Biophysical and Fluoride Release Properties of a Resin Modified Glass Ionomer Cement Enriched with Bioactive Glasses
Abstract
1. Introduction
2. Materials and Methods
2.1. Bond Strength and Microleakage Testing
2.2. Bond Strength Test
2.3. Microleakage Testing
2.4. Fluoride Release Test
2.5. In Vitro Study—Cell Viability Assay
2.6. Scratch Migration Assay
2.7. Statistical Analysis
3. Results
3.1. Bond Strength and Microleakage Test
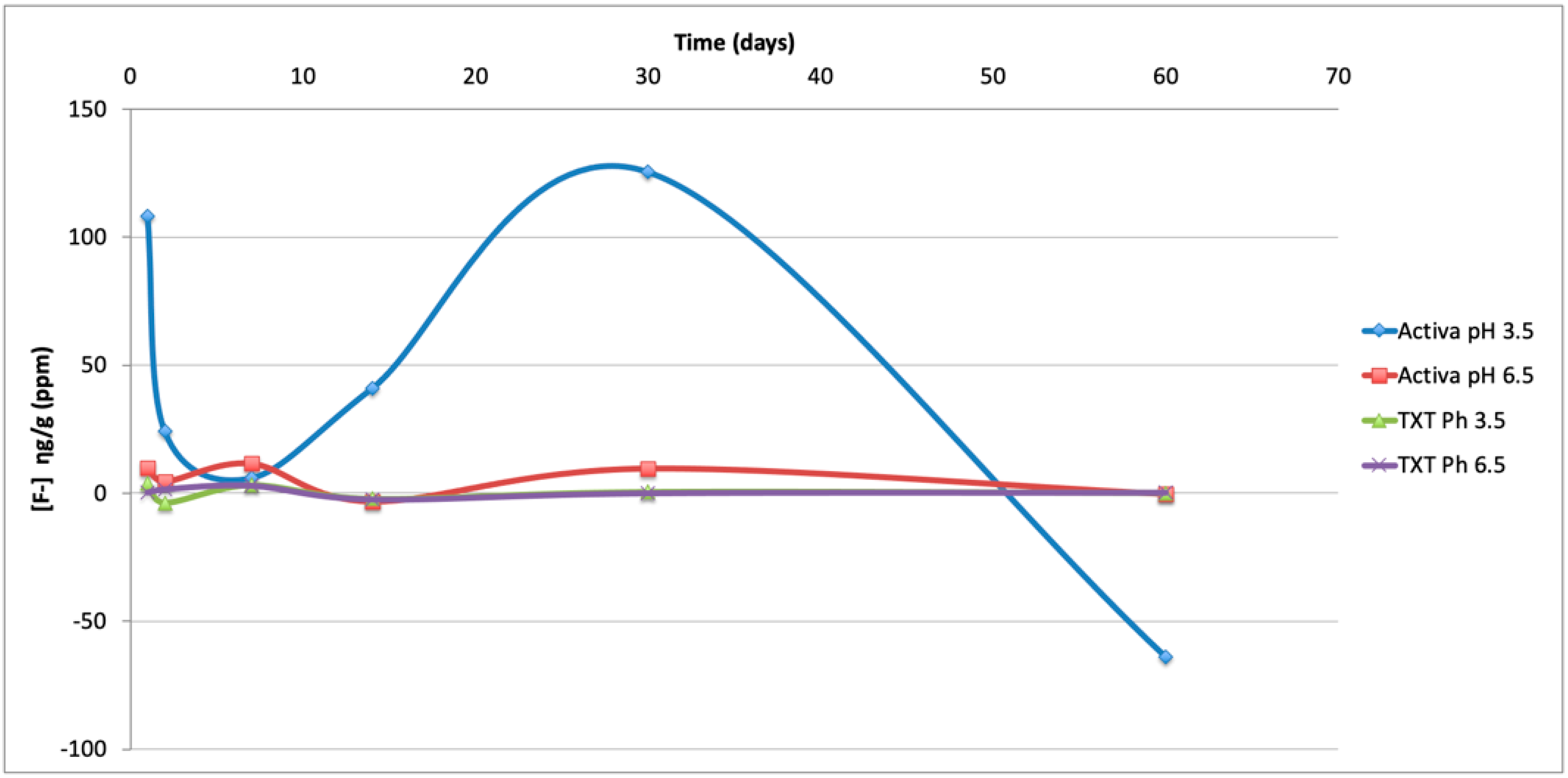
3.2. Fluoride Release
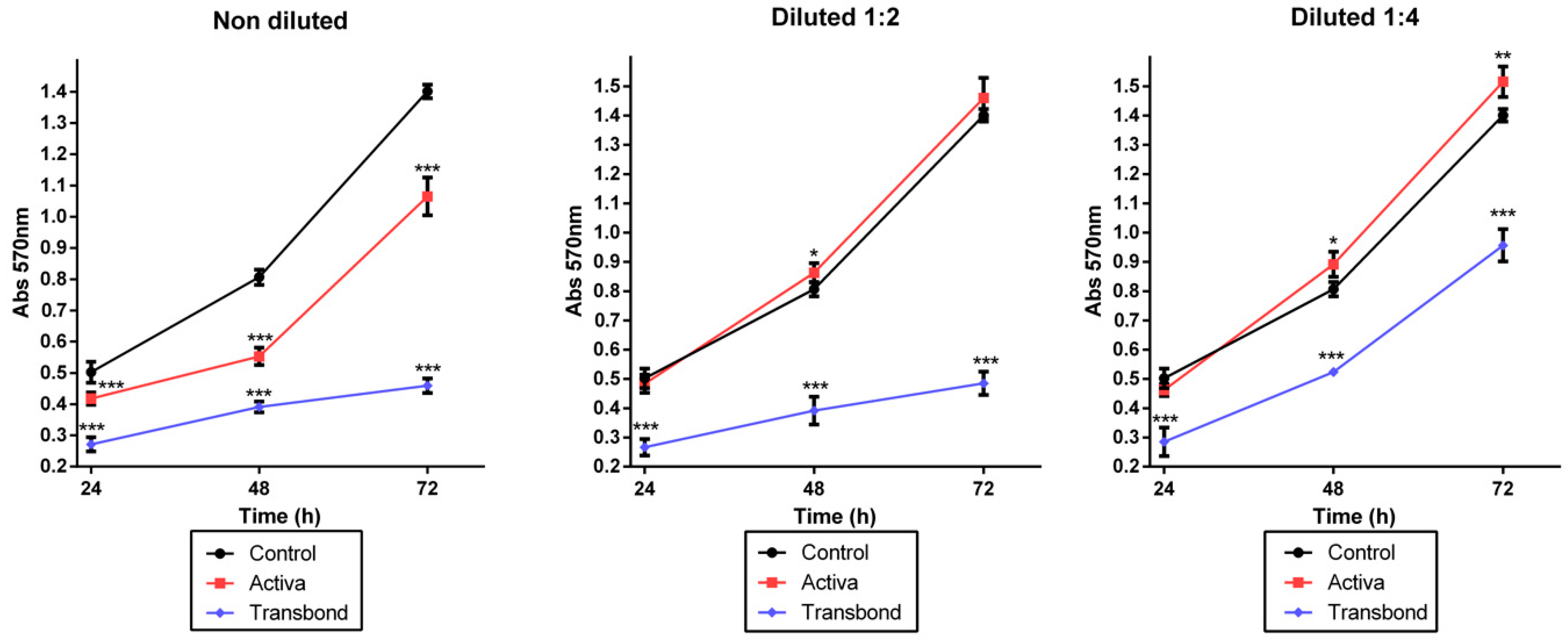
3.3. Cell Viability Assay
3.4. Scratch Migration Assay
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nam, H.J.; Kim, Y.M.; Kwon, Y.H.; Kim, I.R.; Park, B.S.; Son, W.S.; Lee, S.M.; Kim, Y.I. Enamel Surface Remineralization Effect by Fluorinated Graphite and Bioactive Glass-Containing Orthodontic Bonding Resin. Materials 2019, 12, 1308. [Google Scholar] [CrossRef]
- Al-Eesa, N.A.; Wong, F.S.L.; Johal, A.; Hill, R.G. Fluoride containing bioactive glass composite for orthodontic adhesives—Ion release properties. Dent. Mater. 2017, 33, 1324–1329. [Google Scholar] [CrossRef] [PubMed]
- Khoroushi, M.; Keshani, F. A review of glass-ionomers: From conventional glass-ionomer to bioactive glass-ionomer. Dent. Res. J. 2013, 10, 411–420. [Google Scholar]
- Kim, Y.M.; Kim, D.H.; Song, C.W.; Yoon, S.Y.; Kim, S.Y.; Na, H.S.; Chung, J.; Kim, Y.I.; Kwon, Y.H. Antibacterial and remineralization effects of orthodontic bonding agents containing bioactive glass. Korean J. Orthod. 2018, 48, 163–171. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, N.; Weir, M.D.; Bai, Y.; Xu, H.H.K. Novel multifunctional dental cement to prevent enamel demineralization near orthodontic brackets. J. Dent. 2017, 64, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.S.; Syed, M.R. A review of bioceramics-based dental restorative materials. Dent. Mater. J. 2019, 38, 163–176. [Google Scholar] [CrossRef]
- Porenczuk, A.; Jankiewicz, B.; Naurecka, M.; Bartosewicz, B.; Sierakowski, B.; Gozdowski, D.; Kostecki, J.; Nasilowska, B.; Mielczarek, A. A comparison of the remineralizing potential of dental restorative materials by analyzing their fluoride release profiles. Adv. Clin. Exp. Med. 2019, 28, 815–823. [Google Scholar] [CrossRef]
