Spanish Bentonites: A Review and New Data on Their Geology, Mineralogy, and Crystal Chemistry
Abstract
1. Introduction
2. Materials and Methods
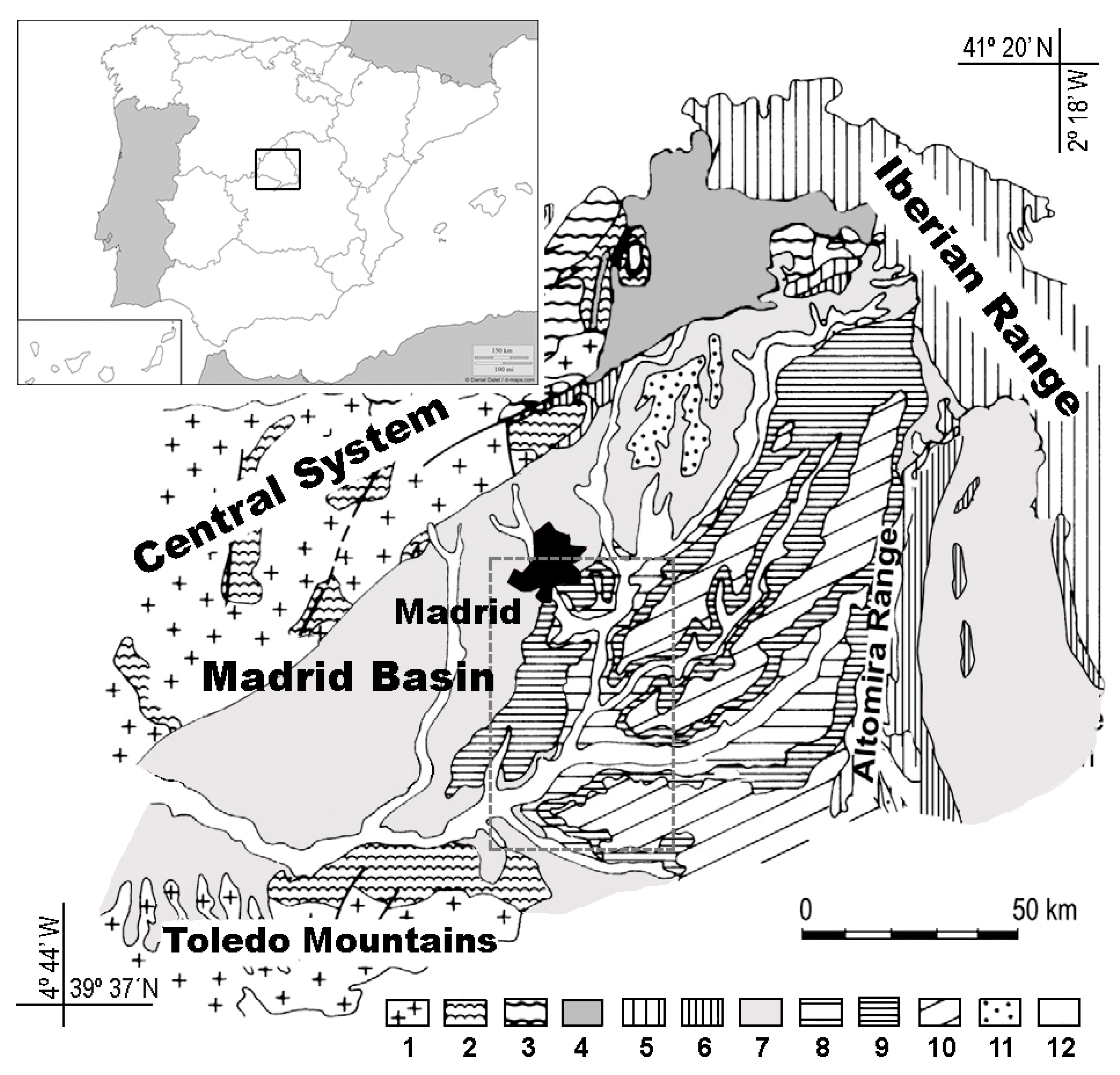
3. Geological Features
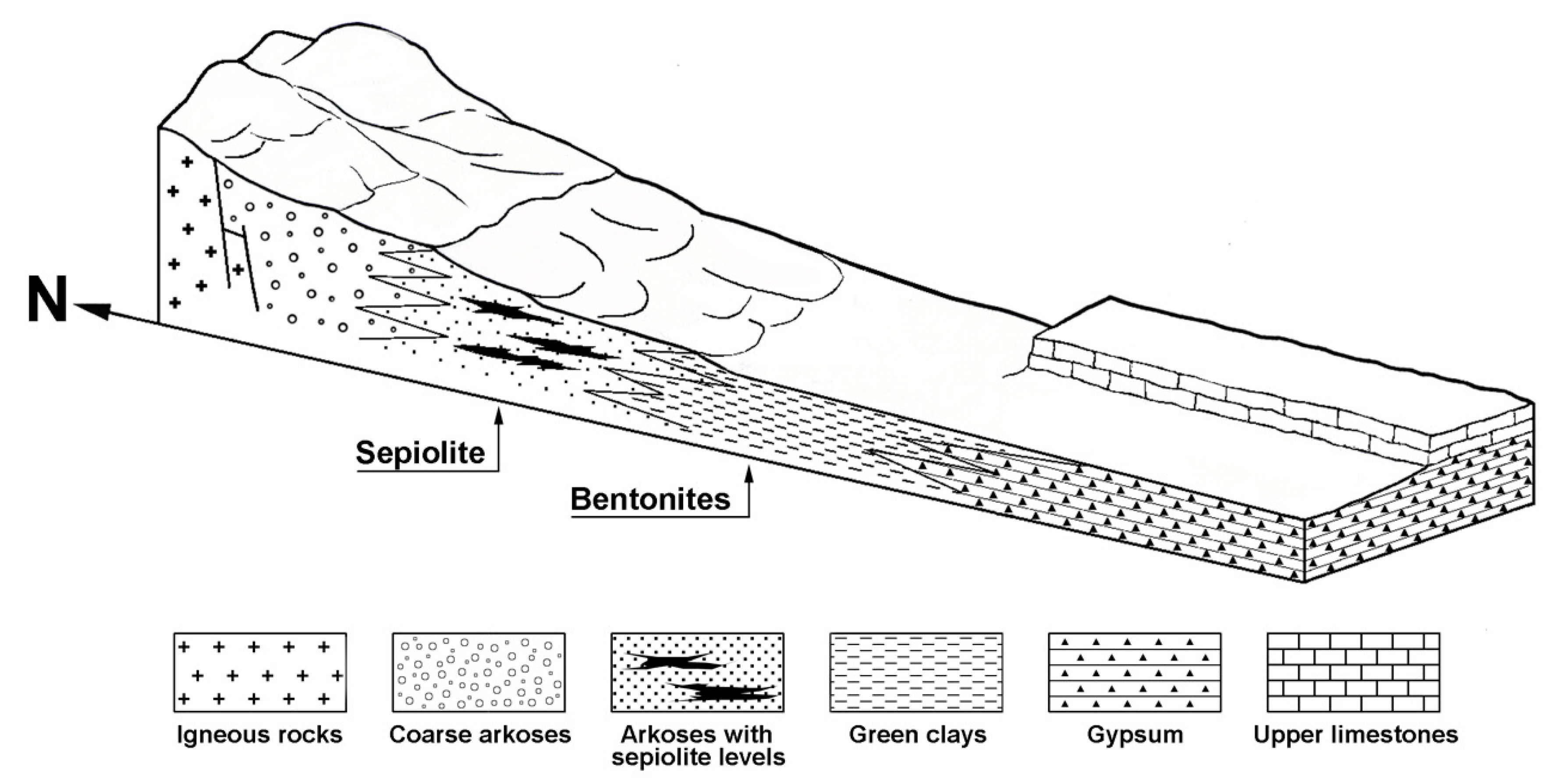
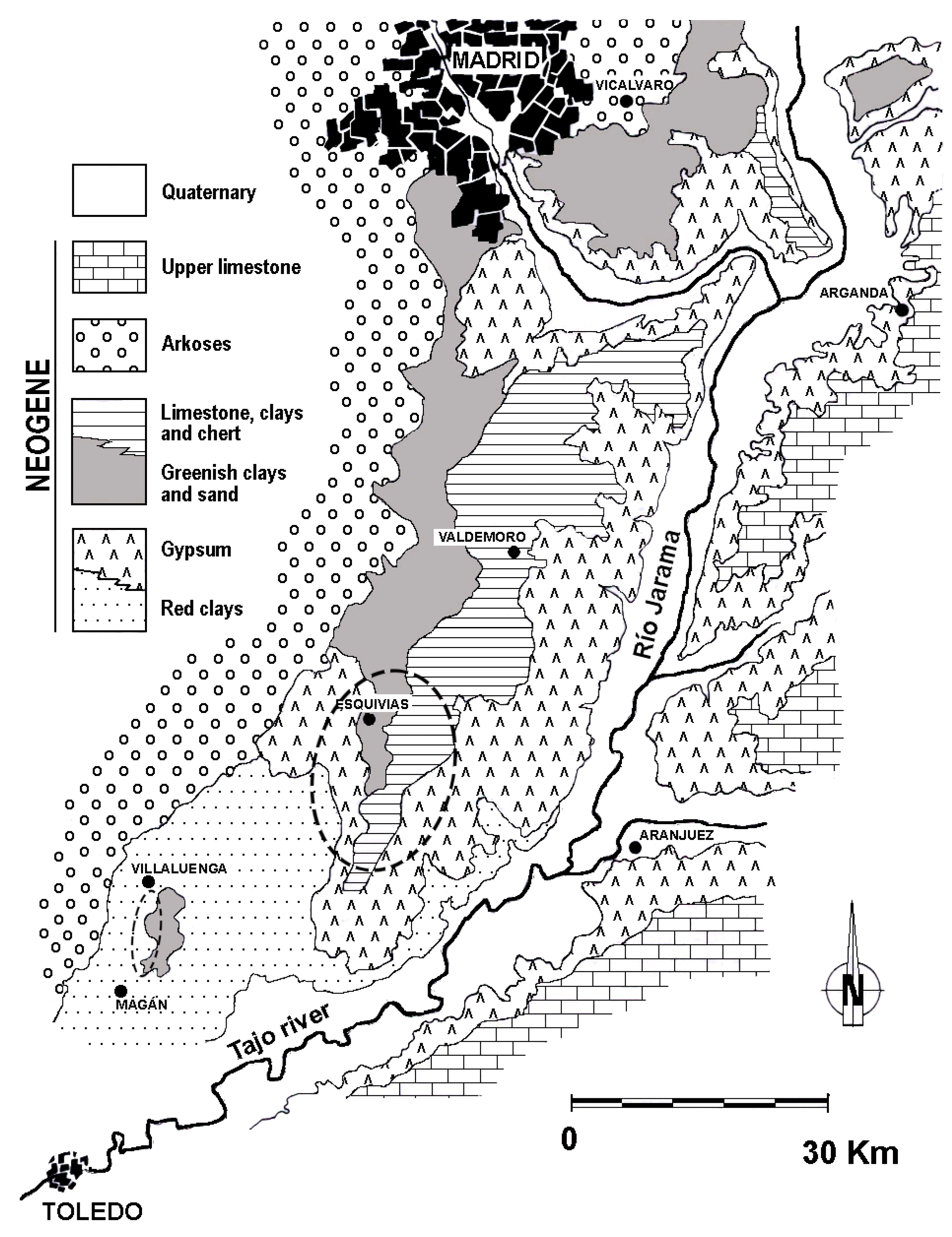
3.1. The Tajo Basin
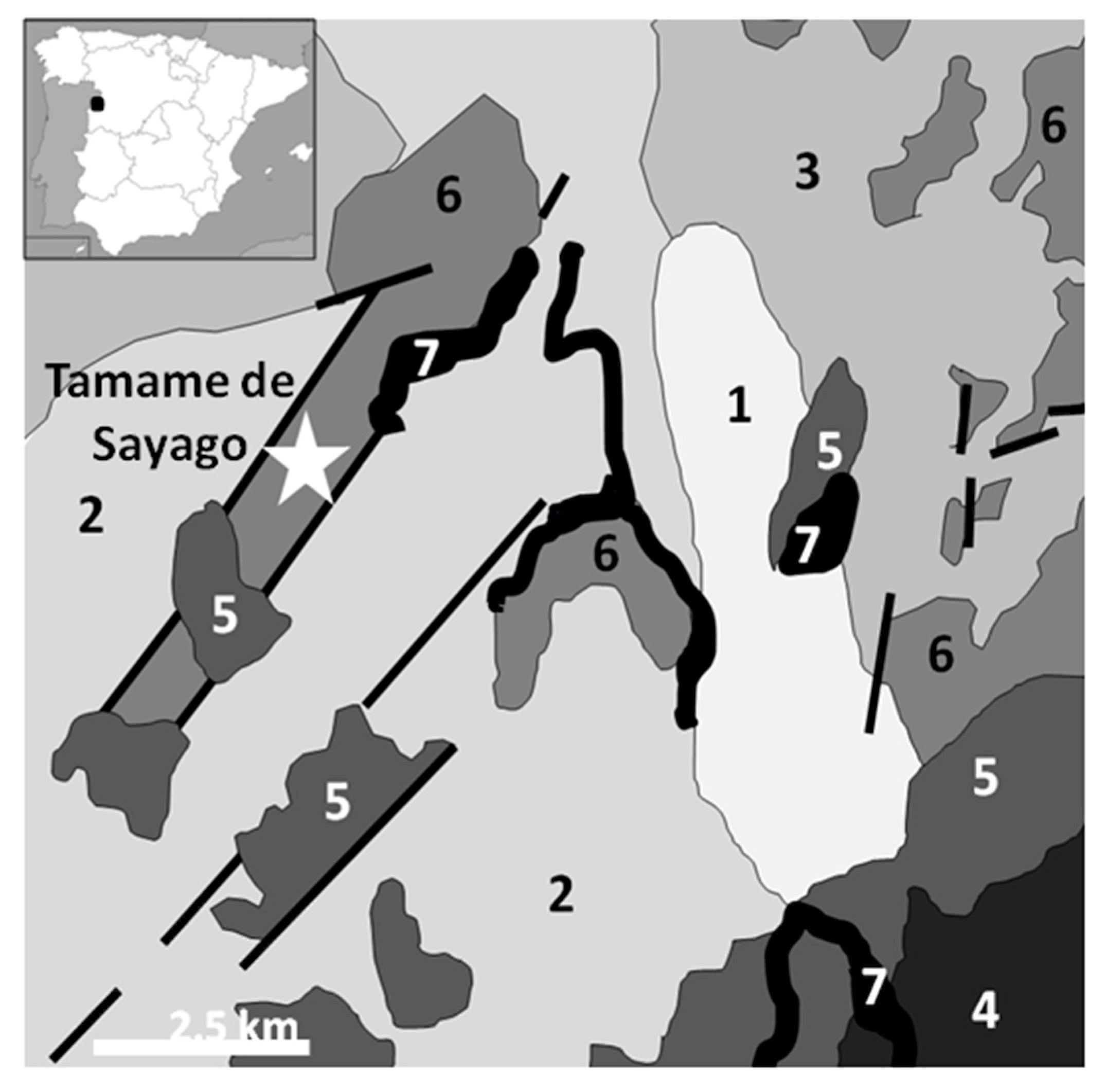
3.2. Tamame de Sayago
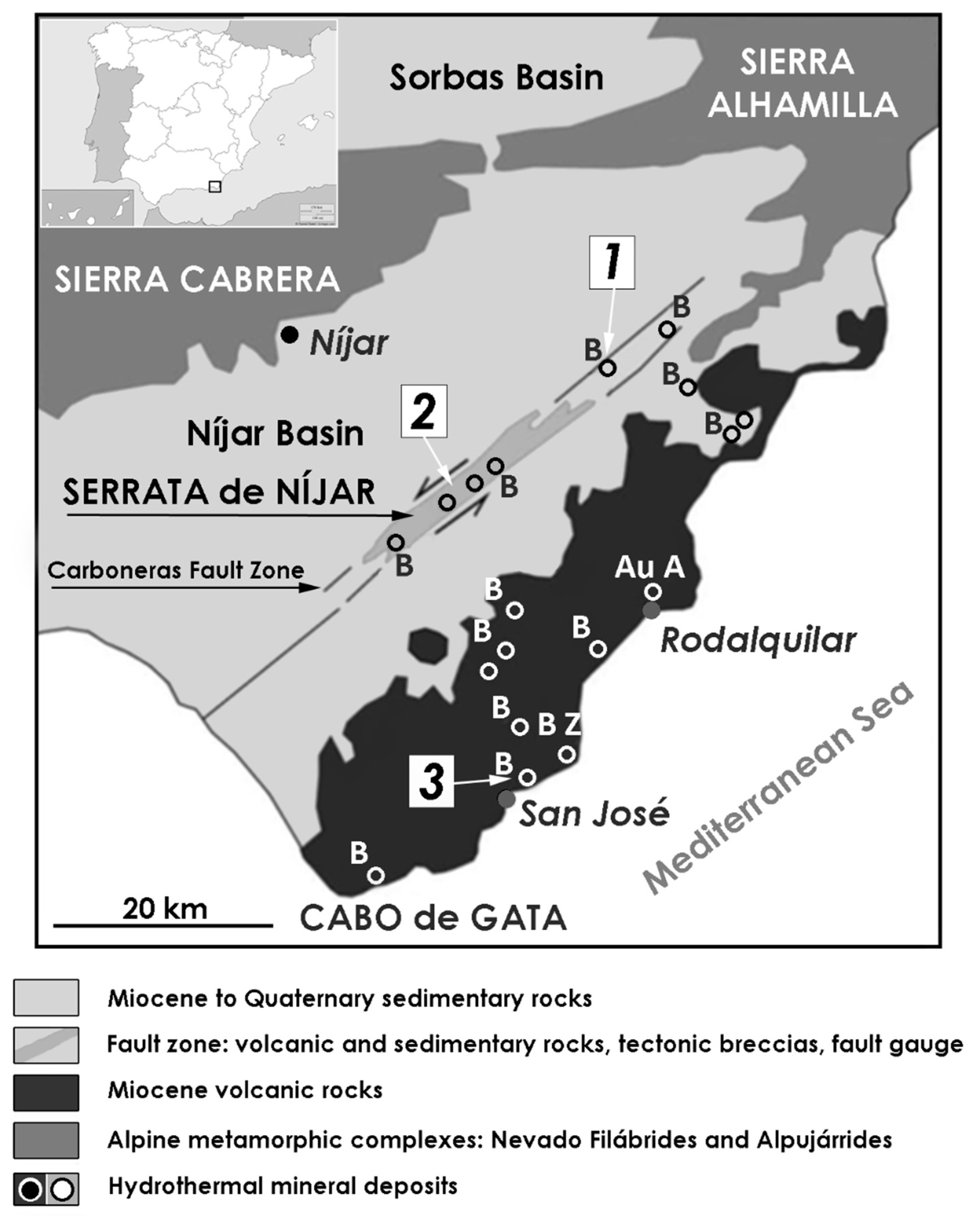
3.3. Cabo de Gata Volcanic Region
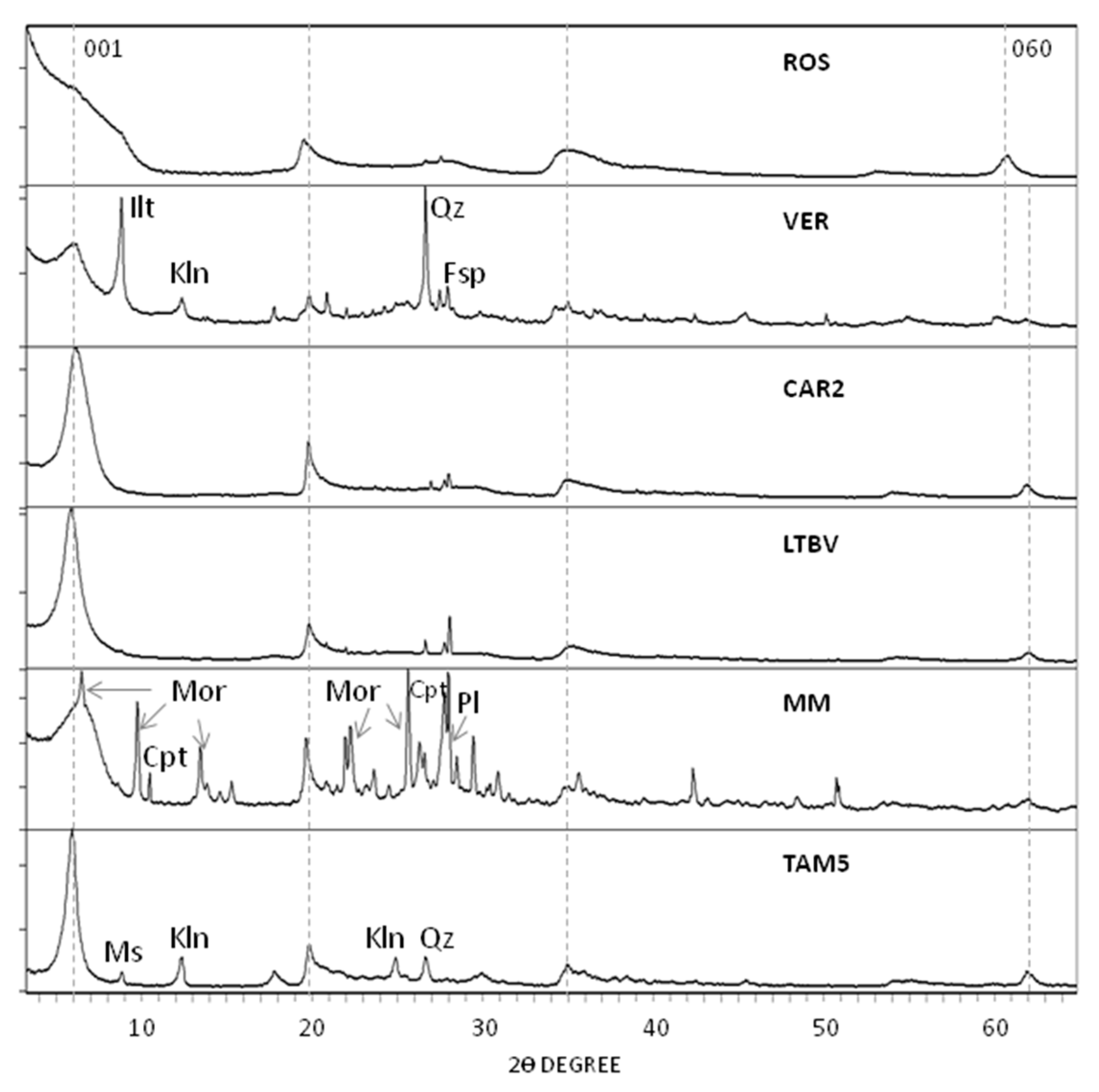
4. Mineralogy and Chemistry of Bentonites
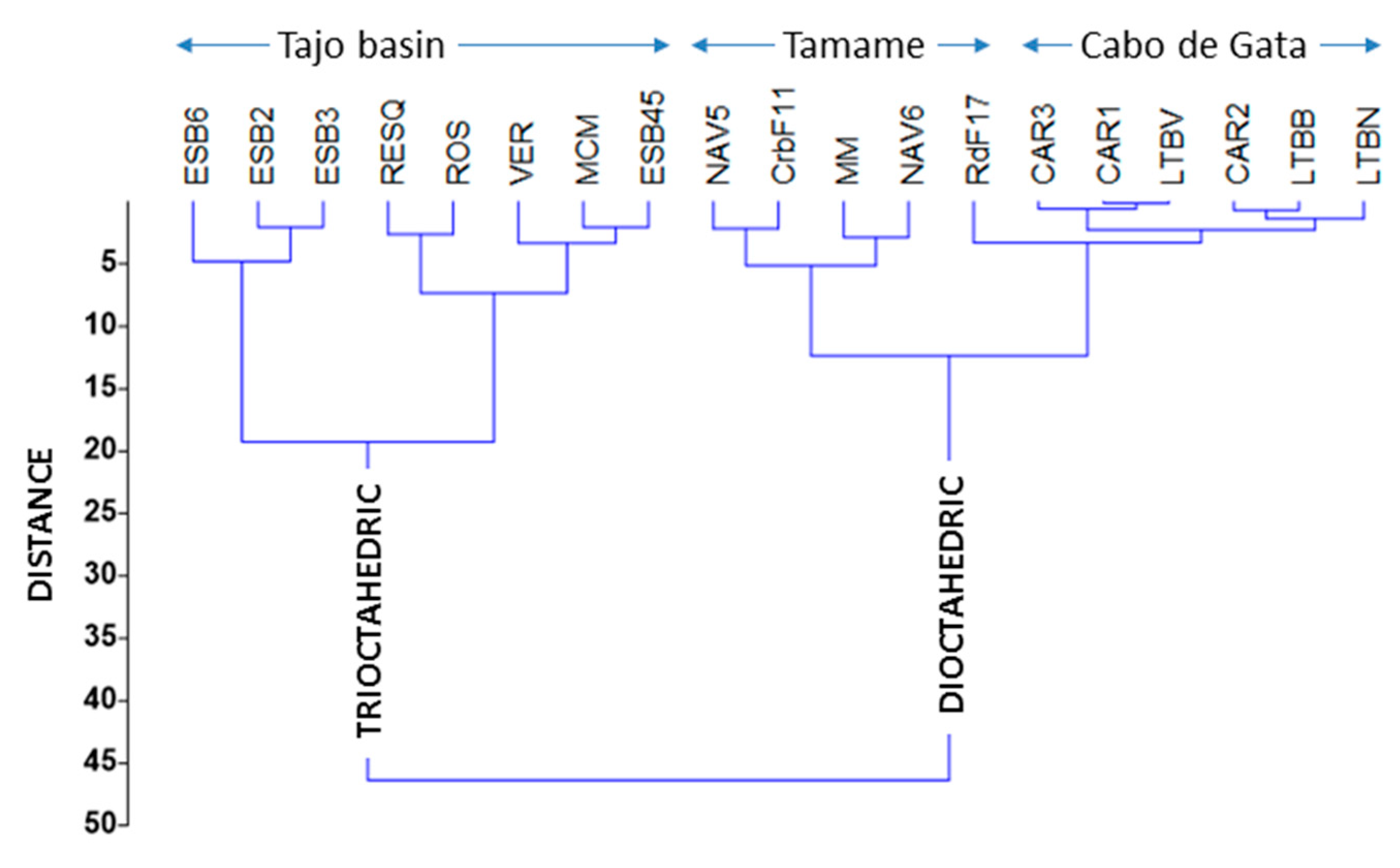
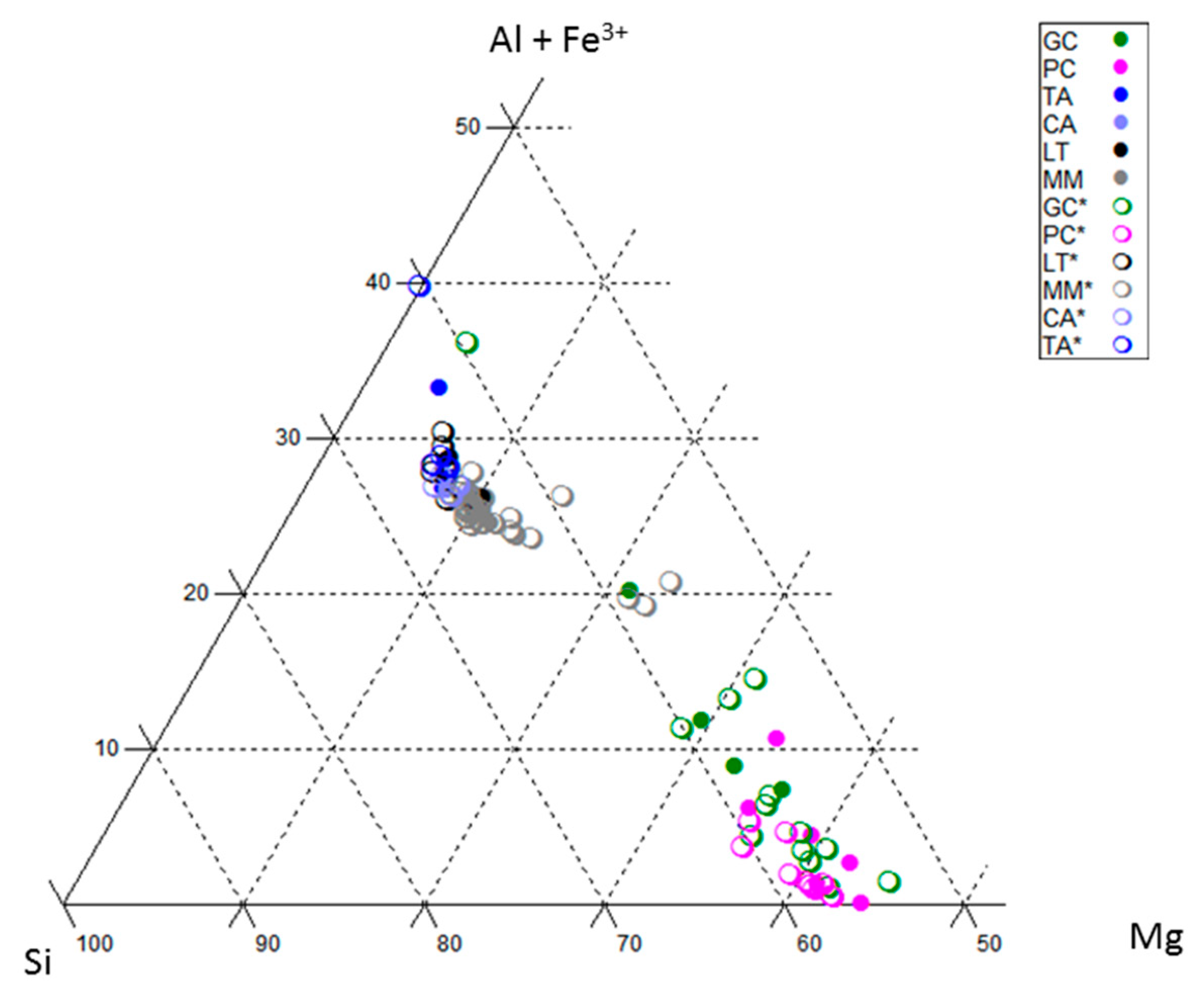
5. Smectite Crystal Chemistry
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Álvarez, A.; Santarén, J.; Esteban-Cubillo, A.; Aparicio, P. Current Industrial Applications of Palygorskite and Sepiolite. Dev. Clay Sci. 2011, 3, 281–298. [Google Scholar]
- Harvey, C.C.; Lagaly, G. Industrial Applications. In Developments in Clay Science; Elsevier: Amsterdam, The Netherlands, 2013; Volume 5, pp. 451–490. [Google Scholar]
- Babu Valapa, R.; Loganathan, S.; Pugazhenthi, G.; Thomas, S.; Varghese, T.O. An Overview of Polymer-Clay Nanocomposites. In Clay-Polymer Nanocomposites; Elsevier: Amsterdam, The Netherlands, 2017; pp. 29–81. [Google Scholar]
- Mohanty, F.; Swain, S.K. Bionanocomposites for Food Packaging Applications. In Nanotechnology Applications in Food: Flavor, Stability Nutrition and Safety; Academic Press: Cambridge, MA, USA, 2017; pp. 363–379. [Google Scholar]
- Rebitski, E.P.; Souza, G.P.; Santana, S.A.A.; Pergher, S.B.C.; Alcântara, A.C.S. Bionanocomposites based on cationic and anionic layered clays as controlled release devices of amoxicillin. Appl. Clay Sci. 2019, 173, 35–45. [Google Scholar] [CrossRef]
- Lee, S.M.; Tiwari, D. Organo and inorgano-organo-modified clays in the remediation of aqueous solutions: An overview. Appl. Clay Sci. 2012, 59–60, 84–102. [Google Scholar] [CrossRef]
- De Brito, B.M.A.; Cartaxo, J.M.; Costa, J.M.R.; Ferreira, H.C.; Neves, G.A. Comparative study of organoclays with ionic surfactant. Mater. Sci. Forum 2014, 798–799, 9–14. [Google Scholar] [CrossRef]
- Singh, P.P.; Ambika, B. Bentonite: A Versatile Clay. In Bentonite: Characteristics, Uses and Implications for the Environment; Mishra, A.K., Ed.; Nova Science Pb. Inc.: London, UK, 2015; pp. 151–172. [Google Scholar]
- Jones, B.F. Clay mineral diagenesis in lacustrine sediments. US Geol. Surv. Bull. 1986, 1578, 291–300. [Google Scholar]
- Darragi, F.; Tardy, Y. Authigenic trioctahedral smectites controlling pH, alkalinity, silica an Mg-concentrations in alkaline lakes. Chem. Geol. 1987, 63, 59–72. [Google Scholar] [CrossRef]
- Hay, R.L.; Pexton, R.E.; Teague, T.T.; Kyser, T.K. Spring-related carbonate rocks, Mg clays, and associated minerals in Pliocene deposits of the Amargosa Desert, Nevada and California. Geol. Soc. Am. Bull. 1986, 97, 1488–1503. [Google Scholar] [CrossRef]
- Fiore, S.; Huertas, F.J.; Huertas, F.; Linares, J. Smectite formation in rhyolitic obsidian as inferred by microscopic (SEM-TEM-AEM) investigation. Clay Miner. 2001, 36, 489–500. [Google Scholar] [CrossRef]
- Birsoy, R. Formation of sepiolite–palygorskite and related minerals from solution. Clays Clay Miner. 2002, 50, 736–745. [Google Scholar] [CrossRef]
- Furquim, S.A.C.; Graham, R.C.; Barbiero, L.; Neto, J.P.D.; Valles, V. Mineralogy and genesis of smectites in an alkaline-saline environment of Panatal Wetland, Brazil. Clays Clay Miner. 2008, 56, 579–595. [Google Scholar] [CrossRef]
- Deocampo, D.M. Authigenic clay minerals in lacustrine mudstones. In Paying Attention to Mudrocks: Priceless; Egenhoff, S., Larsen, D., Fishman, N., Eds.; Geological Society of America Special Paper 515: Colorado, CO, USA, 2015; pp. 49–64. [Google Scholar]
- Shimbashi, M.; Sato, T.; Yamakawa, M.; Fujii, N.; Otake, T. Formation of Fe- and Mg-Rich Smectite under Hyperalkaline Conditions at Narra in Palawan, the Philippines. Minerals 2018, 8, 155. [Google Scholar] [CrossRef]
- Singer, A. Palygorskite in sediments: Detrital, diagenetic or neoformed—A critical review. Geol. Rundsch. 1979, 68, 996–1008. [Google Scholar] [CrossRef]
- Ugolini, F.C. Hydrothermal origin of the clays from the upper slopes of Mauna Kea, Hawaii. Clays Clay Miner. 1974, 22, 189–194. [Google Scholar] [CrossRef]
- Cann, J.R. Metamorphism in the ocean crust. In Maurice Ewing Series; Talwani, M., Harrison, C.G., Hayes, D.E., Eds.; American Geophysical Union: Washington, DC, USA, 1979; pp. 230–238. [Google Scholar]
- Thompson, G. Hydrothermal Fluxes in the Ocean. In Chemical Oceanography; Riley, J.P., Chester, R., Eds.; Elsevier: Amsterdam, The Netherlands, 1983; pp. 271–337. [Google Scholar]
- Aoki, S.; Kohyama, N.; Hotta, H. Hydrothermal clay minerals found in sediment containing yellowish-brown material from the Japan Basin. Mar. Geol. 1996, 129, 331–336. [Google Scholar] [CrossRef]
