Machine Learning for Rupture Risk Prediction of Intracranial Aneurysms: Challenging the PHASES Score in Geographically Constrained Areas
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Acquisition
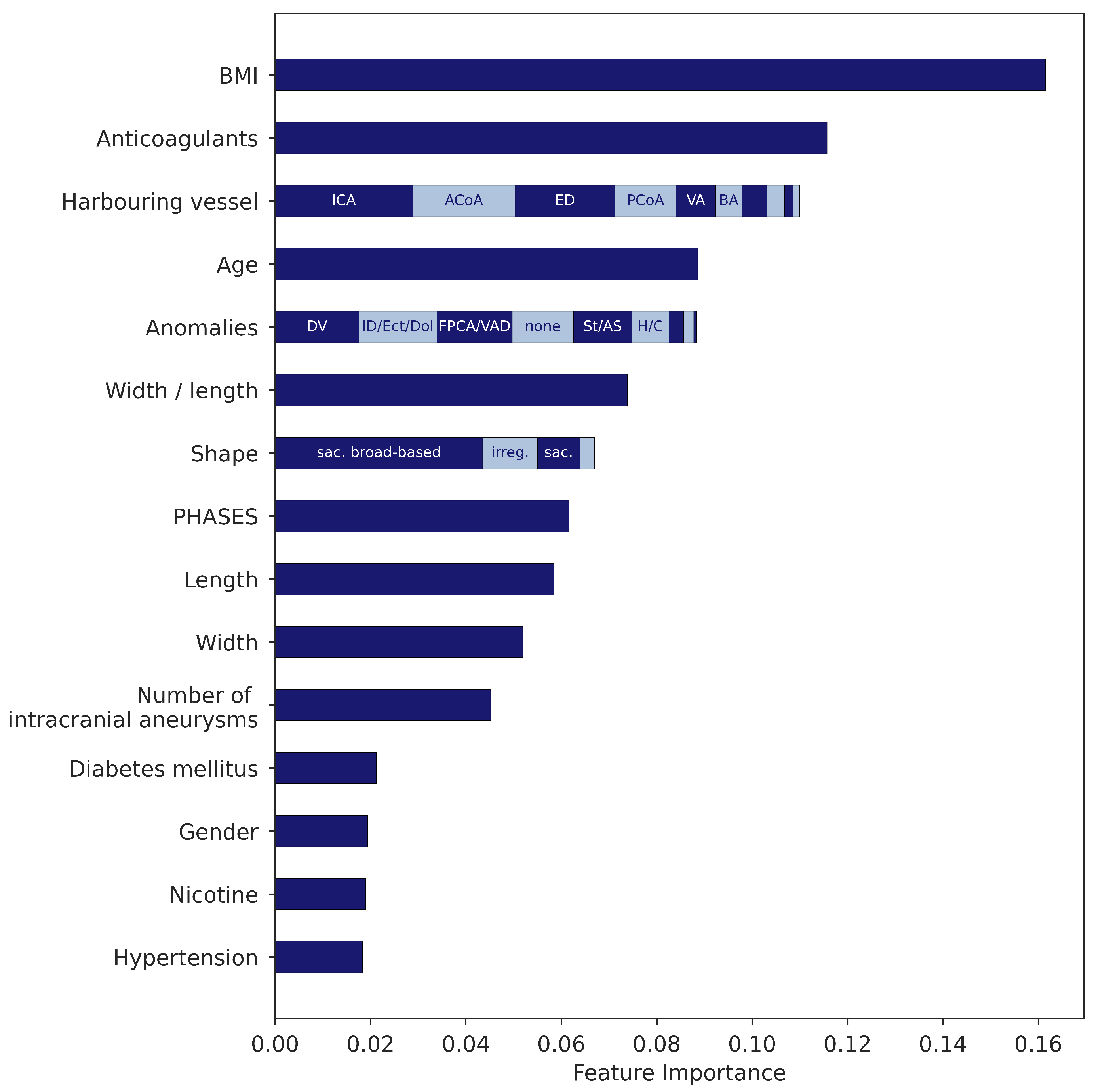
2.2. Clinical Features
2.3. Gradient Boosting Machine
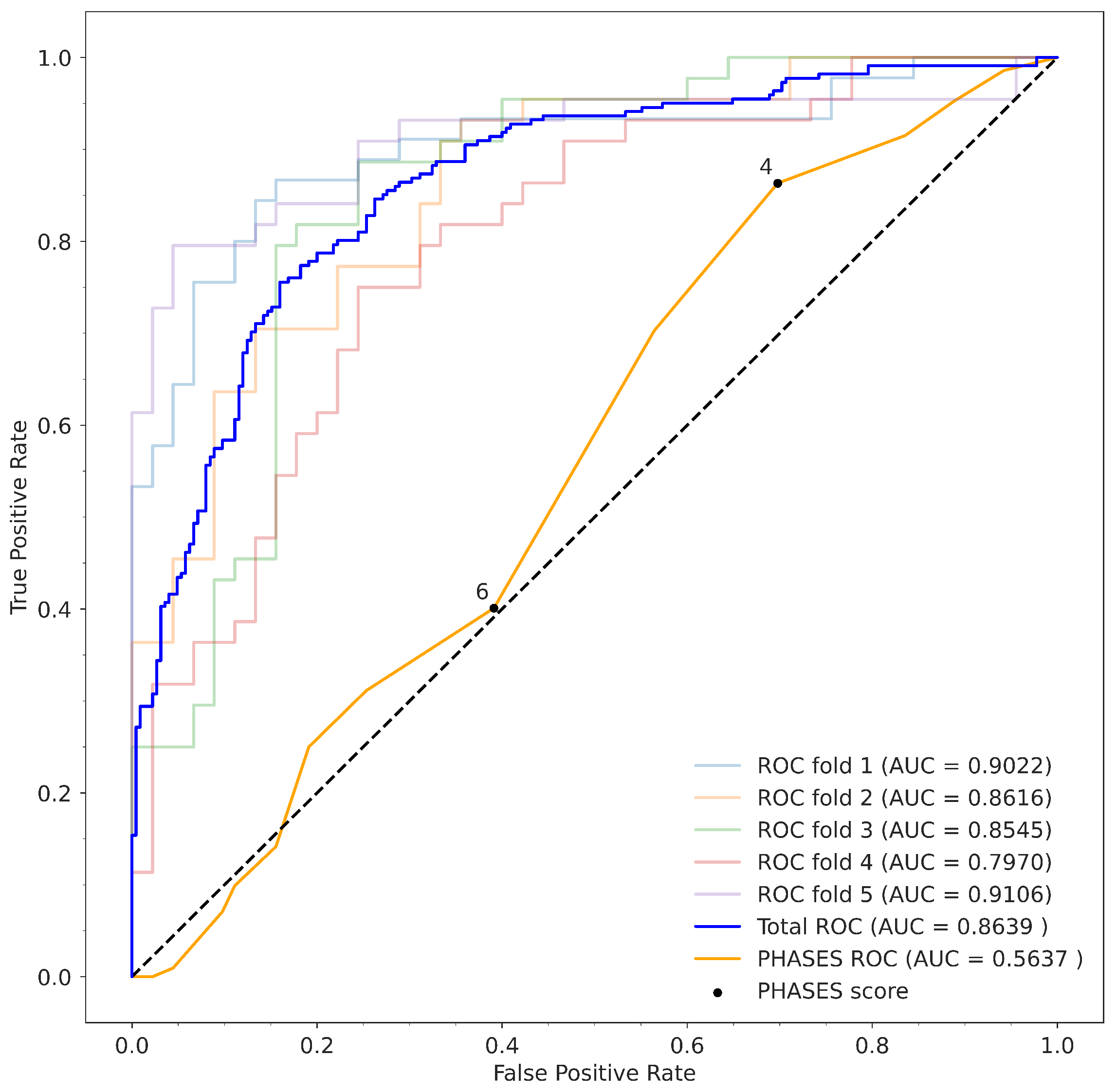
2.4. Evaluation
3. Results
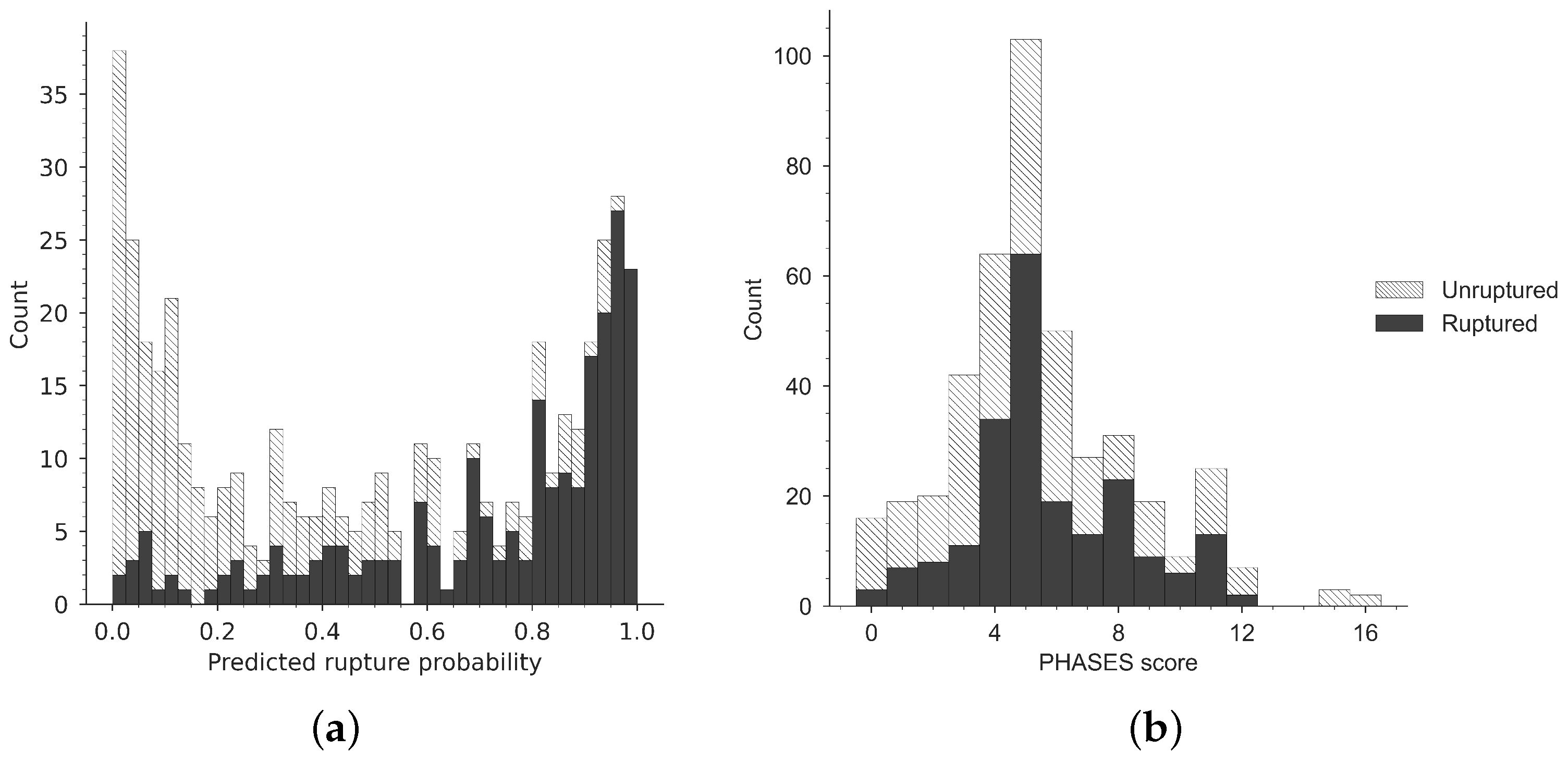
3.1. Gradient Boosting Model
3.2. Comparison to PHASES Score
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| UIA | unruptured intracranial aneurysm |
| SAH | subarachnoid hemorrhage |
| PPV | positive predictive value |
| NPV | negative predictive value |
| TPR | true positive rate |
| TNR | true negative rate |
| ROC | receiver operating characteristic |
| AUC | area under the curve |
| ML | machine learning |
References
- Vlak, M.H.; Algra, A.; Brandenburg, R.; Rinkel, G.J. Prevalence of unruptured intracranial aneurysms, with emphasis on sex, age, comorbidity, country, and time period: A systematic review and meta-analysis. Lancet Neurol. 2011, 10, 626–636. [Google Scholar] [CrossRef]
- Vernooij, M.W.; Ikram, M.A.; Tanghe, H.L.; Vincent, A.J.; Hofman, A.; Krestin, G.P.; Niessen, W.J.; Breteler, M.M.; van der Lugt, A. Incidental Findings on Brain MRI in the General Population. N. Engl. J. Med. 2007, 357, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; van Gelder, J.M. The Probability of Sudden Death from Rupture of Intracranial Aneurysms: A Meta-analysis. Neurosurgery 2002, 51, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Nieuwkamp, D.J.; Setz, L.E.; Algra, A.; Linn, F.H.; de Rooij, N.K.; Rinkel, G.J. Changes in case fatality of aneurysmal subarachnoid haemorrhage over time, according to age, sex, and region: A meta-analysis. Lancet Neurol. 2009, 8, 635–642. [Google Scholar] [CrossRef]
