Evaluation of the Sorption Potential of Mineral Materials Using Tetracycline as a Model Pollutant
Abstract
1. Introduction
2. Materials and Methods
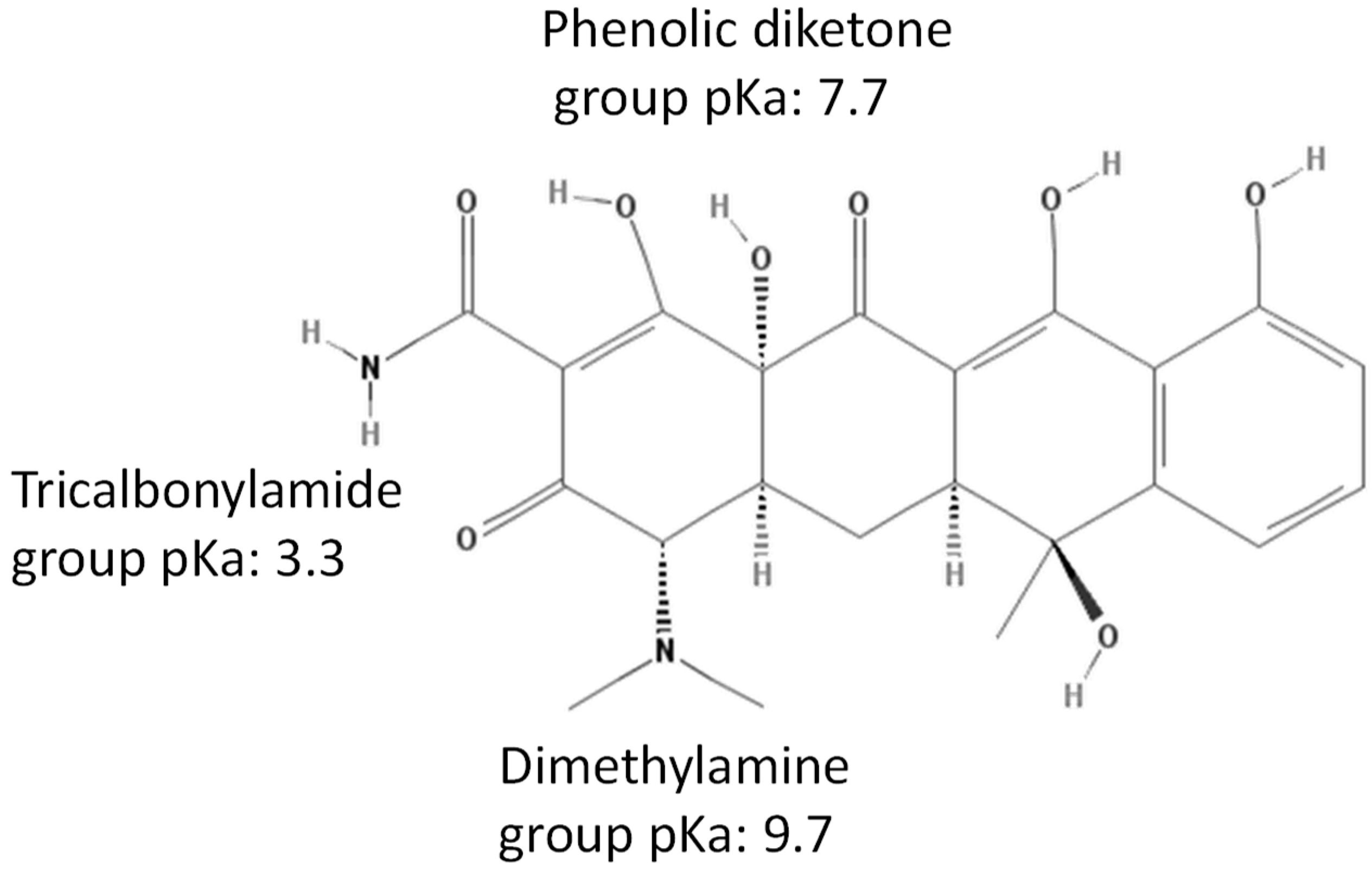
2.1. Tetracycline
2.2. Mineral Sorbents
2.2.1. Clay Minerals
2.2.2. Iron Hydroxides: Ferrihydrite and Goethite
2.2.3. Calcined Sewage Sludge
2.3. Characterization of Materials
2.4. Batch Sorption Experiments
2.4.1. Adsorption Isotherms
2.4.2. Statistical Analysis
3. Results and Discussion
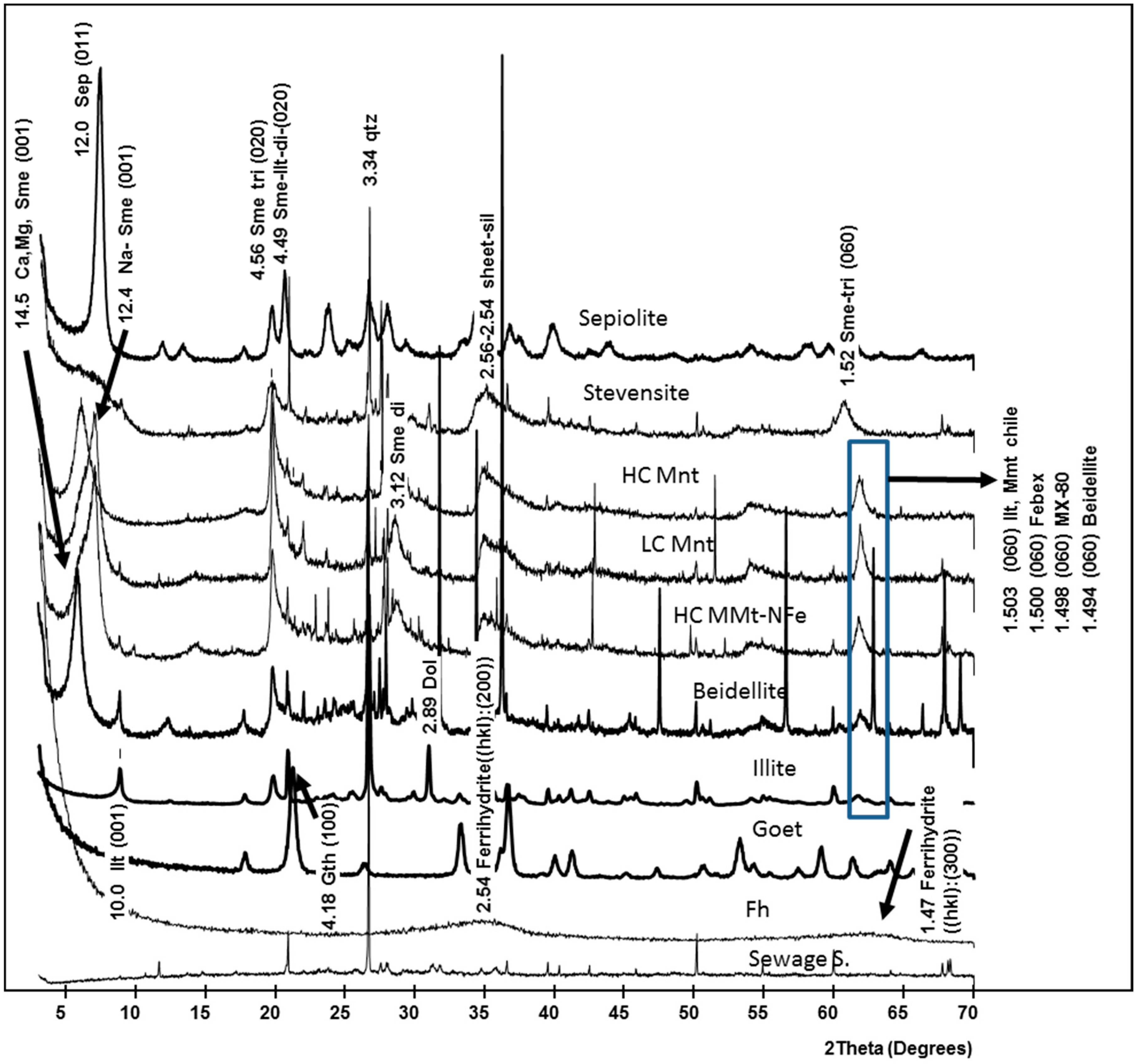
3.1. Physical and Mineralogical Characterization of Synthesized and Natural Sorbent Materials
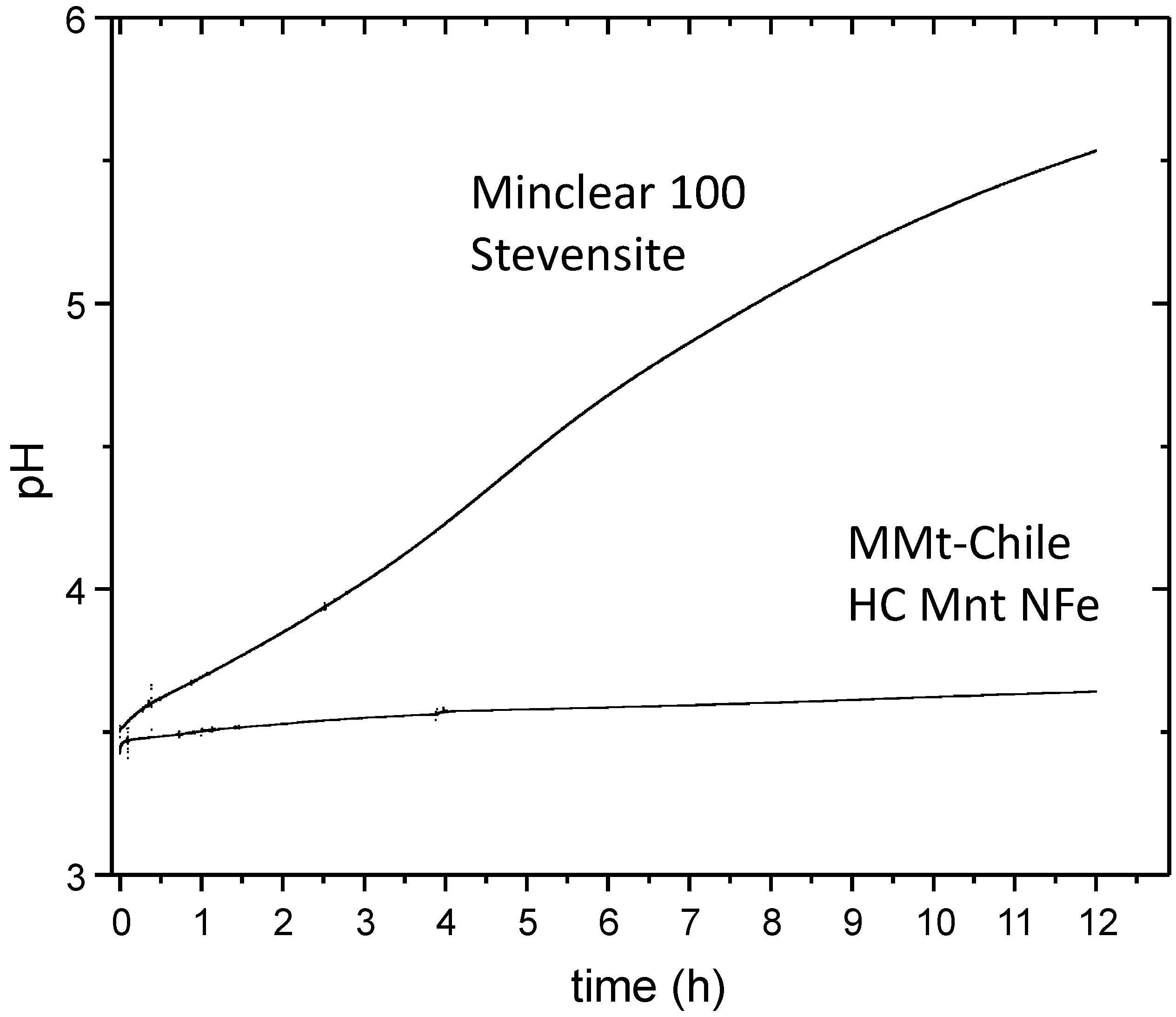
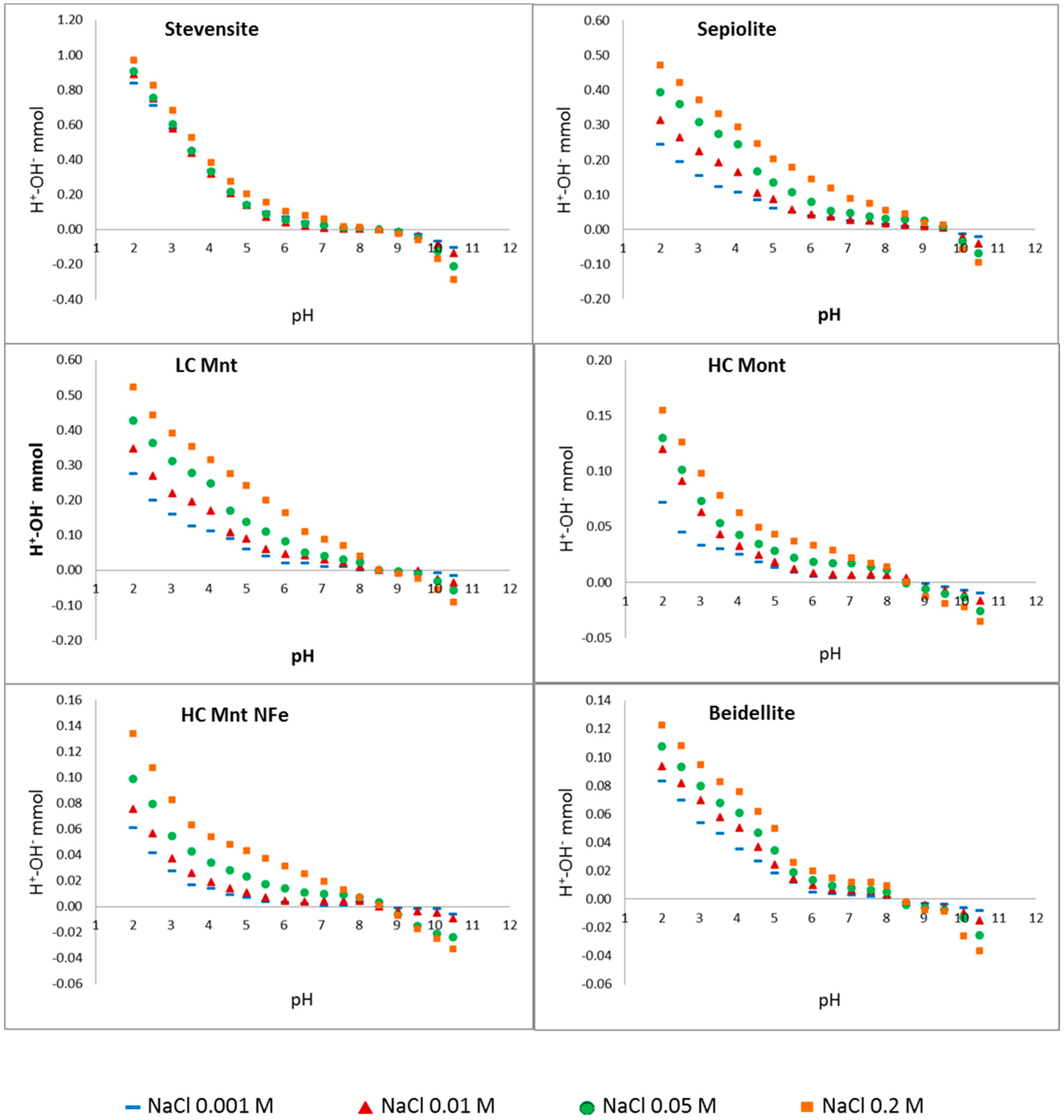
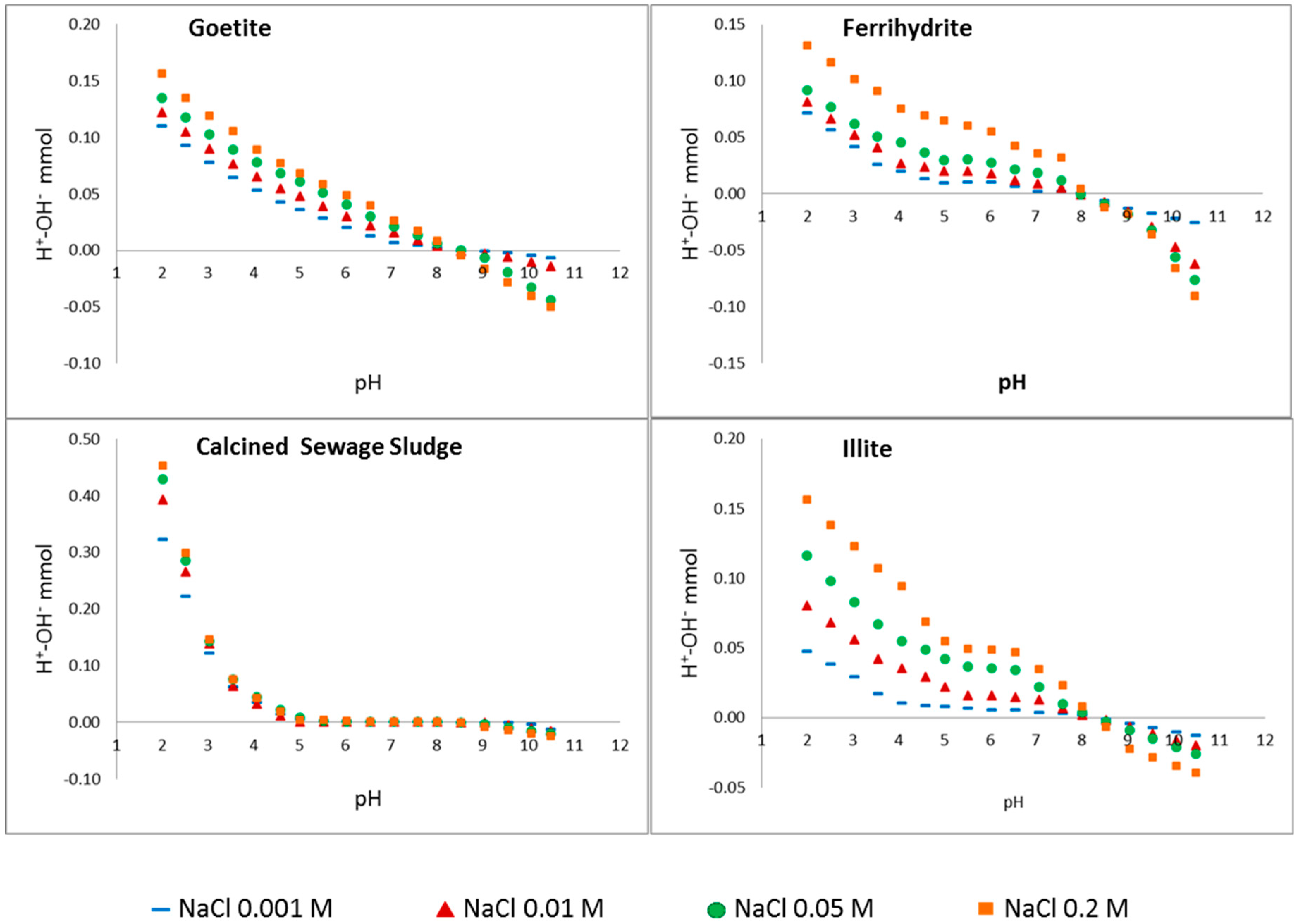
3.2. Point of Zero Salt Effect
3.3. Interaction of Tetracycline with Mineral Sorbents
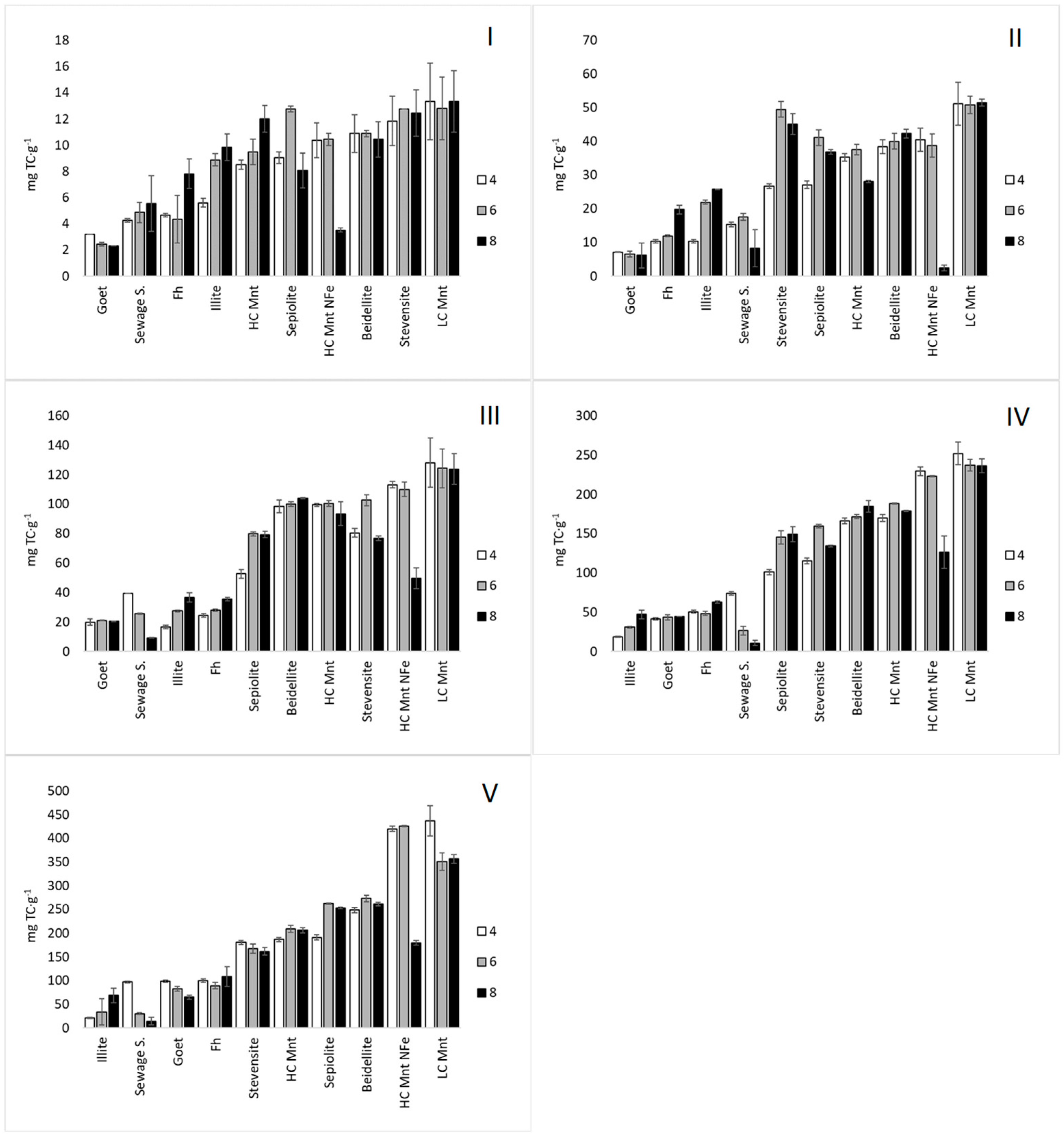
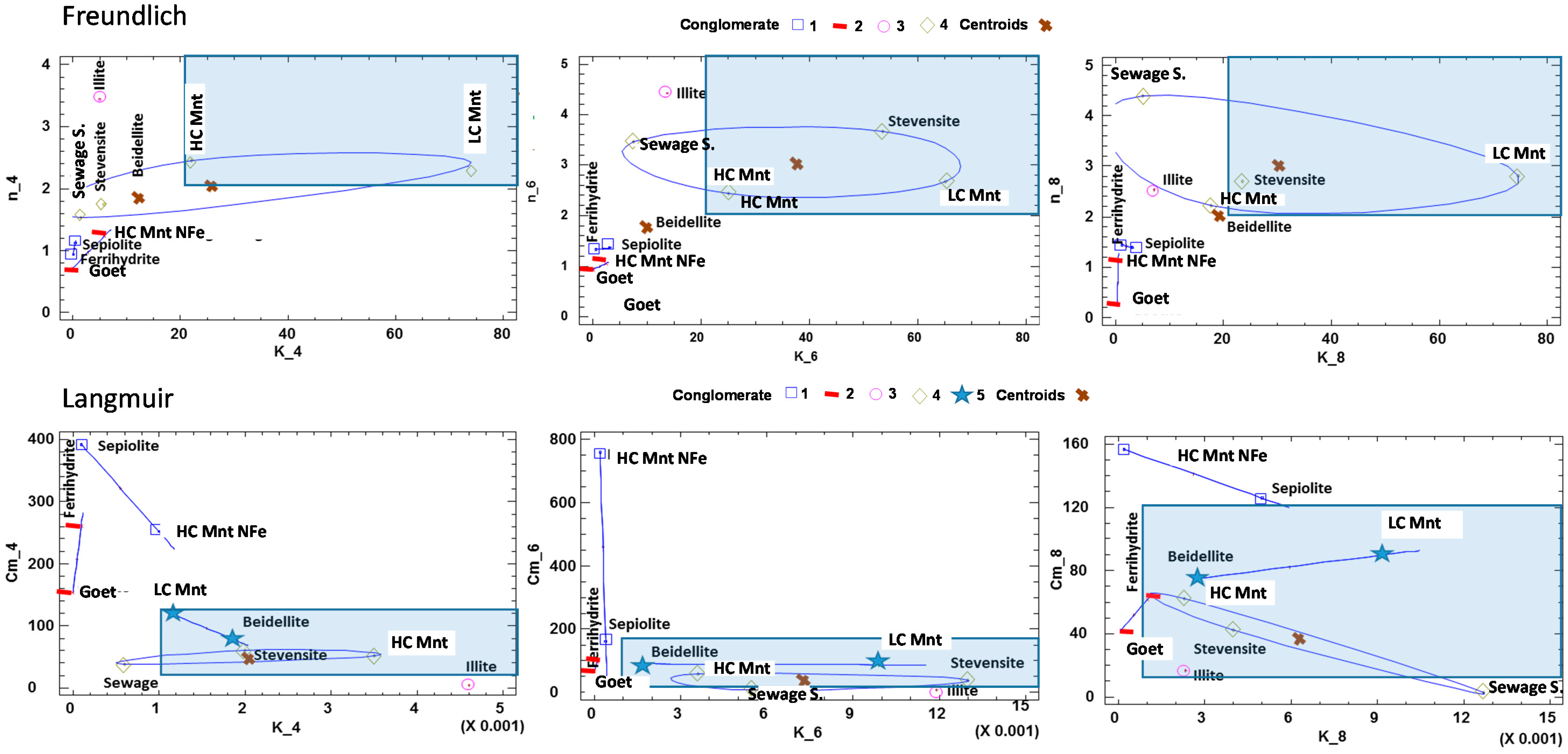
3.3.1. Adsorption Capacity
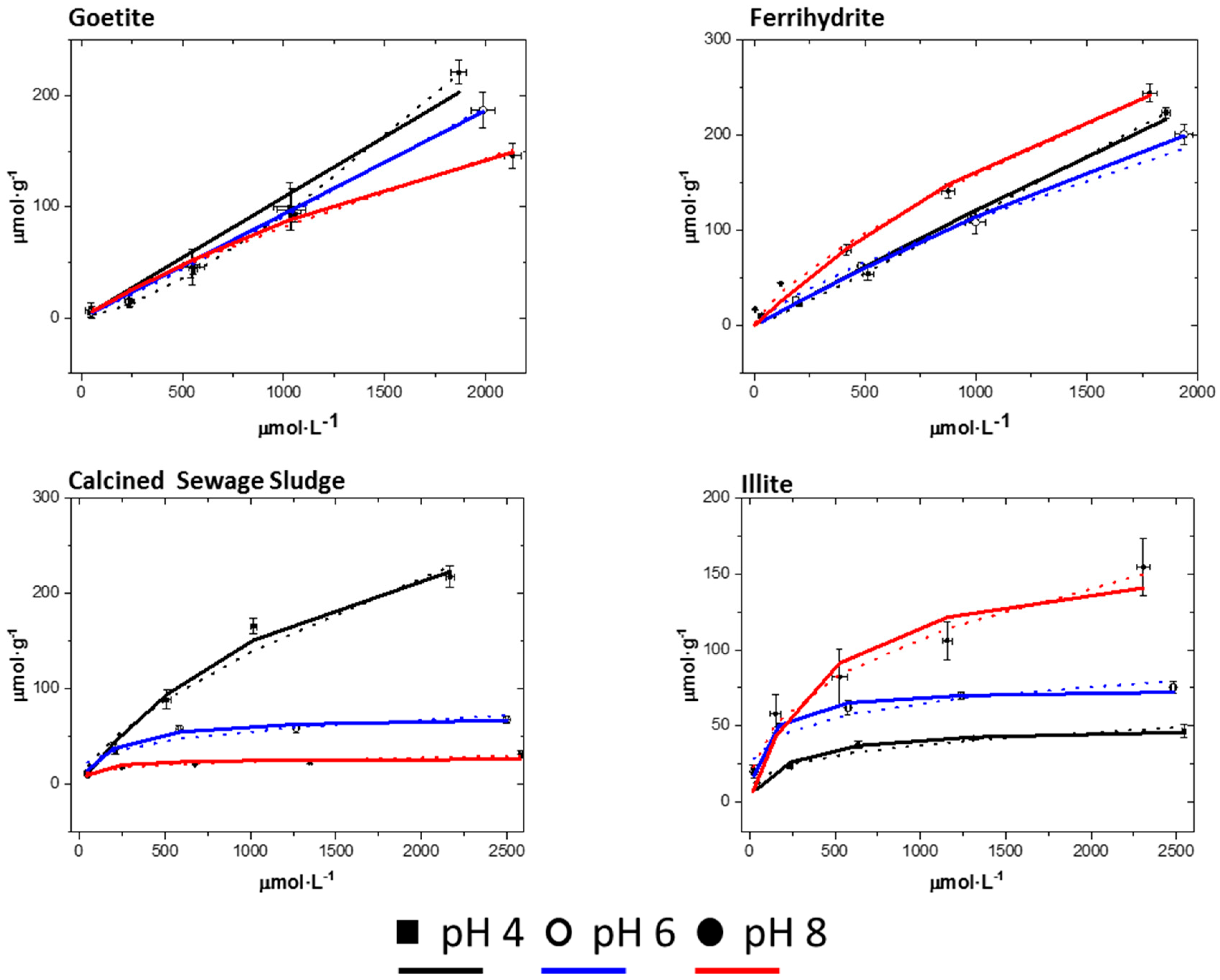
3.3.2. Adsorption Isotherms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- European Medicines Agency, E.S.V.A.C.E. Sales of Veterinary Antimicrobial Agents in 30 European Countries in 2016. (EMA/275982/2018); European Medicines Agency: Amsterdam, The Netherlands, 2018; p. 184. [Google Scholar]
- Stockwell, V.O.; Duffy, B. Use of antibiotics in plant agriculture. Revue Scientifique et Technique-Office International des Epizooties 2012, 31, 199–210. [Google Scholar] [CrossRef]
- Chee-Sanford, J.C.; Mackie, R.I.; Koike, S.; Krapac, I.G.; Lin, Y.F.; Yannarell, A.C.; Maxwell, S.; Aminov, R.I. Fate and transport of antibiotic residues and antibiotic resistance genes following land application of manure waste. J. Environ. Qual. 2009, 38, 1086–1108. [Google Scholar] [CrossRef] [PubMed]
- Osorio, V.; Perez, S.; Ginebreda, A.; Barcelo, D. Pharmaceuticals on a sewage impacted section of a Mediterranean River (Llobregat River, NE Spain) and their relationship with hydrological conditions. Environ. Sci. Pollut. Res. Int. 2012, 19, 1013–1025. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Eichhorn, P.; Jensen, J.N.; Weber, A.S.; Aga, D.S. Removal of antibiotics in wastewater: Effect of hydraulic and solid retention times on the fate of tetracycline in the activated sludge process. Environ. Sci. Technol. 2005, 39, 5816–5823. