The Earliest Generation of Diamond: The First Find of a Diamond Inclusion in Kimberlitic Olivine
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Olivine Composition
3.2. Inclusions Description
3.3. Micro-Raman Spectroscopy Data
3.4. Re-Os Isotope Data
4. Discussion
5. Conclusions
- The crystallization of the diamond inclusion that we found must have proceeded either from silicate melt, enriched with a carbonate component (carbon-bearing fluid), or proceeded from sulfide melt with dissolved carbon (Fe-Ni-Cu-Co-S-C melt).
- Crystallization of the studied diamond occurred at temperatures at least 200–250 °C higher than for the crystallization of later metasomatic diamonds: in the case of silicate melt, at 1400 °C; in the case of sulfide melt, at 1600 °C.
- The diamond we discovered is apparently the oldest to date. The age of the syngenetic inclusion of sulfide is estimated at ~3.6 billion years.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prinz, M.; Manson, D.V.; Hlava, P.F.; Keil, K. Inclusions in diamonds: Garnet lherzolite and eclogite assemblages. Phys. Chem. Earth 1975, 9, 797–815. [Google Scholar] [CrossRef]
- Efimova, E.S.; Sobolev, N.V.; Pospelova, L.N. Sulfide inclusions in diamond and their paragenesis features. Rep. Russ. Mineral. Soc. 1983, 112, 300–310. (In Russian) [Google Scholar]
- Deines, P.; Harris, J.W. Sulfide inclusion chemistry and carbon isotopes of African diamonds. Geochim. Cosmochim. Acta 1995, 59, 3173–3188. [Google Scholar] [CrossRef]
- Schulze, D.J.; Coopersmith, H.G.; Harte, B.; Pizzolato, L.A. Mineral inclusions in diamonds from the Kelsey Lake Mine, Colorado, USA: Depleted Archean mantle beneath the Proterozoic Yavapai province. Geochim. Cosmochim. Acta 2008, 72, 1685–1695. [Google Scholar] [CrossRef]
- Jean, M.M.; Taylor, L.A.; Howarth, G.H.; Peslier, A.H.; Fedele, L.; Bodnar, R.J.; Guan, Y.; Doucet, L.S.; Ionov, D.A.; Logvinova, A.M.; et al. Olivine inclusions in Siberian diamonds and mantle xenoliths: Contrasting water and trace-element contents. Lithos 2016, 265, 31–41. [Google Scholar] [CrossRef]
- Carvalho, L.D.V.; Schnellrath, J.; Medeiros, S.R. Mineral inclusions in diamonds from Chapada Diamantina, Bahia, Brazil: A Raman spectroscopic characterization. REM Int. Eng. J. 2018, 71, 27–35. [Google Scholar] [CrossRef][Green Version]
- Nestola, F.; Zaffiro, G.; Mazzucchelli, M.L.; Nimis, P.; Andreozzi, G.B.; Periotto, B.; Princivalle, F.; Lenaz, D.; Secco, L.; Pasqualetto, L.; et al. Diamond-inclusion system recording old deep lithosphere conditions at Udachnaya (Siberia). Sci. Rep. 2019, 9, 12586. [Google Scholar] [CrossRef]
- Sobolev, N.V.; Logvinova, A.M.; Tomilenko, A.A.; Wirth, R.; Bul’bak, T.A.; Luk’yanova, L.I.; Fedorova, E.N.; Reutsky, V.N.; Efimova, E.S. Mineral and fluid inclusions in diamonds from the Urals placers, Russia: Evidence for solid molecular N-2 and hydrocarbons in fluid inclusions. Geochim. Cosmochim. Acta 2019, 266, 197–219. [Google Scholar] [CrossRef]
- Pokhilenko, N.P.; Sobolev, N.V.; Lavrent’ev, Y.G. Xenoliths of diamondiferous ultramafic rocks from Yakutian kimberlites. In Proceedings of the II International Kimberlite Conference, Santa Fe, NM, USA, 3–7 October 1977; Available online: https://archive.org/details/secondinternatio02inte (accessed on 24 November 2022).
