Trace-Element Geochemistry of Sulfides in Upper Mantle Lherzolite Xenoliths from East Antarctica
Abstract
1. Introduction
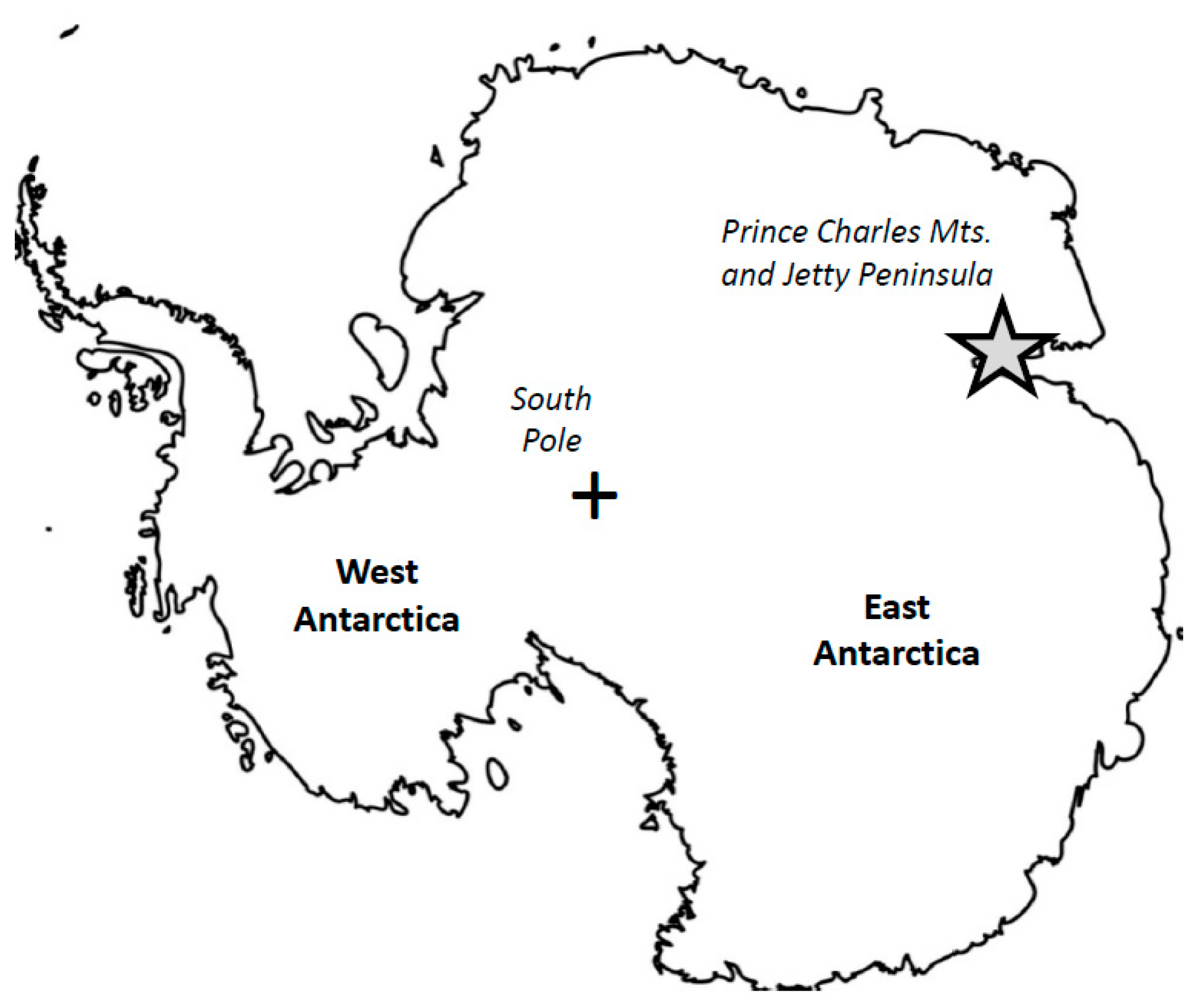
2. Materials and Methods
2.1. Brief Characteristics of Lherzolite Xenoliths
2.2. Analytical Techniques
2.2.1. SEM
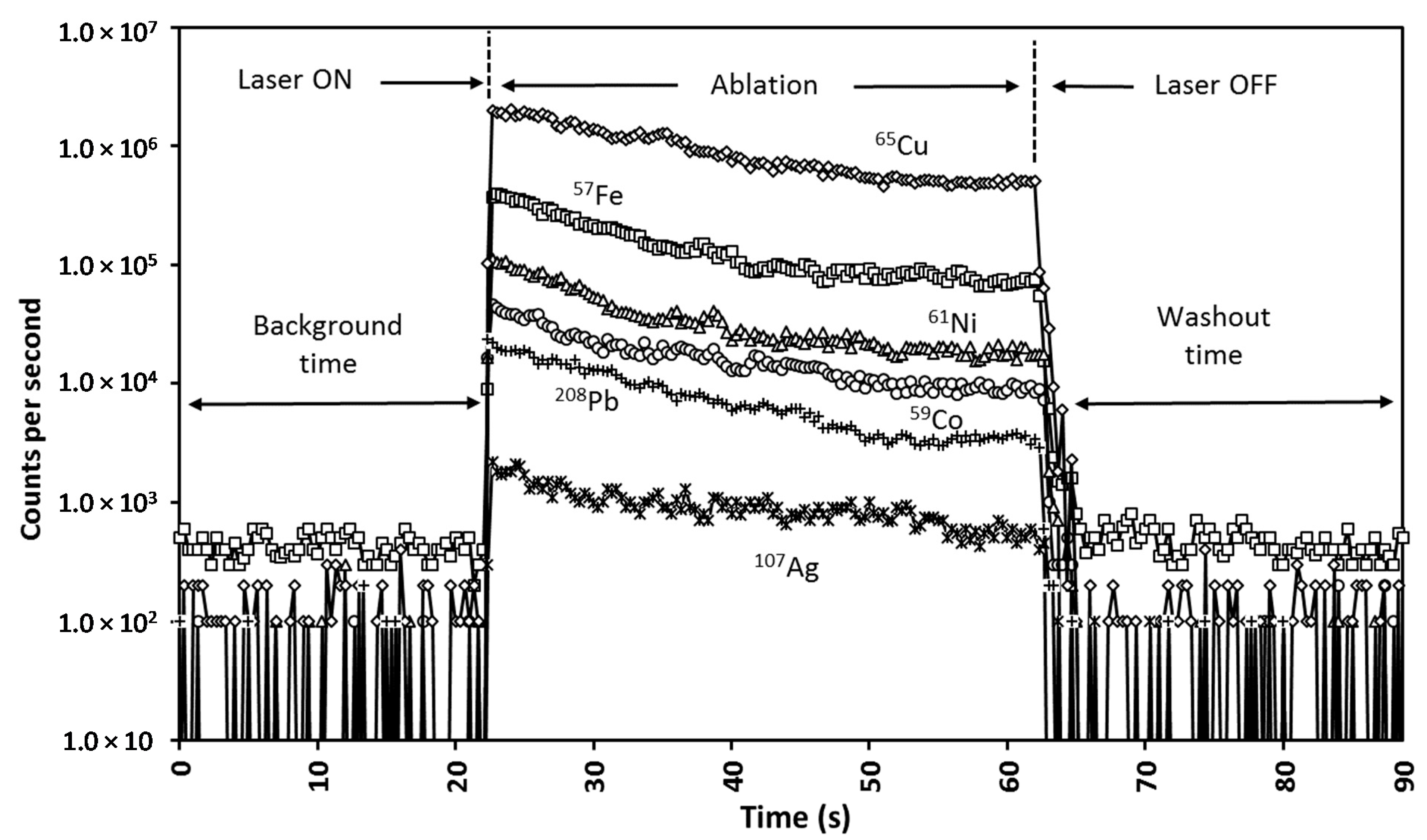
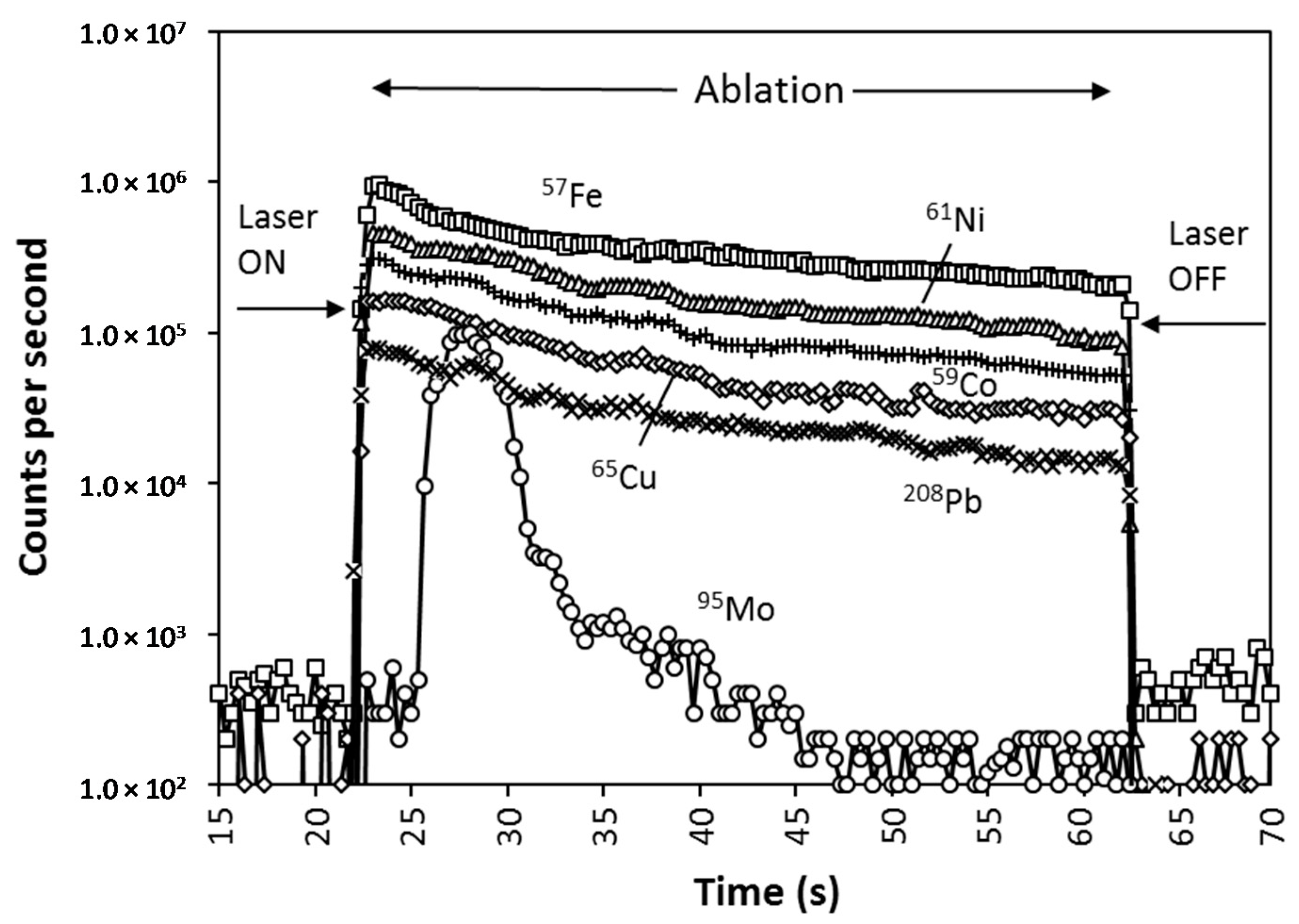
2.2.2. LA-ICP-MS
3. Results
3.1. Sulfide Petrography and Major-Element Composition
3.1.1. Sulfides in Cpx-Poor Spl Lherzolites
3.1.2. Sulfides in Cpx-Rich Spl Lherzolites


3.1.3. Sulfides in Spl-Grt Lherzolites
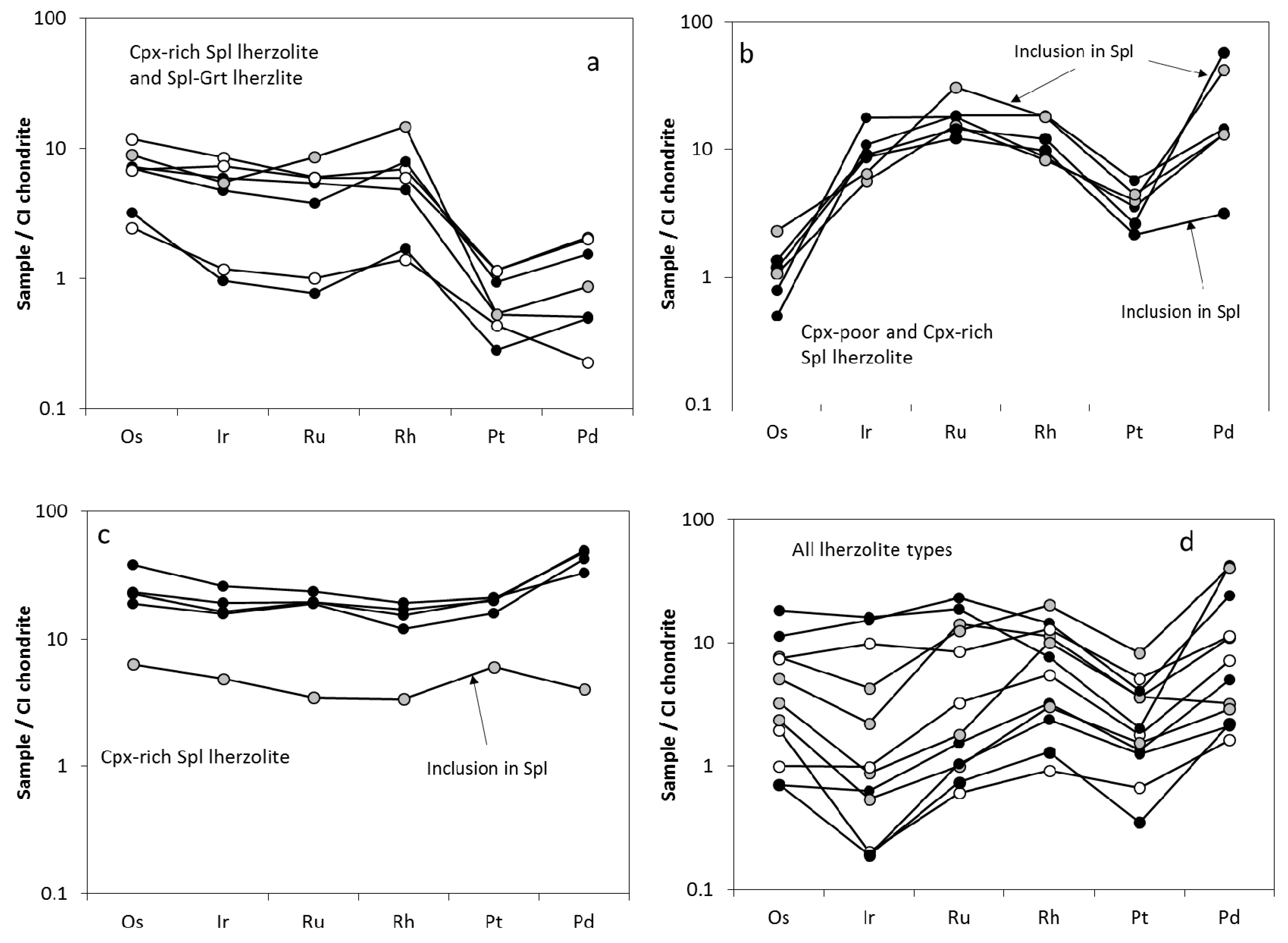
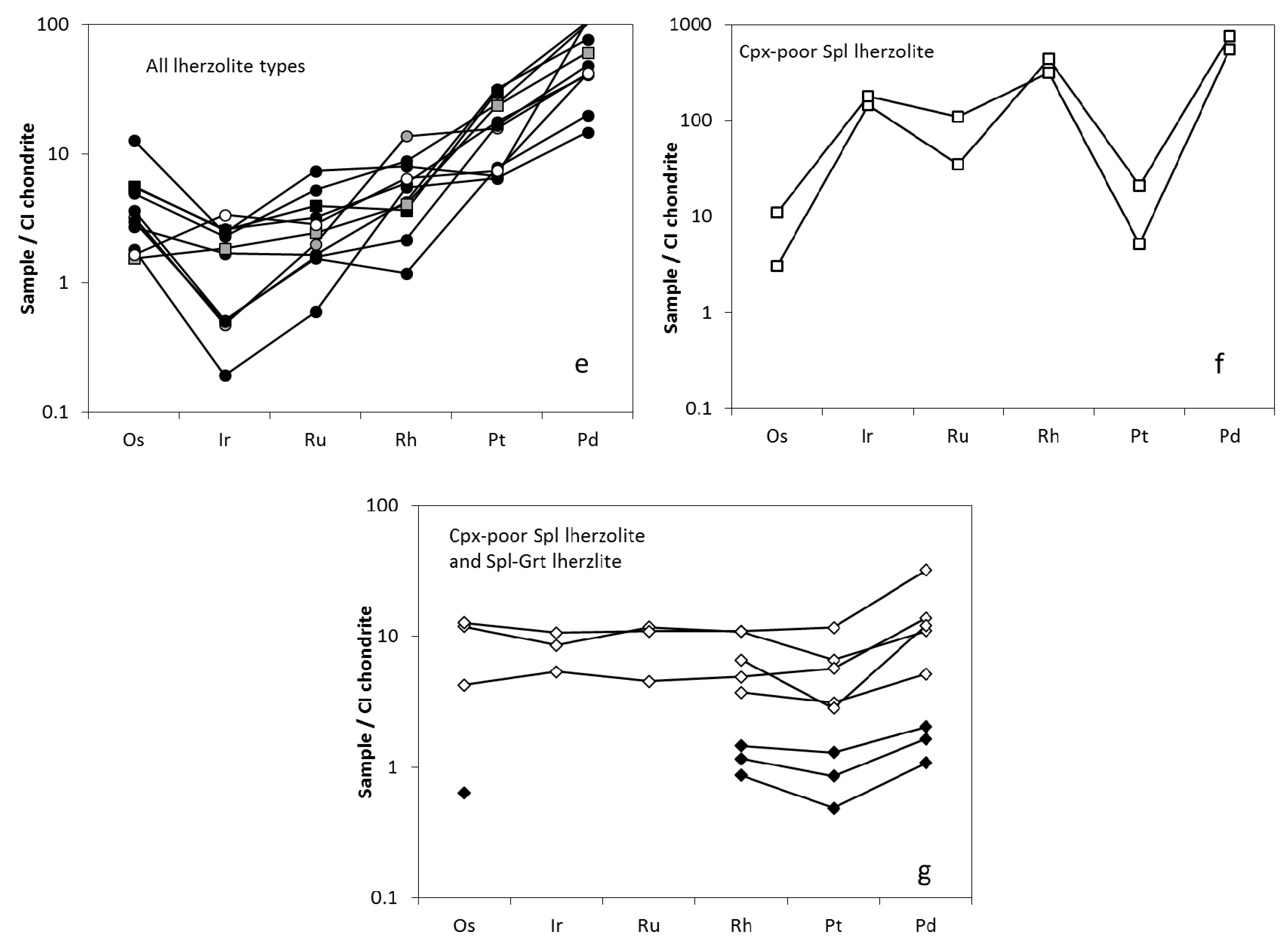
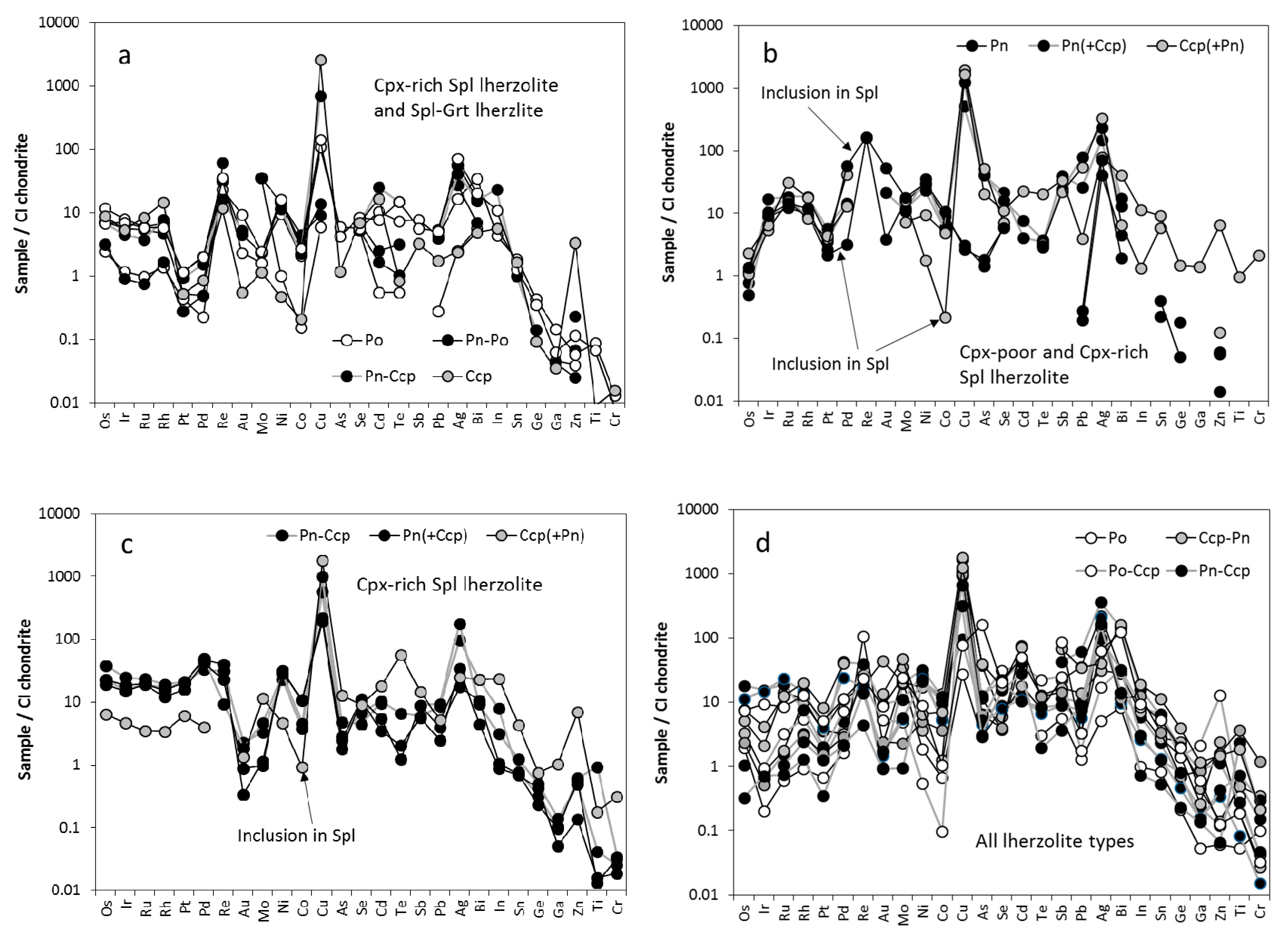
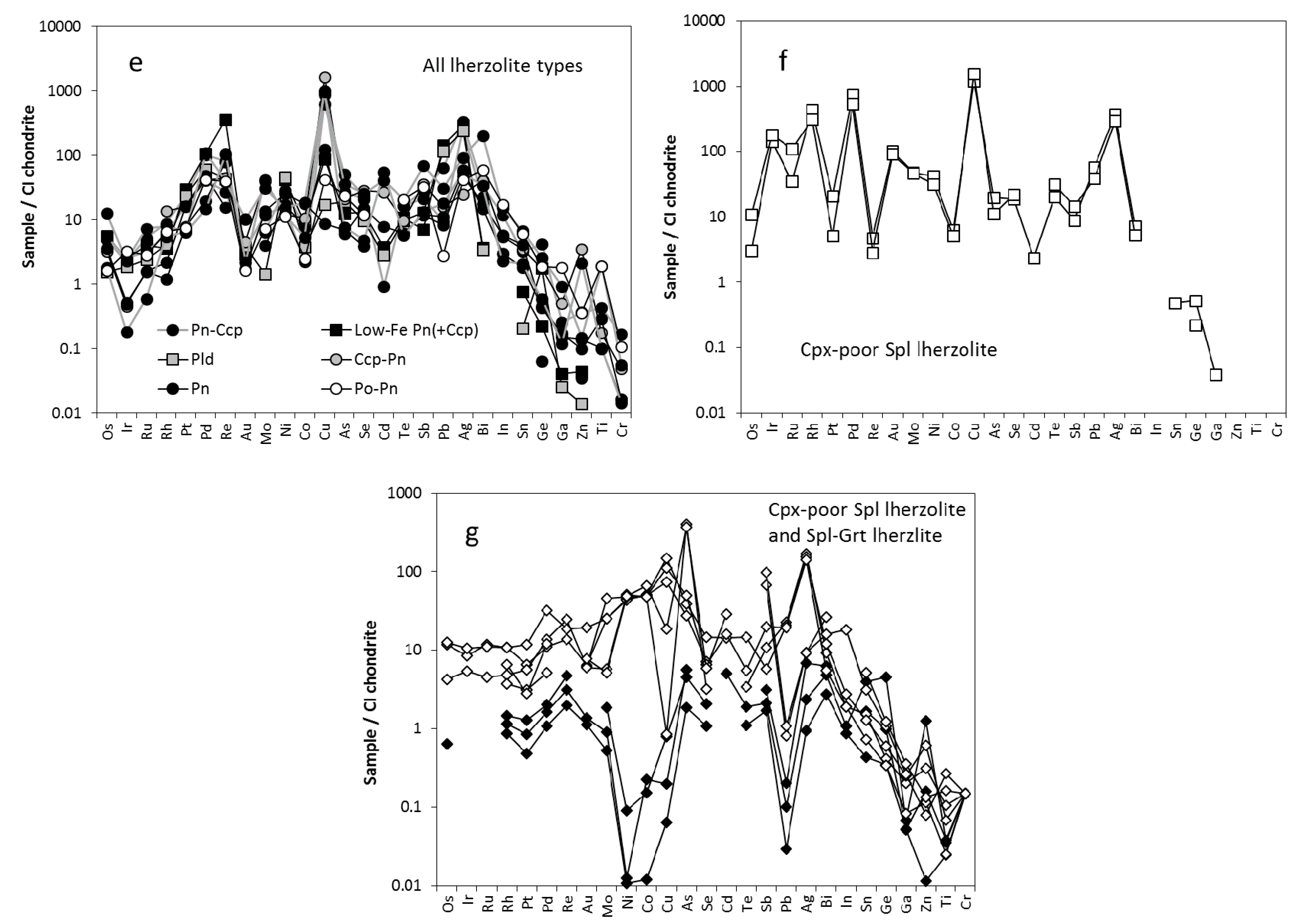
3.2. Trace Element Characteristics of Sulfides
3.2.1. Sulfides in Cpx-Poor Spl Lherzolites
3.2.2. Sulfides in Cpx-Rich Spl Lherzolites
| Rock | Cpx-Poor | Cpx-Rich | |||||||
| Inter | In Spl | In Ol | |||||||
| Sample | U-3/4-2 | U-1/4-3 | UN-1 | XLT-4 | |||||
| Mineral | Pn | Low-Fe Pn | Pld | Cu-Ni ss | Cu-Ni ss | Pn | Pn | Ccp | Pn |
| Ti | 7.08 | nd | nd | nd | nd | nd | nd | 423.7 | 230.2 |
| Cr | 9.04 | nd | nd | nd | nd | nd | nd | 5659 | 401.8 |
| Mn | 4.71 | 3.43 | 3.12 | 1331 | 359.4 | 31.81 | 48.64 | 2122 | 337.7 |
| Co | 2782 | 20.65 | 1880 | 3159 | 2562 | 4506 | 5442.5 | 109.2 | 5989 |
| Ni | 361,633 | 551,976 | 502,298 | 466,104 | 351,212 | 318,362 | 331,828 | 19,670 | 353,381 |
| Cu | 8231 | 23.68 | 21.44 | 152,811 | 194,705 | 387.2 | 335.6 | 212,253 | 11,938 |
| Zn | 7.74 | 4.77 | 4.34 | bdl | bdl | 4.43 | bdl | 2030 | 37.62 |
| Ga | 0.45 | 0.68 | 0.25 | 0.39 | bdl | bdl | bdl | 13.84 | 2.30 |
| Ge | 5.05 | 64.40 | 58.60 | 17.18 | 7.27 | 1.66 | bdl | 48.56 | 22.32 |
| As | 99.14 | 39.38 | 35.84 | 36.65 | 20.79 | 2.64 | 3.36 | 38.35 | 12.07 |
| Se | 511.2 | 197.6 | 179.8 | 352.6 | 413.4 | 112.5 | 108.3 | 204.8 | 167.0 |
| Mo | 1541 | 1.45 | 1.32 | 44.32 | 43.17 | 14.65 | 16.39 | 15.09 | 18.32 |
| Ru | 27.16 | 1.92 | 1.75 | 24.93 | 77.51 | 8.73 | 10.30 | 21.91 | 1.10 |
| Rh | 8.60 | 0.60 | 0.55 | 58.43 | 41.66 | 1.31 | 1.63 | 2.42 | 0.43 |
| Ag | 20.54 | 52.45 | 47.73 | 73.82 | 58.72 | 7.99 | 14.20 | 15.72 | 19.12 |
| Pd | 156.0 | 37.51 | 34.13 | 422.1 | 303.8 | 1.76 | 32.50 | 7.36 | 2.81 |
| Cd | bdl | 2.16 | 1.96 | 1.63 | bdl | bdl | bdl | 15.61 | 8.41 |
| In | 0.06 | bdl | bdl | bdl | bdl | bdl | bdl | 0.92 | 0.47 |
| Sn | 1.08 | 0.39 | 0.36 | 0.84 | bdl | 0.69 | 0.39 | 15.67 | 4.02 |
| Sb | 4.44 | 2.01 | 1.83 | 1.24 | 2.08 | bdl | bdl | 4.85 | 1.65 |
| Te | 86.01 | 24.78 | 22.55 | 46.99 | 74.07 | 6.71 | 7.03 | 47.48 | bdl |
| Re | 0.40 | 1.72 | 1.57 | 0.17 | 0.10 | 5.83 | 6.11 | 1.90 | 0.68 |
| Os | 0.95 | 0.83 | 0.75 | 1.47 | 5.33 | 0.58 | 0.66 | 1.13 | bdl |
| Ir | 0.67 | 0.98 | 0.89 | 68.79 | 86.06 | 4.20 | 4.37 | 3.11 | 0.30 |
| Pt | 10.32 | 25.76 | 23.44 | 20.59 | 5.09 | 2.13 | 2.60 | 4.39 | 1.33 |
| Au | 0.16 | 0.60 | 0.55 | 14.30 | 12.73 | 0.53 | 7.40 | 1.59 | 0.13 |
| Pb | 46.51 | 318.4 | 289.7 | 142.3 | 96.99 | 0.49 | 0.69 | 9.82 | 24.45 |
| Bi | 7.58 | 0.43 | 0.39 | 0.82 | 0.60 | 0.52 | 0.22 | 4.62 | 1.62 |
| Rock | Cpx-rich | Spl-Grt | |||||||
| Inter | |||||||||
| Sample | UN-1 | XLT-4 | DN-1 | ||||||
| Mineral | Pn | Pn | Pn | Po | Ccp | Ccp | Pn | ||
| Ti | 44.43 | 39.07 | 18.16 | nd | 78.03 | 3.87 | 17.61 | ||
| Cr | 43.14 | 76.56 | 157.1 | nd | 71.26 | 42.66 | 109.8 | ||
| Mn | 104.5 | 50.49 | 40.87 | 318.6 | 31.46 | 4.17 | 609.9 | ||
| Co | 8879 | 5077 | 5649 | 76.90 | 4.58 | 106.9 | 3080 | ||
| Ni | 281,295 | 329,743 | 341,584 | 11,073 | bdl | 52.59 | 349,622 | ||
| Cu | 1106 | 136.1 | 104.9 | 763.0 | 370,433 | 324,917 | 8631 | ||
