Abstract
In this study, column leaching experiments were carried out to investigate the extraction of Ni and Co from low-grade limonitic laterites from Agios Ioannis mines in central Greece. Tests were carried out in laboratory Plexiglas columns using H2SO4 as leaching solution. Parameters determining the efficiency of the process, i.e., acid concentration (0.5 M or 1.5 M) and addition of 20 or 30 g/L of sodium sulfite (Na2SO3) in the leaching solution, were also studied. Upflow transport of the leaching solution with the use of peristaltic pumps was carried out, while the pregnant leach solution (PLS) was recycled several times over the entire test duration. The concentration of Ni, Co, Fe, Ca, Al, Mg, and Mn in the PLS was determined by Atomic Absorption Spectroscopy (AAS). The ore and the leaching residues were characterized by different techniques, i.e., X-ray fluorescence (XRF), X-ray diffraction (XRD), Fourier transform infrared (FTIR) spectroscopy, and differential scanning calorimetry and thermogravimetry (DSC/TG). The experimental results showed that (i) Ni and Co extractions increased with the increase of H2SO4 concentration—60.2% Ni and 59.0% Co extractions were obtained after 33 days of leaching with 1.5 M H2SO4; (ii) addition of 20 g/L Na2SO3 in the leaching solution resulted in higher extraction percentages for both metals (73.5% for Ni and 84.1% for Co, respectively), whereas further increase of Na2SO3 concentration to 30 g/L only marginally affected Ni and Co extractions; and (iii) when leaching was carried out with 1.5 M H2SO4 and 20 g/L Na2SO3, its selectivity was improved, as deduced from the ratios Ni/Mg, Ni/Ca and Ni/Al in the PLS; on the other hand, the ratio Ni/Fe dropped as a result of the higher Fe extraction compared with that of Ni.
1. Introduction
Nickel is the fifth most common element on earth and is widely used in many industrial, transport, aerospace, marine, architectural, military, and consumer applications. Its biggest use is in alloying, particularly with chromium and other metals, to produce stainless and heat-resisting steels [1]. It must also be underlined that nickel is considered today as the most important metal by mass in the Li-ion battery cathodes used by electric vehicle manufacturers [2].
Sulphide ores comprise about 30% of the global nickel reserves and result in almost 55% of world metal production [3]. The gradual depletion of high-grade nickel sulphides and the increasing demand for nickel has initiated studies into the exploitation of the huge reserves of nickel laterites [4,5,6].
It is known that conventional mineral processing techniques cannot be readily applied to laterites due to the complex nature of the ores and the fact that nickel is hosted in several mineral phases [7,8]. The treatment of laterites is mainly carried out by pyrometallurgical techniques to produce ferronickel (FeNi) [9,10]. However, due to the high capital and operating costs of pyrometallurgy, the use of hydrometallurgical techniques, including atmospheric, heap, and high-pressure acid leaching (HPAL), as well as bioleaching, has gained more interest [11,12,13,14].
Earlier studies of column leaching indicated that Ni or Co extraction from laterites is strongly dependent on the type of nickel-bearing phases, leaching time, acid type or concentration, and particle size. In several cases, the use of agglomerating was considered to improve permeability of the reaction bed and accelerate leaching of the useful elements [15]. Duyvesteyn et al. [16] suggested that the ore should be crushed and pelletized with the use of sulphuric acid with a concentration of at least 100 g/L to neutralize magnesia. Horizonte, S.G. and Horizonte, D.O. [17] carried out tests in columns with 1 and 4 m height using agglomerates, concentration of H2SO4 20 to 200 g/L, and irrigation rate 10 L/(m2·h). Their results indicated that higher acidity leads to faster kinetics and recoveries of 84% for Ni and 70% for Co after 150 days of leaching. Elliot et al. [18] performed column leaching tests using various Australian nickel laterites, which were agglomerated with the use of water and sulfuric acid in a rotating drum. The leaching solution contained 200 g/L H2SO4 while no recycling was involved. Depending on the ore mineralogy, extractions of 10–98% for Ni and 12–97% for Co were obtained after 120 days of leaching. The same study indicated that Ni and Co extractions were increased for laterites containing low goethite and moderate smectite content. Quast et al. [11] studied the behavior of siliceous goethitic ore and the effect of agglomeration on Ni and Co extractions in column studies, with the use of H2SO4 for a period of 100 days; also in this case, no recycling of the leaching solution was carried out. The authors mentioned that the feed size influences Ni and Co extractions which were, for the finest size used, 90% and 80%, respectively. In the study by Quaicoe et al. [19], column leaching experiments were performed using agglomerates of saprolitic (SAP) and goethitic (G) laterite ores, 200 g/L H2SO4, and an irrigation rate of 8.5 L/(m2·h). Ni and Co extractions for the G ore were, after 100 days, 62.7% and 55.6%, respectively, while for the SAP ore higher extractions of 90.0% Ni and 72.8% Co were obtained. This is due to the fact that serpentine minerals dissolve more rapidly in H2SO4 compared with the more refractory goethite. Other studies have investigated the potential of SO2 as reductant, also in the presence of Cu2+ ions as catalyst, to accelerate the rate of atmospheric acid leaching of laterites [20,21,22,23,24].
Greek laterite deposits are mainly located in three regions, i.e., Evia island and Lokrida in central Greece and Kastoria in northwestern Greece. Evia and Lokrida deposits are of limonitic type, with high iron and low magnesium content, whereas Kastoria laterites are of saprolitic type with high magnesium and low iron content. Greek laterites have been processed for more than 50 years at the Larco S.A. pyrometallurgical plant at Larymna to produce FeNi containing 18–20% Ni [25,26]. The hydrometallurgical treatment of these Greek ores, with atmospheric and pressure H2SO4 leaching, was first studied in the mid-1980s at National Technical University of Athens (Athens, Greece) [27,28,29,30]. Later, at the same institution, research efforts were focused on heap leaching [31,32]. The developed approach, through laboratory and pilot studies, involved the use of H2SO4, purification of the pregnant leach solution (PLS) by chemical precipitation, and, finally, recovery of Ni and Co [33]. No further studies to assess the potential of leaching of Greek laterites were then carried out, while in the last years the quality of the ores has gradually dropped. Thus, the aim of this study is to investigate the potential of column leaching for the treatment of low-grade Greek limonitic laterite and also to assess the effect of the addition of Na2SO3 in the leaching solution on the overall efficiency of leaching.
2. Materials and Methods
The ore used in this study (~50 kg) is a low-grade (0.58 wt % Ni) laterite from Agios Ioannis (LAI), Greece, obtained from Larco S.A mines. It was dried and homogenized by the cone and quarter method, and a representative subsample was crushed to minus 16 mm using a jaw crusher. Chemical and mineralogical analyses of the ore, after pulverization with the use of a FRITSCH-BICO pulverizer (Fritsch, Dresden, Germany), and of the leaching residues, after drying overnight at 80 °C, were carried out with the use of (i) a Bruker-AXS S2 Range type X-ray fluorescence energy dispersive spectrometer (XRF-EDS) (Bruker, Karlsruhe, Germany) and (ii) an X-ray diffractometer (XRD)(Bruker AXS (D8 Advance type), Karlsruhe, Germany) (Cu tube, scanning range from 4° to 70° 2θ, step 0.02° and measuring time 0.2 s/step) with the use of the DIFFRACplus EVA v. 2006 software (Bruker, Karlsruhe, Germany) and the Powder Diffraction File (PDF-2) database(Bruker, Karlsruhe, Germany). A quantity of 2 g of each ore sample and leaching residue was also digested with the use of aqua regia and analyzed with AAS to carry out mass balance calculations [27]. Since the differences between aqua regia digestion and XRF were only minor (±5%), results presented in this paper are those derived by XRF. The functional groups present in laterite and leaching residues were identified through Fourier transform infrared (FTIR) spectroscopy using KBr pellets and a Perkin Elmer Spectrum 1000 spectrometer (Akron, OH, USA); each sample was mixed with KBr at a ratio 1:100 w/w and pressed to obtain a disc. In addition, differential scanning calorimetry and thermogravimetry (DSC/TG) were performed using a Setaram LabSys Evo TG-DTA-DSC analyzer (SETARAM Inc., Cranbury, NJ, USA). The samples were heated in a nitrogen atmosphere from 40 to 1000 °C with a heating rate of 10 °C/min. All analyses were carried out in duplicate and average values are given in this study.
Four leaching tests were carried out in columns to study the effect of H2SO4 concentration and the addition of sodium sulfite (Na2SO3) in the leaching solution on Ni, Co, and other element extractions. The leaching solutions and the process parameters considered are presented in Table 1. Laboratory Plexiglas columns with a diameter of 5 cm and height of 50 cm were used. The bed consisted of 1000 g laterite (bed height 40 cm) and two layers of 2 cm of silica sand placed at both ends of each column to act as filters. Cotton glass was also added at the bottom of each column to prevent transport of ultra-fine particles and precipitates and subsequent blockage of the columns. The leaching solution used, with an initial volume of 10 L, was either 0.5 M or 1.5 M H2SO4. The effect of the addition of 20 or 30 g/L Na2SO3 in the solution containing 1.5 M H2SO4 was also investigated. It is noted that the conditions used in the present study were the ones which were considered optimum after a series of previous tests investigating leaching of saprolitic and limonitic ores were carried out.

Table 1.
Experimental details.
The leaching solution was pumped from plastic vessels with a flowrate of 3 L/day and collected in similar vessels. The solution was pumped upwards using a variable-speed peristaltic pump (Masterflex L/S economy variable-speed drive, Cole-Parmer Instrument Co, Vernon Hills, IL, USA) at a Darcy velocity of 152.9 cm/day. This means that an input flowrate of 2.08 ± 0.1 mL/min was used. This flowrate can be also expressed as ~175.4 L/(m2·h). It is mentioned that in column leaching studies, velocities greater than 20 cm/day minimize the impact of axial (e.g. longitudinal) diffusion on transport [34]. Hence, the total empty bed contact time (EBCT), defined as the ratio of actual bed length to approach velocity, was 15.7 h [35].
The leaching solution was recycled every 3 days. Recirculation started after the entire feed solution passed through the column (laterite bed). The total duration of the tests was 33 days. At the end of the tests, columns were flushed with distilled water so that the final PLS volume was ~10 L. Samples from the outflow were taken initially every day and at later stages at the end of each cycle to determine the concentration of Ni, Co, Fe, Ca, Al, Mg, and Mn by Atomic Absorption Spectroscopy (AAS). pH and Eh measurements were carried out with the use of a WTW pH 7110 inoLab pH/Eh meter. Acid consumption was determined by calculating the acid strength of the initial and the solution at the end of each cycle by a titrimetric method, as proposed by Quaicoe et al. [19]. The residues were dried at 80 °C for 1 day before characterization with the use of analytical techniques.
All leaching tests were done twice and metal concentration values in PLS given in this paper are averages; it is noted that the difference in concentration for all metals was less than 1% in all cases.
