Abstract
An efficient algorithm leading to the Fries canonical structure is presented for benzenoid hydrocarbons. This is a purely topological approach, which is based on adjacency matrices and the Hadamard procedure of matrix multiplication. The idea is presented for naphthalene, as an example. The Fries canonical-structures are also derived for anthracene, coronene, triphenylene, phenanthrene, benz[a]pyrene, and one large benzenoid system. The Fries concept can be convenient for obtaining Clar structures with the maximum number of sextets, which in turn effectively represent π-electron (de)localization in benzenoid hydrocarbons.
1. Introduction
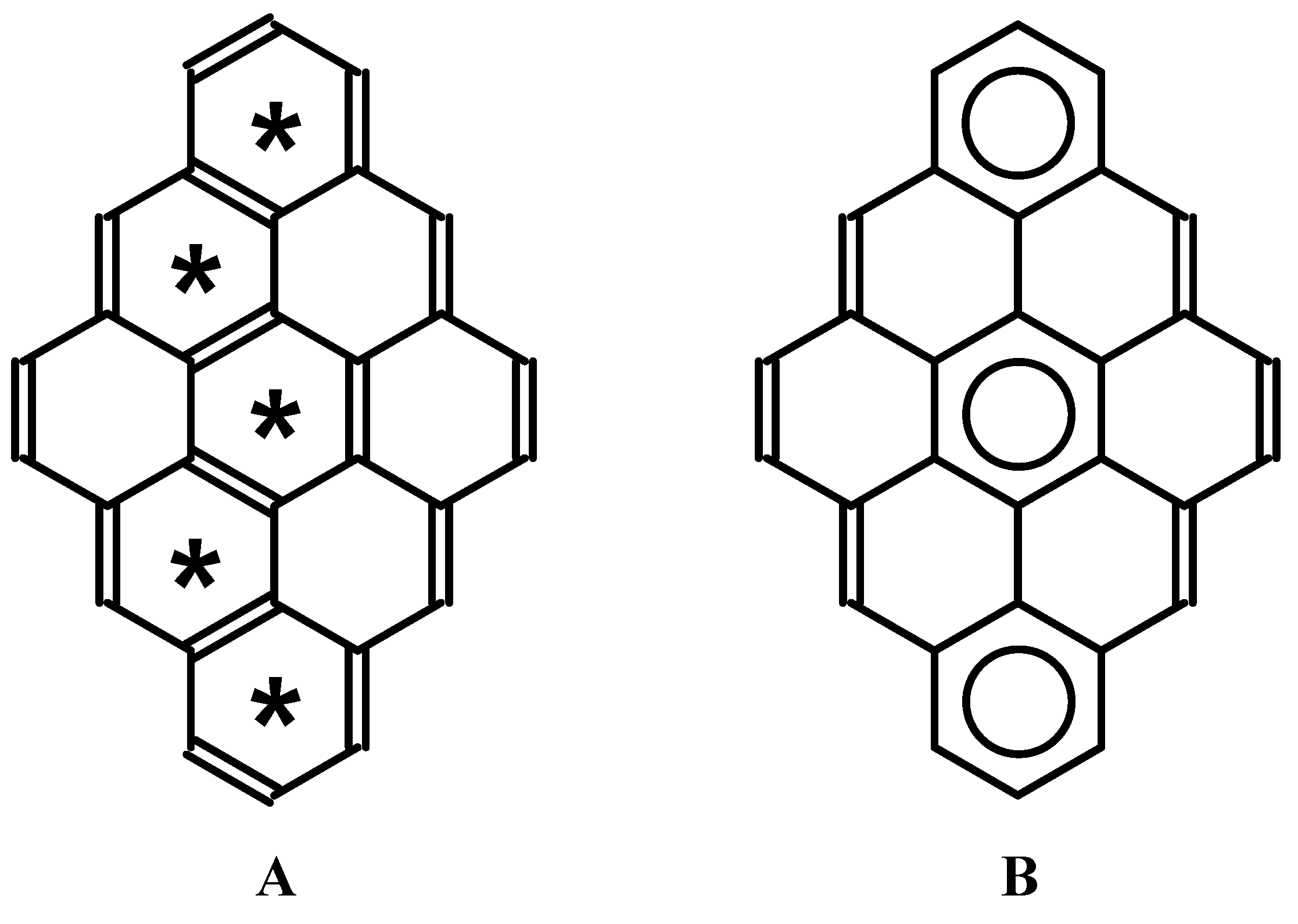
Benzenoid hydrocarbons are probably the most important π-electron systems. They have often been used to analyze various hypotheses concerning chemical or physicochemical behavior, or both, in relation to electron structure and aromaticity. The amount of literature is already vast, and the interested reader may find many excellent recent papers and reviews on this topic [1,2,3,4,5,6,7,8,9]. From a historical perspective, one concept seems to have had an immense impact on research in the field. In the 1960s, Clar introduced a classification of individual rings in polycyclic benzenoid hydrocarbons [10,11], which helped one to understand, in a simple way, their π-electron structure [12,13,14,15,16,17,18,19,20,21]. He noted that resonance contributors that maximize the number of rings with three double-bonds are the most important [22]. Rings which are described by so-called Clar sextets exhibit the highest aromatic character; this character is associated with the ring’s stability [23]. Thirty years before Clar’s suggestion, other interesting ideas in this field had been proposed. To relate with chemical stability, Fries [24] distinguished a resonance contributor that maximizes the number of rings with a benzene-like Kekulé electron structure [9]. Graovac et al. indicated the importance of this structure [25]. To some extent, there is a similarity between Clar’s and Fries’s ideas; in both cases, circles are inscribed inside benzenoid rings that have six π-electrons, but in the case of the Fries structure, these circles may share an edge. An illustrative example of the relation between both concepts is shown in Figure 1. The dibenzo[bc,kl]coronene molecule serves as a good example. Fries structure (Figure 1A) presents a case with the maximum number of Kekulé-type rings. Clar structure(s), with the maximum number of isolated sextets, can be easily deduced from the Fries structure (see Figure 1B).
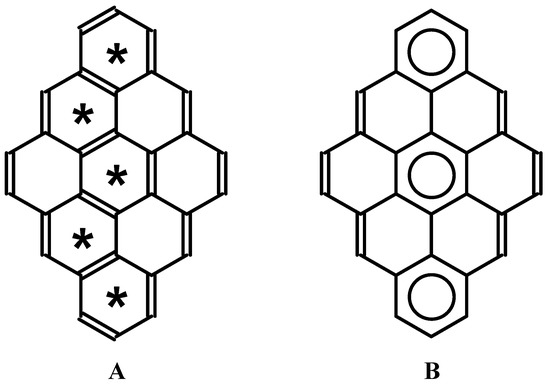
Figure 1.
Dibenzo[bc,kl]coronene molecule. A) Fries structure. Stars denote Kekulé-type six-membered rings. B) Clar structure with maximal number of isolated sextets.
