Group | - | - | San Salvador | Matilde | Buen Suceso | |||||
| Sample | 1322 | 1321 | 1319 | 1329 | 1330 | 1278 | 1280 | 1282 | 1295 | 1329 |
| Analysis | 23 | d | h | 27 | 12 | e | d | b | sec | 27 |
| S (wt. %) | 33.34 | 32.94 | 31.28 | 33.31 | 32.38 | 33.49 | 32.57 | 32.76 | 33.28 | 33.31 |
| Zn | 60.35 | 66.63 | 67.68 | 53.62 | 66.89 | 53.12 | 58.23 | 59.18 | 63.76 | 53.62 |
| Fe | 5.16 | 0.17 | 0.04 | 12.36 | 0.04 | 11.80 | 8.55 | 7.00 | 2.79 | 12.36 |
| Cu | b.d.l. | 0.21 | b.d.l. | 0.17 | b.d.l. | 0.68 | 0.21 | b.d.l. | b.d.l. | 0.17 |
| Sn | 0.25 | b.d.l. | b.d.l. | 0.08 | b.d.l. | 0.60 | b.d.l. | 0.04 | b.d.l. | 0.08 |
| Cd | 0.27 | 0.27 | 0.38 | b.d.l. | 0.38 | 0.31 | 0.18 | 0.17 | 0.21 | b.d.l. |
| Ag | 0.54 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Mn | b.d.l. | 0.10 | b.d.l. | b.d.l. | b.d.l. | 0.03 | b.d.l. | 0.03 | b.d.l. | b.d.l. |
| In | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.27 | 0.04 | b.d.l. | b.d.l. |
| Ge | b.d.l. | b.d.l. | b.d.l. | 0.06 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.06 |
| Ga | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.64 | b.d.l. | b.d.l. |
| Total | 99.91 | 100.32 | 99.39 | 99.59 | 99.69 | 100.02 | 100.00 | 99.87 | 100.04 | 99.59 |
| S (a.p.f.u.) | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Zn | 0.89 | 0.99 | 1.06 | 0.79 | 1.01 | 0.78 | 0.88 | 0.89 | 0.94 | 0.79 |
| Fe | 0.09 | 0.00 | 0.00 | 0.21 | 0.00 | 0.20 | 0.15 | 0.12 | 0.05 | 0.21 |
| Cu | - | 0.00 | - | 0.00 | - | 0.01 | 0.00 | - | - | 0.00 |
| Sn | 0.00 | - | - | 0.00 | - | 0.00 | - | 0.00 | - | 0.00 |
| Cd | 0.00 | 0.00 | 0.00 | - | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | - |
| Ag | 0.00 | - | - | - | - | - | - | - | - | - |
| Mn | - | 0.00 | - | - | - | 0.00 | - | 0.00 | - | - |
| In | - | - | - | - | - | - | 0.00 | 0.00 | - | - |
| Ge | - | - | - | 0.00 | - | - | - | - | - | 0.00 |
| Ga | - | - | - | - | - | - | - | 0.01 | - | - |
| (Zn, Cd, and Mn)S (mol. %) | 90.7 | 99.4 | 99.9 | 78.5 | 99.9 | 78.2 | 84.9 | 87.8 | 95.1 | 78.5 |
| FeS | 9.1 | 0.3 | 0.1 | 21.1 | 0.1 | 19.8 | 14.6 | 12.1 | 4.9 | 21.1 |
| Cu2FeSnS4 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.5 | 0.0 | 0.0 | 0.0 | 0.1 |
| CuInS2 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 0.0 | 0.0 |
| Miner. Style | Polymetallic Vein | |||||||||
| Vein Group | Antón Bravo | |||||||||
| Sample | 1332 | 1334 | 2018-hh3A | 2018-hh3A | 2018-hh14 | 1289 | 1289 | 1289 | 1289 | 1331 |
| Analysis | 2 | a | A-47 | F-08 | A-04 | A-11 | C-01 | C-06 | C-10 | h |
| S (wt. %) | 32.75 | 32.73 | 32.86 | 32.65 | 33.84 | 33.45 | 33.12 | 33.58 | 33.63 | 32.26 |