| Zn | 44.32 | 14.31 | 10.36 | 12.31 | 827.1 | 1048 | 333.8 | ||
| Ga | 1.52 | 1.91 | 0.91 | 0.47 | 2.55 | 0.35 | 0.98 | ||
| Ge | 14.29 | 12.95 | 8.04 | bdl | 33.54 | 3.08 | 18.17 | ||
| As | 12.98 | 8.31 | 6.13 | bdl | 26.54 | 2.20 | 6.20 | ||
| Se | 72.27 | 82.92 | 235.2 | 97.91 | 266.4 | 129.9 | 267.6 | ||
| Mo | 12.71 | 30.07 | 15.44 | 33.31 | 7.85 | 1.08 | 2.28 | ||
| Ru | 1.11 | 1.04 | bdl | bdl | bdl | 6.08 | 21.11 | ||
| Rh | 0.16 | bdl | 0.08 | 0.19 | 1.65 | 1.96 | 0.18 | ||
| Ag | 9.63 | 1.55 | 6.92 | 0.51 | 1.88 | 0.47 | 26.98 | ||
| Pd | 11.15 | 2.85 | 7.51 | 0.13 | 2.17 | 0.48 | 48.02 | ||
| Cd | bdl | bdl | 2.64 | 0.39 | 84.05 | 11.35 | bdl | ||
| In | 0.46 | 0.15 | bdl | bdl | 0.36 | 0.45 | 0.16 | ||
| Sn | 6.96 | 2.53 | 2.09 | bdl | 4.48 | 2.85 | 2.15 | ||
| Sb | 1.71 | 1.62 | 0.70 | bdl | 3.22 | 0.47 | 7.56 | ||
| Te | bdl | bdl | 4.07 | 1.29 | 175.7 | 1.94 | 8.66 | ||
| Re | 2.85 | 0.38 | 6.79 | 1.20 | 0.50 | 0.44 | 0.10 | ||
| Os | 1.77 | 0.42 | bdl | 1.19 | 1.34 | 4.30 | 13.30 | ||
| Ir | 0.25 | 0.21 | 0.10 | 0.57 | bdl | 2.62 | 1.86 | ||
| Pt | 7.72 | 4.34 | 1.30 | 0.43 | 3.62 | 0.52 | 2.45 | ||
| Au | 0.24 | bdl | 0.09 | bdl | 0.70 | 0.08 | bdl | ||
| Pb | 20.48 | 9.04 | 27.24 | 0.70 | 4.87 | 4.38 | 36.19 | ||
| Bi | 2.03 | 1.22 | 2.85 | 0.64 | 2.89 | 0.56 | 1.65 | ||
3.2.3. Sulfides in Spl-Grt Lherzolites
| Rock | Cpx-poor | Spl-Grt | ||||||
|---|---|---|---|---|---|---|---|---|
| Skeletal | Faceted | Massive | Skeletal | |||||
| Sample | 32601-9b | DN-4 | ||||||
| Mineral | Co-Vo | Co-Vo | Co-Vo | Py | Py | Py | Co-Vo | Co-Vo |
| Ti | 46.40 | 29.47 | 11.03 | 15.34 | 16.56 | 10.66 | 117.3 | 71.59 |
| Cr | 889.8 | 62.98 | 149.8 | 397.3 | 97.08 | 7.23 | 735.4 | 371.1 |
| Mn | 168.1 | 30.13 | 23.41 | 4634 | 1529 | 3.70 | 24.59 | 36.91 |
| Co | 24,794 | 25,662 | 24,616 | 113.2 | 76.95 | 0.60 | 24,106 | 32,944 |
| Ni | 487,469 | 480,393 | 521,611 | 137.8 | 1002 | 55.87 | 553,276 | 540,826 |
| Cu | 9332 | 18,647 | 13,891 | 24.84 | 101.1 | 8.03 | 106.2 | 2355 |
| Zn | 97.58 | 189.5 | 35.85 | 395.6 | 49.89 | 3.54 | 24.71 | 42.19 |
| Ga | 2.01 | 2.54 | 0.84 | 0.68 | 0.52 | 0.53 | 2.65 | 3.59 |
| Ge | 13.70 | 19.49 | 11.15 | 148.9 | 32.35 | 11.52 | 35.13 | 40.04 |
| As | 51.71 | 72.89 | 92.34 | 10.57 | 8.40 | 3.46 | 739.2 | 689.6 |
| Se | 134.0 | 273.1 | 122.0 | bdl | 38.79 | 20.04 | 60.21 | 109.0 |
| Mo | 23.38 | 23.59 | 41.87 | 0.85 | 1.73 | 0.49 | 5.36 | 4.85 |
| Ru | 8.40 | 7.81 | 3.22 | bdl | bdl | bdl | bdl | bdl |
| Rh | 1.46 | 1.47 | 0.66 | 0.16 | 0.19 | 0.12 | 0.50 | 0.88 |
| Ag | 33.36 | 30.70 | 27.95 | 0.47 | 1.36 | 0.19 | 1.84 | 1.85 |
| Pd | 6.19 | 17.93 | 7.73 | 0.92 | 1.15 | 0.60 | 2.90 | 6.80 |
| Cd | bdl | 9.88 | 11.12 | 3.46 | bdl | bdl | bdl | 19.75 |
| In | 0.15 | 0.22 | 0.15 | 0.09 | bdl | 0.07 | 1.46 | bdl |
| Sn | 2.62 | 2.19 | 1.25 | 6.79 | 2.85 | 0.74 | 5.36 | 8.90 |
| Sb | 1.55 | 0.82 | 2.81 | 0.30 | 0.45 | 0.24 | 9.48 | 13.72 |
| Te | 7.94 | 33.94 | 12.74 | 4.46 | bdl | 2.56 | bdl | bdl |
| Re | 0.50 | 0.68 | 0.90 | 0.11 | 0.17 | 0.07 | bdl | bdl |
| Os | 5.78 | 6.18 | 2.07 | 0.31 | bdl | bdl | bdl | bdl |
| Ir | 4.13 | 5.15 | 2.58 | bdl | bdl | bdl | 0.48 | 0.61 |
| Pt | 6.53 | 11.60 | 5.65 | 0.85 | 1.28 | 0.48 | 3.08 | 2.79 |
| Au | 0.90 | 2.71 | 0.86 | 0.19 | bdl | 0.16 | 0.85 | 1.11 |
| Pb | 56.59 | 51.24 | 48.62 | 0.25 | 0.51 | 0.07 | 2.03 | 2.68 |
| Bi | 1.06 | 1.38 | 0.63 | 0.55 | 0.71 | 0.31 | 1.81 | 3.03 |
4. Discussion
4.1. Sulfides from Lherzolite Matrix
4.2. Sulfides from Silicate-Carbonate Patches
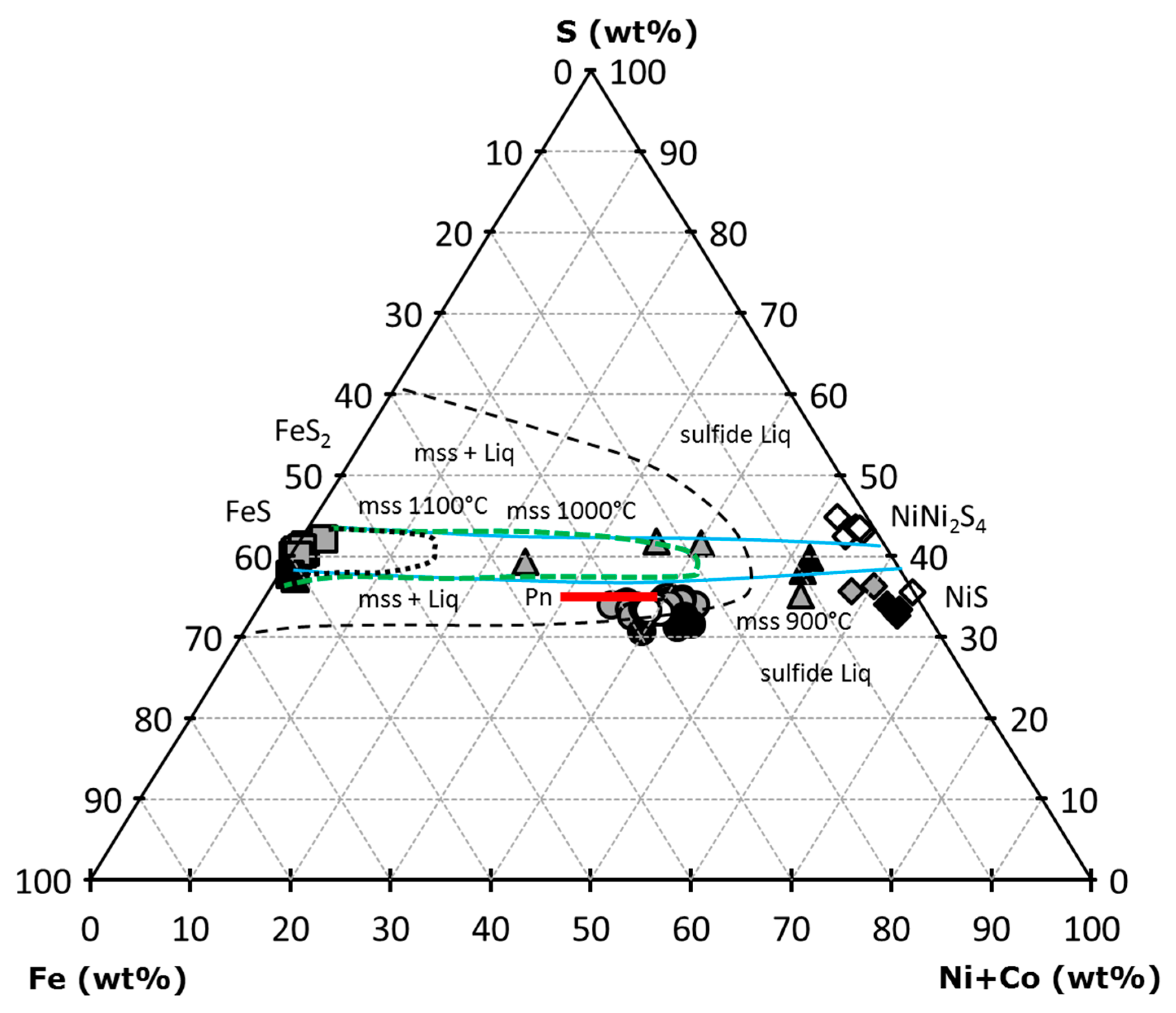
4.3. Sulfides from Metasomatic Veinlets
4.4. Trace Elements in Lherzolite Sulfides
4.4.1. Platinum Group Elements