- Ilupin, I.P.; Efimova, E.S.; Sobolev, N.V.; Usova, L.V.; Savrasov, D.I.; Kharkiv, A.D. Inclusions in diamond from diamondiferous dunite. Dokl. Acad. Nauk SSSR 1982, 264, 454–456. [Google Scholar]
- Sobolev, N.V.; Pokhilenko, N.P.; Efimova, E.S. Xenoliths of diamondiferous peridotite in kimberlites and the origin of diamond. Soviet Geol. Geophys. 1984, 25, 63–80. [Google Scholar]
- Pokhilenko, N.P.; Pearson, D.G.; Boyd, F.R.; Sobolev, N.V. Megacrystalline Dunites: Sources of Siberian Diamonds. In Annual Report of Director Geophysical Laboratory; Carnegie Institution: Washington, DC, USA, 1991; pp. 11–18. [Google Scholar]
- Pokhilenko, N.P.; Sobolev, N.V.; Boyd, F.R.; Pearson, D.G.; Shimizu, N. Megacrystalline pyrope peridotites in the lithosphere of the Siberian platform: Mineralogy, geochemical peculiarities and the problem of their origin. Russ. Geol. Geophys. 1993, 34, 71–84. [Google Scholar]
- Sobolev, N.V. Deep-Seated Inclusions in Kimberlites and the Problem of the Composition of the Upper Mantle; American Geological Union: Washington, DC, USA, 1977; 279p. [Google Scholar]
- Garanin, V.K.; Zaborovskii, V.V.; Kudryavtseva, G.P.; Mikhailichenko, O.A.; Sobolev, N.V. Mineralogy of Diamondiferous Garnet Nodules from the Udachnaya Kimberlite (Yakutia). In Proceedings of the International Symposium Deep Subcontinental Lithosphere: Composition and Processes, Novosibirsk, Russia, 30 May–2 June 1988. [Google Scholar]
- Barashkov, Y.P.; Zudin, N.G. The composition of garnets with diamond inclusions from the Krasnopresnenskaya kimberlite pipe (Yakutia). Russ. Geol. Geophys. 1997, 38, 353–357. [Google Scholar]
- Spetsius, Z.V.; Taylor, L.A. Diamonds of Siberia: Photographic Evidence for their Origin; Tranquility Base Press: Lenoir City, TN, USA, 2008; 278p. [Google Scholar]
- Howarth, G.H.; Sobolev, N.V.; Pernet-Fisher, J.F.; Ketcham, R.A.; Maisano, J.A.; Pokhilenko, L.N.; Taylor, D.; Taylor, L.A. 3-D X-ray tomography of diamondiferous mantle eclogite xenoliths, Siberia. J. Asian Earth Sci. 2015, 101, 39–67. [Google Scholar] [CrossRef]
- Laz’ko, E.E.; Pokhilenko, N.P.; Lavrentiev, Y.G.; Usova, L.V. Compositions of mineral inclusions in olivines from the Udachnaya kimberlite (Yakutia). Soviet Geol. Geophys. 1976, 17, 107–112. [Google Scholar]
- Kamenetsky, V.S.; Kamenetsky, M.B.; Sobolev, A.V.; Golovin, A.V.; Demouchy, S.; Faure, K.; Sharygin, V.V.; Kuzmin, D.V. Olivine in the Udachnaya-East Kimberlite (Yakutia, Russia): Types, Compositions and Origins. J. Petrol. 2008, 49, 823–839. [Google Scholar] [CrossRef]
- Pearson, N.J.; Alard, O.; Griffin, W.L.; Jackson, S.E.; O’Reilly, S.Y. In situ measurement of Re–Os isotopes in mantle sulfides by laser ablation multicollector-inductively coupled plasma mass spectrometry: Analytical methods and preliminary results. Geochim. Cosmochim. Acta 2002, 66, 1037–1050. [Google Scholar] [CrossRef]
- Alard, O.; Griffin, W.L.; Lorand, J.-P.; Jackson, S.E.; O’Reilly, S.Y. Non-chondritic distribution of the highly siderophile elements in mantle sulfides. Nature 2000, 407, 891–894. [Google Scholar] [CrossRef]
