1. Introduction
In the early 1900s, apatite and phlogopite were recovered from dozens of small mines in the Mont-Laurier–Bancroft corridor in the Western Quebec/Eastern Ontario portion of the Grenville Province [
1,
2]. These minerals are found in a calcite matrix, and an association with ultrabasic rocks, pyroxenite or dunite, is prominent in most of these. Among all these deposits, the Parker mica mine, located near Notre-Dame-du-Laus, Quebec (46°05.5’ N, 75°39.7’ W), is among the rare ones which display strikingly euhedral crystals of black spinel associated with dunite. The isolated crystals of spinel are found in a matrix of white to pinkish calcite, along with euhedral forsterite, phlogopite, and apatite [
3]. Van Velthuizen [
4] described the olivine morphologically and compositionally. The Parker mine occurs in what he called “a green rock complex” exposed in an area dominated by marble (his Figure 2). We have characterized one sample of green rock in detail. It consists mostly of an accumulation of magnesian olivine (∼65 vol. %) and spinel crystals. In spite of high values of Mg# (100Mg/(Mg + Fe
2+ + Mn)), close to 90, the forsterite is devoid of Ni and Cr. The presence of an ultrabasic rock in association with apatite and phlogopite mineralization is enigmatic, and its origin has been debated in the past.
In this article, we discuss the geological setting and document the age of the Parker phlogopite mine in the Central Metasedimentary Belt (CMB) of the Grenville Province. We describe the dunite texturally and mineralogically on the basis of large-area image mosaics and 92 maps of key areas in two polished thin sections. We have identified the constituent minerals with the help of their energy-dispersion spectra. We were able to reconstruct the order of primary crystallization and the appearance of secondary minerals as the dunite cooled. Then we focused on a euhedral crystal of spinel enclosed in calcite. We scrutinized the large-area image mosaics and 60 maps of selected areas in the spinel crystal, and were able to recognize early-formed largely anhydrous minerals, and others that represent lower-temperature minerals formed in a hydrous environment. Our observations led to a reconstruction of what transpired at the Parker mine in the late Mesoproterozoic in this portion of the Grenville Province.
6. The Minerals Encountered in the Spinel-Rich Dunite
Our representative specimen of Van Velthuizen’s “green rock complex” consists of fine-grained dunite, i.e., a forsterite-dominant olivine-rich rock locally containing close to 40 vol. % of spinel (
Table 1,
Table 2 and
Table 3,
Figure 3 and
Figure 4). The olivine-group mineral is forsterite of composition Fo
90. Although an apatite-group mineral is present, the olivine is poor in phosphate. Lizardite has a higher Mg# value than the olivine (
Table 1). The greenish spinel commonly shows incipient development of exsolution-induced domains of magnetite. The Mg# value is close to 81.7 (
Table 2). The Mg# value of diopside, magnesio-hastingsite, and phlogopite is greater than that of the olivine (
Table 1 and
Table 3), which indicates that they crystallized from the melt before the olivine. Textural data are presented below that confirm this finding in the case of phlogopite. Ilmenite, an apatite-group mineral, and dolomite are minor primary constituents. Secondary minerals have also been identified in our examination of 92 key areas in the thin sections, as documented below. Calcite and dolomite are distributed interstitially. We consider this ultrabasic assemblage a cumulate, formed by the rapid gravitational settling of dense minerals (ρ of forsterite 3.3, spinel 3.6 g/cm
3, respectively) in a carbonate growth-medium. There is evidence that the crystal mush in the dunitic cumulate has recrystallized to form an aggregate of polygonal grains that share common boundaries and define 120° triple junctions. As in all cumulates, this recrystallization involved solution in and redeposition from the interstitial low-density, low-viscosity liquid before it was buoyantly squeezed out [
15]. It is common for spinel crystals growing in a melt to link into chains [
16]. Conjoined crystals of spinel in
Figure 3 and
Figure 4 seem to have settled in this way into the mush. The dunite contains heterogeneously distributed spinel grains, some areas being almost devoid of spinel. Note the common presence of spinel in forsterite grains. The converse also is found, though less commonly (
Figure 4 and
Figure 5).
The analytical data were acquired with a JEOL-8900L electron microprobe (JEOL USA, Inc., Peabody, MA, USA) at McGill University, operated at 20 kV, 50 nA, with a beam 5 μm in diameter (columns 1, 2), at 15 kV, 20 nA, 10 μm (columns 3, 4), and 20 kV, 20 nA, 5 μm (column 6). The following standards were used: forsterite (Si, Mg, Fe), spessartine (Mn), orthoclase or pyrope (Al), chromite (Cr), NiO (Ni), diopside (Ca), vanadinite (Cl), and fluorapatite (P). The ZAF method of correction was employed. Sought in the forsterite but not found: Al, Cr, and Ni. Mg# is defined as 100Mg/(Fe + Mn + Mg);
n: number of analyses made. Columns 1–4: forsterite, this work; 5: forsterite [
4], 6: lizardite, this work. Lizardite was identified by X-ray diffraction [
4].