- Lopez-Garcia, S.; Pecci-Lloret, M.P.; Pecci-Lloret, M.R.; Onate-Sanchez, R.E.; Garcia-Bernal, D.; Castelo-Baz, P.; Rodriguez-Lozano, F.J.; Guerrero-Girones, J. In Vitro Evaluation of the Biological Effects of ACTIVA Kids BioACTIVE Restorative, Ionolux, and Riva Light Cure on Human Dental Pulp Stem Cells. Materials 2019, 12, 3694. [Google Scholar] [CrossRef]
- Saunders, K.G.; Mattevi, G.; Donly, K.J.; Anthony, R. Enamel Demineralization Adjacent to Orthodontic Brackets Bonded with ACTIVA BioACTIVE-RESTORATIVE. APOS Trends Orthod. 2018, 8, 200–203. [Google Scholar] [CrossRef]
- Alrahlah, A. Diametral Tensile Strength, Flexural Strength, and Surface Microhardness of Bioactive Bulk Fill Restorative. J. Contemp. Dent. Pract. 2018, 19, 13–19. [Google Scholar] [CrossRef]
- Arikan, S.; Arhun, N.; Arman, A.; Cehreli, S.B. Microleakage beneath ceramic and metal brackets photopolymerized with LED or conventional light curing units. Angle Orthod. 2006, 76, 1035–1040. [Google Scholar] [CrossRef]
- Wiegand, A.; Buchalla, W.; Attin, T. Review on fluoride-releasing restorative materials—Fluoride release and uptake characteristics, antibacterial activity and influence on caries formation. Dent. Mater. 2007, 23, 343–362. [Google Scholar] [CrossRef] [PubMed]
- Tiskaya, M.; Al-Eesa, N.A.; Wong, F.S.L.; Hill, R.G. Characterization of the bioactivity of two commercial composites. Dent. Mater. 2019, 35, 1757–1768. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standarization. Dental Materials-Guidance on Testing of Adhesion to Tooth Structure; ISO TR 11405; International Organization for Standarization: Geneva, Switzerland, 1994. [Google Scholar]
- Oncag, G.; Tuncer, A.V.; Tosun, Y.S. Acidic soft drinks effects on the shear bond strength of orthodontic brackets and a scanning electron microscopy evaluation of the enamel. Angle Orthod. 2005, 75, 247–253. [Google Scholar] [CrossRef]
- International Standard. Biological Evaluation of Medical Devices. Part 5. Test for In Vitro Cytotoxicity; International Organization for Standardization: Geneva, Switzerland, 2009. [Google Scholar]
- Arnold, R.W.; Combe, E.C.; Warford, J.H., Jr. Bonding of stainless steel brackets to enamel with a new self-etching primer. Am. J. Orthod. Dentofac. Orthop. 2002, 122, 274–276. [Google Scholar] [CrossRef]
- Vicente, A.; Bravo, L.A.; Romero, M.; Ortiz, A.J.; Canteras, M. Shear bond strength of orthodontic brackets bonded with self-etching primers. Am. J. Dent. 2005, 18, 256–260. [Google Scholar]