- Manuella, F.C.; Carbone, S.; Barreca, G. Origin of Saponite-Rich Clays in a Fossil Serpentinite-Hosted Hydrothermal System in the Crustal Basement of the Hyblean Plateau (Sicily, Italy). Clays Clay Miner. 2012, 60, 18–31. [Google Scholar] [CrossRef]
- Khoury, H.N.; Eberl, D.D.; Jones, B.F. Origin of Magnesium Clays from the Amargosa Desert, Nevada. Clays Clay Miner. 1982, 30, 327–336. [Google Scholar] [CrossRef]
- Bellanca, A.; Calvo, J.P.; Censi, P.; Neri, R.; Pozo, M. Recognition of lake-level changes in Miocene lacustrine units, Madrid Basin, Spain. Evidence from facies analysis, isotope geochemistry and clay mineralogy. Sediment. Geol. 1992, 76, 135–153. [Google Scholar] [CrossRef]
- Cuevas, J.; Pelayo, M.; Rivas, P.; Leguey, S. Characterization of Mg-clays from the Neogene of the Madrid Basin and their potential as backfilling and sealing material in high level radioactive waste disposal. Appl. Clay Sci. 1993, 7, 383–406. [Google Scholar] [CrossRef]
- Ece, Ö.İ.; Çoban, F. Geology, occurrence, and genesis of Eskişehir sepiolites, Turkey. Clays Clay Miner. 1994, 42, 81–92. [Google Scholar] [CrossRef]
- Hay, R.L.; Hughes, R.E.; Kyser, T.K.; Glass, H.D.; Liu, J. Magnesium-Rich Clays of the Meerschaum Mines in the Amboseli Basin, Tanzania and Kenya. Clays Clay Miner. 1995, 43, 455–466. [Google Scholar] [CrossRef]
- Pozo, M.; Casas, J. Origin of kerolite and associated Mg clays in palustrine-lacustrine environments. The Esquivias deposit (Neogene Madrid Basin, Spain). Clay Miner. 1999, 34, 395–418. [Google Scholar] [CrossRef]
- Karakaya, M.Ç.; Karakaya, N.; Temel, A. Mineralogical and geochemical characteristics and genesis of the sepiolite deposits at Polatli Basin (Ankara, Turkey). Clays Clay Miner. 2011, 59, 286–314. [Google Scholar] [CrossRef]
- Kadir, S.; Külah, T.; Önalgil, N.; Erkoyun, H.; Elliott, W.C. Mineralogy, Geochemistry, and Genesis of Bentonites in Miocene Volcanic-sedimentary Units of the Ankara-Çankiri Basin, Central Anatolia, Turkey. Clays Clay Miner. 2017, 65, 64–91. [Google Scholar] [CrossRef]
- Pozo, M.; Calvo, J.P. An Overview of Authigenic Magnesian Clays. Minerals 2018, 8, 520. [Google Scholar] [CrossRef]
- Mayayo, M.J.; Bauluz, B.; Lopez, J.M.G. Variations in the chemistry of smectites from the Calatayud Basin (NE Spain). Clay Miner. 2000, 35, 365–374. [Google Scholar] [CrossRef]
- Harder, H. The role of magnesium in the formation of smectite minerals. Chem. Geol. 1972, 10, 31–39. [Google Scholar] [CrossRef]
- Harder, H. Nontronite synthesis at low temperature. Chem. Geol. 1976, 18, 169–180. [Google Scholar] [CrossRef]
- Harder, H. Synthesis if iron layer silicate minerals under natural conditions. Clays Clay Miner. 1978, 26, 65–72. [Google Scholar] [CrossRef]
- Decarreau, A.; Bonnin, D. Synthesis and crystallogenesis at low temperature of Fe(II)-smectites by evolution of coprecipitated gels: Experiments in partial reducing conditions. Clay Miner. 1986, 21, 861–877. [Google Scholar] [CrossRef]
- Decarreau, A.; Bonnin, D.; Badaut-Trauth, D.; Couty, R.; Kaiser, P. Synthesis and crystallogenesis of ferric smectites by evolution of Si-Fe coprecipitates in oxidizing conditions. Clay Miner. 1987, 22, 207–223. [Google Scholar] [CrossRef]
- Masár, J.; Luptáková, V.; Kuchta, L.; Fajnor, V.S. Hydrothermal synthesis of montmorillonite containing iron in structure. Chem. Pap. 1987, 41, 343–350. [Google Scholar]
- Kloprogge, J.T.; Jansen, J.B.H.; Geus, J.W. Characterization of synthetic Na-beidellite. Clays Clay Miner. 1990, 38, 409–414. [Google Scholar] [CrossRef]
- Tomita, K.; Yamane, H.; Kawano, M. Synthesis of smectites from volcanic glass at low temperature. Clays Clay Miner. 1993, 41, 655–661. [Google Scholar] [CrossRef]
- Huertas, F.J.; Cuadros, J.; Huertas, F.; Linares, J. Experimental study of the hydrothermal formation of smectite in the beidellite-saponite series. Am. J. Sci. 2000, 300, 504–527. [Google Scholar] [CrossRef]
- Fesharaki, O.; García-Romero, E.; Cuevas-González, J.; López-Martínez, N. Clay mineral genesis evolution in the Miocene sediments of Somosaguas, Madrid Basin, Spain. Clay Miner. 2007, 42, 187–201. [Google Scholar] [CrossRef]
- Cuadros, J.; Dekov, V.M.; Arroyo, X.; Nieto, F. Smectite formation in submarine hydrothermal sediments: Samples from the HMS Challenger expedition (1872–1876). Clays Clay Miner. 2011, 59, 147–164. [Google Scholar] [CrossRef]
- Lacoviello, F.; Giorgetti, G.; Nieto, F.; Memmi, I.T. Evolution with depth from detrital to authigenic smectites in sediments from AND-2A drill core (McMurdo Sound, Antarctica). Clay Miner. 2012, 47, 481–498. [Google Scholar] [CrossRef]
- Christidis, G.E.; Dunham, A.C. Compositional Variations in Smectites. Part II: Alteration of Acidic Precursors. A Case Study from Milos Island, Greece. Clay Miner. 1997, 32, 253–270. [Google Scholar] [CrossRef]
- García-Romero, E.; Vegas, J.; Baldonedo, J.L.; Marfil, R. Clay minerals as alteration products in basaltic volcaniclastic deposits of La Palma (Canary Islands, Spain). Sediment. Geol. 2005, 174, 237–253. [Google Scholar] [CrossRef]
