Mechanisms at Different pH for Stabilization of Arsenic in Mine Tailings Using Steelmaking Slag
Abstract
:1. Introduction
2. Materials and Experimental Methods
2.1. Preparation of MTs and Steel Slags
2.2. Batch Experiments for Investigating the Mechanism of As Removal by the Steel Slags
2.3. Batch Extraction Experiment for the Efficiency of As-Extraction Reduction from MTs by Steel Slag
3. Results and Discussion
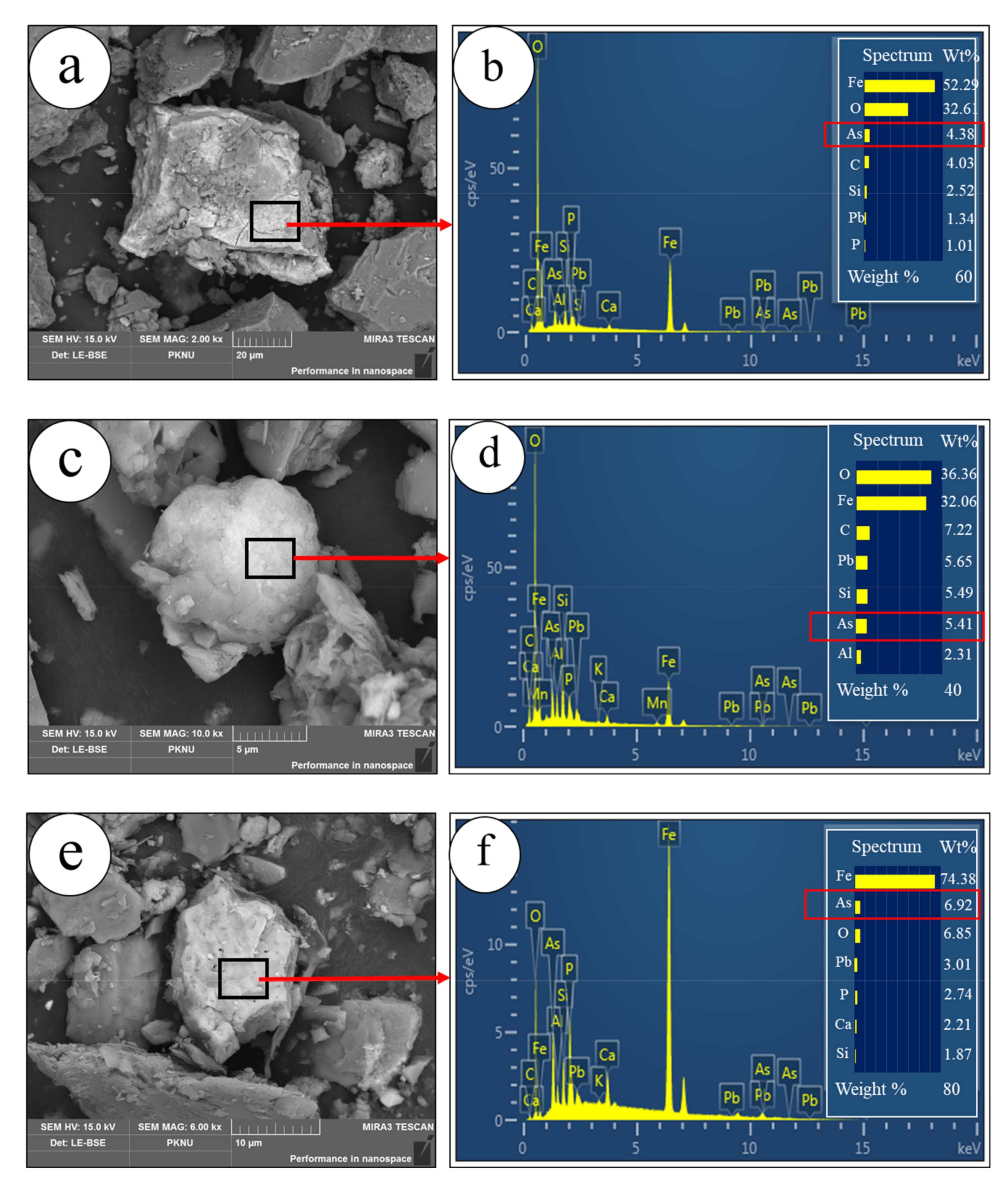
3.1. Evaluation of the Characteristics of the MTs and Steel Slags