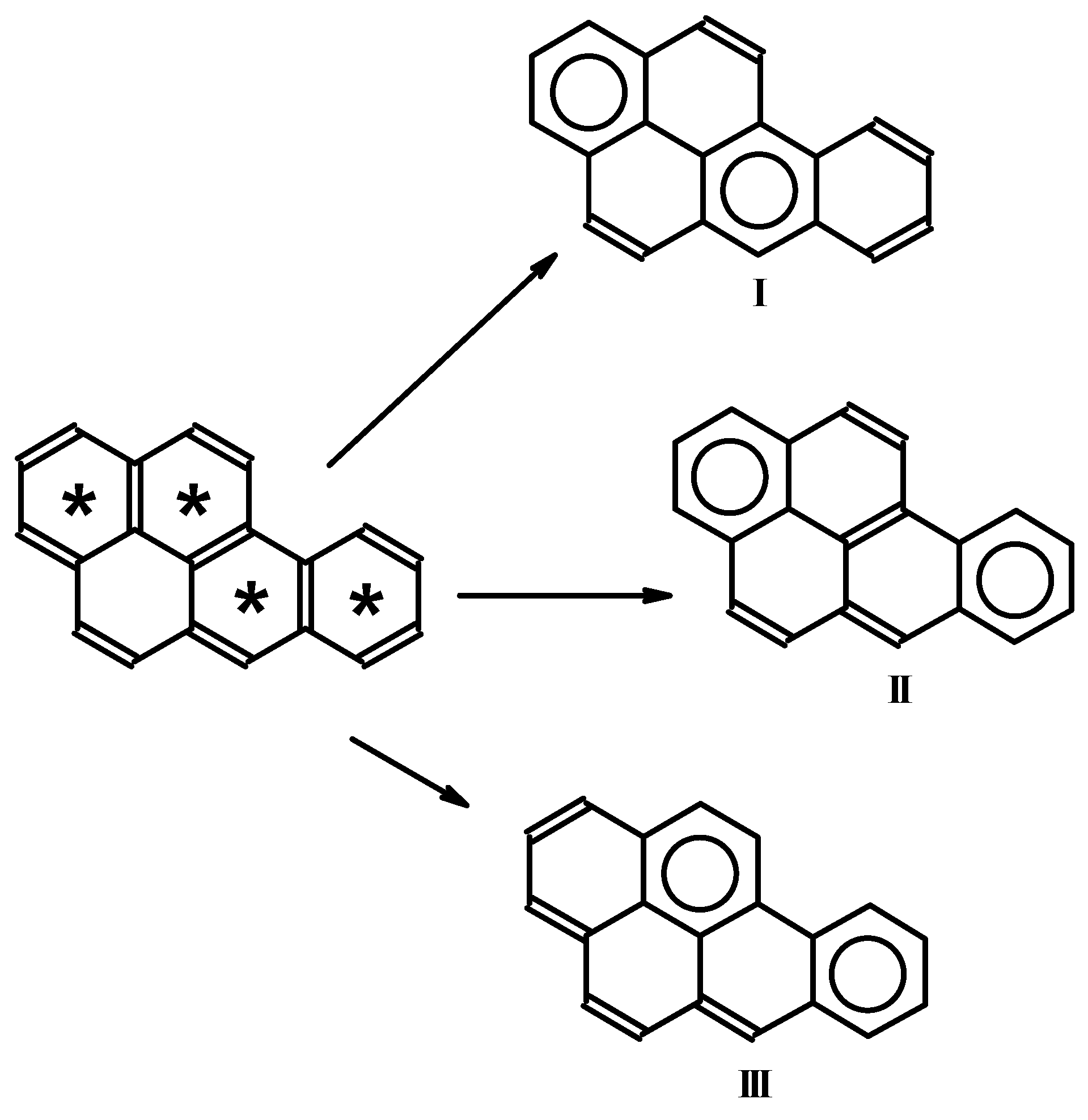
Importantly, from a given Fries structure, one can obtain a few Clar structures with the maximum number of separated sextets. In such a case, additional rules should be considered, such as the number of isolated formal double bonds, which should be separated from each other as much as possible. Figure 2 presents the decomposition of the Fries structure for benzo[a]pyrene into three Clar structures with the maximum number of sextets. In case I, the most efficient separation of double bonds is achieved. Local descriptors of aromaticity (such as HOMA [26] or NICS [27,28]) confirm that the separated sextets in these Clar structures have the most efficient π-electron delocalization [23,29].
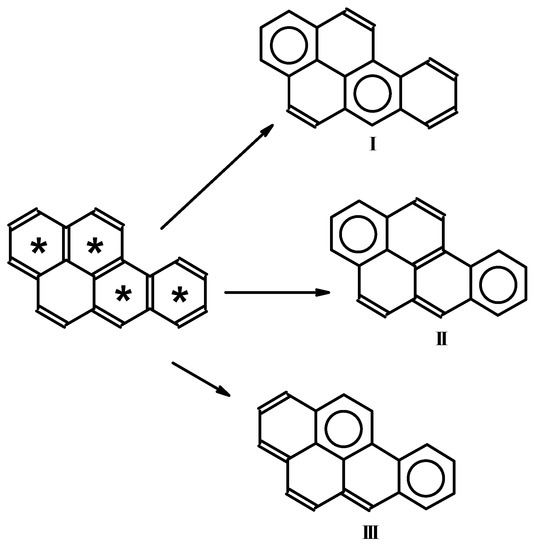
Figure 2.
Decomposition of Fries structure for benzo[a]pyrene into three Clar structures.
It is not always an easy task to manually draw the Clar structure with the maximum number of isolated sextets for a given benzenoid hydrocarbon, especially if a molecule is large and has low symmetry. This however, can be easily achieved from the Fries structure, as such a transformation is straightforward. In this paper, we present a mathematical way of finding this important, canonical structure that is the main contributor for benzenoid hydrocarbons.
2. Method and Applications
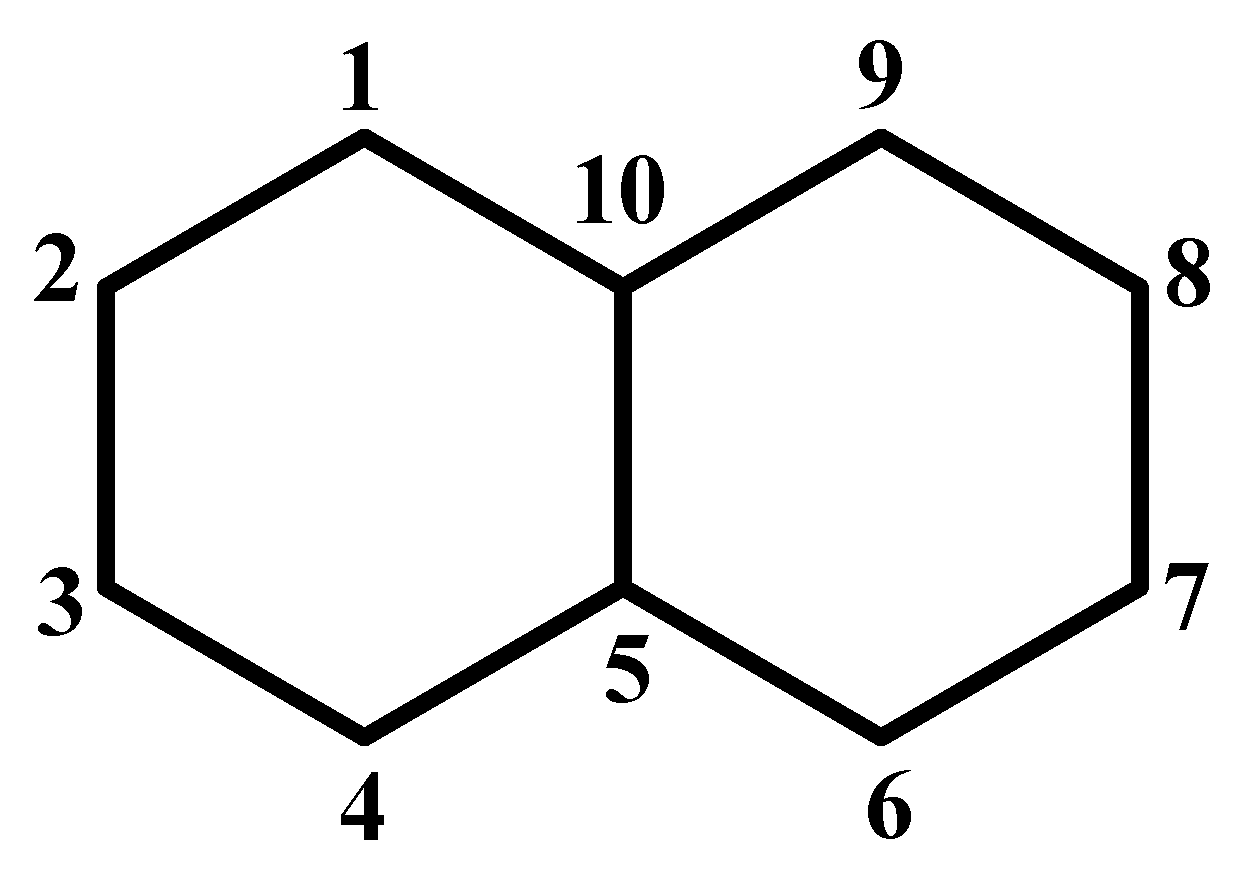
The presentation of the method is illustrated by use of the naphthalene molecule (see Figure 3) as an example.