| Zn | 59.50 | 57.07 | 49.56 | 49.89 | 50.06 | 47.51 | 65.83 | 49.89 | 50.73 | 59.70 |
| Fe | 7.10 | 7.83 | 9.93 | 10.98 | 9.77 | 15.63 | 1.09 | 15.61 | 15.17 | 7.73 |
| Cu | 0.06 | 0.73 | 3.93 | 2.58 | 2.14 | 1.07 | b.d.l. | 0.10 | b.d.l. | b.d.l. |
| Sn | 0.04 | b.d.l. | 1.12 | 0.88 | 0.23 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.14 |
| Cd | 0.28 | 0.35 | 0.74 | 0.75 | 0.70 | 0.32 | 0.21 | 0.42 | 0.40 | 0.12 |
| Ag | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Mn | 0.03 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| In | b.d.l. | 1.05 | 2.50 | 2.99 | 3.49 | b.d.l. | b.d.l. | 0.10 | b.d.l. | 0.09 |
| Ge | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Ga | 0.30 | 0.25 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.63 |
| Total | 100.06 | 100.02 | 100.64 | 100.72 | 100.30 | 98.05 | 100.38 | 99.73 | 100.13 | 100.66 |
| S (a.p.f.u.) | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Zn | 0.89 | 0.86 | 0.74 | 0.75 | 0.73 | 0.70 | 0.97 | 0.73 | 0.74 | 0.91 |
| Fe | 0.12 | 0.14 | 0.17 | 0.19 | 0.17 | 0.27 | 0.02 | 0.27 | 0.26 | 0.14 |
| Cu | 0.00 | 0.01 | 0.06 | 0.04 | 0.03 | 0.02 | - | 0.00 | - | - |
| Sn | 0.00 | - | 0.01 | 0.01 | 0.00 | - | - | - | - | 0.00 |
| Cd | 0.00 | 0.00 | 0.01 | 0.01 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Ag | - | - | - | - | - | - | - | - | - | - |
| Mn | 0.00 | - | - | - | - | - | - | - | - | - |
| In | - | 0.01 | 0.02 | 0.03 | 0.03 | - | - | 0.00 | - | 0.00 |
| Ge | - | - | - | - | - | - | - | - | - | - |
| Ga | 0.00 | 0.00 | - | - | - | - | - | - | - | 0.01 |
| (Zn, Cd, and Mn)S | 87.7 | 84.5 | 73.8 | 74.0 | 76.2 | 71.1 | 98.1 | 73.1 | 74.2 | 86.7 |
| FeS | 12.2 | 13.5 | 16.3 | 18.2 | 17.1 | 27.3 | 1.9 | 26.7 | 25.8 | 13.1 |
| Cu2FeSnS4 | 0.0 | 0.0 | 0.9 | 0.7 | 0.2 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| CuInS2 | 0.0 | 0.9 | 2.1 | 2.6 | 3.1 | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 |
| Vein Group | Matilde | B. Suceso | Antón Bravo | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sample | 1329 | 1329 | 1269 | 2018hh3A | 2018hh3A | 2018hh20 | 2018hh14 | 1289 | 1289 | 1289 |
| Analysis | 140 | 141 | e-38 | A-16 | E-04 | B-01 | A-09 | B-03 | B-04 | B-05 |
| S (wt. %) | 28.90 | 28.92 | 28.25 | 29.62 | 29.26 | 29.55 | 30.61 | 29.30 | 29.14 | 29.33 |
| Cu | 27.62 | 27.79 | 30.02 | 29.21 | 29.11 | 27.74 | 26.64 | 28.37 | 28.41 | 28.28 |
| Sn | 26.55 | 27.17 | 26.01 | 27.74 | 25.96 | 24.47 | 23.97 | 27.95 | 27.76 | 27.86 |
| Fe | 12.45 | 12.36 | 12.77 | 10.93 | 11.00 | 13.04 | 12.88 | 11.56 | 11.52 | 11.27 |