4.4.2. Other Trace Elements
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Laiba, A.A.; Andronikov, A.V.; Egorov, L.S.; Fedorov, L.V. Stock-like and dyke bodies of alakaline-ultramafic composition in Jetty Peninsula (Prince Charles Mountains, East Antarctica). In Geological and Geophysical Investigations in Antarctica; Ivanov, V.L., Grikurov, G.E., Eds.; PGO “Sevmorgeologia”: Leningrad, Russia, 1987; pp. 35–47. (In Russian) [Google Scholar]
- Andronikov, A.V. Spinel-garnet lherzolite nodules from alkaline-ultrabasic rocks of Jetty Peninsula (East Antarctica). Antarctic Sci. 1990, 2, 321–330. [Google Scholar] [CrossRef]
- Andronikov, A.V. The first study of upper mantle inclusions from the Prince Charles Mountains, East Antarctica. In Recent Progress in Antarctic Earth Science; Yoshida, Y., Kaminuma, K., Shiraishi, K., Eds.; Terrapub: Tokyo, Japan, 1992; pp. 163–171. [Google Scholar]
- Foley, S.F.; Andronikov, A.V.; Jacob, D.E.; Melzer, S. Evidence from Antarctic mantle peridotite xenoliths for changes in mineralogy, geochemistry and geothermal gradients beneath a developing rift. Geochim. Cosmochim. Acta 2006, 70, 3096–3120. [Google Scholar] [CrossRef]
- Foley, S.F.; Andronikov, A.V.; Halpin, J.A.; Daczko, N.R.; Jacob, D.E. Mantle rocks in East Antarctica. Geol. Soc. London Mem. 2021, 56. [Google Scholar] [CrossRef]
- Mikhalsky, E.; Andronikov, A.; Leitchenkov, G.; Belyatsky, B. The age of the Earth‘s crust in the Northern Prince Charles Mountains (East Antarctica) as evidenced by zircon xenocrysts from Cretaceous alkaline-ultramafic rocks. Lithos 2020, 368–369. [Google Scholar] [CrossRef]
- Andronikov, A.V.; Beliatsky, B.V. Implication of Sm-Nd isotopic systematics to the events recorded in the mantle-derived xenoliths from the Jetty Peninsula, East Antarctica. Terra Antarct. 1995, 2, 103–110. [Google Scholar]
- Andronikov, A.V. Varying glass compositions in deep-seated inclusions from alkaline-ultamafic rocks of Jetty Peninsula (East Antarctica): Evidence for mantle metasomatism. In The Antarctic Region: Geological Evolution and Processes; Ricci, C.A., Ed.; Terra Antartica Publication: Siena, Italy, 1997; pp. 901–910. [Google Scholar]
- Kogarko, L.N.; Kurat, G.; Ntaflos, T. Henrymeyerite in the metasomatized upper mantle of Eastern Antarctica. Canad. Mineral. 2007, 45, 497–501. [Google Scholar] [CrossRef]
- Buikin, A.I.; Solovova, I.P.; Verchovsky, A.B.; Kogarko, L.N.; Averin, A.A. PVT parameters of fluid inclusions and the C, O, N, and Ar isotopic composition in a garnet lherzolite xenolith from the Oasis Jetty, East Antarctica. Geochem. Intl. 2014, 52, 805–821. [Google Scholar] [CrossRef]
- Solovova, I.P.; Kogarko, L.N.; Averin, A.A. Conditions of sulphide formation in the metasomatized mantle beneath East Antarctica. Petrology 2015, 23, 519–542. [Google Scholar] [CrossRef]
- Beliatsky, B.V.; Andronikov, A.V. Deep-seated lherzolite inclusions from alkaline-ultramafic rocks in Jetty Peninsula: The mineral and chemical composition, P-T conditions and Sr-Nd isotope systematic. In Scientific Results of Russian Geological and Geophysical Researches in Antarctica 2; Mikhalsky, E.V., Laiba, A.A., Eds.; VNIIOkeangeologia: St. Petersburg, Russia, 2009; pp. 89–109, (In Russian with English afterword). [Google Scholar]
- Shindo, K.; Komuro, K.; Hayashi, K. Sulfide minerals in mantle xenoliths from the Kurose reef, Fukuoka Prefecture, Japan. J. Mineral. Petrol. Sci. 2009, 104, 182–187. [Google Scholar] [CrossRef][Green Version]
- Vaughan, D.J.; Corkhill, C.L. Mineralogy of sulfides. Elements 2017, 13, 81–87. [Google Scholar] [CrossRef]
