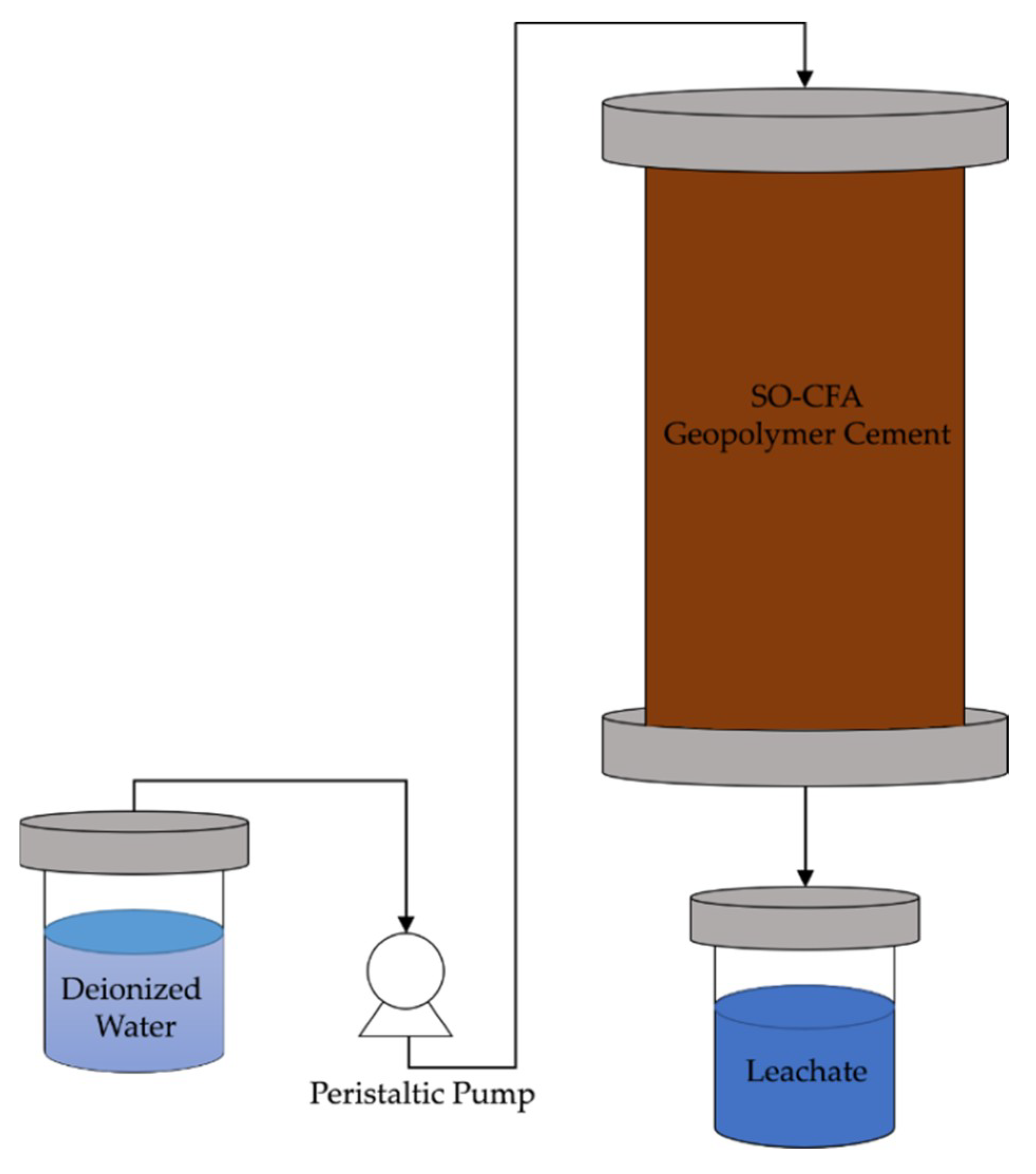
3.2.1. Unconfined Compressive Strength
The control sample soil-fly ash (SO-CFA) geopolymer, not immersed in acidic condition, was tested after the 28-day curing has a recorded unconfined compressive strength of 1.0 MPa. Note that such strength development of the stabilized soil even at ambient curing suggests geopolymerization has occurred. Geopolymeric gels that are formed in the soil can assist the soil particles to form more compact microstructures [
18]. The soil used in this study is nonplastic and cannot be molded to exhibit measurable unconfined compressive strength; however, when mixed as part of one-part geopolymer binder system, it has strength that can be used as backfill/foundation. With the standard stated in the National Structural Code of the Philippines, stabilized soil with an unconfined compressive strength of at least 0.075 MPa (75 kPa) for the SM type soil can be recommended for backfill/foundation applications [
39].