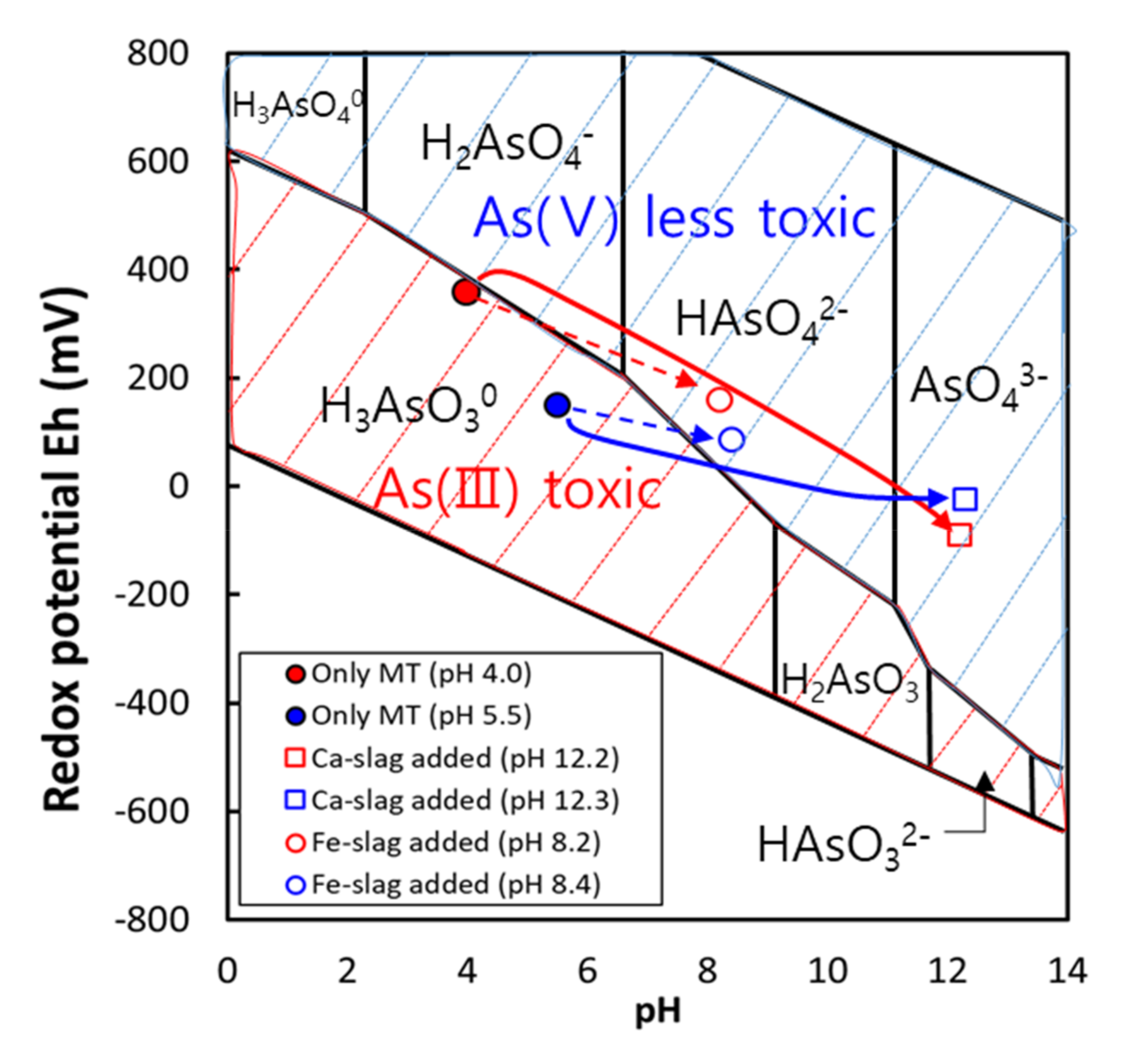
3.2. Batch Experiments for Investigating the Mechanism of Arsenic Removal by Steel Slag
3.3. Batch Extraction Experiment for the Efficiency of As-Extraction Reduction from MTs by Steel Slag
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oremland, R.S.; Stolz, J.F. The ecology of arsenic. Science 2003, 300, 939–944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- USEPA. Available online: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations (accessed on 10 October 2020).