- Ozer, M.; Bayram, M.; Dincyurek, C.; Tokalak, F. Clinical bond failure rates of adhesive precoated self-ligating brackets using a self-etching primer. Angle Orthod. 2014, 84, 155–160. [Google Scholar] [CrossRef]
- Ousehal, L.; El Aouame, A.; Rachdy, Z.; Benkiran, G. Comparison of the efficacy of a conventional primer and a self-etching primer. Int. Orthod. 2016, 14, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Yoo, K.H.; Yoon, S.Y.; Kim, I.R.; Park, B.S.; Son, W.S.; Ko, C.C.; Son, S.A.; Kim, Y.I. Enamel Anti-Demineralization Effect of Orthodontic Adhesive Containing Bioactive Glass and Graphene Oxide: An In-Vitro Study. Materials 2018, 11, 1728. [Google Scholar] [CrossRef]
- Kohda, N.; Iijima, M.; Kawaguchi, K.; Toshima, H.; Muguruma, T.; Endo, K.; Mizoguchi, I. Inhibition of enamel demineralization and bond-strength properties of bioactive glass containing 4-META/MMA-TBB-based resin adhesive. Eur. J. Oral Sci. 2015, 123, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Sauro, S.; Makeeva, I.; Faus-Matoses, V.; Foschi, F.; Giovarruscio, M.; Maciel Pires, P.; Martins Moura, M.E.; Almeida Neves, A.; Faus-Llacer, V. Effects of Ions-Releasing Restorative Materials on the Dentine Bonding Longevity of Modern Universal Adhesives after Load-Cycle and Prolonged Artificial Saliva Aging. Materials 2019, 12, 722. [Google Scholar] [CrossRef]
- Alkhudhairy, F.I.; Ahmad, Z.H. Comparison of Shear Bond Strength and Microleakage of Various Bulk-fill Bioactive Dentin substitutes: An in vitro Study. J. Contemp. Dent. Pract. 2016, 17, 997–1002. [Google Scholar] [CrossRef]
- van Dijken, J.W.V.; Pallesen, U.; Benetti, A. A randomized controlled evaluation of posterior resin restorations of an altered resin modified glass-ionomer cement with claimed bioactivity. Dent. Mater. 2019, 35, 335–343. [Google Scholar] [CrossRef]
- Amaireh, A.I.; Al-Jundi, S.H.; Alshraideh, H.A. In vitro evaluation of microleakage in primary teeth restored with three adhesive materials: ACTIVA, composite resin, and resin-modified glass ionomer. Eur. Arch. Paediatr. Dent. 2019, 20, 359–367. [Google Scholar] [CrossRef]
- Omidi, B.R.; Naeini, F.F.; Dehghan, H.; Tamiz, P.; Savadroodbari, M.M.; Jabbarian, R. Microleakage of an Enhanced Resin-Modified Glass Ionomer Restorative Material in Primary Molars. J. Dent. 2018, 15, 205–213. [Google Scholar]
- Wan Jusoh, W.N.; Matori, K.A.; Mohd Zaid, M.H.; Zainuddin, N.; Ahmad Khiri, M.Z.; Abdul Rahman, N.A.; Abdul Jalil, R.; Kul, E. Incorporation of Hydroxyapatite into Glass Ionomer Cement (GIC) Formulated Based on Alumino-Silicate-Fluoride Glass Ceramics from Waste Materials. Materials 2021, 14, 954. [Google Scholar] [CrossRef]
