Abstract
Although hand grip strength is critical to the daily lives of humans and our arboreal great ape relatives, the human hand has changed in form and function throughout our evolution due to terrestrial bipedalism, tool use, and directional asymmetry (DA) such as handedness. Here we investigate how hand form and function interact in modern humans to gain an insight into our evolutionary past. We measured grip strength in a heterogeneous, cross-sectional sample of human participants (n = 662, 17 to 83 years old) to test the potential effects of age, sex, asymmetry (hand dominance and handedness), hand shape, occupation, and practice of sports and musical instruments that involve the hand(s). We found a significant effect of sex and hand dominance on grip strength, but not of handedness, while hand shape and age had a greater influence on female grip strength. Females were significantly weaker with age, but grip strength in females with large hands was less affected than those with long hands. Frequent engagement in hand sports significantly increased grip strength in the non-dominant hand in both sexes, while only males showed a significant effect of occupation, indicating different patterns of hand dominance asymmetries and hand function. These results improve our understanding of the link between form and function in both hands and offer an insight into the evolution of human laterality and dexterity.
1. Introduction
The hand is essential to how modern humans interact with their environment, as it was for our extinct relatives. The enhanced dexterity of the human hand is unique among living primates and is generally considered to have evolved through both (1) adaptation to bipedalism and a relaxation of locomotor selective pressures on the hands and (2) increasingly more complex tool production and use in hominins (i.e., group consisting of modern humans and our closely related extinct relatives) [1,2]. The use of stone tools would have allowed early hominins to access different and potentially higher-quality foods (e.g., marrow) [3,4]. The manufacture and use of even relatively simple stone tools, such as Oldowan technology (2.6–1.7 million years ago) [5,6], would have required both increased cognitive function (e.g., learning, working memory/future thinking, planning and decision-making etc.) [7,8] and particular biomechanical demands on the anatomy of the hands [9,10,11]. Thus, it is likely that tool production and use played a critical role in shaping both cognitive development (e.g., with the crucial role of social learning) [12] and hand morphology. For example, a long, powerful thumb and relatively short fingers facilitate the forceful precision and power-squeeze gripping that are considered to be unique human abilities [1,13]. Although modern humans are also adept as using their hands for locomotion [14,15], the upper limbs are predominantly used for manipulation.
Humans are unique among primates in the strength of population-level hand directional asymmetry (DA) or laterality (i.e., preference for one hand, called the dominant hand, over the other, non-dominant hand), with 85–90% of humans being right-handed regardless of geographical region and ethnicity [16,17,18,19]. Non-human primates also show population laterality for object manipulation, but not with the same strength as that found in humans ([20,21,22], see [23]) and their laterality can also vary depending on the complexity of the manual task (e.g., bimanual manipulative action versus tool use) [24]. Moreover, motor skill biases for tool use in chimpanzees may be supported by anatomically asymmetric, left-biased brain regions analogous to Broca’s and Wernicke’s area in humans [25], brain regions that are both implicated in the perception and production of speech. Handedness (i.e., side preference for the right or the left hand) in humans is thought to have played an important role in the lateralisation of the human brain for language [26] and the emergence of other complex cognitive functions, including tool use [27,28], manual gestures [29,30], and throwing [31]. Greenfield [32] proposed that it was the motor sequencing for tool use—requiring dexterous hierarchical motor activities—that paved a way for the emergence of language that likely emerged first in the form of hand gestures [33,34]. Thus, more dexterous hands may have increased object manipulation capabilities that, in turn, increased hemispheric specialisation and DA, suggesting that the capacities for tool use and language evolved together [32]. However, when population-level handedness first evolved within the hominin clade remains unclear [35,36].
Hand size, shape [37,38,39,40], and bone morphology are also highly variable among recent human populations [41,42]. How this variability potentially affects hand function may provide insights into the evolution of the human hand. For example, ergonomic studies have shown that handle design is important for hand grip performance (e.g., time to complete the task and strength use) [43] and that hand size strongly affects performance [44], indicating the importance of designing tools in accordance with current anthropometric data. Moreover, individuals with relatively longer fingers and therefore larger joint surfaces require less force during stone tool production than those with shorter fingers [45]. Key and Lycett [46] found that through experimental stone tool use, grip strength was the primary contributing biometric factor for stone cutting efficiency. Therefore, both hand shape and hand strength were likely important factors in the efficient stone tool production and use, and would have played an important role in the evolution of hominin cultural technology.
Hand grip strength is commonly measured in a clinical or sports medicine context as an indicator of overall muscle strength [47,48,49,50,51]. Grip strength reflects the gross power of the hand and has been found to be strongly associated with physical activity [52,53,54,55], as well as anthropometric traits, such as age and sex [56,57,58], hand length and shape [59,60], handedness [61], and body mass index [62,63,64]. For example, males typically have a stronger average grip strength than females [56,65]. In both sexes, hand asymmetry in grip strength was found, with the dominant hand (defined as the hand used most within the context of object manipulation) is approximatively 10% stronger than the non-dominant hand [61], although this difference is more pronounced, and is therefore more of a DA, for right-handed individuals than left-handed individuals [66]. Furthermore, hand size has been shown to be positively correlated with grip strength for both sexes [67,68,69,70]. It was also found that hand shape influences grip strength [59,71], such that, for both sexes, people with bigger hands (i.e., large hand length and width) were significantly stronger than people with smaller hands. Moreover, Carlson [72] proposed that, although variation in hand grip strength primarily reflected differences in soft tissue and skeletal morphology, changes in grip strength across the lifespan were also significantly influenced by neural mechanisms (e.g., central nervous system recruiting motoneurons to mediate the control of coordinated movement). Thus, grip strength can be used as a marker of brain health [72]. Indeed, maximum grip strength provides a discriminating measure of cognitive function, such as how central nervous system disorders (e.g., vascular disorders, structural disorders or degeneration) affect the quality of motor coordination [72]. The rate of decline in cognitive function (e.g., motor and perceptual speed, memory, and spatial functioning) has also been shown to correlate with a decline in grip strength, especially towards the end of life [73].
However, most previous studies of grip strength have focused on specific populations [57,74,75,76], occupations and activities [51,52,54], or sex and age [77,78,79] with the aim to better understand health, but these same methods may also be useful for understanding the broader scope of form and function from an evolutionary perspective. Although informative, these studies do not fully capture the potential variability in the key factors that can affect grip strength, particularly hand size, shape, and daily use. To broaden our understanding of the link between hand form and function, this study aims to evaluate the variation of grip strength in a heterogeneous and international group of human adults across the lifespan. We test the potential influence of age, sex, asymmetry in hand dominance and preference (i.e., right- vs. left-hand), hand shape, and lifestyle factors (i.e., occupation, practice of sport and music) on grip strength. In this context, we explore hand asymmetry in grip strength as an indicator of brain/manual lateralisation, with hand dominance (i.e., significant difference between the dominant and the non-dominant hand, without taking into account the left-right direction) and DA (i.e., pattern of bilateral variation observed when one side, right or left, is significantly stronger than the other). Based on previous studies, we predict that (1) males will be significantly stronger than females; (2) younger participants will be stronger than the oldest participants; (3) hand asymmetry will be found, with the dominant hand significantly stronger than the non-dominant hand and that this effect will be stronger for right-handed compared with left-handed individuals indicating DA; (4) participants with wider hands (i.e., a hand wider than it is long) will be significantly stronger than those with smaller or longer hands (a hand longer than it is wide); and (5) participants that regularly practice sport, music, or occupational activities that engage their hands will be stronger than those who do not.
2. Materials and Methods
2.1. Sample
The participants were visitors to an interactive, three-month public engagement and citizen science collaboration project called Me, Human (www.mehuman.io/, accessed on 10 February 2021) hosted by Live Science at the London Science Museum between 02/07/2019 and 31/09/2019. Me, Human consisted of a series of experiments exploring motor-sensory behavioural biases and cognitive ability. Experiments included measurements of grip strength, dexterity, cognitive puzzles, and functional brain laterality, in which participants could engage in as many of the experiments as they wanted. Volunteer participants first completed a baseline demographic questionnaire, including date of birth, sex, and handedness for writing (our hand asymmetry indicator), before engaging in the experiment(s). More than 1600 individuals participated in the Me, Human experiments, of which n = 1286 took part in the ‘Get a grip’ experiment that measured grip strength and hand size, and collected further information about lifestyle and daily activities using the hands. Within this sample, 719 were classified as ‘adults’ between the ages 17–83 years old because the hand is fully developed (i.e., complete fusion of hand bones) by 17 years of age [80,81]. The remainder of the sample (n = 567) were children and adolescents (6–16 year-olds), and were excluded from this study. Of the total adult sample, 57 participants were removed from the data set due to incomplete data or because they had a recorded hand and/or arm injury within one year of the testing date, making the final sample size n = 662. Of the 662 participants, 89.6% (N = 593) self-reported as right-handed, 9.0% (N = 60) left-handed, and 1.4% (N = 9) as ambidextrous. Participants self-reported as ambidextrous were excluded from subsequent analyses, with the remaining sample containing 653 participants (Table 1; Figure S1). Our sample was divided into 10 age categories (Table 1) of five-year intervals, excluding the first (17–19 years) and last (60 years and older) age categories.