- Loebenstein, J.R. Arsenic: Supply, demand, and the environment. In Mercury and Arsenic Wastes: Removal, Recovery, Treatment and Disposal; U.S. Environmental Protection Agency Pollution Technology Review; Elsevier Science B.V.: Amsterdam, The Netherlands, 1993; pp. 67–71. [Google Scholar]
- Woolson, E.A. Introduction to arsenic chemistry and analysis. In Mercury and Arsenic Wastes: Removal, Recovery, Treatment and Disposal; U.S. Environmental Protection Agency Pollution Technology Review; Elsevier Science B.V.: Amsterdam, The Netherlands, 1993; pp. 60–61. [Google Scholar]
- MIRECO (Mine Reclamation Corporation). Available online: https://www.data.go.kr/data/3077830/fileData.do (accessed on 10 October 2020).
- Ahn, J.S.; Park, Y.S.; Kim, K.W. Mineralogical and geochemical characterization of arsenic in an abandoned mine tailings of Korea. Environ. Geochem. Health 2005, 27, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Luo, X.; Li, D.; Li, Z.; Chen, Z.; Zhu, M. A multidisciplinary investigation on bio-oxidation gold mine tailings from Dandong, China as a potential arsenic pollution source. J. Geochem. Explor. 2019, 196, 33–41. [Google Scholar] [CrossRef]
- Park, J.H.; Choi, H.I.; Yang, I.J. Mine tailings characteristics with a view of prevention of tailings dam failure. J. Korean Soc. Miner. Energy Resour. Eng. 2018, 55, 354–363. (In Korean) [Google Scholar] [CrossRef]
- Raven, K.P.; Jain, A.; Loeppert, R.H. Arsenite and arsenate adsorption on ferrihydrite: kinetics, equilibrium, and adsorption envelopes. Environ. Sci. Technol. 1998, 32, 344–349. [Google Scholar] [CrossRef]
- Chiu, V.Q.; Hering, J.G. Arsenic adsorption and oxidation at manganite surfaces. 1. method for simultaneous determination of adsorbed and dissolved arsenic species. Environ. Sci. Technol. 2000, 34, 2029–2034. [Google Scholar] [CrossRef]
- Alam, M.G.M.; Tokunaga, S.; Maekawa, T. Extraction of arsenic in a synthetic arsenic-contaminated soil using phosphate. Chemosphere 2001, 43, 1035–1041. [Google Scholar] [CrossRef]
- Li, J.; Ding, Y.; Wang, K.; Li, N.; Qian, G.; Xu, Y.; Zhang, J. Comparison of humic and fulvic acid on remediation of arsenic contaminated soil by electrokinetic technology. Chemosphere 2020, 241, 125038. [Google Scholar] [CrossRef]
- USEPA. Available online: https://clu-in.org/download/remed/542r02004/arsenic_report.pdf (accessed on 10 October 2020).
