Pyrometamorphic Rocks in the Molinicos Basin (Betic Cordillera, SE Spain): Insights into the Generation of Cordierite Paralavas
Abstract
1. Introduction
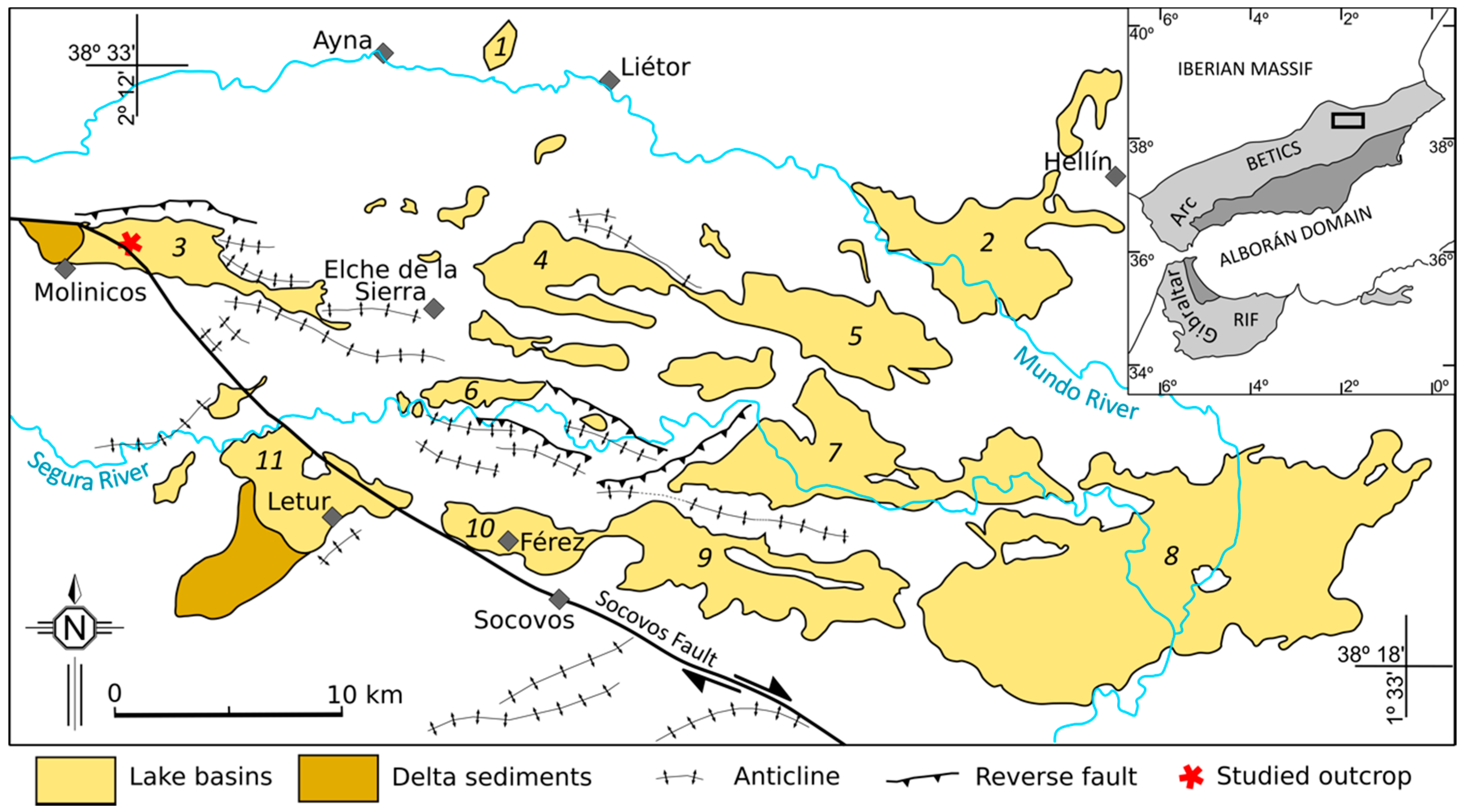
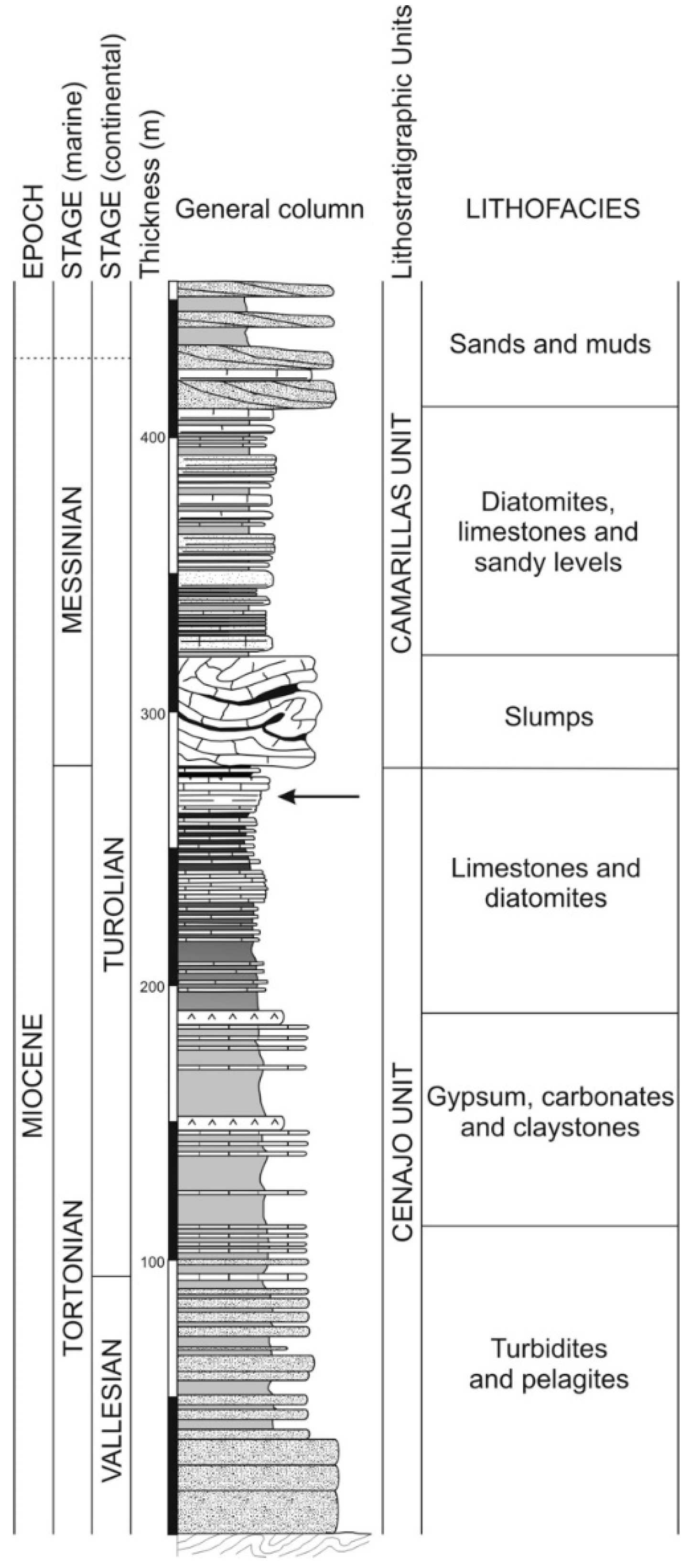
2. Geological Setting
3. Materials and Methods
4. Results
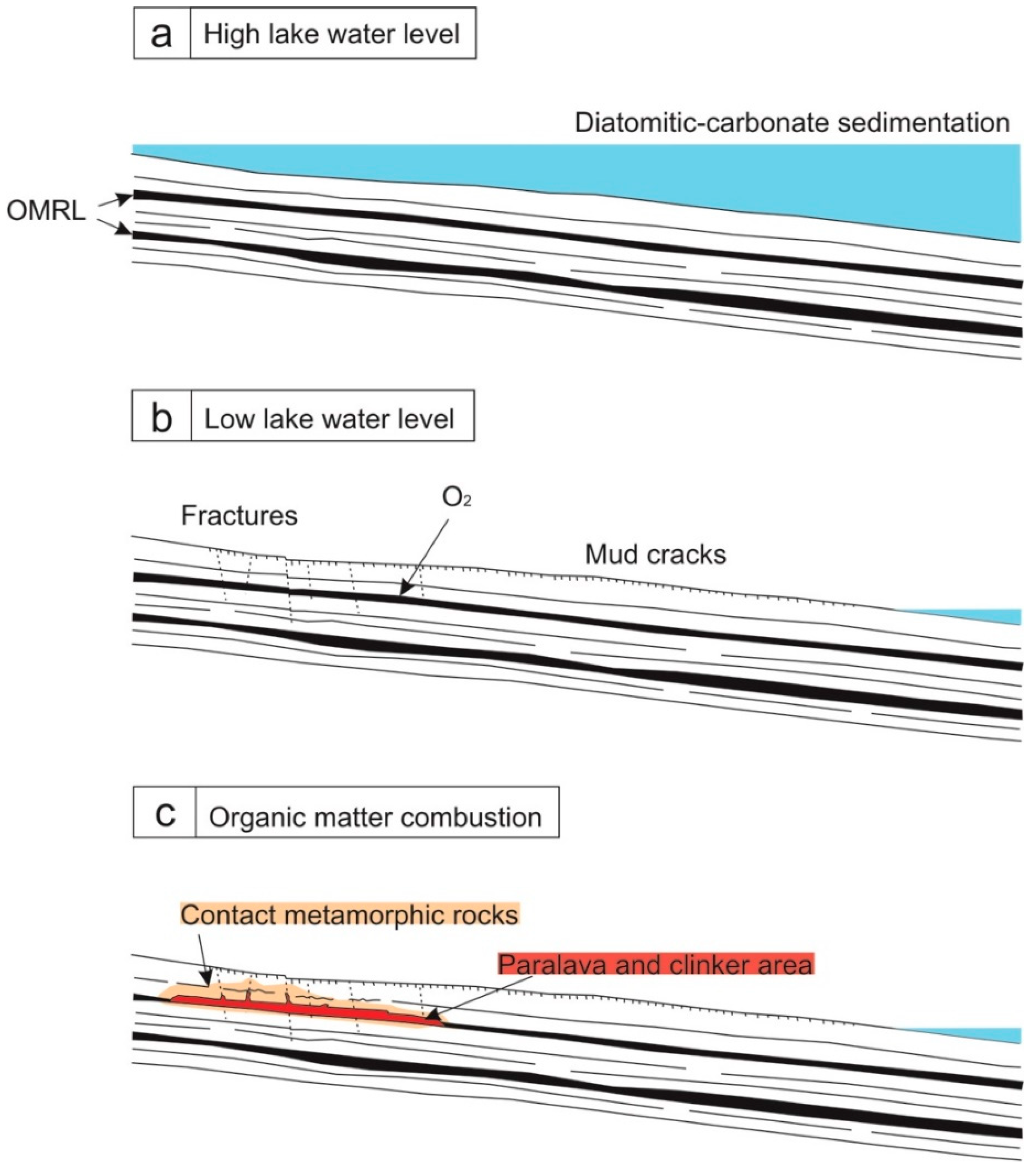
4.1. Field Relationships
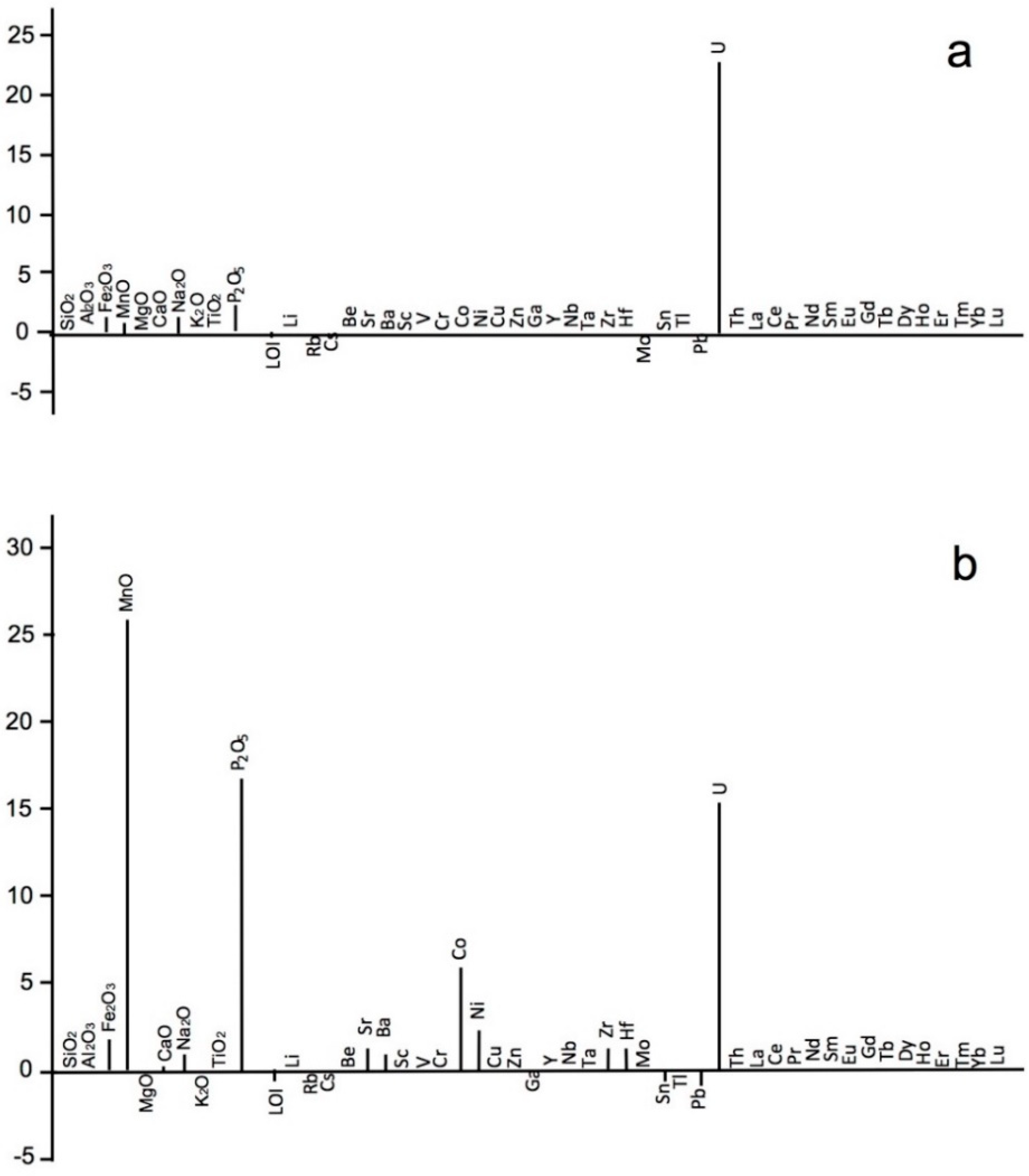
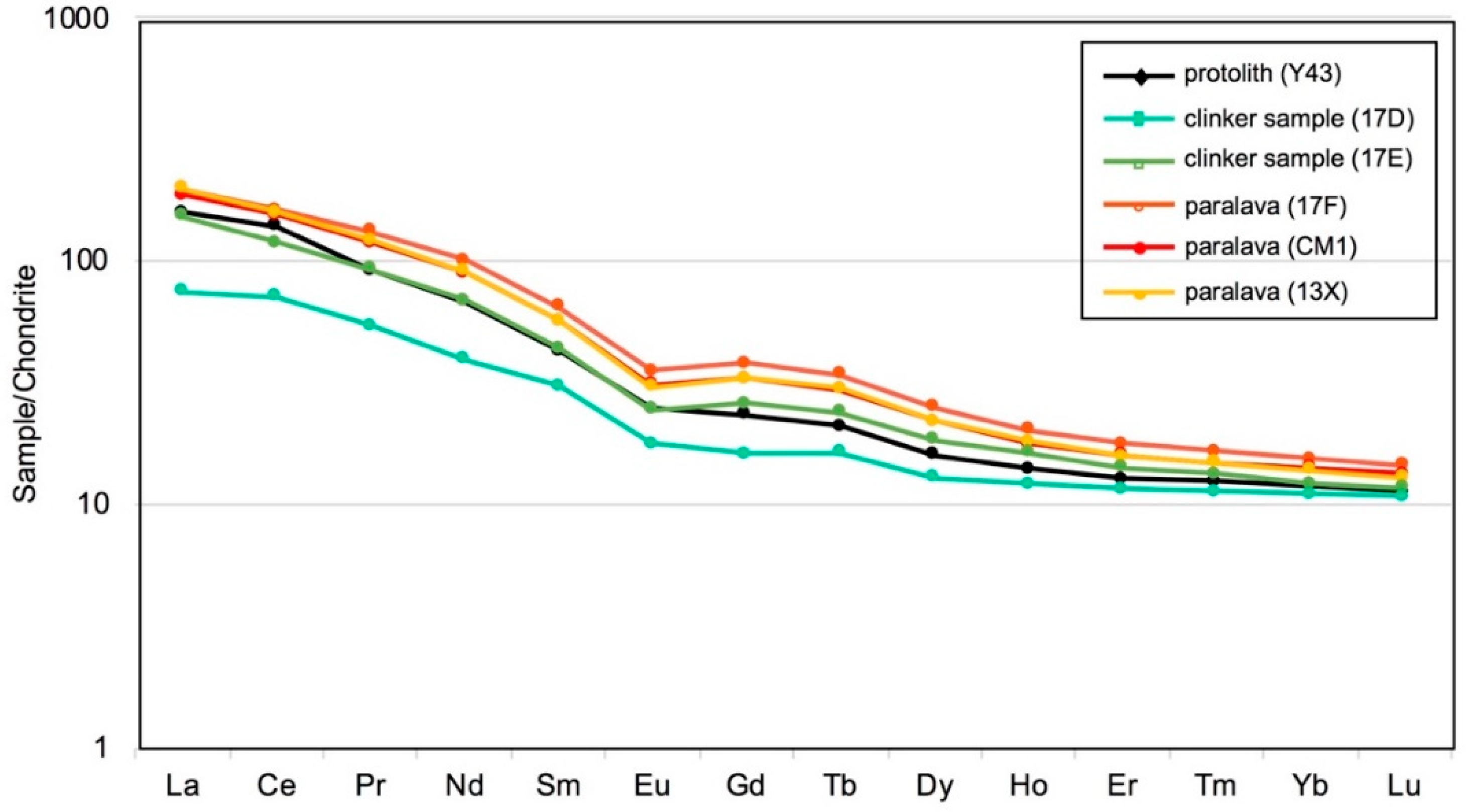
4.2. Bulk-Rock Chemical Composition and Total Organic Carbon (TOC) Contents
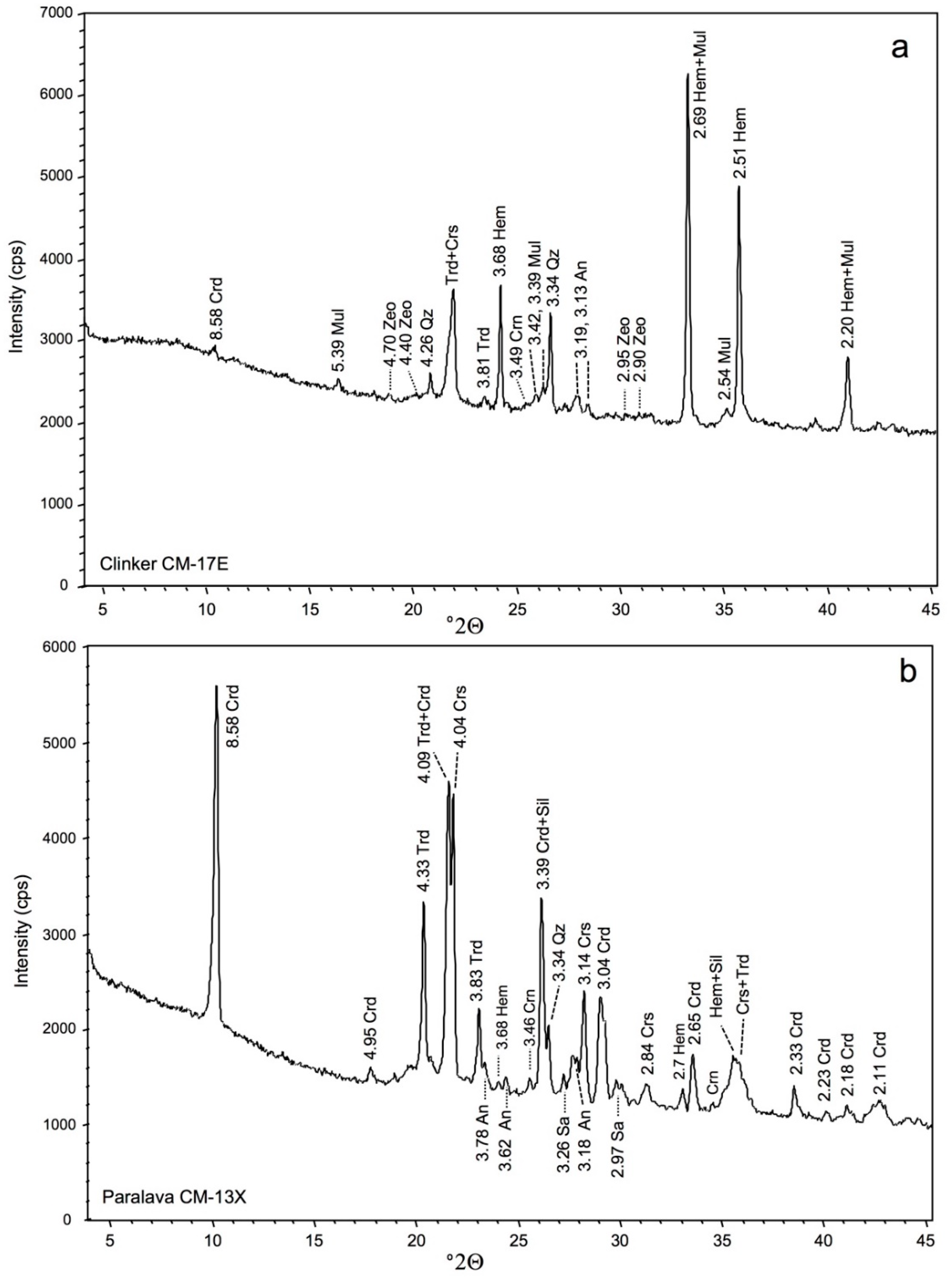
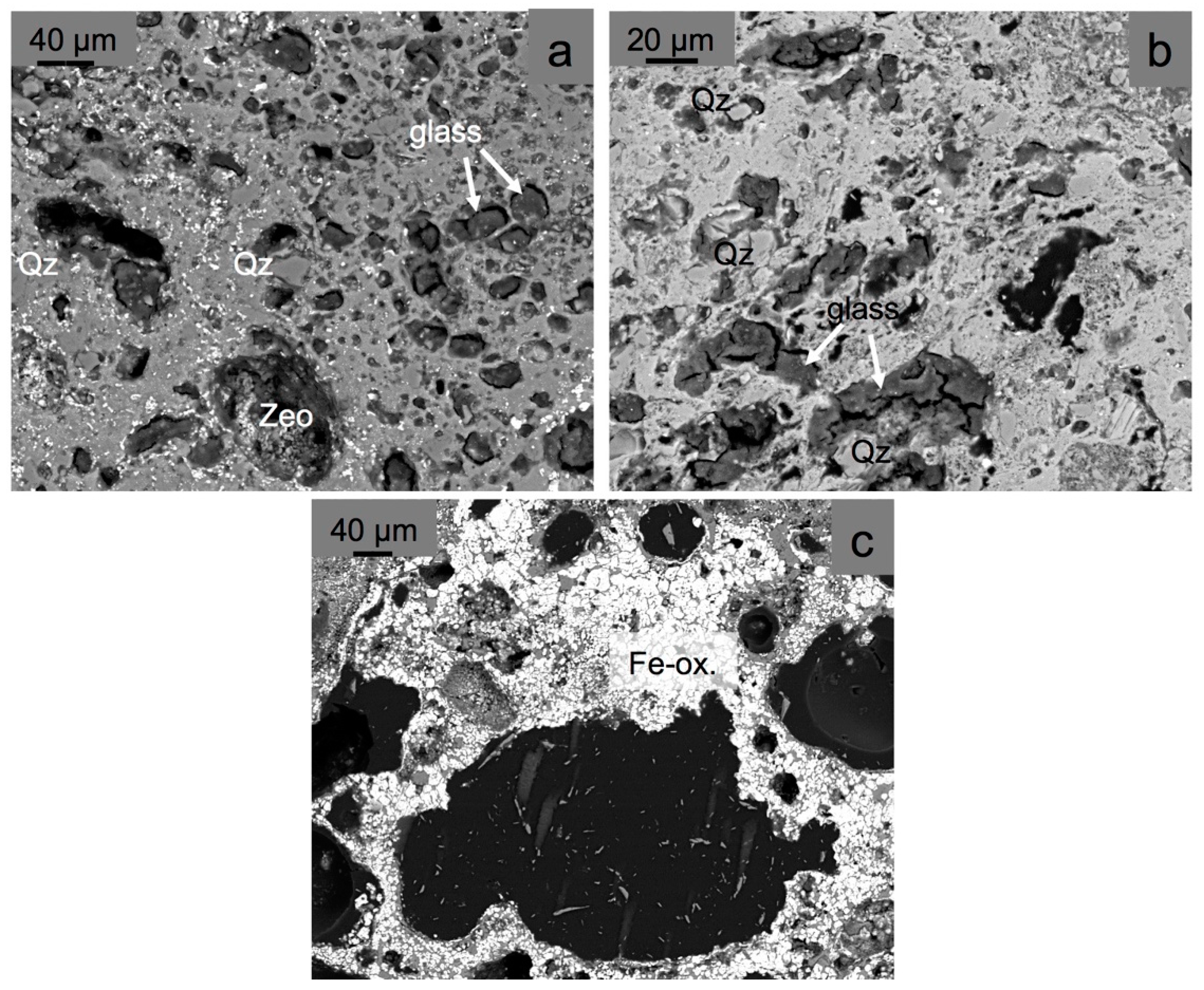
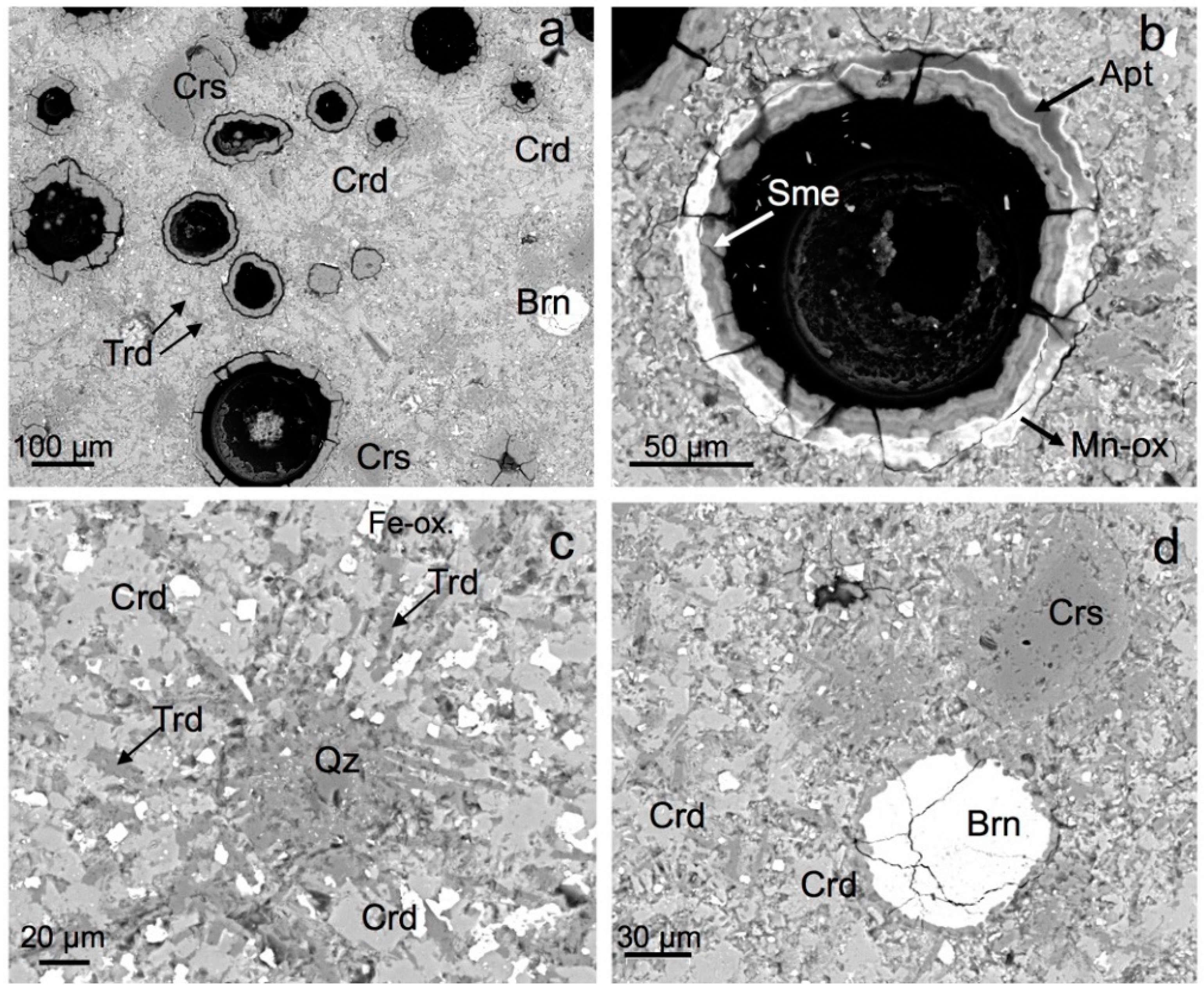
4.3. Mineralogy (XRD Data and SEM Characterization)
5. Discussion
5.1. Origin of Pyrometamorphism
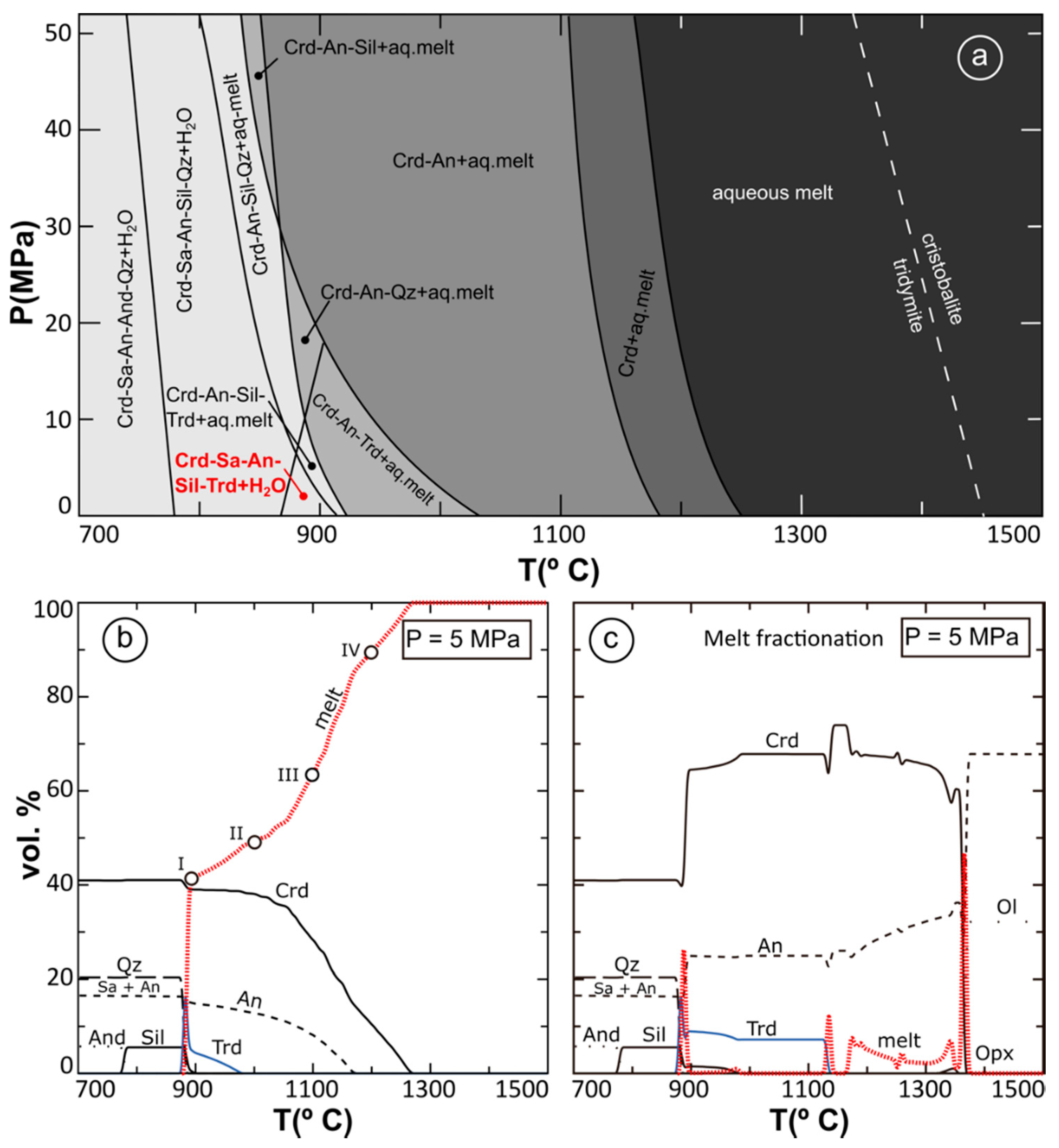
5.2. Pressure and Temperature Conditions of Pyrometamorphism
5.3. Tridymite and Cristobalite Stability
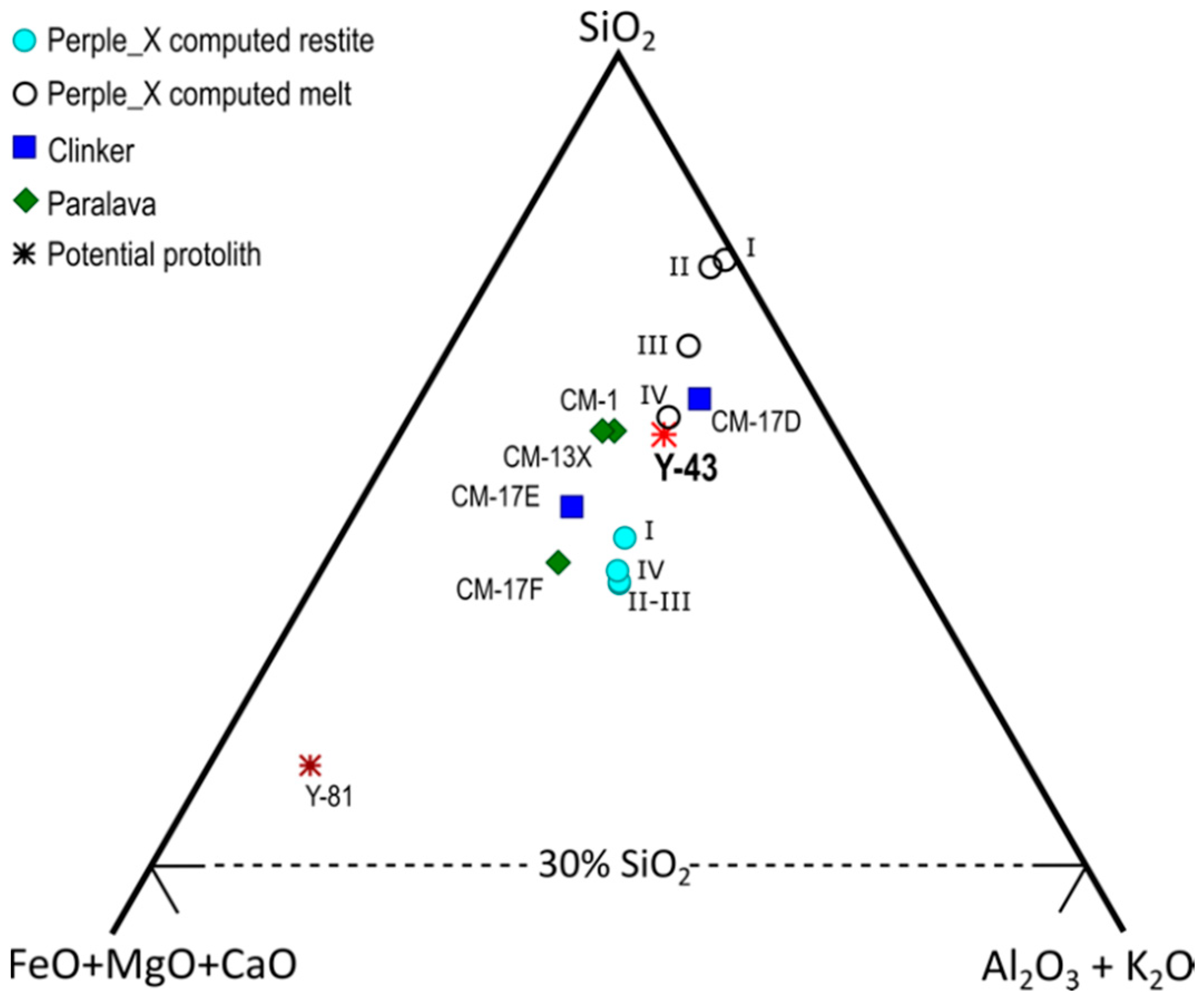
5.4. Clinker and Paralava Composition: the Role of Melting Conditions and Fractionation
5.5. Changes in Effective Bulk Composition and Oxygen Fugacity
5.6. Cooling Evolution
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grapes, R. Pyrometamorphism; Springer: Berlin, Germany, 2011; p. 365. [Google Scholar]
- Sokol, E.V.; Volkova, N.I. Combustion metamorphic events resulting from natural coal fires. In GSA Reviews in Engineering Geology XVIII: Geology of Coal Fires: Case Studies from Around the World; Stracher, G.B., Ed.; Geological Society of America: Boulder, GO, USA, 2007; pp. 97–115. [Google Scholar]
- Moreno, L.; Jiménez, M.E.; Aguilera, H.; Jiménez, P.; de la Losa, A. The 2009 Smouldering Peat Fire in Las Tablas de Daimiel National Park (Spain). Fire Technol. 2011, 47, 519–538. [Google Scholar] [CrossRef]
- Rein, G. Smoldering-Peat Megafires: The Largest Fires on Earth. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 4, pp. 1–11. [Google Scholar] [CrossRef]
- Prat-Guitart, N.; Nugent, C.; Mullen, E.; Mitchell, F.J.G.; Hawthorne, D.; Belcher, C.M.; Yearsley, J.M. History of irish peatland fires. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; Volume 5, pp. 452–482. [Google Scholar] [CrossRef]
- Akulov, N.I.; Akulova, V.V.; Khudogonova, E.V. Pyrogenic metamorphism of the carbonaceous rocks in the south of the Siberian platform. In Coal Combustion Research; Grace, C.T., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2011; pp. 219–234. [Google Scholar]
- Kim, A.G. Coal Formation and the Origin of Coal Fires. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier: Amsterdam, The Netherlands, 2011; Volume 1, pp. 1–28. [Google Scholar] [CrossRef]