- Kiseeva, E.S.; Wood, B.J. The effects of composition and temperature on chalcophile and lithophile element partitioning into magmatic sulphides. Earth Planet. Sci. Lett. 2015, 424, 280–294. [Google Scholar] [CrossRef]
- Kiseeva, E.S.; Fonseca, R.O.C.; Smythe, D.J. Chalcophile elements and sulfides in the upper mantle. Elements 2017, 13, 111–116. [Google Scholar] [CrossRef]
- Alard, O.; Griffin, W.L.; Lorand, J.-P.; Jackson, S.E.; O’Reilly, S.Y. Non-chondritic distribution of the highly siderophile elements in mantle sulfides. Nature 2000, 407, 891–894. [Google Scholar] [CrossRef] [PubMed]
- Griffin, W.L.; Graham, S.; O’Reilly, S.Y.; Pearson, N.J. Lithosphere Evolution beneath the Kaapvaal Craton: Re-Os systematics of sulfides in mantle-derived peridotites. Chem. Geol. 2004, 208, 89–118. [Google Scholar] [CrossRef]
- Dromgoole, E.L.; Pasteris, J.D. Interpretation of the sulfide assemblages in a suite of xenoliths from Kilbourne Hole, New Mexico. Geol. Soc. Amer. Spec. Paper 1987, 215, 25–46. [Google Scholar]
- Szabo, C.; Bodnar, R.J. Chemistry and origin of mantle sulfides in spinel peridotite xenoliths from alkaline basaltic lavas, Nógrád Gömör Volcanic Field, northern Hungary and southern Slovakia. Geochim. Cosmochim. Acata 1995, 59, 3917–3927. [Google Scholar] [CrossRef]
- Guo, J.; Griffin, W.L.; O’Reilly, S.Y. Geochemistry and origin of sulphide minerals in mantle xenoliths: Qilin, Southeastern China. J. Petrol. 1999, 40, 1125–1149. [Google Scholar] [CrossRef]
- Lorand, J.P.; Gregoire, M. Petrogenesis of base metal sulfide assemblages of some peridotites from the Kaapvaal Craton (South Africa). Contrib. Mineral. Petrol. 2006, 151, 521–538. [Google Scholar] [CrossRef]
- O’Reilly, S.Y.; Griffin, W.L. Imaging global chemical and thermal heterogeneity in the subcontinental lithospheric mantle with garnets and xenoliths: Geophysical implications. Tectonophysics 2006, 416, 289–309. [Google Scholar] [CrossRef]
- Sen, I.S.; Bizimis, M.; Sen, G. Geochemistry of sulfides in Hawaiian garnet pyroxenite xenoliths: Implications for highly siderophile elements in the oceanic mantle. Chem. Geol. 2010, 273, 180–192. [Google Scholar] [CrossRef]
- Delpech, G.; Lorand, J.-P.; Gregoire, M.; Cottin, J.-Y.; O’Reilly, S.Y. In-situ geochemistry of sulfides in highly metasomatized mantle xenoliths from Kerguelen, southern Indian Ocean. Lithos 2012, 154, 296–314. [Google Scholar] [CrossRef]
- Lorand, J.P.; Luguet, A.; Alard, O. Platinum-group element systematics and petrogenetic processes of the continental mantle: A review. Lithos 2013, 164, 2–21. [Google Scholar] [CrossRef]
- Westner, K.J.; Beier, C.; Klemd, R.; Osbahr, I.; Brooks, N. In situ chalcophile and siderophile element behavior in sulfides from Moroccan Middle Atlas spinel peridotite xenoliths during metasomatism and weathering. Minerals 2019, 9, 276. [Google Scholar] [CrossRef]
- Andronikov, A.V.; Egorov, L.S. Mesozoic alkaline-ultramafic magmatism of Jetty Peninsula. In Gondwana Eight: Assembly, Evolution and Dispersal; Findlay, R.H., Unrug, R., Banks, M.R., Veevers, J.J., Eds.; Balkema; Brookfield: Rotterdam, The Netherlands, 1993; pp. 547–557. [Google Scholar]
- Foley, S.F.; Andronikov, A.V.; Melzer, S. Petrology of ultramafic lamprophyres from the Beaver Lake area of Eastern Antarctica and their relation to the breakup of the Gondwanaland. Mineral. Petrol. 2002, 74, 361–384. [Google Scholar] [CrossRef]
- Andronikov, A.V.; Sheraton, J.W. The redox state of the lithospheric upper mantle beneath the East Antarctic Shield. Terra Antarct. 1996, 3, 39–48. [Google Scholar]