| Zn | 1.15 | 1.35 | b.d.l. | 2.30 | 2.08 | 3.30 | 4.53 | 2.39 | 2.60 | 3.04 |
| Ag | 2.08 | 1.47 | b.d.l. | 0.08 | 1.88 | 0.74 | 0.24 | 0.68 | 0.52 | 0.55 |
| In | 0.06 | b.d.l. | b.d.l. | 0.03 | 0.03 | 2.64 | 1.25 | 0.05 | 0.13 | 0.05 |
| Total | 98.81 | 99.06 | 97.22 | 99.01 | 99.32 | 101.48 | 100.12 | 100.30 | 100.08 | 100.38 |
| S (a.p.f.u.) 1 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 |
| Cu | 1.93 | 1.94 | 2.14 | 1.99 | 2.01 | 1.89 | 1.76 | 1.95 | 1.97 | 1.95 |
| Sn | 0.99 | 1.01 | 0.99 | 1.01 | 0.96 | 0.89 | 0.85 | 1.03 | 1.03 | 1.03 |
| Fe | 0.99 | 0.98 | 1.04 | 0.85 | 0.86 | 1.01 | 0.97 | 0.91 | 0.91 | 0.88 |
| Zn | 0.08 | 0.09 | - | 0.15 | 0.14 | 0.22 | 0.29 | 0.16 | 0.18 | 0.20 |
| Ag | 0.09 | 0.06 | - | 0.00 | 0.08 | 0.03 | 0.01 | 0.03 | 0.02 | 0.02 |
| In | 0.00 | - | - | 0.00 | 0.00 | 0.10 | 0.05 | 0.00 | 0.01 | 0.00 |
| Zn/(Fe + Zn)2 | 0.07 | 0.09 | 0.00 | 0.15 | 0.14 | 0.18 | 0.23 | 0.15 | 0.16 | 0.19 |
| Mineral | Galena | Tetrahedrite | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variety | Apthonite | Argentian Tetrahedrite | |||||||||
| Vein Group | Antón Bravo | Buen Suceso | Matilde | Antón Bravo | |||||||
| Sample | 1315 | 1274 | 1274 | 1270 | 1270 | 1328 | 1328 | 2018hh3 | 2018hh3 | 2018hh3 | 2018hh3 |
| Analysis | 8 | 96 | 97 | 67 | 73 | 30 | 31 | 65 | 66 | 86 | 97 |
| S (wt. %) | 13.75 | 12.84 | 12.85 | 12.63 | 12.93 | 23.78 | 23.75 | 22.43 | 22.25 | 21.63 | 22.02 |
| Sb | 4.82 | 0.85 | 2.62 | b.d.l. | b.d.l. | 27.89 | 27.79 | 27.22 | 29.17 | 26.46 | 27.51 |
| As | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.85 | 0.92 | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Pb | 80.93 | 84.39 | 82.49 | 84.72 | 78.97 | 0.09 | b.d.l. | 0.03 | b.d.l. | 0.05 | b.d.l. |
| Cu | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 32.52 | 32.00 | 20.03 | 17.51 | 18.90 | 20.47 |
| Ag | 0.16 | 0.35 | 0.91 | 0.79 | 1.94 | 8.13 | 8.17 | 24.46 | 25.57 | 27.48 | 24.33 |
| Zn | b.d.l. | b.d.l. | b.d.l. | 0.12 | 2.01 | 5.15 | 5.34 | 1.57 | 1.49 | 1.80 | 1.67 |
| Fe | 0.06 | 0.26 | 0.20 | 0.04 | 0.16 | 0.40 | 0.39 | 4.86 | 4.15 | 4.49 | 4.78 |
| Bi | 0.23 | 1.11 | 1.13 | 1.94 | 4.68 | 0.05 | b.d.l. | 0.07 | b.d.l. | 0.08 | b.d.l. |
| Sn | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| In | b.d.l. | 0.03 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Ge | 0.25 | 0.53 | 0.21 | b.d.l. | b.d.l. | b.d.l. | 0.03 | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Ga | 0.10 | 0.08 | 0.09 | 0.12 | 0.15 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Total | 100.30 | 100.44 | 100.50 | 100.55 | 101.01 | 98.98 | 98.52 | 100.75 | 100.27 | 100.94 | 100.86 |