Moreover, our study indicates that strength development of soil-fly ash (SO-CFA) geopolymer still progressing even if the specimen is immersed in 5% (percentage by weight) sulfuric acid solution. The measured unconfined compressive strength of soil-fly ash geopolymer cement immersed in acidic condition for 28 and 56 days were 2.6 and 3.2 MPa, respectively. The results of the compressive strength show that the matrix of soil-fly ash geopolymer cement seemed not to be affected by the presence of an acid. Likewise, the strength of soil-fly ash geopolymer immersed in acidic condition has also been found to increase as the length of immersion increases.
As for the product designed for fit to purpose, the soil-fly ash geopolymer developed in this study has an unconfined compressive strength comparable to soil stabilized with lime and/or cement despite its exposure to a more aggressive environment as shown in
Table 3. Based on several studies, soil treated with lime has a range of unconfined compressive strength value of 0.01–3.0 MPa. Conversely, soil stabilized with cement can have unconfined compressive strength values ranging from 0.10 to 0.90 MPa.
The increase in unconfined compressive strength of this study during the sulfuric acid immersion was not in agreement with the result of the study conducted in [
25], wherein a loss in strength was recorded. The mechanism that may occur during sulfuric acid immersion involves an attack of acid on aluminosilicate bonding present in the geopolymer matrix. This behavior may result in deterioration of the geopolymeric network, particularly attacking the aluminosilicate bonding, which could result in an increase in the formation of Si–OH and Al–OH, and thereby a loss in compressive strength. However, the strength loss was not observed in this study. In fact, an increase in strength was manifested. This may be attributed to the type of gel matrix formed and may be closely related to the intact geopolymer network structure of the Si–O and Al–O in the matrix, which makes the soil-fly ash geopolymer cement in this study an acid resistant. Similar findings were observed in [
19], in which the geopolymer was a better acid resistant due to the intact matrix of the geopolymeric network and to the possible neutralization of Na-rich gel with acidic media.
3.2.2. Structural Bonding Property
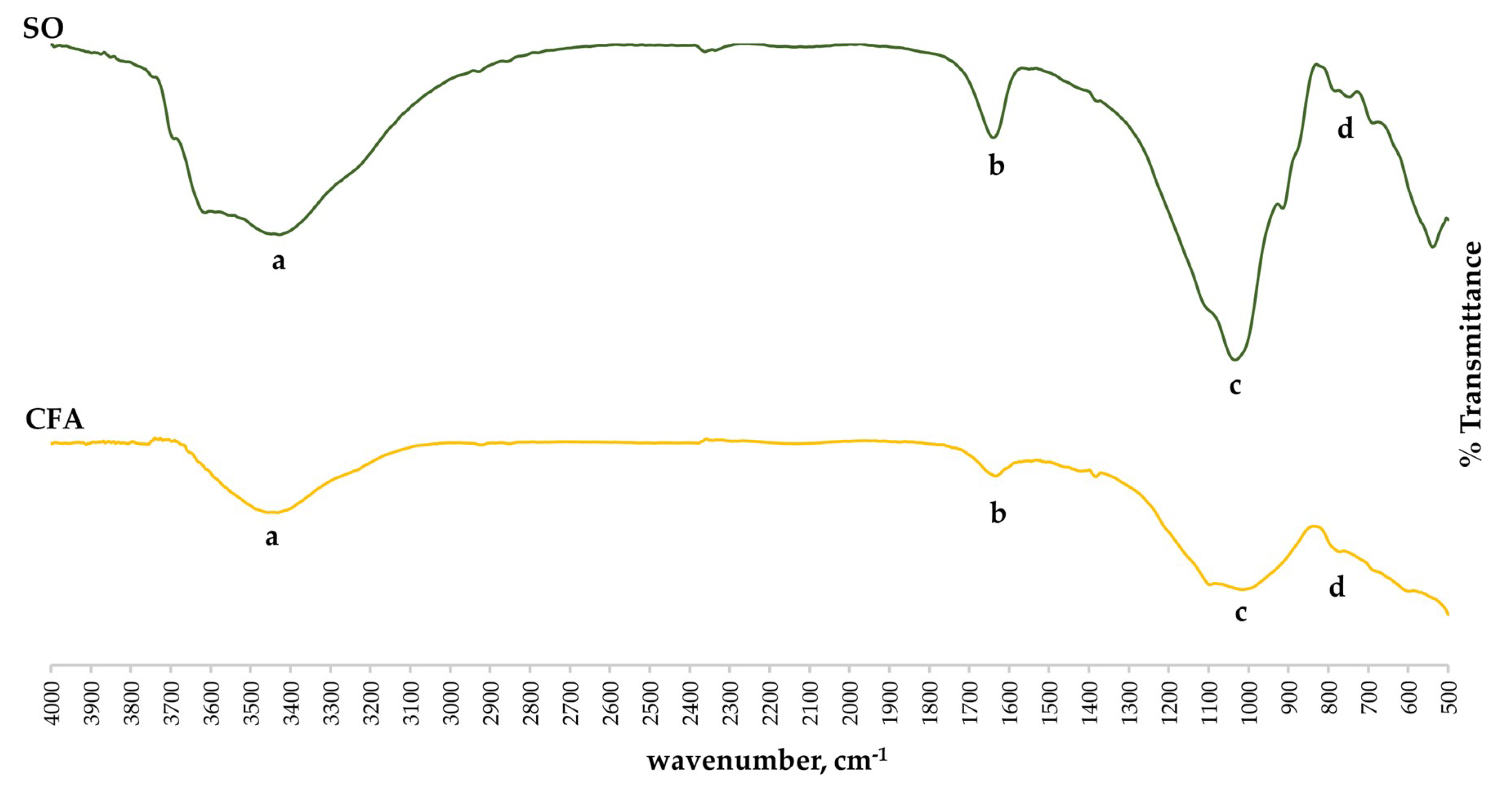
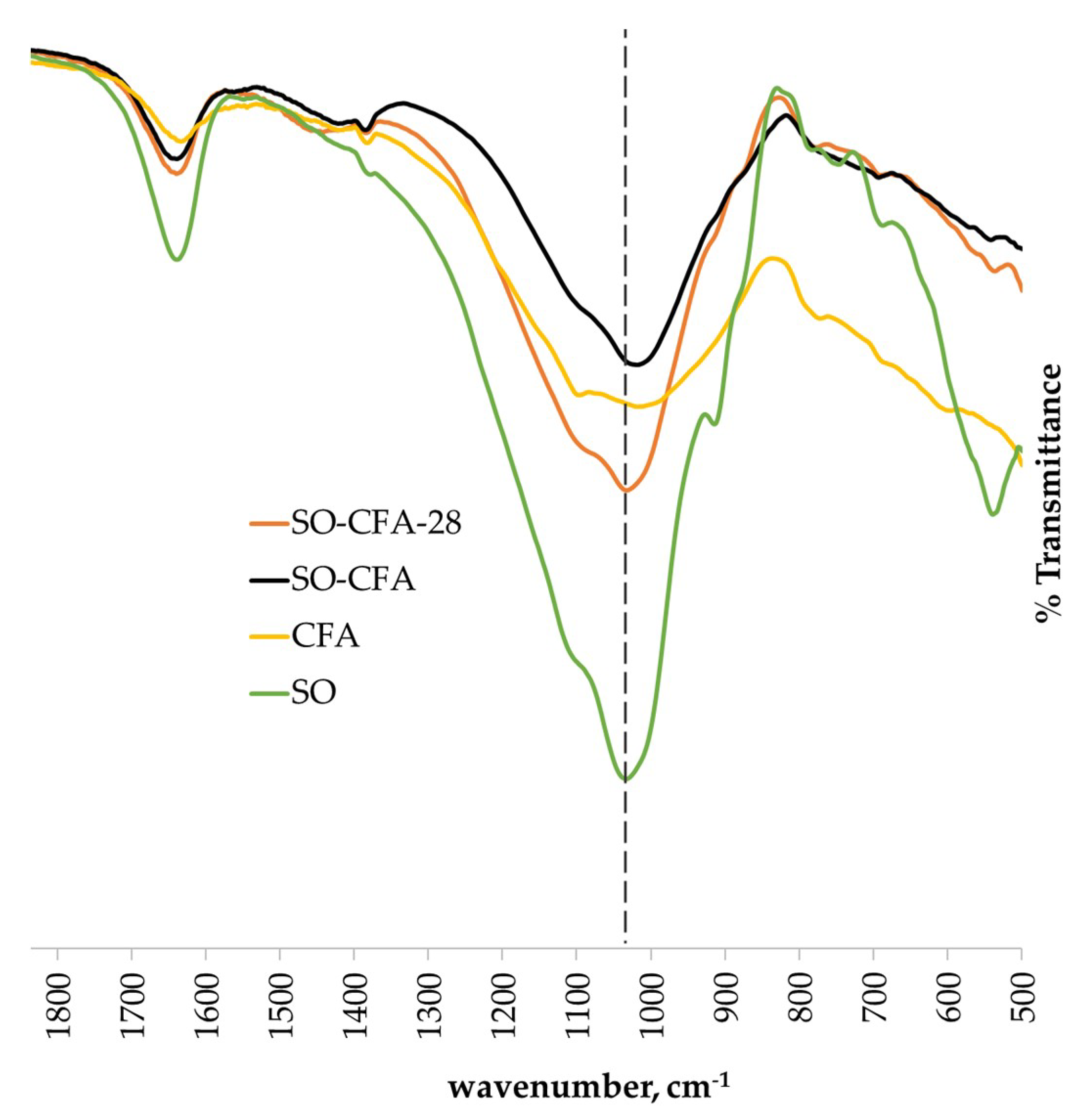
The structural bonding of the control soil-fly ash geopolymer cement (SO-CFA) and acid-immersed soil-fly ash geopolymer cement (SO-CFA-28) were investigated using FTIR analysis as presented in
Figure 5.
Table 4 shows the values of the observed peak of raw materials, SO-CFA, and SO-CFA-28. The O–H antisymmetric was observed at 1637–1643 cm
−1 [
46]. Main structural bonds are located at peaks between 900 and 1099 cm
−1 which indicates Si–O–T (T, either Al or Si) anti-asymmetric stretching [
36].
As shown in