- Christidis, G.E. Genesis and compositional heterogeneity of smectites. Part III: Alteration of basic pyroclastic rocks—A case study from the Troodos Ophiolite Complex, Cyprus. Am. Mineral. 2006, 91, 685–701. [Google Scholar] [CrossRef]
- Suarez, M.; Robert, M.; Elsass, F.; Martín-Pozas, J.M. Evidence of a precursor in the neoformation of palygorskite—New data by analytical electron microscopy. Clay Miner. 1994, 29, 255–264. [Google Scholar] [CrossRef]
- Christidis, G.E. Formation and growth of smectites in bentonites: A case study from Kimolos Island, Aegean, Greece. Clays Clay Miner. 2001, 49, 204–215. [Google Scholar] [CrossRef]
- Dias, M.I.; Suárez, M.; Prates, S. Las bentonitas de Benavila (Portugal). Caracterización mineralógica y propiedades. Geogaceta 2004, 35, 99–102. [Google Scholar]
- Christidis, G.E.; Huff, W.D. Geological Aspects and Genesis of Bentonites. Elements 2009, 5, 93–98. [Google Scholar] [CrossRef]
- Instituto Geológico y Minero de España (IGME). Panorama Minero; IGME: Madrid, Spain, 2019. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST. Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- De Vicente, G.; Cloething, S.; Van Wees, J.D.; Cunha, P.P. Tectonic classification of Cenozoic Iberian foreland basins. Tectonohysics 2011, 502, 38–61. [Google Scholar] [CrossRef]
- Calvo, J.P.; Jones, B.J.; Bustillo, M.A.; Fort, R.; Alonso-Zarza, A.M.; Kendall, C. Sedimentology and geochemistry of carbonates from lacustrine sequences in the Madrid Basin, Central Spain. Chem. Geol. 1995, 123, 173–191. [Google Scholar] [CrossRef]
- Martín Escorza, C. Actividad Tectónica durante el Mioceno en las fracturas del basamento de la fosa del Tajo. Estud. Geol. 1976, 32, 509–522. [Google Scholar]
- Alberdi, M.T.; Hoyos, M.; Junco, F.; López Martinez, N.; Morales, J.; Sese, C.; Soria, D. Bioiestratigraphie et évolution sedimentaire de l’aire de Madrid. Colloquium Mediterranean Neogene Continental Paleoenvironments and Paleoclimatic evolution. Monpellier 1983, 12–83. [Google Scholar]
- Junco, F.; Calvo, J.P. Cuenca de Madrid. In Libro Jubilar JM Ríos; Instituto Geológico y Minero de España, Ríos, J.M., Eds.; Geologico y Minero de España: Madrid, Spain, 1983; pp. 534–542. [Google Scholar]
- Mejías, A.G.; Ordoñez, S.; Calvo, J.P. Nuevas aportaciones al conocimiento geológico de la Cuenca de Madrid. Rev. Mater. Procesos Geol. 1983, 1, 163–191. [Google Scholar]
- Portero, J.M.; Aznar, J.M. Evolución morfotectónica y sedimentación terciarias en el Sistema Central y Cuencas Limítrofes (Duero y Tajo). I Congr. Esp. Geol. 1984, 3, 147–153. [Google Scholar]
- Torres, T.; Junco, F.; Zapata de La Vega, S.L.; Plaza, J.M. Similitud de los procesos sedimentarios del Neogeno de la cuenca del Tajo y en la Depresión intermedia. I Congr. Esp. Geol. 1984, 1, 285–300. [Google Scholar]
- Calvo, J.P.; Ordoñez, S.; Hoyos, M.; García del Cura, M.A. Caracterización sedimentológica de la Unidad Intermedia del Mioceno de la zona Sur de Madrid Rev. Mater. Procesos Geol. 1984, 2, 145–176. [Google Scholar]
- Calvo, J.P.; Alonso-Zarza, A.M.; García del Cura, M.A. Models of marginal lacustrine sedimentation in response to varied source areas in the Madrid Basin (Central Spain). Paleogeogr. Paleoclimatol. Paleoecol. 1989, 70, 199–214. [Google Scholar] [CrossRef]
- Brell, J.M.; Doval, M.; Carames, M. Clay minerals distribution in the evaporitic Miocene sediments of the Tajo Basin, Spain. Mineral. Petrogr. Acta 1985, 29, 267–276. [Google Scholar]
- Doval, M.; García Santiago, P.; Domínguez Díaz, M.C.; Brell, J.M. Mineralogía de las arcillas de las facies evaporíticas de la Cuenca del Tajo. Trab. Geol. 1985, 15, 267–274. [Google Scholar]
- Doval, M.; Domínguez Díaz, M.C.; Brell, J.M.; García-Romero, E. Mineralogía y sedimentología de las facies distales del borde norte de la cuenca del Tajo. Bol. Soc. Esp. Mineral. 1985, 8, 257–269. [Google Scholar]
- Galán, E.; Álvarez, A.; Esteban, M.A. Characterization and technical properties of a Mg-rich bentonite. Appl. Clay Sci. 1986, 1, 295–309. [Google Scholar] [CrossRef]
- Garcia-Romero, E. Estudio Mineralógico y Estratigráfico de las Arcillas de las Facies Centrales del Neógeno del Borde sur de la Cuenca del Tajo. Ph.D. Thesis, Universidad complutense de Madrid, Madrid, Spain, 1988; p. 436. [Google Scholar]
- García-Romero, E.; Brell, J.M.; Doval, M.; Navarro, J.V. Caracterización mineralógica y estratigráfica de las formaciones neógenas del borde sur de la cuenca del Tajo (Comarca de la Sagra). Bol. Geol. Min. 1990, 101–106, 945–956. [Google Scholar]
- Pozo, M.; Moreno, A.; Casas, J.; Martín Rubí, J.A. Estudio geoquímico de litofacies con arcillas magnésicas en depósitos lacutres-palustres de la Cuenca de Madrid. Bol. Soc. Esp. Mineral. 1996, 19, 71–83. [Google Scholar]
- De Santiago, C.; Suárez, M.; Garcia Romero, E.; Domínguez Díaz, M.C.; Doval, M. Electron microscopic study of the illie-smectite transformation in the bentonites from Cerro del Águila (Toledo, Spain). Clay Miner. 1998, 33, 501–510. [Google Scholar]