| S (a.p.f.u.) 1 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 13.00 | 13.00 | 12.52 | 11.59 | 12.42 | 12.16 |
| Sb | 0.09 | 0.02 | 0.05 | - | - | 4.01 | 4.01 | 4.00 | 4.00 | 4.00 | 4.00 |
| As | - | - | - | - | - | 0.20 | 0.22 | - | - | - | - |
| Pb | 0.91 | 1.02 | 0.99 | 1.04 | 0.95 | 0.01 | - | 0.00 | - | 0.00 | - |
| Cu | - | - | - | - | - | 8.97 | 8.84 | 5.64 | 4.60 | 5.47 | 5.70 |
| Ag | 0.00 | 0.01 | 0.02 | 0.02 | 0.04 | 1.32 | 1.33 | 4.06 | 3.96 | 4.69 | 3.99 |
| Zn | - | - | - | 0.00 | 0.08 | 1.38 | 1.43 | 0.43 | 0.38 | 0.51 | 0.45 |
| Fe | 0.00 | 0.01 | 0.01 | 0.00 | 0.01 | 0.12 | 0.12 | 1.56 | 1.24 | 1.48 | 1.52 |
| Bi | 0.00 | 0.01 | 0.01 | 0.02 | 0.06 | 0.00 | - | 0.01 | - | 0.01 | - |
| Sn | - | - | - | - | - | - | - | - | - | - | - |
| In | - | 0.00 | - | - | - | - | - | - | - | - | - |
| Ge | 0.01 | 0.02 | 0.01 | - | - | - | 0.01 | - | - | - | - |
| Ga | 0.00 | 0.00 | 0.00 | 0.00 | 0.01 | - | - | - | - | - | - |
| As/(As+Sb) 2 | - | - | - | - | - | 0.05 | 0.05 | 0.00 | 0.00 | 0.00 | 0.00 |
| Ag/(Ag+Cu) 2 | - | - | - | - | - | 0.13 | 0.13 | 0.42 | 0.46 | 0.46 | 0.41 |
| Zn/(Zn+Fe) 2 | - | - | - | - | - | 0.92 | 0.92 | 0.22 | 0.23 | 0.26 | 0.23 |
| Mineral | Andorite | Diaphorite | Staročeskéite | Jalpaite | Miargyrite | Pyrargyrite | Franckeite | |||
| Vein Group | Antón Bravo | Antón Bravo | Antón Bravo | Matilde | Antón Bravo | Antón Bravo | Matilde | |||
| Sample | 1315 | 1334 | 2018hh3A | 1329 | 2018hh3A | 2018hh3A | 2018hh3A | 2018hh3A | 1330 | 1323 |
| Analysis | B ssa | 4 | A-38 | 15 | A-36 | A-37 | A-17 | B-41 | 26 | 2 |
| S (wt. %) | 21.48 | 19.11 | 19.89 | 13.50 | 21.45 | 21.61 | 17.42 | 17.47 | 19.57 | 20.27 |
| Sb | 40.25 | 26.21 | 29.69 | b.d.l. | 41.97 | 41.72 | 22.09 | 22.96 | 9.77 | 9.98 |
| As | 0.47 | 0.32 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.13 | 0.46 |
| Pb | 24.08 | 29.10 | 32.85 | 0.10 | 0.05 | 0.18 | 0.15 | 0.09 | 53.96 | 49.32 |
| Ag | 11.49 | 23.50 | 8.91 | 75.53 | 36.18 | 36.59 | 59.89 | 59.54 | 0.41 | 0.93 |
| Sn | 0.73 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 12.43 | 14.59 |
| Cu | 0.02 | 0.62 | 0.03 | 4.88 | 0.33 | 0.07 | 0.13 | 0.16 | b.d.l. | 0.02 |
| Zn | b.d.l. | 0.00 | 0.05 | 4.31 | 0.05 | b.d.l. | b.d.l. | 0.09 | b.d.l. | 1.89 |
| Fe | 0.35 | 0.58 | 0.33 | 0.10 | 0.07 | b.d.l. | b.d.l. | 0.03 | 2.52 | 2.61 |
| Cd | b.d.l. | b.d.l. | 0.23 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Bi | 0.12 | 0.16 | 5.84 | b.d.l. | 0.05 | 0.08 | 0.14 | 0.05 | 0.21 | 0.22 |