Figure 5, the narrowing of peaks was observed between the spectra of raw materials and soil-fly ash (SO-CFA) geopolymer cement. This may be due to the further stretching of the Si–O–T bonds. It was also observed that a shift in peaks occurred on the main bands of SO, CFA, and SO-CFA samples toward a decreasing wavenumber, from 1099 to 1018 cm
−1. The shift of this band is indicative of the geopolymerization in the solid matrix [
47,
48]. Additionally, peaks within the main band for SO-CFA-28 were clearly sharper than those of the SO-CFA sample. This is an indication of a change in the structural matrix of geopolymer brought by either the attack of the acid or the further development of the geopolymer matrix. A shift in peaks to increasing wavenumbers, from 1018 to 1032 cm
−1, of the SO-CFA and SO-CFA-28 samples may indicate partial depolymerization of the amorphous gel matrix due to acid attack, thereby dissolving some of the amorphous content of the gel matrix [
48]. Similar findings were reported in [
14], wherein the shift in peaks was caused by the depolymerization due to acid attack. Partial dissolution of the gel matrix could result in a compressive loss of the geopolymer. However, with the increasing unconfined compressive strength as reported in
Section 3.2.1, this mechanism is not evident with the SO-CFA-28 sample. This partial depolymerization that may have occurred still has not caused significant impact to affect the unconfined compressive strength of soil-fly ash geopolymer cement developed in this study. This observation can be correlated with the hypothesis on the possible intact network that had been formed on the SO-CFA-28 sample, making it an acid-resistance material [
19].