- Griffin, W.L.; Powell, W.J.; Pearson, N.J.; O’Reilly, S.Y. GLITTER: Data reduction software for laser ablation ICP-MS. In Laser Ablation ICP-MS in the Earth Sciences: Current Practices and Outstanding Issues; Sylvester, P.J., Ed.; Mineralogical Association of Canada Short Course Series; Short Course 40; Mineralogical Association of Canada: Vancouver, BC, Canada, 2008; pp. 308–311. [Google Scholar]
- Pokhilenko, L.N. The Fluid Regime of Lithospheric Mantle in the Siberian Craton (from Mantle Xenoliths in Kimberlites). Author’s Abstract. Ph.D. Thesis, Institute of Geology & Mineralogy, Novosibirsk, Russia, 2006; 16p. (In Russian). [Google Scholar]
- Sobolev, N.V.; Logvinova, A.M.; Zedgenizov, D.A.; Pokhilenko, N.P.; Malygina, E.V.; Kuzmin, D.V.; Sobolev, A.V. Petrogenetic significance of minor elements in olivines from diamonds and peridotite xenoliths from kimberlites of Yakutia. Lithos 2009, 112, 701–713. [Google Scholar] [CrossRef]
- Pokhilenko, L.N.; Mal’kovets, V.G.; Kuz’min, D.V.; Pokhilenko, N.P. New Data on the Mineralogy of Megacrystalline Pyrope Peridotite from the Udachnaya Kimberlite Pipe, Siberian Craton, Yakutian Diamondiferous Province. Dokl. Earth Sci. 2014, 454, 179–184. [Google Scholar] [CrossRef]
- Arndt, N.T.; Guitreau, M.; Boullier, A.-M.; Le Roex, A.; Tommasi, A.; Cordier, P.; Sobolev, A.V. Olivine, and the origin of kimberlite. J. Petrol. 2010, 51, 573–602. [Google Scholar] [CrossRef]
- Donnelly, C.L.; Stachel, T.; Creighton, S.; Muehlenbachs, K.; Whiteford, S. Diamonds and their mineral inclusions from the A154 South pipe, Diavik Diamond Mine, Northwest territories, Canada. Lithos 2007, 98, 160–176. [Google Scholar] [CrossRef]
- Sobolev, N.V.; Logvinova, A.M.; Zedgenizov, D.A.; Pokhilenko, N.P.; Kuzmin, D.V.; Sobolev, A.V. Olivine inclusions in Siberian diamonds: High-precision approach to minor elements. Eur. J. Mineral. 2008, 20, 305–315. [Google Scholar] [CrossRef]
- Van Rythoven, A.D.; Schulze, D.J. In situ analysis of diamonds and their inclusions from the Diavik Mine, Northwest Territories, Canada: Mapping diamond growth. Lithos 2009, 112, 870–879. [Google Scholar] [CrossRef]
- Shatsky, V.S.; Zedgenizov, D.A.; Ragozin, A.L.; Kalinina, V.V. Diamondiferous subcontinental lithospheric mantle of the northeastern Siberian Craton: Evidence from mineral inclusions in alluvial diamonds. Gondwana Res. 2015, 28, 106–120. [Google Scholar] [CrossRef]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies: Tables and Charts, 3rd ed.; John Wiley and Sons, Ltd.: Chichester, UK, 2004; 368p. [Google Scholar]
- Griffin, W.L.; Spetsius, Z.V.; Pearson, N.J.; O’Reilly, S.Y. In situ Re–Os analysis of sulfide inclusions in kimberlitic olivine: New constraints on depletion events in the Siberian lithospheric mantle. Geochem. Geophys. Geosyst. 2002, 3, 1069. [Google Scholar] [CrossRef]