| In | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Ge | 0.09 | b.d.l. | b.d.l. | b.d.l. | 0.03 | b.d.l. | b.d.l. | b.d.l. | 0.10 | 0.08 |
| Ga | b.d.l. | 0.04 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.09 | b.d.l. |
| Total | 99.08 | 99.64 | 97.82 | 98.42 | 100.18 | 100.25 | 99.82 | 100.40 | 99.19 | 100.37 |
| S (a.p.f.u.) 1 | 6.00 | 8.00 | 6.00 | 2.00 | 2.00 | 2.00 | 3.00 | 3.00 | 14.00 | 14.00 |
| Sb | 2.96 | 2.89 | 2.36 | - | 1.03 | 1.02 | 1.00 | 1.04 | 1.84 | 1.81 |
| As | 0.06 | 0.06 | - | - | - | - | - | - | 0.04 | 0.14 |
| Pb | 1.04 | 1.88 | 1.53 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 5.97 | 5.27 |
| Ag | 0.95 | 2.92 | 0.80 | 3.33 | 1.00 | 1.01 | 3.07 | 3.04 | 0.09 | 0.19 |
| Sn | 0.06 | - | - | - | - | - | - | - | 2.40 | 2.72 |
| Cu | 0.00 | 0.13 | 0.00 | 0.36 | 0.02 | 0.00 | 0.01 | 0.01 | - | 0.01 |
| Zn | - | - | 0.01 | 0.31 | 0.00 | - | - | 0.01 | - | 0.64 |
| Fe | 0.06 | 0.14 | 0.06 | 0.01 | 0.00 | - | - | 0.00 | 1.03 | 1.03 |
| Cd | - | - | 0.02 | - | - | - | - | - | - | - |
| Bi | 0.01 | 0.01 | 0.27 | - | 0.00 | 0.00 | 0.00 | 0.00 | 0.02 | 0.02 |
| In | - | - | - | - | - | - | - | - | - | - |
| Ge | 0.01 | - | - | - | 0.00 | - | - | - | 0.03 | 0.02 |
| Ga | - | 0.01 | - | - | - | - | - | - | 0.03 | - |
| Mineral | Semseyite | Jamesonite | ||||||||
| Vein Group | Antón Bravo | Antón Bravo | ||||||||
| Sample | 1315 | 1315 | 1315 | 1315 | 1315 | 1315 | 1315 | 1315 | 1315 | 1334 |
| Analysis | 13 | 14 | 15 | 16 | b | 2 | 3 | 11 | 13b | 16b |
| S (wt. %) | 18.97 | 18.82 | 20.82 | 21.11 | 20.88 | 20.96 | 20.84 | 20.86 | 20.89 | 21.12 |
| Sb | 31.08 | 29.47 | 34.30 | 34.54 | 34.61 | 34.76 | 34.67 | 34.62 | 34.56 | 34.44 |
| As | 0.32 | 0.30 | 0.46 | 0.29 | 0.39 | 0.35 | 0.45 | 0.36 | 0.51 | 0.48 |
| Pb | 48.78 | 50.80 | 41.15 | 40.95 | 40.88 | 40.59 | 40.90 | 40.25 | 41.00 | 40.89 |
| Ag | b.d.l. | 0.18 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Sn | 0.20 | 0.15 | 0.26 | 0.22 | 0.20 | 0.26 | 0.28 | 0.40 | 0.27 | 0.27 |
| Cu | b.d.l. | b.d.l. | b.d.l. | b.d.l. | 0.02 | 0.03 | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Zn | b.d.l. | 0.04 | b.d.l. | b.d.l. | 0.06 | 0.20 | 0.19 | 0.48 | b.d.l. | b.d.l. |
| Fe | 0.04 | 0.03 | 2.57 | 2.55 | 2.53 | 2.62 | 2.62 | 2.54 | 2.58 | 2.55 |
| Cd | b.d.l. | b.d.l. | b.d.l. | 0.17 | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Bi | 0.14 | 0.19 | 0.05 | 0.12 | 0.11 | 0.11 | 0.04 | 0.15 | 0.11 | 0.08 |
| In | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. | b.d.l. |
| Ge | 0.10 | 0.65 | 0.54 | 0.12 | 0.08 | 0.08 | 0.17 | 0.10 | 0.14 | 0.07 |
| Ga | 0.04 | 0.10 | 0.06 | 0.04 | 0.05 | 0.05 | 0.04 | b.d.l. | b.d.l. | 0.06 |