- Moon, D.H.; Dermatas, D.; Menounou, N. Arsenic immobilization by calcium–arsenic precipitates in lime treated soils. Sci. Total Environ. 2004, 330, 171–185. [Google Scholar] [CrossRef]
- Kumpiene, J.; Lagerkvist, A.; Maurice, C. Stabilization of As, Cr, Cu, Pb and Zn in soil using amendments—A review. Waste Manag. 2008, 28, 215–225. [Google Scholar] [CrossRef]
- Lee, M.; Jeon, J. Study for the stabilization of arsenic in the farmland soil by using steel making slag and limestone. Econ. Environ. Geol. 2010, 43, 305–314. (In Korean) [Google Scholar]
- Suda, A.; Makino, T. Functional effects of manganese and iron oxides on the dynamics of trace elements in soils with a special focus on arsenic and cadmium: A review. Geoderma 2016, 270, 68–75. [Google Scholar] [CrossRef]
- Lewińska, K.; Karczewska, A.; Siepak, M.; Gałka, B. Potential of Fe-Mn wastes produced by a water treatment plant for arsenic immobilization in contaminated soils. J. Geochem. Explor. 2018, 184, 226–231. [Google Scholar] [CrossRef]
- Hartley, W.; Edwards, R.; Lepp, N.W. Arsenic and heavy metal mobility in iron oxide-amended contaminated soils as evaluated by short- and long-term leaching tests. Environ. Pollut. 2004, 131, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Masscheleyn, P.H.; Delaune, R.D.; Patrick, W.H. Effect of redox potential and pH on arsenic speciation and solubility in a contaminated soil. Environ. Sci. Technol. 1991, 25, 1414–1419. [Google Scholar] [CrossRef]
- Proctor, D.M.; Fehling, K.A.; Shay, E.C.; Wittenborn, J.L.; Green, J.J.; Avent, C.; Bigham, R.D.; Connolly, M.; Lee, B.; Shepker, T.O.; et al. Physical and chemical characteristics of blast furnace, basic oxygen furnace, and electric arc furnace steel industry slags. Environ. Sci. Technol. 2000, 34, 1576–1582. [Google Scholar] [CrossRef]
- Van Oss, H.G. Slag-iron and steel. In Minerals Yearbook; U.S. Geological Survey: Reston, VA, USA, 2002. [Google Scholar]
- Park, J.B.; Lee, B.C.; Jang, M.H.; Na, H.H. Environmental characteristics of leachates from steel slag. J. Korean Geosynth. Soc. 2012, 11, 31–38. (In Korean) [Google Scholar] [CrossRef]
- MOE (Ministry of Environment). The Guideline to Recycling for Steel Slag and Coal Ash Discharger, Korea; Notification 2016-217; MOE: Sejong-si, Korea, 2016. (In Korean)
- Claveau-Mallet, D.; Wallace, S.; Comeau, Y. Removal of phosphorus, fluoride and metals from a gypsum mining leachate using steel slag filters. Water Res. 2013, 47, 1512–1520. [Google Scholar] [CrossRef]
- Kim, S.H.; Jeong, S.; Chung, H.; Nam, K. Stabilization mechanism of arsenic in mine waste using basic oxygen furnace slag: The role of water contents on stabilization efficiency. Chemosphere 2018, 208, 916–921. [Google Scholar] [CrossRef]
- Edwin, B. Major steel-producing countries 2017 and 2018. In World Steel in Figures 2019; World Steel Association: Brussel, Belgium, 2019; Available online: https://worldsteel.org/steel-by-topic/statistics/top-producers (accessed on 10 October 2020).