- Dressano, D.; Palialol, A.R.; Xavier, T.A.; Braga, R.R.; Oxman, J.D.; Watts, D.C.; Marchi, G.M.; Lima, A.F. Effect of diphenyliodonium hexafluorophosphate on the physical and chemical properties of ethanolic solvated resins containing camphorquinone and 1-phenyl-1,2-propanedione sensitizers as initiators. Dent. Mater. 2016, 32, 756–764. [Google Scholar] [CrossRef] [PubMed]
- Huddleston, J.G.; Visser, A.E.; Reichert, W.M.; Willauer, H.D.; Broker, G.A.; Rogers, R.D. Characterization and comparison of hydrophilic and hydrophobic room temperature ionic liquids incorporating the imidazolium cation. Green Chem. 2001, 3, 156–164. [Google Scholar] [CrossRef]
- Carey, C.M.; Spencer, M.; Gove, R.J.; Eichmiller, F.C. Fluoride release from a resin-modified glass-ionomer cement in a continuous-flow system. Effect of pH. J. Dent. Res. 2003, 82, 829–832. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Chersoni, S.; Acquaviva, G.L.; Piana, G.; Prati, C.; Mongiorgi, R. Fluoride release and absorption at different pH from glass-ionomer cements. Dent. Mater. 2006, 22, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Schmalz, G.; Widbiller, M.; Galler, K.M. Material Tissue Interaction--From Toxicity to Tissue Regeneration. Oper. Dent. 2016, 41, 117–131. [Google Scholar] [CrossRef]
- Abou ElReash, A.; Hamama, H.; Grawish, M.; Saeed, M.; Zaen El-Din, A.M.; Shahin, M.A.; Zhenhuan, W.; Xiaoli, X. A laboratory study to test the responses of human dental pulp stem cells to extracts from three dental pulp capping biomaterials. Int. Endod. J. 2021. [Google Scholar] [CrossRef] [PubMed]
- Marrelli, M.; Codispoti, B.; Shelton, R.M.; Scheven, B.A.; Cooper, P.R.; Tatullo, M.; Paduano, F. Dental Pulp Stem Cell Mechanoresponsiveness: Effects of Mechanical Stimuli on Dental Pulp Stem Cell Behavior. Front. Physiol. 2018, 9, 1685. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, G.; Codispoti, B.; Marrelli, M.; Rengo, C.; Rengo, S.; Tatullo, M. Commitment of Oral-Derived Stem Cells in Dental and Maxillofacial Applications. Dent. J. 2018, 6, 72. [Google Scholar] [CrossRef]
- Toy, E.; Yuksel, S.; Ozturk, F.; Karatas, O.H.; Yalcin, M. Evaluation of the genotoxicity and cytotoxicity in the buccal epithelial cells of patients undergoing orthodontic treatment with three light-cured bonding composites by using micronucleus testing. Korean J. Orthod. 2014, 44, 128–135. [Google Scholar] [CrossRef]