- García-Romero, E.; Bustillo, A.; Suarez, M. Characteristics of a Mg-palygorskite in Miocene rocks, Madrid basin (Spain). Clays Clay Miner. 2004, 52, 484–494. [Google Scholar]
- Bustillo, M.A.; Alonso-Zarza, A.M. Overlapping of pedogenesis and meteoric diagenesis in distal alluvial and shallow lacustrine deposits in the Madrid Mioceno Basin, Spain. Sediment. Geol. 2007, 198, 255–271. [Google Scholar] [CrossRef]
- García-Romero, E. Génesis de arcillas magnésicas en la cuenca de Madrid: Interrogantes planteados. Bol. Geol. Min. 2004, 115, 629–640. [Google Scholar]
- Dominguez, M.C.; Brell Parladé, J.M.; Doval Montoya, M.; Garccía-Romero, E. Análisis de los minerales de la arcilla y sus procesos genéticos en las formaciones arcillosas de la Cuenca del Tajo. Estud. Geol. 1997, 53, 185–196. [Google Scholar] [CrossRef]
- Pozo, M.; Casas, J.; Martín De Vidales, J.L.; Medina, J.A.; Martín Rubí, J.A. Características texturales y composicionales en depósitos de arcillas magnésicas de la Cuenca de Madrid. I) Kerolitas (sector de Esquivias y Pinto). Bol. Geol. Min. 1999, 110–111, 77–102. [Google Scholar]
- García-Rivas, J.; Suárez, M.; Torres, T.; Sánchez-Palencia, Y.; García-Romero, E.; Ortíz, J.E. Geochemistry and Biomarker Analysis of the Bentonites from Esquivias (Toledo, Spain). Minerals 2018, 8, 291. [Google Scholar] [CrossRef]
- De Santiago, C.; Suárez, M.; Garcia Romero, E.; Doval, M. Mg-rich smectite “precursor” phase in the Tagus Basin, Spain. Clays Clay Miner. 2000, 48, 366–373. [Google Scholar]
- Martínez, F.J.; Julivert, M.; Sebastián, A.; Arboleda, M.L.; Gil-Ibarguchi, J.I. Structural and thermal evolution of metamorphism. In Pre-Mesozoic Geology of Iberia; Dallmeyer, R.D., Martínez García, E., Eds.; Springer: Berlin, Germany, 1988; pp. 207–211. [Google Scholar]
- López-Moro, F.J.; López-Plaza, M. Monzonitic series from the Variscan Tormes Dome (Central Iberian Zone): Petrogenetic evolution from monzogabbro to granite magmas. Lithos 2004, 72, 19–44. [Google Scholar] [CrossRef]
- Dias, G.; Leterrier, J.; Mendes, A.; Simões, P.P.; Bertrand, J.M. U–Pb zircon and monazite geochronology of post-collisional Hercynian granitoids from the Central Iberian Zone (Northern Portugal). Lithos 1988, 45, 349–369. [Google Scholar] [CrossRef]
- Molina, E.; Cantano, M.; Vicente, M.A.; García Rodríguez, P. Someaspects of paleoweathering in the Iberian Hercynian Massif. Catena 1990, 17, 333–346. [Google Scholar] [CrossRef]
- Siemcalsa. Mapa Geológico y Minero de Castilla y León: Escala 1:400.000; Siemcalsa: Valladolid, Spain, 1997. [Google Scholar]
- Manchado, E.; Suarez, M.; Garcia Romero, E. A three-dimensional study of the kaolin deposit from Tamame de Sayago (Zamora, Spain). In Proceedings of the Euroclay 2007, Aveiro, Portugal, 22–27 July 2007; p. 73. [Google Scholar]
- Manchado, E.M.; García-Romero, E.; Suárez, M. Génesis del Yacimiento de arcillas especiales de Tamame de Sayago (Zamora). Macla 2012, 16, 94–95. [Google Scholar]
- Manchado, E.M. Arcillas Especiales de Tamame de Sayago (Zamora): Mineralogía, Génesis y Propiedades. Ph.D. Thesis, Universidad de Salamanca, Salamanca, Spain, 2012; p. 526. [Google Scholar]
- Manchado, E.M.; Suárez, M.; García-Romero, E. Minerales del Grupo de la Alunita en el Yacimiento de Caolinita de Tamame de Sayago (Zamora). Macla 2008, 9, 151–152. [Google Scholar]
- Doblas, M.; Oyarzun, R. Neogene extensional collapse in the western Mediterranean (Betic-Rif Alpine orogenic belt): Implications for the genesis of the Gibraltar Arc and magmatic activity. Geology 1989, 17, 430–433. [Google Scholar] [CrossRef]
- Platt, J.P.; Vissers, R.L.M. Extensional collapse of thickened continental lithosphere: A working hypothesis for the Alborán Sea and Gibraltar Arc. Geology 1989, 17, 540–543. [Google Scholar] [CrossRef]
- López Ruíz, J.; Cebriá, J.M.; Doblas, M. Cenozoic volcanism I: The Iberian Pensinsula. In The Geology of Spain; Gibbons, W., Moreno, T., Eds.; Geologiacal Society: London, UK, 2002; pp. 417–438. [Google Scholar]
- García-Romero, E.; Suárez, M.; Oyarzun, R.; López-García, J.A.; Regueiro. Fault-hosted palygorskite from the Serrata de Nijar Deformation Zone (SE Spain). Clays Clay Miner. 2006, 54, 324–332. [Google Scholar]
- Huibregtse, P.; Alebeek, H.V.; Mattijs, Z.; Biermann, C. Palaeostress analysis of the northern Nijar and southern Vera basins: Constraints for the Neogene displacement history of major strike-slip faults in the Betic Cordilleras, SE Spain. Tectonophysics 1998, 300, 79–101. [Google Scholar] [CrossRef]
- Keller, J.V.A.; Hall, S.H.; McClay, K.R. Shear fracture pattern and microstructural evolution in transpres-sional fault zones from field and laboratory studies. J. Struct. Geol. 1997, 19, 1173–1187. [Google Scholar] [CrossRef]
- Reyes, E.; Caballero, E.; Huertas, F.; Linares, J.A. Bentonite deposits from Cabo de Gata Region, Almería, SE. Spain. Guidebook for excursions. In Proceedings of the Sixth Meeting of the European Clay Groups, Sevilla, Spain, 7–10 September 1987. [Google Scholar]