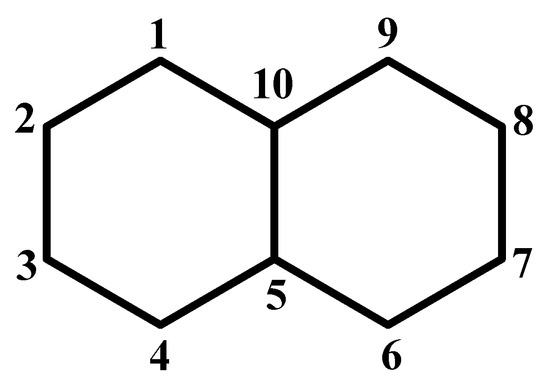
Figure 3.
Numbering of atoms for naphthalene used in this paper.
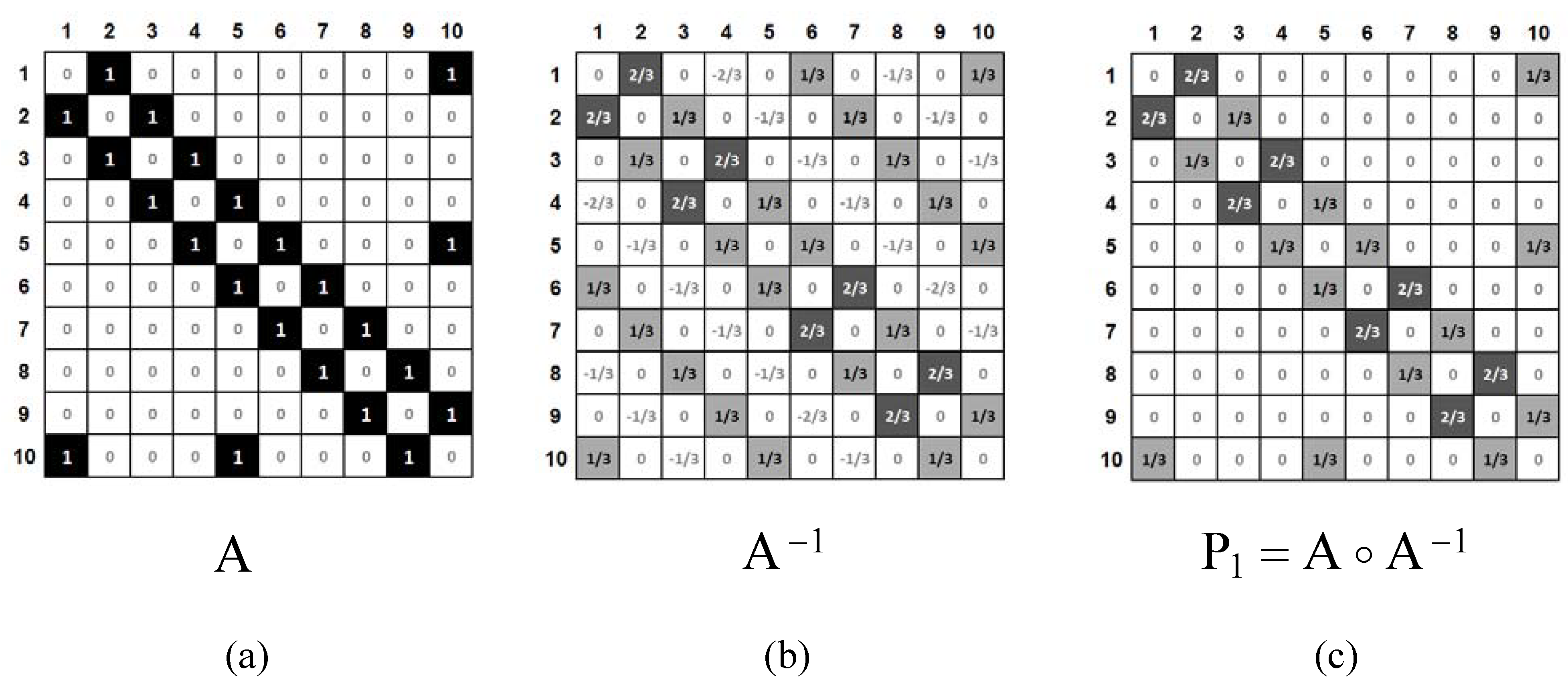
For any benzenoid hydrocarbon, one can define an adjacency matrix, A, the elements of which, aij, represent CC bonds. The adjacency matrix is schematically presented in Figure 4a.
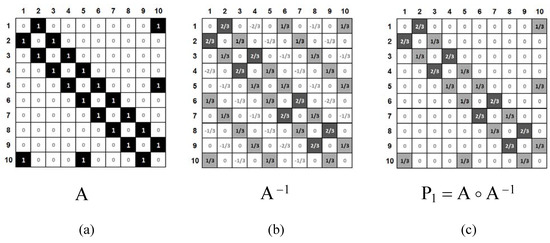
Figure 4.
A) The adjacency matrix, A, for naphthalene (1 and 0 are represented by black boxes and white boxes, respectively). B) The inverse matrix A−1 (light-gray boxes and dark-gray boxes correspond to the values of 1/3 and 2/3, respectively; white boxes correspond to negative or zero values). C) The Hadamard product of matrices A and A−1 (light-gray boxes and dark-gray boxes correspond to the values of 1/3 and 2/3, respectively; white boxes correspond to values of zero).
It is well known that for benzenoid hydrocarbons, the inverse matrix A−1 (Figure 4b) contains the Pauling bond orders [30]. The Hadamard product [31,32] (for definition, see Eq. 1) of matrices A and A−1, gives matrix P1, which contains exclusively Pauling bond orders, prs.
where aij, bij, and cij are elements of (n×m) matrices A, B, and C, respectively.