Table 1.
Sample used in the analyses with details on the number of participants for each sex by age range and self-reported handedness, type of job, and according to thus practicing a musical instrument and sport.
All participants gave their written informed consent before participating in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Department of Psychological Sciences Ethics Committee at Birkbeck (ref: 181996), University of London.
2.2. Data Collection Procedure
2.2.1. Questionnaire
Participants were asked first if they had a hand or arm injury in the 12 months prior to the test date, and only those participants who answered “no” were allowed to continue the experiment. Participants were then asked several multiple-choice binary questions about their type of occupation, if they regularly played musical instruments (e.g., violin, guitar, piano, saxophone, flute, drums), or engaged in sport using their hands (e.g., rock climbing, bouldering, gymnastics, acrobatics, archery, racket sports, lifting, cricket, golf, hand ball games and bike riding (including commuting to work)). Regarding occupation, participants could choose between (1) office job or work that requires limited manual strength (e.g., typing, shop teller); (2) precision manual work (e.g., jeweller, dressmaker, artist, lab technician); or (3) forceful manual labour (e.g., builder, carpenter, farmer). We considered as “office job/work” students and stay-at-home parents, who would use their hands for a variety of tasks, but none that were specialised enough to be considered “precision” or “forceful” manual labourer. From this questionnaire we created three measurements: music with two levels (yes/no), sport with two levels (yes/no), and occupation with three levels (office/precision/forceful).
2.2.2. Grip Strength
Grip strength was measured using a Jamar dynamometer (Sammons Preston, USA) while the participant was in a seated position. The size of the grip span was adjusted according to the hand size of the participant, visually evaluated by the experimenter, and tested by the participant before they did the grip test. The experimenter first demonstrated the appropriate sitting position and how to hold and use the dynamometer with the elbow bent at 90 degrees, as recommended by the American Society of Hand Therapy (ASHT) [82]. A poster demonstrating the appropriate posture and arm/hand position was also visible to the participant (Figure 1A). The participant was asked to squeeze the dynamometer to their maximum ability for two seconds. Grip strength was measured (lbs) for the left and right hand and after a rest of approximately one minute, each hand was measured again. An average of both measures for each hand was used in the analysis. All staff and volunteers of the Me, Human project were trained to measure grip strength with the same protocol to limit measurement error.
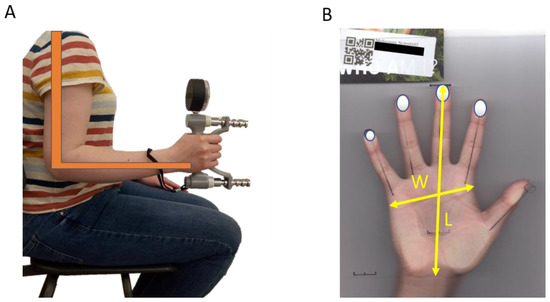
Figure 1.
Appropriate posture and arm/hand position for the grip strength on the poster showed to participant (A) and the measurements taken on hand scans (B). W = hand width, L = hand length.
2.2.3. Hand Size and Shape
We measured hand size and shape from scans using a flatbed scanner (Epson Perfection V39). Participants were asked to place each hand palm side down, lining up their fingers to fit within an outline drawn on a transparent plastic sheet and keeping the fingers and palm flat (Figure 1B). Two differently sized outline transparencies for intermediate and large hands were available to allow a participant to best align their hand in a standardised manner (Figure 2). A 2 cm scale was included in each transparent plastic sheet to facilitate the accurate measurement of hand size from the scans. Hand size and shape were measured from each scan using freeware tpsDig2 software version 2.31 [83]. The hand width (W) was measured from the radial side of the second metacarpal joint to the ulnar side of the fifth metacarpal joint and hand length (L) from midline of the distal wrist crease to the tip of the middle finger (Figure 1B), following [59]. A ratio of hand width to length (W/L) was used as an indicator of hand shape, such as in [59]. We denoted hands with a ratio >0.5 as ‘wide’ hands and hands with a ratio <0.5 as ‘long’ hands. To correct the potential effect of hand size on grip strength, W*L was used as an estimate of hand area to quantify the relative grip strength (i.e., grip strength/hand area). All measurements were taken by one researcher (KT) and to test interobserver error, 20% of both right and left hands (n = 266) were measured by a second researcher (AB). An intraclass correlation coefficient (ICC) was used to test for interobserver reliability in R (R Core Team, 2020) with the “irr” package [84]. Both measurements of right and left hands were consistent between the two observers (ICC = 0.981, p < 0.0001), indicating an excellent reproducibility and repeatability of the measurements. Therefore, we only used the measurements of the first researcher for all subsequent analyses.
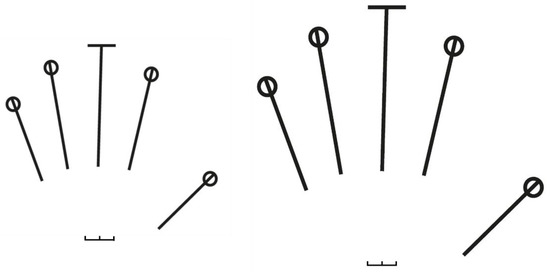
Figure 2.
The two different sizes of outline transparencies (intermediate to the left and for large hands to the right) used on the flatbed scanner to allow the participant to best align their hand in a standardised manner. The scale of 2 cm was placed in the middle of the palm.
2.3. Statistical Analyses
Shapiro–Wilk Normality tests (p > 0.05) revealed that all data were normally distributed. We used an ANOVA to test our prediction that males would be stronger than females and that the dominant hand (defined here as the hand used to write) would be significantly stronger than the non-dominant hand (dominant hand asymmetry) using, first, absolute grip strength and, second, relative grip strength (i.e., grip strength/hand area). A Levene’s test was performed to test the homogeneity of variance between males and females and for both hands. An ANOVA was also used to assess the difference in absolute grip strength between both hands across age categories within (1) males, (2) females, and (3) right and left-handers (sexes pooled) (DA).
Next, we fitted four linear multiple regressions to predict the four outcome measures of: (1) male dominant hand, (2) male non-dominant hand, (3) female dominant hand, (4) female non-dominant hand. Our predictor variables were age, occupation, hand shape, hand preference, playing music, and playing sport. These six predictor variables were considered as fixed effects and grip strength was considered a random effect. The function “predictorEffects” from the package “effect” [85] was used to graphically represent the model effects. An ANOVA was performed for each model to statistically test the effect of the predictor variables on grip strength. Tukey corrections were used for post-hoc analyses. All tests were performed with R 3.6.3 [86] with level of significance set at p ≤ 0.05.
3. Results
First, we tested whether there were differences in grip strength between males and females and, for each sex, between both hands to test the effect of hand dominance asymmetry using ANOVAs. We investigated potential differences in both absolute grip strength and relative grip strength, in which hand area was used as a proxy for size. The results of the ANOVA showed that males were significantly stronger than females for both absolute grip strength (F(1, 1303) = 1782.72, p < 0.0001, η2 = 0.58) (Table 2 and Figure 3) and relative grip strength (F(1, 1303) = 820.36, p < 0.0001, η2 = 0.39), and both males and females were significantly stronger with their dominant hand compared with their non-dominant hand (grip strength, F(1, 1303) = 16.16, p < 0.0001, η2 = 0.005; relative grip strength, F(1, 1303) = 16.88, p < 0.0001, η2 = 0.007), indicating dominant hand asymmetry. When looking at the strength of this asymmetry, we found a mean difference in grip strength between the two hands of 5.5% for males, ranging between 1.9% (35–39 years old) and 11.4% (55–59 years old), and 4.2% for females, ranging between 0.4% (40–44 years old) and 8.8% (60 years and older) (Table S1). As a result, we searched for possible differences in the strength of hand dominance asymmetry across ages, and we found no significant interaction between age category and grip strength difference between dominant and non-dominant hands for either males (F(9, 494) = 0.134, p > 0.05) or females (F(9, 772) = 0.207, p > 0.05) (Table S1). We tested for the homogeneity of variance between males and females, and for both hands, males showed significantly more variation than females across all age categories (F(3, 1302) = 41.822, p < 0.0001, η2 = 0.09).

