Carmeltazite, ZrAl2Ti4O11, a New Mineral Trapped in Corundum from Volcanic Rocks of Mt Carmel, Northern Israel
Abstract
:1. Introduction
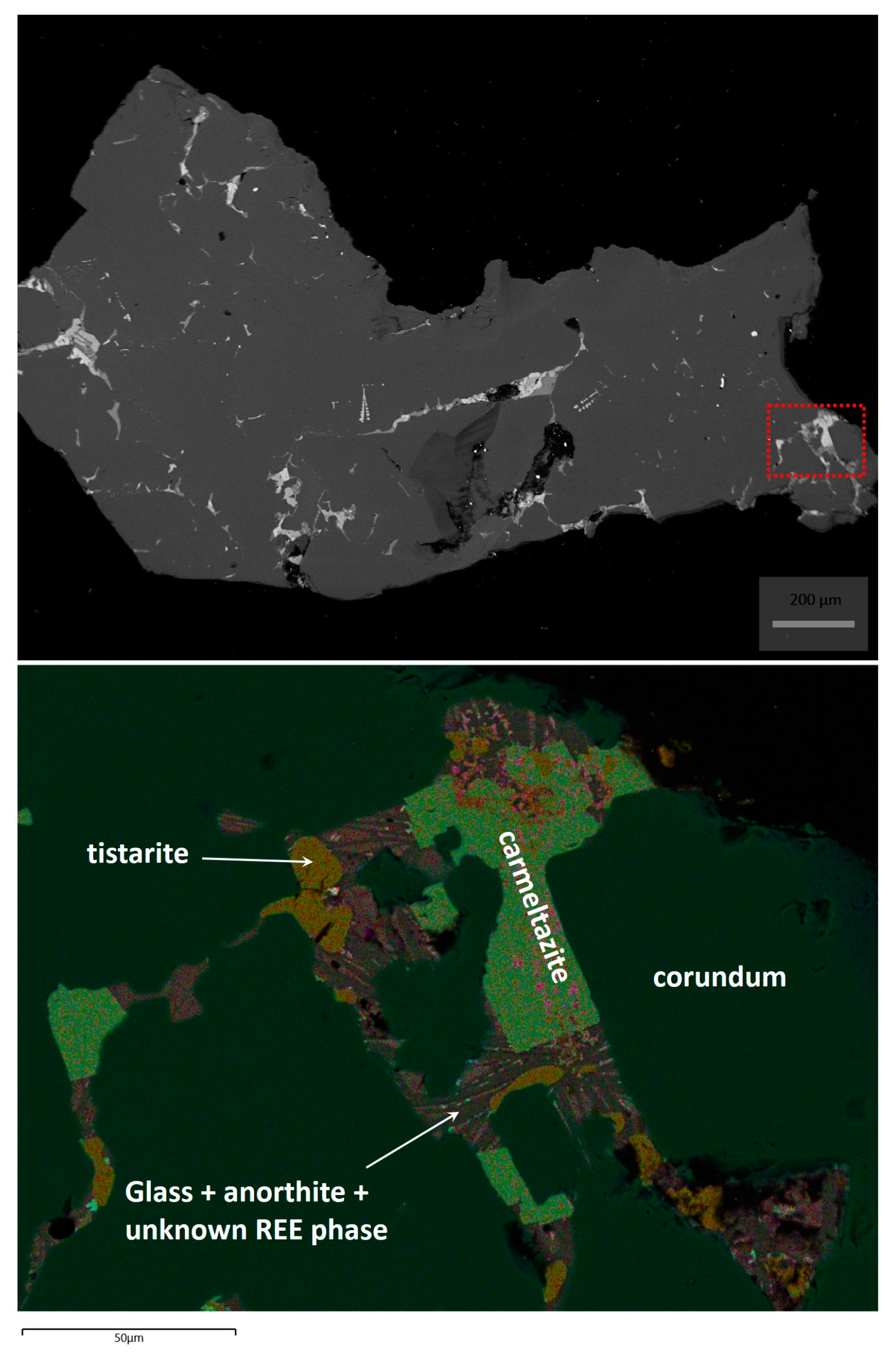
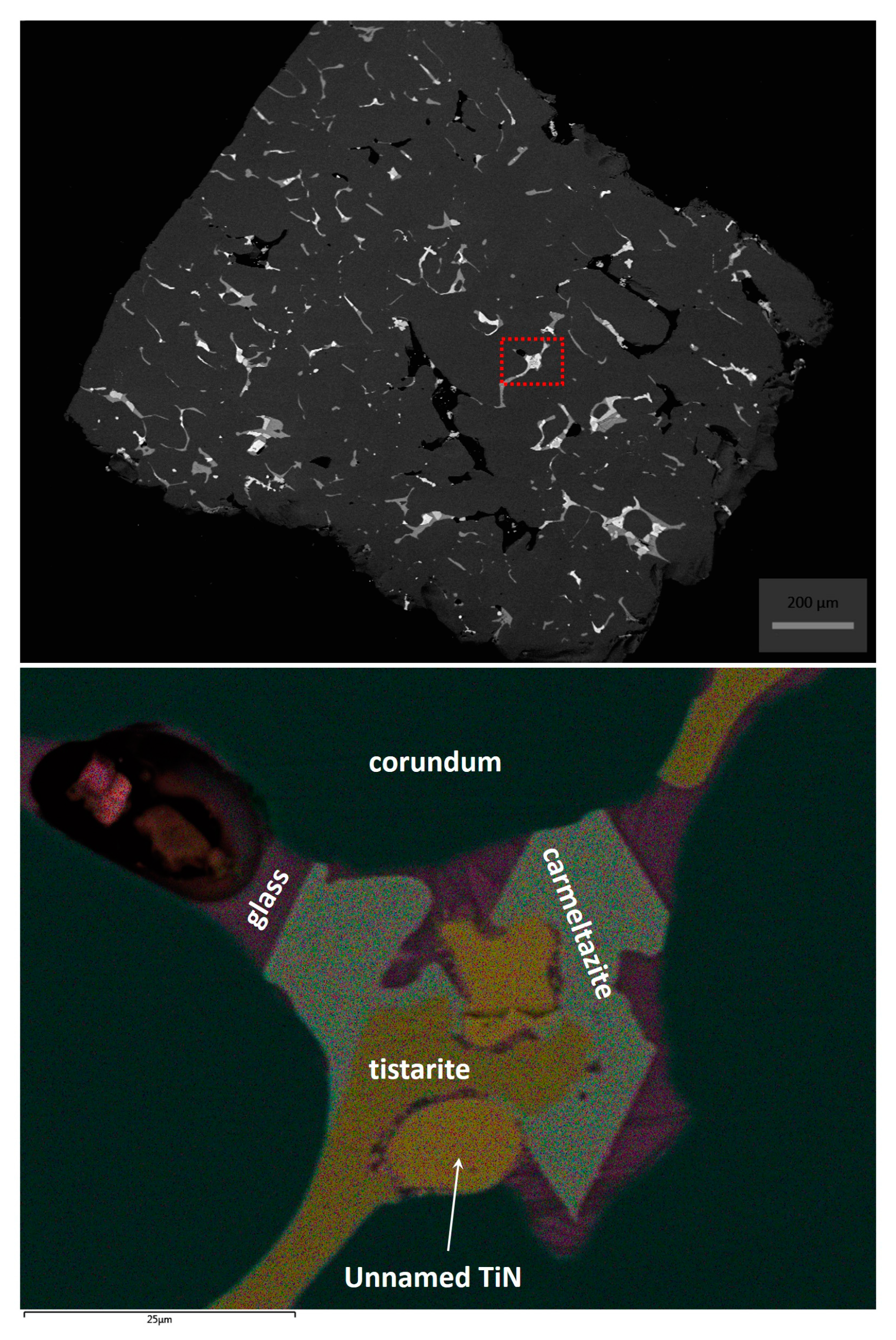
2. Occurrence of Carmeltazite
3. Mineral Description and Physical Properties
4. Chemical Data
5. X-ray Crystallography
6. Results and Discussion
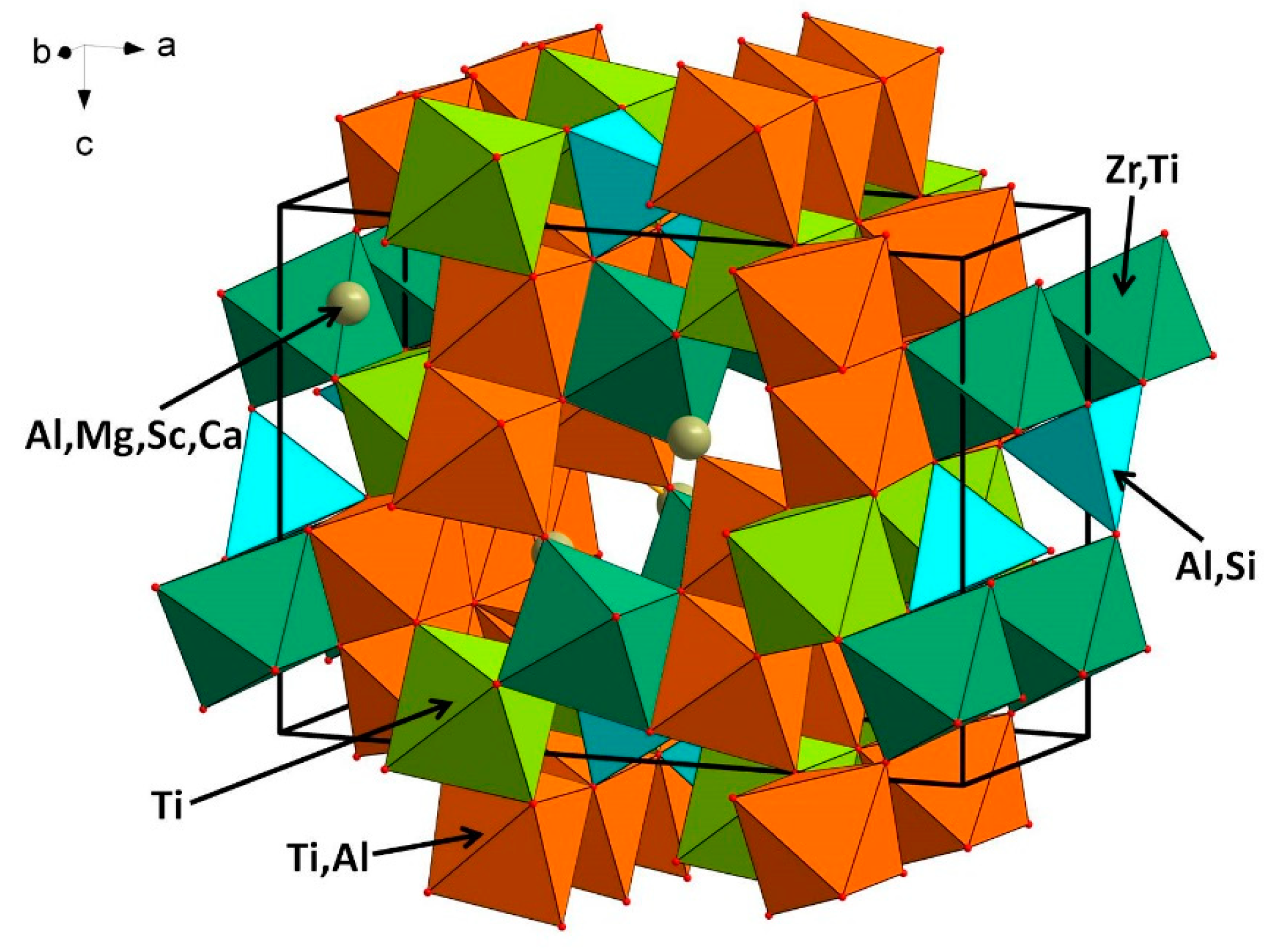
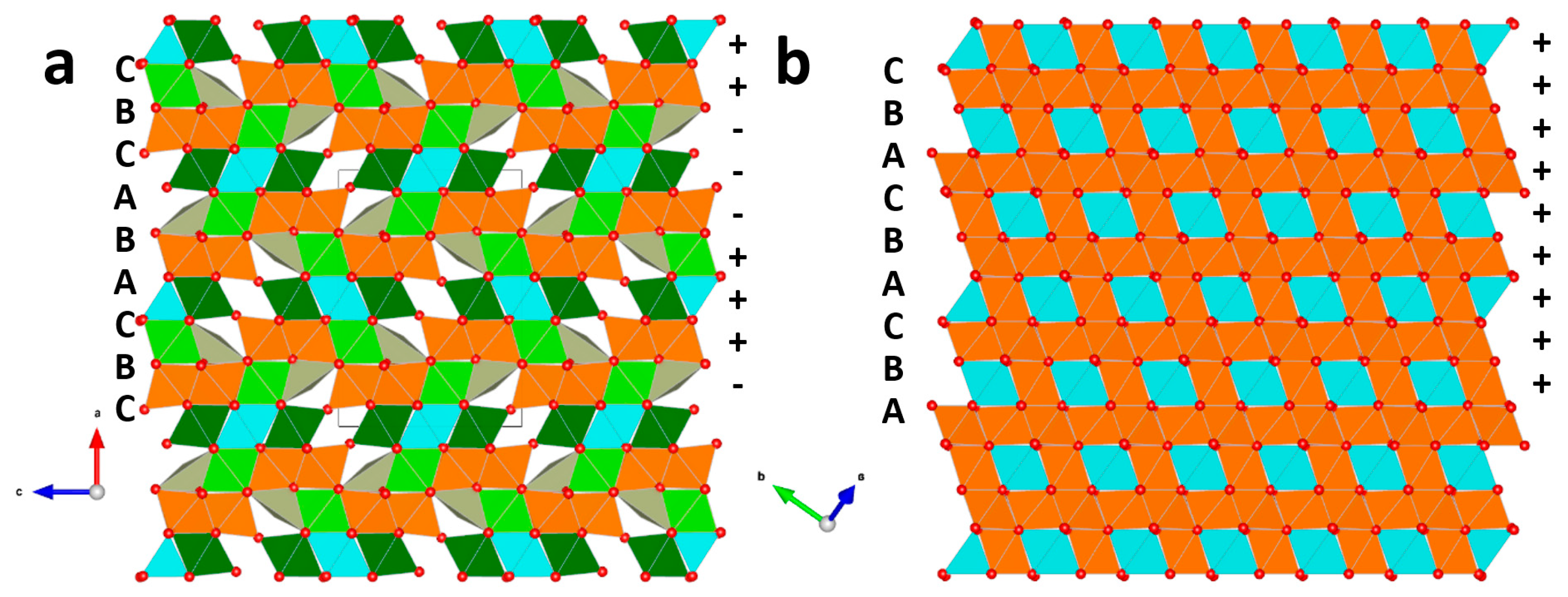
6.1. Description of the Crystal Structure
6.2. Origin of Carmeltazite
7. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Griffin, W.L.; Gain, S.E.M.; Huang, J.-X.; Saunders, M.; Shaw, J.; Toledo, V.; O’Reilly, S.Y. A terrestrial magmatic hibonite-grossite-vanadium assemblage: Desilication and extreme reduction in a volcanic plumbing system, Mt Carmel, Israel. Am. Miner. 2019. [Google Scholar] [CrossRef]
- Griffin, W.L.; Gain, S.E.M.; Adams, D.T.; Huang, J.-X.; Saunders, M.; Toledo, V.; Pearson, N.J.; O’Reilly, S.Y. First terrestrial occurrence of tistarite (Ti2O3): Ultra-low oxygen fugacity in the upper mantle beneath Mount Carmel, Israel. Geology 2016, 44, 815–818. [Google Scholar] [CrossRef]
- Xiong, Q.; Griffin, W.L.; Huang, J.-X.; Gain, S.E.M.; Toledo, V.; Pearson, N.J.; O’Reilly, S.Y. Super-reduced mineral assemblages in “ophiolitic” chromitites and peridotites: The view from Mount Carmel. Eur. J. Mineral. 2017, 29, 557–570. [Google Scholar] [CrossRef]