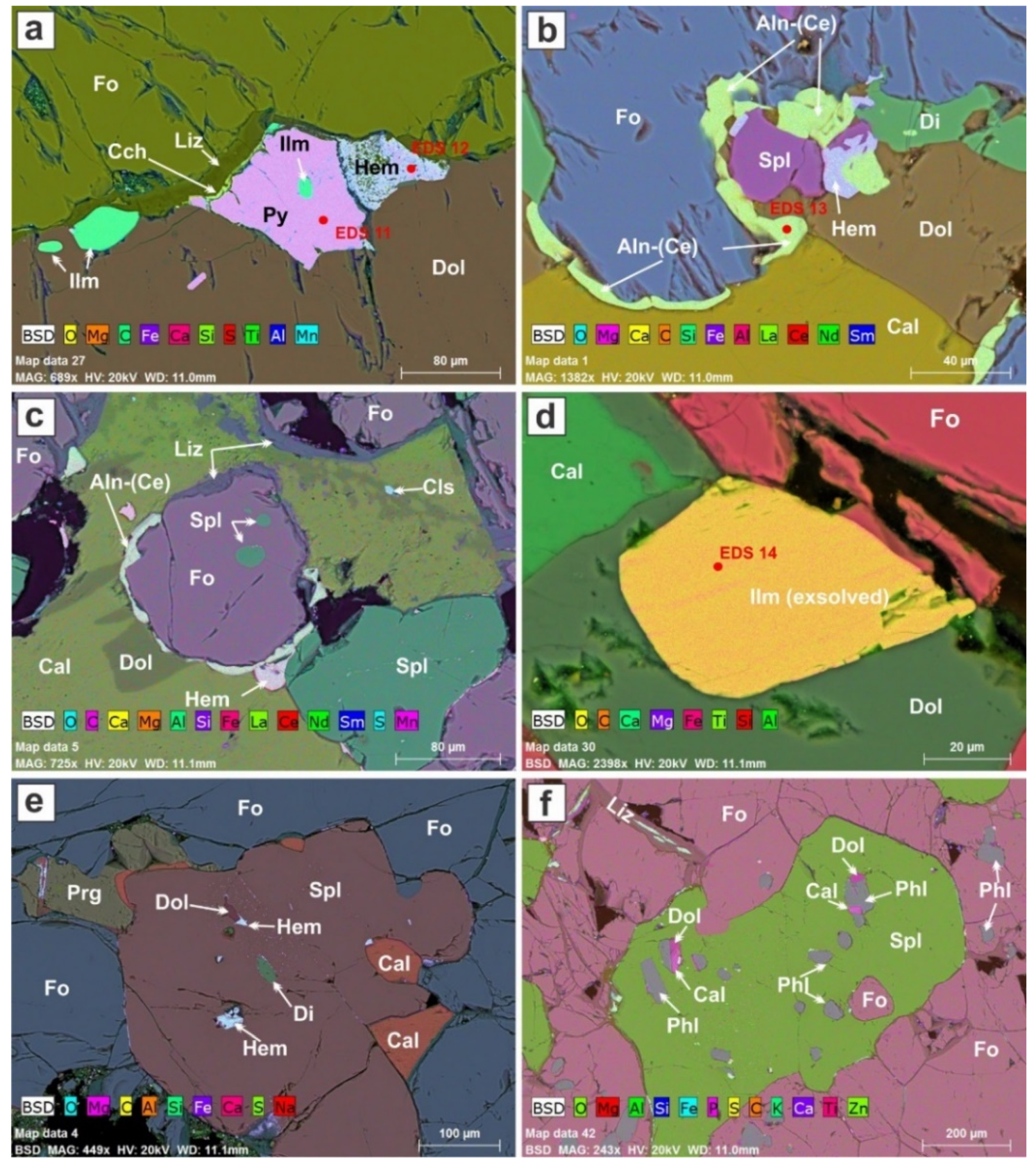
The minerals in the selected high-resolution areas have been identified on the basis of their energy-dispersive spectrum (EDS). Forsterite and spinel are both present in each of the six areas illustrated (
Figure 4a–f), but proportions are quite variable. Each area contains one or two carbonate minerals in interstices, again in quite variable proportions. Calcite and dolomite share a boundary in
Figure 4a; both crystallized from a single (Ca,Mg) carbonate melt. The calcite invariably contains a small amount of Mg, but in the area labeled Mg-rich calcite in
Figure 4a, the Mg peak in the EDS spectrum is noticeably enhanced (
Figure S4). In this suite, it is clear that not all the dolomite crystallized directly from a melt. In
Figure 4d,e, for example, dolomite developed by the replacement of calcite, presumably by reaction of the melt with adjacent forsterite and perhaps also spinel. In
Figure 4b, the carbonate melt crystallized magnesite and dolomite, which share a boundary. This is one of two occurrences of magnesite that we encountered. The lack of a uniform interstitial carbonate assemblage is symptomatic of a poorly blended melt phase.
Among the minor “primary” constituents encountered in the dunite are phlogopite, diopside, and a magnesio-hastingsitic amphibole (
Table 3). Phlogopite nucleated early in the carbonate melt, as it is entrapped in forsterite (
Figure 4e) and spinel (
Figure 5f). It was encountered in 28 of the 92 thin-section areas investigated. Diopside invariably forms a ribbon-like interface between forsterite and interstitial carbonate (
Figure 4a,d,e and
Figure 5b). Note that diopside does not occur at all forsterite–carbonate interfaces. We see evidence of such a reaction in 17 of the 92 areas investigated. Even more rarely encountered, only in nine cases of the 92, is a magnesio-hastingsitic amphibole. In
Figure 4f, a relatively large crystal of this amphibole is judged to have formed relatively late, as it poikilitically encloses forsterite, spinel, calcite with a dolomitized border, and dolomite. Apparently homogeneous ilmenite occurs interstitially, associated with pyrite (
Figure 5a).
Also present in the dunitic cumulate are hematite and a serpentine-group mineral inferred to be lizardite (present in 42 and 28 areas of the 92 investigated, respectively). As is clear in
Figure 4 and
Figure 5, these secondary minerals are volumetrically rather minor. Interstitial ilmenite is visibly exsolved in
Figure 5d. Allanite-(Ce) locally decorates forsterite–carbonate grain boundaries, along with lizardite, hematite, and clinochlore (
Figure 5a–c). They were probably coeval, and filled contraction-induced cracks. A speck of celestine is found in secondary dolomite (
Figure 5d).