3.2.3. Mineralogical Phase
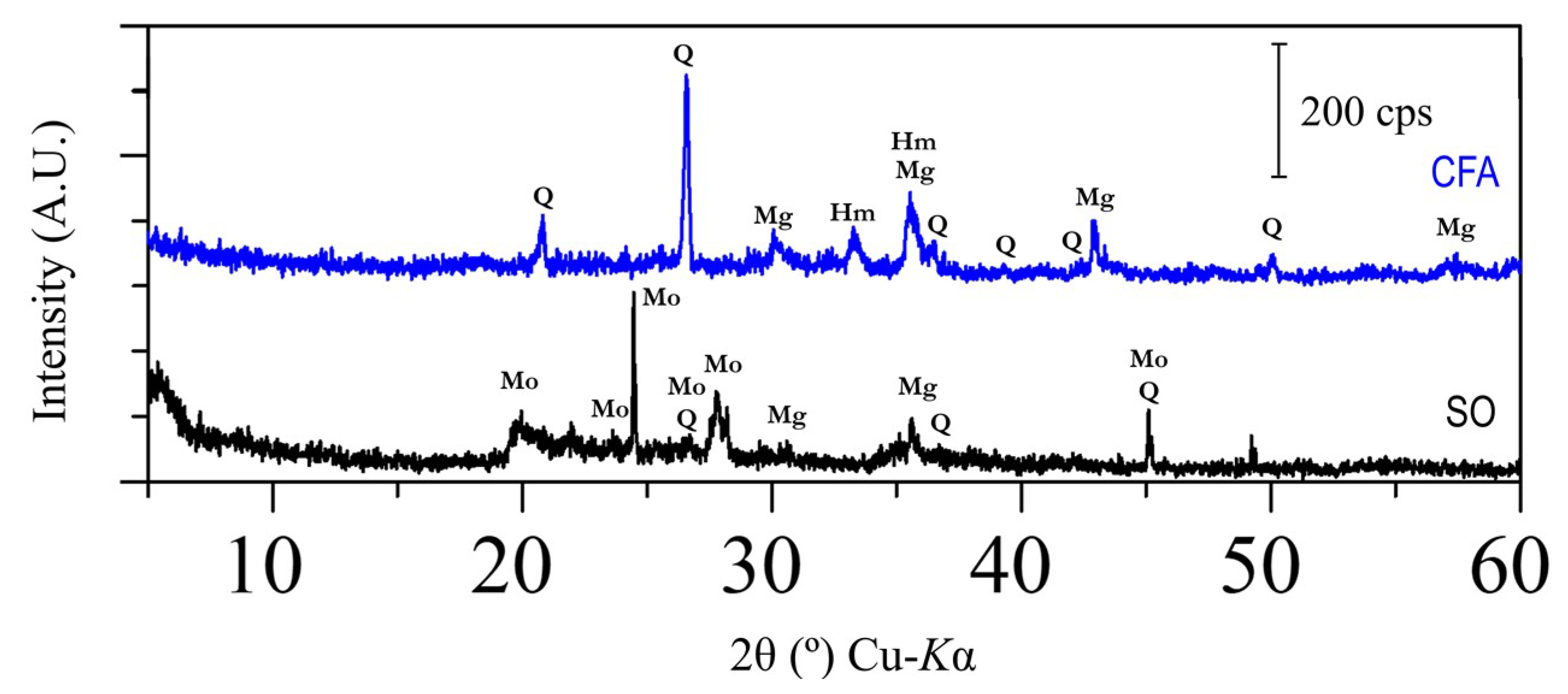
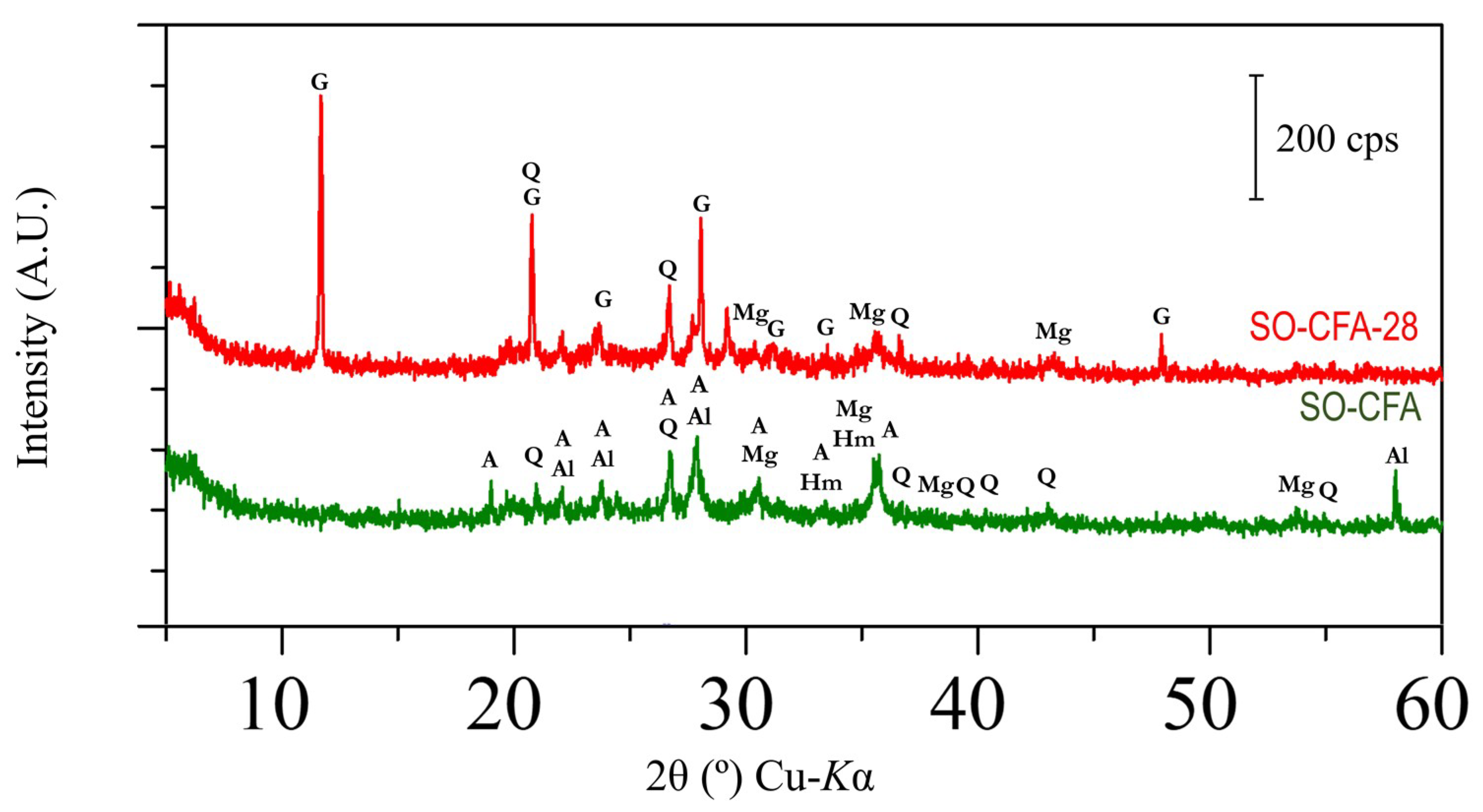
The difference in mineralogical phase between soil, coal fly ash, SO-CFA, and SO-CFA-28 has been demonstrated in
Figure 6. The presence of minerals such as quartz (SiO
2), hematite (Fe
2O
3), and magnetite (Fe
3O
4) are expected given that the soil and coal fly ash contain oxides of silica and iron.
