1. Introduction
Copper is considered the third most important metal for industry after iron and aluminum, with a yearly production of 10 Mt [
1]. In the north of Chile there are many mining industries that produce copper in cathodes from oxidized minerals with the traditional process of leaching, followed by solvent extraction (SX), and electrowinning (LIX-SX-EW). Due to the deepening of pits [
2], depletion of oxidized ores and the presence of sulfide ore is noted, a situation that puts at risk the continuity of hydrometallurgical processes due to the lack of leachable mineral resources. Hence, different leaching techniques for copper sulfide ores, especially chalcopyrite, need to be investigated in the future.
Presently, the mining industry faces the challenge of extracting copper from low-grade copper ores. In 2002 the average copper ore grade was 1.13%, while in 2011 it was 0.84% [
3].
Many chalcopyrite leaching studies have been performed with leachants such as ferric sulfate [
4,
5,
6,
7], ferric chloride [
7,
8], sodium nitrate-sulfuric acid [
9], hydrochloric acid [
10], and sodium chloride-sulfuric acid [
11], among others [
10].
The slow copper from chalcopyrite dissolution has been attributed to the formation of a passivating layer on the mineral surface. This phenomenon has been widely studied, with no consensus regarding its cause or how to avoid its formation [
12,
13]. So far, three possible causes have been identified: (i) the formation of a layer of precipitated ferric iron that prevents contact between the chalcopyrite and the leaching solution [
14,
15], (ii) the formation of a layer of sulfur, which reduces the flow of electrons and the diffusion of the leaching agent to the mineral surface [
4,
16,
17], and/or (iii) the formation of an intermediate layer during the dissolution of chalcopyrite, such as a sulfide, disulfide, or polysulfide compounds, which have a slow kinetic dissolution [
12,
18,
19]. Recent studies showed that passivation of chalcopyrite is dependent on redox potential (Eh) since high redox potentials (high concentration of ferric ions) promote passivation of chalcopyrite [
5]. On the other hand, ferrous ions may increase the leaching kinetics significantly compared to ferric ions [
6,
20,
21]. This is in contrast through to other leaching processes, such as the bacterial leaching of gold sulfide concentrates, where ferric iron promotes the dissolution of pyrite and arsenopyrite [
22].
In addition, numerous studies showed that leaching of chalcopyrite in chloride media results in higher copper extractions than in sulfate media [
16,
23,
24]. The main reasons are the formation of chloride complexes, which change the electrochemical behavior of chalcopyrite passivation and/or the higher porosity of the mineral surface [
8,
16,
25]. For example, Dutrizac [
7] associated the high percentages of copper extraction in a chloride medium to the lower activation energy of the medium, approximately 42 kJ/mol, compared with that in a sulfate medium (75 kJ/mol).
Li et al. [
26] and Kaplun et al. [
27] demonstrated that the redox potential (Eh) significantly affects the leaching rate of chalcopyrite. To understand the effects of passivation during leaching processes, the passivation potential (Epp) has been used to determine the adequate potential to effectively leach the chalcopyrite. Chalcopyrite leached in a medium with sulfuric acid maintains its reactivity under 685 mV (SHE) regardless of impurities, acidity, or temperature. Between 685 and 755 mV (SHE), chalcopyrite may be in between the passive and active state, depending on the form at which that potential was reached. At 755 mV (SHE) the leaching of chalcopyrite becomes slow due to the effect of passivation [
28]. Sandström et al. [
29] suggested that chalcopyrite leaching should be carried out in sulfuric acid medium at 620 mV SHE, rather than at 800 mV SHE.
The leaching of chalcopyrite at high potentials (high ferric concentration) has been questioned because some studies have shown that ferric ions inhibit the leaching of chalcopyrite [
5] while ferrous ions increase its leaching kinetics [
6]. Kametani and Aoki [
21] established a critical reduction potential of 420 mV versus Ag/AgCl at which chalcopyrite dissolution reaches its maximum, demonstrating that there is an optimal concentration of ferrous ions that increase the leaching of chalcopyrite [
6,
30]. In addition, temperature is one of the most important parameters in the kinetics of chalcopyrite leaching [
31], with the dissolution rate increasing at higher temperatures [
23,
32].
Nicol and coworkers [
33,
34,
35] performed chalcopyrite leaching experiments in chloride solutions containing cupric ions and dissolved oxygen. As part of that overall program on the fundamental aspects of the heap leaching of copper sulfide minerals, Nicol [
36] compared the anodic behavior of chalcopyrite at high potentials (transpassive region) in both sulfate and chloride solutions. Nicol et al. [
37] studied the voltammetric characteristics of chalcopyrite under various conditions appropriate to low temperature leaching with acidic chloride solutions while Nicol and Basson [
38] described the anodic behavior of covellite over a wide range of chloride concentrations at potentials relevant to its dissolution under ambient conditions similar to heap leaching. The other important aspect in column or heap leaching is the solution flowrate [
39]. According to Ilankoon and Neethling [
40], the way the solution spreads from drippers and the subsequent development of flow paths and any associated channeling needs to be properly investigated.
Actually, the current heap leaching process at mining companies involves a curing stage with the addition of 12 kg of sulfuric acid per ton of ore and a determined amount of Intermediate Pregnant Leach Solution (IPLS). The agglomerated ore is transported by conveyor belts to form 107 leach pads weighing a total of approximately 200,000 tons. Once the heap is formed, irrigation begins with a continuous leaching cycle of approximately 300 days. The modification proposed in this study includes the addition of reagents such as sodium chloride and ferrous sulfate in the agglomeration stage, together with the acid, and carrying out the leaching process at an average temperature of approximately 30 °C to increase the percentage of copper extraction from primary sulfide ore.
