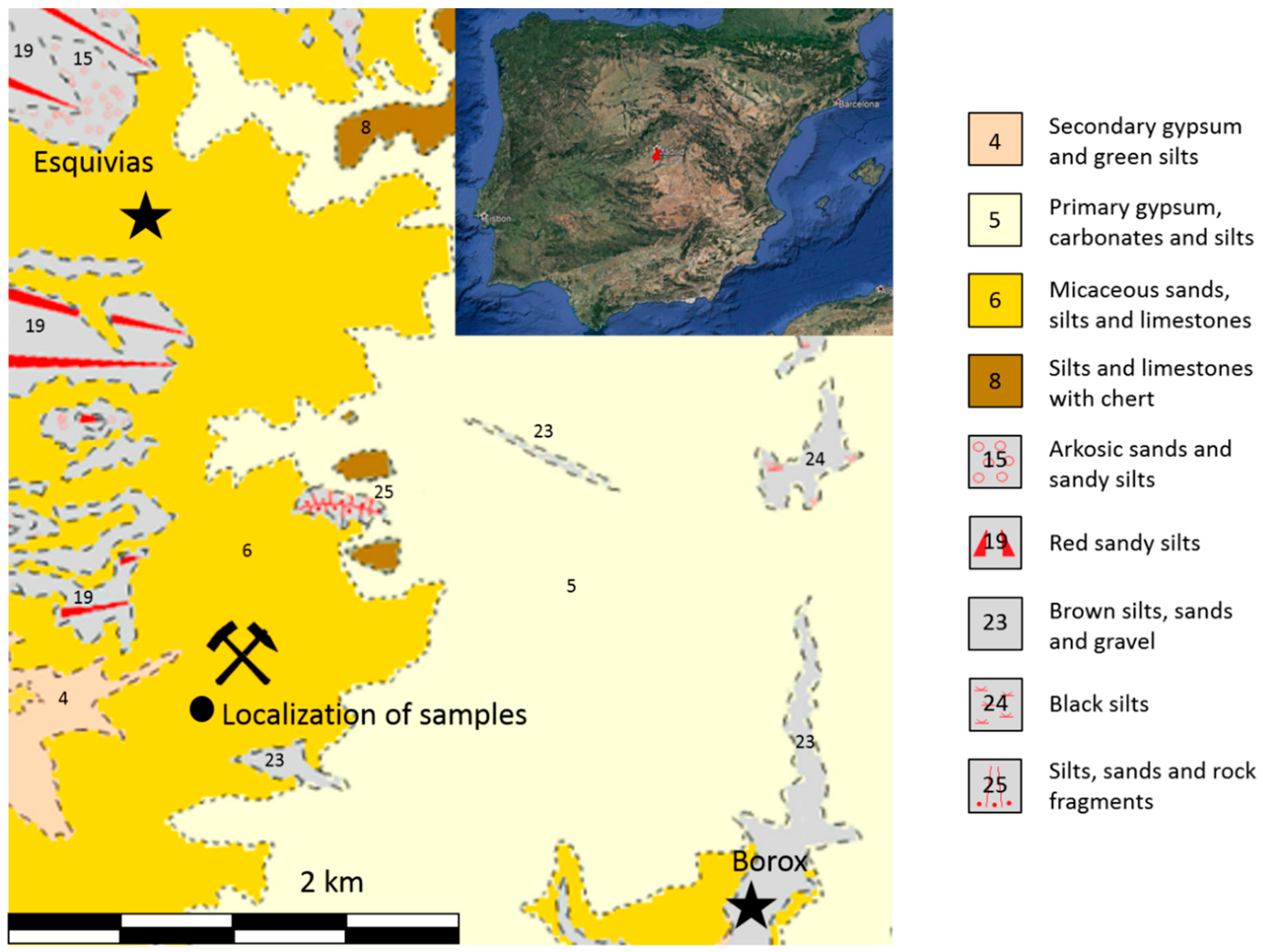
3.2. Geochemistry
We analyzed both major, including F (
Table 2), and trace elements through ICP-MS. The trace elements considered were the high field strength elements (HFSE), transition trace elements (TTE), large-ion litophile elements (LILE) and rare earth elements (REE), as well as other trace elements such as Be, Ga, Ge, As, Y, Mo, Ag, In, Sn, Sb, W and Bi (
Table 3), of 31 representative samples distributed vertically and horizontally. These analyses allowed the interpretation of the origin of the detrital materials through different means, such as the content in heavy non-soluble elements or different relations such as Th/U.
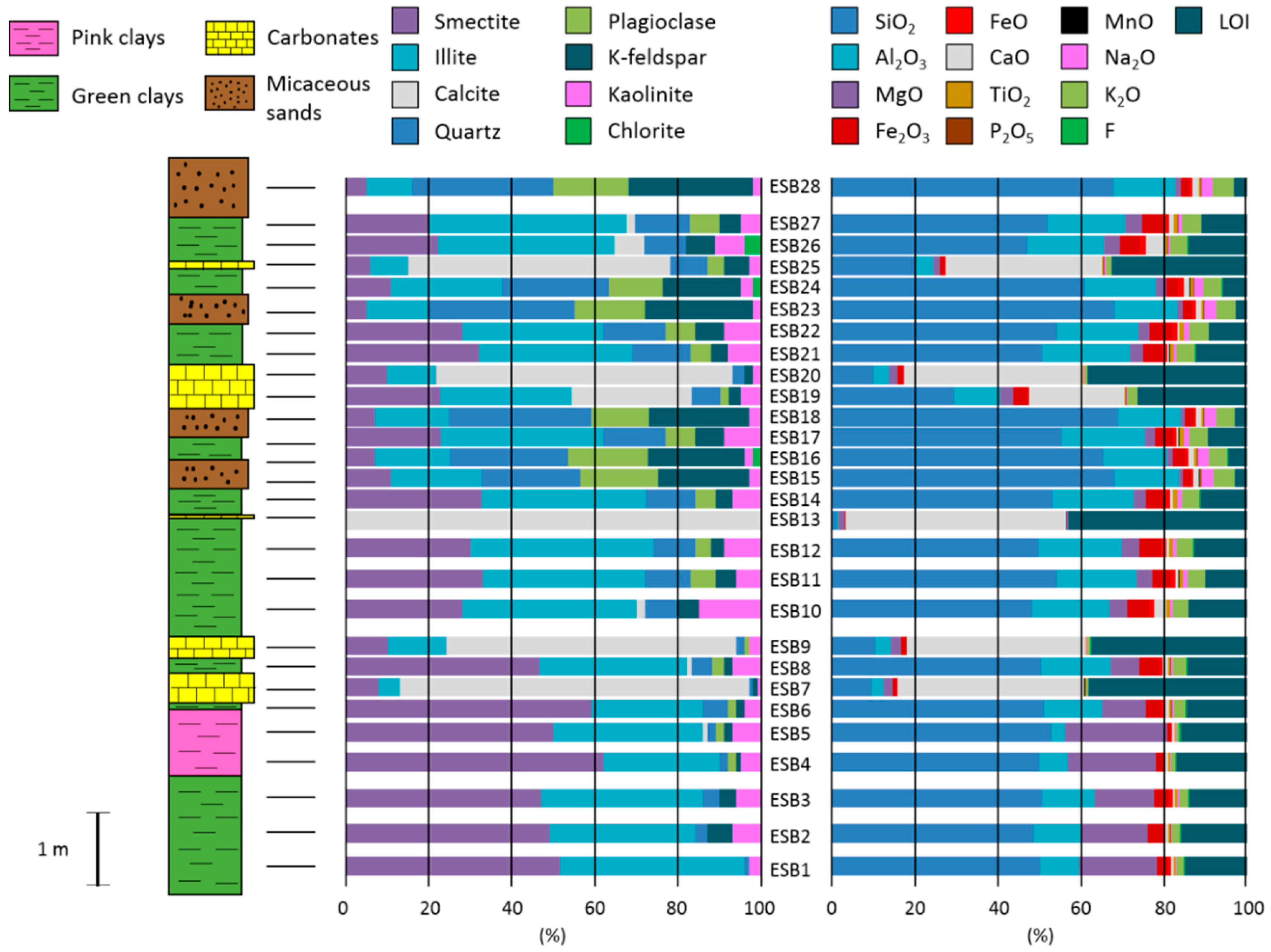
Regarding major elements, we observed that the composition matches with the mineralogy (
Figure 2). Sandy samples are characterized by a higher concentration of SiO
2 while carbonatic samples have the highest content of CaO and MnO. Illitic samples present the highest content in Al
2O
3 and TiO
2, and also tend to have a higher concentration of Fe
2O
3 and FeO (
Table 2), although these last two majors also show high concentrations in some bentonitic and sandy samples respectively, playing an important role in their color due to hematite pigment formation [
31,
32]. Bentonitic samples show higher contents in MgO and F over all the other groups, matching the common references of F being related to authigenic processes [
24,
25]. Regarding the differences of major elements between pink and green clays, we can observe significant higher percentages of MgO and F in pink clays over green clays and of FeO, Fe
2O
3, Al
2O
3, K
2O, TiO
2 and P
2O
5 in green clays over pink clays (
Table 2), allowing the discrimination between the two types of bentonites.
HFSE show higher concentrations in both sandy and illitic samples (
Table 3), indicating a correlation with the detrital minerals. Sandy samples in general have higher concentrations of Zr, Hf and Th, while illitic samples have higher concentrations of Nb, Ta and U. The concentration of these elements in these samples is explained by their relative immobility and their association with heavy and more resistant minerals [
33,
34], in good agreement with the detrital origin proposed. Considering only the bentonitic samples, we find differences in the concentrations of Nb, Tl and Th between green and pink clays, being higher in the former than in the latter, indicating a more detrital character. Therefore, pink clays show a more authigenic character.