Table 2.
Summary statistics for grip strength (Ibs) of the dominant and non-dominant hand according to the sex and the age categories.
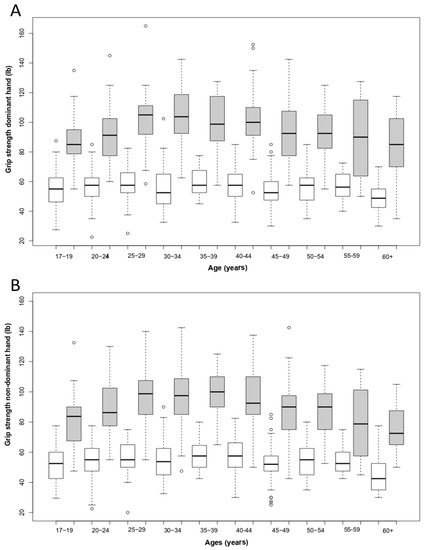
Figure 3.
Grip strength (lbs) performance in males (grey) and females (white) for the dominant hand (A) and the non-dominant hand (B). The boxplots show medians (solid line) and interquartile ranges of grip strength according to age groups. The dotted lines indicate variability outside the upper and lower quartiles, except for “outliers” (dots).
We then tested differences in handedness, as an indicator of DA, in absolute grip strength (sexes pooled). There were no significant differences in grip strength between right- and left-handed participants for either hand (F(1, 1302) = 0.180, p > 0.05), although left-handed individuals had, on average, a higher hand dominance asymmetry with a stronger difference in grip strength between their dominant and non-dominant hands (males = 8.2%, females = 6.4%) compared with right-handed individuals (males = 5.2%, females = 4.0%). No significant interaction was found between age category, handedness and grip strength difference between dominant and non-dominant hands (F(9, 1266) = 0.028, p > 0.05) (Table S1).
We tested for the effect of the predictor variables (age, occupation, hand shape, hand preference, playing music, and playing sport) on grip strength using Fitting linear models. Results revealed that male grip strength of the dominant hand showed a trend towards being affected by hand shape (Figure 4) but not significantly so (F(1, 249) = 3.562, p = 0.06). However, for the non-dominant hand, male grip strength was significantly affected by hand shape (F(1, 249) = 9.489, p < 0.01, η2 = 0.034), such that males with wider hands were stronger than males with longer hands (Figure 4). Grip strength for the non-dominant hand was also significantly affected by occupation (F(2, 249) = 5.278, p < 0.01, η2 = 0.038), such that males doing forceful manual labour were significantly stronger than males doing an office job (post-hoc analyses, p < 0.05), and males who practiced sports (F(1, 249) = 4.125, p < 0.05, η2 = 0.015) were stronger than males who did not (Figure 4).
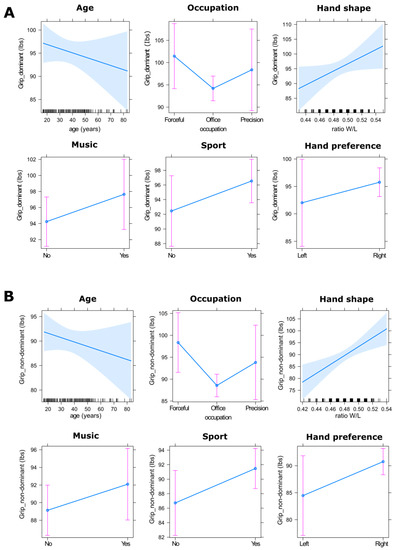
Figure 4.
Predictor effect plots for the fitting linear models in males for the dominant hand (A) and non-dominant hand (B). On the age and hand shape graphics, the blue shaded area is a pointwise confidence band for the fitted values at a level of confidence of 95%. The rug plots at the bottom of the graphs shows the location of the age values and the ratio W/L values. On the other graphics, the pink bars represent the confidence intervals at a level of 95%.
For females, linear modelling revealed that grip strength of the dominant hand was significantly affected by hand shape (F(1, 388) = 4.733, p < 0.05, η2 = 0.017), with females with wider hands being stronger than females with longer hands, and by age (F(1, 388) = 5.369, p < 0.05, η2 = 0.013), such that younger females (~<30 age) were significantly stronger than older females (~>50 age; Figure 5). For the non-dominant hand, female grip strength was also significantly affected by hand shape (F(1, 388) = 5.891, p < 0.02, η2 = 0.014) and by age (F(1, 388) = 4.463, p < 0.05, η2 = 0.011), following the same pattern as the dominant hand. However, females practicing hand sports also had a significantly stronger non-dominant hand (F(1, 388) = 4.858, p < 0.05, η2 = 0.012) than females who did not (Figure 5). Given the effect of age on female grip strength, we tested which factors potentially interacted with age. Linear modelling revealed that for both hands, a significant interaction was found for grip strength between age (continuous) and hand shape (dominant hand, F(1, 388) = 4.123, p < 0.05, η2 = 0.010; non-dominant hand, F(1, 388) = 6.092, p < 0.05, η2 = 0.015). Moreover, while younger females (~<30 age) showed a similar grip strength regardless of differences in hand shape, older females (~>50 age) with wider hands were stronger than older females with longer hands (Figure S2).
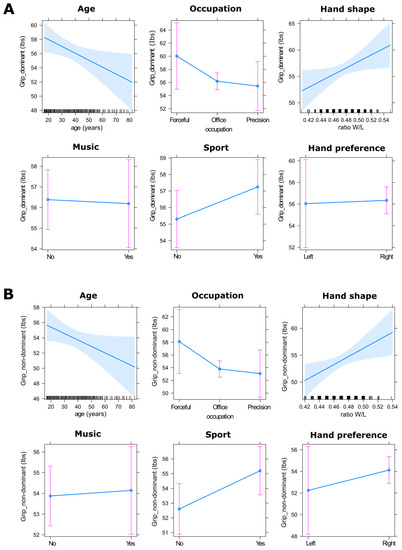
Figure 5.
Predictor effect plots for the fitting linear models in females for the dominant hand (A) and non-dominant hand (B). On the age and hand shape graphics, the blue shaded area is a pointwise confidence band for the fitted values at a level of confidence of 95%. The rug plots at the bottom of the graphs shows the location of the age values and the ratio W/L values. On the other graphics, the pink bars represent the confidence intervals at a level of 95%.
4. Discussion
This study investigated different factors that predict hand grip strength in a large, heterogenous adult human sample. While some of the results support the findings of previous studies, our study sheds new light on the variability in grip strength relative to sex, age, hand shape, hand dominance asymmetry (i.e., laterality) and daily hand use. We discuss these results below and their implications for understanding the evolution of the human hand.
Consistent with findings from previous studies [78,79] and our predictions, we found that males were stronger than females. However, our study also investigated relative grip strength and found that males remained significantly stronger than females even when accounting for variation in hand size. This result is consistent with Leyk et al. [62], who showed that untrained males were, on average, stronger than highly trained female athletes. We also found a significant effect of hand dominance asymmetry, in which the dominant hand was significantly stronger than the non-dominant hand in both sexes, with an average difference being slightly higher for males (5.5%) than for females (4.2%) across all age categories. These mean differences were lower than the reported average of 10% higher grip strength for the dominant hand compared with the non-dominant hand for both sexes reported in previous studies [61,74,87]. Interestingly, we found that the difference in hand strength between the hands seemed to vary with age in both sexes (Table S1), but the differences were not significant. This result could be due to the uneven distributions of the sample across the age categories (e.g., more participants in the 20–24 age category than the 60 years and older), but also requires further investigation through a larger study to examine this general premise of 10% difference in grip strength between the dominant and nondominant hands by examining different age categories.