- Shirey, S.B.; Walker, R.J. The Re–Os isotope system in cosmochemistry and high-temperature geochemistry. Annu. Rev. Earth Planet. Sci. 1998, 26, 423–500. [Google Scholar] [CrossRef]
- Pearson, D.G.; Shirey, S.B.; Carlson, R.W.; Boyd, F.R.; Pokhilenko, N.P.; Shimizu, N. Re–Os, Sm–Nd, and Rb–Sr isotope evidence for thick Archaean lithospheric mantle beneath the Siberian craton modified by multistage metasomatism. Geochim. Cosmochim. Acta 1995, 59, 959–977. [Google Scholar] [CrossRef]
- Klein-Ben David, O.; Logvinova, A.M.; Schrauder, M.; Spetius, Z.V.; Weiss, Y.; Hauri, E.H.; Kaminsky, F.V.; Sobolev, N.V.; Navon, O. High-Mg carbonatitic microinclusions in some Yakutian diamonds—A new type of diamond-forming fluid. Lithos 2009, 112, 648–659. [Google Scholar] [CrossRef]
- Klein-BenDavid, O.; Pearson, D.G.; Nowell, G.M.; Ottley, C.; McNeill, J.C.R.; Logvinova, A.M.; Sobolev, N.V. The sources and time-integrated evolution of diamond-forming fluids—Trace elements and isotopic evidence. Geochim. Cosmochim. Acta 2014, 125, 146–169. [Google Scholar] [CrossRef]
- Logvinova, A.M.; Wirth, R.; Fedorova, E.N.; Sobolev, N.V. Nanometre-sized mineral and fluid inclusions in cloudy Siberian diamonds: New insights on diamond formation. Eur. J. Mineral. 2008, 20, 317–331. [Google Scholar] [CrossRef]
- Logvinova, A.M.; Wirth, R.; Tomilenko, A.A.; Afanasiev, V.P.; Sobolev, N.V. The phase composition of crystal-fluid nanoinclusions in alluvial diamonds in the northeastern Siberian Platform. Russ. Geol. Geophys. 2011, 52, 1286–1297. [Google Scholar] [CrossRef]
- Zedgenizov, D.A.; Rege, S.; Griffin, W.L.; Kagi, H.; Shatsky, V.S. Composition of trapped fluids in cuboid fibrous diamonds from the Udachnaya kimberlite: LAM-ICPMS analysis. Chem. Geol. 2007, 240, 151–162. [Google Scholar] [CrossRef]
- Pokhilenko, N.P.; Agashev, A.M.; Litasov, K.D.; Pokhilenko, L.N. Carbonatite metasomatism of peridotite lithospheric mantle: Implications for diamond formation and carbonatite-kimberlite magmatism. Russ. Geol. Geophys. 2015, 56, 280–295. [Google Scholar] [CrossRef]
- Pal’yanov, Y.N.; Sokol, A.G.; Borzdov, Y.M.; Khokhryakov, A.F.; Sobolev, N.V. Diamond formation through carbonate-silicate interaction. Am. Mineral. 2002, 87, 1009–1013. [Google Scholar] [CrossRef]
- Litvin, Y.A.; Bobrov, A.V.; Vasili’ev, P.G.; Okoemova, V.Y.; Kuzyura, A.V. Parental media of natural diamonds and primary mineral inclusions in them: Evidence from physicochemical experiment. Geochem. Int. 2012, 50, 726–759. [Google Scholar] [CrossRef]
- Shirey, S.B.; Cartigny, P.; Frost, D.J.; Keshav, S.; Nestola, F.; Nimis, P.; Pearson, D.G.; Sobolev, N.V.; Walter, M.J. Diamonds and the geology of mantle carbon. Rev. Mineral. Geochem. 2013, 75, 355–421. [Google Scholar] [CrossRef]
- Litasov, K.D. Physicochemical Conditions of Melting in the Mantle in the Presence of Volatiles (from Experimental Data). Ph.D. Thesis, Institute of Geology & Mineralogy, Novosibirsk, Russia, 2011; 35p. (In Russian). [Google Scholar]