Th and U are commonly fractioned during external processes [
35], being the Th/U ratios high in weathered profiles due to the relative high solubility of U in oxidizing conditions in comparison with relative immobility of Th. Under reducing conditions, U is insoluble and therefore the Th/U ratio shows lower values. We observe remarkable differences between the different types of samples (
Figure 3), this ratio being higher than 1 in sandy and illitic samples, although this last group presents two samples with a value slightly lower than 1. The illitic association is a mixture of detrital quartz, illite, feldspar and neoformed smectite, therefore explaining its intermediate character between sandy and bentonitic samples. Bentonitic and carbonatic samples both present values below 1, except two bentonitic samples. Focusing on the differences between pink and green clays, we can observe a clear discrimination, having green clays values comprised between 0.6 and 1.15, while the values of this ratio for pink clays range between 0.2 and 0.55. As previously stated, lower values of the Th/U ratio are linked to reducing conditions and higher values to oxidizing conditions, therefore the interpretation of this ratio for bentonitic samples matches previous studies which indicate that pink clays were formed in reductive palustrine environments and green clays in oxidizing lacustrine environments [
22].
From the TTE, Ni cannot be taken into consideration, because all samples present values at or under the limit of detection. Samples from the illitic association show higher concentrations in all of these elements (
Table 2), suggesting a correlation with elements belonging to this group, with the exception of V, which shows a wide variability within all the groups. We can observe within bentonitic samples that green clays present higher concentrations of Sc and Zn than pink clays.
LILE show a wider distribution than all the previous elements mentioned before (
Table 3). Pb shows a high variability within each of the mineralogical associations, thus not being directly associated with any of them. Carbonatic samples present the highest amount of Sr, which can go up to 1622 µg/g as in sample ESB13. Illitic samples have higher concentrations of Cs and sandy samples present higher concentrations of Ba, while Rb is equally concentrated in these two groups of samples. These elements show remarkable differences in the concentrations of bentonitic samples, except Sr and Pb, being more concentrated in green than pink clays.