- van Achterbergh, E.; Ryan, C.G.; Jackson, S.E.; Griffin, W.L. Data reduction software for LA-ICP-MS. In Laser-Ablation-ICPMS in the Earth Sciences: Principles and Applications; Sylvester, P., Ed.; Mineral. Assoc. Canada Short Course Series; Mineralogical Association of Canada: Quebec City, QC, Canada, 2001; Volume 29, pp. 239–243. [Google Scholar]
- Griffin, W.L.; Powell, W.J.; Pearson, N.J.; O’Reilly, S.Y. GLITTER: Data reduction software for laser ablation ICP-MS. In Laser Ablation ICP–MS in the Earth Sciences: Current Practices and Outstanding Issues; Sylvester, P., Ed.; Mineralogical Association of Canada Short Course Series; Mineralogical Association of Canada: Quebec City, QC, Canada, 2008; Volume 40, pp. 308–311. [Google Scholar]
- Wilson, S.A.; Koenig, A.E.; Ridley, W.I. Development of sulfide calibration standards for the laser ablation inductively-coupled plasma mass spectrometry. J. Anal. Atom. Spectrom. 2002, 17, 406–409. [Google Scholar] [CrossRef]
- Savard, D.; Bouchard-Boivin, B.; Barnes, S.-J.; Garbe-Schönberg, D. UQAC-FeS: A new series of base metal sulfide quality control reference material for LA-ICP-MS analysis. In Proceedings of the 10th International Conference on the Analysis of Geological and Environmental Materials, Sydney, Australia, 8–13 July 2018. [Google Scholar]
- Duran, C.J.; Dube-Loubert, H.; Page, P.; Barnes, S.J.; Roy, M.; Savard, D.; Cave, B.J.; Arguin, J.-P.; Mansur, E.T. Applications of trace element chemistry of pyrite and chalcopyrite in glacial sediments to mineral exploration targeting: Example from the Churchill Province, northern Quebec, Canada. J. Geochem. Explor. 2019, 196, 105–130. [Google Scholar] [CrossRef]
- Mathieu, L.; Racicot, D. Petrogenetic Study of the Multiphase Chibougamau Pluton: Archaean Magmas Associated with Cu–Au Magmato-Hydrothermal Systems. Minerals 2019, 9, 174. [Google Scholar] [CrossRef]
- Ballhaus, C.; Sylvester, P. 2000. Noble metal enrichment processes in the Merensky Reef, Bushveld Complex. J. Petrol. 2000, 41, 546–561. [Google Scholar] [CrossRef]
- Cook, N.; Ciobanu, C.L.; George, L.; Zhu, Z.-Y.; Wade, B.; Ehrig, K. Trace Element Analysis of Minerals in Magmatic-Hydrothermal Ores by Laser Ablation Inductively-Coupled Plasma Mass Spectrometry: Approaches and Opportunities. Minerals 2016, 6, 111. [Google Scholar] [CrossRef]
- Pettke, T. Analytical protocol for element concentration and isotope ratio measurement in fluid inclusions by LA–(MC–)ICP–MS. In Laser Ablation–ICP–MS in the Earth Sciences; Sylvester, P., Ed.; Mineralogical Association of Canada Short Course; Mineralogical Association of Canada: Quebec City, QC, Canada, 2008; Volume 40, pp. 189–217. [Google Scholar]
- Hughes, H.S.R.; McDonald, I.; Faithfull, J.W.; Upton, B.G.J.; Loocke, M. Cobalt and precious metals in sulphides of peridotite xenoliths and inferences concerning their ditribution accordying to geodynamic environment: A case study from the Scottish lithospheric mantle. Lithos 2016, 240–243, 202–227. [Google Scholar] [CrossRef]
- Anders, E.; Grevesse, N. Abundances of the elements: Meteoritic and solar. Geochim. Cosmochim. Acta 1989, 53, 197–214. [Google Scholar] [CrossRef]
- Mitchell, R.H.; Keays, R.R. Abundance and distribution of gold, palladium and iridium in some spinel and garnet lherzolites: Implications for the nature and origin of precious metal-rich intergranular components in the upper mantle. Geochim. Cosmochim. Acta 1981, 45, 2425–2445. [Google Scholar] [CrossRef]
- Ebel, D.S.; Naldrett, A.J. Fractional crystallization of sulfide ore liquids at high temperature. Econom. Geol. 1996, 91, 607–621. [Google Scholar] [CrossRef]
- Ebel, D.S.; Naldrett, A.J. Crystallization of sulfide liquids and interpretation of ore composition. Canad. J. Earth Sci. 1997, 34, 352–365. [Google Scholar] [CrossRef]