- Oxford Diffraction. CrysAlis RED (Version 1.171.31.2) and ABSPACK in CrysAlis RED; Oxford Diffraction Ltd.: Abingdon, Oxfordshire, UK, 2006. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.J.C. International Tables for Crystallography. Volume C: Mathematical, Physical and Chemical Tables; Kluwer Academic: Dordrecht, The Netherland, 1992. [Google Scholar]
- Brese, N.E.; O’Keeffe, M. Bond-valence parameters for solids. Acta Cryst. 1991, B47, 192–197. [Google Scholar] [CrossRef]
- Tillmanns, E.; Hofmeister, W.; Baur, W.H. Variations on the theme of closest packing: The structural chemistry of barium titanate compounds. J. Solid State Chem. 1985, 58, 14–28. [Google Scholar] [CrossRef]
- Tillmanns, E.; Wendt, I. Die Kristallstruktur eines Bariumtitanlithiumoxids, Ba2Ti9,25Li3O22. Zeit. Kristallogr. 1976, 144, 16–31. [Google Scholar] [CrossRef]
- Imaz, I.; Péchev, S.; Koseva, I.; Bourée, F.; Gravereau, P.; Peshev, P.; Chaminade, J.-P. Structural filiations in the new complex titanates SrLiMTi4O11 (M = Cr, Fe). Acta Cryst. 2007, B63, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Griffin, W.L.; Huang, J.-X.; Thomassot, E.; Gain, S.E.M.; Toledo, V.; O’Reilly, S.Y. Super-reducing conditions in ancient and modern volcanic systems: Sources and behaviour of carbon-rich fluids in the lithospheric mantle. Min. Petr. 2018, 112, 1–14. [Google Scholar] [CrossRef]