- Day, S.; Bainbridge, N.; Carras, J.; Lilley, W.; Roberts, C.; Saghafi, A.; Williams, D. Spontaneous Combustion in Open-Cut Coal Mines: Australian Experience and Research. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 2–36. [Google Scholar] [CrossRef]
- Nolter, M.A.; Aurand, H.W.; Vice, D.H. Anthracite Coal-Mine Fires of Northeastern Pennsylvania. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 652–665. [Google Scholar] [CrossRef]
- Novikova, S.A.; Sokol, E.V.; Novikov, I.S.; Travin, A.V. Ancient Coal Fires on the Southwestern Periphery of the Kuznetsk Basin, West Siberia, Russia: Geology and Geochronology. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 510–541. [Google Scholar] [CrossRef]
- Vice, D.H.; Stracher, G.B.; Eckert, A. Coal Fires of the Pacific Northwest, USA. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 671–680. [Google Scholar] [CrossRef]
- Sharygin, V.V.; Sokol, E.V.; Belakovsky, D.I. Mineralogy and Origin of Fayalite-Sekaninaite Paralava: Ravat Coal Fire, Central Tajikistan. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 582–607. [Google Scholar] [CrossRef]
- Ciesielczuk, J. Coal Mining and Combustion in the Coal Waste Dumps of Poland. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 464–473. [Google Scholar] [CrossRef]
- Misz-Kennan, M.; Tabor, A. The Thermal History of Select Coal-Waste Dumps in the Upper Silesian Coal Basin, Poland. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 432–462. [Google Scholar] [CrossRef]
- Ribeiro, J.; Ania, C.O.; Suárez-Ruiz, I.; Flores, D. The spontaneous combustion of coal-mine waste and stream effects in the El Bierzo Coalfield, Spain. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2019; Volume 5, pp. 98–124. [Google Scholar] [CrossRef]
- Hovland, M.; Hill, A.; Stokes, D. The structure and geomorphology of the Dashgil mud volcano, Azerbaijan. Geomorphology 1997, 21, 1–15. [Google Scholar] [CrossRef]
- Sokol, E.; Novikov, I.; Zateeva, S.; Vapnik, Y.; Shagam, R.; Kozmenko, O. Combustion metamorphism in the Nabi Musa dome: New implications for a mud volcanic origin of the Mottled Zone, Dead Sea area. Basin Res. 2010, 22, 414–438. [Google Scholar] [CrossRef]
- Vapnik, Y.; Khesin, B.; Itkis, S. Geophysical Studies of Pyrometamorphic and Hydrothermal Rocks of the Nabi Musa Mottled Zone, Vicinity of the Dead Sea Transform, Israel. In Coal and Peat Fires: A Global Perspective; Stracher, G.B., Prakash, A., Sokol, E.V., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 3, pp. 318–337. [Google Scholar] [CrossRef]
- McLintock, W.F.P. On the metamorphism produced by the combustion of hydrocarbons in the Tertiary sediments of south-west Persia. Mineral. Mag. 1932, 23, 207–227. [Google Scholar] [CrossRef]