| Total | 99.68 | 100.73 | 100.21 | 100.11 | 99.82 | 100.01 | 100.23 | 99.76 | 100.07 | 99.95 |
| S (a.p.f.u.) 1 | 21.00 | 21.00 | 14.00 | 14.00 | 14.00 | 14.00 | 14.00 | 14.00 | 14.00 | 14.00 |
| Sb | 9.06 | 8.66 | 6.07 | 6.03 | 6.11 | 6.11 | 6.13 | 6.12 | 6.10 | 6.01 |
| As | 0.15 | 0.15 | 0.13 | 0.08 | 0.11 | 0.10 | 0.13 | 0.10 | 0.15 | 0.14 |
| Pb | 8.36 | 8.77 | 4.28 | 4.20 | 4.24 | 4.19 | 4.25 | 4.18 | 4.25 | 4.19 |
| Ag | - | 0.06 | - | - | - | - | - | - | - | - |
| Sn | 0.06 | 0.05 | 0.05 | 0.04 | 0.04 | 0.05 | 0.05 | 0.07 | 0.05 | 0.05 |
| Cu | - | - | - | - | 0.01 | 0.01 | - | - | - | - |
| Zn | - | 0.02 | - | - | 0.02 | 0.06 | 0.06 | 0.16 | - | - |
| Fe | 0.03 | 0.02 | 0.99 | 0.97 | 0.97 | 1.00 | 1.01 | 0.98 | 0.99 | 0.97 |
| Cd | - | - | - | 0.03 | - | - | - | - | - | - |
| Bi | 0.02 | 0.03 | 0.00 | 0.01 | 0.01 | 0.01 | 0.00 | 0.02 | 0.01 | 0.01 |
| In | - | - | - | - | - | - | - | - | - | - |
| Ge | 0.05 | 0.32 | 0.16 | 0.03 | 0.03 | 0.02 | 0.05 | 0.03 | 0.04 | 0.02 |
| Ga | 0.02 | 0.05 | 0.02 | 0.01 | 0.02 | 0.01 | 0.01 | - | - | 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torró, L.; Melgarejo, J.C.; Gemmrich, L.; Mollinedo, D.; Cazorla, M.; Martínez, Á.; Pujol-Solà, N.; Farré-de-Pablo, J.; Camprubí, A.; Artiaga, D.; et al. Spatial and Temporal Controls on the Distribution of Indium in Xenothermal Vein-Deposits: The Huari Huari District, Potosí, Bolivia. Minerals 2019, 9, 304. https://doi.org/10.3390/min9050304
Torró L, Melgarejo JC, Gemmrich L, Mollinedo D, Cazorla M, Martínez Á, Pujol-Solà N, Farré-de-Pablo J, Camprubí A, Artiaga D, et al. Spatial and Temporal Controls on the Distribution of Indium in Xenothermal Vein-Deposits: The Huari Huari District, Potosí, Bolivia. Minerals. 2019; 9(5):304. https://doi.org/10.3390/min9050304
Chicago/Turabian StyleTorró, Lisard, Joan Carles Melgarejo, Laura Gemmrich, Diva Mollinedo, Malena Cazorla, Álvaro Martínez, Núria Pujol-Solà, Júlia Farré-de-Pablo, Antoni Camprubí, David Artiaga, and et al. 2019. "Spatial and Temporal Controls on the Distribution of Indium in Xenothermal Vein-Deposits: The Huari Huari District, Potosí, Bolivia" Minerals 9, no. 5: 304. https://doi.org/10.3390/min9050304
APA StyleTorró, L., Melgarejo, J. C., Gemmrich, L., Mollinedo, D., Cazorla, M., Martínez, Á., Pujol-Solà, N., Farré-de-Pablo, J., Camprubí, A., Artiaga, D., Torres, B., Alfonso, P., & Arce, O. (2019). Spatial and Temporal Controls on the Distribution of Indium in Xenothermal Vein-Deposits: The Huari Huari District, Potosí, Bolivia. Minerals, 9(5), 304. https://doi.org/10.3390/min9050304