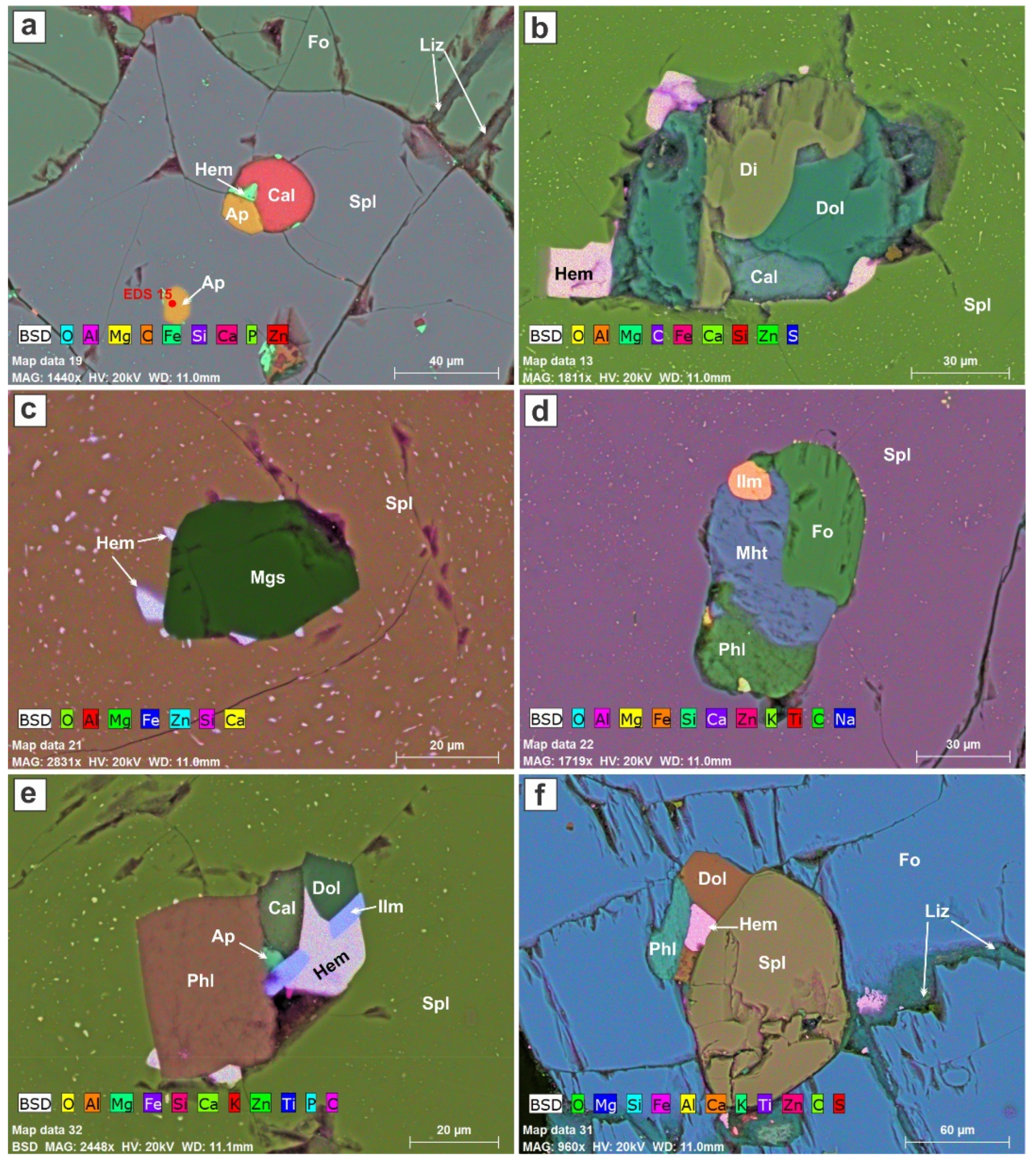
High-magnification images reveal the presence of polymineralic globules in spinel and forsterite (
Figure 5c,e,f and
Figure 6). The minerals seem to have grown in situ in these globules after trapping in the host mineral prior to gravitational settling. In
Figure 6a, the shape of the apatite and calcite conforms to the globule boundary; the two minerals share a border, which suggests that they grew together, possibly from a single droplet of melt. We believe that hematite nucleated in the carbonate melt at that stage and became attached to the globule. A second globule in
Figure 6a seems to consist only of apatite + hematite, but recall that this is a slice through a three-dimensional object.
Hematite flakes also appear stuck on the polymineralic globules trapped in spinel in
Figure 6b,c,e. We contend that the flakes of hematite, up to 20 μm across, appeared at the same time as the dusting of magnetite specks in the spinel. These specks are attributed to oxidation-induced exsolution of the spinel solid-solution. These iron oxide specks form “halos” around the globules (see also Atlas 5 BBV datasets).
The polymineralic inclusion in
Figure 6b consists of calcite, dolomite, diopside, and flakes of hematite. The first three minerals could have formed from a droplet of silicocarbonatitic magma.
Figure 6e also contains calcite and dolomite, along with apatite, phlogopite, ilmenite, and a flake of hematite. The association is recurrent, and again could have crystallized in situ from a droplet of melt.
Figure 6d shows a cluster of forsterite, ilmenite, magnesio-hastingsite, and phlogopite entrapped in spinel. Forsterite thus was present in the carbonate melt as the spinel was growing.
Figure 6f shows a granule of spinel on which dolomite and phlogopite have nucleated, along with a flake of hematite, before being entrapped in forsterite.
7. The Minerals Encountered in the Carbonate Dikes and Segregations