- Levorsen, A.I. Geology of Petroleum; W.H. Freeman: San Francisco, CA, USA, 1954; p. 703. [Google Scholar]
- Basi, M.A.; Jassim, S.Z. Baked and fused Miocene sediments from the Injana area, Hemrin South, Iraq. J. Geol. Soc. Iraq 1974, 7, 1–14. [Google Scholar]
- Bentor, Y.K.; Kastner, M.; Perlman, I.; Yelin, Y. Combustion metamorphism of bituminous sediments and the formation of melts of granitic and sedimentary composition. Geochim. Cosmochim. Acta 1981, 45, 2229–2255. [Google Scholar] [CrossRef]
- Mathews, W.H.; Bustin, R.M. Why do the Smoking Hills smoke? Can. J. Earth Sci. 1984, 21, 737–742. [Google Scholar] [CrossRef]
- Cosca, M.A.; Essene, E.J.; Geissman, J.W.; Simmons, W.B.; Coates, D.A. Pyrometamorphic rocks associated with naturally burned coal beds, Powder River Basin, Wyoming. Am. Mineral. 1989, 74, 85–100. [Google Scholar]
- Eichhubl, P.; Aydin, A.; Lore, J. Opening-mode fracture in siliceous mudstone at high homologous temperature-effect of surface forces. Geophys. Res. Lett. 2001, 28, 1299–1302. [Google Scholar] [CrossRef]
- Lore, J.S.; Eichhubl, P.; Aydin, A. Alteration and fracturing of siliceous mudstone during in situ combustion, Orcutt field, California. J. Petrol. Sci. Eng. 2002, 36, 169–182. [Google Scholar] [CrossRef]
- Eichhubl, P.; Aydin, A. Ductile opening-mode fracture by pore growth and coalescence during combustion alteration of siliceous mudstone. J. Struct. Geol. 2003, 25, 121–134. [Google Scholar] [CrossRef]
- Planke, S.; Svensen, H.; Hovland, M.; Banks, D.A.; Jamtveit, B. Mud and fluid migration in active mud volcanoes in Azerbaijan. Geo-Mar. Lett. 2003, 23, 258–268. [Google Scholar] [CrossRef]
- Svensen, H.; Dysthe, D.G.; Bandlien, E.H.; Sacko, S.; Coulibaly, H.; Planke, S. Subsurface combustion in Mali: Refutation of the active volcanism hypothesis in West Africa. Geology 2003, 31, 581–584. [Google Scholar] [CrossRef]
- Geller, Y.I.; Burg, A.; Halicz, L.; Kolodny, L. System closure during the combustion metamorphic “Mottled Zone” event, Israel. Chem. Geol. 2012, 334, 25–36. [Google Scholar] [CrossRef]
- Clark, B.H.; Peacor, D.R. Pyrometamorphism and partial melting of shales during combustion metamorphism: Mineralogical, textural, and chemical effects. Contrib. Mineral. Petrol. 1992, 112, 558–568. [Google Scholar] [CrossRef]
- Sokol, E.; Volkova, N.; Lepezin, G. Mineralogy of pyrometamorphic rocks associated with naturally burned coal-bearing spoilheaps of the Chelyabinsk coal basin, Russia. Eur. J. Mineral. 1998, 10, 1003–1014. [Google Scholar] [CrossRef]
- Grapes, R.; Zhang, K.; Peng, Z.I. Paralava and clinker products of coal combustion, Yellow River, Shanxi Province, China. Lithos 2009, 113, 831–843. [Google Scholar] [CrossRef]