C = A○B cij = aij·bij
The Pauling bond orders can also be obtained in an alternative way. How many times a given bond is double in all Kekulé structures is counted, and then this number is divided by the total number of canonical structures.
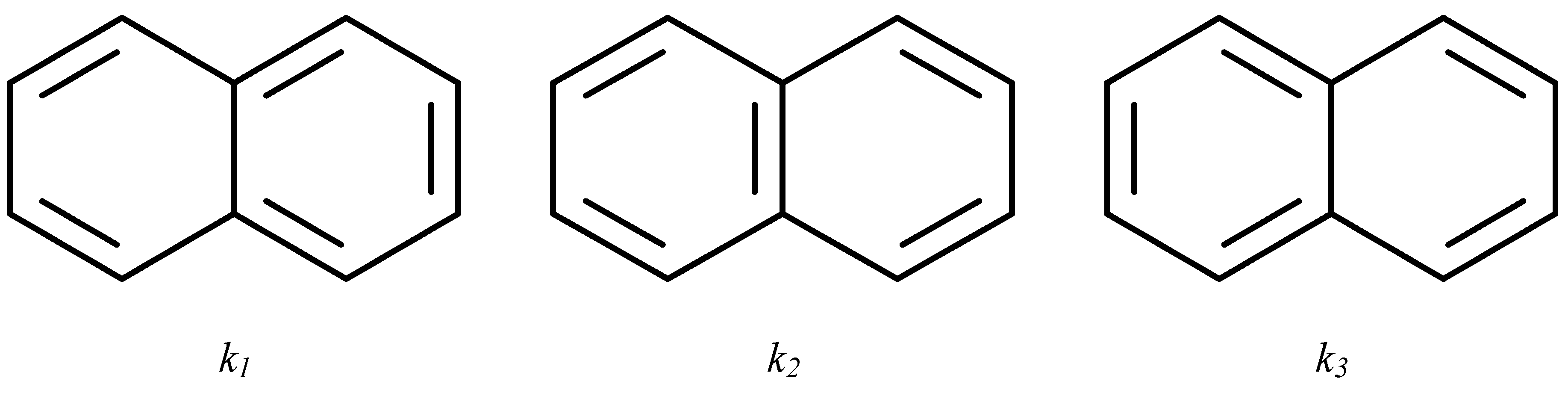
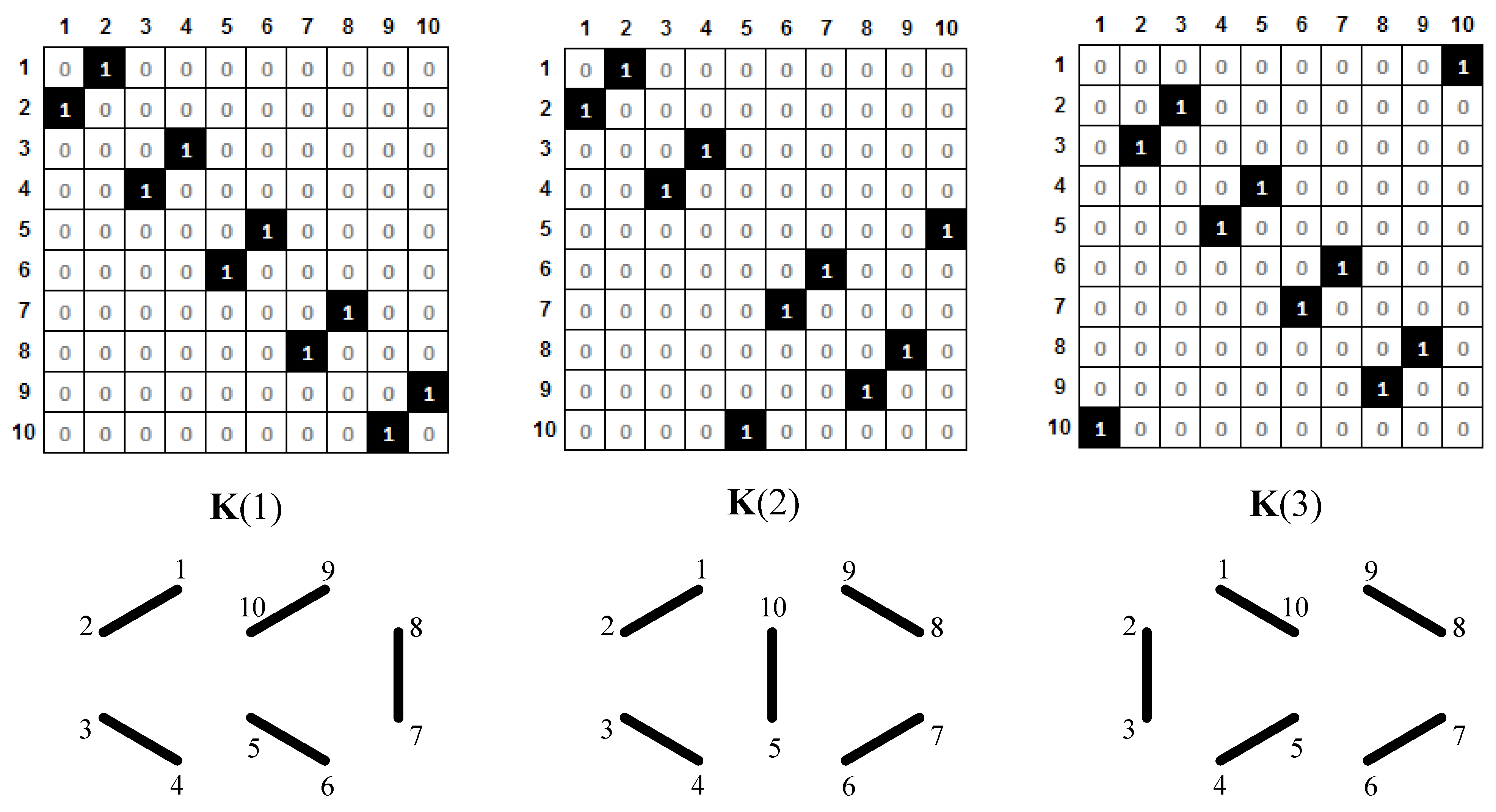
Each of the canonical structures of the naphthalene molecule (Figure 5) contains a Pauling bond order of either 1 or 0. This information may be presented by a matrix with elements 0 or 1. Therefore, we define, for each canonical structure, k1, k2, or k3, matrices K(1), K(2), and K(3), which contain full information about the positions of double bonds. These matrices are shown in Figure 6.
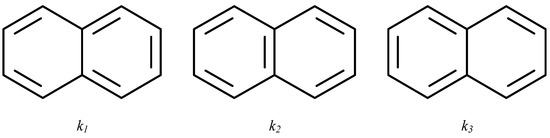
Figure 5.
Three canonical structures of naphthalene.
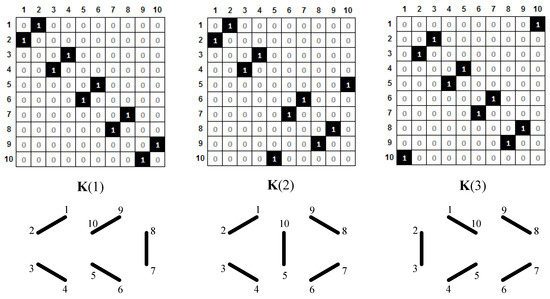
Figure 6.