- Linares, J.; Huertas, F.; Reyes, E.; Caballero, E.; Barahona, E.; Guardiola, J.L.; Yañez, J.; Romero, E.; Delgado, A. Investigaciones de Bentonitas Como Material de Sellado Para Almacenamiento de Residuos Radiactivos de Alta Actividad; ENRESA Thechnical Publication: Madrid, Spain, 1993; p. 324. [Google Scholar]
- Leone, G.; Reyes, E.; Cortecci, G.; Pochini, A.; Linares, J. Genesis of bentonites from Cabo de Gata, Almería, Spain: A stable isotope study. Clay Miner. 1983, 18, 227–238. [Google Scholar] [CrossRef]
- Caballero, E.; Reyes, E.; Linares, J.; Huertas, F. Hydrothermal solutions related to bentonite génesis, Cabo de Gata región, Almería, SE Spain. Mineral. Petrograhica Acta 1985, 29, 187–196. [Google Scholar]
- Caballero, E.; Reyes, E.; Yusta, A.; Huertas, F.; Linares, J. Las bentonitas de la zona del Cabo de Gata, Almería. Geoquímica y mineralogía. Acta Geol. Hisp. 1985, 20, 267–287. [Google Scholar]
- Reyes, E.; Huertas, F.; Linares, J. Bentonitas de Andalucía (España): Yacimientos hidrotermales del norte de Rodalquilar (Almería). In Proceeding of the First International Congress on Bentonites, 1978; Gallizzi: Sassari, Italy, 1978; Volume 1, pp. 125–147. [Google Scholar]
- Reyes, E.; Huertas, F.; Linares, J. Génesis y geoquímica de esmectitas de Andalucia, España. In Proceeding of the First International Congress on Bentonites, 1978; Gallizzi: Sassari, Italy, 1978; Volume 1, pp. 149–173. [Google Scholar]
- Delgado, A. Estudio Isotópico de los Procesos Diagenéticos e Hidrotermales Relacionados con la Génesis de Bentonitas (Cabo de Gata, Almería). Ph.D. Thesis, Universidad de Granada, Granada, Spain, 1993; p. 413. [Google Scholar]
- Caballero, E.; Jimenez de Cisneros, C.; Huertas, F.J.; Huertas, F.; Pozzuoli, A.; Linares, J. Bentonites from Cabo de Gata, Almería, Spain: A mineralogical and geochemical overview. Clay Miner. 2005, 40, 463–480. [Google Scholar] [CrossRef]
- Martín Vivaldi, J.L.; Linares, J. Las bentonitas de cabo de Gata. I: Yacimientos de Los Trancos y Majada de Las Vacas. Bol. Geol. Min. 1968, 79, 513–523. [Google Scholar]
- Cuadros, J.; Delgado, A.; Cardenete, A.; Reyes, E.; Linares, J. Kaolinite/montmorillonite resembles beidellite. Clays Clay Miner. 1994, 42, 643–651. [Google Scholar] [CrossRef]
- Pelayo, M.; García-Romero, E.; Labajo, M.A.; Pérez del Villar, L. Occurence of Fe-Mg-rich smectites and corrensite in the Morrón de Mateo bentonite deposit (Cabo de Gata region, Spain): A natural analogue of the bentonite barrier in a radwaste repository. Appl. Geochem. 2011, 26, 1135–1168. [Google Scholar] [CrossRef][Green Version]
- Di Battistini, G.; Toscani, L.; Iaccarino, S.; Villa, I.M. K/Ar ages and the geological setting of calc-alkaline volcanic rocks from Sierra de Gata. SE Spain. Neues Jahrb. Miner. Mon. 1987, 8, 369–383. [Google Scholar]
- Fernández Soler, J.M. El Volcanismo Calco-Alcalino de Cabo de Gata (Almería). Ph.D. Thesis, Universidad de Granada, Granada, Spain, 1992; p. 243. [Google Scholar]
- Pelayo, M.; García-Romero, E.; Labajo, M.A.; Pérez del Villar, L. Evidence of montmorillonite/Fe-rich smectite transformation in the Morrón de Mateo bentonite deposit (Spain): Implications for the clayey barrier behaviour. Appl. Clay Sci. 2016, 131, 59–70. [Google Scholar] [CrossRef]
- Martín de Vidales, J.L.; Pozo, M.; Alia, J.M.; García Navarro, F.; Rull, R. Kerolite-stevensite mixed-layers from The Madrid Basin, Central Spain. Clay Miner. 1991, 26, 329–342. [Google Scholar] [CrossRef]
- Göktürk, H.; Dyer, A. Sodium, rubidium and caesium uptake on Los Trancos clay. J. Radioanal. Nucl. Chem. Lett. 1994, 188, 231–241. [Google Scholar] [CrossRef]
- Cuevas, J.; Vigil de La Villa, R.; Ramírez, S.; Petit, S.; Meunier, A.; Leguey, S. Chemistry of Mg smectites in lacustrine sediments from the Vicálvaro sepiolite deposit, Madrid Neogene basin (Spain). Clays Clay Miner. 2003, 5, 457–472. [Google Scholar] [CrossRef]
- Newman, A.C.D.; Bown, G. Chemistry of Clays and Clay Minerals; Mineralogical Society: London, UK, 1987; p. 480. [Google Scholar]
- Christidis, G.E. The concept of layer charge of smectites and its implications for important smectite-water properties. EMU Notes Mineral. 2011, 11, 239–260. [Google Scholar]
- Cuevas, J. Caracterización de Esmectitas Magnésicas de la Cuenca de Madrid Como Materiales de Sellado: Ensayos de Alteración Hidrotermal. Ph.D. Thesis, UAM, Madrid, Spain, 1990; p. 357. [Google Scholar]
- Pozo, M.; Cuevas, J.; Moreno, A.; Redondo, R.; y Leguey, S. Caracterización de arcillas magnésicas bentoníticas en la zona de Yuncos (Toledo). Bol. Geol. Min. 1991, 102–106, 893–904. [Google Scholar]
- Augustin, V. Estudio Geoquímico de las Alteraciones de Rocas Volcánicas en el Sector del Pozo de los Frailes, Cabo de Gata (Almería). Bachelor’s Thesis, Granada University, Granada, Spain, 1973; p. 260. [Google Scholar]
- Caballero, E. Quimismo del Proceso de Bentonitización en la Región Volcánica de Cabo de Gata (Almería). Ph.D. Thesis, Universidad de Granada, Granada, Spain, 1985; p. 198. [Google Scholar]