We also found that the grip strength of both hands decreased significantly with age in females but not in males, which partially supported our expectations. Previous studies have shown an effect of age on grip strength for both sexes [77,78,79]. The non-significant effect of age for both hands in males in our study may reflect the greater variability in grip strength (Figure 3). Middle-aged participants (ages 35–39 for males and 40–44 for females) showed more hand symmetry with limited differences in grip strength between both hands, while younger and older individuals showed greater asymmetry in hand dominance. Although previous studies have shown that laterality decreased with age [88], this was not the case for grip strength in our male sample. The dominant hand was always significantly stronger than the non-dominant hand for both sexes, even for participants in older age categories (Table S1). One reason why this might be the case is that previous studies have typically only assessed differences in the average grip strength across all ages [61,89,90], and thus we demonstrate for the first time, to our knowledge, important grip strength variation at specific life stages for both sexes.
Our results showed that grip strength in our sample was not an indicator of handedness. We did not find a significant effect of self-reported hand preference, although left-handed participants tended to show a larger difference of grip strength between the dominant and non-dominant hand (males = 8.2%, females = 6.4%) compared with right-handed participants (males = 5.2%, females = 4.0%; Table S1). This result did not support our expectation or previous research showing more symmetry in grip strength between the hands in left-handed compared to right-handed people showing more DA [66,75,91]. However, previous work has yielded mixed results, with some studies finding that left-handed individuals had a relatively stronger non-dominant hand [92,93]. The results of our study (and previous research) may be biased by differences in sample size (n = 60 left-handed vs. n = 594 right-handed participants), given the much lower proportion of left-handed individuals across human populations [16,17,18,19]. Left-handed individuals may also be expected to show more symmetry in grip strength between both hands (i.e., a relatively stronger right hand), because the world is adapted for right-handed individuals. Thus, our results are somewhat unexpected and require further investigation through a larger study of left-handed people across their lifespan to better understand the potential differences in grip strength between right- and left-handed individuals.
We found that grip strength of the non-dominant hand was also significantly influenced by hand shape in both males and females, and in both hands for females only. Participants with wider hands were stronger than participants with longer hands, which is consistent with previous studies that reported that people with wider hands tend to have greater muscular strength (when controlling for height) [67,68,69]. The fact that hand shape did not significantly influence grip strength of the dominant hand in males may also reflect the greater variability in grip strength for males compared with females. Interestingly, we found that the effect of hand shape is stronger for older females (~>50 age) than younger females (~<30 age), with older females with wider hands being stronger than older females with longer hands. This variation by age may reflect younger females being, on average, more active than older females and potentially using both hands more frequently and/or with more muscular force during a variety of daily activities. In contrast, older females are more likely to develop osteoarthritis within the hand [94,95], and patients with this disease show weaker grip strength in the affected hand than healthy individuals [96]. As humans with longer digits appear to have relatively larger articular areas [45], females with longer digits could be less susceptible to osteoarthritis, and thus could show less reduction in hand strength than females with shorter fingers. Additional research is needed to investigate the potential effect of hand shape on grip strength in older females and the potential clinical implications of this.
Variation in hand dominance asymmetry and hand function was observed according to the lifestyle factors (i.e., occupation, practice of sport and music). We found that the type of occupation had a significant effect on grip strength for males but not for females, which is consistent with the findings of Hossain et al. [76]. The female result could be explained by the relatively fewer number of female participants doing, for example, forceful manual labour (22 females against 36 males), which potentially could have affected the analysis. In particular, we found that males engaging in forceful manual work were significantly stronger than those doing ‘office work’. This result supports that of previous studies [52,54] (but see [76]). However, we found an effect only for the non-dominant hand. This result likely reflects the fact that manual labour occupations often involve using both hands more forcefully and frequently than office work does, thus increasing muscle strength [54]. Indeed, middle-aged males doing forceful manual work showed greater similarities in grip strength between the dominant and non-dominant hand compared to office workers (Table S1). We found similar results for the practice of manual sports, which significantly affected grip strength in the non-dominant hand for both sexes, while there was no effect from practicing a musical instrument. Together, these results suggest that middle-aged individuals practicing regular manual activities that require the forceful use of both hands have less strength difference between the two hands (i.e., greater symmetry), while office and precision workers, who are doing more fine motor manipulation and using general tools more often with their dominant hand, may have a greater asymmetry in grip strength between the two hands.
Our findings have interesting implications for the study of human evolution. Both hands are important for modern human daily activities; however, experimental studies have demonstrated the importance of having two strong upper limbs, and hands in particular, for prehistoric activities, such as tool production/use behaviours [1,9,10,11,97,98,99], carrying [100], hunting, picking fruit, or dismembering an animal carcass [101]. For some hominins, powerful grip strength in both hands would be critical for climbing as well [102,103,104]. Thus, there would likely be negative selection for having weak hand grip strength throughout hominin evolution [105,106,107]. We found a significant influence of hand shape on grip strength such that individuals with wider hands were significantly stronger than those with longer hands. If this relationship held true in the past, there may have been increased selection for relatively shorter fingers and proportionally wider hands. Indeed, fossil evidence demonstrates that hand proportions have changed throughout human evolution (e.g., [103,108]) and that these changes likely improved dexterity [109,110,111] and potentially grip strength [112,113].
In modern hunter-gatherer populations, greater grip strength in the dominant hand is associated with better hunting outcomes among Hadza males, but not for Yali males [114]. It would be interesting to also measure the strength of the non-dominant hand in hunter-gatherer populations to test the hypothesis of the importance of high grip strength in both hands for hunting and other manual activities. An increase in sedentism among recent (non-foraging) humans correlates with a decrease in cortical bone strength (reviewed in [115]) and a reduction in trabecular bone density [116,117,118] throughout the skeleton, including the hands [116,117]. Given bone’s ability to reflect variation in loading throughout life via (re-)modelling, this research suggests that recent, more sedentary humans have a reduced level of forceful manual activity compared with that of hunter-gatherers and/or that increased sedentism has resulted in systemic changes to bone structure throughout the skeleton.
It is possible that the population-level hand asymmetry or bias well-documented in modern human populations, inferred in Neandertals [28,36] and potentially earlier Homo species [119], is related to advances in technological and cultural innovations [28,120] or, more generally, to task complexity [24,119,121]. In turn, more frequent use of a dominant hand, rather than both hands, for diverse activities could have favoured an increase in hemispheric specialisation and vice versa. However, early hominins (e.g., Australopithecus, Homo habilis), and particularly those that likely still used their hands for locomotion, may have been under stronger selection for bimanual manipulative ability and grip strength, such as in extant great apes [119]. Thus, it is important to investigate both hands in studies of grip strength and laterality to provide a broader evolutionary understanding. In their research on lateralisation through prehistorical tools, Steele and Uomini [122] also highlighted the importance of studying the roles of both hands during bimanual activities (i.e., what they call a “Complementary Role Differentiation” model). We also require a greater appreciation of the effect of lateralisation of specific behaviours on upper limb and hand bone morphology [123,124,125,126,127] to better understand the evolution of human dexterity related to strength of laterality.
There are some limitations to this study which should be considered. As these data were collected as part of the larger Me, Human project and within the rules of the Live Science scheme of the London Science Museum, we were limited to a specific amount of time in which we could keep participants at any one experimental station. As a result, we were not able to collect more detailed data on daily hand use (e.g., the number of musical instruments or specific sports played and for how long). Moreover, although participants were not selected and came voluntarily to the Live Science experiment, potential selection bias could be present and may have affected our results. First, we had more young participants and parents accompanying their children compared to participants from 55 years and older. Moreover, the experiments took place during the summer months when the museum was more likely to attract international visitors. We did not record the ethnicity of the participants while previous, more targeted, studies have shown variation in grip strength across different populations [57,74,75,76]. Future research on large and more diverse groups of humans providing greater detail on ethnicity and specific hand use activities may provide a more nuanced understanding of the links between performance (e.g., grip strength), hand asymmetry, and hand shape or how variation in hand shape and size impacts dexterity.