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.; Cormican, M.; Cummins, E. The effect of conventional wastewater treatment on the levels of antimicrobial-resistant bacteria in effluent: A meta-analysis of current studies. Environ. Geochem. Health 2012, 34, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Michael, I.; Rizzo, L.; McArdell, C.S.; Manaia, C.M.; Merlin, C.; Schwartz, T.; Dagot, C.; Fatta-Kassinos, D. Urban wastewater treatment plants as hotspots for the release of antibiotics in the environment: A review. Water Res. 2013, 47, 957–995. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Schulz, L.; Ackley, C.; Fenske, N. Adsorption of tetracycline on kaolinite with pH-dependent surface charges. Journal of colloid and interface science 2010, 351, 254–260. [Google Scholar] [CrossRef]
- Chang, P.-H.; Li, Z.; Jean, J.-S.; Jiang, W.-T.; Wang, C.-J.; Lin, K.-H. Adsorption of tetracycline on 2:1 layered non-swelling clay mineral illite. Appl. Clay Sci. 2012, 67–68, 158–163. [Google Scholar] [CrossRef]
- Antón-Herrero, R.; García-Delgado, C.; Alonso-Izquierdo, M.; García-Rodríguez, G.; Cuevas, J.; Eymar, E. Comparative adsorption of tetracyclines on biochars and stevensite: Looking for the most effective adsorbent. Appl. Clay Sci. 2018, 160, 162–172. [Google Scholar] [CrossRef]
- Sithole, B.B.; Guy, R.D. Models For Tetracycline In Aquatic Environments. l. lnteraction with Bentonite Clay Systems. Water Air Soil Pollut. 1987, 32, 303–314. [Google Scholar] [CrossRef]
- Chang, P.-H.; Li, Z.; Jiang, W.-T.; Kuo, C.-Y.; Jean, J.-S. Adsorption of tetracycline on montmorillonite: Influence of solution pH, temperature, and ionic strength. Desalinat. Water Treat. 2014, 1–13. [Google Scholar] [CrossRef]
- Aristilde, L.; Lanson, B.; Miehe-Brendle, J.; Marichal, C.; Charlet, L. Enhanced interlayer trapping of a tetracycline antibiotic within montmorillonite layers in the presence of Ca and Mg. J. Colloid Interface Sci. 2016, 464, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-J.; Jia, D.-A.; Sun, R.-J.; Zhu, H.-W.; Zhou, D.M. Adsorption and Cosorption of Tetracycline and Copper(II) on Montmorillonite as Affected by Solution pH. Environ. Sci. Technol. 2008, 42, 3254–3259. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.H.; Li, Z.; Yu, T.L.; Munkhbayer, S.; Kuo, T.H.; Hung, Y.C.; Jean, J.S.; Lin, K.H. Sorptive removal of tetracycline from water by palygorskite. J. Hazard. Mater. 2009, 165, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Borghi, A.A.; Palma, M.S.A. Tetracycline: Production, waste treatment and environmental impact assessment. Brazilian J. Pharmaceut. Sci. 2014, 50, 25–40. [Google Scholar] [CrossRef]