Matrices K(1), K(2), and K(3), which correspond to Kekulé structures k1, k2, and k3. Double CC bonds (represented in a graphical way below each matrix) are denoted by the entry 1.
The matrices K(1), K(2), and K(3), are so-called self-inverse matrices with the property
It is known that for any benzenoid system, the square-root of the absolute value of the determinant of the adjacency matrix is equal to the number of its canonical structures, so that [30]. In the case of any K matrix representing a canonical structure, because, for one structure, K = 1.
In order to generate the K matrix that represents a Fries canonical-structure, we construct from matrices A and P, as presented in Figure 4, a recurrence function, denoted as a Fries structure generating function (FGF). This function is defined in Equation 3.
A stands for an adjacency matrix, and P for the matrix of Pauling bond orders. The multiplication follows the Hadamard rule, and n is a number of steps in the recurrence procedure [31,32]. In Figure 7, a graphical illustration is presented of the procedure as applied to the naphthalene molecule.

Figure 7.
Graphical illustration of the procedure for obtaining the Fries structure of naphthalene in four cycles of iteration. The elements of matrices are shown in a symbolic way as varying degrees of grayness.
For the iteration with n = 4, we obtain the matrix K(2), which represent the Fries structure for naphthalene (Figure 4).
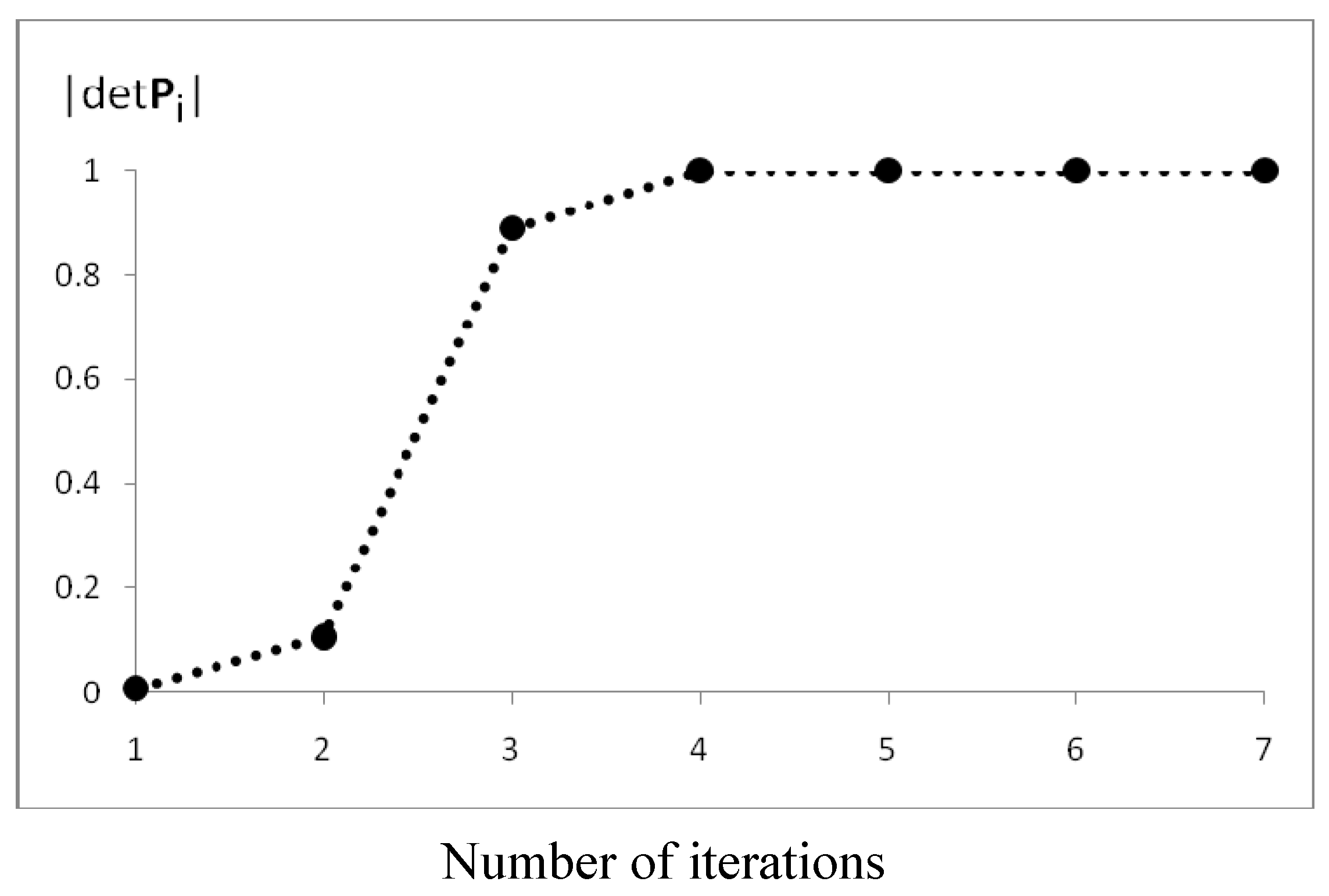
The efficiency of the algorithm for this case is presented in Figure 8. After n = 7 iterations, we obtained the Fries canonical structure for which |detPn| = 1. Note that, in subsequent iterations, because of the property given in Equation 2, the Pn matrix is closer to the K matrix. The determinant of P is a useful measure of the FGF convergence.
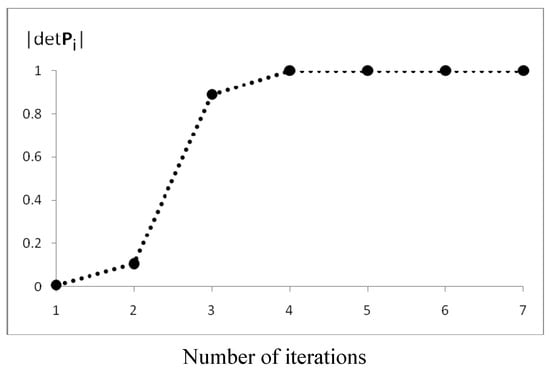
Figure 8.
Convergence of the FGF procedure for naphthalene.