- Grapes, R.; Korzhova, S.; Sokol, E.; Seryotkin, Y. Paragenesis of unusual Fe-cordierite (sekaninaite)-bearing paralava and clinker from the Kuznetsk coal basin, Siberia, Russia. Contrib. Mineral. Petrol. 2011, 162, 253–273. [Google Scholar] [CrossRef]
- Cosca, M.A.; Essene, J. Paralava chemistry and conditions of formation, Powder River Basin, Wyoming (abstr.). EOS 1985, 66, 396. [Google Scholar]
- Calvo, J.P.; Rodríguez-Pascua, M.A.; Gómez-Gras, D. Sedimentary features indicative of seismogenetic tectonic activity. Las Minas and Cenajo Neogene basins (External Prebetic Zone, SE Spain). Rev. Soc. Geol. España 2014, 27, 205–221. [Google Scholar]
- Elizaga, E. Análisis de facies sedimentarias y petrología de los depósitos lacustres de edad Neógeno superior de la Zona Prebética, Albacete, España; Instituto Estudios Albacetenses Serie I Estudios: Albacete, Spain, 1994; Volume 74, p. 216. [Google Scholar]
- Ortí, F.; Rosell, L.; Gibert, L.; Moragas, M.; Playà, E.; Inglès, M.; Rouchy, J.M.; Calvo, J.P.; Gimeno, D. Evaporite sedimentation in a tectonically active basin: The lacustrine Las Minas Gypsum unit (Late Tortonian, SE Spain). Sediment. Geol. 2014, 311, 17–42. [Google Scholar] [CrossRef]
- Calvo, J.P.; Elízaga, E. The Cenajo and Las Minas-Camarillas Basins (Miocene), Southeastern Spain. In A Global Geological Record of Lake Basins; Gierlowski-Kordesch, E., Kelts, K., Eds.; Cambridge University Press: Cambridge, UK, 1994; pp. 319–324. [Google Scholar]
- Jerez Mir, L. Geología de la zona Prebética en la transversal de Elche de la Sierra y sectores adyacentes (provincias de Albacete y Murcia). Ph.D. Thesis, Univ. Granada, Granada, Spain, 1973. [Google Scholar]
- Pérez-Valera, L.A.; Sánchez-Gómez, M.; Pérez-Valera, F.; Azor, A.; Villalaín, J.J. Evolución tectónica de la Falla de Socovos desde el Mioceno hasta la actualidad. Geotemas 2012, 13, 429–432. [Google Scholar]
- Pérez-Valera, L.A.; Rosenbaum, G.; Sánchez-Gómez, M.; Azor, A.; Fernández-Soler, J.M.; Pérez-Valera, F.; Vasconcelos, P.M. Vasconcelos. Age distribution of lamproites along the Socovos Fault (southern Spain) and lithospheric scale tearing. Lithos 2013, 180–181, 252–263. [Google Scholar] [CrossRef]
- Sánchez-Gómez, M.; Pérez-Valera, L.A.; Pérez-Valera, F.; Azor, A. Segmentation of the Socovos Fault based on geological data. In First Iberian Meeting on Active Faults and Paleoseismology; Insua-Arévalo, J.M., Martín-González, F., Eds.; Instituto Geológico y Minero de España: Guadalajara, Spain, 2010; pp. 137–140. [Google Scholar]
- Foucault, A.; Calvo, J.P.; Elizaga, E.; Rouchy, J.M.; Servant-Vildary, S. Place des dépôts lacustres d’âge miocène supérieur de la région de Hellín (province de Albacete, Espagne) dans l’évolution géodynamique des Cordillères Bétiques. C. R. de l’Académie des Sci. Ser. II 1987, 305, 1163–1166. [Google Scholar]