- Boyd, F.R.; Pokhilenko, N.P.; Pearson, D.G.; Mertzman, S.A.; Sobolev, N.V.; Finger, L.W. Composition of the Siberian cratonic mantle: Evidence from Udachnaya peridotite xenoliths. Contrib. Mineral. Petrol. 1997, 128, 228–246. [Google Scholar] [CrossRef]
- Stachel, T.; Harris, J. The origin of cratonic diamonds–constraints from mineral inclusions. Ore Geol. Rev. 2008, 34, 5–32. [Google Scholar] [CrossRef]
- Botkunov, A.I.; Garanin, V.K.; Gotovtsev, V.V.; Kudryavtseva, G.P. Sulfide inclusions in olivine from the Udachnaya kimberlite. Dokl. Akad. Nauk SSSR 1979, 247, 929–932. (In Russian) [Google Scholar]
- Meyer, H.O.A. Inclusions in Diamond. In Mantle Xenoliths; Nixon, P.H., Ed.; John Wiley and Sons: Chichester, UK; New York, NY, USA, 1987; pp. 501–523. [Google Scholar]
- Bulanova, G.P.; Spetsius, Z.V.; Leskova, N.V. Sulfides in Diamonds and Xenoliths from Kimberlitic Pipes of Yakutia; Nauka: Novosibirsk, Russia, 1990; 120p. (In Russian) [Google Scholar]
- Shushkanova, A.V.; Litvin, Y.A. Experimental liquid immiscibility in sulphide-silicate pyrrhotite-garnet system at 7 GPa: Implication to origin of diamond and syngenetic inclusions. Lithos 2004, 73, 101. [Google Scholar]
- Zhimulev, E.I.; Shein, M.A.; Pokhilenko, N.P. Crystallization of diamond in the system Fe-S-C. Dokl. Earth Sci. 2013, 451, 729–731. [Google Scholar] [CrossRef]
- Spetsius, Z.V. The form, nature and conditions of partial melting of eclogites from the kimberlite pipes. Vestn. MGU Geol. 1980, 4, 101–105. [Google Scholar]
- Chepurov, A.I.; Zhimulev, E.I.; Sonin, V.M.; Chepurov, A.A.; Pokhilenko, N.P. Crystallization of diamond in metal-sulfide melts. Dokl. Earth Sci. 2009, 428, 1139–1141. [Google Scholar] [CrossRef]
- Zhimulev, E.I.; Chepurov, A.I.; Sinyakova, E.F.; Sonin, V.M.; Chepurov, A.A.; Pokhilenko, N.P. Diamond crystallization in the Fe-Co-S-C and Fe-Ni-S-C systems and the role of sulfide-metal melts in the genesis of diamond. Geochem. Int. 2012, 50, 205–216. [Google Scholar] [CrossRef]
- Pal’yanov, Y.N.; Borzdov, Y.M.; Khokhryakov, A.F.; Kupriyanov, I.N.; Sobolev, N.V. Sulfide melts-graphite interaction at HPHT conditions: Implications for diamond genesis. Earth. Planet. Sci. Lett. 2006, 250, 269–280. [Google Scholar] [CrossRef]
- Pal’yanov, Y.N.; Sokol, A.G.; Borzdov, Y.M.; Khokhryakov, A.F.; Shatsky, A.F.; Sobolev, N.V. The diamond growth from Li2CO3, Na2CO3, K2CO3 and Cs2CO3 solvent-catalysts at P = 7 GPa and T = 1700–1750 °C. Diamond Relat. Mater. 1999, 8, 1118–1124. [Google Scholar] [CrossRef]
- Tomlinson, E.; Jones, A.; Milledge, J. High-pressure experimental growth of diamond using C–K2CO3–KCl as an analogue for Cl-bearing carbonate fluid. Lithos 2004, 77, 287–294. [Google Scholar] [CrossRef]
- Akaishi, M.; Yamaoka, S. Crystallization of diamond from C–O–H fluids under high-pressure and high-temperature conditions. J. Cryst. Growth 2000, 209, 999–1003. [Google Scholar] [CrossRef]
- Sokol, A.G.; Pal’yanov, Y.N.; Pal’yanova, G.A.; Khokhryakov, A.F.; Borzdov, Y.M. Diamond and graphite crystallization from C–O–H fluids under high pressure and high temperature conditions. Diamond Relat. Mater. 2001, 10, 2131–2136. [Google Scholar] [CrossRef]