- Mungall, J.A. Crystallization of magmatic sulfides: An empirical model and application to Sudbury ores. Geochim. Cosmochim. Acta 2007, 71, 2809–2819. [Google Scholar] [CrossRef]
- Nkoumbou, C.; Villieras, F.; Barbey, P.; Ngoune, C.-Y.; Joussemet, R.; Diot, F.; Njopwouo, D.; Yvon, J. Ni-Co sulphide segregation in the Mamb pyroxenite intrusion, Cameroon. Comptes Rendus Geosci. 2009, 341, 517–525. [Google Scholar] [CrossRef]
- Kitakaze, A.; Komatsu, R. Phase relations of some sulfide systems-(5). Especially Fe-Ni-S system. In Memoirs of the Faculty of Engenieering, Yamaguchi University; Yamaguchi University: Yamaguchi, Japan, 2020; Volume 70, pp. 1–22. [Google Scholar]
- Kullerud, G.; Yund, R.A.; Moh, G. Phase relation in the Cu-Fe-S and Cu-Ni-S systems. Econ. Geol.Monogr. 1969, 4, 323–343. [Google Scholar]
- Kelly, D.P.; Vaughan, D.J. Pyrrhotine-pentlandite ore textures: A mechanistic approach. Mineral. Mag. 1983, 47, 453–463. [Google Scholar] [CrossRef]
- Wang, H.; Pring, A.; Ngothai, Y.; O’Neil, B. A low-temperature kinetic study of the exsolution of pentlandite from the monosulfide solid solution using a refined Avrami method. Geochim. Cosmochim. Acta 2005, 69, 415–425. [Google Scholar] [CrossRef]
- Bishop, F.C.; Smith, J.V.; Dawson, J.B. Pentlandite-magnetite intergrowth in the De Beers spinel lherzolite: Review of sulfide in nodules. Phys. Chem. Earth 1975, 9, 323–337. [Google Scholar] [CrossRef]
- Raghavan, V. Phase diagram evaluation: Section II. Fe-Ni-S (Iron-Nickol-Sulfur). J. Phase Equilibria Diffus. 2004, 25, 373–381. [Google Scholar] [CrossRef]
- Krymsky, R.S.; Antonov, A.V.; Belyatsky, B.V.; Sushchevskaya, N.M.; Sergeev, S.A. The age and the evolution of the lithospheric mantle in the East Antarctic Craton: Osmium isotope composition and the distribution of the platinum group elements in spinel lherzolite nodules. Doklady Earth Sci. 2019, 485, 444–449. [Google Scholar] [CrossRef]
- Zaccarini, F.; Pushkarev, E.; Garuti, G.; Bindi, L. Multi-Analytical Characterization of Minerals of the Bowieite–Kashinite Series from the Svetly Bor Complex, Urals, Russia, and Comparison with Worldwide Occurrences. Canad. Mineral. 2016, 54, 461–473. [Google Scholar] [CrossRef]
- Kiseeva, E.S.; Wood, B.J. A simple model for chalcopyrite element partitioning between sulphide and silicate liquids with geochemical applications. Earth Planet. Sci. Lett. 2013, 383, 68–81. [Google Scholar] [CrossRef]
- Brenan, J.M. Re-Os fractionation by sulfide melt-silicate melt partitioning: A new spin. Chem. Geol. 2008, 248, 140–165. [Google Scholar] [CrossRef]
- Brenan, J.M. Se-Te fractionation by sulfide-silicate melt partitioning: Implications for the composition of mantle-derived magmas and their melting residues. Earth Planet. Sci. Lett. 2015, 299, 328–338. [Google Scholar] [CrossRef]
- Li, Y.; Audetat, A. Partitioning of V, Mn, Co, Ni, Cu, Zn, As, Mo, Ag, Sn, Sb, W, Au, Pb and Bi between sulfide phases and hydrous basanite melt at upper mantle conditions. Earth Planet. Sci. Lett. 2012, 355–356, 327–340. [Google Scholar] [CrossRef]
- Greaney, A.T.; Rudnick, R.L.; Helz, R.T.; Gaschnig, R.M.; Piccoli, P.M.; Ash, R.D. The behavior of chalcophile elements during magmatic differentiation as observed in Kilauea Iki lava lake, Hawaii. Geochim. Cosmochim. Acta 2017, 210, 71–96. [Google Scholar] [CrossRef]
- Steenstra, E.S.; Trautner, V.T.; Berndt, J.; Klemme, S.; van Westrenen, W. Trace element partitioning between sulfide-, metal-, and silicate melts at highly reduced conditions: Insights into the distribution of volatile elements during core formation in reduced bodies. Icarus 2020, 335, 113408. [Google Scholar] [CrossRef]