- Permanyer, A.; Ortí, F.; Inglès, M.; Rosell, L.; Salvany, J.M. Contenidos de materia orgánica de formaciones evaporíticas peninsulares. Geogaceta 1991, 10, 48–52. [Google Scholar]
- Reolid, M.; Abad, I.; Selva, A. Elementos Traza y Tierras Raras en el Azufre Rojo de Hellín (Albacete): Interpretación Paleoambiental. Macla 2010, 13, 181–182. [Google Scholar]
- Lindtke, J.; Ziegenbalg, S.B.; Brunner, B.; Rouchy, J.M. Authigenesis of native sulfur and dolomite in a lacustrine evaporitic setting (Hellín Basin, Late Miocene, SE Spain). Geol. Mag. 2011, 148, 655–669. [Google Scholar] [CrossRef]
- Abad, I.; Nieto, F.; Gutiérrez-Alonso, G. Textural and chemical changes in slate-forming phyllosilicates across the external-internal zones transition in the low-grade metamorphic belt of the NW Iberian Variscan Chain. Swiss Bull. Mineral. Petrol. 2003, 83, 63–80. [Google Scholar]
- Connolly, J.A.D. The geodynamic equation of state: What and how. Geochem. Geophy. Geosy. 2009, 10. [Google Scholar] [CrossRef]
- Holland, T.; Powell, R. An internally consistent thermodynamic data set for phases of petrological interest. J. Metamorph. Geol. 1998, 16, 309–343. [Google Scholar] [CrossRef]
- Holland, T.; Powell, R. Calculation of phase relations involving haplogranitic melts using an internally consistent thermodynamic dataset. J. Petrol. 2001, 42, 673–683. [Google Scholar] [CrossRef]
- Holland, T.; Powell, R. Thermodynamics of order-disorder in minerals. 2. Symmetric formalism applied to solid solutions. Am. Mineral. 1996, 81, 1425–1437. [Google Scholar] [CrossRef]
- White, R.W.; Powell, R.; Holland, T. Calculation of partial melting equilibria in the system Na2O-CaO-K2O-FeO-MgO-Al2O-SiO2-H2O (NCKFMASH). J. Metamorph. Geol. 2001, 19, 139–153. [Google Scholar] [CrossRef]
- Holland, T.; Baker, J.; Powell, R. Mixing properties and activity-composition relationships of chlorites in the system MgO-FeO-Al2O3-SiO2-H2O. Eur. J. Mineral. 1998, 10, 395–406. [Google Scholar] [CrossRef]
- Powell, R.; Holland, T. Relating formulations of the thermodynamics of mineral solid solutions: Activity modelling of pyroxenes, amphiboles, and micas. Am. Mineral. 1999, 84, 1–14. [Google Scholar] [CrossRef]
- White, R.W.; Powell, R.; Holland, T.; Johnson, T.E.; Green, E.C.R. New mineral activity–composition relations for thermodynamic calculations in metapelitic systems. J. Metamorph. Geol. 2014, 32, 261–286. [Google Scholar] [CrossRef]
- Fuhrman, M.L.; Lindsley, D.H. Ternary-Feldspar Modeling and Thermometry. Am. Mineral. 1988, 3, 201–215. [Google Scholar]