The specimen of marble that we chose to investigate is representative; it was specifically chosen because the centimetric euhedral crystal of spinel trapped visible inclusions of calcite. It has a much coarser grain-size (
Figure 2b,
Figure 7a, and
Figure S3, see also Atlas 5 BBV datasets) than the greenish dunite rock. Oxidation-induced exsolution lamellae of magnetite and specks of hematite or magnetite are found in the spinel (
Figure 7b,c). The specks form “halos” around the polymineralic inclusions (
Figure 7c). Idiomorphic crystals of forsterite [
4] and phlogopite greater than 10 cm across are present in the carbonate dikes.
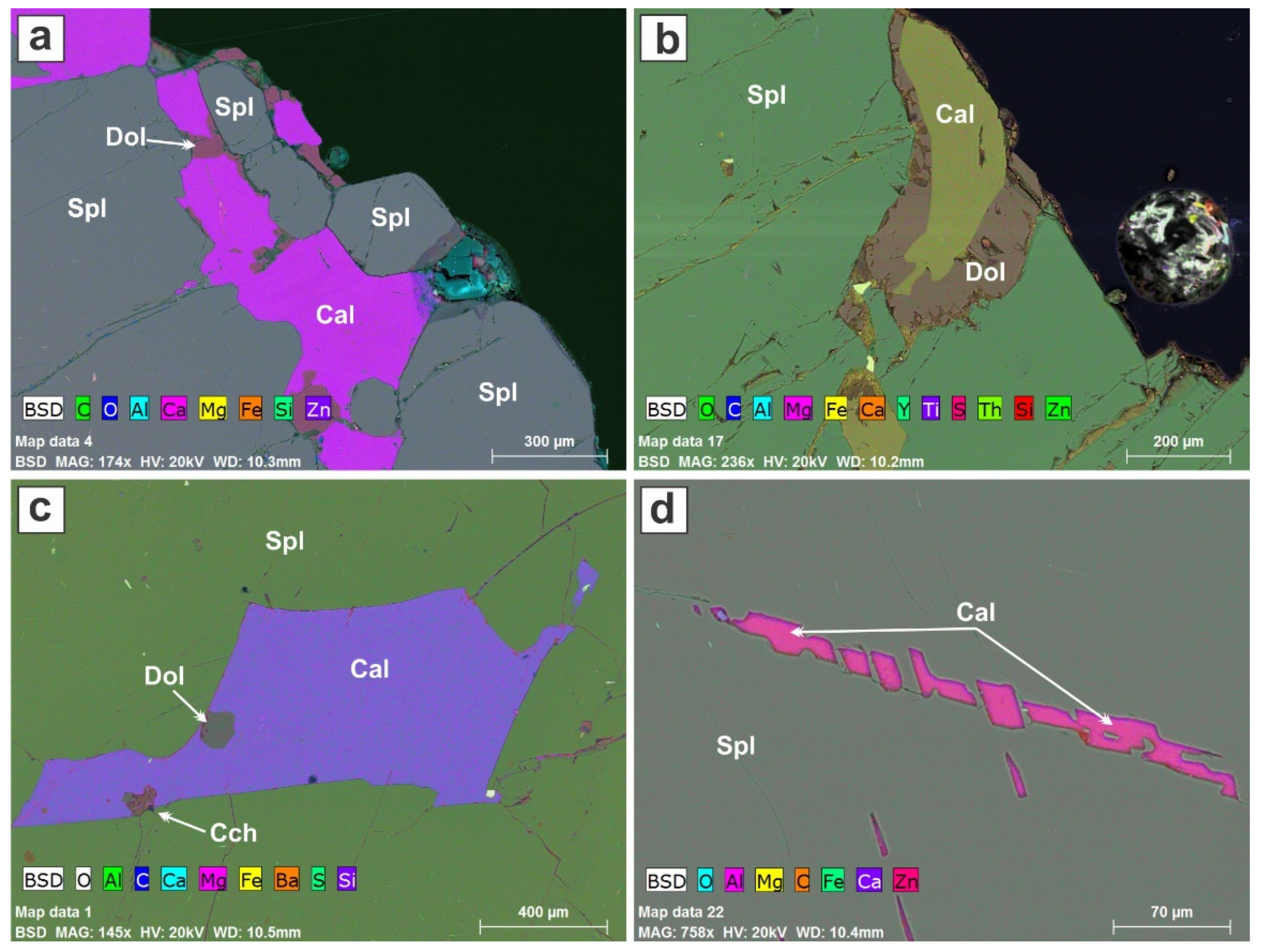
The outer boundary of the investigated crystals of spinel contains remnants of the calcite matrix from which it grew (
Figure 8a,b). Smaller crystals of spinel can also be observed in this calcite (
Figure 8a). A large inclusion of calcite can be found in the center of the crystal (
Figure 8c and
Figure S3). Close to the outer perimeter of the spinel crystal that has escaped oxidation-induced exsolution of magnetite, monomineralic calcite inclusions do occur; some form groups in a string-like arrangement (
Figure 8d). Although these might be construed to be aligned individual rhombs trapped during growth, we prefer to interpret these as melt inclusions filling negative crystal cavities in the spinel. We are influenced by the presence of conformable films of calcite below the main string, perhaps indicative of tensional gashes.
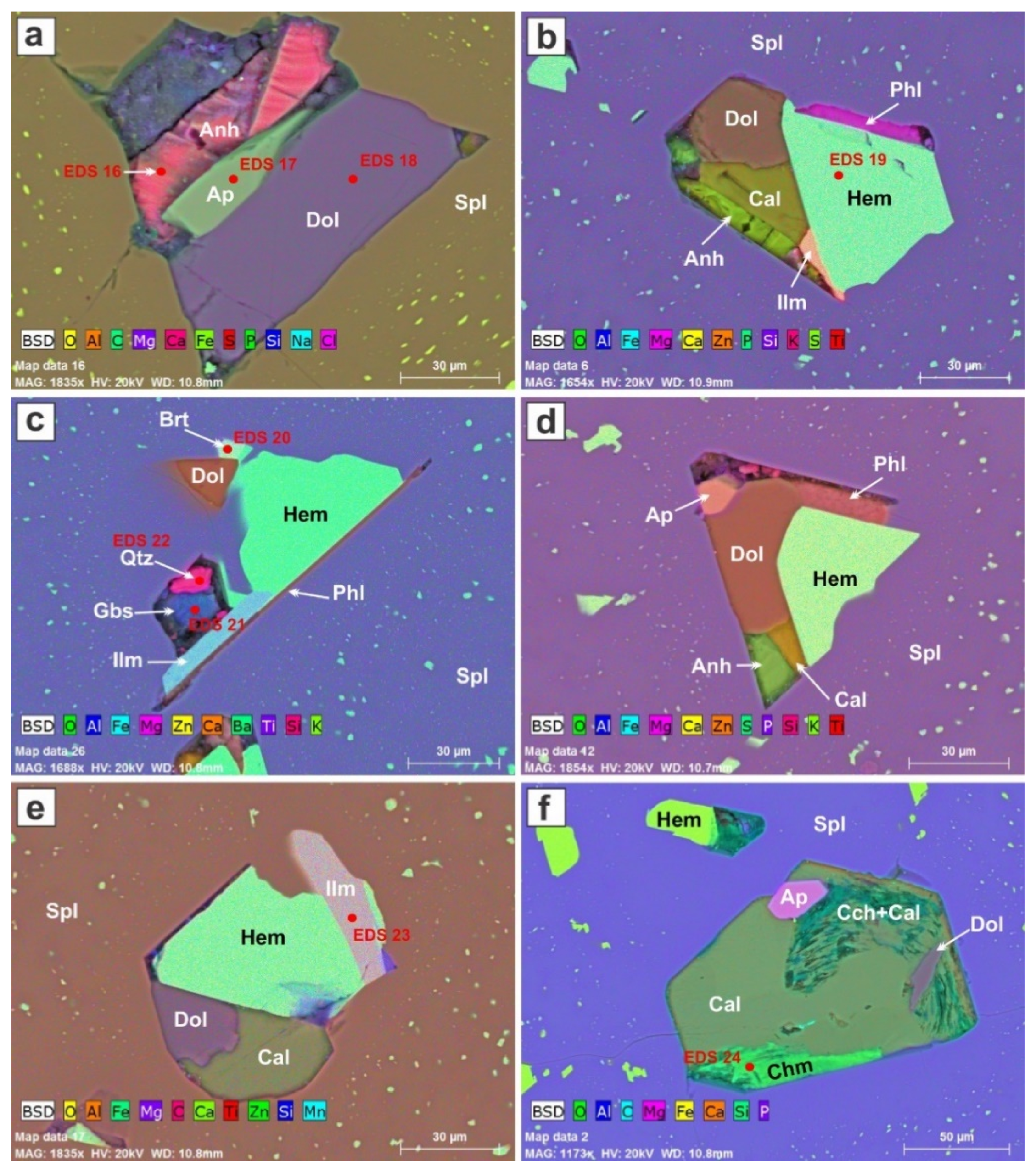
We studied 60 polymineralic inclusions trapped by the spinel crystal, and identified the minerals present on the basis of their energy-dispersive spectra. Compared to the assemblages encountered in the cumulates, we note that diopside and forsterite are absent among the minerals trapped in the spinel. The early-formed minerals (
Figure 9) consist of ilmenite, phlogopite, an apatite-group mineral, anhydrite, calcite and dolomite.