3. Results and Discussion
3.1. Ore Characterization
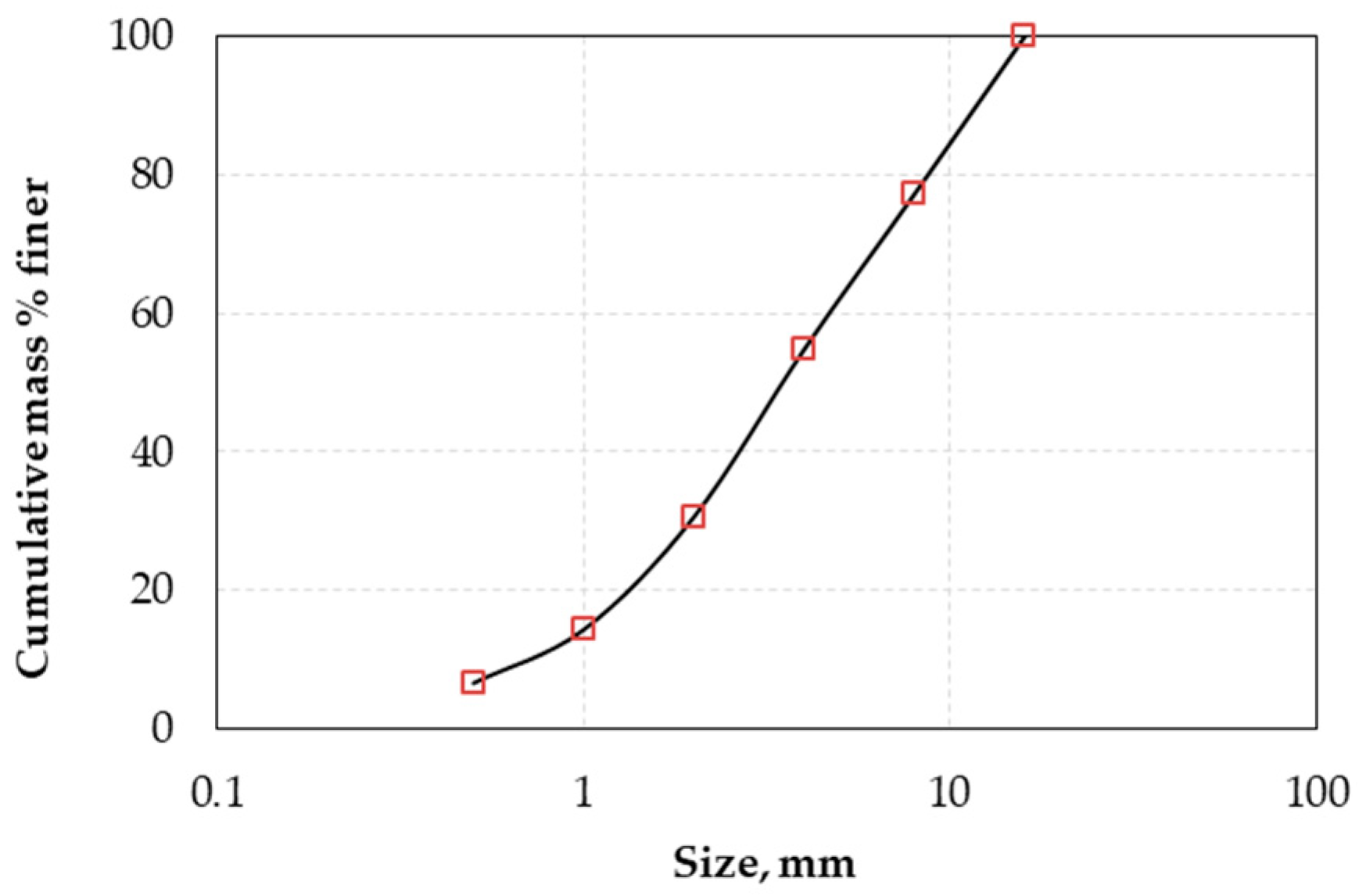
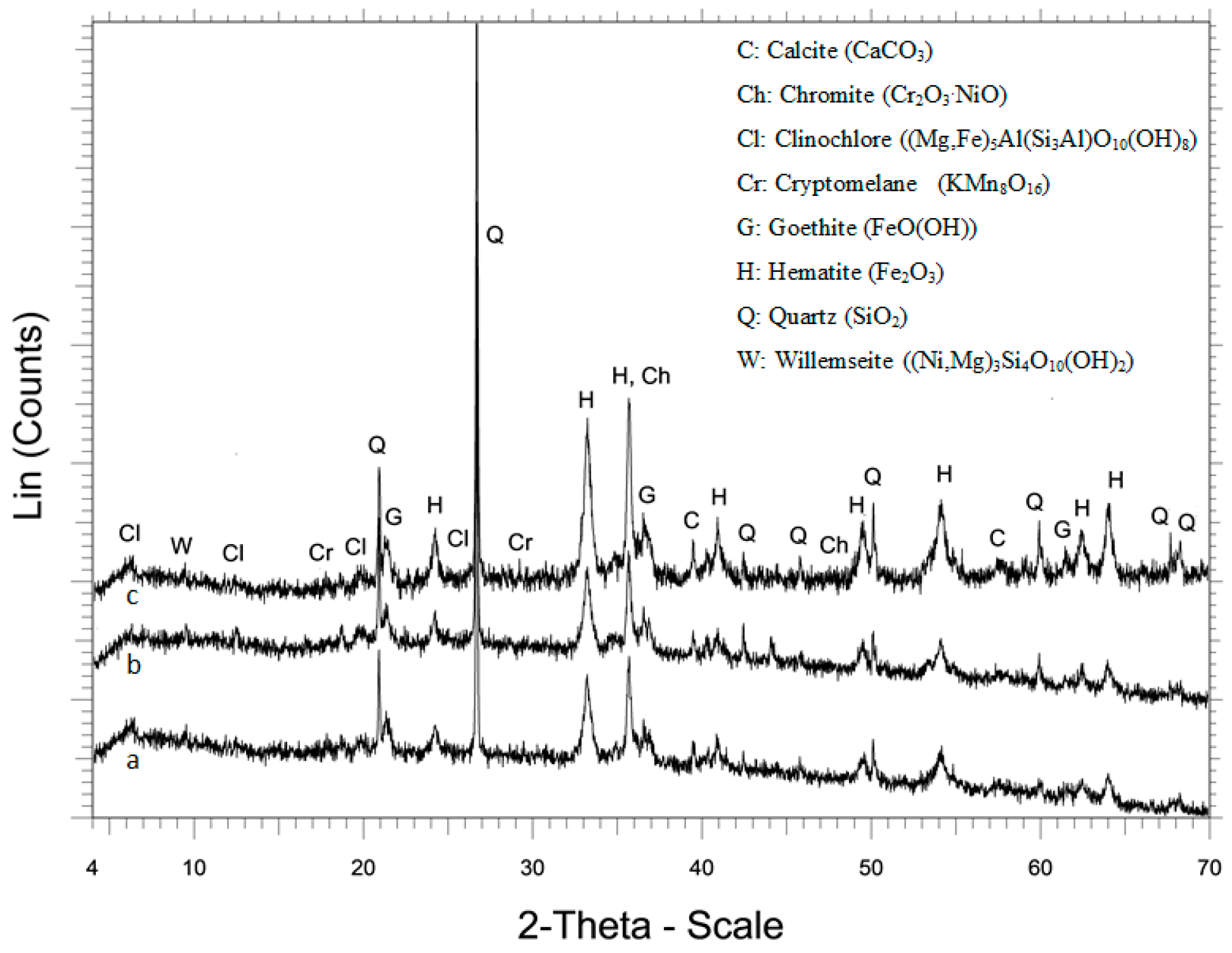
The product of the crusher was dry sieved using a series of screens with an aperture ratio of 2 and the determined particle size distribution is presented in Figure 1. The results showed that the 80% passing size (d80) of the raw material was 8.8 mm. The chemical composition of each size fraction is shown in Table 2. It is seen that grinding is not selective and does not result in noticeable nickel upgrade in the finer fractions. The XRD pattern of LAI and selected leaching residues is shown in Figure 2. Chemical and mineralogical analyses showed that the ore is of limonitic type (Fe content > 32% and MgO < 10%). The main mineral phases identified in the ore are hematite (Fe2O3), goethite (FeO(OH)), and quartz (SiO2), while calcite (CaCO3) and cryptomelane (KMn8O16) are minor phases. The main Ni-bearing phases of the ore are chromite (Cr2O3·NiO), clinochlore (Mg,Fe)5Al(Si3Al)O10(OH)8), and willemseite ((Ni,Mg)3Si4O10(OH)2). The mineralogy of the laterites used in the present study is quite similar to the mineralogy of other limonitic laterites originating either from the same area or from other countries and investigated in earlier studies [36,37].
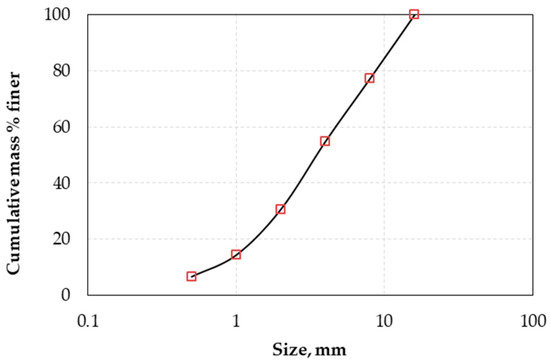
Figure 1.
Particle size distribution of laterite from Agios Ioannis (LAI) used in column leaching tests.

Table 2.
Chemical composition (% w/w) of LAI size fractions.
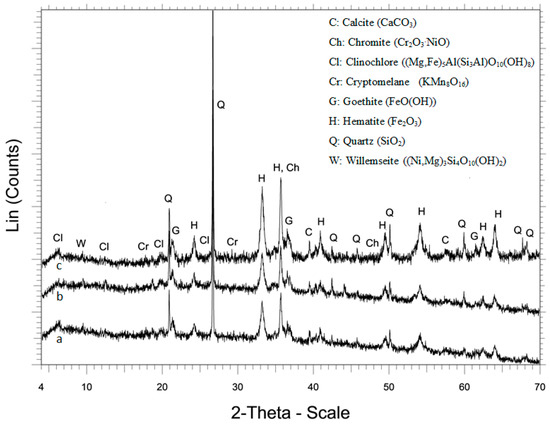
Figure 2.
XRD patterns of (a) LAI, (b) residue after leaching with 1.5 M H2SO4, and (c) residue after leaching with 1.5 M H2SO4 and 20 g/L Na2SO3.
The XRD patterns of the leaching residues, resulting from H2SO4 leaching with and without the use of Na2SO3, are shown in Figure 2b,c. The main difference in these patterns is the higher intensity of hematite peaks, especially when Na2SO3 was added in the leaching solution. No gypsum was detected in the residues, as in the case of reactor leaching of the same ore [38], due to the conditions prevailing in columns, i.e., low temperature and high H2SO4 concentration [39]; some gypsum may be formed but not enough to be detected by XRD.