In general, for any benzenoid hydrocarbon, the final result of the iteration leads to the matrix, K(F), of its Fries structure
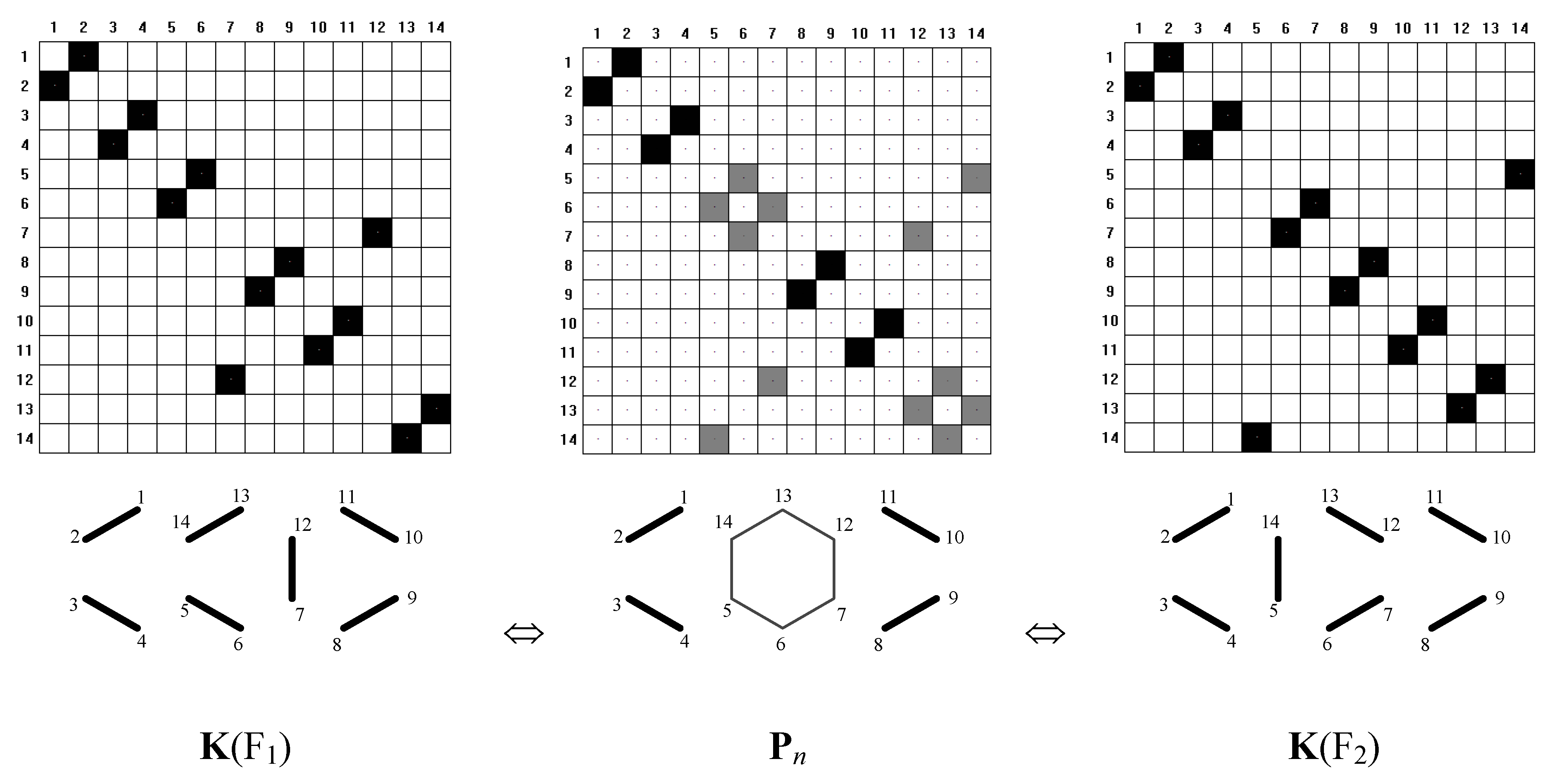
To illustrate the method, we have chosen a few typical benzenoid hydrocarbons. Consider first the case of anthracene, in which we can describe two symmetry dependent canonical-structures, which can be regarded as Fries structures. In this case, the FGF function converges to the matrix Pn that is a superposition of matrices K(F1) and K(F2) (see Equation 6 and Figure 9 and Figure 10).

Figure 9.
Two symmetry-equivalent Fries canonical structures for the anthracene molecule.
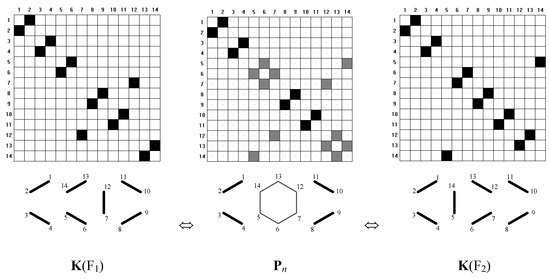
Figure 10.
Illustration of superposition of k1 and k2 with matrices below, K(F1), Pn, and K(F2). Solid lines and black boxes visualize prs = 1, dashed lines and gray boxes indicate prs = 0.5.
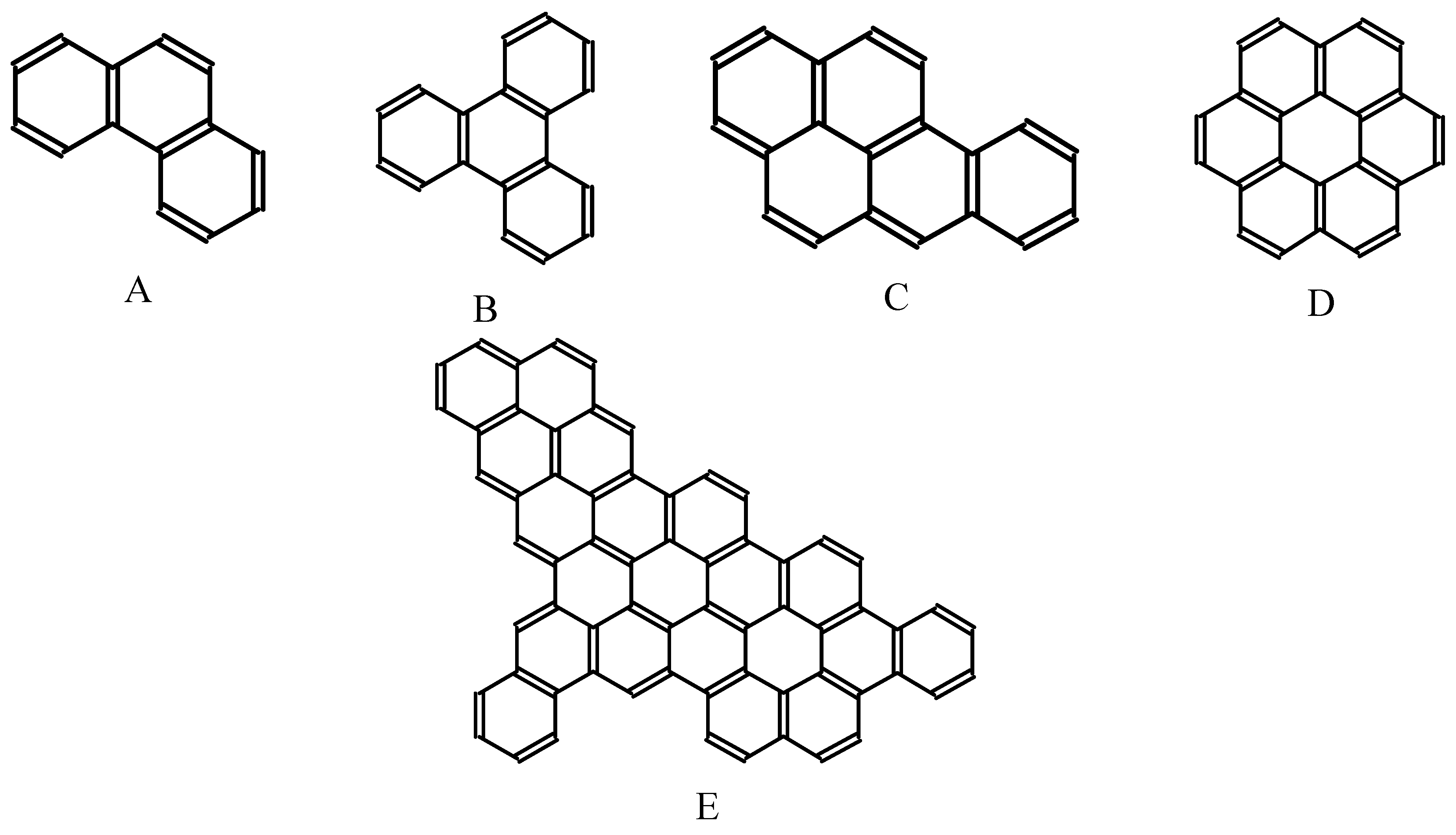
In Figure 11, the Fries structures generated by the FGF algorithm are presented for a few important benzenoid hydrocarbons: phenanthrene, triphenylene, benz[a]pyrene, coronene, and a lager benzenoid structure with the formula C64H26. These structures can be easily transformed into Clar structures with the maximum number of sextets, as shown in Figure 12.