- Ertem, G.; Steudel, A.; Emmerich, K.; Lagaly, G.; Schuhmann, R. Correlation between the extent of catalytic activity and charge density of montmorillonites. Astrobiology 2010, 10, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Kraepiel, A.M.L.; Keller, K.; Morel, F.M.M. On the Acid-Base Chemistry of Permanently Charged Minerals. Environ. Sci. Technol. 1998, 32, 2829–2838. [Google Scholar] [CrossRef]
- Chang, P.-H.; Li, Z.; Jiang, W.-T.; Jean, J.-S. Adsorption and intercalation of tetracycline by swelling clay minerals. Appl. Clay Sci. 2009, 46, 27–36. [Google Scholar] [CrossRef]
- Bandura, L.; Woszuk, A.; Kołodyńska, D.; Franus, W. Application of Mineral Sorbents for Removal of Petroleum Substances: A Review. Minerals 2017, 7, 37. [Google Scholar] [CrossRef]
- Fernández, R.; Ruiz, A.I.; Cuevas, J. The role of smectite composition on the hyperalkaline alteration of bentonite. Appl. Clay Sci. 2014, 95, 83–94. [Google Scholar] [CrossRef]
- Ruiz, A.I.; Fernandez, R.; Sanchez Jimenez, N.; Rodriguez Rastrero, M.; Regadio, M.; de Soto, I.S.; Cuevas, J. Improvement of attenuation functions of a clayey sandstone for landfill leachate containment by bentonite addition. Sci. Total Environ. 2012, 419, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, J.; Leguey, S.; Garralon, A.; Rastrero, M.; Procopio, J.; Sevilla, M.; Jimenez, N.; Abad, R.; Garrido, A. Behavior of kaolinite and illite-based clays as landfill barriers. Appl. Clay Sci. 2008. [Google Scholar] [CrossRef]
- Cuevas, J.; Fernández, R.; Ortega, A.; Ruiz, A.I. Comparison of Alternative Bentonites for Potential use as Buffer and Sealing Materials in the Swiss Concept for Radioactive Waste Disposal. Part 2: Results; Nagra ed. CH-5430: Wettingen, Switzerland, 2014; p. 90. [Google Scholar]
- Cuevas, J.; De La Villa, R.V.; Ramirez, S.; Petit, S.; Meunier, A.; Leguey, S. Chemistry of Mg smectites in lacustrine sediments from the vicalvaro sepiolite deposit, madrid neogene basin (Spain). Clays Clay Miner. 2003, 51, 457–472. [Google Scholar] [CrossRef]
- Álvarez, A.; Santarén, J.; Esteban-Cubillo, A.; Aparicio, P. Current industrial applications of palygorskite and sepiolite. Dev. Clay Sci. 2011, 3, 281–298. [Google Scholar] [CrossRef]