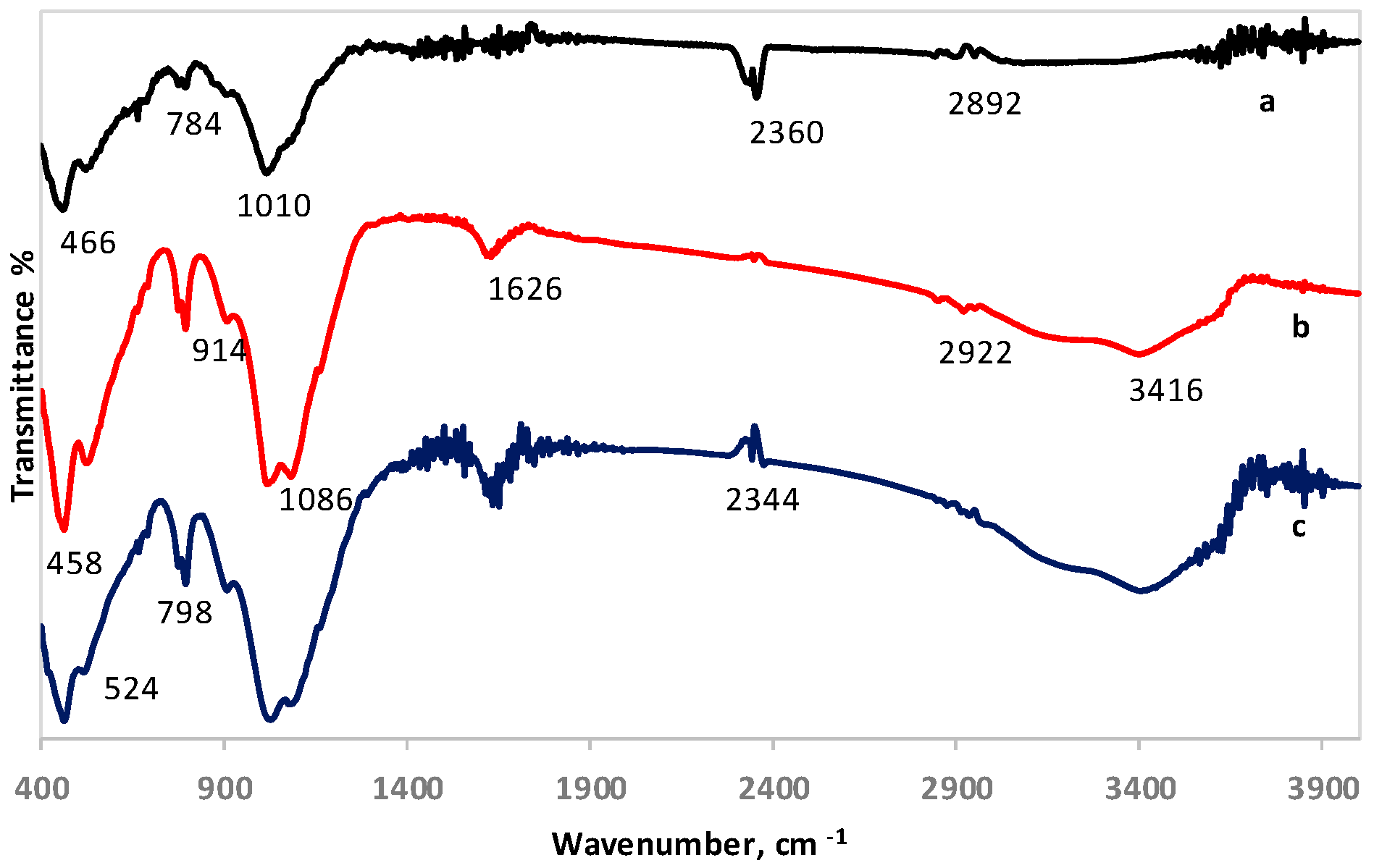
The FTIR spectra of LAI and its residues after column leaching with 1.5 M H2SO4 and 1.5 M H2SO4 with the addition of 20 g/L Na2SO3 are shown in Figure 3. In all spectra, three regions can be identified, including several low-, mid-, and higher-frequency weaker bands. The band seen at 466 cm−1 in LAI (Figure 3a), which was slightly shifted to 458 cm−1 in leaching residues (Figure 3b,c), can be attributed to the bending motions of the Al- and Si-containing phases and the formation of Fe phases. The peak at 784 cm−1 in LAI is mainly due to Si–O–Si symmetric stretching of bridging oxygen between SiO4 tetrahedra. The band seen at 1010 cm−1 in LAI, which has been shifted to higher values (1086 cm−1) in the leaching residues (Figure 3b,c), is attributed to asymmetric stretching vibrations of the silicate tetrahedral network [40]. The weak band shown at 1626 cm−1 in LAI, which becomes more intense in the leaching residues and especially in the one obtained after leaching with the addition of Na2SO3, is probably due to –OH bending vibrations. The small band seen around 2360 cm−1 in LAI is mainly associated with the infrared band position of HCO3− ions. The band shown between 2892 and 2922 cm−1 is due to hydrocarbon stretches. The broad band seen at 3416 cm−1 only in leaching residues (Figure 3b,c) corresponds to –OH stretching vibrations [41]. The band seen at 3416 cm−1 only in leaching residues may be assigned to Fe3+–OH–Fe3+ stretching and deformation vibrations [42,43].
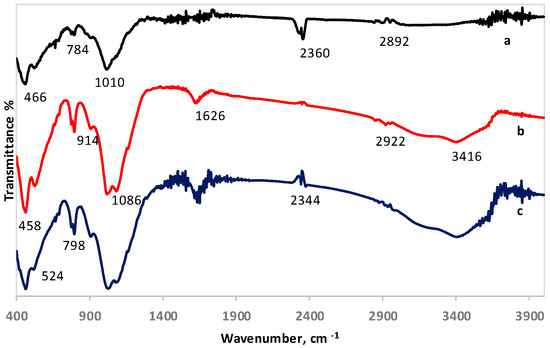
Figure 3.
FTIR spectra of (a) LAI, (b) residue after column leaching with 1.5 M H2SO4, and (c) residue after column leaching with 1.5 M H2SO4 with 20 g/L Na2SO3.
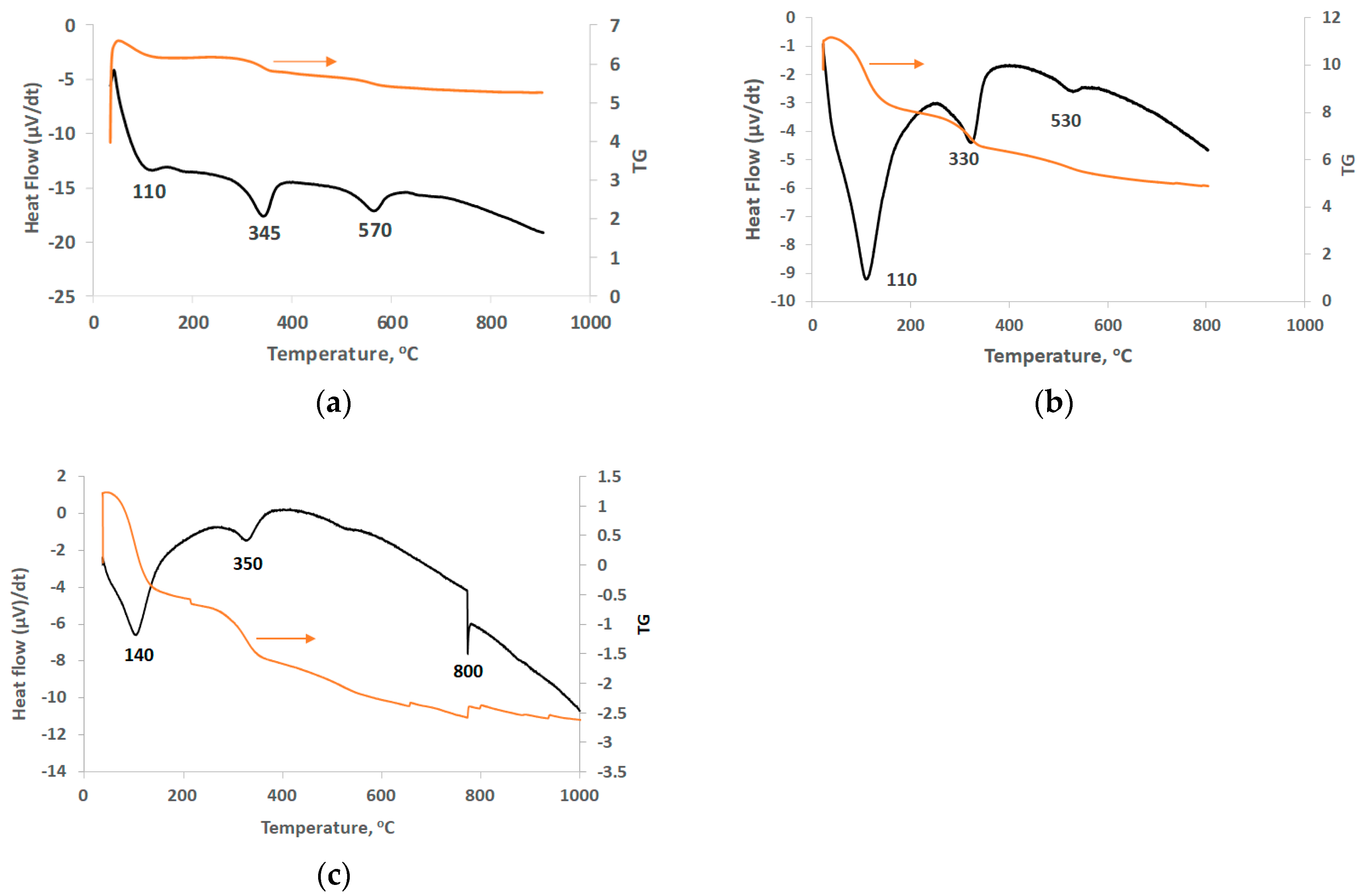
The behavior of LAI and its residues during heating was investigated through DSC/TG analysis (Figure 4). The peaks at 110 °C and 140 °C in all samples (Figure 4a–c) are due to loss of free water. The peaks seen between 330 and 350 °C, also in all samples, are associated with the removal of crystalline water and OH− group from the structure of goethite as well as the formation of hematite [44]. This peak was shifted to a higher temperature (350 °C) in the residue obtained after leaching with the use of Na2SO3, probably due to the higher degree of crystallinity of goethite [45]. The peaks at 530 and 570 °C (Figure 4a,b) are due to phase transformations of iron, silica, and calcium compounds; these peaks are almost invisible in the leaching residue obtained after leaching with the addition of Na2SO3. The exothermic peak at 800 °C (Figure 4c) is associated with recrystallization of forsterite (Mg2SiO4) and transformation of NiSO4 and CoSO4 to NiO and CoO, respectively [45,46]. Concerning TG analysis, the total weight loss of LAI was almost 15% (Figure 4a) and increased to 40% and 63% for the residues obtained after leaching with 1.5 M H2SO4 in the absence/presence of 20 g/L Na2SO3 (Figure 4b,c, respectively).
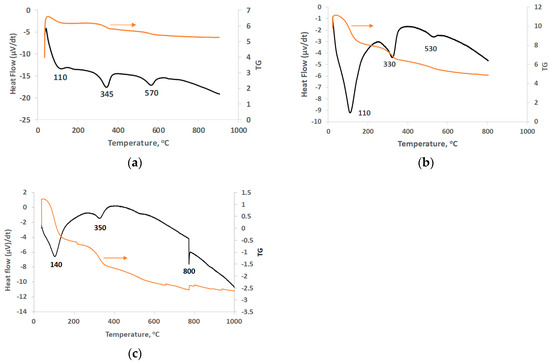
Figure 4.
DSC/TG curves of (a) LAI, (b) residue after leaching with 1.5 M H2SO4, and (c) residue after leaching with 1.5 M H2SO4 with 20 g/L Na2SO3.
3.2. Leaching Efficiency
Figure 5, Figure 6, Figure 7, Figure 8, Figure 9, Figure 10 and Figure 11 show the evolution of metal extractions (Ni, Co, Fe, Mg, Al, Ca, and Mn) versus time (in days) during leaching for the different column tests. It is seen that the extraction percentage for each metal increases with time but the leaching rate differs and depends on the conditions prevailing in each column.