The disappearance of some peaks between the raw material and the soil-geopolymer cement (SO-CFA and SO-CFA-28) is worth noting. The disappearance of peaks, as for the case of montmorillonite ((Na,Ca)
0.3(Al,Mg)
2Si
4O
10(OH)
2·nH
2O) and hematite (Fe
2O
3), may be associated with the concealing of amorphous phase over the crystalline phase. This may also imply that crystalline components, for instance, quartz and magnetite initially present in the precursor, have not participated in geopolymerization [
47]. Interestingly, geopolymerization using soil, CFA, and solid alkali activators has led to the formation of new crystal–anorthite (CaAl
2Si
2O
8) and albite (NaAlSi
3O
8) as shown in the SO-CFA pattern. This is an indication that the components such as SiO
2 and Al
2O
3 present in raw materials and alkali activators have reacted. Given the acidic media contains sulfate ions, the presence of gypsum (CaSO
4∙2H
2O) in SO-CFA-28 is expected.
After further exposure to the acidic environment, the mineralogical phase of soil-fly ash geopolymer cement (SO-CFA-28) was examined. The disappearance of albite and anorthite peaks have been observed and may be explained similarly to the mechanism behind weathering. The disappearance of albite peaks may be associated with its dissolution in acidic media. This results in the exchange of hydrogen ions for sodium ions at the surface of the albite, breaking Al–O bonds and forming an Si-rich precursor, and the hydrolysis of Si–O bonds [
49]. On the other hand, anorthite weathering requires only the breaking of the Al–O bonds [
50]. The framework of the anorthite with an Si/Al ratio of 1, upon exposure to acidic media, may deteriorate due to the attack of the hydrogen ions. This deterioration may result in the detachment of calcium and aluminum, thereby leaving tetrahedra of silica [
50]. The presence of available calcium ions, which resulted from the dissolution of anorthite and sulfate ions from acidic media, leads to the formation of a new crystal, with peaks of gypsum (CaSO
4∙2H
2O ) at 11.60° and 20.70° 2θ, in the SO-CFA-28 sample [
19]. This finding is similar to that found in [
19], in which the formation of a new crystal such as gypsum was observed after immersion in a sulfuric acid solution.
Exposure of anorthite in the acidic environment could result in (i) the dissolution of a framework resulting in amorphous polymer and (ii) the further formation of N–A–S–H and C(–A)–S–H gel. The increase in unconfined compressive strength upon exposure in the acidic environment may be explained by the idea that the crystalline geopolymer was more stable than amorphous geopolymer when exposed to an acidic environment [
32]. In this case, the dissolution of anorthite may have caused an increase in strength. Similar findings were observed in [
51]. The results of that study showed a disappearance and formation of new peaks in XRD analysis. Notable findings such as an increase in strength of geopolymer binders over time and a decrease in strength of a cement-based binder under acidic media were also observed. The N–A–S–H gel phase present in geopolymer binder is believed to neutralize the acid, thereby preventing the matrix from further deterioration [
19].
In the case of cement-based binder, deterioration is attributed to the attack of an acid on the calcium hydroxide in the cement paste and on the C–S–H framework, which could lead to the formation of more gypsum in the matrix. The presence of gypsum in the matrix can be detrimental because of its solubility in water [
52]. This could affect the long-term durability, as the presence of this mineral can cause expansions and cracks. This may explain the advantage of a matrix with a N–A–S–H/C(–A)–S–H gel over one with a C–S–H gel that is typically found in ordinary Portland cement-based binder systems. This also supports our hypothesis that an intact geopolymeric network has been developed in this study, which results in an increase in compressive strength despite the presence of gypsum. The result of the XRD pattern is also in agreement with the result of FTIR analysis, wherein a change in structure was evident with the shift of peaks, which corresponds to the formation of new crystals/component in the soil–geopolymer cement matrix.