- Guven, N. Bentonites—Clays for molecular engineering. Elements 2009, 5, 89–92. [Google Scholar] [CrossRef]
- Tosca, N.J.; Wright, V.P. Diagenetic pathways linked to labile Mg-clays in lacustrine carbonate reservoirs: A model for the origin of secondary porosity in the Cretaceous pre-salt Barra Velha Formation, offshore Brazil. Geolog. Soc. Lond. Spec. Publicat. 2018, 435, 33–46. [Google Scholar] [CrossRef]
- Benhammou, A.; Yaacoubi, A.; Nibou, L.; Tanouti, B. Adsorption of metal ions onto Moroccan stevensite: Kinetic and isotherm studies. J. Colloid Interface Sci. 2005, 282, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Emmerich, K.; Wolters, F.; Kahr, G.; Lagaly, G. Clay profiling: The classification of montmorillonites. Clays Clay Miner. 2009, 57, 104–114. [Google Scholar] [CrossRef]
- Vázquez, M.; Jiménez-Millán, J. Clay raw materials from the Triassic Red Beds (Northern Jaén, Spain) for making ceramic construction materials. Materiales de Construcción 2004, 54, 5–20. [Google Scholar] [CrossRef]
- Yunta, F.; Di Foggia, M.; Bellido-Diaz, V.; Morales-Calderon, M.; Tessarin, P.; Lopez-Rayo, S.; Tinti, A.; Kovacs, K.; Klencsar, Z.; Fodor, F.; et al. Blood meal-based compound. Good choice as iron fertilizer for organic farming. J. Agricult. Food Chem. 2013, 61, 3995–4003. [Google Scholar] [CrossRef]
- Sánchez, J.M.; Murillo, J.M.; Otero, J.; Plaza, F.; Garralon, G. Thermal valorisation of dried sewage sludge through gasification. In Proceedings of the 10th International Conference on Environmental Science and Technology 2007, Kos Island, Grrece, 5–7 September 2007; pp. A1277–A1286. [Google Scholar]
- Parolo, M.E.; Savini, M.C.; Vallés, J.M.; Baschini, M.T.; Avena, M.J. Tetracycline adsorption on montmorillonite: pH and ionic strength effects. Appl. Clay Sci. 2008, 40, 179–186. [Google Scholar] [CrossRef]
- Guler, U.A.; Sarioglu, M. Removal of tetracycline from wastewater using pumice stone: Equilibrium, kinetic and thermodynamic studies. J. Environ. Health Sci. Eng. 2014, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Liu, X.; He, H.; Wang, L.; Dai, G. Quantitatively modeling soil-water distribution coefficients of three antibiotics using soil physicochemical properties. Chemosphere 2012, 89, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Avisar, D.; Primor, O.; Gozlan, I.; Mamane, H. Sorption of sulfonamides and tetracyclines to montmorillonite clay. Water Air Soil Pollut. 2009, 209, 439–450. [Google Scholar] [CrossRef]