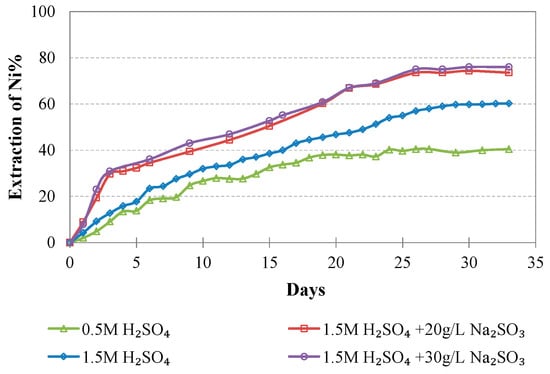
Figure 5.
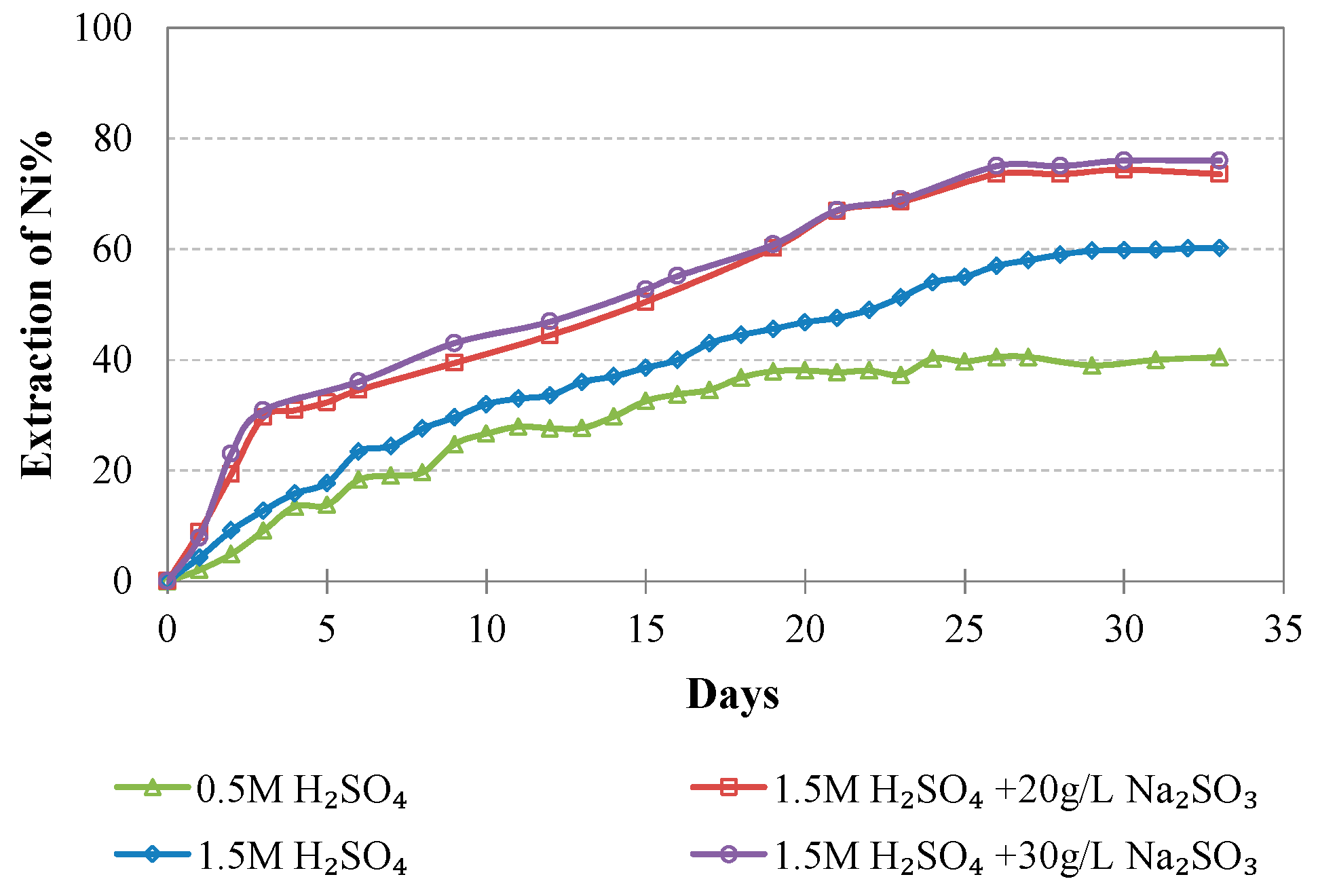
Evolution of % Ni extraction vs time during LAI leaching for different tests.
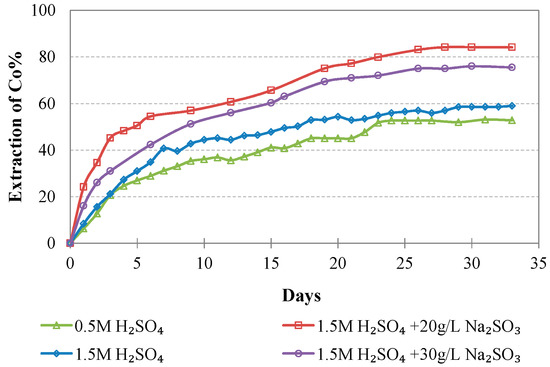
Figure 6.
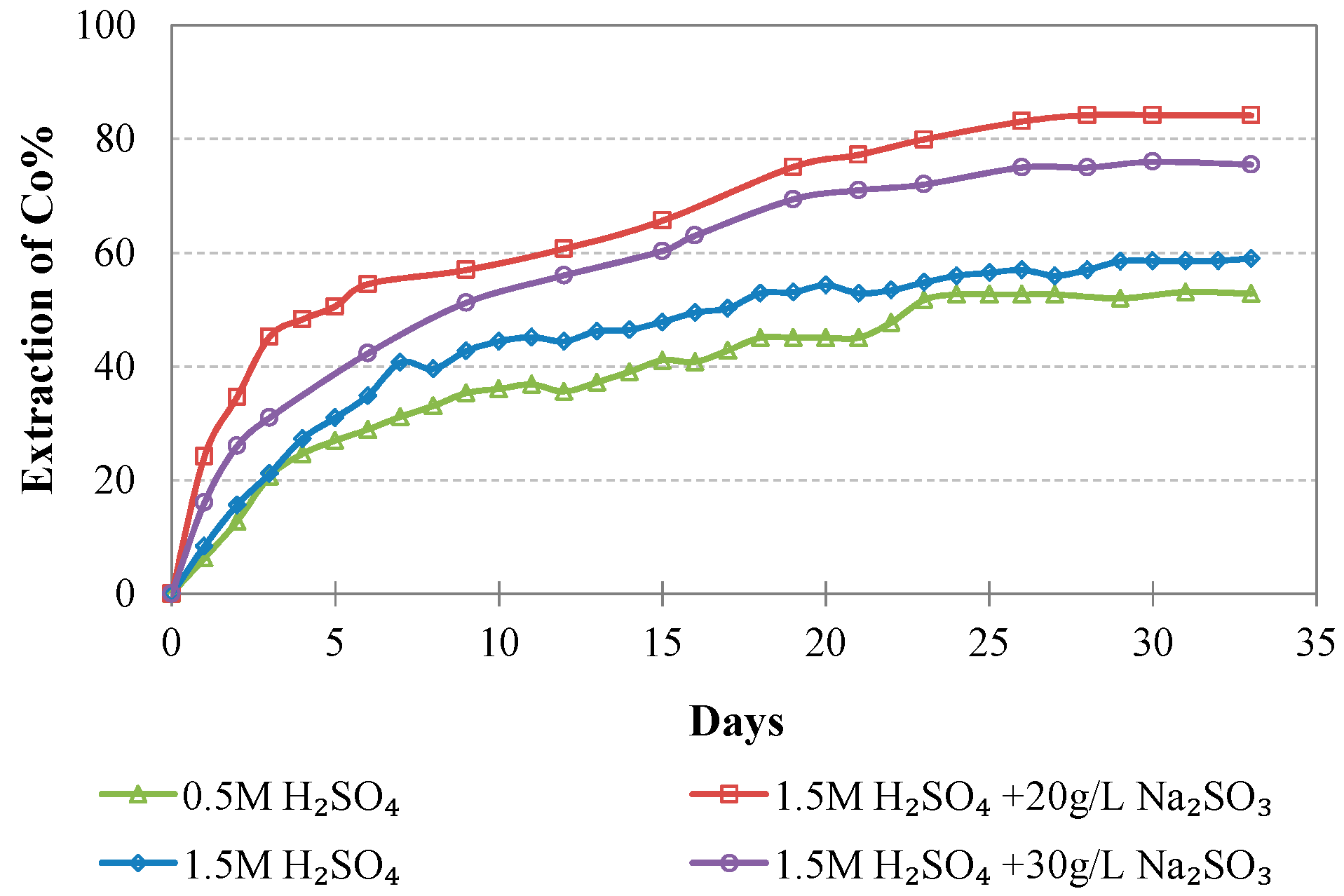
Evolution of % Co extraction vs time during LAI leaching for different tests.
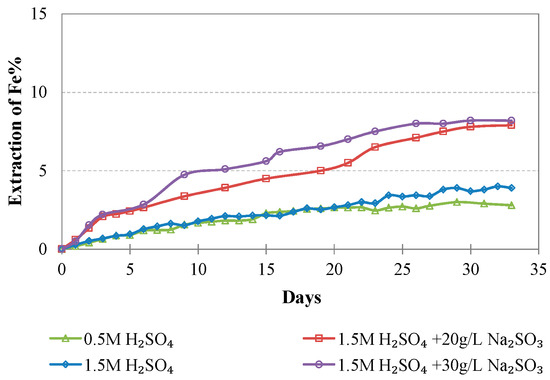
Figure 7.