5. Conclusions
To conclude, we found that adult human grip strength was influenced by a variety of factors, including age, sex, hand shape, and hand dominance asymmetry (i.e., laterality), consistent with previous studies. We also demonstrated for the first time that (1) grip strength varies throughout the lifespan, with more pronounced differences at specific life stages and (2) that the practice of different manual activities through occupation and sport also influence grip strength, particularly in males. These results emphasise the importance of physical manual activities for the attenuation of age-related grip strength loss in a clinical context. These findings may also inform ergonomic research on modern anthropotechnical systems that rely on grip strength data [128,129]. Our results highlight the importance of studying the grip strength of both hands, rather than just the dominant hand, in relation to the above factors, to better understand the link between form and function of the hand, in both modern populations and in our evolutionary past.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/sym13071142/s1, Figure S1: Distribution of participants used in the analyses according to ages groups and with right-handed (R) and left-handed (L) participants, Figure S2: Predictor effect plots for the fitting linear models of the interaction between age and hand shape for grip strength in female, for the dominant hand (A) and the non-dominant hand (B), Table S1: Details of the difference in percentage between hand grip strength in dominant and non-dominant hands for each sex (M = males; F = females) and age groups, according to right and left-handers, practicing of sport and the different occupations.
Author Contributions
Conception and Design of the research, G.S.F., G.D., T.E., T.L.K., A.B., H.B., C.S.; Data Acquisition, G.S.F., G.D., T.E., T.L.K., A.B., H.B., C.S., K.T.; Analysis, A.B., K.T.; Writing—Original Draft Preparation, A.B.; Writing—Review and Editing, A.B., T.L.K., G.S.F., G.D., T.E., H.B., C.S.; Project Administration, G.S.F.; Funding Acquisition, G.S.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by The Waterloo Foundation, grant number 917-3756.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Department of Psychological Sciences Ethics Committee at Birkbeck (ref: 181996, 14/05/2019), University of London.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Acknowledgments
We thank the London Science Museum and the Live Science scheme for allowing us to conduct this study, and the visitors who agreed to be part of the citizen science collaboration project called Me, Human. We thank the volunteers and research assistants: M. Ali, M. Diletta, R. Filippi, L. Fish, E. Forrester, S. Forrester, A. Ghanemi, C. Grevel, J. Hall, E. Jackson, C. Marsh, B. Meyer, S. Pem, E. Periche, E. Ranzato, Y. Slaveva, B. Stewart, and B. Todd for their help in collecting participant data. The authors thank the reviewers for their helpful comments, and Antoine Balzeau for the invitation to submit our work in the special issues “Symmetry in Human Evolution, From Biology to Behaviours”.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Marzke, M.W.; Wullstein, K.L.; Viegas, S.F. Evolution of the power (squeeze) grip and its morphological correlates in hominids. Am. J. Phys. Anthropol. 1992, 89, 283–298. [Google Scholar] [CrossRef]
- Marzke, M.W. Tool making, hand morphology and fossil hominins. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2013, 368, 20120414. [Google Scholar] [CrossRef]
- Toth, N.; Schick, K. Early stone industries and inferences regarding language and cognition. In Tools, Language and Cognition in Human Evolution; Gibson, K.R., Ingold, T., Eds.; Cambridge University Press: Cambridge, UK, 1993; pp. 346–362. [Google Scholar]
- McPherron, S.P.; Alemseged, Z.; Marean, C.W.; Wynn, J.G.; Reed, D.; Geraads, D.; Bobe, R.; Béarat, H.A. Evidence for stone-tool-assisted consumption of animal tissues before 3.39 million years ago at Dikika, Ethiopia. Nature 2010, 466, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Toth, N.; Schick, K. The Oldowan: The tool making of early hominins and chimpanzees compared. Annu. Rev. Anthropol. 2009, 38, 289–305. [Google Scholar] [CrossRef]
- Braun, D.R.; Aldeias, V.; Archer, W.; Arrowsmith, J.R.; Baraki, N.; Campisano, C.J.; Deino, A.L.; DiMaggio, E.N.; Dupont-Nivet, G.; Engda, B.; et al. Earliest known Oldowan artifacts at> 2.58 Ma from Ledi-Geraru, Ethiopia, highlight early technological diversity. Proc. Natl. Acad. Sci. USA 2019, 116, 11712–11717. [Google Scholar] [CrossRef]
- Stout, D.; Toth, N.; Schick, K.; Chaminade, T. Neural correlates of Early Stone Age toolmaking: Technology, language and cognition in human evolution. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 1939–1949. [Google Scholar] [CrossRef]
- Toth, N.; Schick, K. An overview of the cognitive implications of the Oldowan Industrial Complex. Azania Archaeol. Res. Afr. 2018, 53, 3–39. [Google Scholar] [CrossRef]
- Williams, E.M.S.; Gordon, A.D.; Richmond, B.G. Upper limb kinematics and the role of the wrist during stone tool production. Am. J. Phys. Anthropol. 2010, 43, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.M.S.; Gordon, A.D.; Richmond, B.G. Hand pressure distribution during Oldowan stone tool production. J. Hum. Evol. 2012, 62, 520–532. [Google Scholar] [CrossRef]
- Williams-Hatala, E.M.; Hatala, K.G.; Gordon, M.; Key, A.; Kasper, M.; Kivell, T.L. The manual pressures of stone tool behaviors and their implications for the evolution of the human hand. J. Hum. Evol. 2018, 119, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Stout, D.; Hecht, E.E. Evolutionary neuroscience of cumulative culture. Proc. Natl. Acad. Sci. USA 2017, 114, 7861–7868. [Google Scholar] [CrossRef] [PubMed]
- Feix, T.; Romero, J.; Schmiedmayer, H.B.; Dollar, A.M.; Kragic, D. The grasp taxonomy of human grasp types. IEEE Trans. Hum. Mach. Syst. 2015, 46, 66–77. [Google Scholar] [CrossRef]
- Venkataraman, V.V.; Kraft, T.S.; Dominy, N.J. Tree climbing and human evolution. Proc. Natl. Acad. Sci. USA 2013, 110, 1237–1242. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, B.; Kukla, M.; Warguła, Ł. The symmetric nature of the position distribution of the human body center of gravity during propelling manual wheelchairs with innovative propulsion systems. Symmetry 2021, 13, 154. [Google Scholar] [CrossRef]
- Annett, M. Left, Right, Hand and Brain: The Right Shift Theory; LEA Publishers: London, UK, 1985; 416p. [Google Scholar]
- Porac, C.; Coren, S. Lateral Preferences and Human Behavior; Springer: New York, NY, USA, 1981; 283p. [Google Scholar]
- McManus, I.C. The history and geography of human handedness. In Language Lateralization and Psychosis; Sommer, I.E.C., Kahn, R.S., Eds.; Cambridge University Press: Cambridge, UK, 2009; pp. 37–57. [Google Scholar]
- Llaurens, V.; Raymond, M.; Faurie, C. Why are some people left-handed? An evolutionary perspective. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 881–894. [Google Scholar] [CrossRef] [PubMed]