- Ciarlone, A.E.; Fry, B.W.; Ziemer, D.M. Some observations on the adsorption of tetracyclines to glass and plastic labware. Microchem. J. 1990, 42, 250–255. [Google Scholar] [CrossRef]
- Wammer, K.H.; Slattery, M.T.; Stemig, A.M.; Ditty, J.L. Tetracycline photolysis in natural waters: Loss of antibacterial activity. Chemosphere 2011, 85, 1505–1510. [Google Scholar] [CrossRef] [PubMed]
- Bolster, C.H. Microsoft Excel Spreadsheets for Fitting Sorption Data; USDA-ARS: Madison, WI, USA, 2010. [Google Scholar]
- Sposito, G. The Chemistry of Soils, 2nd ed.; Oxford University Press, Inc.: New York, NY, USA, 2008; p. 342. [Google Scholar]
- Masel, R.I. Principles of Adsorption and Reaction on Solid Surfaces; John Wiley & Sons, Inc.: New York, NY, USA, 1996; p. 793. [Google Scholar]
- Brindley, G.W.; Brown, G. Crystal Structures of Clay Minerals and Their X-ray Identification; Mineralogical Society: London, UK, 1980; Volume 5. [Google Scholar]
- Hibino, T.; Tsunashima, A.; Yamazaki, A.; Otsuka, R. Model calculation of sepiolite surface areas. Clays Clay Miner. 1995, 43, 391–396. [Google Scholar] [CrossRef]
- Jambor, J.L.; Dutrizac, J.E. Occurrence and constitution of natural and synthetic ferrihydrite, a widespread iron oxyhydroxide. Chem. Rev. 1998, 98, 2549–2585. [Google Scholar] [CrossRef]
- Dzombak, D.A.; Morel, F.M.M. Surface Complexation Modeling: Hydrous Ferric Oxide; John Wiley & Sons: New York, NY, USA, 1990; p. 416. [Google Scholar]
- Kosmulski, M. pH-dependent surface charging and points of zero charge. IV. Update and new approach. J. Colloid Interface Sci. 2009, 337, 439–448. [Google Scholar] [CrossRef]
- Antelo, J.; Avena, M.; Fiol, S.; Lopez, R.; Arce, F. Effects of pH and ionic strength on the adsorption of phosphate and arsenate at the goethite-water interface. J. Colloid Interface Sci. 2005, 285, 476–486. [Google Scholar] [CrossRef]
- Kriaa, A.; Hamdi, N.; Srasra, E. Acid–Base Chemistry of montmorillonitic and beidellitic-montmorillonitic smectite. Russian J. Electrochem. 2007, 43, 167–177. [Google Scholar] [CrossRef]
- Tombácz, E.; Szekeres, M. Colloidal behavior of aqueous montmorillonite suspensions: The specific role of pH in the presence of indifferent electrolytes. Appl. Clay Sci. 2004, 27, 75–94. [Google Scholar] [CrossRef]