- Ballhaus, C.; Tredoux, M.; Späth, A. Phase relations in the Fe–Ni–Cu–PGE–S system at magmatic temperature and application to massive sulphide ores of the Sudbury igneous complex. J. Petrol. 2001, 42, 1911–1926. [Google Scholar] [CrossRef]
- Lorand, J.P.; Luguet, A.; Alard, O. Platinum-group elements: A new set of key tracers for the earth’s interior. Elements 2008, 4, 247–252. [Google Scholar] [CrossRef]


| Inter | In Ol | |||||||||||
| Sample | U-3/4-2 | U-1/3-4 | ||||||||||
| Mineral | Pn | Pn | Ccp | Ccp | Pn | Pn | Pn | Ccp | Ccp | Pld | Pld | |
| Zn | 0.08 | 0.02 | 0.01 | |||||||||
| Cu | 0.44 | 0.12 | 30.64 | 30.29 | 0.29 | 0.62 | 0.18 | 31.47 | 32.51 | 0.56 | 0.09 | |
| Ni | 37.98 | 36.48 | 0.38 | 0.09 | 42.97 | 43.18 | 43.36 | 2.36 | 0.76 | 49.76 | 52.20 | |
| Co | 0.38 | 0.01 | 0.01 | 0.39 | 0.40 | 0.32 | 0.02 | 0.08 | 0.79 | 0.54 | ||
| Fe | 25.56 | 25.76 | 28.31 | 29.62 | 22.36 | 22.30 | 21.95 | 28.77 | 29.71 | 6.45 | 3.04 | |
| S | 32.34 | 31.00 | 33.70 | 34.29 | 32.65 | 32.95 | 32.67 | 33.97 | 34.32 | 39.23 | 41.01 | |
| Total | 96.32 | 93.82 | 93.04 | 94.33 | 98.67 | 99.45 | 98.48 | 96.60 | 97.38 | 96.79 | 96.88 | |
| Inter | ||||||||||||
| Sample | 32601-9b | U-1/3-4 | ||||||||||
| Mineral | Ccp | Ccp | Pld | Pld | Pld | Pn | Pn | Pld | Ccp | Cu-Ni ss | Cu-Ni ss | Cu-Ni ss |
| Zn | 0.05 | 0.13 | 0.08 | 0.16 | 0.07 | |||||||
| Cu | 33.09 | 30.79 | 0.50 | 0.62 | 0.38 | 0.18 | 0.23 | 0.15 | 31.75 | 13.09 | 22.04 | 7.58 |
| Ni | 0.47 | 1.21 | 54.18 | 53.25 | 53.78 | 37.77 | 37.80 | 49.17 | 1.06 | 44.93 | 38.11 | 48.03 |
| Co | 0.05 | 0.19 | 1.25 | 1.92 | 1.69 | 0.26 | 0.46 | 0.39 | 0.01 | 0.54 | 0.18 | 0.89 |
| Fe | 30.05 | 27.52 | 1.20 | 1.71 | 1.31 | 26.26 | 26.48 | 5.62 | 29.55 | 1.06 | 1.18 | 0.95 |
| S | 34.92 | 34.06 | 41.91 | 42.49 | 42.35 | 32.47 | 32.56 | 40.70 | 34.50 | 37.80 | 36.29 | 39.37 |
| Total | 98.63 | 93.90 | 99.04 | 99.99 | 99.51 | 96.94 | 97.61 | 96.03 | 97.03 | 97.49 | 97.80 | 96.82 |
| In Ol | In Spl | ||||||||||
| Sample | UN-1 | ||||||||||
| Mineral | Po | Po | Pn | Pn | Ccp | Po | Pn | Pn | Ccp | Ccp | mss |
| Zn | 0.09 | 0.31 | 0.01 | 0.14 | |||||||
| Cu | 0.16 | 0.06 | 0.13 | 0.08 | 32.47 | 0.09 | 0.21 | 33.30 | 33.52 | 2.92 | |
| Ni | 0.94 | 1.14 | 35.18 | 33.78 | 0.52 | 2.27 | 37.52 | 35.42 | 0.36 | 0.27 | 20.28 |
| Co | 0.07 | 0.45 | 0.09 | 0.62 | 0.66 | 0.08 | 0.04 | 0.22 | |||
| Fe | 54.54 | 54.69 | 27.78 | 27.68 | 28.18 | 51.86 | 25.74 | 27.69 | 28.61 | 28.57 | 31.41 |
| S | 38.96 | 37.51 | 31.48 | 31.70 | 32.28 | 39.28 | 31.07 | 30.47 | 32.82 | 32.81 | 33.44 |
| Total | 94.60 | 93.56 | 95.02 | 93.24 | 93.85 | 93.50 | 95.16 | 94.24 | 95.18 | 95.35 | 88.27 |
| Inter | |||||||||||
| Sample | UN-1 | XLT-4 | |||||||||
| Mineral | Po | Po | Pn | Pn | Ccp | Ccp | Pn | Pn | Ccp | Ccp | Mlr? |
| Zn | 0.07 | ||||||||||
| Cu | 0.15 | 0.04 | 31.98 | 32.38 | 0.82 | 0.53 | 34.12 | 33.14 | 0.39 | ||
| Ni | 0.54 | 0.40 | 37.55 | 38.36 | 0.33 | 0.45 | 41.62 | 40.89 | 0.47 | 0.64 | 52.22 |
| Co | 0.15 | 0.11 | 0.63 | 0.55 | 0.01 | 0.55 | |||||
| Fe | 57.48 | 56.68 | 26.73 | 26.36 | 29.28 | 29.11 | 21.44 | 24.62 | 29.87 | 30.16 | 11.19 |
| S | 39.33 | 39.10 | 32.48 | 32.84 | 34.76 | 34.02 | 32.35 | 33.55 | 34.80 | 35.09 | 34.38 |
| Total | 97.65 | 96.33 | 97.39 | 98.11 | 96.42 | 95.97 | 96.78 | 99.59 | 99.26 | 99.03 | 98.18 |
| In Ol | |||||||||||
| Sample | XLT-4 | ||||||||||
| Mineral | Pn | Pn | Ccp | Ccp | mss | mss | Mlr | Mlr | |||
| Zn | |||||||||||
| Cu | 0.38 | 0.37 | 34.36 | 33.25 | 1.14 | 1.44 | 0.46 | 0.06 | |||
| Ni | 39.72 | 39.35 | 0.11 | 0.51 | 34.16 | 30.47 | 57.17 | 58.60 | |||
| Co | 0.63 | 0.63 | |||||||||