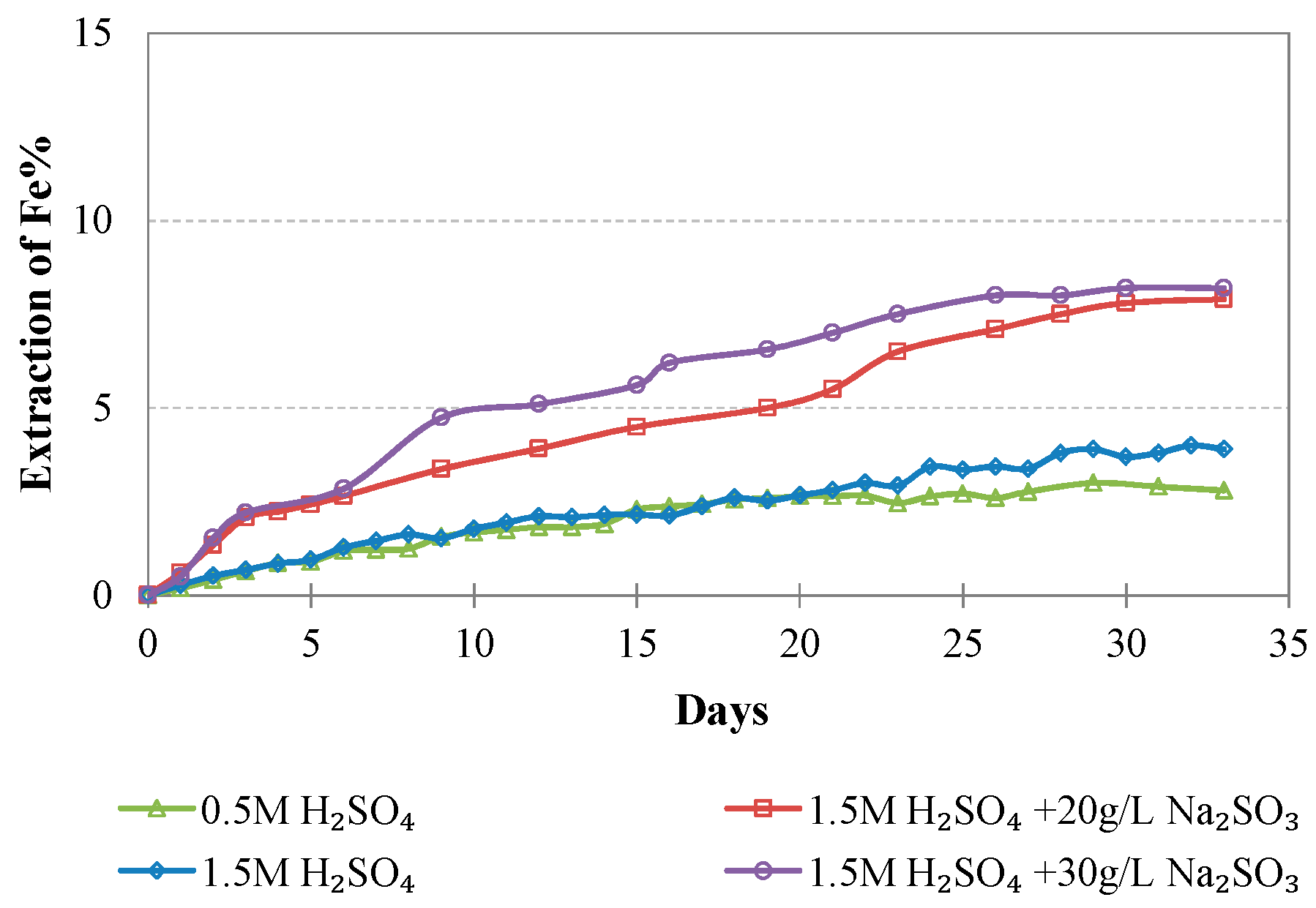
Evolution of % Fe extraction vs time during LAI leaching for different tests.
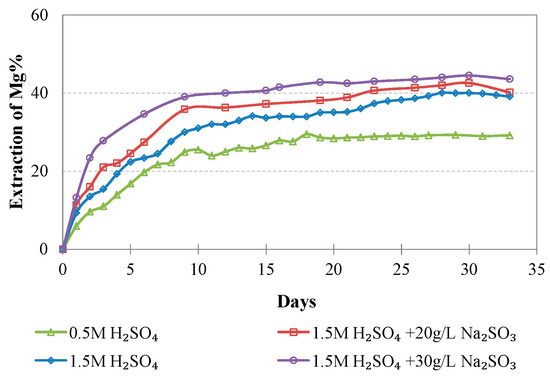
Figure 8.
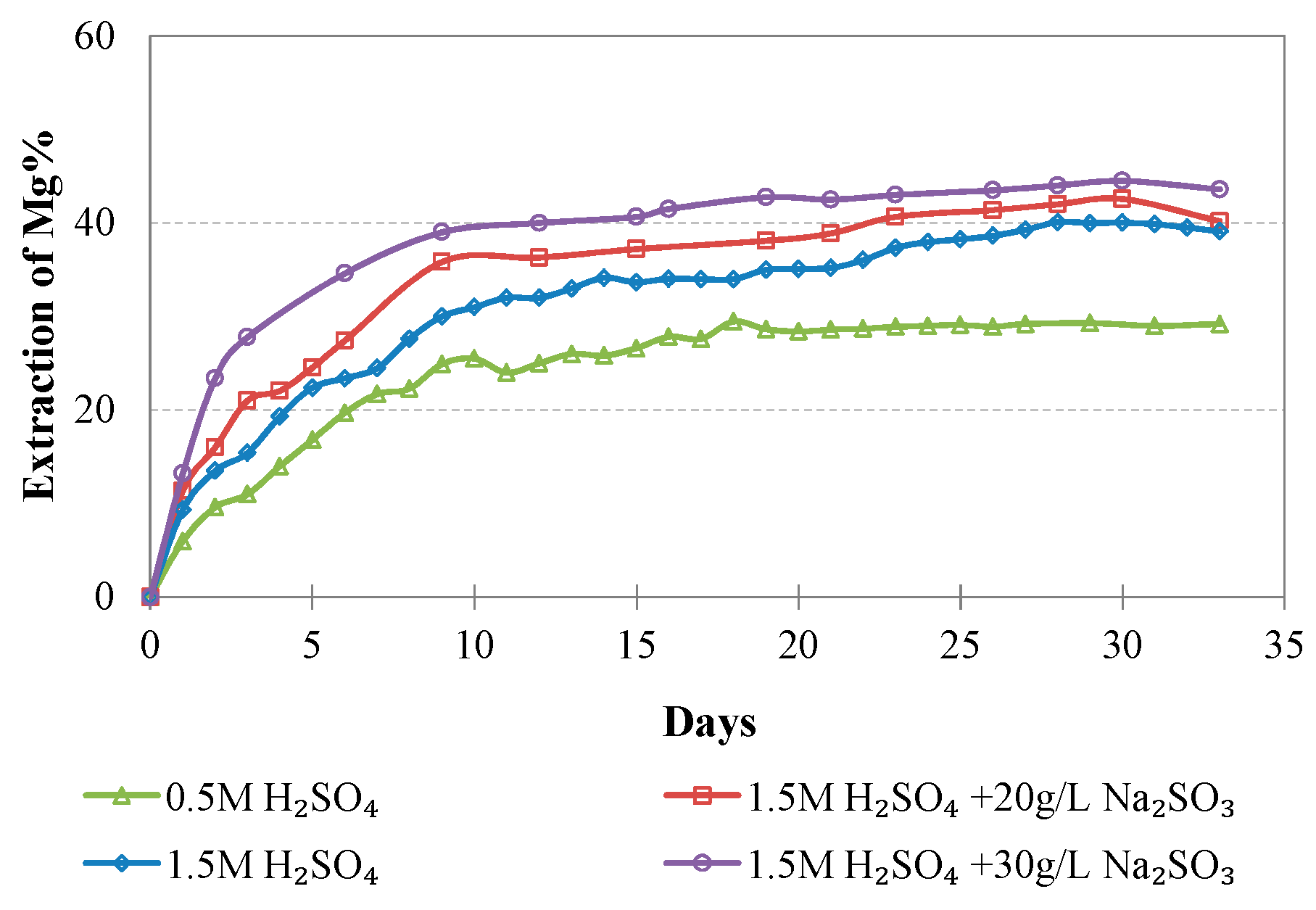
Evolution of % Mg extraction vs time during LAI leaching for different tests.
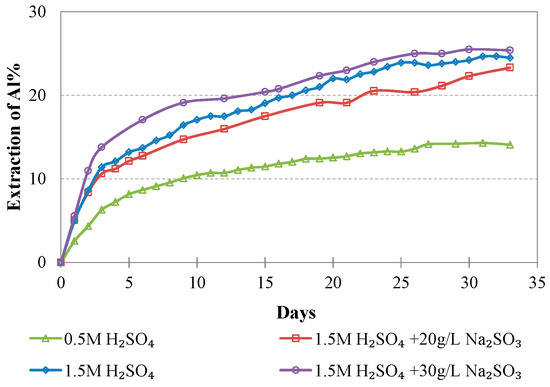
Figure 9.
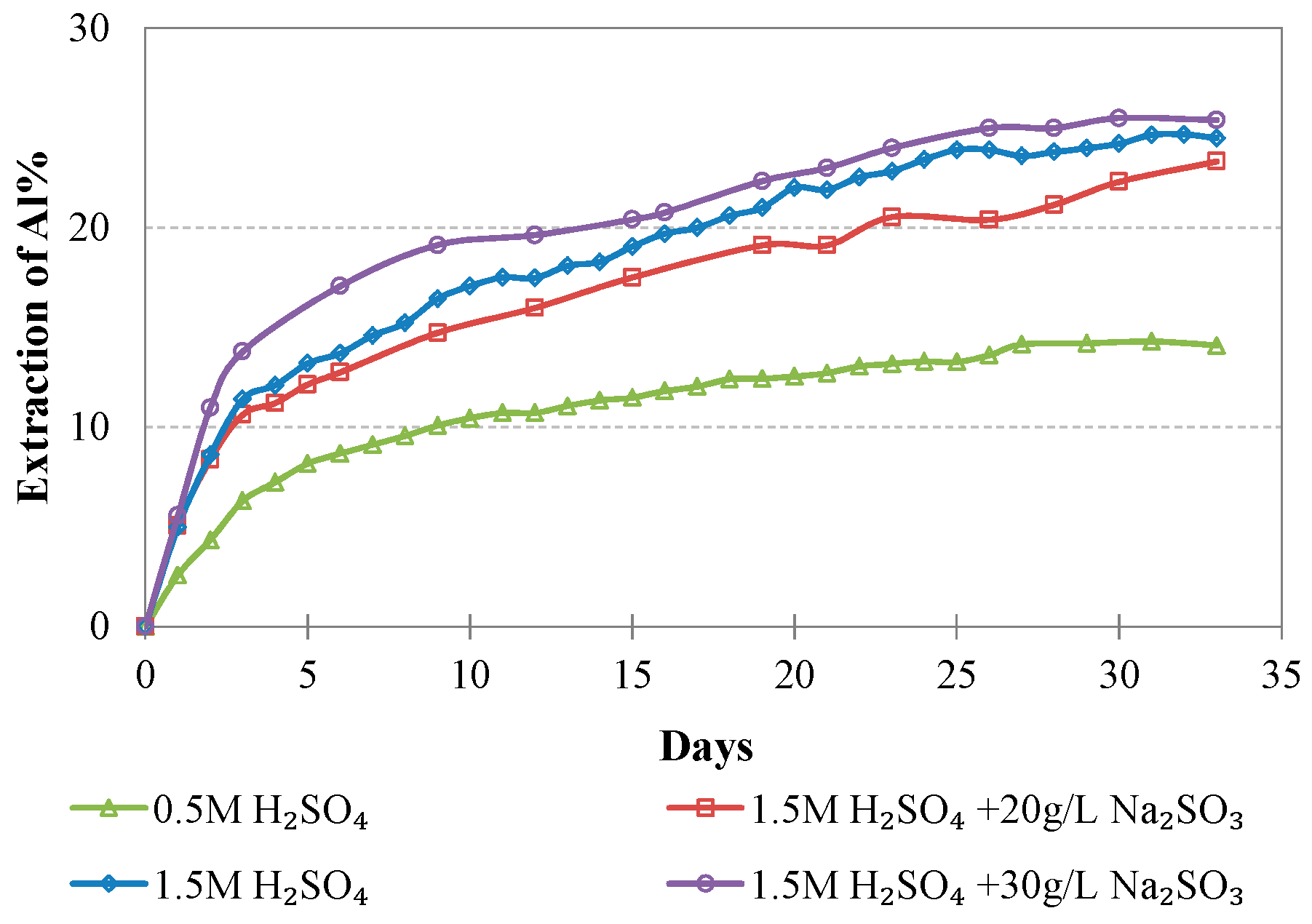
Evolution of % Al extraction vs time during LAI leaching for different tests.