The inclusions in
Figure 9 are relatively pristine. Other than the hematite flakes, the minerals seem to have crystallized from a single melt within the confines of the globule’s volume, which in some cases has a morphology controlled by the spinel host (
Figure 9a,d). One gets the impression that the hematite nucleated independently in the melt, and continued to grow once trapped in the spinel by late dissolution of specks of hematite within radius of 10 μm from the included clusters.
Figure 9b,d contain a silicocarbonate assemblage: calcite–dolomite–anhydrite–phlogopite–ilmenite and calcite–dolomite–anhydrite–phlogopite–apatite.
Figure 9a is devoid of a silicate. Recall that these images illustrate random cuts in three-dimensional objects; we are thus not sampling the minerals in their correct volumetric proportions.
In
Figure 9c, a plate of phlogopite seems to have acted as a substrate on which ilmenite, quartz, gibbsite (or corundum), and then hematite nucleated. Both the quartz and gibbsite (or corundum) seem to have been partially etched, as there is empty space around them. The presence of quartz in a system containing forsterite is a clear indication of metastability. Hematite and ilmenite crystals growing on a crystal face of spinel seem to have acted as the site at which droplets of carbonate melt can attach.
Figure 9e shows an inclusion in which calcite and dolomite crystallized in a globular shape, whereas the subhedral hematite and ilmenite crystals fill the other half of the inclusion. Calcite containing a subhedral inclusion of apatite and a domain of dolomite (
Figure 9f) also has been etched, and now contains areas of clinochlore and chamosite deposited from the ambient fluid medium.
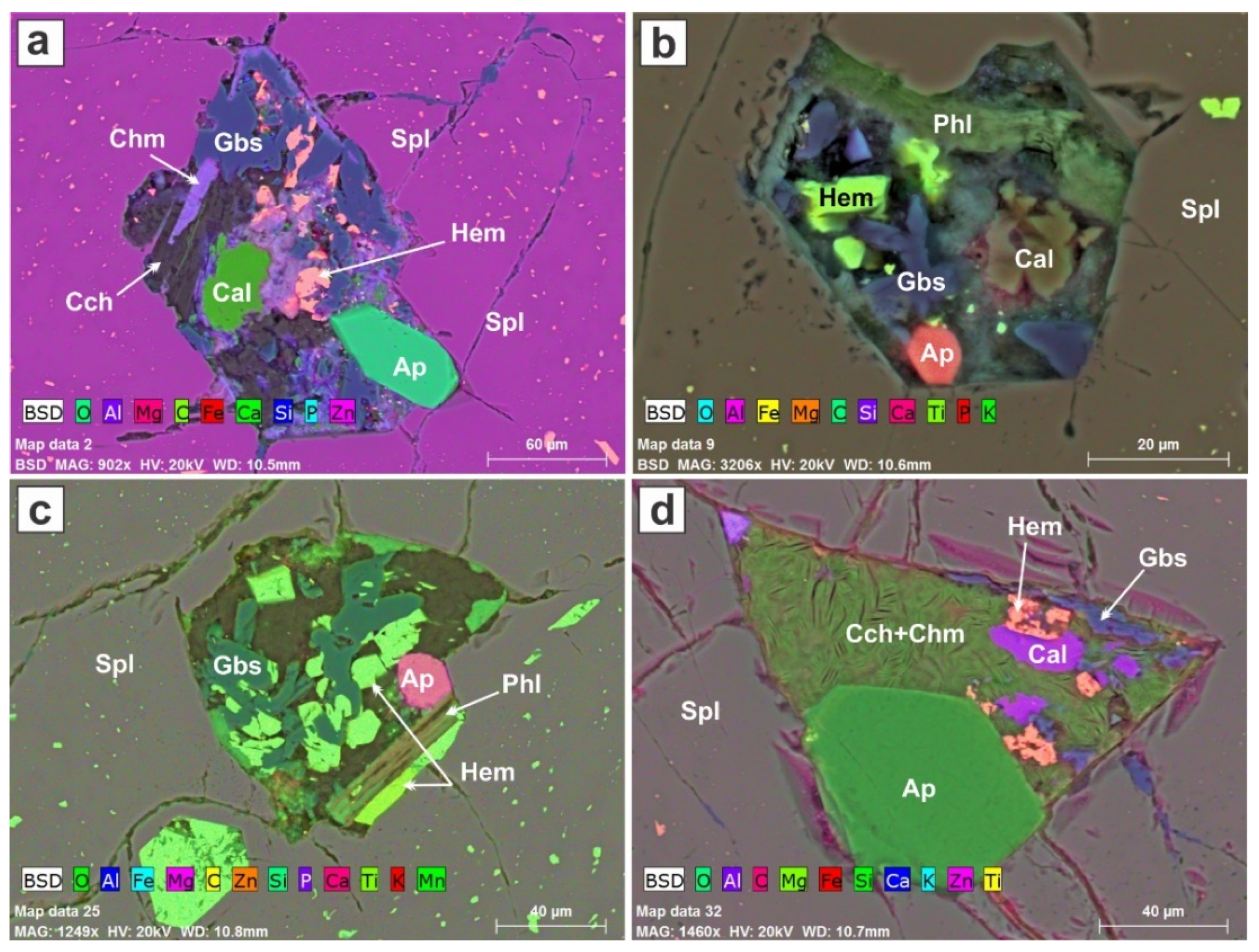
Many polymineralic inclusions show the effects of a hydrothermal overprint (
Figure 10); in some instances, not much of the primary minerals is left. In
Figure 10a, the apatite seems to have survived well, but the calcite has been partly dissolved. Clinochlore, chamosite, and gibbsite (or another polymorph of Al(OH)
3) are prominent. In
Figure 10b, the calcite and phlogopite crystals seem to have been attacked. Phlogopite is coated by hematite in
Figure 10b. Apatite has survived in the triangular inclusion in
Figure 10d (presumably tetrahedral in three dimensions), but the calcite has been attacked, and the empty space has been filled with a mixture of clinochlore and chamosite.