- Bindi, L.; Cámara, F.; Griffin, W.L.; Huang, J.-X.; Gain, S.E.M.; Toledo, V.; O’Reilly, S. Solar nebula conditions on Earth produced the first natural hydride. Sci. Rep. 2019. submitted. [Google Scholar]
- Ma, C.; Tschauner, O.; Beckett, J.R.; Rossman, G.R.; Liu, W. Panguite, (Ti4+,Sc,Al,Mg,Zr,Ca)1.8O3, a new ultra-refractory titania mineral from the Allende meteorite: Synchrotron micro-diffraction and EBSD. Am. Mineral. 2012, 97, 1219–1225. [Google Scholar] [CrossRef]
- Ma, C.; Tschauner, O.; Beckett, J.R.; Rossman, G.R.; Liu, W. Kangite, (Sc,Ti,Al,Zr,Mg,Ca,□)2O3, a new ultra-refractory scandian mineral from the Allende meteorite: Synchrotron micro-Laue diffraction and electron backscatter diffraction. Am. Mineral. 2013, 98, 870–878. [Google Scholar] [CrossRef]
- Ma, C.; Beckett, J.R.; Rossman, G.R. Allendeite (Sc4Zr3O12) and hexamolybdenum (Mo,Ru,Fe), two new minerals from an ultrarefractory inclusion from the Allende meteorite. Am. Mineral. 2015, 99, 654–666. [Google Scholar] [CrossRef]

| Oxide | wt (%) (n = 8) | Range | Standard Deviation |
|---|---|---|---|
| SiO2 | 1.50 | 1.24–1.70 | 0.24 |
| ZrO2 | 24.9 | 23.7–27.9 | 1.45 |
| HfO2 | 0.53 | 0.48–0.67 | 0.07 |
| UO2 | 0.16 | 0.00–0.40 | 0.15 |