- Forrester, G.S.; Quaresmini, C.; Leavens, D.A.; Mareschal, D.; Thomas, M.S.C. Human handedness: An inherited evolutionary trait. Behav. Brain Res. 2013, 237, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Forrester, G.S. Hand, limb and other motor preferences: Methodological Considerations. In Lateralized Brain Functions; Vallortigara, G., Rogers, L., Eds.; Humana Press: New York, NY, USA, 2017; pp. 121–152. [Google Scholar]
- Hopkins, W.D.; Phillips, K.A.; Bania, A.; Calcutt, S.E.; Gardner, M.; Russell, J.; Schaeffer, J.; Lonsdorf, E.V.; Ross, S.R.; Schapiro, S.J. Hand preferences for coordinated bimanual actions in 777 great apes: Implications for the evolution of handedness in hominins. J. Hum. Evol. 2011, 60, 605–611. [Google Scholar] [CrossRef]
- Papademetriou, E.; Sheu, C.F.; Michel, G.F. A meta-analysis of primate hand preferences, particularly for reaching. J. Comp. Psychol. 2005, 119, 33–48. [Google Scholar] [CrossRef]
- Bardo, A.; Pouydebat, E.; Meunier, H. Do bimanual coordination, tool use, and body posture contribute equally to hand preferences in bonobos? J. Hum. Evol. 2015, 82, 159–169. [Google Scholar] [CrossRef]
- Hopkins, W.D.; Russell, J.L.; Cantalupo, C. Neuroanatomical correlates of handedness for tool use in chimpanzees (Pan troglodytes): Implication for theories on the evolution of language. Psychol. Sci. 2007, 18, 971–977. [Google Scholar] [CrossRef]
- Corballis, M.C. Cerebral asymmetry: Motoring on. Trends Cogn. Sci. 1998, 2, 152–157. [Google Scholar] [CrossRef]
- Gibson, K.R.; Ingold, T. Tools, Language and Cognition in Human Evolution; Cambridge University Press: Cambridge, UK, 1993; 496p. [Google Scholar]
- Uomini, N.T. The prehistory of handedness: Archaeological data and comparative ethology. J. Hum. Evol. 2009, 57, 411–419. [Google Scholar] [CrossRef]
- Corballis, M.C. From mouth to hand: Gesture, speech and the evolution of right- handedness. Behav. Brain Sci. 2003, 26, 199–260. [Google Scholar] [CrossRef]
- Meguerditchian, A.; Vauclair, J.; Hopkins, W.D. On the origins of human handedness and language: A comparative review of hand preferences for bimanual coordinated actions and gestural communication in nonhuman primates. Dev. Psychobiol. 2013, 55, 637–650. [Google Scholar] [CrossRef]
- Calvin, W.H. A stone’s throw and its launch window: Timing precision and its implication for language and hominids brain. J. Theor. Biol. 1983, 104, 121–135. [Google Scholar] [CrossRef]
- Greenfield, P.M. Language, tools and brain: The ontogeny and phylogeny of hierarchically organized sequential behavior. Behav. Brain Sci. 1991, 14, 531–595. [Google Scholar] [CrossRef]
- Bradshaw, J.L.; Nettleton, N.C. Language lateralization to the dominant hemisphere: Tool use, gesture and language in hominid evolution. Curr. Psychol. 1982, 2, 171–192. [Google Scholar] [CrossRef]
- Armstrong, D.F.; Stokoe, W.C.; Wilcox, S.E. Gesture and the Nature of Language; Cambridge University Press: Cambridge, UK, 1995; 270p. [Google Scholar]
- Uomini, N.T. Handedness in Neanderthals. In Neanderthal Lifeways, Subsistence and Technology; Conard, N.J., Richter, J., Eds.; Springer: Dordrecht, Netherlands, 2011; pp. 139–154. [Google Scholar]
- Estalrrich, A.; Rosas, A. Handedness in Neandertals from the El Sidrón (Asturias, Spain): Evidence from instrumental striations with ontogenetic inferences. PLoS ONE 2013, 8, e62797. [Google Scholar] [CrossRef] [PubMed]
- Courtney, A.J. Hand anthropometry of Hong Kong Chinese females compared to other ethnic groups. Ergonomics 1984, 27, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Okunribido, O.O. A survey of hand anthropometry of female rural farm workers in Ibadan, Western Nigeria. Ergonomics 2000, 43, 282–292. [Google Scholar] [CrossRef]
- Kar, S.K.; Ghosh, S.; Manna, I.; Banerjee, S.; Dhara, P. An investigation of hand anthropometry of agricultural workers. J. Hum. Ecol. 2003, 14, 57–62. [Google Scholar] [CrossRef]
- Imrhan, S.N.; Sarder, M.D.; Mandahawi, N. Hand anthropometry in Bangladeshis living in America and comparisons with other populations. Ergonomics 2009, 52, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Lazenby, R.; Smashnuk, A. Osteometric variation in the Inuit second metacarpal: A test of Allen’s Rule. Int. J. Osteoarchaeol. 1999, 9, 182–188. [Google Scholar] [CrossRef]
- Betti, L.; Lycett, S.J.; von Cramon-Taubadel, N.; Pearson, O.M. Are human hands and feet affected by climate? A test of Allen’s rule. Am. J. Phys. Anthropol. 2015, 158, 132–140. [Google Scholar] [CrossRef]
- Dianat, I.; Nedaei, M.; Nezami, M.A.M. The effects of tool handle shape on hand performance, usability and discomfort using masons’ trowels. Int. J. Ind. Ergon. 2015, 45, 13–20. [Google Scholar] [CrossRef]
- Kong, Y.K.; Lowe, B.D. Optimal cylindrical handle diameter for grip force tasks. Int. J. Ind. Ergon. 2005, 35, 495–507. [Google Scholar] [CrossRef]
- Rolian, C.; Lieberman, D.E.; Zermeno, J.P. Hand biomechanics during simulated stone tool use. J. Hum. Evol. 2011, 61, 26–41. [Google Scholar] [CrossRef]
- Key, A.J.; Lycett, S.J. Investigating interrelationships between Lower Palaeolithic stone tool effectiveness and tool user biometric variation: Implications for technological and evolutionary changes. Archaeol. Anthrop. Sci. 2018, 10, 989–1006. [Google Scholar] [CrossRef]
- Mathiowetz, V.; Kashman, N.; Volland, G.; Weber, K.; Dowe, M.; Rogers, S. Grip and pinch strength: Normative data for adults. Arch. Phys. Med. Rehabil. 1985, 66, 69–74. [Google Scholar]
- Bohannon, R.W. Dynamometer measurements of hand-grip strength predict multiple outcomes. Percept. Mot. Skills 2001, 93, 323–328. [Google Scholar] [CrossRef]
- Bohannon, R.W. Hand-grip dynamometry predicts future outcomes in aging adults. J. Geriat. Phys. Ther. 2008, 31, 3–10. [Google Scholar] [CrossRef]
- Cuesta-Vargas, A.; Hilgenkamp, T. Reference values of grip strength measured with a Jamar dynamometer in 1526 adults with intellectual disabilities and compared to adults without intellectual disability. PLoS ONE 2015, 10, e0129585. [Google Scholar] [CrossRef] [PubMed]
- Zaggelidis, G. Maximal isometric handgrip strength (HGS) in Greek elite male judo and karate athletes. Sport Sci. Rev. 2016, 25, 320. [Google Scholar] [CrossRef]
- Josty, I.C.; Tyler, M.P.H.; Shewell, P.C.; Roberts, A.H.N. Grip and pinch strength variations in different types of workers. J. Hand Surg. 1997, 22, 266–269. [Google Scholar] [CrossRef]
- Nevill, A.M.; Holder, R.L. Modelling handgrip strength in the presence of confounding variables: Results from the Allied Dunbar National Fitness Survey. Ergonomics 2000, 43, 1547–1558. [Google Scholar] [CrossRef]
- Saremi, M.; Rostamzadeh, S. Hand Dimensions and Grip Strength: A Comparison of Manual and Non-manual Workers. In Proceedings of the 20th Congress of the International Ergonomics Association, Florence, Italy, 26–30 August 2018; Bagnara, S., Tartaglia, R., Albolino, S., Alexander, T., Fujita, Y., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 520–529. [Google Scholar]