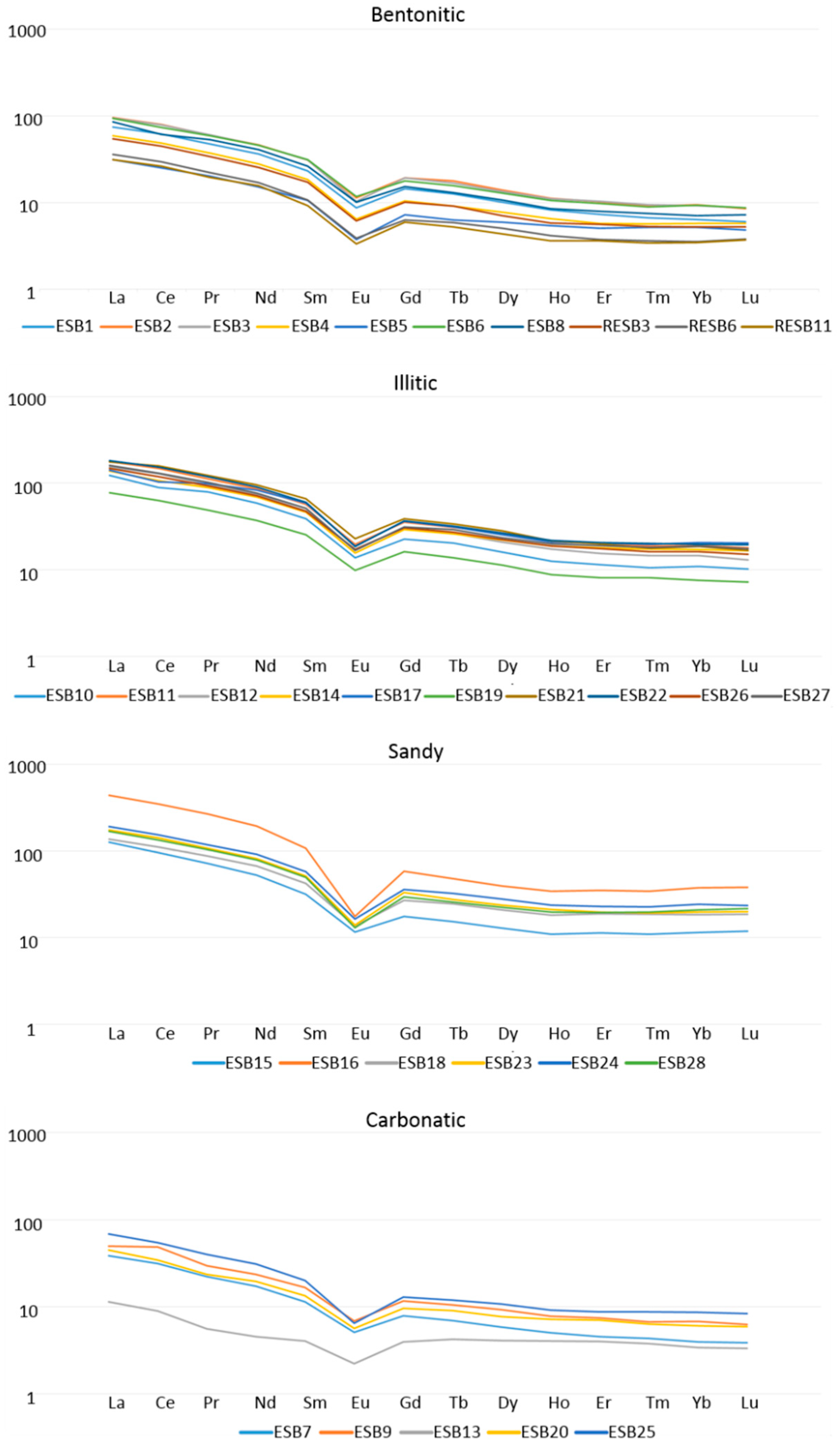
All the REE present higher concentrations at sandy and illitic samples, due to their low solubility and their preferential accumulation in the terrigenous fraction of the sediments [
36]. As expected, green clays present higher concentrations of REE than pink clays. After chondrite normalization [
37] (
Figure 4), we can observe that all the samples show a relative enrichment in light REE (LREE) and a relative depletion in heavy REE (HREE), regardless of their mineral association. These relative enrichments and depletions are probably originated by a fractioning of the REE. We can also observe that there is always a negative Eu anomaly and that, in some bentonitic and illitic samples, there is a negative Ce anomaly, indicating a granitic rock source. Some carbonatic samples present positive Ce anomaly, linked to alkaline, carbonate-rich, aerobic lake waters [
38].
The other trace elements show different associations, in one part because many of them show values below their detection limit (Mo, In, Ag and Bi) and in another due to the lack of correlatable characteristics. Be, Ga, Sn, W and Bi are present in higher concentrations in illitic samples (
Table 3). Ge presents the highest concentrations in illitic and sandy samples. Y and Ag show their highest values in sandy samples, although illitic samples also show similar values. Mo shows a wide variation in its concentration, but it is more abundant at illitic and carbonatic samples. Sb also shows a wide variation, but in this case its content does not show remarkable contents in any of the mineral associations established. The lowest concentrations of As are found in the bentonitic samples, more precisely in pink clays, while in the remaining samples it appeared with similar concentrations values. Finally, In shows similar values within all the mineralogical associations, due to the fact that half the samples present contents in this element below de detection limits. The concentrations of these trace elements show notable differences between green and pink clays, except in the case of Be, Mo and Sb where some values overlap, and in the case of Ag, which does not allow any kind of differentiation of these two types of bentonitic samples, because they are all at or below the detection limit, except sample ESB8.