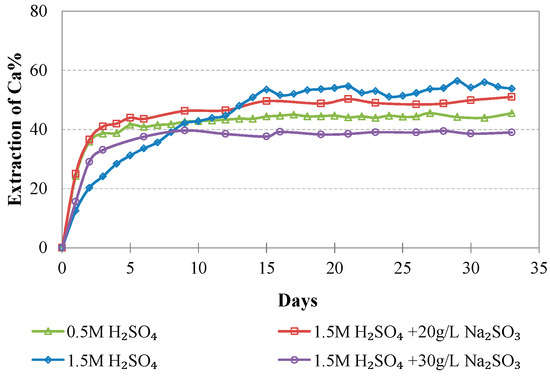
Figure 10.
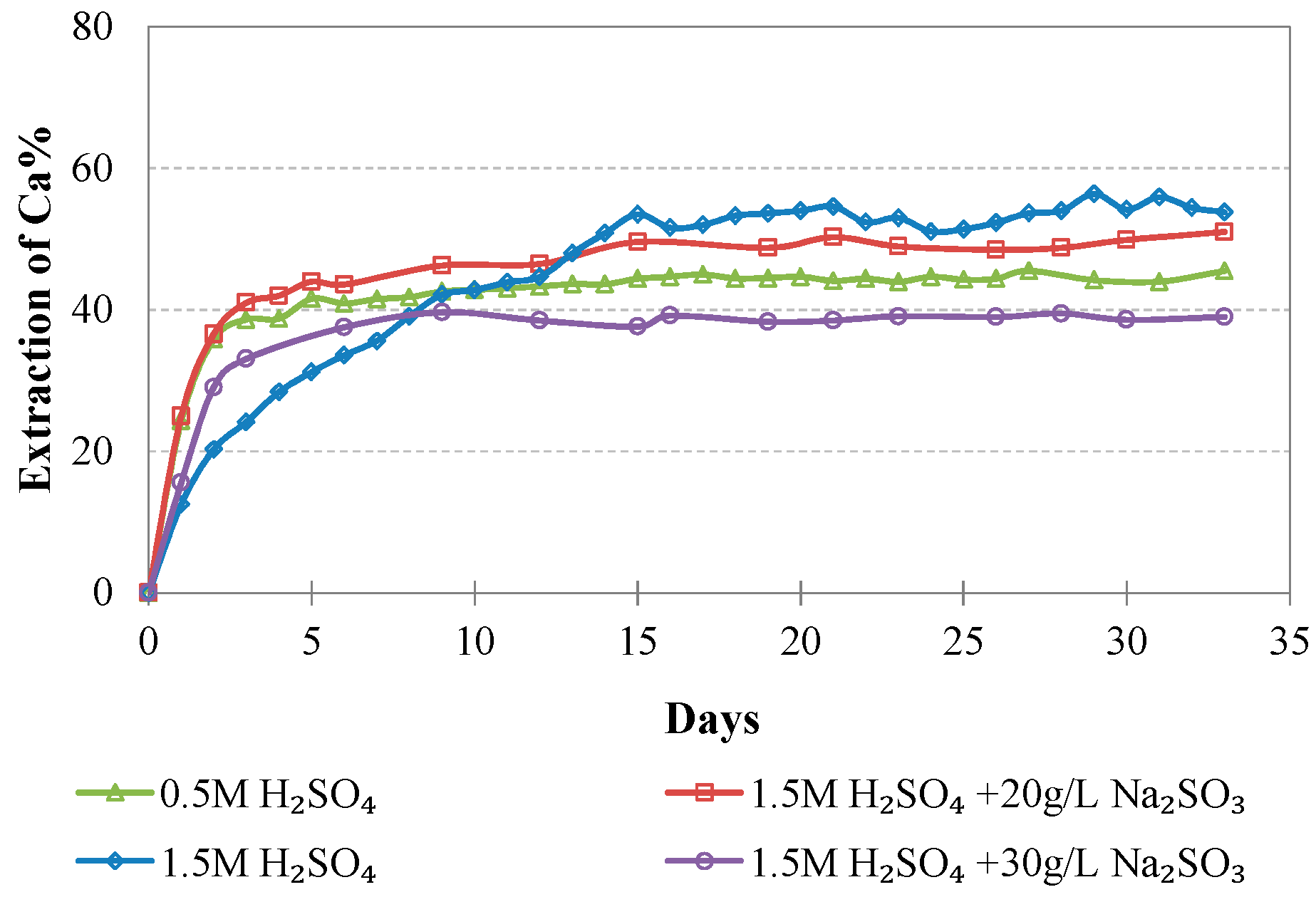
Evolution of % Ca extraction vs time during LAI leaching for different tests.
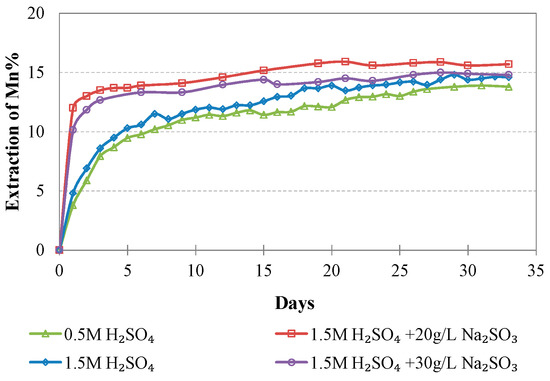
Figure 11.
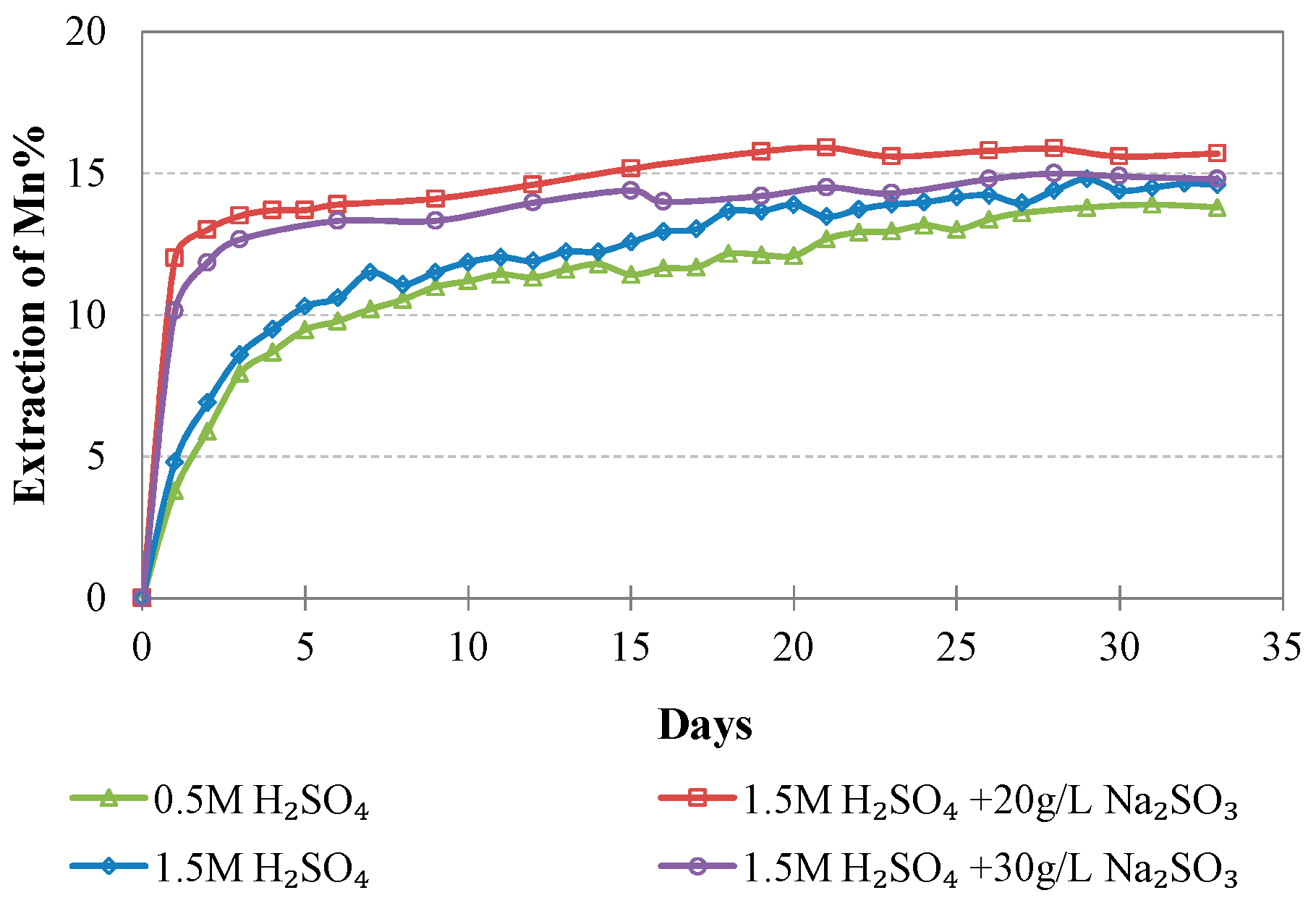
Evolution of % Mn extraction vs time during LAI leaching for different tests.
The extraction of Ni and Co when 0.5 M H2SO4 is used is rather low and does not exceed 40 and 55%, respectively; this is also due to the fact that some acid is consumed for the solubilization of Mg and Ca compounds present in the ore. Increase of the acid strength to 1.5 M improves Ni and Co extractions to 60.2% and 59.0% for Ni and Co, respectively. Further increase of the Ni and Co extractions is shown when 20 g/L of Na2SO3 is added to the leaching solution, reaching 73.5% and 84.1%, respectively. Increase of the addition of Na2SO3 to 30 g/L only marginally affects extraction of Ni and Co: Ni extraction increases from 73.5 to 76.0%, whereas Co extraction decreases from 84.1 to 75.5%; this decrease may be due to the formation of neophases and their precipitation in the residues.
The extraction of Ni and Co is increased in the presence of Na2SO3 as a result of the following reactions. In the presence of H2SO4, Na2SO3 reacts with H+ ions to form H2SO3, as shown in Reaction (1). Then, H2SO3 is dissociated according to Reaction (2).
Goethite reacts according to the following reactions ((3) and (4)) and, thus, nickel associated to this mineral phase is extracted.
The SO2(aq) generated from Reaction (2) will lower the potential of Reactions (3) and (4) and accelerate Fe extraction and Ni liberation from goethite. An in-depth analysis of the leaching chemistry of laterites with the use of H2SO4 in the presence of H2SO3 is given in a recent paper [24].
It is seen from Reactions (3) and (4) that less acid is required for the initial dissolution of goethite, namely 0.5 or 1 mole of H2SO4 per mole of goethite, compared with the consumption of acid without the presence of H2SO3, which is 1.5 moles per mole of goethite [47], as shown in Reaction (5).
Iron extraction is, in general, very low; however, the increase of the acid strength and the addition of Na2SO3 result in an increase of Fe extraction which does not exceed 8.2%. It is known that Fe extraction during laterite leaching is the main parameter that defines the selectivity of the process. In this study, the low Fe extraction is due to the room temperature used, the use of columns instead of stirred reactors, the coarser feed size used in comparison to the size used in agitated leaching, and the precipitation of iron compounds during leaching. In the study carried out by Luo et al. [24] and involving leaching of a limonitic laterite with liquid/solid (L/S) ratio 10:1 and 12% (w/w) H2SO4 at 90 °C with or without the use of Na2SO3, Fe extraction after 6 h was much higher and reached 70% and 55%, respectively. It is known that in the conditions prevailing in our experiments, iron can be removed through the formation of hydronium jarosites and ferrihydrites. The formation of hydronium jarosites is favored at very low pH values, i.e., pH = 0.2, whereas hematite can only be formed when tests are carried out in much higher temperatures [44].