- McDonough, W.F.; Sun, S.S. The composition of the Earth. Chem. Geol. 1995, 120, 223–253. [Google Scholar] [CrossRef]
- Suan, G.; Rolleau, L.; Mattioli, E.; Suchéras-Marx, B.; Rousselle, B.; Pittet, B.; Vincent, P.; Martin, J.E.; Léna, A.; Spangenberg, J.E.; et al. Palaeoenvironmental significance of Toarcian black shales and event deposits from southern Beaujolais, France. Geol. Mag. 2013, 150, 728–742. [Google Scholar] [CrossRef]
- Whitney, D.L.; Evans, B.W. Abbreviations for names of rock-forming minerals. Am. Mineral. 2010, 95, 185–187. [Google Scholar] [CrossRef]
- Del Moro, S.; Renzulli, A.; Tribandino, M. Pyrometamorphic processes at the magma hydrothermal system interface of active volcanoes: Evidences from Buchite ejecta of Stromboli (Aeolian Islands, Italy). J. Petrol. 2011, 52, 541–564. [Google Scholar] [CrossRef]
- Sharygin, V.V. A hibonite-spinel-corundum-hematite assemblage in plagioclase-clinopyroxene pyrometamorphic rocks, Hatrurim Basin, Israel: Mineral chemistry, genesis and formation temperatures. Mineral. Mag. 2019, 83, 123–135. [Google Scholar] [CrossRef]
- Galuskin, E.V.; Galuskina, I.O.; Gfeller, F.; Krüger, B.; Kusz, J.; Vapnik, Y.; Dulski, M.; Dzierzanowski, P. Silicocarnotite, Ca5[(SiO4)(PO4)](PO4), a new “old’’ mineral from the Negev Desert, Israel, and the ternesite–silicocarnotite solid solution: Indicators of high-temperature alteration of pyrometamorphic rocks of the Hatrurim Complex, Southern Levant. Eur. J. Mineral. 2016, 28, 105–123. [Google Scholar] [CrossRef]





| Samples | Clinker | Paralavas | Clay Layer | Marly Diatomite | |||
|---|---|---|---|---|---|---|---|
| CM-17D | CM-17E | CM-17F | CM-1 | CM-13X | Y-43 | Y-81 | |
| SiO2 | 58.87 | 49.79 | 46.56 | 56.51 | 56.77 | 52.27 | 26.98 |
| Al2O3 | 26.73 | 20.22 | 21.93 | 20.91 | 20.32 | 23.4 | 8.39 |
| Fe2O3* | 4.23 | 18.78 | 19.8 | 10.47 | 11.64 | 5.44 | 3.64 |
| MnO | 0.02 | 0.02 | 0.02 | 0.43 | 0.36 | 0.01 | 0.02 |
| MgO | 1.84 | 1.09 | 1.46 | 1.25 | 1.26 | 1.90 | 1.13 |
| CaO | 1.34 | 2.66 | 4.33 | 3.84 | 3.59 | 2.91 | 30.50 |
| Na2O | 0.10 | 0.30 | 0.17 | 0.15 | 0.12 | 0.09 | 0.04 |
| K2O | 2.87 | 1.92 | 2.16 | 1.21 | 1.23 | 2.42 | 0.98 |
| TiO2 | 1.24 | 0.99 | 1.13 | 1.15 | 1.11 | 1.04 | 0.44 |
| P2O5 | 0.09 | 0.25 | 0.19 | 1.22 | 1.20 | 0.05 | 0.07 |
| LOI | 2.30 | 3.44 | 1.75 | 2.47 | 1.78 | 10.29 | 27.52 |
| TOTAL | 99.60 | 99.46 | 99.50 | 99.61 | 99.38 | 99.83 | 99.70 |
| Li | 34.75 | 30.64 | 30.71 | 44.39 | 35.80 | 32.47 | 12.40 |
| Rb | 44.82 | 61.90 | 90.37 | 33.49 | 68.67 | 150.63 | 63.30 |
| Cs | 9.11 | 8.23 | 8.39 | 6.38 | 8.58 | 12.33 | 4.34 |
| Be | 3.16 | 3.60 | 1.87 | 3.27 | 3.47 | 2.65 | 1.59 |
| Sr | 76.45 | 124.09 | 142.81 | 198.58 | 191.41 | 85.46 | 567.77 |