The results of the geochemical analyses match the predictable tendency for both major and trace elements. We are able to distinguish elements associated with a detrital origin more concentrated in illitic and sandy samples as well as elements of neoformation linked to bentonitic and carbonatic samples. We can also observe these differences within the bentonitic samples, being able to determine a higher detrital character of green clays with respect to pink clays. Bearing this in mind, we proceeded to perform a statistical treatment of the data.
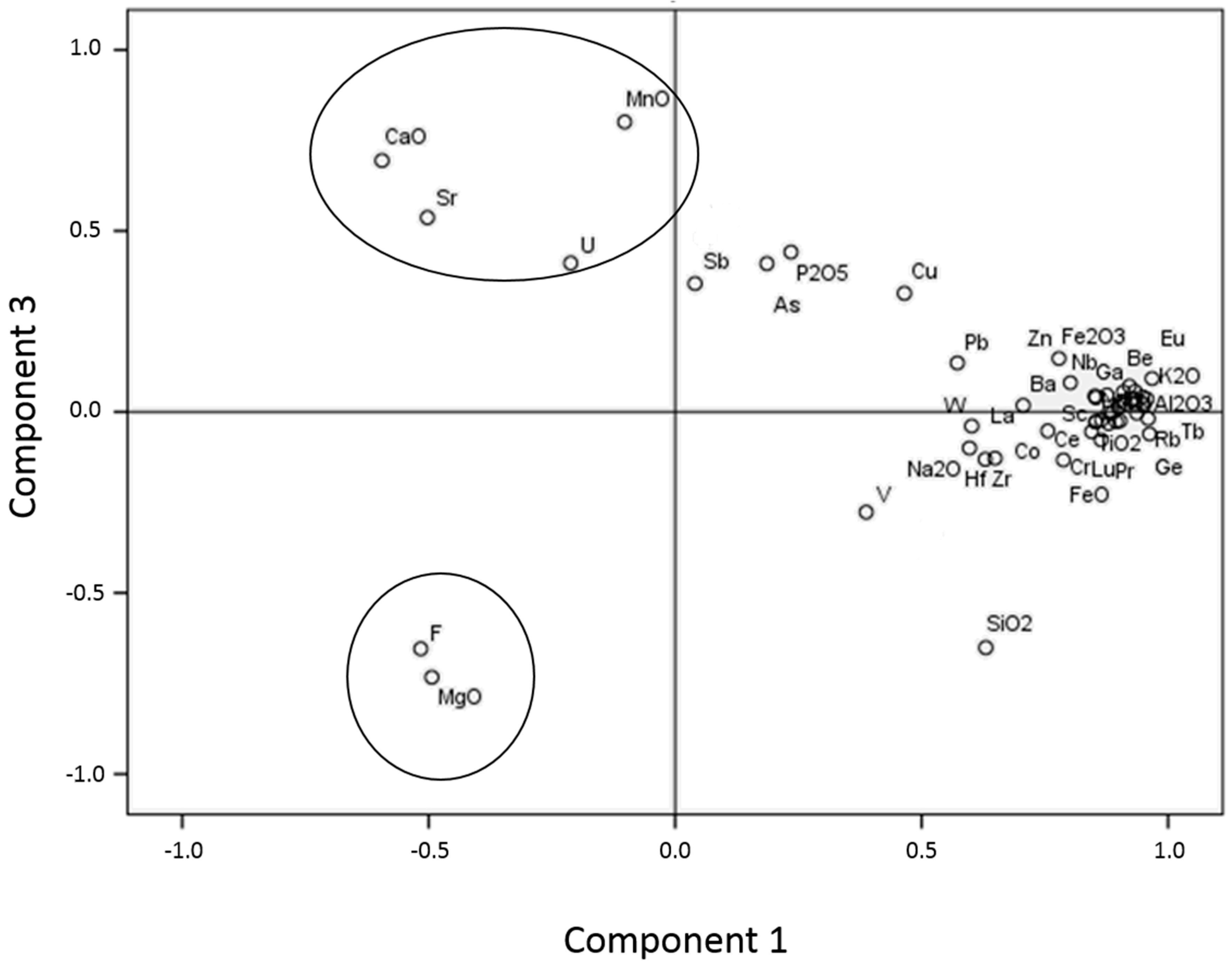
A principal component analysis (PCA) was performed (
Figure 5) considering in the X axis the principal component 1 and in the Y axis the principal component 3. This statistical procedure helps analyze large size data sets with a large number of interrelated variables, reducing its dimensions while retaining as much as possible of the variation present in the data set [
39]. It is important to remark that several elements which are below the detection limit within several samples (Mo, In, Ag and Bi) have not been considered for this type of analysis. The components used in the PCA were chosen after carefully examining a bivariate correlation matrix, which determines the empirical relationship between the different variables, and considering the comparison between them the most reliable. The resulting groups obtained through this method were easily assigned to the mineral associations established when considering the bivariate correlation matrix previously mentioned. F and MgO show significant correlation coefficients with each other as well as with smectite, being the only elements analyzed, along with V and W, which show this tendency (
Table 4). CaO, MnO and Sr show significant correlation coefficients with each other as well as with calcite, in contrast to the rest of the elements analyzed (
Table 5). U, which falls within this group, only shows a significant correlation with MnO. Considering the PCA as well as the bivariate correlation matrixes, we can state that only elements which show significant correlation coefficients with each other and with a certain mineral should be considered linked to that precise mineral. Therefore, MgO and F are attributed to the bentonitic samples and CaO, MnO and Sr to carbonatic samples. The rest of the elements, which fall within a range which is hardly differentiable at some points, are attributed to the illitic and sandy samples. While the groups linked to bentonitic and carbonatic samples are interpreted as being linked to neoformation processes, the other group is considered to have a detrital origin. This matches the associations of HFSE, Y and Ti being related to heavy and immobile minerals, such as monazite and zircon, present as accessory minerals in small quantities within sandy samples [