The maximum extractions of the other three elements that affect the selectivity of leaching, namely, Mg, Al, and Ca, do not exceed 40.2, 23.3, and 51.0%, respectively, in the optimum conditions (1.5 M H2SO4, 20 g/L Na2SO3). The low extraction percentages of these elements during laterite leaching with the use of H2SO4 is an advantage of column leaching, since the solubility of metal sulphates in water increases with the increase of temperature; on the other hand, inverse solubilities may be noticed at elevated temperatures. It is known that limonitic laterites have lower content of Ca and Mg and higher content of Al compounds compared with saprolitic ores; this affects the selectivity of the process, especially when leaching is carried out in stirred reactors and higher temperature [38].
pH values were very low, close to zero, in all column leaching tests; the only exception was noted in the use of 0.5 M H2SO4, where pH increased with time to 0.36 at the end of the test. On the other hand, final Eh values decreased slightly with the addition of Na2SO3 and varied between 410 and 370 mV when 0.5 M H2SO4 and 1.5 M H2SO4 with 30 g/L Na2SO3 were used, respectively.
Table 3 presents the concentration of Ni and other main elements in the PLS in order to assess the selectivity of leaching in each case by comparing the concentration ratios of Ni/Fe, Ni/Mg, Ni/Ca, and Ni/Al. The comparisons done do not include the use of 0.5 M H2SO4, which does not result in high Ni and Co extractions, or the use of 30g/L Na2SO3, which does not seem to improve the efficiency of leaching compared with the addition of 20 g/L.

Table 3.
Comparison of pregnant leach solution (PLS) quality and selectivity of each leaching medium.
It is seen from the results that the addition of 20 g/L Na2SO3 in the leaching medium improves the selectivity of leaching compared with the test which involves only the addition of 1.5 M H2SO4, as the ratios Ni/Mg, Ni/Ca, and Ni/Al increase by almost 18, 30, and 28%, respectively. The only exception is the ratio Ni/Fe which decreases by almost 40%; this is mainly due to the much higher concentration of Fe in the PLS, which increases from 1.56 to 2.88 g/L, compared with the concentration of Ni, which also increases from 0.40 to 0.45 g/L. It is worth mentioning that the purification of PLS for the recovery of Ni and Co as well as of other useful elements including Mg, has been extensively investigated [48,49,50].
Table 4 compares the results of this study with those of other leaching studies pertinent to the leaching of limonitic laterites carried out over the last 25 years, as derived from an extensive literature search. Some data presented in this table were derived after calculations done by the authors of this study.

Table 4.
Comparison of results of various column leaching studies.
Agatzini-Leonardou and Dimaki [31] performed large column leaching tests on low-grade limonitic laterites using 0.5 M H2SO4. Extractions of 80% for Ni and 60% for Co were achieved after 90 days of leaching, while acid consumption was around 380 kg H2SO4/tonne ore. In another leaching study involving different types of laterites, Agatzini-Leonardou and Zafiratos [32] noted that the high calcite content in the ore reduces the permeability of the heap and adversely affects leaching. The recoveries achieved for the limonitic type after 10 days of leaching reached 60% for Ni and 45% for Co. The Fe/Ni ratio was reduced from 37:1 in the limonitic ore to 5:1 in the PLS. The acid consumption was around 284 kg H2SO4/tonne ore. Other studies investigated the leaching behavior of goethitic (G) and siliceous goethitic laterites (SG) [4,11,51] and found that the particle size of the agglomerated ore (<15, <2, and <0.038 mm) had a significant impact on Ni and Co extractions which increased with decreasing feed size, also resulting in reduction of acid consumption. Quite similar results were obtained in the study by Quaicoe et al. [19] who performed column leaching tests using <2 mm agglomerated goethitic ore. Extractions of 62.7% for Ni and 55.6% for Co were achieved after 100 days of leaching. The most interesting results of the present study, which involved leaching of a very poor laterite (0.58% Ni), are the good extractions of Ni and Co, which exceed 70 and 80%, respectively, when Na2SO3 is added in the leaching solution; the very low extraction of Fe, which remains below 8%; and the short duration (33 days). It is worth mentioning that the higher acid consumption noted in the present study may be decreased if bigger quantities of ore are leached in larger columns with the use of the same volume of leaching solution; this is an issue that is currently under study.
Leaching results of the present and earlier studies indicated that the variability in the efficiency of column leaching of limonitic laterites depends on the complexity of the ore and the strong bonding of Ni with iron minerals, compared with clay-like and high-magnesium ores (i.e., saprolitic) [52,53]. As mentioned in an excellent recent study [54], a full and comprehensive mineralogical characterization of the ore is necessary, especially at the mineral grain scale, in order to fully understand the overall leach behavior. The same authors also proved that the experimental scale noticeably affects the leaching behavior of ores at ambient conditions and proposed that the use of synchrotron methods is required to ascertain the coordination chemistry of key elements within both the fresh ore and leaching residues, in order to elucidate mineral formation and identify the major leaching mechanisms.
Other crucial factors that affect the economics of the process are the efficient control of Fe and other secondary elements’ co-extraction and the possibility to recover saleable byproducts, such as MgO and MnO2, during PLS purification. Finally, in order to minimize the environmental impacts and improve the environmental footprint of the process, the leaching residues may be alkali activated for the production of inorganic polymers. By taking into account that the residues contain sufficient amounts of SiO2 and Al2O3, the residues may be transformed into solid matrices comprising a Si–O–Al network and exhibiting beneficial physical and chemical properties, including high early strength, low shrinkage and porosity, as well as high fire and corrosion resistance. These new materials may find several applications, mainly in the construction sector [55,56].
4. Conclusions
The results of the present study are considered very promising and confirm the potential of column leaching of very low-grade Greek limonitic laterites with the use of 1.5 M H2SO4 and the addition of 20 g/L Na2SO3. These types of laterites cannot be treated economically with the use of pyrometallurgical techniques due to the extremely high energy requirements. The experimental results indicated high extractions of Ni and Co, namely, 73.5 and 84.1%, respectively, while the extractions of Fe, Mg, Al, and Ca were quite low, namely, 7.9, 40.2, 23.3, and 51.0%, respectively. The low extractions of the unwanted elements are the result of the ore granulometry and the low temperature used.
Further studies are required though to optimize leaching efficiency with the use of larger columns and ore quantities in order to enrich the quality of the PLS, improve selectivity, and reduce acid consumption. The use of agglomerates and the presence of catalysts to accelerate the leaching rate are also issues which are currently under study. In addition, a full and comprehensive mineralogical characterization of the ore and the leaching residues is necessary in order to fully elucidate the overall leach behavior and identify the major leaching mechanisms.
In order to minimize the environmental impacts and improve the environmental footprint of the process, the leaching residues can be alkali activated for the production of inorganic polymers and their potential use in the construction sector. This subject is also under study.
Author Contributions
K.K. designed the experiments, critically analyzed results, and reviewed the paper. E.P. performed a literature search, carried out experiments and analytical techniques, analyzed data, and wrote a first draft of the paper. O.P. carried out experiments and analyzed data. A.K. carried out analytical techniques and analyzed data.
Acknowledgments
The authors would like to acknowledge the financial support of European Commission in the frame of Horizon 2020 project “Metal recovery from low-grade ores and wastes”, www.metgrowplus.eu, Grant Agreement n° 690088.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kursunoglu, S.; Kaya, M. Atmospheric pressure acid leaching of Caldag lateritic nickel ore. Int. J. Miner. Process. 2016, 150, 1–8. [Google Scholar] [CrossRef]
- Zeng, X.; Zhan, C.; Lu, J.; Amine, K. Stabilization of a high capacity and high-power nickel-based cathode for Li-Ion batteries. Chem 2018, 4, 690–704. [Google Scholar] [CrossRef]
- Mohammadreza, F.; Mohammad, N.; Ziaeddin, S.S. Nickel extraction from low grade laterite by agitation leaching at atmospheric pressure. Int. J. Min. Sci. Technol. 2014, 24, 543–548. [Google Scholar] [CrossRef]
- Nosrati, A.; Quast, K.; Xu, D.; Skinner, W.; Robinson, D.J.; Addai-Mensah, J. Agglomeration and column leaching behaviour of nickel laterite ores: Effect of ore mineralogy and particle size distribution. Hydrometallurgy 2014, 146, 29–39. [Google Scholar] [CrossRef]
- MacCarthy, J.; Nosrati, A.; Skinner, W.; Addai-Mensah, J. Atmospheric acid leaching mechanisms and kinetics and rheological studies of a low grade saprolitic nickel laterite ore. Hydrometallurgy 2016, 160, 26–37. [Google Scholar] [CrossRef]
- Dalvi, A.D.; Bacon, W.G.; Osborne, R.C. The past and the future of nickel laterites. In Proceedings of the Prospectors & Developers Association of Canada (PDAC) 2004 International Convention, Trade Show & Investors Exchange, Toronto, ON, Canada, 7–10 March 2004. [Google Scholar]
- Girgin, I.; Obut, A.; Ucyildiz, A. Dissolution behaviour of a Turkish lateritic nickel ore. Miner. Eng. 2011, 24, 603–609. [Google Scholar] [CrossRef]
- Onal, M.A.R.; Topkaya, Y.A. Pressure acid leaching of Çaldağ lateritic nickel ore: An alternative to heap leaching. Hydrometallurgy 2014, 142, 98–107. [Google Scholar] [CrossRef]
- Takeda, O.; Lu, X.; Miki, T.; Nakajima, K. Thermodynamic evaluation of elemental distribution in a ferronickel electric furnace for the prospect of recycling pathway of nickel. Resour. Conserv. Recycl. 2018, 133, 362–368. [Google Scholar] [CrossRef]