- Darsaut, T.E.; Findlay, J.M.; Magro, E.; Kotowski, M.; Roy, D.; Weill, A.; Bojanowski, M.W.; Chaalala, C.; Iancu, D.; Lesiuk, H.; et al. Surgical clipping or endovascular coiling for unruptured intracranial aneurysms: A pragmatic randomised trial. J. Neurol. Neurosurg. Psychiatry 2017, 88, 663–668. [Google Scholar] [CrossRef]
- Etminan, N.; Rinkel, G.J. Unruptured intracranial aneurysms: Development, rupture and preventive management. Nat. Rev. Neurol. 2016, 12, 699–713. [Google Scholar] [CrossRef]
- Pierot, L.; Barbe, C.; Ferré, J.C.; Cognard, C.; Soize, S.; White, P.; Spelle, L. Patient and aneurysm factors associated with aneurysm rupture in the population of the ARETA study. J. Neuroradiol. 2020, 47, 292–300. [Google Scholar] [CrossRef]
- AlMatter, M.; Bhogal, P.; Pérez, M.A.; Schob, S.; Hellstern, V.; Bäzner, H.; Ganslandt, O.; Henkes, H. The Size of Ruptured Intracranial Aneurysms. Clin. Neuroradiol. 2017, 29, 125–133. [Google Scholar] [CrossRef]
- Greving, J.P.; Wermer, M.J.H.; Brown, R.D.; Morita, A.; Juvela, S.; Yonekura, M.; Ishibashi, T.; Torner, J.C.; Nakayama, T.; Rinkel, G.J.E.; et al. Development of the PHASES score for prediction of risk of rupture of intracranial aneurysms: A pooled analysis of six prospective cohort studies. Lancet Neurol. 2014, 13, 59–66. [Google Scholar] [CrossRef]
- Wende, T.; Kasper, J.; Wilhelmy, F.; Prasse, G.; Quäschling, U.; Haase, A.; Meixensberger, J.; Nestler, U. Comparison of the unruptured intracranial aneurysm treatment score recommendations with clinical treatment results—A series of 322 aneurysms. J. Clin. Neurosci. 2022, 98, 104–108. [Google Scholar] [CrossRef]
- Hernández-Durán, S.; Mielke, D.; Rohde, V.; Malinova, V. Is the unruptured intracranial aneurysm treatment score (UIATS) sensitive enough to detect aneurysms at risk of rupture? Neurosurg. Rev. 2020, 44, 987–993. [Google Scholar] [CrossRef] [Green Version]