| ThO2 | 0.06 | 0.00–0.13 | 0.05 |
| Al2O3 | 18.8 | 18.0–20.1 | 0.78 |
| Cr2O3 | 0.02 | 0.00–0.08 | 0.03 |
| Ti2O3 | 50.6 | 48.8–52.2 | 1.30 |
| Sc2O3 | 0.76 | 0.59–1.24 | 0.27 |
| Y2O3 | 0.39 | 0.30–0.51 | 0.08 |
| MgO | 1.89 | 1.50–2.93 | 0.50 |
| CaO | 0.51 | 0.29–1.45 | 0.43 |
| Total | 100.12 | 98.87–100.5 | 0.28 |
| Crystal Data | |
| Crystal size (mm3) | 0.060 × 0.075 × 0.080 |
| Cell setting, space group | Orthorhombic, Pnma |
| a, b, c (Å) | 14.0951 (9), 5.8123 (4), 10.0848 (7) |
| V (Å3) | 826.2 (1) |
| Z | 4 |
| Data Collection and Refinement | |
| Radiation, wavelength (Å) | Mo Kα, λ = 0.71073 |
| Temperature (K) | 293 |
| 2θmax (°) | 63.95 |
| Measured reflections | 12368 |
| Unique reflections | 1546 |
| Reflections with Fo > 4σ (Fo) | 1165 |
| Rint | 0.0134 |
| Rσ | 0.0428 |
| Range of h, k, l | −20 ≤ h ≤ 20, −8 ≤ k ≤ 8, −15 ≤ l ≤ 15 |
| R (Fo > 4σ (Fo)) | 0.0216 |
| R (all data) | 0.0242 |
| wR (on F2) | 0.1426 |
| GooF | 0.911 |
| Number of least-square parameters | 103 |
| Maximum and minimum residuals (e/Å3) | 0.49 (at 1.53 from M1), −0.51 (at 1.02 from O4) |
| Atom | x/a | y/b | z/c | Uiso |
|---|---|---|---|---|
| M1 | 0.14578(9) | ¼ | 0.14765(13) | 0.0400(4) |
| M2 | 0.00052(3) | ¼ | 0.77052(8) | 0.0502(3) |
| M3 | 0.33102(5) | ¼ | 0.89039(8) | 0.0374(3) |
| M4 | 0.33492(5) | ¼ | 0.42841(8) | 0.0336(3) |