- García-Romero, E.; Huertas, J. Bentonites from Cabo de Gata (Almería, Spain). Fieldtrip Guide. In Proceedings of 16th International Clay Conference; Sociedad Española de Arcillas (SEA): Granada, Spain, 17– July 2017; p. 36. [Google Scholar]
- Caballero, E.; Fernandez Porto, M.J.; Linares, J.; Huertas, F.; Reyes, E. Las bentonitas de la Serrata de Nijar (Almería): Mineralogía, geoquímica y mineralogénesis. Estud. Geol. 1983, 39, 121–140. [Google Scholar]




| Location | Quarry | Label | Impurities | Geological Origin | |
|---|---|---|---|---|---|
| Tajo Basin | Esquivias | Green clays | ESB 2 | Ilt, (Kln, Qz) Fsp * | Sedimentary |
| ESB 3 | Ilt, (Kln, Qz) Fsp * | ||||
| ESB 6 | Ilt, Qz (Kln, Fsp) | ||||
| Pink clays | PB 10 | (Ilt) | |||
| PB 11 | (Ilt, Qz) | ||||
| RESQ | Qz * | ||||
| ESB 45 | (Qz, Ilt) | ||||
| Magán Hill | Pink clays | MCM | (Qz, Pl) | ||
| ROS | Qz *, Pl * | ||||
| Green C. | VER | Ilt, Qz (Kln, Fsp) | |||
| Cabo de Gata | Cortijo de Archidona | CAR 1 | (Pl) | Hydrothermal alteration of volcanic rocks | |
| CAR 2 | (Pl) Qz * | ||||
| CAR 3 | Pl | ||||
| Los Trancos | LT BB | (Pl) | |||
| LTBV | (Pl) Qz * | ||||
| LTBN | (Pl) | ||||
| Morrón de Mateo | MM | Mor, Cpt, (Pl) | |||
| Tamame de Sayago | Roderica | RodF17 | (Kln) | Hydrothermal alteration of kaolinitized granite | |
| Navalacruz | NAV 5 | Kln, Qz, (Ms, Fsp) | |||
| NAV 6 | Qz, (Ms) | ||||
| Carboneras | CarbF11 | Qz, (Ms, Fsp) | |||
| SiO2 | Al2O3 | Fe2O3 | FeO | MnO | MgO | CaO | Na2O | K2O | TiO2 | P2O5 | LOI | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ESB 2 | 48.55 | 11.52 | 3.70 | 0.40 | 0.04 | 15.90 | 0.67 | 0.27 | 2.02 | 0.52 | 0.11 | 15.96 |
| ESB 3 | 50.80 | 12.76 | 3.49 | 0.90 | 0.04 | 14.58 | 0.53 | 0.40 | 2.22 | 0.60 | 0.08 | 14.09 |
| ESB 6 | 50.75 | 14.01 | 3.88 | 0.90 | 0.03 | 10.40 | 0.77 | 0.48 | 2.91 | 0.48 | 0.20 | 14.56 |
| ESB 45 | 51.62 | 5.15 | 1.51 | 0.25 | 0.02 | 23.07 | 0.68 | 0.16 | 0.90 | 0.24 | 0.08 | 16.67 |
| MCM | 52.08 | 6.21 | 1.79 | <0.1 | 0.03 | 20.41 | 0.56 | 0.39 | 1.54 | 0.22 | 0.04 | 15.49 |
| VER | 48.85 | 6.64 | 2.33 | 0.17 | 0.03 | 20.96 | 0.68 | 0.22 | 1.24 | 0.31 | 0.11 | 17.52 |
| ROS | 52.15 | 2.11 | 0.71 | <0.01 | 0.01 | 25.39 | 2.63 | 0.05 | 0.40 | 0.11 | 0.05 | 17.03 |
| RESQ | 48.56 | 1.61 | 0.71 | <0.01 | 0.02 | 24.52 | 2.76 | 0.03 | 0.33 | 0.08 | 0.04 | 21.66 |
| NAV 5 | 57.79 | 19.88 | 1.53 | 0.00 | 0.00 | 2.03 | 1.52 | 0.18 | 0.93 | 0.21 | 0.37 | 15.20 |
| NAV 6 | 60.58 | 16.21 | 1.77 | 0.00 | 0.00 | 3.02 | 1.96 | 0.13 | 0.79 | 0.29 | 0.16 | 14.63 |
| RodF17 | 52.37 | 19.51 | 2.10 | 0.00 | 0.00 | 3.87 | 1.88 | 0.11 | 0.49 | 0.09 | 0.18 | 20.17 |
| CarbF11 | 57.49 | 17.01 | 2.49 | 0.00 | 0.01 | 3.10 | 1.61 | 0.11 | 1.29 | 0.40 | 0.10 | 17.38 |
| CAR 1 | 51.42 | 16.92 | 2.52 | <0.1 | 0.09 | 4.71 | 1.31 | 0.94 | 0.55 | 0.17 | 0.02 | 19.93 |
| CAR 2 | 51.99 | 16.83 | 3.26 | <0.1 | 0.02 | 4.31 | 1.10 | 1.17 | 0.44 | 0.16 | 0.03 | 19.27 |
| CAR 3 | 50.73 | 17.30 | 2.46 | <0.1 | 0.04 | 4.72 | 1.30 | 0.98 | 0.35 | 0.15 | 0.03 | 20.36 |
| LTBB | 52.12 | 17.75 | 2.41 | <0.1 | 0.11 | 4.56 | 1.29 | 0.51 | 0.14 | 0.20 | 0.03 | 20.77 |
| LTBV | 51.25 | 17.01 | 2.31 | <0.1 | 0.08 | 4.78 | 1.32 | 0.51 | 0.43 | 0.17 | 0.06 | 20.61 |
| LTBN | 53.47 | 17.21 | 2.21 | <0.1 | 0.09 | 4.87 | 1.05 | 1.07 | 0.27 | 0.16 | 0.03 | 19.42 |
| MM | 57.38 | 13.84 | 2.48 | 0.20 | 0.02 | 2.28 | 4.15 | 2.32 | 0.90 | 0.36 | 0.08 | 14.87 |
| Mean of the Chemical Analyses | Tetrahedral Cations (TC) | Octahedral Cations (OC) | Interlayer Cations (IC) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | Fe2O3 | TiO2 | MgO | CaO | NaO | K2O | Si | AlIV | Fe3+ | AlVI | Fe3+ | Mg | Ti | Ʃo | Ca | K | Na | TC | OC | IC | ||
| ESB 2 | M | 60.62 | 7.82 | 4.44 | 26.19 | 0.48 | 0.46 | 7.43 | 0.57 | 0.56 | 0.41 | 4.78 | 5.75 | 0.02 | 0.07 | −0.57 | 0.47 | 0.11 | |||||
| n: 18 | SD | 2.29 | 4.57 | 3.43 | 5.52 | 0.23 | 0.67 | ||||||||||||||||
| ESB 3 | M | 63.23 | 8.19 | 3.59 | 23.86 | 0.52 | 0.04 | 0.56 | 7.66 | 0.34 | 0.83 | 0.33 | 4.31 | 5.47 | 0.07 | 0.09 | 0.01 | −0.34 | 0.10 | 0.24 | |||
| n: 19 | SD | 1.94 | 3.27 | 4.59 | 5.14 | 0.16 | 0.38 | 0.36 | |||||||||||||||
| ESB 6 | M | 61.91 | 12.27 | 2.71 | 20.89 | 0.84 | 0.10 | 1.28 | 7.50 | 0.50 | 1.25 | 0.25 | 3.77 | 5.27 | 0.11 | 0.20 | 0.02 | −0.49 | 0.04 | 0.44 | |||
| n: 14 | SD | 2.38 | 4.25 | 1.11 | 3.89 | 0.70 | 0.12 | 0.91 | |||||||||||||||
| ESB 45 | M | 66.16 | 5.51 | 1.19 | 26.32 | 0.39 | 0.36 | 7.94 | 0.06 | 0.72 | 0.11 | 4.71 | 5.54 | 0.05 | 0.06 | −0.06 | −0.09 | 0.16 | |||||
| n: 36 | SD | 3.48 | 4.01 | 1.46 | 4.24 | 0.19 | 0.62 | ||||||||||||||||
| MCM | M | 59.91 | 5.91 | 1.70 | 30.96 | 0.50 | 0.24 | 0.76 | 7.34 | 0.66 | 0.19 | 0.16 | 5.65 | 6.00 | 0.07 | 0.12 | 0.06 | −0.66 | 0.35 | 0.32 | |||
| n: 19 | SD | 1.80 | 1.08 | 0.61 | 1.59 | 0.20 | 0.70 | 1.31 | |||||||||||||||
| PB 10 | M | 61.30 | 6.02 | 2.85 | 0.08 | 28.17 | 1.02 | 0.55 | 7.46 | 0.54 | 0.32 | 0.26 | 5.21 | 0.01 | 5.80 | 0.13 | 0.09 | −0.54 | 0.16 | 0.35 | |||