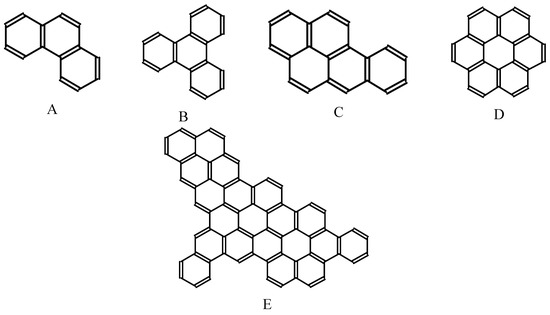
Figure 11.
Fries structures for phenanthrene (A), triphenylene (B), benz[a]pyrene (C), coronene (D), and one large benzenoid hydrocarbon (E) obtained form FGF procedure in 10 cycles of interaction.
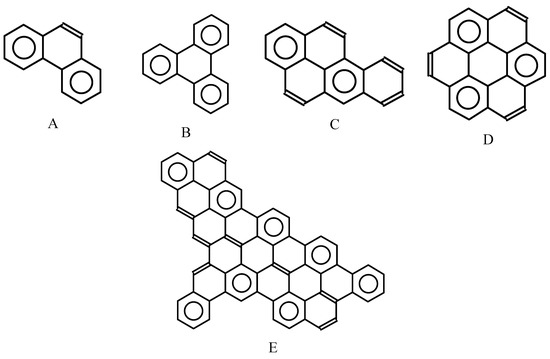
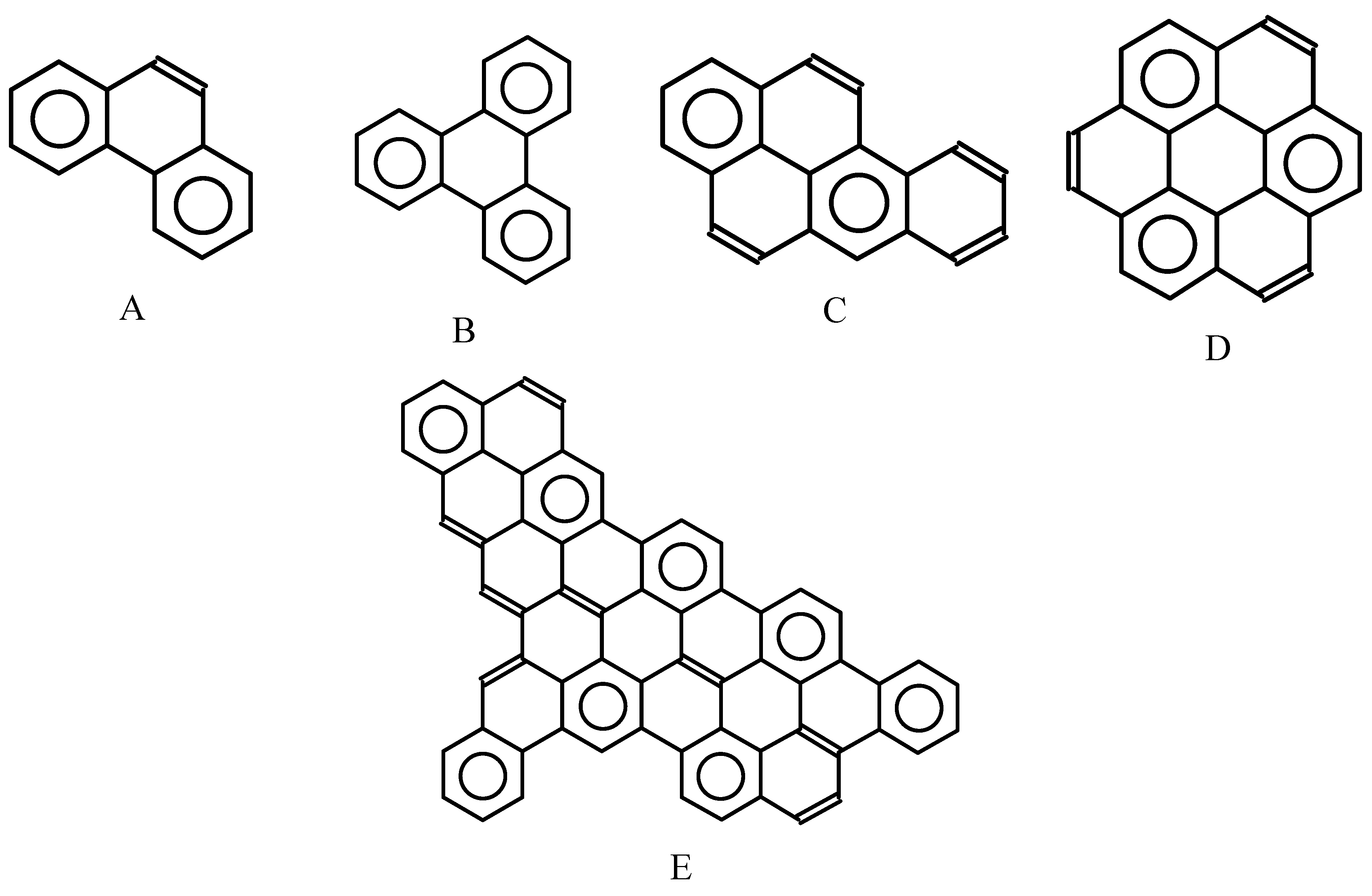
Figure 12.
Clar structure with the maximal number of sextets deduced from the Fries structure for phenanthrene (A), triphenylene (B), benz[a]pyrene (C), coronene (D), and one large benzenoid hydrocarbon (E).
3. Conclusions
In this paper, we have proposed an efficient algorithm leading to an assignment of the Fries canonical structure to any benzenoid hydrocarbon. One of the examples, E, presented in Figure 11 and Figure 12, supports the view that even large and nonsymmetrical systems can be analyzed in this way. Therefore, the Fries concept can be very convenient for obtaining Clar structures with the maximum number of sextets, which well represent π-electron (de)localization in benzenoid hydrocarbons [23].
Acknowledgements
Financial support from BST-120000-501/64-143157 (University of Warsaw) is gratefully acknowledged.