- Burdukiewicz, A.; Pietraszewska, J.; Andrzejewska, J.; Chromik, K.; Stachoń, A. Asymmetry of Musculature and Hand Grip Strength in Bodybuilders and Martial Artists. Int. J. Environ. Res. Public Health 2020, 17, 4695. [Google Scholar] [CrossRef]
- Desrosiers, J.; Bravo, G.; Hẻbert, R.; Dutil, E. Normative data forgrip strength of elderly men and women. Am. J. Occup. Ther. 1995, 49, 637–644. [Google Scholar] [CrossRef]
- Vaz, M.; Hunsberger, S.; Diffey, B. Prediction equations for hand grip strength in healthy Indian male and female subjects encompassing a wide age range. Ann. Hum. Biol. 2002, 29, 131–141. [Google Scholar] [CrossRef]
- Bohannon, R.W.; Bear-Lehman, J.; Desrosiers, J.; Massy-Westropp, N.; Mathiowetz, V. Average grip strength: A meta-analysis of data obtained with a Jamar dynamometer from individuals 75 years or more of age. J. Geriatr. Phys. Ther. 2007, 30, 28–30. [Google Scholar] [CrossRef] [PubMed]
- Clerke, A.M.; Clerke, J.P.; Adams, R.D. Effects of hand shape on maximal isometric grip strength and its reliability in teenagers. J. Hand Ther. 2005, 18, 19–29. [Google Scholar] [CrossRef]
- Hone, L.S.; McCullough, M.E. 2D: 4D ratios predict hand grip strength (but not hand grip endurance) in men (but not in women). Evol. Hum. Behav. 2012, 33, 780–789. [Google Scholar] [CrossRef]
- Petersen, P.; Petrick, M.; Connor, H.; Conklin, D. Grip strength and hand dominance: Challenging the 10% rule. Am. J. Occup. Ther. 1989, 43, 444–447. [Google Scholar] [CrossRef] [PubMed]
- Leyk, D.; Gorges, W.; Ridder, D.; Wunderlich, M.; Rüther, T.; Sievert, A.; Essfeld, D. Hand-grip strength of young men, women and highly trained female athletes. Eur. J. Appl. Physiol. 2007, 99, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Angst, F.; Drerup, S.; Werle, S.; Herren, D.B.; Simmen, B.R.; Goldhahn, J. Prediction of grip and key pinch strength in 978 healthy subjects. BMC Musculoskelet. Disord. 2010, 11, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Massy-Westropp, N.M.; Gill, T.K.; Taylor, A.W.; Bohannon, R.W.; Hill, C.L. Hand Grip Strength: Age and gender stratified normative data in a population-based study. BMC Res. Notes 2011, 4, 1–5. [Google Scholar] [CrossRef]
- Hanten, W.P.; Chen, W.-Y.; Austin, A.A.; Brooks, R.E.; Carter, H.C.; Law, C.A.; Morgan, M.K.; Sanders, D.J.; Swan, C.A.; Vanderslice, A.L. Maximum grip strength in normal subjects from 20 to 64years of age. J. Hand. Ther. 1999, 12, 193–200. [Google Scholar] [CrossRef]
- Bohannon, R.W. Grip strength: A summary of studies comparing dominant and nondominant limb measurements. Percept. Mot. Ski. 2003, 96, 728–730. [Google Scholar] [CrossRef] [PubMed]
- Aghazadeh, F.; Waikar, A.M.; Lee, K.S.; Backhouse, T.; Davis, P. Impact of anthropometric variables and sex on grip strength. In Advances in Industrial Ergonomics and Safety; Mital, A., Ed.; Taylor & Francis: London, UK, 1989; Volume I, pp. 501–505. [Google Scholar]
- Yakou, T.; Yamamoto, K.; Koyama, M.; Hyodo, K. Sensory evaluation of grip using cylindrical objects. JSME Int. J. Ser. C Mech. Syst. Mach. Elem. Manuf. 1997, 40, 730–735. [Google Scholar] [CrossRef][Green Version]
- Crawford, J.O.; Wabine, E.; Nayak, L. The interaction between lid diameter, height and shape on wrist torque exertion in younger and older adults. Ergonomics 2002, 45, 922–933. [Google Scholar] [CrossRef]
- Kong, Y.K.; Kim, D.M. The relationship between hand anthropometrics, total grip strength and individual finger force for various handle shapes. Int. J. Occup. Saf. Ergon. 2015, 21, 187–192. [Google Scholar] [CrossRef]
- Barut, Ç.; Demirel, P.; Kiran, S. Evaluation of hand anthropometric measurements and grip strength in basketball, volleyball and handball players. Anatomy 2008, 2, 55–59. [Google Scholar] [CrossRef]
- Carson, R.G. Get a grip: Individual variations in grip strength are a marker of brain health. Neurobiol. Aging 2018, 71, 189–222. [Google Scholar] [CrossRef] [PubMed]
- Praetorius Björk, M.; Johansson, B.; Hassing, L.B. I forgot when I lost my grip-strong associations between cognition and grip strength in level of performance and change across time in relation to impending death. Neurobiol. Aging 2016, 38, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Lau, V.W.-S.; Ip, W.-Y. Comparison of power grip and lateral pinch strengths between the dominant and non-dominant hands for normal Chinese male subjects of different occupational demand. Hong Kong Physiother. J. 2006, 24, 16–22. [Google Scholar] [CrossRef]
- Mitsionis, G.; Pakos, E.E.; Stafilas, K.S.; Paschos, N.; Papakostas, T.; Beris, A.E. Normative data on hand grip strength in a Greek adult population. Int. Orthop. 2009, 33, 713–717. [Google Scholar] [CrossRef]
- Hossain, M.G.; Zyroul, R.; Pereira, B.P.; Kamarul, T. Multiple regression analysis of factors influencing dominant hand grip strength in an adult Malaysian population. J. Hand Surg. 2012, 37, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W.; Peolsson, A.; Massy-Westropp, N.; Desrosiers, J.; Bear-Lehman, J. Reference values for adult grip strength measured with a Jamar dynamometer: A descriptive meta-analysis. Physiotherapy 2006, 92, 11–15. [Google Scholar] [CrossRef]
- Nicolay, C.W.; Walker, A.L. Grip strength and endurance: Influences of anthropometric variation, hand dominance, and gender. Int. J. Ind. Ergon. 2005, 35, 605–618. [Google Scholar] [CrossRef]
- Wang, Y.C.; Bohannon, R.W.; Li, X.; Sindhu, B.; Kapellusch, J. Hand-grip strength: Normative reference values and equations for individuals 18 to 85 years of age residing in the United States. J. Orthop. Sports Phys. Ther. 2018, 48, 685–693. [Google Scholar] [CrossRef]
- Cardoso, H.F.V.; Severino, R.S.S. The chronology of epiphyseal union in the hand and foot from dry bone observations. Int. J. Osteoarchaeol. 2010, 20, 737–746. [Google Scholar] [CrossRef]
- Cunningham, C.; Scheuer, L.; Black, S. Developmental Juvenile Osteology, 2nd ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 283–350. [Google Scholar]
- Fess, E.E. Grip strength. In Clinical Assessment Recommendations, 2nd ed.; Casanova, J.S., Ed.; American Society of Hand Therapists: Chicago, IL, USA, 1992; pp. 41–45. [Google Scholar]
- Rohlf, F.J. tpsDig2 Software; Version 2.31; The State University of New York at Stony Brook: Stony Brook, NY, USA, 2017; Available online: http://www.sbmorphometrics.org/soft-dataacq.html (accessed on 12 October 2020).
- Gamer, M.; Lemon, J.; Fellows, I.; Singh, P. irr: Various Coefficients of Interrater Reliability and Agreement; R Package Version 0.84.1.; The R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://CRAN.R-project.org/package=irr (accessed on 3 March 2021).
- Fox, J.; Weisberg, S. Visualizing Fit and Lack of Fit in Complex Regression Models with Predictor Effect Plots and Partial Residuals. J. Stat. Softw. 2018, 87, 1–27. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 6 June 2020).