- Tournassat, C.; Davis, J.; Chiaberge, C.; Grangeon, S.; Bourg, U.C. Modeling the Acid-Base Properties of Montmorillonite Edge Surfaces. Environ. Sci. Technol. 2016, 50, 13436–13445. [Google Scholar] [CrossRef] [PubMed]
- Arfaoui, S.; Hamdi, N.; Frini-Srasra, N.; Srasra, E. Determination of point of zero charge of PILCS with single and mixed oxide pillars prepared from Tunisian-smectite. Geochem. Int. 2012, 50, 447–454. [Google Scholar] [CrossRef]
- Christidis, G.E.; Blum, A.E.; Eberl, D.D. Influence of layer charge and charge distribution of smectites on the flow behaviour and swelling of bentonites. Appl. Clay Sci. 2006, 34, 125–138. [Google Scholar] [CrossRef]
- Wilson, M.J.; Wilson, L.; Patey, I. The influence of individual clay minerals on formation damage of reservoir sandstones: A critical review with some new insights. Clay Miner. 2014, 49, 147–164. [Google Scholar] [CrossRef]
- González-Santamaría, D.E.; López, E.; Ruiz, A.I.; Fernández, R.; Ortega, A.; Cuevas, J. Adsorption of phenantrene by stevensite and sepiolite. Clay Miner. 2017. In press. [Google Scholar]
- Pokrovsky, O.S.; Golubev, S.V.; Schott, J. Dissolution kinetics of calcite, dolomite and magnesite at 25 °C and 0 to 50 atm pCO2. Chem. Geol. 2005, 217, 239–255. [Google Scholar] [CrossRef]
- Santos, S.C.R.; Boaventura, R.A.R. Adsorption of cationic and anionic azo dyes on sepiolite clay: Equilibrium and kinetic studies in batch mode. J. Environ. Chem. Eng. 2016, 4, 1473–1483. [Google Scholar] [CrossRef]
- Villalobos, M.; Antelo, J. A unified surface structural model for ferrihydrite: Proton charge, electrolyte binding, and arsenate adsorption. Revista Internacional de Contaminación Ambiental 2011, 27, 139–151. [Google Scholar]
- MacEwan, D.M.C.; Wilson, M.J. Interlayer and intercalation complexes of clay minerals. In Crystal Structures of Clay minerals and their X-Ray Identification; Brindley, G.W., Brown, G., Eds.; Mineralogical Society: London, UK, 1980; Volume 5, pp. 197–248. [Google Scholar]
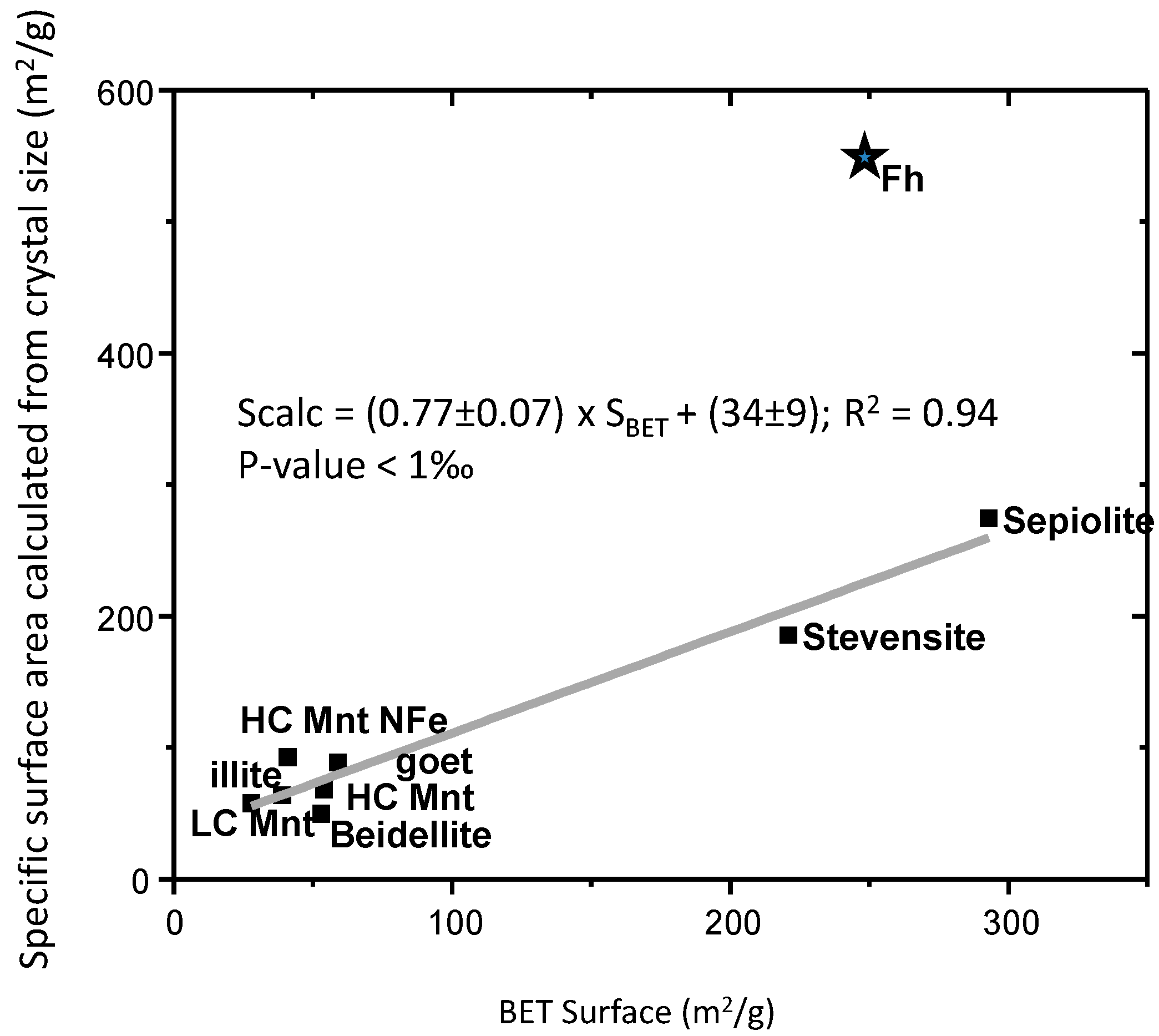

| Material | Cell Parameters (Å) | Cell Volume/Density | Crystal Thickness | % Clay Mineral | BET Surface Area | Calculated Surface Area | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| (hkl) * | a | b | c | (Å3) | g/cm3 | c (001) ** 2:1 (Å) | b (060) ** 2:1 (Å) | wt. % | m2/g | m2/g |