- Manojlovic, D.; Dramicanin, M.D.; Miletic, V.; Mitic-Culafic, D.; Jovanovic, B.; Nikolic, B. Cytotoxicity and genotoxicity of a low-shrinkage monomer and monoacylphosphine oxide photoinitiator: Comparative analyses of individual toxicity and combination effects in mixtures. Dent. Mater. 2017, 33, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Yang, H.; Cao, M.; Li, L.; Cai, Q. Correlating cytotoxicity to elution behaviors of composite resins in term of curing kinetic. Mater. Sci Eng. C Mater. Biol. Appl. 2017, 78, 413–419. [Google Scholar] [CrossRef]
- Abou ElReash, A.; Hamama, H.; Abdo, W.; Wu, Q.; Zaen El-Din, A.; Xiaoli, X. Biocompatibility of new bioactive resin composite versus calcium silicate cements: An animal study. BMC Oral Health 2019, 19, 194. [Google Scholar] [CrossRef] [PubMed]


| Product | Composition | %/wt |
|---|---|---|
| 3M UnitekTransbond XT Light Cure Adhesive (MSDS) | Silane Treated Quartz | 70–80 |
| Bisphenol A Diglycidyl Ether Dimethacrylate (BISGMA) | 10–20 | |
| Bisphenol A bis(2-Hydroxyethyl Ether) Dimethacrylate | 5–10 | |
| Silane Treated Silica | <2 | |
| Diphenyliodonium Hexafluorophosphate | <1 | |
| ACTIVA Bioactive Restorative [7] | Blend of diurethane and other methacrylates with modified polyacrylic acid | 44.6 |
| Reactive glass filler | 21.8 | |
| Inorganic filler | 56 | |
| patented rubberised resin (Embrace) | ||
| Water | ||
| Silica, amorphous | 6.7 | |
| Sodium fluoride | 0.75 |
| Group | Mean | Median | Standard Deviation | 95% C.I. | |
|---|---|---|---|---|---|
| TSEP/TXT | 9.41 | 9.56 | 2.86 | 8.06, 10.75 | A |
| TSEP/ACTIVA | 8.72 | 8.39 | 1.34 | 8.09, 9.35 | A |
| Phosphoric acid/ACTIVA | 11.89 | 11.29 | 2.41 | 10.76, 13.02 | B |
| Group | Enamel-Adhesive | Adhesive-Bracket | ||||
|---|---|---|---|---|---|---|
| Mean ± SD | Median | Range | Mean ± SD | Median | Range | |
| TSEP/TXT | 0.26 ± 0.38 | 0 | 1.19 | 0.07 ± 0.22 | 0 | 1 |
| TSEP/ACTIVA | 0.32 ± 0.75 | 0 | 2.60 | 0.02 ± 0.09 | 0 | 0.4 |
| Phosphoric acid/ACTIVA | 0.25 ± 0.67 | 0 | 2.60 | 0.00 ± 0.00 | 0 | 0 |
| 24 h χ (±SD) | 48 h χ (±SD) | 7 days χ (±SD) | 14 days χ (±SD) | 30 days χ (±SD) | 60 days χ (±SD) | |
|---|---|---|---|---|---|---|
| ACTIVA pH 3.5 | 108.09 ± 76.85 a | 130.01 ± 72.28 a | 150.97 ± 26.13 a | 440.57 ± 115.67 a | 2399.01 ± 988.26 | 483.88 ± 180.21 a |