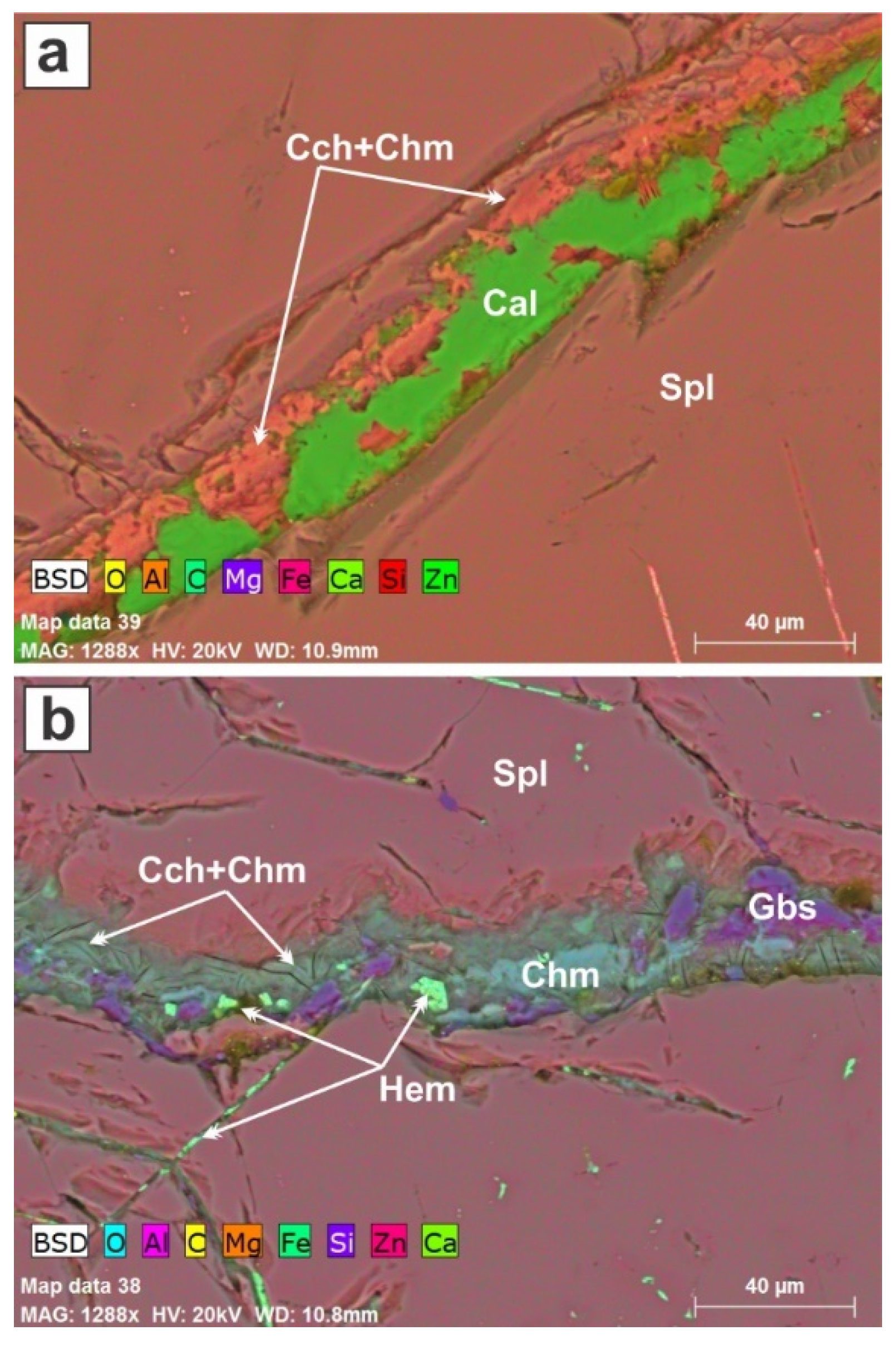
Late fractures cut the spinel;
Figure 11a,b show the minerals that line the fractures, deposited from the ambient fluid phase. Calcite is prominent, as is a mixture of clinochlore and chamosite. On the scale of the hand specimen (
Figure 2b), the fluid phase is in contact with forsterite, calcite and spinel.
Figure 11b shows well how the spinel is dissolved along the cracks. The Al supplied to the fluid phase accounts for the precipitation of chlorite-group minerals instead of a serpentine.
8. Stable Isotope Data
According to our preliminary data, the oxygen isotope composition (δ
18O) of forsterite, spinel, phlogopite, and calcite is in the range 15–17‰ (
Table 4, SMOW standard). Such values clearly are crustal. Values of the coexisting forsterite and spinel are far removed from the value expected for an upper-mantle-derived assemblage, in the range 5.5 to 5.9‰ [
17]. On the other hand, the values found in calcite approach values encountered in calcite of regionally developed marble in the area. At the Yates prospect near Otter Lake, Quebec (
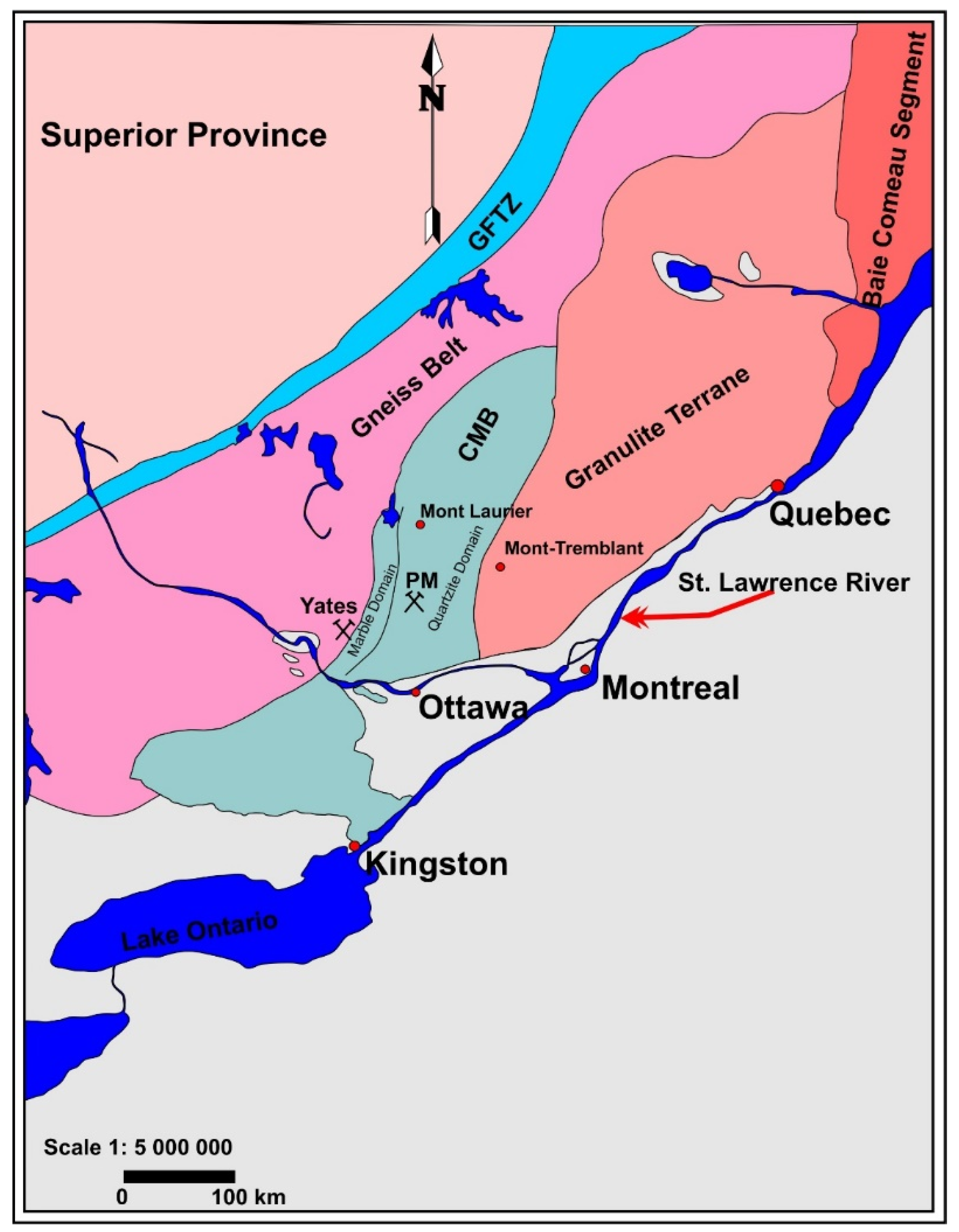
Figure 1), Kretz [
18] found δ
18O values of calcite in the range 17 to 28‰, and a lower value (14.5‰) where the marble is metasomatized. Peck et al. [
19] also documented a range in δ
18O in calcite of 17 to 28‰ at Mont Tremblant (
Figure 1), with the lower values characteristic of rocks more strongly affected by decarbonation reactions.