| M5 | 0.17178(4) | 0.00094(10) | 0.66151(5) | 0.0356(2) |
| T | 0.0406(1) | ¼ | 0.42210(15) | 0.0435(5) |
| O1 | 0.0871(2) | ¼ | 0.5842(4) | 0.0504(8) |
| O2 | 0.2458(2) | ¼ | 0.7435(3) | 0.0478(9) |
| O3 | 0.2620(3) | ¼ | 0.2536(3) | 0.0473(9) |
| O4 | 0.4168(2) | ¼ | 0.0695(3) | 0.0412(7) |
| O5 | 0.4336(3) | ¼ | 0.5616(4) | 0.0550(9) |
| O6 | 0.2446(3) | 0.0018(3) | 0.9994(2) | 0.0485(8) |
| O7 | 0.4146(2) | 0.0071(5) | 0.3259(2) | 0.0513(7) |
| O8 | 0.4141(2) | −0.0288(5) | 0.8169(2) | 0.0420(5) |
| M1-O3 | 1.956(4) | M4-O5 | 1.933(4) |
| M1-O8 | 2.298(3) (×2) | M4-O6 | 1.978(3) (×2) |
| M1-O6 | 2.500(3) (×2) | M4-O3 | 2.041(4) |
| mean | 2.310 | M4-O7 | 2.079(3) (×2) |
| mean | 2.015 | ||
| M2-O5 | 1.938(4) | ||
| M2-O7 | 1.995(3) (×2) | M5-O3 | 1.965(2) |
| M2-O8 | 2.210(2) (×2) | M5-O2 | 1.966(2) |
| M2-O1 | 2.241(4) | M5-O6 | 2.015(3) |
| mean | 2.098 | M5-O1 | 2.031(3) |
| M5-O7 | 2.057(2) | ||
| M3-O2 | 1.908(3) | M5-O4 | 2.132(2) |
| M3-O8 | 2.132(3) (×2) | mean | 2.028 |
| M3-O4 | 2.173(3) | ||
| M3-O6 | 2.184(3) (×2) | T-O4 | 1.747(3) |
| mean | 2.119 | T-O1 | 1.761(4) |
| T-O8 | 1.785(3) (×2) | ||
| mean | 1.770 |
| Atom | M1 | M2 | M3 | M4 | M5 | T | Σ anions |
|---|---|---|---|---|---|---|---|
| O1 | - | 0.41 | - | - | 0.53×2 | 0.69 | 2.16 |
| O2 | - | - | 0.78 | - | 0.63×2 | - | 2.04 |