3.2.4. Surface Morphology
Morphological characteristics of soil-fly ash geopolymer cement without immersion (SO-CFA sample) and with immersion in 5% by weight sulfuric acid for 28 days (SO-CFA-28 sample) were carried out using SEM-EDX analysis. The SEM images (
Figure 7a,b) of the SO-CFA samples clearly contain a number of spherical particles similar to the morphology of coal fly ash. The spherical particles that cause large voids and pores in the matrix may be attributed to the unreacted coal fly ash (
Figure 7c–e). Partial polymerization of CFA particles has also been observed as shown in
Figure 7f. Furthermore, the presence of needle-like structures was observed in
Figure 7e, which also indicates partially dissolved coal fly ash in the sample [
53]. Additionally, the magnification of SO-CFA SEM images reveals that some of the spherical particles are covered with a corrugated surface (
Figure 8a) [
54]. As confirmed by EDX analyses presented in
Figure 8b, spikes of iron and oxygen have been noted. These resemble the morphology of hematite (Fe
2O
3) and magnetite (Fe
3O
4) [
55]. This is consistent with the result of the XRD pattern of the SO-CFA sample in
Section 3.2.3.
On the other hand, SO-CFA-28 has a homogeneous phase structure as evidently illustrated in
Figure 9a–d, relative to the SO-CFA sample. It also contains a lesser amount of unreacted coal fly ash. The smaller voids and pores on the morphology led to a well compacted and cemented surface. A rod-like structure from a broken sphere was also observed (
Figure 9e). Based on the EDX analysis, this morphology has a high iron content. Another study [
56] showed similar characteristics in which such a surface was described as having a prismatic needle feature. This may indicate the partial dissolution of coal fly ash, resulting in the exposure of smaller particles.
Moreover, hexagonal shape structures were also evident in the sample (
Figure 9f). As confirmed by EDX analysis (
Figure 10), these structures are rich in calcium and sulfur. This resembles gypsum (CaSO
4∙2H
2O). Formation of gypsum may be explained by the mechanism behind the sulfate attack in the matrix. This involves the reaction of sulfate ions from acidic media with the free calcium present in the geopolymeric gel. Evidence of gypsum formation was also revealed in the XRD patterns of SO-CFA-28 (
Figure 6).
Based on the EDX results, SO-CFA and SO-CFA-28 samples were composed of Si, Al, Na, Ca, Fe, O, and C. Geopolymeric gel microstructures were investigated by calculating the following molar ratios based on elemental composition: Si/Al, Na/Al, Na/Si, Ca/Si, and Fe/Si. Results are summarized in
Table 5.
Based on the calculated molar ratios, SO-CFA dominantly composed Na-rich gels, while SO-CFA-28 sample comprises of gels with Na and Ca. The coexistence of these gel matrices, which are believed to be sodium aluminosilicate hydrate (N–A–S–H) and calcium (alumino) silicate hydrate (C(–A)–S–H) can cause significant improvement in the compressive strength [
57]. This is consistent with the result of the unconfined compressive strength as presented in
Section 3.2.1, in which the UCS of SO-CFA-28 significantly increased.
The coexistence of these gel matrices has also been observed in [
58]. The results showed that the amount of available calcium ions present in the sample has a significant effect on the formation of the gel matrix. Projection of gel chemistry showed that a lower content of calcium in the proportion mix could produce a N–A–S–H gel. Similar findings were observed in [
59], wherein the formation of N–A–S–H gel demonstrates that some sodium replaces calcium in the C–S–H matrix. On the other hand, a higher content of calcium could produce a chain silicate of C(–A)–S–H gel. This can be correlated with the XRD pattern of SO-CFA-28 in which there was a potential increase in available calcium ions due to the disappearance of anorthite peaks. An increase in calcium ions could favor the development of C(–A)–S–H and hence an increase in Ca/Si molar ratios. This gel formation mechanism may also explain the coexistence of both gels probably present in the SO-CFA-28 sample.
The high amount of iron present in the matrix is also worth noting. It is believed that the iron component participates in geopolymerization [
56]. This can be correlated with the result of XRD analysis of raw materials and geopolymer, as presented in
Figure 6. Peak intensities of iron-rich crystals almost disappeared after further geopolymerization. Given the molar ratios summarized in
Table 5, these gel compositions resemble a typical poly(ferro-sialate-siloxo) and poly(ferro-sialate-disiloxo) type of binder with a formula relatively close to [Ca,Na]–[–Fe–O–]
x–[Si–O–Al–O–]
1−x–[–Si–O–]
y with x values less than or equal to 0.5 and y values between 0 and 25 [
60].