- Neuhold, S.; van Zomeren, A.; Dijkstra, J.J.; van der Sloot, H.A.; Drissen, P.; Algermissen, D.; Mudersbach, D.; Schüler, S.; Griessacher, T.; Raith, J.G.; et al. Investigation of possible leaching control mechanisms for chromium and vanadium in electric arc furnace (EAF) slags using combined experimental and modeling approaches. Minerals 2019, 9, 525. [Google Scholar] [CrossRef] [Green Version]
- Cui, M.; Lee, Y.; Choi, J.; Kim, J.; Han, Z.; Son, Y.; Khim, J. Evaluation of stabilizing materials for immobilization of toxic heavy metals in contaminated agricultural soils in China. J. Clean. Prod. 2018, 193, 748–758. [Google Scholar] [CrossRef]
- Fisher, L.V.; Barron, A.R. The recycling and reuse of steelmaking slags—A review. Resour Conserv Recycl 2019, 146, 244–255. [Google Scholar] [CrossRef] [Green Version]
- Herbelin, M.; Bascou, J.; Lavastre, V.; Guillaume, D.; Benbakkar, M.; Peuble, S.; Baron, J.-P. Steel slag characterisation-benefit of coupling chemical, mineralogical and magnetic techniques. Minerals 2020, 10, 705. [Google Scholar] [CrossRef]
- MOE (Ministry of Environment). The Soil Environmental Conservation Law, Korea; Notification 2017-22; MOE: Sejong-si, Korea, 2017. (In Korean)
- USEPA. Available online: https://www.epa.gov/hw-sw846/sw-846-test-method-1311-toxicity-characteristic-leaching-procedure (accessed on 10 October 2020).
- USEPA. Available online: https://www.epa.gov/hw-sw846/sw-846-test-method-1312-synthetic-precipitation-leaching-procedure (accessed on 10 October 2020).
- Nesbitt, H.W.; Canning, G.W.; Bancroft, G.M. XPS study of reductive dissolution of 7Å-birnessite by H3AsO3, with constraints on reaction mechanism. Geochim. Cosmochim. Acta 1998, 62, 2097–2110. [Google Scholar] [CrossRef]
- USDA. Available online: https://www.nrcs.usda.gov/wps/portal/nrcs/detail/soils/survey/?cid=nrcs142p2_054167 (accessed on 10 October 2020).
- Guan, X.; Dong, H.; Ma, J.; Jiang, L. Removal of arsenic from water: Effects of competing anions on As(III) removal in KMnO4-Fe(II) process. Water Res. 2009, 43, 3891–3899. [Google Scholar] [CrossRef]
- Zobrist, J.; Dowdle, P.R.; Davis, J.A.; Oremland, R.S. Mobilization of arsenite by dissimilatory reduction of adsorbed arsenate. Environ. Sci. Technol. 2000, 34, 4747–4753. [Google Scholar] [CrossRef]
- Korte, N.E.; Fernando, Q. A review of arsenic (III) in groundwater. Environ. Sci. Technol. 1991, 21, 1–39. [Google Scholar] [CrossRef]
- Bothe, J.V.; Brown, P.W. Arsenic immobilization by calcium arsenate formation. Environ. Sci. Technol. 1999, 33, 3806–3811. [Google Scholar] [CrossRef]
- Zhu, Y.N.; Zhang, X.H.; Xie, Q.L.; Wang, D.Q.; Cheng, G.W. Solubility and stability of calcium arsenates at 25 °C. Water Air Soil Pollut. 2006, 169, 221–238. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Majzlan, J.; Königsberger, E. Thermodynamic properties for arsenic minerals and aqueous species. Rev. Miner. Geochem 2014, 79, 217–255. [Google Scholar] [CrossRef]
- Beak, D.G.; Basta, N.T.; Scheckel, K.G.; Traina, S.J. Bioaccessibility of arsenic (V) bound to ferrihydrite using a simulated gastrointestinal system. Environ. Sci. Technol. 2006, 40, 1364–1370. [Google Scholar] [CrossRef] [PubMed]