- Haase, A.; Schob, S.; Quäschling, U.; Hoffmann, K.T.; Meixensberger, J.; Nestler, U. Epidemiologic and anatomic aspects comparing incidental and ruptured intracranial aneurysms: A single centre experience. J. Clin. Neurosci. 2020, 81, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Hu, B.; Schoepf, U.; Savage, R.; Dargis, D.; Pan, C.; Li, X.; Ni, Q.; Lu, G.; Zhang, L. Artificial Intelligence in the Management of Intracranial Aneurysms: Current Status and Future Perspectives. Am. J. Neuroradiol. 2020, 41, 373–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, M.A.; Patel, J.; Kavouridis, V.; Gallerani, T.; Beers, A.; Chang, K.; Hoebel, K.V.; Brown, J.; See, A.P.; Gormley, W.B.; et al. Machine Learning Models can Detect Aneurysm Rupture and Identify Clinical Features Associated with Rupture. World Neurosurg. 2019, 131, e46–e51. [Google Scholar] [CrossRef]
- Team, T. Pandas development Pandas-Dev/Pandas: Pandas. Zenodo 2020, 21, 1–9. [Google Scholar] [CrossRef]
- McKinney, W. Data Structures for Statistical Computing in Python. In Proceedings of the 9th Python in Science Conference (SciPy 2010), Austin, TX, USA, 28 June–3 July 2010; pp. 56–61. [Google Scholar]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Bijlenga, P.; Gondar, R.; Schilling, S.; Morel, S.; Hirsch, S.; Cuony, J.; Corniola, M.V.; Perren, F.; Rüfenacht, D.; Schaller, K. PHASES Score for the Management of Intracranial Aneurysm. Stroke 2017, 48, 2105–2112. [Google Scholar] [CrossRef] [PubMed]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef]
- Breiman, L. Manual on Setting up, Using, and Understanding Random Forests V3.1; Statistics Department University of California Berkeley: Berkeley, CA, USA, 2002; Volume 1, pp. 3–42. [Google Scholar]