33,
34,
40,
41,
42], and Rb and Ba to potassium feldspar [
43].
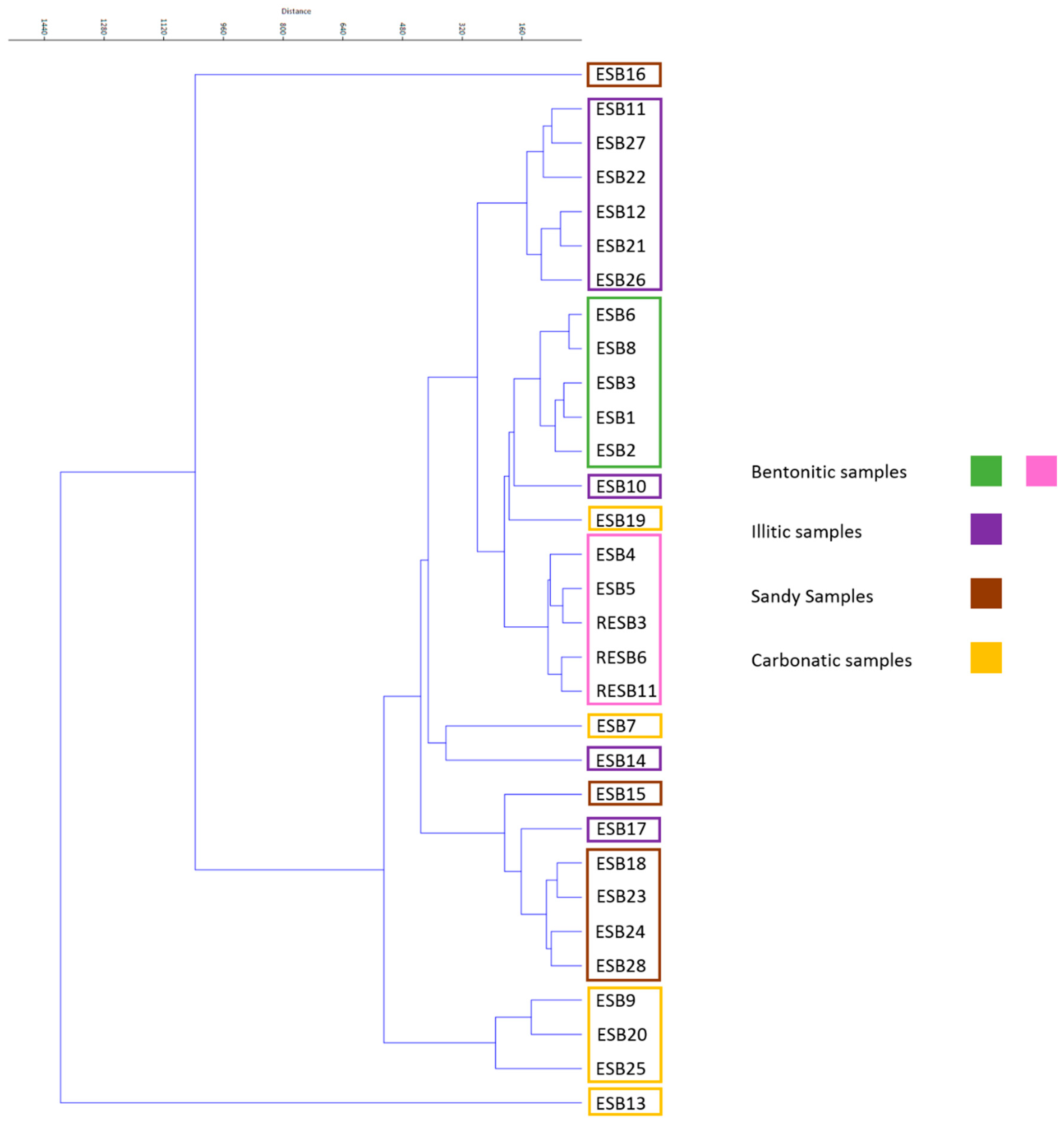
Multivariate clustering was performed with all the geochemical data (
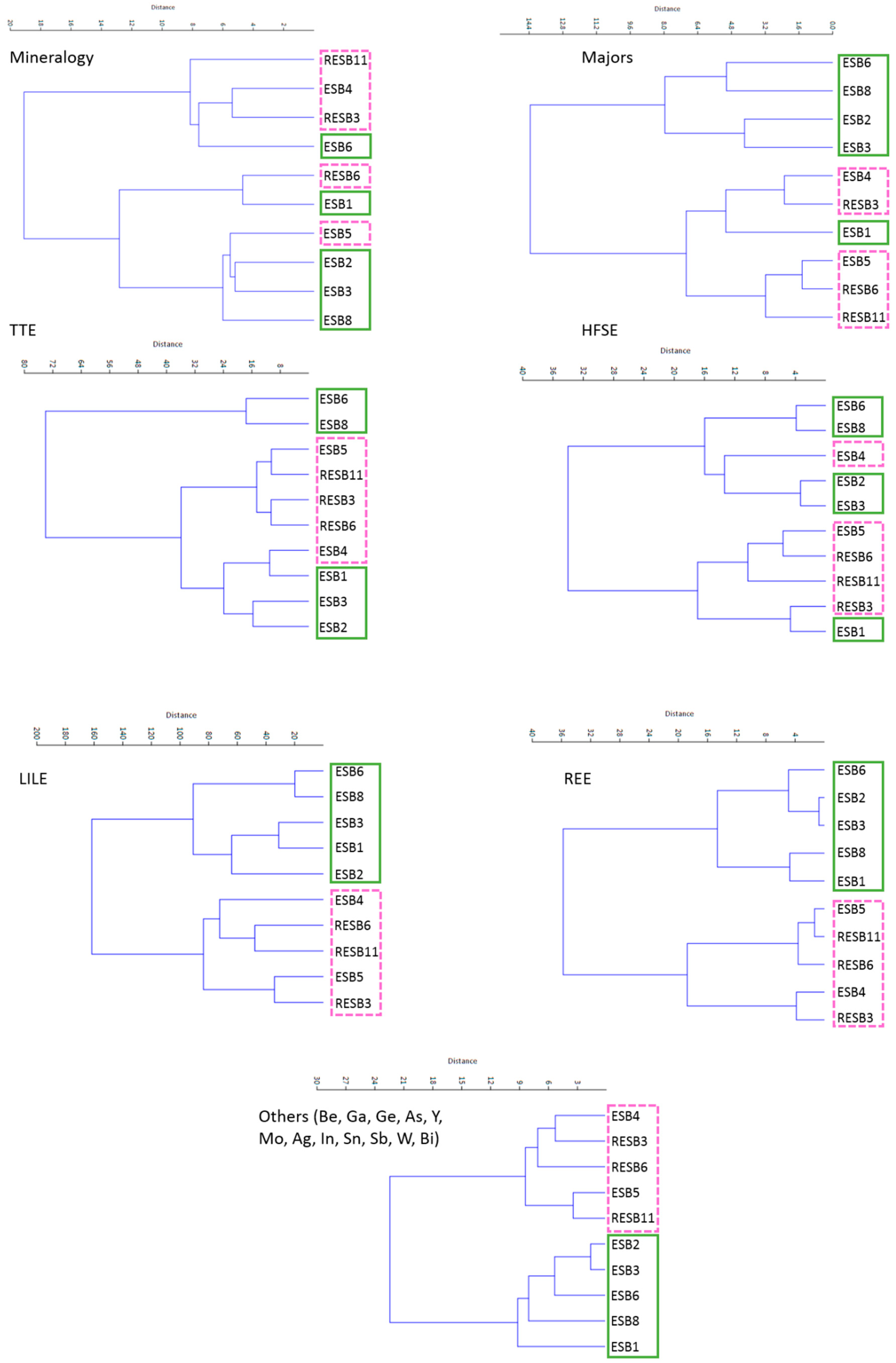
Figure 6), obtaining pretty good grouping of the different mineral associations established according to the XRD data. There are some samples that do not group, which is probably due to its composition being close to the mineralogical limits marking the different groups. As the main purpose of this paper is to obtain discrimination criteria between the two types of bentonitic samples (green and pink clays), we also performed multivariate clusters. Green and pink clays (
Figure 7) cannot be separated when we consider the mineralogy, major elements, HFSE and TTE, but when the clustering is performed considering LILE, REE and the other trace elements analyzed, we observe a good grouping of these samples. It is important to remark that, when performing the cluster considering the other trace elements we have not considered the elements that have a high percentage of samples below the detection limit (Ag, In, Mo and Bi).
All these mineralogical associations established correspond either to detrital/inherited levels (sandy and illitic) or to probable authigenic levels (bentonitic and carbonatic), corresponding to different sedimentary environments, such as periods of a higher energy (detrital input) or low energy. The bentonitic association includes both green and pink clays.
3.3. Biogeochemistry
We analyzed
n-alkanes (
Table 6) and
n-alakanoic acids (
Table 7) in all the samples from the sampled section through gas chromatography–mass spectrometry (GC–MS). The biomarkers indicated low mature organic matter as expected, i.e., clays studied here were not buried substantially and diagenetic processes were not significant. Therefore, the information provided by the biomarkers here is centered in the origin and the degree of degradation of the organic matter. In the case of
n-alkanes, the chain lengths analyzed went from 15 to 36 carbon atoms while the
n-alkanoic acids had chain lengths from 12 to 33 carbon atoms.
Several indexes were employed in the study of the
n-alkanes, such as the carbon preference index (CPI) [
44], predominant chain length (PCL), average chain length (ACL) [
45],
Paq [
46] and the terrigenous/aquatic ratio for hydrocarbons (TAR
HC) [
47]. The proxies employed for
n-alkanoic acids were the PCL and the terrigenous/aquatic acid ratio for fatty acids (TAR
FA) [
48,
49].