| Ba | 195.14 | 224.98 | 358.28 | 522.94 | 474.19 | 236.22 | 146.16 |
| Sc | 16.32 | 13.95 | 18.98 | 13.40 | 13.70 | 19.81 | 7.72 |
| V | 478.79 | 700.91 | 519.69 | 535.03 | 523.21 | 414.29 | 60.66 |
| Cr | 181.97 | 144.84 | 199.34 | 135.76 | 153.95 | 146.23 | 64.78 |
| Co | 5.60 | 7.19 | 5.84 | 56.05 | 48.80 | 5.89 | 5.40 |
| Ni | 35.47 | 33.68 | 37.70 | 93.78 | 112.29 | 26.88 | 32.74 |
| Cu | 50.05 | 33.39 | 66.41 | 39.72 | 64.08 | 51.78 | 14.21 |
| Zn | 36.89 | 47.94 | 17.25 | 44.57 | 75.37 | 33.42 | 33.42 |
| Ga | 34.60 | 28.04 | 7.23 | 28.42 | 29.76 | 34.11 | 11.90 |
| Y | 15.63 | 27.58 | 31.68 | 27.47 | 28.92 | 22.00 | 16.79 |
| Nb | 26.36 | 19.62 | 25.36 | 25.34 | 26.95 | 19.25 | 11.01 |
| Ta | 2.22 | 1.62 | 1.98 | 1.85 | 1.75 | 1.54 | 0.67 |
| Zr | 191.97 | 133.99 | 222.62 | 240.32 | 214.12 | 142.77 | 94.16 |
| Hf | 5.52 | 3.88 | 6.03 | 6.72 | 6.15 | 3.79 | 2.07 |
| Mo | 64.97 | 340.81 | 1113.46 | 38.79 | 40.25 | 415.55 | 12.49 |
| Sn | 4.60 | 2.71 | 2.74 | 0.24 | 0.47 | 5.85 | 2.16 |
| Tl | 3.57 | 3.85 | 0.29 | 1.89 | 3.39 | 3.27 | 0.94 |
| Pb | 17.47 | 4.57 | 0.39 | 0.36 | 0.08 | 30.46 | 11.68 |
| U | 165.86 | 64.35 | 200.33 | 14.58 | 23.26 | 4.86 | 2.57 |
| Th | 9.24 | 12.40 | 13.34 | 12.75 | 13.15 | 13.21 | 7.11 |
| La | 17.79 | 36.17 | 47.37 | 44.35 | 47.48 | 37.87 | 25.13 |
| Ce | 43.90 | 73.26 | 100.07 | 95.45 | 96.99 | 85.63 | 49.04 |
| Pr | 5.06 | 8.60 | 12.30 | 11.04 | 11.31 | 8.57 | 5.83 |
| Nd | 18.13 | 31.65 | 46.05 | 40.84 | 41.43 | 31.20 | 20.65 |
| Sm | 4.56 | 6.50 | 9.70 | 8.44 | 8.47 | 6.34 | 4.35 |
| Eu | 1.01 | 1.39 | 2.00 | 1.75 | 1.71 | 1.40 | 0.99 |
| Gd | 3.23 | 5.18 | 7.59 | 6.57 | 6.60 | 4.64 | 3.23 |
| Tb | 0.59 | 0.87 | 1.24 | 1.08 | 1.09 | 0.76 | 0.54 |
| Dy | 3.18 | 4.55 | 6.19 | 5.42 | 5.45 | 3.96 | 2.83 |
| Ho | 0.67 | 0.90 | 1.11 | 0.97 | 0.99 | 0.77 | 0.57 |
| Er | 1.86 | 2.27 | 2.87 | 2.56 | 2.55 | 2.05 | 1.53 |
| Tm | 0.28 | 0.33 | 0.41 | 0.37 | 0.37 | 0.31 | 0.23 |
| Yb | 1.80 | 1.96 | 2.49 | 2.29 | 2.23 | 1.95 | 1.45 |
| Lu | 0.27 | 0.29 | 0.36 | 0.33 | 0.32 | 0.28 | 0.20 |
| Minerals | Clinker Samples (r) | Paralava Samples (*) | |||
|---|---|---|---|---|---|
| CM-17D | CM-17E | CM-17F | CM-1 | CM-13X | |
| Cordierite | X | XX | XXX | XXX | |
| Corundum | X | X | X | ||
| Cristobalite | XX | XXX | XX | ||
| Feldspars | X | X | XX | XX | X |
| Hematite | X | XXX | XX | X | |
| Hercynite | X | X | |||
| Mullite | XX | X | XX | ||
| Quartz | XX | X | X | X | |
| Sillimanite | XX | X | |||
| Tridymite | X | X | X | XX | XX |
| Zeolites | X | X | |||
| Analyzed Phases | wt. % | Calculated Average Formula | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Na | Mg | Al | Si | K | Ca | Mn | Ti | Fe | O | ||