| O3 | 0.43 | - | - | 0.51 | 0.63×2 | - | 2.20 |
| O4 | - | - | 0.38 | - | 0.40×2 | 0.71 | 1.89 |
| O5 | - | 0.94 | - | 0.69 | - | - | 1.63 |
| O6 | 0.10×2 | - | 0.37×2 | 0.61×2 | 0.55 | - | 1.63 |
| O7 | - | 0.80×2 | - | 0.46×2 | 0.49 | - | 1.75 |
| O8 | 0.17×2 | 0.45×2 | 0.43×2 | - | - | 0.65×2 | 1.70 |
| Σ cations | 0.97 | 3.85 | 2.76 | 3.34 | 3.23 | 2.70 | - |
| 1 | 2 | |||
|---|---|---|---|---|
| hkl | dobs | Iest | dcalc | Icalc |
| 101 | - | - | 8.2017 | 31 |
| 201 | 5.78 | 20 | 5.7768 | 21 |
| 002 | 5.04 | 65 | 5.0424 | 35 |
| 011 | 5.0358 | 33 | ||
| 210 | - | - | 4.4841 | 17 |
| 202 | - | - | 4.1008 | 23 |
| 211 | 4.09 | 60 | 4.0973 | 56 |
| 212 | - | - | 3.3508 | 13 |
| 401 | - | - | 3.3266 | 11 |
| 410 | - | - | 3.0133 | 15 |
| 312 | 2.961 | 100 | 2.9587 | 100 |
| 013 | - | - | 2.9100 | 21 |
| 020 | - | - | 2.9062 | 17 |
| 411 | 2.885 | 40 | 2.8871 | 38 |
| 303 | 2.732 | 30 | 2.7339 | 32 |
| 213 | - | - | 2.6897 | 28 |
| 220 | - | - | 2.6867 | 18 |
| 221 | 2.597 | 20 | 2.5961 | 23 |
| 122 | - | - | 2.4787 | 18 |
| 413 | - | - | 2.2438 | 14 |
| 322 | - | - | 2.2193 | 14 |
| 421 | - | - | 2.1886 | 11 |
| 611 | - | - | 2.1289 | 10 |
| 404 | 2.051 | 25 | 2.0504 | 31 |
| 422 | 2.047 | 60 | 2.0486 | 54 |
| 504 | - | - | 1.8793 | 11 |
| 522 | - | - | 1.8779 | 16 |
| 332 | - | - | 1.6878 | 15 |