- Fendorf, S.; Eick, M.J.; Grossl, P.; Sparks, D.L. Arsenate and chromate retention mechanisms on Goethite. 1. surface structure. Environ. Sci. Technol. 1997, 31, 315–320. [Google Scholar] [CrossRef]
- Tokunaga, S.; Hakuta, T. Acid washing and stabilization of an artificial arsenic- contaminated soil. Chemosphere 2002, 46, 31–38. [Google Scholar] [CrossRef]
- Kosmulski, M. The pH dependent surface charging and points of zero charge. VII. Update. Adv. Colloid Interface Sci. 2018, 251, 115–138. [Google Scholar] [CrossRef]
- Alexandratos, S.D. New polymer-supported ion-complexing agents: Design, precipitation and metal ion affinities of immobilized ligands. J. Hazard. Mater. 2007, 139, 467–470. [Google Scholar] [CrossRef]
- Lee, J.; Kim, J.; Cho, J.; Hwang, J.; Lee, M. Feasibility study of the stabilization for the arsenic contaminated farmland soil by using amendments at Samkwang abandoned mine. Econ. Environ. Geol. 2011, 44, 217–228. (In Korean) [Google Scholar]






| Material Type | pH | Component Portion from XRF Analysis (wt.%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | K2O | Fe2O3 | MgO | MnO | CaO | P2O5 | SO3 | ||
| MTs | 5.5 | 76.5 | 12.8 | 5.4 | 2.6 | 1.3 | - | 0.6 | 0.4 | - |
| Ca-slag | 12.4 | 13.1 | 2.0 | - | 26.6 | 5.8 | 3.1 | 46.9 | 0.2 | 0.8 |
| Fe-slag | 8.4 | 29.7 | 12.1 | - | 24.3 | 4.3 | 6.7 | 19.3 | - | 0.6 |
| Material Type | Concentration (mg/kg) | ||||
|---|---|---|---|---|---|
| Zn | Cu | As | Cd | Pb | |
| MTs | 133.2 | 33.0 | 2225.3 | 3.5 | 39.6 |
| Ca-slag | 3.1 | - | - | - | 5.1 |
| Fe-slag | 222.2 | 172.7 | - | - | 97.9 |
| KSPWL at Area 3 | 2000 | 2000 | 200 | 700 | 700 |
| Type of Steel Slag | Leaching Test Type | Concentration (mg/L) | ||||
|---|---|---|---|---|---|---|
| Zn | Cu | As | Cd | Pb | ||
| Ca-slag | TCLP | DL | DL | DL | DL | DL |
| SPLP | DL | DL | DL | DL | DL | |
| Fe-slag | TCLP | 4.545 | 0.024 | DL | DL | 0.107 |
| SPLP | DL | 0.005 | DL | DL | DL | |
| Extraction Tolerance Limit | 3 | 1.5 | 0.3 | 3 | ||
| Steel Slag Type | The pH before the Reaction | The pH after the Reaction | Initial As Concentration (mg/L) | As Concentration after the Reaction (mg/L) | As Removal Efficiency (%) |
|---|---|---|---|---|---|
| Ca-slag | 4.0 | 12.2 | 100.0 | 3.0 | 97.0 |
| 5.5 | 12.3 | 2.1 | 97.9 | ||
| Fe-slag | 4.0 | 8.2 | 100.0 | 21.3 | 78.7 |
| 5.5 | 8.4 | 20.4 | 79.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.; Kim, S.; Tak, H.; Kim, K.; Chung, C.-W.; Lee, M. Mechanisms at Different pH for Stabilization of Arsenic in Mine Tailings Using Steelmaking Slag. Minerals 2020, 10, 900. https://doi.org/10.3390/min10100900
Kim T, Kim S, Tak H, Kim K, Chung C-W, Lee M. Mechanisms at Different pH for Stabilization of Arsenic in Mine Tailings Using Steelmaking Slag. Minerals. 2020; 10(10):900. https://doi.org/10.3390/min10100900
Chicago/Turabian StyleKim, Taehyoung, Seonhee Kim, Hyunji Tak, Kyeongtae Kim, Chul-Woo Chung, and Minhee Lee. 2020. "Mechanisms at Different pH for Stabilization of Arsenic in Mine Tailings Using Steelmaking Slag" Minerals 10, no. 10: 900. https://doi.org/10.3390/min10100900