The main objective of this work is to determine the effects of the modifications of the agglomeration-curing stages and the temperature on the copper extractions of the sulfidic ore.
4. Conclusions
The effect of the modification of the agglomeration-curing stages and temperature on copper leaching kinetics of a copper sulfide ore was reached using H2SO4, NaCl, and FeSO4.
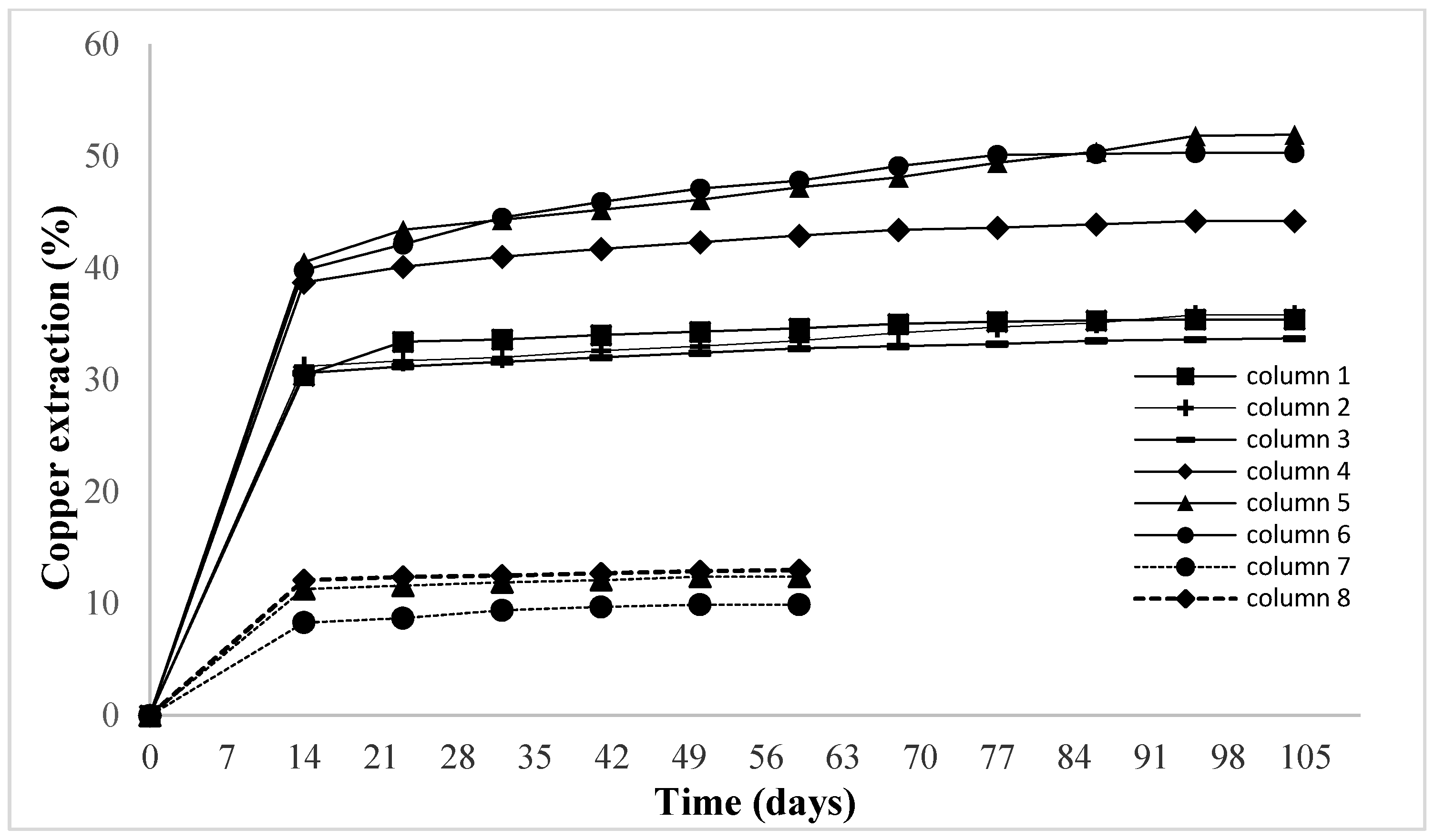
In the column tests, the largest copper extractions were obtained when leaching was carried out at a temperature of 32.9 °C. As temperature increases, for the same agglomeration conditions, extraction is up to five times higher compared to leaching at 14.5 °C.
The highest copper extraction was obtained in column 5 ore agglomerated with H2SO4, NaCl, and 1% w/w FeSO4, in which an extraction of 50.23% of copper was achieved, followed by column 6 (agglomerated with H2SO4, NaCl, and 2% w/w FeSO4), where an extraction of 48.5% was obtained.
The next highest extraction, 41.84%, was obtained in column 4 (agglomerated with H2SO4 and NaCl). Copper extractions in the columns leached at 14.5 °C did not exceed 15%, despite the addition of NaCl and FeSO4.
This medium, using ferrous-chloride, could be applied in the agglomerate stage, prior to heap leaching of copper sulfide minerals at an industrial scale.