| Ferrihydrite (100) | 5.08 | 5.08 | 9.4 | 210 | 3.96 | 13 | 100 | 253 | 561 | |
| Goethite (100) | 4.596 | 9.957 | 3.021 | 138 | 4.29 | 157 | 100 | 59 | 88 | |
| Illite | 5.20 | 9.02 | 10.0 | 469 | 2.80 | 251 | 197 | 70 | 39 | 64 |
| Beidellite | 5.20 | 8.97 | 14.5 | 676 | 2.80 | 191 | 217 | 60 | 61 | 50 |
| Mnt HC NFe | 5.20 | 9.02 | 13.0 | 610 | 2.80 | 112 | 175 | 90 | 47 | 93 |
| Mnt LC | 5.20 | 8.99 | 12.5 | 584 | 2.80 | 107 | 280 | 90 | 34 | 58 |
| Mnt HC | 5.20 | 9.00 | 14.5 | 679 | 2.80 | 54 | 249 | 95 | 54 | 68 |
| Stevensite | 5.30 | 9.15 | 14.5 | 703 | 2.60 | 19 | 94 | 90 | 223 | 186 |
| Sepiolite (110) | 5.28 | 26.95 | 13.4 | 1902 | 2.40 | 141 | 95 | 293 | 274 | |
| Material | CEC (cmol(+)·kg−1) | PZSE (pH) |
|---|---|---|
| Illite (Dolomite < 5%) | 13(1) | 8.2 ** |
| Beidellite | 61(2) | 8,3 ** |
| Stevensite (Dolomite < 5%) | 60(3) | 8.2 ** |
| Sepiolite | 20(3) | 9.8 ** |
| LC Mnt | 83(4) | 8.3 ** |
| HC Mnt | 102(4) | 8.4 ** |
| HC Mnt NFe | 96(4) | 8.3 ** |
| Sewage S. | N.D. | 5–8.5 ** |
| Fh | N.D. | 8.0 ** |
| Goet | N.D. | 8.3 ** |
| Mineral Sorbent | Kf (L·g−1) | n | R2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| pH 4 | pH 6 | pH 8 | pH 4 | pH 6 | pH 8 | pH 4 | pH 6 | pH 8 | |
| Stevensite | 5.79 | 53.53 | 1.75 | 1.75 | 3.67 | 2.70 | 0.967 | 0.928 | 0.970 |
| Sepiolite | 0.50 | 3.03 | 1.12 | 1.12 | 1.37 | 1.39 | 0.997 | 0.995 | 0.997 |
| LC Mnt | 73.3 | 65.09 | 2.36 | 2.36 | 2.68 | 2.82 | 0.964 | 0.953 | 0.971 |
| HC Mnt | 22.0 | 25.08 | 2.45 | 2.45 | 2.44 | 2.22 | 0.833 | 0.830 | 0.835 |
| HC Mnt N Fe | 5.90 | 2.41 | 1.24 | 1.24 | 1.05 | 1.11 | 0.965 | 0.983 | 0.889 |
| Beidellite | 12.1 | 10.48 | 1.88 | 1.88 | 1.75 | 2.06 | 0.978 | 0.990 | 0.963 |
| Goet | 0.01 | 0.059 | 0.72 | 0.72 | 0.94 | 0.29 | 0.998 | 0.998 | 0.970 |
| Fh | 0.07 | 0.62 | 0.94 | 0.94 | 1.33 | 1.44 | 0.998 | 0.995 | 0.992 |
| Sewage S. | 1.55 | 7.56 | 1.54 | 1.54 | 3.46 | 4.39 | 0.966 | 0.860 | 0.956 |
| Illite | 5.04 | 13.68 | 3.44 | 3.44 | 4.43 | 2.54 | 0.957 | 0.929 | 0.987 |
| Mineral Sorbent | K (L·μmol−1) × 10−3 | * Cm (cmol·kg−1) | CEC (cmol·kg−1) | R2 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| pH 4 | pH 6 | pH 8 | pH 4 | pH 6 | pH 8 | pH 4 | pH 6 | pH 8 | ||
| Stevensite | 2.00 | 13.0 | 4.00 | 60.7 | 39.1 | 42.6 | 60 | 0.971 | 0.979 | 0.965 |