Regarding the
n-alkanes, we observe an odd-over-even predominance of carbon number, indicating immature samples. This observation coincides with the interpretation of the carbon preference index (CPI; [
44]), calculated as ½[(ΣC
i + C
i+2 + ... + C
i+8)/(ΣC
i−1 + C
i+1 + ... + C
i+7) + (ΣC
i + C
i+2 + ... + C
i+8)/(ΣC
i+1 + C
i+3 + ... + C
i+9)], with i = 25, which is commonly used to discriminate between mature and immature organic matter in sediments, because it indicates the predominance of odd-over even numbered
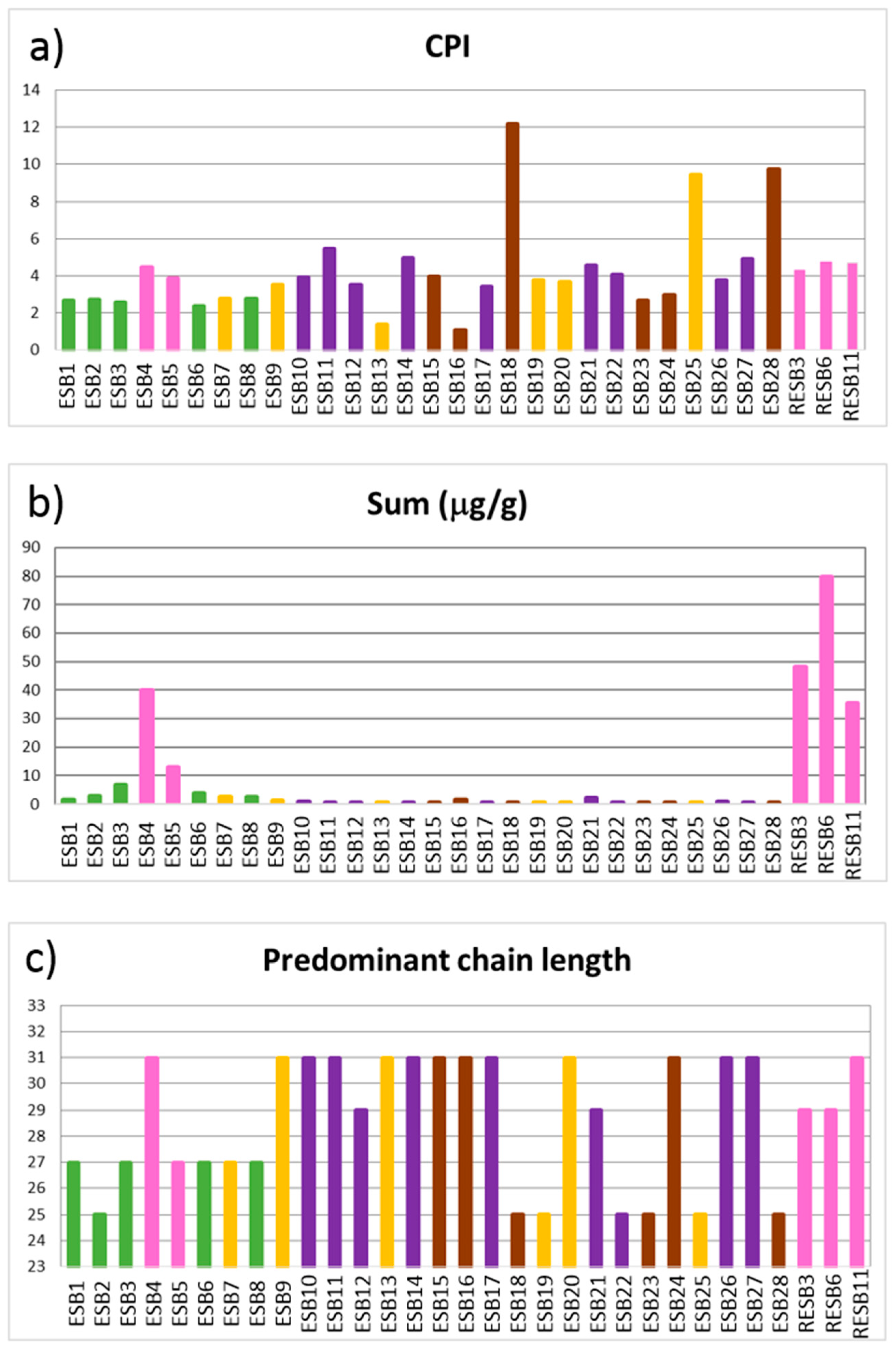
n-alkanes of a certain chain length. The CPI ratio shows values superior to 2, except in samples ESB13 and ESB16 (
Figure 8a), that present values close to 1. Samples with CPI values > 2 are interpreted as not having suffered an important maturity of the organic matter, while when the CPI value is close to 1, microbial degradation or diagenetic processes are responsible for this [
50]. This interpretation matches with the macroscopial aspect of sample ESB13, which is a non-lithified vein clearly formed due to post-sedimentary processes, being composed by calcite in its totality.
The predominant
n-alkane chain varies between C
25, C
27, C
29 or C
31. (
Figure 8c) indicating a major input from aquatic macrophytes and terrestrial plants as the C
25 homologue is predominant in the former [
46], and C
27, C
29 or C
31 are abundant in the latter [
45,
51,
52,
53,
54]. This interpretation coincides with that of the average chain length (ACL; [
55], calculated as [(C
i × i + C
i+1 × (i + 1) + C
i+2 × (i + 2) ... + C
n × n))/(ΣC
n+1 + C
n+2 + ... + C
n), with i = 13, n = 33], which is a good proxy for distinguishing between the predominance of low vs. high molecular weight
n-alkanes [
54,
56]. The ACL oscillates between 25 and 29 (
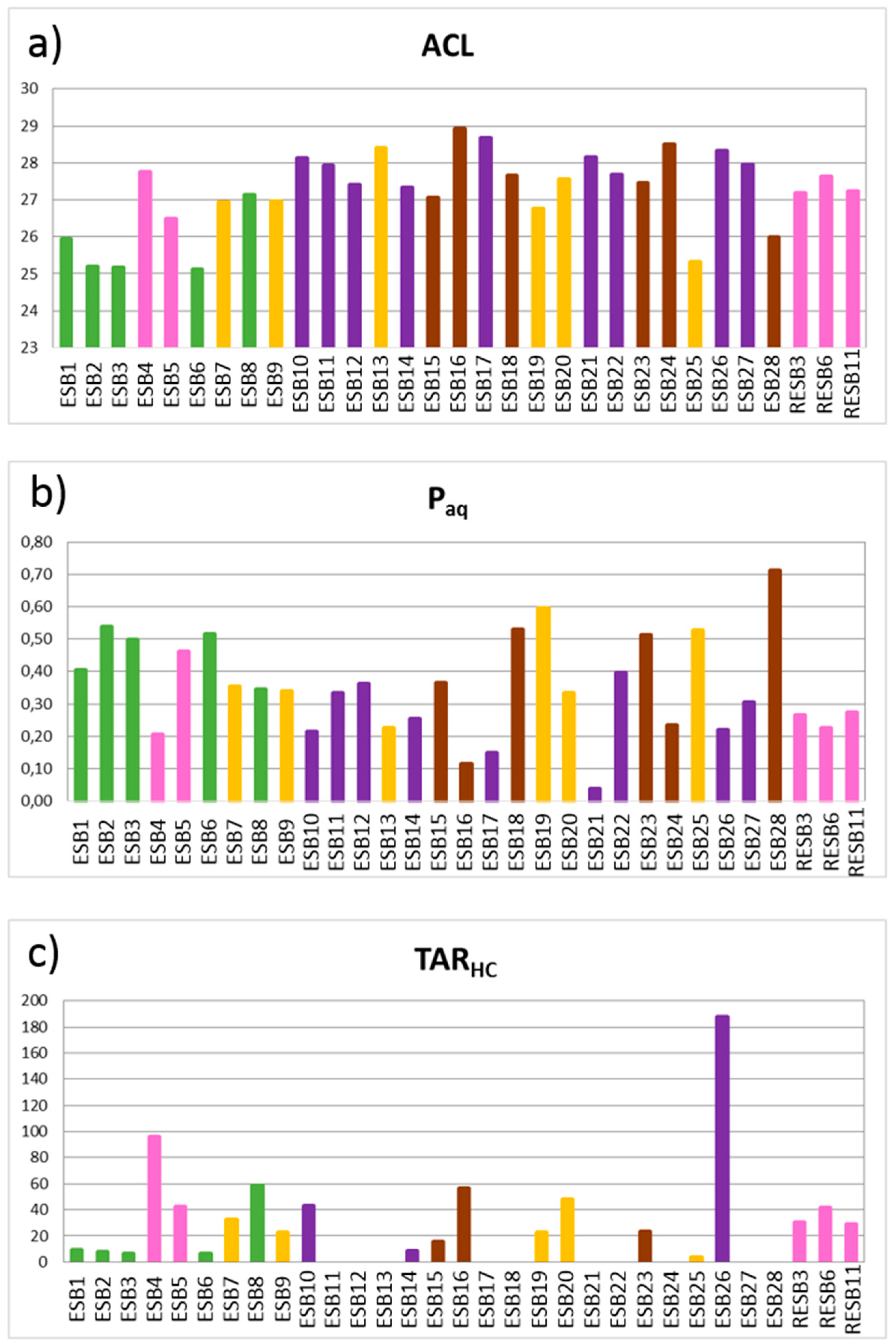
Figure 9a), linked to a major input from aquatic macrophytes and terrestrial plants. However, there is no evidence of a relationship with the mineralogy because these values differ in samples from within the same association.