- Breiman, L.; Friedman, J.; Olshen, R.; Stone, C. Classification and Regression Trees, 1st ed.; Chapman & Hall: London, UK, 1984. [Google Scholar]
- Neulen, A.; Pantel, T.; König, J.; Brockmann, M.A.; Ringel, F.; Kantelhardt, S.R. Comparison of Unruptured Intracranial Aneurysm Treatment Score and PHASES Score in Subarachnoid Hemorrhage Patients with Multiple Intracranial Aneurysms. Front. Neurol. 2021, 12, 445. [Google Scholar] [CrossRef]
- Brinjikji, W.; Pereira, V.M.; Khumtong, R.; Kostensky, A.; Tymianski, M.; Krings, T.; Radovanovich, I. PHASES and ELAPSS Scores Are Associated with Aneurysm Growth: A Study of 431 Unruptured Intracranial Aneurysms. World Neurosurg. 2018, 114, e425–e432. [Google Scholar] [CrossRef]
- Backes, D.; Rinkel, G.J.; Greving, J.P.; Velthuis, B.K.; Murayama, Y.; Takao, H.; Ishibashi, T.; Igase, M.; TerBrugge, K.G.; Agid, R.; et al. ELAPSS score for prediction of risk of growth of unruptured intracranial aneurysms. Neurology 2017, 88, 1600–1606. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Chen, G.; Mao, L.; Li, X.; Zhou, C.; Xia, S.; Zhang, Y.; Zhang, B.; Hu, B.; Lu, G.; et al. Machine Learning-Based Prediction of Small Intracranial Aneurysm Rupture Status Using CTA-Derived Hemodynamics: A Multicenter Study. Am. J. Neuroradiol. 2021, 42, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Ou, C.; Liu, J.; Qian, Y.; Chong, W.; Zhang, X.; Liu, W.; Su, H.; Zhang, N.; Zhang, J.; Duan, C.Z.; et al. Rupture Risk Assessment for Cerebral Aneurysm Using Interpretable Machine Learning on Multidimensional Data. Front. Neurol. 2020, 11, 570181. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Jiang, P.; Jiang, Y.; Ge, H.; Li, S.; Jin, H.; Li, Y. Prediction of Aneurysm Stability Using a Machine Learning Model Based on PyRadiomics-Derived Morphological Features. Stroke 2019, 50, 2314–2321. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.C.; Rhim, J.K.; Ahn, J.H.; Park, J.J.; Moon, J.U.; Hong, E.P.; Kim, M.R.; Kim, S.G.; Lee, S.H.; Jeong, J.H.; et al. Machine Learning Application for Rupture Risk Assessment in Small-Sized Intracranial Aneurysm. J. Clin. Med. 2019, 8, 683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heo, J.; Park, S.J.; Kang, S.H.; Oh, C.W.; Bang, J.S.; Kim, T. Prediction of Intracranial Aneurysm Risk using Machine Learning. Sci. Rep. 2020, 10, 6921. [Google Scholar] [CrossRef]
- Zheng, J.; Xu, R.; Sun, X.; Zhang, X. Small vs. Large Unruptured Cerebral Aneurysm: Concerns with the Age of Patient. Front. Neurol. 2021, 12, 735456. [Google Scholar] [CrossRef]
- Chen, S.; Mao, J.; Chen, X.; Li, Z.; Zhu, Z.; Li, Y.; Jiang, Z.; Zhao, W.; Wang, Z.; Zhong, P.; et al. Association Between Body Mass Index and Intracranial Aneurysm Rupture: A Multicenter Retrospective Study. Front. Aging Neurosci. 2021, 13, 515. [Google Scholar] [CrossRef]
- Nembrini, S.; König, I.R.; Wright, M.N. The Revival of the Gini Importance? Bioinformatics 2018, 34, 3711–3718. [Google Scholar] [CrossRef] [Green Version]
- Ohkuma, H.; Tabata, H.; Suzuki, S.; Islam, M.S. Risk Factors for Aneurysmal Subarachnoid Hemorrhage in Aomori, Japan. Stroke 2003, 34, 96–100. [Google Scholar] [CrossRef] [Green Version]
| Hyperparameter | Domain Space | Optimum after Grid Search |
|---|---|---|
| n_estimators | {100, 200, 300, 400, 500, 600, 700, 800, 900, 1000} | 100 |
| learning_rate | {0.1, 0.01, 0.001, 0.0001} | 0.1 |
| criterion | {‘friedman_mse’, ’mse’, ’mae’} | ‘friedman_mse’ |
| max_features | {‘auto’, ‘sqrt’, ‘log2’} | ‘sqrt’ |
| max_depth | {5, 15, 25, 35, 45, 55, 65, 75, 85, 95, None} | 5 |
| min_samples_split | {2, 4, 6, 8, 10} | 2 |
| min_samples_leaf | {2, 4, 6, 8, 10} | 2 |