| n: 14 | SD | 2.93 | 3.50 | 1.99 | 0.18 | 3.50 | 0.40 | 0.74 | |||||||||||||||
| PB 11 | M | 59.09 | 11.22 | 5.42 | 0.20 | 21.88 | 0.51 | 0.02 | 1.67 | 7.28 | 0.72 | 0.91 | 0.50 | 4.02 | 0.02 | 5.43 | 0.07 | 0.26 | −0.72 | 0.27 | 0.40 | ||
| n: 19 | SD | 2.94 | 6.84 | 2.90 | 0.47 | 7.42 | 0.21 | 0.08 | 1.31 | ||||||||||||||
| VER | M | 57.99 | 18.99 | 6.65 | 14.27 | 0.42 | 1.65 | 7.09 | 0.91 | 1.83 | 0.61 | 2.60 | 5.04 | 0.06 | 0.26 | −0.91 | 0.52 | 0.38 | |||||
| n: 39 | SD | 3.64 | 8.14 | 3.95 | 8.82 | 0.64 | 1.37 | ||||||||||||||||
| ROS | M | 65.83 | 1.50 | 0.57 | 31.55 | 0.44 | 0.11 | 7.93 | 0.07 | 0.14 | 0.05 | 5.67 | 5.86 | 0.06 | 0.02 | −0.07 | −0.09 | 0.14 | |||||
| n: 50 | SD | 1.62 | 1.24 | 0.63 | 2.40 | 0.27 | 0.22 | ||||||||||||||||
| RESQ | M | 63.90 | 1.30 | 0.40 | 34.03 | 0.23 | 0.02 | 0.12 | 7.74 | 0.19 | 0.04 | 6.14 | 6.14 | 0.03 | 0.02 | −0.35 | 0.28 | 0.08 | |||||
| n: 76 | SD | 1.45 | 1.66 | 0.21 | 1.74 | 0.39 | 0.15 | 0.48 | |||||||||||||||
| NAV 5 | M | 60.32 | 31.50 | 2.97 | 2.80 | 1.31 | 1.09 | 7.14 | 0.86 | 3.53 | 0.26 | 0.49 | 4.28 | 0.17 | 0.16 | −0.86 | 0.35 | 0.50 | |||||
| n: 11 | SD | 3.42 | 4.41 | 1.86 | 1.05 | 0.43 | 0.59 | ||||||||||||||||
| NAV 6 | M | 65.15 | 26.32 | 1.12 | 4.72 | 2.50 | 0.17 | 7.63 | 0.37 | 3.26 | 0.10 | 0.82 | 4.18 | 0.31 | 0.03 | −0.37 | −0.28 | 0.65 | |||||
| n: 29 | SD | 1.92 | 1.75 | 0.64 | 0.93 | 0.90 | 0.22 | ||||||||||||||||
| RodF17 | M | 64.35 | 23.88 | 2.85 | 5.14 | 2.76 | 1.02 | 7.64 | 0.36 | 2.98 | 0.25 | 0.91 | 4.14 | 0.35 | 0.15 | −0.36 | −0.49 | 0.85 | |||||
| n: 14 | SD | 3.19 | 3.94 | 1.46 | 1.15 | 1.92 | 0.82 | ||||||||||||||||
| CarbF11 | M | 65.92 | 21.30 | 4.32 | 5.18 | 2.42 | 0.87 | 7.83 | 0.17 | 2.81 | 0.39 | 0.92 | 4.12 | 0.31 | 0.13 | −0.17 | −0.56 | 0.75 | |||||
| n: 19 | SD | 2.46 | 1.85 | 1.81 | 0.90 | 0.61 | 0.61 | ||||||||||||||||
| CAR 1 | M | 65.58 | 22.45 | 3.83 | 6.38 | 1.02 | 0.26 | 0.48 | 7.74 | 0.26 | 2.86 | 0.34 | 1.12 | 4.32 | 0.13 | 0.07 | 0.06 | −0.26 | −0.15 | 0.39 | |||
| n: 42 | SD | 1.25 | 1.16 | 1.68 | 0.93 | 0.30 | 0.38 | 0.40 | |||||||||||||||
| CAR 2 | M | 67.89 | 19.97 | 5.26 | 5.53 | 0.87 | 0.55 | 8.01 | 2.78 | 0.47 | 0.97 | 4.22 | 0.11 | 0.08 | 0.04 | −0.32 | 0.30 | ||||||
| n: 27 | SD | 0.92 | 1.82 | 2.27 | 0.49 | 0.21 | 0.53 | ||||||||||||||||
| CAR 3 | M | 65.48 | 21.02 | 4.39 | 6.95 | 1.39 | 0.44 | 0.33 | 7.76 | 0.24 | 2.70 | 0.39 | 1.23 | 4.32 | 0.18 | 0.05 | 0.10 | −0.24 | −0.27 | 0.51 | |||
| n: 27 | SD | 1.05 | 0.74 | 1.41 | 0.37 | 0.24 | 0.33 | 0.14 | |||||||||||||||
| LTBB | M | 65.74 | 25.68 | 2.36 | 4.88 | 1.17 | 0.09 | 0.08 | 7.68 | 0.32 | 3.22 | 0.21 | 0.85 | 4.28 | 0.15 | 0.01 | 0.02 | −0.32 | −0.02 | 0.33 | |||
| n: 28 | SD | 2.48 | 2.95 | 1.19 | 1.19 | 0.96 | 1.21 | 0.21 | |||||||||||||||
| LTBV | M | 66.42 | 22.24 | 2.28 | 6.87 | 1.66 | 0.04 | 0.48 | 7.81 | 0.19 | 2.89 | 0.20 | 1.20 | 4.29 | 0.21 | 0.07 | 0.01 | −0.19 | −0.33 | 0.50 | |||
| n: 19 | SD | 1.81 | 2.55 | 1.62 | 1.81 | 0.35 | 0.23 | 0.88 | |||||||||||||||
| LTBN | M | 65.60 | 21.59 | 4.79 | 7.06 | 0.74 | 0.09 | 0.14 | 7.75 | 0.25 | 2.76 | 0.43 | 1.24 | 4.43 | 0.09 | 0.02 | 0.02 | −0.25 | 0.05 | 0.22 | |||
| n: 26 | SD | 2.73 | 1.65 | 4.46 | 0.98 | 0.20 | 0.17 | 0.09 | |||||||||||||||
| MM | M | 65.04 | 17.18 | 8.07 | 7.83 | 0.97 | 0.17 | 0.72 | 7.82 | 0.18 | 2.26 | 0.73 | 1.40 | 4.39 | 0.13 | 0.11 | 0.04 | −0.18 | −0.23 | 0.41 | |||
| n: 19 | SD | 4.47 | 3.06 | 5.72 | 2.37 | 0.69 | 0.38 | 0.42 | |||||||||||||||
| Ref. | Tetrahedral Cations | Octahedral Cations | Interlayer Cations | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Si | AlIV | AlVI | Fe3+ | Mg | Ti | Ʃo | Ca | K | Na | X+ | |||
| Tajo Basin | Green Clays | [67] * | 7.40 | 0.60 | 0.40 | 0.05 | 5.40 | 5.85 | 0.30 | ||||
| [71] * | 7.52 | 0.48 | 0.88 | 0.52 | 3.62 | 5.02 | 0.10 | 0.38 | |||||
| [71] * | 6.18 | 1.80 | 3.58 | 0.16 | 0.46 | 4.20 | 1.58 | ||||||
| [71] * | 6.70 | 1.30 | 1.10 | 0.46 | 3.62 | 5.18 | 0.06 | 0.50 | |||||
| [25] | 7.33 | 0.67 | 0.33 | 0.25 | 5.00 | 0.02 | 5.60 | 0.40 | 0.05 | ||||
| [25] | 7.26 | 0.74 | 0.52 | 0.35 | 4.55 | 0.03 | 5.45 | 0.39 | 0.13 | ||||
| [114] | 7.44 | 0.56 | 0.54 | 0.26 | 4.64 | 5.44 | 0.04 | 0.41 | |||||
| [115] | 7.60 | 0.40 | 0.30 | 0.24 | 4.60 | 5.14 | 0.15 | 0.29 | |||||
| [115] | 7.54 | 0.46 | 0.26 | 0.18 | 5.20 | 5.64 | 0.04 | 0.33 | |||||
| [28] | 7.53 | 0.47 | 0.19 | 0.15 | 5.29 | 0.02 | 5.65 | 0.03 | 0.04 | ||||
| [28] | 7.68 | 0.32 | 0.13 | 5.68 | 0.01 | 5.82 | 0.01 | 0.04 | |||||
| [75] | 7.00 | 1.00 | 0.18 | 5.92 | 6.10 | 0.12 | 0.03 | ||||||
| [75] | 6.40 | 1.60 | 0.88 | 0.82 | 3.68 | 0.1 | 5.48 | 0.18 | 0.46 | ||||
| Pink Clays | [78] | 8.01 | 0.36 | 0.11 | 4.76 | 5.26 | 0.01 | 0.01 | |||||
| [25] | 7.69 | 0.31 | 0.09 | 0.08 | 5.61 | 5.78 | 0.29 | 0.01 | |||||
| [28] | 7.88 | 0.12 | 0.02 | 0.03 | 5.88 | 0.01 | 5.93 | 0.02 | |||||
| [28] | 8.00 | 0.10 | 0.05 | 5.66 | 5.81 | 0.04 | 0.04 | ||||||
| [28] | 7.90 | 0.10 | 0.03 | 0.03 | 5.89 | 0.01 | 5.95 | 0.02 | 0.01 | ||||