- Pal’yanov, Y.N.; Sokol, A.G.; Tomilenko, A.A.; Sobolev, N.V. Conditions of diamond formation through carbonate–silicate interaction. Eur. J. Mineral. 2005, 2, 207–214. [Google Scholar] [CrossRef]
- Marx, P.C. Pyrrhotine and the origin of terrestrial diamonds. Miner. Mag. 1972, 38, 636–638. [Google Scholar] [CrossRef]
- Pal’yanov, Y.N.; Borzdov, Y.M.; Bataleva, Y.V.; Sokol, A.G.; Pal’yanova, G.A.; Kupriyanov, I.N. Reducing role of sulfides and diamond formation in the Earth’s mantle. Earth. Planet. Sci. Lett. 2007, 260, 242–256. [Google Scholar] [CrossRef]
- Walker, R.; Carlson, R.; Shirey, S.; Boyd, F. Os, Sr, Nd, and Pb isotope systematics of southern African peridotite xenoliths and implications for the chemical evolution of the subcontinental mantle. Geochim. Cosmochim. Acta 1989, 53, 1583–1595. [Google Scholar] [CrossRef]
- Pokhilenko, N.P.; Sobolev, N.V.; Kuligin, S.S.; Shimizu, N. Peculiarities of distribution of pyroxenite paragenesis garnets in Yakutian kimberlites and some aspects of the evolution of the Siberian craton lithospheric mantle. In Proceedings of the 7th International Kimberlite Conference, Cape Town, South Africa, 1–17 April 1998. [Google Scholar] [CrossRef]
- Shimizu, N.; Sobolev, N.V. Young peridotitic diamonds from the Mir kimberlite pipe. Nature 1995, 375, 394–397. [Google Scholar] [CrossRef]
- Shimizu, N.; Pokhilenko, N.P.; Boyd, F.R.; Pearson, D.G. Geochemical characteristics of mantle xenoliths from Udachnaya kimberlite pipe. Geol. Geophys. 1997, 38, 194–205. Available online: https://repository.geologyscience.ru/handle/123456789/20596 (accessed on 24 November 2022).
- Shimizu, N.; Pokhilenko, N.P.; Boyd, F.R.; Pearson, D.G. Trace element characteristics of garnet dunites/harzburgites, host rocks for Siberian peridotitic diamonds. In Proceedings of the 7th International Kimberlite Conference, Cape Town, South Africa, 1–17 April 1998. [Google Scholar] [CrossRef]
- Malkovets, V.G.; Griffin, W.L.; O’Reilly, S.Y.; Wood, B.J. Diamond, subcalcic garnet, and mantle metasomatism: Kimberlite sampling patterns define the link. Geology 2007, 35, 339–342. [Google Scholar] [CrossRef]
- Griffin, W.L.; Belousova, E.A.; O’Neill, C.; O’Reilly, S.Y.; Malkovets, V.; Pearson, N.J.; Spetsius, S.; Wilde, S.A. The world turns over: Hadean–Archean crust–mantle evolution. Lithos 2014, 189, 2–15. [Google Scholar] [CrossRef]
- Howarth, G.H.; Barry, P.H.; Pernet-Fisher, J.F.; Baziotis, I.P.; Pokhilenko, N.P.; Pokhilenko, L.N.; Bodnar, R.J.; Taylor, L.A.; Agashev, A.M. Superplume metasomatism: Evidence from Siberian mantle xenoliths. Lithos 2014, 184–187, 209–224. [Google Scholar] [CrossRef]
- McCammon, C.A.; Griffin, W.L.; Shee, S.R.; O’Neill, H.S.C. Oxidation during metasomatism in ultramafic xenoliths from the Wesselton kimberlite, South Africa: Implications for the survival of diamond. Contrib. Mineral. Petrol. Contrib. Mineral. Petrol. 2001, 141, 287–296. [Google Scholar] [CrossRef]