| (a) Gradient Boosting Model | ||||
|---|---|---|---|---|
| Rupture | ||||
| Positive | Negative | Total | ||
| Rupture prediction | Positive | 191 | 67 | 258 |
| Negative | 30 | 158 | 188 | |
| Total | 221 | 225 | 446 | |
| (b) PHASES Score | ||||
| Rupture | ||||
| Positive | Negative | Total | ||
| Rupture prediction | Positive | 183 | 157 | 340 |
| Negative | 29 | 68 | 97 | |
| Total | 212 | 225 | 437 | |
| Statistical Measure | Model () | PHASES Score |
|---|---|---|
| Accuracy | 0.7825 | 0.5744 |
| F1-Score | 0.7975 | 0.6630 |
| Sensitivity | 0.8643 | 0.8632 |
| Specificity | 0.7022 | 0.3022 |
| PPV | 0.7403 | 0.5382 |
| NPV | 0.8404 | 0.7010 |
| AUC | 0.8639 | 0.5637 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walther, G.; Martin, C.; Haase, A.; Nestler, U.; Schob, S. Machine Learning for Rupture Risk Prediction of Intracranial Aneurysms: Challenging the PHASES Score in Geographically Constrained Areas. Symmetry 2022, 14, 943. https://doi.org/10.3390/sym14050943
Walther G, Martin C, Haase A, Nestler U, Schob S. Machine Learning for Rupture Risk Prediction of Intracranial Aneurysms: Challenging the PHASES Score in Geographically Constrained Areas. Symmetry. 2022; 14(5):943. https://doi.org/10.3390/sym14050943
Chicago/Turabian StyleWalther, Georg, Christian Martin, Amelie Haase, Ulf Nestler, and Stefan Schob. 2022. "Machine Learning for Rupture Risk Prediction of Intracranial Aneurysms: Challenging the PHASES Score in Geographically Constrained Areas" Symmetry 14, no. 5: 943. https://doi.org/10.3390/sym14050943
APA StyleWalther, G., Martin, C., Haase, A., Nestler, U., & Schob, S. (2022). Machine Learning for Rupture Risk Prediction of Intracranial Aneurysms: Challenging the PHASES Score in Geographically Constrained Areas. Symmetry, 14(5), 943. https://doi.org/10.3390/sym14050943