- Wang, Z.; Chu, M.; Liu, Z.; Wang, H.; Zhao, W.; Gao, L. Preparing Ferro-Nickel Alloy from Low-Grade Laterite Nickel Ore Based on Metallized Reduction–Magnetic Separation. Metals 2017, 7, 313. [Google Scholar] [CrossRef]
- Quast, K.; Xu, D.; Skinner, W.; Nosrati, A.; Hilder, T.; Robinson, D.J.; Addai-Mensah, J. Column leaching of nickel laterite agglomerates: Effect of feed size. Hydrometallurgy 2013, 134, 144–149. [Google Scholar] [CrossRef]
- Oxley, A.; Smith, M.E.; Caceres, O. Why heap leach nickel laterites? Miner. Eng. 2016, 88, 53–60. [Google Scholar] [CrossRef]
- Alibhai, K.A.K.; Dudeney, A.W.L.; Leak, D.J.; Agatzini, S.; Tzeferis, P. Bioleaching and bioprecipitation of nickel and iron from laterites. FEMS Microbiol. Rev. 1993, 11, 87–95. [Google Scholar] [CrossRef]
- du Plessis, C.A.; Slabbert, W.; Hallberg, K.B.; Johnson, D.B. Ferredox: A biohydrometallurgical processing concept for limonitic nickel laterites. Hydrometallurgy 2011, 109, 221–229. [Google Scholar] [CrossRef]
- Watling, H.R.; Elliot, A.D.; Fletcher, H.M.; Robinson, D.J.; Sully, D.M. Ore mineralogy of nickel laterites: Controls on processing characteristics under simulated heap-leach conditions. Aust. J. Earth Sci. 2011, 58, 725–744. [Google Scholar] [CrossRef]
- Duyvesteyn, W.P.C.; Liu, H.; Davis, M.J. Heap Leaching of Nickel Containing Ore. U.S. Patent 6,312,500 B1, 6 November 2001. [Google Scholar]
- Horizonte, S.G.; Horizonte, D.O. Process for Extraction of Nickel, Cobalt, and Other Base Metals from Laterite Ores by Using Heap Leaching and Product Containing Nickel, Cobalt, and Other Metals from Laterite Ores. European Patent EP 1790739 A1, 21 June 2007. [Google Scholar]
- Elliot, A.; Fletcher, H.; Li, J.; Watling, H.; Robinson, D.J. Heap leaching of nickel laterites—A challenge and an opportunity. In Hydrometallurgy of Nickel and Cobalt 2009, Proceeding of the 39th Annual Hydrometallurgy Meeting Held in the Conjunction with the 48th Annual Conference of Metallurgists, Sudbury, ON, Canada, 23–26 August 2009; Budac, J.J., Fraser, R., Mihaylov, I., Papangelakis, V.G., Robinson, D.J., Eds.; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2009; pp. 537–549. [Google Scholar]
- Quaicoe, I.; Nosrati, A.; Skinner, W.; Addai, M.J. Agglomeration and column leaching behaviour of goethitic and saprolitic nickel laterite ores. Miner. Eng. 2014, 65, 1–8. [Google Scholar] [CrossRef]
- Das, G.K.; de Lange, J.A.B. Reductive atmospheric acid leaching of West Australian smectitic nickel laterite in the presence of sulphur dioxide and copper (II). Hydrometallurgy 2011, 105, 264–269. [Google Scholar] [CrossRef]
- Lee, H.Y.; Kim, S.G.; Oh, J.K. Electrochemical leaching of nickel from low-grade laterites. Hydrometallurgy 2005, 77, 263–268. [Google Scholar] [CrossRef]
- Senanayake, G.; Das, G.K. A comparative study of leaching kinetics of limonitic laterite and synthetic iron oxides in sulfuric acid containing sulfur dioxide. Hydrometallurgy 2004, 72, 59–72. [Google Scholar] [CrossRef]
- Youzbashi, A.A.; Dixit, S.G. Leaching of nickel from supported nickel waste catalyst using aqueous sulfur dioxide solution. Metall. Trans. B 1991, 22, 775–781. [Google Scholar] [CrossRef]
- Luo, J.; Li, G.; Rao, M.; Peng, Z.; Zhang, Y.; Jiang, T. Atmospheric leaching characteristics of nickel and iron in limonitic laterite with sulfuric acid in the presence of sodium sulfite. Miner. Eng. 2015, 78, 38–44. [Google Scholar] [CrossRef]
- Zevgolis, E.; Zografidis, C.; Halikia, I. The reducibility of the Greek nickeliferous laterites: A review. Miner. Process. Extract. Metall. 2010, 119, 9–17. [Google Scholar] [CrossRef]
- Bartzas, G.; Komnitsas, K. Life cycle assessment of FeNi production in Greece: A. case study. Resour. Conserv. Recycl. 2015, 105, 113–122. [Google Scholar] [CrossRef]
- Komnitsas, K. Kinetic Study of Laterite Leaching with Sulfuric Acid at Atmospheric Pressure. Bachelor’s Thesis, National Technical University of Athens, Athens, Greece, 1983. (In Greek). [Google Scholar]
- Komnitsas, K. High Pressure acid Leaching of Laterites. Ph.D. Thesis, National Technical University of Athens, Athens, Greece, 1988. (In Greek). [Google Scholar]
- Kontopoulos, A.; Komnitsas, K. Sulphuric acid Pressure Leaching of Low-Grade Greek Laterites. In Proceedings of the 1st International Symposium on Hydrometallurgy, Beijing, China, 12–15 October 1988; Zheng, Y.L., Xu, J.Z., Eds.; Pergamon Press: Oxford, UK, 1988; pp. 140–144. [Google Scholar]
- Panagiotopoulos, N.; Agatzini, S.; Kontopoulos, A. Extraction of nickel and cobalt from serpentinic type laterites by atmospheric pressure sulphuric acid leaching. In Proceedings of the Technical Sessions at the 115th TMS-AIME Annual Meeting, New Orleans, LA, USA, 2–6 March 1986; p. A86-30. [Google Scholar]
- Agatzini-Leonardou, S.; Dimaki, D. Heap leaching of poor nickel laterites by sulphuric acid at ambient temperature. In Hydrometallurgy ´94; Springer: Dordrecht, The Netherlands, 1994; pp. 193–208. [Google Scholar]
- Agatzini-Leonardou, S.; Zafiratos, I.G. Beneficiation of a Greek serpentinic nickeliferous ore part II. Sulphuric acid heap and agitation leaching. Hydrometallurgy 2004, 74, 267–275. [Google Scholar] [CrossRef]
- Agatzini-Leonardou, S.; Zafiratos, J.G.; Spathis, D. Beneficiation of a Greek serpentinic nickeliferous ore—Part I: Mineral processing. Hydrometallurgy 2004, 74, 259–265. [Google Scholar] [CrossRef]
- Komnitsas, K.; Bartzas, G.; Paspaliaris, I. Inorganic contaminant fate assessment in zero-valent iron treatment walls. Environ. Forensics 2006, 7, 207–217. [Google Scholar] [CrossRef]
- Komnitsas, K.; Bartzas, G.; Paspaliaris, I. Modeling of reaction front progress in fly ash permeable reactive barriers. Environ. Forensics 2006, 7, 219–231. [Google Scholar] [CrossRef]
- Alevizos, G. Mineralogy, Geochemistry and Genesis of the Sedimentary Nickeliferous Iron-Ores of Locris (Central Greece). Ph.D. Thesis, Technical University of Crete, Chania, Greece, 1997. [Google Scholar]
- Li, G.; Rao, M.; Jiang, T.; Huang, Q.; Peng, Z. Leaching of limonitic laterite ore by acidic thiosulfate solution. Miner. Eng. 2011, 24, 859–863. [Google Scholar] [CrossRef]
- Mystrioti, C.; Papassiopi, N.; Xenidis, A.; Komnitsas, K. Comparative Evaluation of Sulfuric and Hydrochloric Acid Atmospheric Leaching for the Treatment of Greek Low Grade Nickel Laterites. In Extraction 2018, Proceedings of the First Global Conference on Extractive Metallurgy, The Minerals, Metals & Materials Series, Ottawa, QC, Canada, 26–29 August 2018; Davis, B.R., Moats, M.S., Wang, S., Gregurek, D., Kapusta, J., Battle, T.P., Schlesinger, M.E., Flores, G.R.A., Jak, E., Goodall, G., et al., Eds.; Springer: Cham, Switzerland, 2018; pp. 1753–1764. [Google Scholar]
- Farrah, H.E.; Lawrance, G.A.; Wanless, E.J. Solubility of calcium sulfate salts in acidic manganese sulfate solutions from 30 to 105 °C. Hydrometallurgy 2007, 86, 13–21. [Google Scholar] [CrossRef]
- Rinaudo, C.; Gastaldi, D.; Belluso, E. Characterization of chrysotile, antigorite, and lizardite by FT-Raman spectroscopy. Can. Mineral. 2003, 41, 883–890. [Google Scholar] [CrossRef]
- Madejov, J.; Janek, M.; Komadel, P.; Herbert, H.J.; Moog, H.C. FTIR analyses of water in MX-80 bentonite compacted from high salinary salt solution systems. Appl. Clay Sci. 2002, 20, 255–271. [Google Scholar] [CrossRef]
- Petit, S.; Decarreau, A. Hydrothermal (200 °C) synthesis and crystal chemistry of iron rich kaolinites. Clay Miner. 1990, 25, 181–196. [Google Scholar] [CrossRef]
- Delineau, T.; Allard, T.; Muller, J.P.; Barres, O.; Yvon, J.; Cases, J.M. FTIR reflectance vs. EPR studies of structural iron in kaolinites. Clays Clay Miner. 1994, 42, 308–320. [Google Scholar] [CrossRef]
- Stopić, S.; Friedrich, B.; Fuchs, R. Sulphuric acid leaching of the Serbian nickel lateritic ore. Erzmetall 2003, 56, 198–203. [Google Scholar]
- Ma, B.; Yang, W.; Pei, Y.; Wang, C.; Jin, B. Effect of activation pretreatment of limonitic laterite ores using sodium fluoride and sulfuric acid on water leaching of nickel and cobalt. Hydrometallurgy 2017, 169, 411–417. [Google Scholar] [CrossRef]
- Luo, W.; Feng, Q.; Ou, L.; Zhang, G.; Chen, Y. Kinetics of saprolitic laterite leaching by sulphuric acid at atmospheric pressure. Miner. Eng. 2010, 23, 458–462. [Google Scholar] [CrossRef]
- Georgiou, D.; Papangelakis, V. Sulphuric acid pressure leaching of a limonitic laterite: Chemistry and kinetics. Hydrometallurgy 1998, 49, 23–46. [Google Scholar] [CrossRef]
- Agatzini-Leonardou, S.; Tsakiridis, P.E.; Oustadakis, P.; Karidakis, T.; Katsiapi, A. Hydrometallurgical process for the separation and recovery of nickel from sulphate heap leach 271 liquor of nickeliferous laterite ores. Miner. Eng. 2009, 22, 1181–1192. [Google Scholar] [CrossRef]
- Mihaylov, I.; Krause, E.; Okita, Y.; Perraud, J.J. The Development of a novel hydrometallurgical process for nickel and cobalt recovery from Goro laterite ore. CIM Bull. 2000, 93, 124–130. [Google Scholar]
- Ritcey, G.M.; Hayward, N.L.; Salinovich, T. The Recovery of Nickel and Cobalt from Lateritic Ores. Australian Patent AU-B-40890, 18 July 1996. [Google Scholar]
- Xu, D.; Liu, L.X.; Quast, K.; Addai-Mensah, J.; Robinson, D.J. Effect of nickel laterite agglomerate properties on their leaching performance. Adv. Powder Technol. 2013, 24, 750–756. [Google Scholar] [CrossRef]
- McDonald, R.G.; Whittington, B.I. Atmospheric acid leaching of nickel laterites review. Part I. Sulphuric acid technologies. Hydrometallurgy 2008, 91, 35–55. [Google Scholar] [CrossRef]
- MacCarthy, J.; Nosrati, A.; Skinner, W.; Addai-Mensah, J. Temperature Influence of Atmospheric Acid Leaching Behaviour of Saprolitic Nickel Laterite Ore. In Proceedings of the Chemeca 2013: Challenging Tomorrow, Brisbane, Australia, 29 September–2 October 2013; Wang, L., Ed.; Engineers Australia: Brisbane, Australia, 2013; pp. 439–443. [Google Scholar]
- Hunter, H.M.A.; Herrington, R.J.; Oxley, E.A. Examining Ni-laterite leach mineralogy & chemistry—A holistic multi-scale approach. Miner. Eng. 2013, 54, 100–119. [Google Scholar]
- Komnitsas, K.; Zaharaki, D. Geopolymerisation: A review and prospects for the minerals industry. Miner. Eng. 2007, 20, 1261–1277. [Google Scholar] [CrossRef]
- Zaharaki, D.; Komnitsas, K. Valorization of construction and demolition (C&D) and industrial wastes through alkali activation. Constr. Build. Mater. 2016, 121, 686–693. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).