- Goncharov, A.G.; Ionov, D.A.; Doucet, L.S.; Pokhilenko, L.N. Thermal state, oxygen fugacity and C-O–H fluid speciation in cratonic lithospheric mantle: New data on peridotite xenoliths from the Udachnaya kimberlite, Siberia. Earth Planet. Sci. Lett. 2012, 357–358, 99–110. [Google Scholar] [CrossRef]
- Tomilenko, A.; Chepurov, A.; Pal’yanov, Y.; Pokhilenko, L.; Shebanin, A. Volatile components in the upper mantle (based on data on fluid inclusion studies). Russ. Geol. Geophys. 1997, 38, 276–285. [Google Scholar]
- Sharygin, I.S.; Golovin, A.V.; Pokhilenko, N.P. Djerfisherite in xenoliths of sheared peridotite in the Udachnaya-East pipe (Yakutia): Origin and relationship with kimberlitic magmatism. Russ. Geol. Geophys. 2012, 53, 247–261. [Google Scholar] [CrossRef]
- Pearson, D.G.; Shirey, S.B.; Bulanova, G.P.; Carlson, R.W.; Milledge, H.J. Re–Os isotope measurements of single sulfide inclusions in a Siberian diamond and its nitrogen aggregation systematics. Geochim. Cosmochim. Acta 1999, 63, 703–711. [Google Scholar] [CrossRef]
- Pearson, D.G.; Bulanova, G.P.; Shirey, S.B.; Carlson, R.W.; Milledge, H.J.; Barashkov, Y.P. Re-Os isotope constraints on the ages of Siberian diamonds. J. Conf. Abstr. 2000, 5, 776. [Google Scholar]

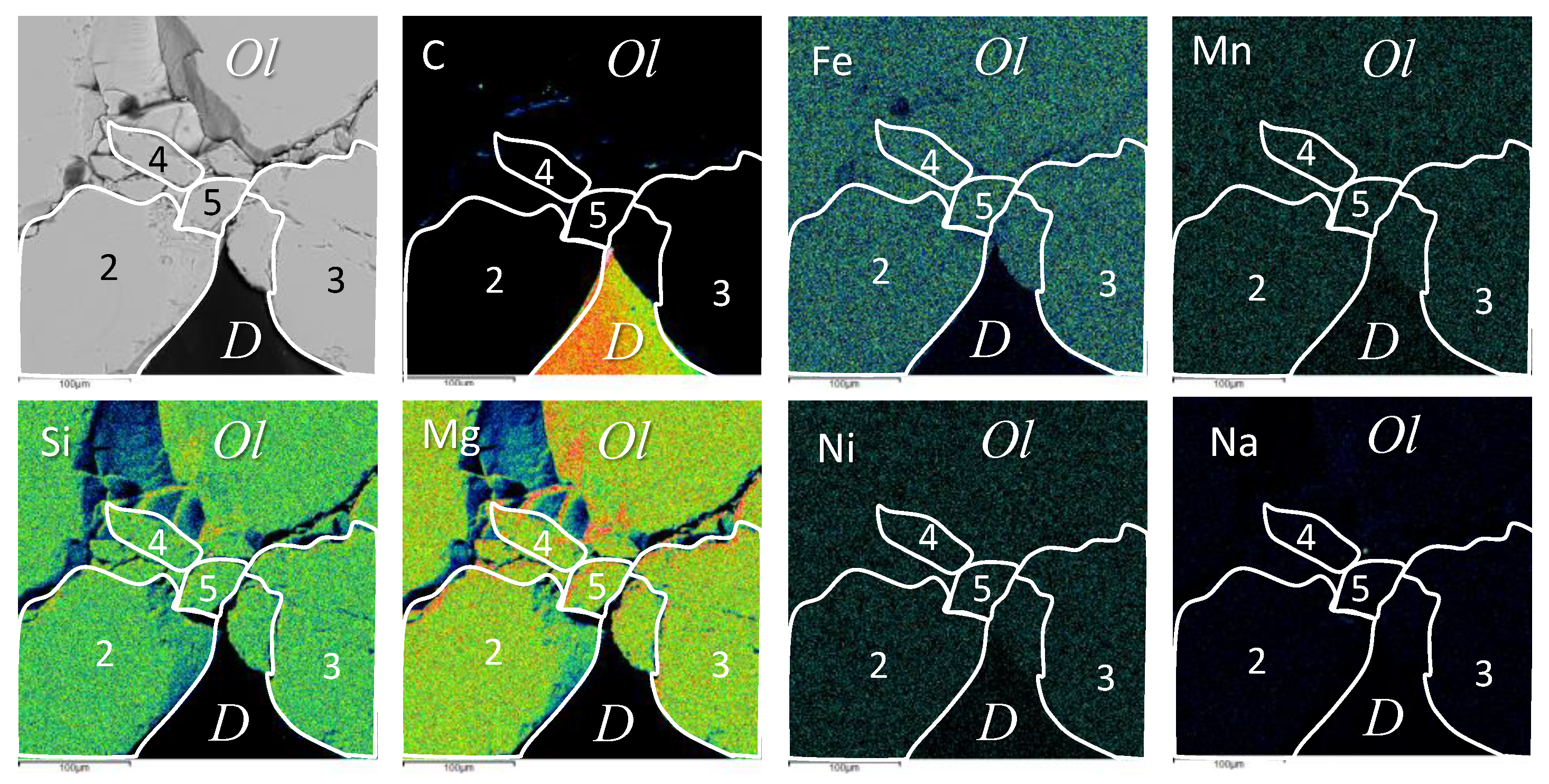


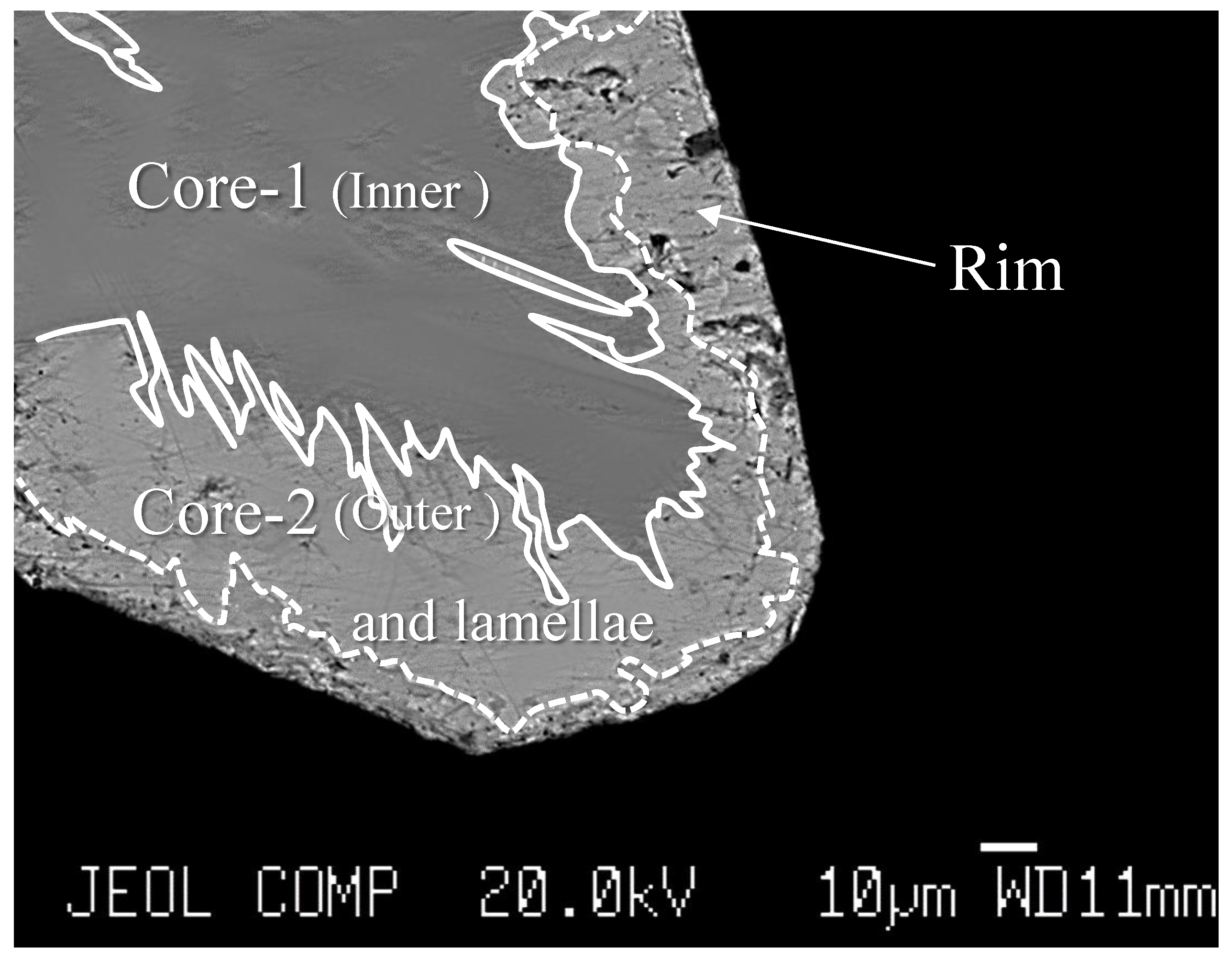
| N | 1–1 | 1–2 | 1–3 | 1–4 | 1–5 | Average |
|---|---|---|---|---|---|---|
| SiO2 | 42.11 | 42.11 | 41.79 | 41.89 | 41.84 | 41.95 |
| Cr2O3 | 0.045 | 0.046 | 0.045 | 0.049 | 0.046 | 0.046 |
| FeO | 6.76 | 6.74 | 6.75 | 6.74 | 6.74 | 6.75 |
| MnO | 0.081 | 0.082 | 0.083 | 0.081 | 0.084 | 0.082 |
| MgO | 51.31 | 51.21 | 50.94 | 51.12 | 51.04 | 51.12 |
| CaO | 0.013 | 0.013 | 0.011 | 0.010 | 0.013 | 0.012 |
| NiO | 0.348 | 0.346 | 0.347 | 0.347 | 0.352 | 0.348 |
| Total | 100.7 | 100.5 | 99.97 | 100.2 | 100.1 | 100.3 |
| N | 2 | 3 | 4 | 5 |
|---|---|---|---|---|
| SiO2 | 40.93 | 40.9 | 41.31 | 41.18 |
| Al2O3 | 0.11 | 0.11 | 0.09 | 0.13 |
| Cr2O3 | 0.06 | 0.04 | 0.06 | 0.06 |
| FeO | 6.63 | 6.65 | 6.73 | 6.64 |
| MnO | 0.08 | 0.09 | 0.09 | 0.05 |
| MgO | 51.09 | 51.03 | 51.47 | 51.36 |
| Na2O | 0 | 0.05 | 0.05 | 0.04 |
| NiO | 0.38 | 0.39 | 0.39 | 0.39 |
| Total | 99.27 | 99.28 | 100.2 | 99.86 |
| wt% | Core-1 (Inner Part) | ||||||||
| N | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
| S | 39.31 | 39.58 | 39.20 | 39.15 | 39.30 | 39.49 | 39.75 | 39.24 | 39.22 |
| Fe | 52.50 | 52.62 | 51.86 | 51.77 | 49.90 | 49.90 | 49.09 | 52.87 | 50.22 |
| Cr | 0.20 | 0.18 | 0.21 | 0.22 | 0.24 | 0.13 | 0.15 | 0.18 | 0.16 |
| Co | 0.18 | 0.13 | 0.13 | 0.15 | 0.13 | 0.16 | 0.28 | 0.11 | 0.13 |
| Ni | 7.75 | 7.35 | 8.04 | 8.03 | 10.46 | 10.37 | 10.02 | 7.13 | 10.09 |
| Cu | 0.02 | 0.00 | 0.01 | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 | 0.00 |
| Total | 99.95 | 99.86 | 99.45 | 99.32 | 100.04 | 100.05 | 99.29 | 99.53 | 99.83 |
| at% | Core-1 (Inner Part) | ||||||||
| N | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