| ACTIVA pH 6.4 | 9.96 ± 5.45 a,b | 14.48 ± 29.58 a,b,c | 72.28 ± 21.40 | 49.04 ± 17.00 a,b | 202.87 ± 111.16 | 191.77 ± 43.25 |
| p value | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| TXT pH 3.5 | 3.90 ± 6.06 | 0.15 ± 0.22 b,c | 15.95 ± 1.11 | 0.51 ± 0.60 b,c | 7.97 ± 20.99 | 11.51 ± 9.86 |
| TXT pH 6.4 | 0.35 ± 0.47 c | 1.91 ± 4.78 c | 16.80 ± 2.60 | 0.14 ± 0.31 b,c | 0.11 ± 0.60 b,c | 7.80 ± 3.32 |
| p value | =0.006 | =0.054 | =0.427 | =0.007 | =0.031 | =0.275 |
| Material | 1 day χ (±SD) | 2 days χ (±SD) | 7 days χ (±SD) | 14 days χ (±SD) | 30 days χ (±SD) | 60 days χ (±SD) |
|---|---|---|---|---|---|---|
| ACTIVA pH 3.5 | 108.09 ± 76.85 b | 24.08 ± 127.01 | 5.92 ± 13.24 a | 40.91 ± 16.28 | 125.46 ± 57.74 | −63.89 ± 29.47 a |
| ACTIVA pH 6.4 | 9.96 ± 5.45 + | 4.51 ± 32,40 a,c,d | 11.56 ±7.45 | −3.32 ± 1.28 a,c,d,+ | 9.61± 7.52 + | −0.37 ± 4.05 + |
| TXT pH 3.5 | 3.90 ± 6.06 + | −3.75 ± 6.15 c,d | 3.16 ± 0.22 | −2.21 ± 0.16 b,c,d,+ | 0.47 ± 1.30 + | 0.12 ± 0.70 + |
| TXT pH 6.4 | 0.35 ± 0.47 +,# | 1.56 ± 4.88 c | 2.98 ± 1.14 | −2.42 ± 0.40 b,c,d,+ | 0.002 ± 0.05 c,+,# | 0.26 ± 0.11 + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vicente, A.; Rodríguez-Lozano, F.J.; Martínez-Beneyto, Y.; Jaimez, M.; Guerrero-Gironés, J.; Ortiz-Ruiz, A.J. Biophysical and Fluoride Release Properties of a Resin Modified Glass Ionomer Cement Enriched with Bioactive Glasses. Symmetry 2021, 13, 494. https://doi.org/10.3390/sym13030494
Vicente A, Rodríguez-Lozano FJ, Martínez-Beneyto Y, Jaimez M, Guerrero-Gironés J, Ortiz-Ruiz AJ. Biophysical and Fluoride Release Properties of a Resin Modified Glass Ionomer Cement Enriched with Bioactive Glasses. Symmetry. 2021; 13(3):494. https://doi.org/10.3390/sym13030494
Chicago/Turabian StyleVicente, Ascensión, Francisco Javier Rodríguez-Lozano, Yolanda Martínez-Beneyto, María Jaimez, Julia Guerrero-Gironés, and Antonio J. Ortiz-Ruiz. 2021. "Biophysical and Fluoride Release Properties of a Resin Modified Glass Ionomer Cement Enriched with Bioactive Glasses" Symmetry 13, no. 3: 494. https://doi.org/10.3390/sym13030494
APA StyleVicente, A., Rodríguez-Lozano, F. J., Martínez-Beneyto, Y., Jaimez, M., Guerrero-Gironés, J., & Ortiz-Ruiz, A. J. (2021). Biophysical and Fluoride Release Properties of a Resin Modified Glass Ionomer Cement Enriched with Bioactive Glasses. Symmetry, 13(3), 494. https://doi.org/10.3390/sym13030494