In this regard, some bentonitic (ESB1, ESB2, ESB3 and ESB6), carbonatic (ESB25) and sandy samples (ESB28) have low ACL values, probably indicating a similar source of the organic matter. According to bibliographical references [
54], these samples are linked to important aquatic macrophytes input. Therefore, samples having an aquatic origin belong to different groups of samples, according to these data. The remaining samples show higher ACL values, indicating a terrestrial origin, although with certain influence of aquatic macrophytes.
Focusing on the abundance of
n-alkanes, the highest values are linked to the betonitic samples, more obviously in the pink clays (
Figure 8b). The correlation matrix between mineralogy and biomarker concentrations shows that the only mineral which presents a positive significant correlation is smectite (
Table 8), justifying the higher concentrations at the bentonitic samples. This higher concentration within bentonitic samples, especially within pink clays, can be justified by the high specific surface area of these materials, being higher in pink clays [
19] than in green clays [
57].
The
Paq index, calculated as the (C
23 + C
25)/(C
23 + C
25 + C
29 + C
31) ratio [
46], was postulated to reflect the relative contribution of emergent and submerged/floating aquatic macrophytes, which typically maximize at C
23 and C
25. In general, the
Paq index (
Figure 9b) shows values comprised between 0.1 and 0.6, indicating a mixed input of aquatic macrophytes and terrestrial plants [
46], coinciding with the interpretation of the
n-alkane predominant chain length and ACL values. However, an illitic (ESB21) and a sandy sample (ESB28) fall out of this range.
The terrigenous/aquatic ratio (TAR
HC) index (
Figure 9c), calculated as (C
31 + C
29+ C
27)/(C
15 + C
17 + C
19) [
47], was studied in order of distinguishing between land plants and algal input, its being higher values associated with a higher input of land plants and lower values with a higher algal input. It must be highlighted that it was not possible to obtain this proxy for several samples, due to lack of low molecular weight
n-alkanes. There are two families of samples, characterized by values over and below 20, regardless of their mineralogy. Samples with lower values are bentonitic (ESB1, ESB2, ESB3 and ESB6), illitic (ESB14), sandy (ESB15) and carbonatic (ESB25), and are interpreted according to [
47] as having a mixed origin of organic matter, while samples with a higher value are interpreted as having a more important input of terrestrial organic matter.
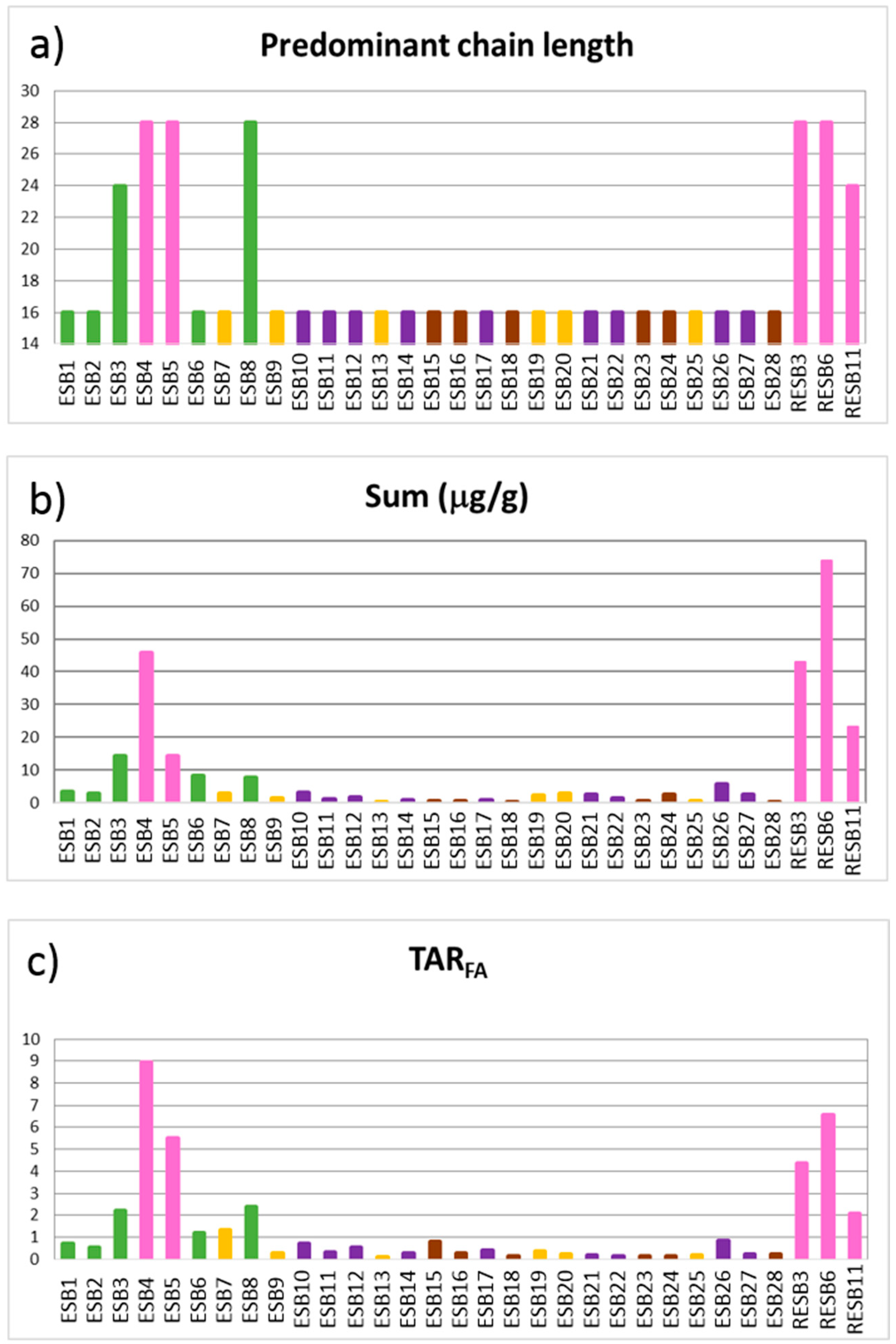
The
n-alkanoic acid predominant chain length show a unimodal distribution maximizing at C
16 (
Figure 10a), although several samples show a bimodal distribution maximizing at C
24 and C
28. The samples with a bimodal distribution are bentonitic, both pink and green clays. It is important to remark that not all of the bentonitic samples show the same distribution. The lack of correspondence between the PCL and ACL of
n-alkanes and
n-alkanoic acids (
Figure 9 and
Figure 10) suggests a certain preferential microbial synthesis of long-chain saturated fatty acids from primary organic matter (OM) and/or bacterial activity producing an increase in short chain homologues (mainly C
16 and C
18), i.e.,
n-alkanoic acids in lake sediments typically originate from multiple sources (algae, aquatic macrophytes, land plants), but they are more sensitive to degradation and modification than other types of lipid biomarker [
58]. Thus, the
n-alkanoic acid content reveals not only the OM source but can also be used to evaluate the degree of preservation, especially when the
n-alkanes distribution does not coincide with the
n-alkanoic acid one.