| S | 53.19 | 53.50 | 53.28 | 53.28 | 53.19 | 53.40 | 53.97 | 53.28 | 53.19 |
| Fe | 40.78 | 40.83 | 40.47 | 40.45 | 38.77 | 38.73 | 38.26 | 41.20 | 39.10 |
| Cr | 0.16 | 0.15 | 0.18 | 0.18 | 0.20 | 0.11 | 0.13 | 0.15 | 0.14 |
| Co | 0.13 | 0.09 | 0.10 | 0.11 | 0.10 | 0.12 | 0.21 | 0.08 | 0.09 |
| Ni | 5.72 | 5.42 | 5.96 | 5.97 | 7.73 | 7.65 | 7.43 | 5.29 | 7.47 |
| Cu | 0.01 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 |
| Total | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| wt% | Core-2 (Outer Part) and Lamellae | Rim | |||||||
| N | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 1 |
| S | 32.97 | 33.33 | 33.27 | 33.11 | 33.46 | 33.49 | 33.08 | 33.29 | 34.92 |
| Fe | 27.22 | 27.16 | 27.17 | 27.14 | 27.52 | 27.93 | 27.27 | 27.30 | 30.75 |
| Cr | 0.03 | 0.04 | 0.04 | 0.04 | 0.17 | 0.18 | 0.19 | 0.05 | 0.04 |
| Co | 0.97 | 1.08 | 0.95 | 0.90 | 0.50 | 0.72 | 0.55 | 0.97 | 0.06 |
| Ni | 38.96 | 38.71 | 38.68 | 38.69 | 38.64 | 37.94 | 38.61 | 38.64 | 1.09 |
| Cu | 0.04 | 0.00 | 0.00 | 0.03 | 0.00 | 0.04 | 0.00 | 0.02 | 33.38 |
| Total | 100.17 | 100.32 | 100.12 | 99.90 | 100.29 | 100.30 | 99.71 | 100.27 | 100.23 |
| at% | Core-2 (Outer Part) and Lamellae | Rim | |||||||
| N | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 1 |
| S | 46.81 | 47.16 | 47.17 | 47.06 | 47.30 | 47.33 | 47.10 | 47.14 | 49.84 |
| Fe | 22.19 | 22.06 | 22.11 | 22.15 | 22.34 | 22.66 | 22.29 | 22.18 | 25.20 |
| Cr | 0.02 | 0.04 | 0.04 | 0.03 | 0.15 | 0.16 | 0.17 | 0.04 | 0.03 |
| Co | 0.75 | 0.83 | 0.73 | 0.69 | 0.38 | 0.55 | 0.43 | 0.75 | 0.05 |
| Ni | 30.21 | 29.92 | 29.94 | 30.04 | 29.83 | 29.28 | 30.02 | 29.88 | 0.85 |
| Cu | 0.03 | 0.00 | 0.00 | 0.02 | 0.00 | 0.03 | 0.00 | 0.01 | 24.03 |
| Total | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| 1 | 2 | 3 |
|---|---|---|
| Os | 731 ppm | |
| 187Re/188Os | 0.05261 | 0.00039 |
| 187Os/188Os | 0.105736 | 0.000045 |
| 187Os/188Os360 | 0.105419 | |
| ɣOs360 | −17.1 | |
| T (MA) | 3.58 | 0.03 |
| T (RD)360 | 3.15 | 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pokhilenko, L.; Pokhilenko, N.; Malkovets, V.; Alifirova, T. The Earliest Generation of Diamond: The First Find of a Diamond Inclusion in Kimberlitic Olivine. Minerals 2023, 13, 36. https://doi.org/10.3390/min13010036
Pokhilenko L, Pokhilenko N, Malkovets V, Alifirova T. The Earliest Generation of Diamond: The First Find of a Diamond Inclusion in Kimberlitic Olivine. Minerals. 2023; 13(1):36. https://doi.org/10.3390/min13010036
Chicago/Turabian StylePokhilenko, Lyudmila, Nikolay Pokhilenko, Vladimir Malkovets, and Taisia Alifirova. 2023. "The Earliest Generation of Diamond: The First Find of a Diamond Inclusion in Kimberlitic Olivine" Minerals 13, no. 1: 36. https://doi.org/10.3390/min13010036
APA StylePokhilenko, L., Pokhilenko, N., Malkovets, V., & Alifirova, T. (2023). The Earliest Generation of Diamond: The First Find of a Diamond Inclusion in Kimberlitic Olivine. Minerals, 13(1), 36. https://doi.org/10.3390/min13010036