| 225 | - | - | 1.6130 | 12 |
| 722 | - | - | 1.5726 | 22 |
| 026 | 1.456 | 30 | 1.4550 | 25 |
| 040 | - | - | 1.4531 | 21 |
| 822 | - | - | 1.4436 | 18 |
| 1 | 3.9377 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Griffin, W.L.; Gain, S.E.M.; Bindi, L.; Toledo, V.; Cámara, F.; Saunders, M.; O’Reilly, S.Y. Carmeltazite, ZrAl2Ti4O11, a New Mineral Trapped in Corundum from Volcanic Rocks of Mt Carmel, Northern Israel. Minerals 2018, 8, 601. https://doi.org/10.3390/min8120601
Griffin WL, Gain SEM, Bindi L, Toledo V, Cámara F, Saunders M, O’Reilly SY. Carmeltazite, ZrAl2Ti4O11, a New Mineral Trapped in Corundum from Volcanic Rocks of Mt Carmel, Northern Israel. Minerals. 2018; 8(12):601. https://doi.org/10.3390/min8120601
Chicago/Turabian StyleGriffin, William L., Sarah E. M. Gain, Luca Bindi, Vered Toledo, Fernando Cámara, Martin Saunders, and Suzanne Y. O’Reilly. 2018. "Carmeltazite, ZrAl2Ti4O11, a New Mineral Trapped in Corundum from Volcanic Rocks of Mt Carmel, Northern Israel" Minerals 8, no. 12: 601. https://doi.org/10.3390/min8120601
APA StyleGriffin, W. L., Gain, S. E. M., Bindi, L., Toledo, V., Cámara, F., Saunders, M., & O’Reilly, S. Y. (2018). Carmeltazite, ZrAl2Ti4O11, a New Mineral Trapped in Corundum from Volcanic Rocks of Mt Carmel, Northern Israel. Minerals, 8(12), 601. https://doi.org/10.3390/min8120601