- Golden, C.J.; Espe-Pfeifer, P.; Wachsler-Felder, J. Neuropsychological Interpretation of Objective Psychological Tests. Motor and Sensory Tests; Springer: New York, NY, USA, 2002; 243p. [Google Scholar]
- Porac, C. Are age trends in adult hand preference best explained by developmental shifts or generational differences? Can. J. Exp. Psychol. 1993, 47, 697–713. [Google Scholar] [CrossRef]
- Armstrong, C.A.; Oldham, J.A. A comparison of dominant and non-dominant hand strengths. J. Hand Surg. Eur. Vol. 1999, 24, 421–425. [Google Scholar] [CrossRef] [PubMed]
- Crosby, C.A.; Wehbé, M.A. Hand strength: Normative values. J. Hand Surg. 1994, 19, 665–670. [Google Scholar] [CrossRef]
- Incel, N.A.; Ceceli, E.; Durukan, P.B.; Erdem, H.R.; Yorgancioglu, Z.R. Grip strength: Effect of hand dominance. Singap. Med. J. 2002, 43, 234–237. [Google Scholar]
- Lewandowski, L.; Kobus, D.A.; Church, K.L.; Van Orden, K. Neuropsychological implications of hand preference versus hand grip performance. Percep. Mot. Skills 1982, 55, 311–314. [Google Scholar] [CrossRef]
- Ziyagil, M.A.; Gürsoy, R.; Dane, Ş.; Türkmen, M.; Çebi, M. Effects of handedness on the hand grip strength asymmetry in Turkish athletes. Compr. Psychol. 2015, 4, 20. [Google Scholar] [CrossRef]
- Bagis, S.; Sahin, G.; Yapici, Y.; Cimen, Ö.B.; Erdogan, C. The effect of hand osteoarthritis on grip and pinch strength and hand function in postmenopausal women. Clin. Rheumatol. 2003, 22, 420–424. [Google Scholar] [CrossRef]
- Fontana, L.; Neel, S.; Claise, J.M.; Uqhetto, S.; Catilina, P. Osteoarthritis of the thumb carpometacarpal joint in women and occupational risk factors: A case-control study. J. Hand. Surg. Am. 2007, 32, 459–465. [Google Scholar] [CrossRef]
- Ceceli, E.; Gül, S.; Borman, P.N.; Uysal, S.R.; Okumuş, M. Hand function in female patients with hand osteoarthritis: Relation with radiological progression. Hand 2012, 7, 335–340. [Google Scholar] [CrossRef]
- Marzke, M.W.; Toth, N.; Schick, K.; Reece, S.; Steinberg, B.; Hunt, K.; Linscheid, R.L.; An, K.N. EMG study of hand muscle recruitment during hard hammer percussion manufacture of Oldowan tools. Am. J. Phys. Anthropol. 1998, 105, 315–332. [Google Scholar] [CrossRef]
- Key, A.J.; Dunmore, C.J. The evolution of the hominin thumb and the influence exerted by the non-dominant hand during stone tool production. J. Hum. Evol. 2015, 78, 60–69. [Google Scholar] [CrossRef]
- Shaw, C.N.; Hofmann, C.L.; Petraglia, M.D.; Stock, J.T.; Gottschall, J.S. Neandertal humeri may reflect adaptation to scraping tasks, but not spear thrusting. PLoS ONE 2012, 7, e40349. [Google Scholar] [CrossRef]
- Key, A.J. Manual loading distribution during carrying behaviors: Implications for the evolution of the hominin hand. PLoS ONE 2016, 11, e0163801. [Google Scholar] [CrossRef]
- Eshed, V.; Gopher, A.; Galili, E.; Hershkovitz, I. Musculoskeletal stress markers in Natufian hunter-gatherers and Neolithic farmers in the Levant: The upper limb. Amer. J. Phys. Anth. 2004, 123, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Ward, C.V. Interpreting the posture and locomotion of Australopithecus afarensis: Where do we stand? Am. J. Phys. Anthropol. 2002, 119 (Suppl. 35), 185–215. [Google Scholar] [CrossRef]
- Kivell, T.L. Evidence in hand: Recent discoveries and the early evolution of human manual manipulation. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2015, 370, 20150105. [Google Scholar] [CrossRef] [PubMed]
- Carlson, K.J.; Green, D.J.; Jashashvili, T.; Pickering, T.R.; Heaton, J.L.; Beaudet, A.; Stratford, D.; Crompton, R.; Kuman, K.; Bruxelles, L.; et al. The pectoral girdle of StW 573 (‘Little Foot’) and its implications for shoulder evolution in the Hominina. J. Hum. Evol. 2021, 102983. [Google Scholar] [CrossRef]
- Cartmill, M. Climbing. In Functional Vertebrate Morphology; Hildebrand, M., Bramble, D.M., Liem, K.F., Wake, D.B., Eds.; Harvard University Press: Cambridge, MA, USA, 1985; pp. 73–88. [Google Scholar]
- Preuschoft, H. What does “arboreal locomotion” mean exactly and what are the relationships between “climbing”, environment and morphology? Z. Morphol. Anthropol. 2002, 83, 171–188. [Google Scholar]
- Kappelman, J.; Ketcham, R.A.; Pearce, S.; Todd, L.; Akins, W.; Colbert, M.W.; Feseha, M.; Maisano, J.A.; Witzel, A. Perimortem fractures in Lucy suggest mortality from fall out of tall tree. Nature 2016, 537, 503–507. [Google Scholar] [CrossRef]
- Rolian, C.; Gordon, A.D. Reassessing manual proportions in Australopithecus afarensis. Am. J. Phys. Anthropol. 2013, 152, 393–406. [Google Scholar] [CrossRef]
- Niewoehner, W.A.; Bergstrom, A.; Eichele, D.; Zuroff, M.; Clark, J.T. Manual dexterity in Neanderthals. Nature 2003, 422, 395. [Google Scholar] [CrossRef]
- Feix, T.; Kivell, T.L.; Pouydebat, E.; Dollar, A.M. Estimating thumb–index finger precision grip and manipulation potential in extant and fossil primates. J. R. Soc. Interface 2015, 12, 20150176. [Google Scholar] [CrossRef]
- Almécija, S.; Alba, D.M. On manual proportions and pad-to-pad precision grasping in Australopithecus afarensis. J. Hum. Evol. 2014, 73, 88–92. [Google Scholar] [CrossRef]
- Niewoehner, W.A. Neanderthal hands in their proper perspective. In Neanderthals Revisited: New Approaches and Perspectives; Harvati, K., Harrison, T., Eds.; Springer: Berlin, Germany, 2008; pp. 157–190. [Google Scholar]
- Bardo, A.; Moncel, M.H.; Dunmore, C.J.; Kivell, T.L.; Pouydebat, E.; Cornette, R. The implications of thumb movements for Neanderthal and modern human manipulation. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Misiak, M.; Butovskaya, M.; Oleszkiewicz, A.; Sorokowski, P. Digit ratio and hand grip strength are associated with male competition outcomes: A study among traditional populations of the Yali and Hadza. Am. J. Hum. Biol. 2020, 32, e23321. [Google Scholar] [CrossRef]
- Ruff, C.B.; Larsen, C.S. Long bone structural analyses and the reconstruction of past mobility: A historical review. In Reconstructing Mobility; Carlson, K.J., Marchi, D., Eds.; Springer: Boston, MA, USA, 2014; pp. 13–29. [Google Scholar]
- Chirchir, H.; Kivell, T.L.; Ruff, C.B.; Hublin, J.J.; Carlson, K.J.; Zipfel, B.; Richmond, B.G. Recent origin of low trabecular bone density in modern humans. Proc. Natl. Acad. Sci. USA 2015, 112, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Chirchir, H.; Ruff, C.B.; Junno, J.A.; Potts, R. Low trabecular bone density in recent sedentary modern humans. Am. J. Phys. Anthropol. 2017, 162, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Ryan, T.M.; Shaw, C.N. Gracility of the modern Homo sapiens skeleton is the result of decreased biomechanical loading. Proc. Natl. Acad. Sci. USA 2015, 112, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Cashmore, L.; Uomini, N.; Chapelain, A. The evolution of handedness in humans and great apes: A review and current issues. J. Anthropol. Sci. 2008, 86, 7–35. [Google Scholar]
- Provins, K.A. The specificity of motor skill and manual asymmetry: A review of the evidence and its implications. J. Mot. Behav. 1997, 29, 183–192. [Google Scholar] [CrossRef]
- Fagot, J.; Vauclair, J. Manual laterality in nonhuman primates: A distinction between handedness and manual specialization. Psychol. Bull. 1991, 109, 76. [Google Scholar] [CrossRef]
- Steele, J.; Uomini, N. Can the archaeology of manual specialization tell us anything about language evolution? A survey of the state of play. Cambr. Arch. J. 2009, 19, 97–110. [Google Scholar] [CrossRef][Green Version]
- Lazenby, R.A. Identification of sex from metacarpals: Effect of side asymmetry. J. Forensic Sci. 1994, 39, 1188–1194. [Google Scholar] [CrossRef] [PubMed]
- Lazenby, R.A.; Cooper, D.M.L.; Angus, S.; Hallgrmsson, B. Articular constraint, handedness, and directional asymmetry in the human second metacarpal. J. Hum. Evol. 2008, 54, 857–885. [Google Scholar] [CrossRef] [PubMed]
- Stephens, N.B.; Kivell, T.L.; Gross, T.; Pahr, D.H.; Lazenby, R.A.; Hublin, J.J.; Hershkovitz, I.; Skinner, M.M. Trabecular architecture in the thumb of Pan and Homo: Implications for investigating hand use, loading, and hand preference in the fossil record. Am. J. Phys. Anthropol. 2016, 161, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Boonsaner, K.; Louthrenoo, W.; Meyer, S.; Schumacher, H.R., Jr. Effect of dominancy on severity in rheumatoid arthritis. Br. J. Rheumatol. 1992, 31, 77–80. [Google Scholar] [CrossRef]
- Sarringhaus, L.A.; Stock, J.T.; Marchant, L.F.; McGrew, W.C. Bilateral asymmetry in the limb bones of the chimpanzee (Pan troglodytes). Am. J. Phys. Anthropol. 2005, 128, 840–845. [Google Scholar] [CrossRef]
- Warguła, Ł.; Kukla, M.; Krawiec, P.; Wieczorek, B. Impact of Number of Operators and Distance to Branch Piles on Woodchipper Operation. Forests 2020, 11, 598. [Google Scholar] [CrossRef]
- Wieczorek, B.; Kukla, M. Biomechanical Relationships Between Manual Wheelchair Steering and the Position of the Human Body’s Center of Gravity. J. Biomech. Eng. 2020, 142. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).