We calculated the terrigenous/aquatic ratio for fatty acids (TAR
FA) as (C
24 + C
26+ C
28)/(C
12 + C
14 + C
16) [
47] to distinguish between algal (low TAR
FA values) vs. land plant sources (high TAR
FA values). However, selective degradation and diagenetic processes commonly overprint
n-alkanoic acid distributions. Short-chain acids are often preferentially degraded by microbes during early diagenesis [
59,
60] and they produce higher TAR
FA values [
49]. On the other hand, the microbial synthesis of secondary fatty acids from primary OM produces short-chain components [
60] and can depress TAR
FA values. The low TAR
FA values (
Figure 10c), along with the unimodal distribution of
n-alkanoic acids, maximizing mainly at C
16, and the most abundant alkanes being C
25 to C
31, point to the occurrence of microbial degradation of high molecular weight
n-alkanoic acids from primary organic matter, a process that produces short-chain homologues [
61], with the exception of samples ESB3, ESB4, ESB5, ESB8, RESB3, RESB6 and RESB11, as will be further discussed.
The TAR
FA index (
Figure 10c) shows two different groups of samples. On one hand, we have samples with values lower than 1 and, on the other, samples whose values are higher than 1. The latter group is formed by eight bentonitic samples (ESB3, ESB4, ESB5, ESB6, ESB8, RESB3, RESB6 and RESB11) and one carbonatic sample (ESB7). The values of the TAR
FA index indicate that the organic matter present in these samples did not suffer important degradation processes as in the other samples [
48,
49].
As in the case of the
n-alkanes, it is possible to observe a higher concentration of
n-alkanoic acids in the bentonitic samples, and especially in the pink clays (
Figure 10b). The correlation values of the mineralogy and biomarkers also show the same tendency (
Table 9), smectite being the only mineral significantly correlated to the alkanoic acids.
Summarizing the interpretations of the biomarkers analyzed, we can observe several clashes between them. Different proxies employed for the study of n-alkanes reveal that pink clays have a more important input of organic matter of terrestrial origin, while green clays show a higher input of aquatic macrophytes (PCL, ACL and Paq). The Paq index values indicate that a pink clay (ESB5) has more aquatic macrophytes input than the rest of the pink clays, clashing with all the other proxies studied.
The n-alkanoic acid PCL indicates that three green clays (ESB1, ESB2 and ESB6) have suffered important organic matter degradation, while the TARFA values show that only two green clays (ESB1, ESB2) have undergone important organic matter degradation. The rest of the bentonitic samples are interpreted, according to both proxies, as not having suffered important organic matter degradation.
The PCL of the n-alkanes shows all pink clays and one green clay (ESB2) as having a major input of organic matter of terrestrial origin, which is corroborated by the n-alkanoic acid PCL values, these latter also indicate that all the pink clays and one green clay (ESB8) did not suffer important organic matter degradation. This is confirmed with the low TARHC values in all pink clays and one green clay (ESB8), together with TARFA values, showing that all pink clays and three green clays (ESB3, ESB6, ESB8) have a major input from land plants and suffered less important organic matter degradation). These differences are probably caused by selective degradation and diagenetic processes, which commonly overprint n-alkanoic acid distributions.
A multivariate cluster analysis of the biomarkers data was performed (
n-alkanes +
n-alkanoic acids) (
Figure 11a), not grouping well in respect to the mineralogical associations established. This indicates that the biomarker content is not related to the mineralogical characteristics of the samples, in an opposite way to the geochemical data. Performing a multivariate analysis including biomarkers and geochemical data (
Figure 11b), it is possible to observe the same cluster than when considering only the geochemical data (
Figure 6), reinforcing the idea that biomarkers do not help in the discrimination between the different mineralogical associations established.
Thus, it seems evident that biomarkers, in contrast to the geochemical analyses, are not related to the mineralogy. Only the total concentration of both
n-alkanes and
n-alkanoic acids seems to be directly correlated to smectite, and therefore to the bentonitic samples. The interpretation of biomarkers also discards the idea that samples having similar mineral associations belong to similar sedimentary environments, as can be seen from different proxies obtained from the
n-alkanes analysis. Biomarkers also give contradictory sedimentological interpretations with those from bibliographical references of the Tajo Basin, such as pink clays being formed in a reducing palustrine environment and green clays in an oxidizing lacustrine one [
22], which agree with the geochemical analysis presented in this paper; therefore, having different ratios of organic matter degradation, clashing with the interpretation of the TAR
FA proxy of the
n-alkanoic acids. It must be highlighted that organic matter incorporates during the sedimentation processes many sources (algal, aquatic macrophytes, land plants) and also has variable degree of preservation. During diagenesis original organic matter suffers alteration when sinking to the lake bottom, but biomarkers retain key information about their origin [
58]. Thus, biomarkers reveal not only the organic matter source but can also be used to evaluate the degree of preservation and diagenesis.