| [28] | 8.00 | 0.10 | 0.05 | 5.66 | 5.81 | 0.04 | 0.01 | 0.04 | |||||
| [28] | 7.90 | 0.10 | 0.03 | 0.03 | 5.89 | 0.01 | 5.96 | 0.02 | 0.01 | ||||
| [75] | 7.98 | 0.02 | 0.64 | 0.06 | 4.80 | 5.50 | 0.07 | 0.04 | |||||
| [109] | 8.00 | 0.13 | 0.05 | 5.60 | 5.78 | 0.08 | 0.06 | 0.04 | |||||
| [111] | 7.94 | 0.06 | 0.15 | 0.09 | 5.34 | 0.01 | 5.58 | 0.18 | 0.01 | 0.08 | |||
| [111] | 7.51 | 0.49 | 0.43 | 0.16 | 4.95 | 0.01 | 5.54 | 0.35 | 0.08 | ||||
| [28] | 7.80 | 0.20 | 0.06 | 0.06 | 5.56 | 0.01 | 5.68 | 0.02 | 0.06 | ||||
| Cabo de Gata | Los Trancos | [101] | 7.81 | 0.19 | 2.96 | 0.12 | 1.00 | 4.09 | 0.94 | ||||
| [101] | 7.74 | 0.26 | 3.06 | 0.21 | 0.79 | 4.06 | 0.85 | ||||||
| [103] | 7.36 | 0.64 | 3.18 | 0.32 | 0.69 | 4.19 | 0.77 | ||||||
| [99,100] | 7.53 | 0.45 | 3.08 | 0.17 | 0.76 | 4.01 | 0.89 | ||||||
| [95] | 7.59 | 0.41 | 3.02 | 0.26 | 0.87 | 4.15 | 0.84 | ||||||
| [95] | 7.56 | 0.44 | 3.27 | 0.19 | 0.77 | 4.23 | 0.16 | 0.01 | 0.03 | ||||
| Morrón de Mateo | [101] | 7.68 | 0.32 | 2.71 | 0.47 | 1.06 | 4.24 | 0.66 | |||||
| [116] | 7.79 | 0.21 | 2.26 | 0.87 | 1.00 | 4.13 | 0.82 | ||||||
| [117] | 7.79 | 0.21 | 2.35 | 0.67 | 1.17 | 4.19 | 0.83 | ||||||
| [95] | 7.62 | 0.38 | 2.57 | 0.56 | 1.11 | 4.24 | 0.91 | ||||||
| [107] * | 7.77 | 0.23 | 2.21 | 0.75 | 1.44 | 4.40 | 0.17 | 0.12 | 0.01 | ||||
| [107] * | 7.92 | 0.08 | 2.56 | 0.44 | 1.29 | 4.29 | 0.08 | 0.06 | 0.27 | ||||
| [107] * | 6.87 | 1.13 | 2.28 | 0.75 | 1.71 | 4.74 | 0.10 | 0.20 | 0.19 | ||||
| [107] * | 7.46 | 0.54 | 2.09 | 0.85 | 1.49 | 4.43 | 0.27 | 0.07 | 0.15 | ||||
| [107] * | 7.57 | 0.43 | 1.95 | 0.90 | 1.74 | 4.59 | 0.20 | 0.08 | 0.05 | ||||
| [107] * | 7.52 | 0.48 | 1.96 | 0.87 | 1.58 | 4.41 | 0.24 | 0.24 | 0.13 | ||||
| [107] * | 7.70 | 0.30 | 2.20 | 0.71 | 1.58 | 4.49 | 0.15 | 0.09 | 0.04 | ||||
| [107,108] * | 7.91 | 0.09 | 2.63 | 0.42 | 1.21 | 4.26 | 0.16 | 0.07 | 0.15 | ||||
| [107,108] * | 7.91 | 0.09 | 2.53 | 0.47 | 1.21 | 4.21 | 0.16 | 0.22 | 0.12 | ||||
| [107,108] * | 7.86 | 0.14 | 2.56 | 0.52 | 1.22 | 4.30 | 0.13 | 0.05 | 0.18 | ||||
| [107,108] * | 7.96 | 0.04 | 2.56 | 0.46 | 1.23 | 0.01 | 4.26 | 0.14 | 0.05 | 0.16 | |||
| [108] * | 7.60 | 0.40 | 2.22 | 0.89 | 1.24 | 4.35 | 0.12 | 0.09 | 0.29 | ||||
| [108] * | 7.95 | 0.05 | 2.64 | 0.54 | 1.04 | 4.22 | 0.16 | 0.07 | |||||
| [108] * | 7.76 | 0.24 | 2.50 | 0.53 | 1.26 | 4.29 | 0.15 | 0.07 | 0.25 | ||||
| [108] * | 7.95 | 0.05 | 2.43 | 0.53 | 1.29 | 4.25 | 0.17 | 0.11 | 0.11 | ||||
| [108] * | 7.78 | 0.22 | 2.41 | 0.53 | 1.34 | 4.28 | 0.17 | 0.07 | 0.30 | ||||
| [108] * | 7.65 | 0.35 | 2.31 | 0.71 | 1.26 | 4.28 | 0.08 | 0.21 | 0.38 | ||||
| [108] * | 7.83 | 0.17 | 2.26 | 0.80 | 1.19 | 0.03 | 4.28 | 0.11 | 0.11 | 0.19 | |||
| [108] * | 7.77 | 0.23 | 2.47 | 0.63 | 1.18 | 4.28 | 0.17 | 0.14 | 0.20 | ||||
| [108] * | 7.80 | 0.20 | 2.66 | 0.53 | 1.09 | 0.01 | 4.29 | 0.12 | 0.10 | 0.07 | |||
| [108] * | 7.48 | 0.52 | 2.61 | 0.65 | 1.06 | 0.01 | 4.33 | 0.12 | 0.12 | 0.19 | |||
| [108] * | 7.73 | 0.27 | 2.43 | 0.65 | 1.20 | 4.28 | 0.18 | 0.11 | 0.14 | ||||
| [108] * | 7.72 | 0.28 | 2.32 | 0.72 | 1.22 | 4.26 | 0.19 | 0.12 | 0.23 | ||||
| [108] * | 7.81 | 0.19 | 2.42 | 0.65 | 1.17 | 0.01 | 4.25 | 0.12 | 0.13 | 0.24 | |||
| [108] * | 7.03 | 0.97 | 0.77 | 1.54 | 2.76 | 0.02 | 5.09 | 0.10 | 0.26 | ||||
| [108] * | 7.01 | 0.99 | 0.82 | 1.51 | 2.59 | 4.92 | 0.08 | 0.11 | 0.58 | ||||
| [108] * | 6.61 | 1.39 | 0.91 | 1.53 | 2.78 | 5.22 | 0.14 | 0.02 | 0.20 | ||||
| Cortijo Archidona | [101] | 7.74 | 0.26 | 2.69 | 0.55 | 1.04 | 4.28 | 0.74 | |||||
| [95] | 7.77 | 0.22 | 2.78 | 0.33 | 1.03 | 4.14 | 0.81 | ||||||
| [118] * | 8.00 | 2.68 | 0.56 | 0.89 | 4.13 | ||||||||
| [119] | 7.71 | 0.22 | 2.73 | 0.52 | 0.86 | 4.11 | 0.11 | 0.83 | |||||
| Tamame | [86] * | 6.64 | 1.36 | 4.07 | 0.29 | 4.36 | 0.09 | 0.10 | |||||
| [86] * | 7.63 | 0.37 | 3.23 | 0.17 | 0.79 | 4.19 | 0.26 | 0.07 | |||||
| [86] * | 7.74 | 0.26 | 3.12 | 0.21 | 0.77 | 4.10 | 0.30 | 0.07 | 0.03 | ||||
| [86] * | 7.58 | 0.42 | 3.07 | 0.23 | 0.88 | 4.18 | 0.32 | 0.11 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Romero, E.; María Manchado, E.; Suárez, M.; García-Rivas, J. Spanish Bentonites: A Review and New Data on Their Geology, Mineralogy, and Crystal Chemistry. Minerals 2019, 9, 696. https://doi.org/10.3390/min9110696
García-Romero E, María Manchado E, Suárez M, García-Rivas J. Spanish Bentonites: A Review and New Data on Their Geology, Mineralogy, and Crystal Chemistry. Minerals. 2019; 9(11):696. https://doi.org/10.3390/min9110696
Chicago/Turabian StyleGarcía-Romero, Emilia, Eva María Manchado, Mercedes Suárez, and Javier García-Rivas. 2019. "Spanish Bentonites: A Review and New Data on Their Geology, Mineralogy, and Crystal Chemistry" Minerals 9, no. 11: 696. https://doi.org/10.3390/min9110696
APA StyleGarcía-Romero, E., María Manchado, E., Suárez, M., & García-Rivas, J. (2019). Spanish Bentonites: A Review and New Data on Their Geology, Mineralogy, and Crystal Chemistry. Minerals, 9(11), 696. https://doi.org/10.3390/min9110696