Carbon with a δ
13C value of −5‰ is proposed as the isotopic composition signature of the mantle [
20]. However, a ±3‰ range of values is commonly encountered. Values of δ
13C in inorganic limestone are expected to be close to +2‰, whereas in limestone containing organic carbon, δ
13C is expected to be as low as −25‰, all with reference to the PDB standard. The calcite matrix to the forsterite, spinel and phlogopite at the Parker mine, with a δ
13C value in the range 0.6–0.8‰, is close to what is expected in limestone. The range of δ
13C values in calcite in marble at the Yates prospect, is −2 to 6‰ (0.23‰ where the marble is modified by metasomatism, according to Kretz [
18]), whereas at Mont Tremblant, it is −1.9 to 2.5‰ [
19].
9. Discussion
In the context of Grenville studies in Quebec, Ontario, and New York, the Parker phlogopite mine is the site of a classic “vein-dike” association [
21,
22]. Plates of phlogopite up to 15 cm across were extracted from the calcite-cored dikes. As stated earlier, the Parker mine is distinct from the norm in being associated with forsterite rather than diopside, such that the peridotite component of the vein-dike association is dunite rather than clinopyroxenite. In addition, the locality is well known for aesthetic black spinel crystals enclosed in the calcite matrix (
Figure 2a).
There are very few options when it comes to explaining the formation of dunite. We know of three scenarios. On the seafloor, dunite may form simply by infiltration of heated seawater in harzburgite, as enstatite is very efficiently converted to forsterite by incongruent dissolution [
23]. A second possibility in the oceanic crust is the localized reaction expected along the interface between an ascending basaltic magma and a harzburgitic wallrock (e.g., [
24]).
The third alternative, which must be the case here, involves the gravitational settling of olivine crystals, as seen in stratiform layered complexes. In our case, the same process applies, but the olivine crystals grew not in a silicate magma, but in a crustally derived carbonate magma. We have documented what seems to be the first example of a dunitic cumulate formed from a crustally derived carbonatite. Viewed in this light, the absence of chromium and nickel makes sense.
We consider the statement that the carbonatitic melt has a crustal derivation to be firmly established on the basis of our preliminary oxygen-isotope values on calcite, forsterite, spinel, and phlogopite (
Table 4. Values of δ
18O are far from the “magmatic box” [
17]. Values of +15–17‰ with respect to the SMOW standard are intermediate between values of up to 28‰ encountered regionally in the white marble of the Otter Lake and Mont Tremblant areas, to the west and east of our study area, respectively, and the value expected for an upper-mantle-derived assemblage, in the range 5.5 to 5.9‰ [
17]. Were mantle-derived fluids perhaps involved as a mechanism of heat transfer to promote the fusion of marble and to shift the δ
18O values of all four minerals to such an intermediate range? We plan to acquire more δ
18O data at this location and at other sites of “vein-dike” activity in the Grenville Province to address this question. If there was an involvement of mantle-derived fluids at the Parker mine, it is not evident in an enrichment of nickel, chromium, rare-earth elements or high field-strength elements.
The δ
18O values of forsterite, spinel and phlogopite at the Parker mine are quite similar, which implies a high temperature of equilibration among these minerals. Application of the forsterite–calcite geothermometer [
25] indicates an equilibrium temperature of the order of 1100 °C if one assumes equilibrium between forsterite and the enclosing calcite. Note that calcite is a reactive mineral; its δ
18O values (
Table 4) could have been modified during late fluid-circulation events, such that an attempt at geothermometry would be compromised. On the other hand, there can be no doubt that the assemblage formed at a high temperature. A subsolidus modification of the oxygen-isotope signature of centimetric crystals of spinel, forsterite and phlogopite is not considered possible.
Were the regionally developed temperatures sufficient to get calcite to melt in the presence of a mixed CO
2–H
2O gas phase? At a pressure of one kilobar, the temperature of the calcite + portlandite + liquid + vapor assemblage is 675 °C [
26], and signs of melting of calcite have been noted at 650 °C [
27]. Regionally developed temperatures to the north, in the Bondy gneiss complex, attained 950 °C at a pressure close to 10 kbar during the Shawinigan stage of deformation. Corriveau (Table 9 in reference [
7]) quoted an estimated temperature of equilibration of 650 °C in four samples of marble and one calc-silicate rock from the Quartzite domain, and an age of metamorphism measured on titanite (U–Pb) ranging from 1165 to 1110 Ma. We conclude that regionally developed temperatures in the area were sufficient to melt marble.