References and Notes
- Garratt, P.J. Aromaticity; Wiley Inc.: New York, NY, USA, 1986. [Google Scholar]
- Lloyd, D. The Chemistry of Conjugated Cyclic Compounds: To Be or Not to Be like Benzene; J. Wiley: New York, NY, USA, 1990. [Google Scholar]
- Minkin, V.I.; Glukhovtsev, M.N.; Simkin, B.Y. Aromaticity and Antiaromaticity; J. Wiley: New York, NY, USA, 1994. [Google Scholar]
- Hopf, H. Classics in Hydrocarbon Chemistry: Syntheses, Concepts, Perspectives; Wiley – VCH: Weinheim, Germany, 2000. [Google Scholar]
- Pascal, R.A., Jr. Twisted Acenes. Chem. Rev. 2006, 106, 4809–4819. [Google Scholar] [CrossRef] [PubMed]
- Schleyer, P.v.R. Special Issue of the Journal Devoted to Aromaticity. Chem. Rev. 2001, 101, 1115–1566. [Google Scholar] [CrossRef] [PubMed]
- Lazzeretti, P.; Corminboeuf, C.; Heine, T.; Seifert, G.; Schleyer, P.v.R.; Weber, J.; De Proft, F.; Geerlings, P.; Hiberty, P.C.; Shaik, S.; Rassat, A.; Soncini, A.; Fowler, P.W.; Jenneskens, L.W.; Havenith, R.W.A.; Engelberts, J.J.; Fowler, P.W.; Steiner, E.; van Lenthe, J.; Poater, J.; Garcia-Cruz, I.; Illas, F.; Solà, M.; Diudea, M.V.; Taylor, R.; Steiner, E.; Fowler, P.W.; Steiner, E.; Shetty, S.; Kanhere, D.; Pal, S.; Krygowski, T.M.; Cyrański, M.K. Lectures at the European Science Foundation Workshop: New Perspectives on Aromaticity, Exeter, UK, 5–9 July, 2003. Phys. Chem. Chem. Phys. 2005, 6, 217–339. [Google Scholar] [CrossRef]
- Schleyer, P.v.R. Special Issue of the Journal Devoted to Delocalization Sigma and Pi. Chem. Rev. 2005, 105, 3433–3948. [Google Scholar] [CrossRef]
- Randić, M. Aromaticity of Polycyclic Conjugated Hydrocarbons. Chem. Rev. 2003, 103, 3449–3605. [Google Scholar] [CrossRef]
- Clar, E. Polycyclic Hydrocarbons; Academic Press: London, UK, 1964; Volume 1–2. [Google Scholar]
- Clar, E. The aromatic Sextet; Wiley: New York, NY, USA, 1972. [Google Scholar]
- Poater, J.; Solà, M.; Duran, M.; Fradera, X. The Calculation of Electron Localization and Delocalization Indices at the Hartree-Fock, Density Functional and Post-Hartree-Fock Levels of Theory. Theor. Chem. Acc. 2002, 107, 362–371. [Google Scholar] [CrossRef]
- Poater, J.; Fradera, X.; Duran, M.; Solà, M. The Delocalization Index as an Electronic Aromaticity Criterion: Application to a Series of Planar Polycyclic Aromatic Hydrocarbons. Chem. Eur. J. 2003, 9, 400–406. [Google Scholar] [CrossRef]
- Portella, G.; Poater, J.; Solà, M. Assessment of Clar’ s Aromatic π-Sextet Rule by Means of PDI, NICS and HOMA Indicators of Local Aromaticity. J. Phys. Org. Chem. 2005, 18, 785–791. [Google Scholar] [CrossRef]
- Faglioni, F.; Ligabue, A.; Pelloni, S.; Soncini, A.; Viglione, R.G.; Ferraro, M.B.; Zanasi, R.; Lazzaretti, P. Why Downfield Proton Chemical Shifts Are Not Reliable Aromaticity Indicators? Org. Lett. 2005, 7, 3457–3460. [Google Scholar] [CrossRef]
- Maksić, Z.B.; Barić, D.; Muller, T. Clar’s Sextet Rule Is a Consequence of the σ-Electron Framework. J. Phys. Chem. 2006, 110, 10135–10147. [Google Scholar] [CrossRef]
- Bultinck, P.; Ponec, R.; Gallegos, A.; Fias, S.; Van Damme, S.; Carbó-Dorca, R. Generalized Polansky Index as an Aromaticity Measure in Polycyclic Aromatic Hydrocarbons. Croat. Chem. Acta 2006, 79, 363–371. [Google Scholar]
- Bultinck, P. Critical Analysis of the Local Aromaticity Concept in Polyaromatic Hydrocarbons. Farad. Disc. 2007, 135, 347–365. [Google Scholar] [CrossRef]
- Alonso, M.; Herradon, B. Neural Networks as a Tool to Classify Compounds According to Aromaticity Criteria. Chem. Eur. J. 2007, 13, 3913–3923. [Google Scholar] [CrossRef] [PubMed]
- Fias, S.; van Damme, S.; Bultinck, P. Multidimensionality of Delocalization Indices and Nucleus Independent Chemical Shifts in Polycyclic Aromatic Hydrocarbons. J. Comput. Chem. 2008, 29, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Fias, S.; Fowler, P.W.; Delgado, J.L.; Hahn, U.; Bultinck, P. Correlation of Delocalization Indices and Current-Density Maps in Polycyclic Aromatic Hydrocarbons. Chem. Eur. J. 2008, 14, 3093–3099. [Google Scholar] [CrossRef] [PubMed]
- Balaban, A.T.; Schleyer, P.v.R.; Rzepa, H.S. Crocker, Not Armit and Robinson, Begat the Six Aromatic Electrons. Chem. Rev. 2005, 105, 3436–3447. [Google Scholar] [CrossRef]
- Balaban, A.T.; Klein, D.J. Claromatic Carbon Nanostructures. J. Phys. Chem. C 2009, 113, 19123–19133. [Google Scholar] [CrossRef]
- Fries, K. Über bicyclische Verbindungen und ihren Vergleich mit dem Naphtalin. Justus Liebigs Ann. Chem. 1927, 454, 121–324. [Google Scholar] [CrossRef]
- Graovac, A.; Gutman, I.; Randic, M.; Trinajstic, N. Kekule index for valence bond structures of conjugated polycyclic systems. J. Am. Chem. Soc. 1973, 95, 6267–6273. [Google Scholar] [CrossRef]
- Krygowski, T.M.; Cyrański, M.K. Structural Aspects of Aromaticity. Chem. Rev. 2001, 101, 1385–1419. [Google Scholar] [CrossRef]
- Schleyer, P.v.R.; Maerker, C.; Dransfeld, A.; Jiao, H.J.; Hommes, N.J.R.V. Nucleus-independent chemical shifts: A simple and efficient aromaticity probe. J. Am. Chem. Soc. 1996, 118, 6317–6318. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wannere, C.S.; Corminboeuf, C.; Puchta, R.; Schleyer, P.v.R. Nucleus-Independent Chemical Shifts (NICS) as an Aromaticity Criterion. Chem. Rev. 2005, 105, 3842–3888. [Google Scholar] [CrossRef] [PubMed]
- Ciesielski, A.; Krygowski, T.M.; Cyrański, M.K.; Dobrowolski, M.A.; Aihara, J.-I. Graphtopological approach to magnetic properties of benzenoid hydrocarbons. Phys. Chem. Chem. Phys. 2009, 11, 11447–11445. [Google Scholar] [CrossRef] [PubMed]
- Trinajstić, N. Chemical Graph Theory, 2nd ed.; CRC press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Horn, R.A.; Johnson, C.R. Matrix Analysis; Cambridge University Press: Cambridge, UK, 1985. [Google Scholar]
- Horn, R.A.; Johnson, C.R.; Johnson, C.R. Topics in Matrix Analysis; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).