| Anorthite | 0.1 | - | 15.3 | 17.6 | 0.3 | 7.5 | 0.1 | 0.1 | 0.3 | 58.9 | Si2.1Al1.9Ca0.9O8 |
| (n = 12) | 0.1 | - | 0.7 | 0.7 | 0.2 | 0.6 | 0.0 | 0.0 | 0.1 | 0.6 | |
| Cordierite | - | 2.2 | 14.6 | 18.1 | 0.3 | 0.1 | 0.2 | 0.1 | 4.9 | 59.7 | Si5Al4(Fe1.3Mg0.6Mn0.1)K0.1O18 |
| (n = 21) | - | 0.4 | 1.0 | 1.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.3 | 2.3 | |
| Corundum | - | - | 38.8 | 2.1 | - | 0.0 | 0.1 | 0.1 | 3.1 | 56.4 | Al1.8Fe0.1Si0.1O3 |
| (n = 3) | - | - | 2.0 | 1.6 | - | 0.1 | 0.0 | 0.1 | 1.0 | 0.85 | |
| Hematite | - | 0.8 | 3.3 | 0.4 | 0.0 | 0.2 | 0.7 | 1.4 | 39.5 | 54.2 | Fe1.7Al0.2Ti0.1O3 |
| (n = 16) | - | 0.6 | 0.8 | 0.4 | 0.0 | 0.2 | 0.8 | 0.7 | 1.9 | 1.9 | |
| Sillimanite | - | - | 25.0 | 14.1 | 0.4 | 0.1 | - | 0.3 | 1.0 | 59.0 | Al1.9Fe0.1Si1.0O5 |
| (n = 3) | - | - | 1.6 | 2.2 | 0.3 | 0.0 | - | 0.0 | 0.4 | 0.7 | |
| Silica phases | - | - | 0.7 | 34.2 | 0.1 | 0,3 | - | 0.1 | 0.24 | 64.33 | SiO2 |
| (n = 18) | - | - | 1.2 | 2.9 | 0.2 | 0.5 | - | 0.1 | 0.33 | 1.36 | |
| Smectite | - | 0.5 | 13.6 | 19.4 | 0.1 | 0.8 | - | 0.2 | 2.4 | 63.2 | (Si3.3Al0.7)(Fe0.4Mg0.1Al1.67)Ca0.1O10(OH)2 |
| (n = 12) | - | 0.2 | 2.4 | 0.8 | 0.0 | 0.2 | - | 0.2 | 1.9 | 0.5 | |
| Zeolite | - | 0.5 | 11.8 | 16.1 | 0.1 | 0.5 | - | 0.1 | 3.4 | 67.4 | Si2.9Al2.1Fe0.6Mg0.1Ca0.1O10 |
| (n = 4) | - | 0.5 | 12.0 | 16.0 | 0.0 | 0.5 | - | 0.1 | 3.8 | 67.0 | |
| Vitreous phase | - | - | 1.7 | 33.8 | 0.2 | 0.6 | - | 0.1 | 0.5 | 62.9 | |
| (n = 14) | - | - | 1.3 | 3.9 | 0.2 | 0.8 | - | 0.1 | 0.4 | 4.6 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abad, I.; Sánchez-Gómez, M.; Reolid, M.; López Sánchez-Vizcaíno, V. Pyrometamorphic Rocks in the Molinicos Basin (Betic Cordillera, SE Spain): Insights into the Generation of Cordierite Paralavas. Minerals 2019, 9, 748. https://doi.org/10.3390/min9120748
Abad I, Sánchez-Gómez M, Reolid M, López Sánchez-Vizcaíno V. Pyrometamorphic Rocks in the Molinicos Basin (Betic Cordillera, SE Spain): Insights into the Generation of Cordierite Paralavas. Minerals. 2019; 9(12):748. https://doi.org/10.3390/min9120748
Chicago/Turabian StyleAbad, Isabel, Mario Sánchez-Gómez, Matías Reolid, and Vicente López Sánchez-Vizcaíno. 2019. "Pyrometamorphic Rocks in the Molinicos Basin (Betic Cordillera, SE Spain): Insights into the Generation of Cordierite Paralavas" Minerals 9, no. 12: 748. https://doi.org/10.3390/min9120748
APA StyleAbad, I., Sánchez-Gómez, M., Reolid, M., & López Sánchez-Vizcaíno, V. (2019). Pyrometamorphic Rocks in the Molinicos Basin (Betic Cordillera, SE Spain): Insights into the Generation of Cordierite Paralavas. Minerals, 9(12), 748. https://doi.org/10.3390/min9120748