In terms of the evolution of the Grenville collision zone [
9], subduction and compressive stresses were actively developed during the Shawinigan episode over the interval 1200–1160 Ma. We concur with McLelland et al. [
9,
28] that the emplacement of the AMCG suites, for example the Morin anorthosite east of Mont Tremblant at 1150 Ma, marks a time of distension in the Earth’s crust. The Parker mine crustal silicocarbonatite and associated dunite were emplaced at 1140 Ma. Any fluids rising into the crust at 1140 Ma may have had a link with the progressively sinking subducted plate or the rising asthenopheric mantle. There is no sign of pervasive deformation in the rocks at the Parker mine, nor is there at other sites of “vein-dike” activity to our knowledge. The relatively young ages of marble quoted by Corriveau, in the lower part of the 1160–1110 Ma interval (Table 9 in reference [
7]), may well have been taken at sites like the Parker mine where the marble experienced an additional thermal pulse at a time of tectonic relaxation and orogenic collapse.
The dunite seems to have formed quickly. The distribution of the two dense minerals is rather chaotic; the volume proportion of spinel, the denser mineral, is highly variable (
Figure 3 and
Figure 4). The ambient interstitial low-density carbonatitic melt into which these minerals sank has largely been squeezed upward to form the carbonatitic segregations and the phlogopite-bearing dikes. The rising melt was enriched in dissolved H
2O because of the massive crystallization of forsterite and spinel, which are devoid of hydroxyl. This dissolved H
2O was necessarily liberated upon crystallization of the melt. This explains the hydrothermal overprint that seriously affected the micro-inclusions trapped in the spinel (
Figure 10). The dunitic cumulate cooled in a relatively anhydrous environment, to judge from the limited extent of serpentinization (
Figure 6a,f).
Inclusions of spinel in forsterite and vice versa (
Figure 4 and
Figure 6) point to their virtually simultaneous nucleation in the hydrous silicocarbonatitic magma. The necessary production of a gas phase during crystallization [
26,
27] provides the explanation of such rapid crystallization; the low-viscosity melt degassed quickly, and hydrogen [
29] diffused out of the system. The net loss of hydrogen caused the melt to become intrinsically oxygenated, and to crystallize hematite. The spinel solid-solution also exsolved magnetite as the system cooled. The absence of sodium-dominant minerals suggests that sodium also may have left the system via the escaping gas phase.
The development of polymineralic inclusions (
Figure 6), in which several minerals seem to have crystallized together within the confines of a tiny enclosure, suggests an origin by incorporation of aliquots of a boundary-layer melt developed as the host mineral grew, much as we found in the fluorapatite prisms at the Yates prospect, Quebec [
30]. Such a melt contained all constituents rejected by the rapidly growing host, either forsterite or spinel. This enrichment explains how the melt inclusion crystallized to an intergrown assemblage of a sulfate (anhydrite), a phosphate (apatite), a silicate (quartz, phlogopite), along with the carbonates (
Figure 6). The carbonate component consists of calcite (dominant) and dolomite, but in some instances, magnesite is present (e.g.,
Figure 6c, Atlas 5 BBV datasets). This may indicate that in the time available, the melt had not become well blended.
There are also subtle indications of disequilibrium in the system. For example, quartz (or another polymorph of SiO
2) is present in micro-inclusions in spinel although forsterite crystals are present close by. Quartz seems to coexist with corundum (?) or gibbsite (
Figure 9c). In granulite-grade rocks, the coexistence of quartz and corundum is considered a sign of metastability [
31,
32]. Finally, ribbons of diopside are developed along only selected interfaces of forsterite with the interstitial (Ca, Mg) carbonate melt (
Figure 4a,d,e and
Figure 5b). This reaction is probably relevant to the conversion of lherzolite to wehrlite in the mantle; a primary dolomitic melt is expected to react with lherzolite as it migrates upward, causing the carbonatite to become enriched in Ca as Mg is consumed through the metasomatic “wehrlitization” of the conduit [
33,
34]. Why the reaction is not developed along all forsterite–(Ca, Mg) carbonate interfaces in the dunite is likely a reflection of the rapid crystallization of the assemblage upon loss of the gas phase. Presumably the reaction could only occur where the carbonate was still molten.
10. Summary and Conclusions
We describe an unusual case of a “vein-dike” association in which the peridotite member of the association is dunite rather than pyroxenite. The phlogopite-bearing carbonate dikes and accompanying dunite were emplaced 1140 million years ago, after the Shawinigan episode of subduction and deformation had come to an end. The crust was in a state of distension. In the area near Notre-Dame-du-Laus, there was perhaps a localized thermal pulse that raised the temperature significantly over that achieved during the Shawinigan orogenic event. Transfer of a fluid phase is the most efficient mechanism of heat transfer. We believe that marble melted in response to such an influx of fluid that had equilibrated with a silica-poor deep source. The prevalence of forsterite and spinel indicates that the activity of silica in that melt was quite low, and insufficient for enstatite to crystallize.
The dunite is a cumulate, largely of forsterite and spinel, but also with diopside, magnesio-hastingsite, phlogopite, ilmenite and apatite. Vestiges of the interstitial carbonate remain. The main minerals have values of δ18O in the range 15–17‰, which means that they formed from a crustally derived melt. What we have is a dunite with a metasedimentary antecedent, possibly a first! The melt was initially hydrous, and became more enriched in H2O owing to the crystallization of the anhydrous minerals. The fluxed carbonate magma was rapidly emplaced above the cumulate and developed strikingly euhedral centimetric crystals of spinel, forsterite, and phlogopite prior to freezing. Degassing of the system led to a preferential loss of hydrogen; the magma did crystallize hematite, and the spinel underwent oxidation-induced exsolution of magnetite.
More studies of this type in similar Grenvillian occurrences are necessary to test fully the validity of our petrogenetic scheme. Did melting of marble occur only at point sources, or is the scale of marble melting regional? What seems clear is that the emplacement of “vein-dikes” occurred at different times in different places. The noncommittal term “vein-dike” was favored by Moyd [
21] because of his hesitation to conclude that marble did melt in the Ontario portion of the Grenville Province. It is fair to say that most metamorphic petrologists still are not aware that marble can melt. Interestingly, the first claim that it does melt where the conditions are right was made by Ortega-Gutierrez [
35] on Grenvillian rocks exposed in the province of Oaxaca, Mexico. Such dikes of crustally derived carbonatite seem to be rather common in the Grenville Province.
At the Yates prospect, near Otter Lake, Quebec, the melting of marble occurred close to one billion years ago, at the terminal stage of the Grenville collisional orogeny [
30]. It was in the Bancroft area, Ontario [
36], and particularly in explorations with Ray McDougall at the Bear Lake diggings, that the first author started to wonder about the possibility of crustally derived carbonatites. They can be expected in any collision zone. Excellent examples do exist in the major Early Paleozoic collision that occurred late in the pan-African orogeny [
37,
38], although most still view them as skarns, just as in the Grenville Province. In each of these environments, there were likely many episodes of collision between continental plates. We visualize a period of quiescence and distension after each one, at which stage localized melting can be expected in the thickened crust.