| Fe | 24.23 | 24.85 | 29.17 | 28.82 | 15.48 | 19.19 | 6.00 | 3.54 | |||
| S | 33.56 | 33.66 | 34.23 | 34.22 | 35.19 | 35.64 | 34.95 | 35.30 | |||
| Total | 97.89 | 98.23 | 97.87 | 96.80 | 86.60 | 87.37 | 98.58 | 97.50 | |||
| Inter | In Ol | |||||||||
| Sample | DK-8/3 | DN-4 | ||||||||
| Mineral | Pn | Ccp | Ccp | Ccp | Fe-Mlr | Fe-Mlr | Fe-Mlr | Pn | Ccp | Pld |
| Zn | 0.12 | 0.03 | 0.01 | |||||||
| Cu | 1.14 | 33.32 | 33.31 | 33.25 | 0.63 | 0.68 | 0.34 | 0.20 | 32.83 | |
| Ni | 39.85 | 0.33 | 0.45 | 0.84 | 60.55 | 62.00 | 62.61 | 42.08 | 0.28 | 50.51 |
| Co | 0.53 | 0.08 | 0.07 | 0.80 | 0.52 | 0.25 | 0.37 | 0.42 | ||
| Fe | 23.12 | 30.28 | 30.58 | 30.99 | 3.31 | 2.39 | 2.95 | 23.77 | 29.40 | 7.99 |
| S | 32.93 | 32.75 | 31.69 | 31.91 | 32.87 | 32.21 | 31.75 | 31.65 | 33.79 | 38.60 |
| Total | 97.57 | 96.80 | 96.14 | 97.06 | 98.16 | 97.80 | 97.90 | 98.07 | 96.31 | 97.52 |
| In kelyphite | ||||||||||
| Sample | DN-1 | |||||||||
| Mineral | Po | Po | Pn | Pn | Ccp | Ccp | ||||
| Zn | 0.05 | 0.04 | ||||||||
| Cu | 0.06 | 0.06 | 33.62 | 33.29 | ||||||
| Ni | 1.89 | 1.18 | 41.18 | 38.24 | 0.60 | 0.50 | ||||
| Co | 0.03 | 0.05 | 0.35 | 0.26 | 0.04 | 0.13 | ||||
| Fe | 58.58 | 58.41 | 24.46 | 28.26 | 31.50 | 29.94 | ||||
| S | 35.73 | 36.06 | 29.69 | 29.27 | 32.15 | 31.69 | ||||
| Total | 96.29 | 95.70 | 95.73 | 96.09 | 97.95 | 95.55 | ||||
| Rock | Cpx-poor | Spl-Grt | ||||||||
| Type | Skeletal | |||||||||
| Loation | Veinlet | |||||||||
| Sample | 32601-9b | DN-4 | ||||||||
| Mineral | Co-Mlr | Sgn | Sgn | Co-Vo | Co-Vo | Co-Vo | Co-Mlr | Co-Mlr | Co-Mlr | Co-Mlr |
| Zn | 0.04 | |||||||||
| Cu | 0.08 | 0.01 | ||||||||
| Ni | 60.05 | 44.14 | 36.68 | 53.29 | 53.24 | 45.08 | 60.38 | 59.21 | 58.93 | 61.79 |
| Co | 2.47 | 11.36 | 17.09 | 2.15 | 1.75 | 8.61 | 1.85 | 2.04 | 3.16 | 0.96 |
| Fe | 0.04 | 0.31 | 0.98 | 1.09 | 1.10 | 2.74 | 1.23 | 1.32 | 1.43 | 1.13 |
| S | 33.13 | 42.04 | 41.19 | 40.58 | 40.72 | 40.77 | 34.07 | 33.99 | 33.73 | 34.01 |
| Total | 95.69 | 97.85 | 95.98 | 97.11 | 96.81 | 97.20 | 97.61 | 96.56 | 97.25 | 97.90 |
| Rock | Spl-Grt | |||||||||
| Type | Massive | Faceted | ||||||||
| Location | Patch | Patch | ||||||||
| Sample | DN-4 | DN-4 | ||||||||
| Mineral | Py | Py | Py | Py | Py | Py | Py | Py | Py | |
| Zn | 0.16 | 0.04 | 0.15 | 0.06 | 0.02 | |||||
| Cu | 0.10 | 0.05 | ||||||||
| Ni | 0.05 | 0.07 | 0.06 | 0.03 | 0.39 | 0.04 | ||||
| Co | 0.12 | 0.03 | 0.04 | 0.07 | 0.09 | 0.05 | 0.09 | 0.05 | 0.01 | |
| Fe | 45.25 | 45.87 | 45.22 | 45.41 | 45.25 | 45.14 | 45.15 | 41.18 | 42.99 | |
| S | 49.96 | 48.93 | 49.67 | 49.15 | 48.85 | 49.24 | 49.16 | 43.88 | 43.26 | |
| Total | 95.64 | 94.83 | 95.04 | 94.63 | 94.34 | 94.60 | 94.43 | 85.50 | 86.32 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andronikov, A.V.; Andronikova, I.E.; Sidorinova, T. Trace-Element Geochemistry of Sulfides in Upper Mantle Lherzolite Xenoliths from East Antarctica. Minerals 2021, 11, 773. https://doi.org/10.3390/min11070773
Andronikov AV, Andronikova IE, Sidorinova T. Trace-Element Geochemistry of Sulfides in Upper Mantle Lherzolite Xenoliths from East Antarctica. Minerals. 2021; 11(7):773. https://doi.org/10.3390/min11070773
Chicago/Turabian StyleAndronikov, Alexandre V., Irina E. Andronikova, and Tamara Sidorinova. 2021. "Trace-Element Geochemistry of Sulfides in Upper Mantle Lherzolite Xenoliths from East Antarctica" Minerals 11, no. 7: 773. https://doi.org/10.3390/min11070773
APA StyleAndronikov, A. V., Andronikova, I. E., & Sidorinova, T. (2021). Trace-Element Geochemistry of Sulfides in Upper Mantle Lherzolite Xenoliths from East Antarctica. Minerals, 11(7), 773. https://doi.org/10.3390/min11070773