| Sepiolite | 0.10 | 0.40 | 5.00 | 392 | 160 | 125 | 20 | 0.997 | 0.993 | 0.998 |
| LC Mnt | 1.20 | 9.90 | 9.20 | 116 | 87.6 | 89.8 | 83 | 0.997 | 0.999 | 0.998 |
| HC Mnt | 3.50 | 3.60 | 2.30 | 52.0 | 58.6 | 62.1 | 102 | 0.951 | 0.945 | 0.901 |
| HC Mnt N Fe | 1.00 | 0.20 | 0.20 | 251 | 759 | 156 | 96 | 0.978 | 0.986 | 0.905 |
| Beidellite | 1.90 | 1.60 | 2.70 | 76.2 | 89.7 | 74.8 | 61 | 0.996 | 0.996 | 0.999 |
| Goet | 0.00 | 0.00 | 0.10 | 153 | 67.8 | 42.1 | n.d. | 0.970 | 0.998 | 0.982 |
| Fh | 0.10 | 0.10 | 1.00 | 261 | 95.5 | 61.6 | n.d. | 0.994 | 0.998 | 0.991 |
| Sewage S. | 0.60 | 5.50 | 12.7 | 38.6 | 7.20 | 2.72 | n.d. | 0.989 | 0.983 | 0.742 |
| Illite | 4.60 | 11.9 | 2.30 | 4.96 | 7.47 | 16.8 | 13 | 0.975 | 0.986 | 0.932 |
| Intensity/Capacity | Optimum Properties (1) | High Intensity at Low TC Concentrations | |
|---|---|---|---|
| Low pH | Neutral pH (6–8) | ||
| Freundlich n > 2; K > 10; | LC Mnt HC Mnt | Stevensite, LC Mnt, HC Mnt | Illite, Sewage S. (pH 6–8) |
| Langmuir Cm(10–120 cmol/kg) (2); Klangmuir > 1 | MX-80, Beidellite, HC Mnt, Stevensite | LC Mnt, Beidellite, HC Mnt, Stevensite | Sepiolite, Fh pH (8) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuevas, J.; Dirocie, N.; Yunta, F.; García Delgado, C.; González Santamaría, D.E.; Ruiz, A.I.; Fernández, R.; Eymar, E. Evaluation of the Sorption Potential of Mineral Materials Using Tetracycline as a Model Pollutant. Minerals 2019, 9, 453. https://doi.org/10.3390/min9070453
Cuevas J, Dirocie N, Yunta F, García Delgado C, González Santamaría DE, Ruiz AI, Fernández R, Eymar E. Evaluation of the Sorption Potential of Mineral Materials Using Tetracycline as a Model Pollutant. Minerals. 2019; 9(7):453. https://doi.org/10.3390/min9070453
Chicago/Turabian StyleCuevas, Jaime, Nisael Dirocie, Felipe Yunta, Carlos García Delgado, Daniel E. González Santamaría, Ana Isabel Ruiz, Raúl Fernández, and Enrique Eymar. 2019. "Evaluation of the Sorption Potential of Mineral Materials Using Tetracycline as a Model Pollutant" Minerals 9, no. 7: 453. https://doi.org/10.3390/min9070453
APA StyleCuevas, J., Dirocie, N., Yunta, F., García Delgado, C., González Santamaría, D. E., Ruiz, A. I., Fernández, R., & Eymar, E. (2019). Evaluation of the Sorption Potential of Mineral Materials Using Tetracycline as a Model Pollutant. Minerals, 9(7), 453. https://doi.org/10.3390/min9070453










