Visual Lateralization in the Cephalopod Mollusk Octopus vulgaris
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Scoring of Animals’ Responses Toward the Target
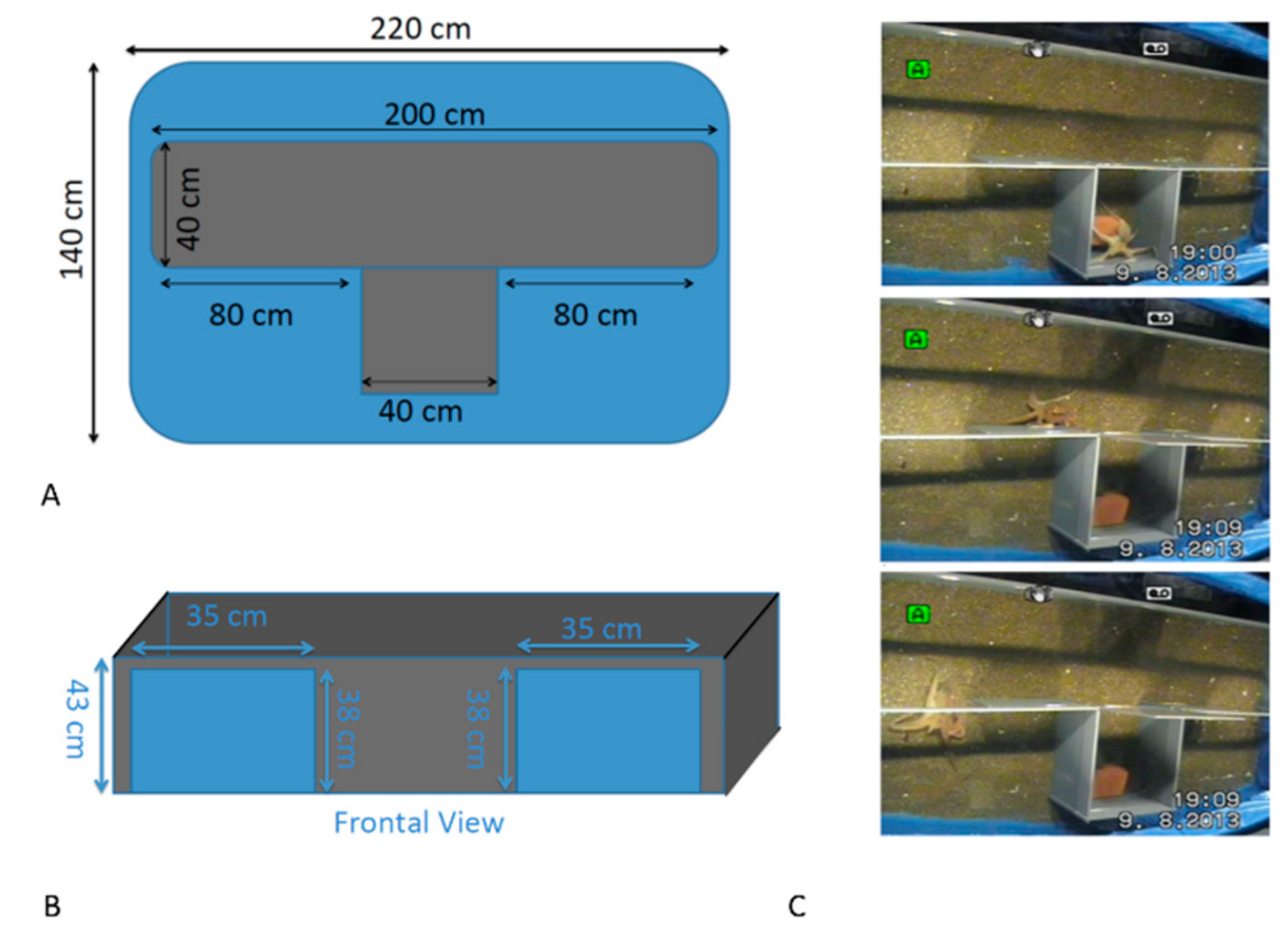
2.3. Experimental Procedure
2.3.1. Testing Asymmetry in Predatory Response: Natural Stimulus
2.3.2. Testing Asymmetry in Predatory Response: Artificial Stimulus
2.3.3. Testing Asymmetry in Turning: The T-Maze
2.4. Data Analysis
3. Results
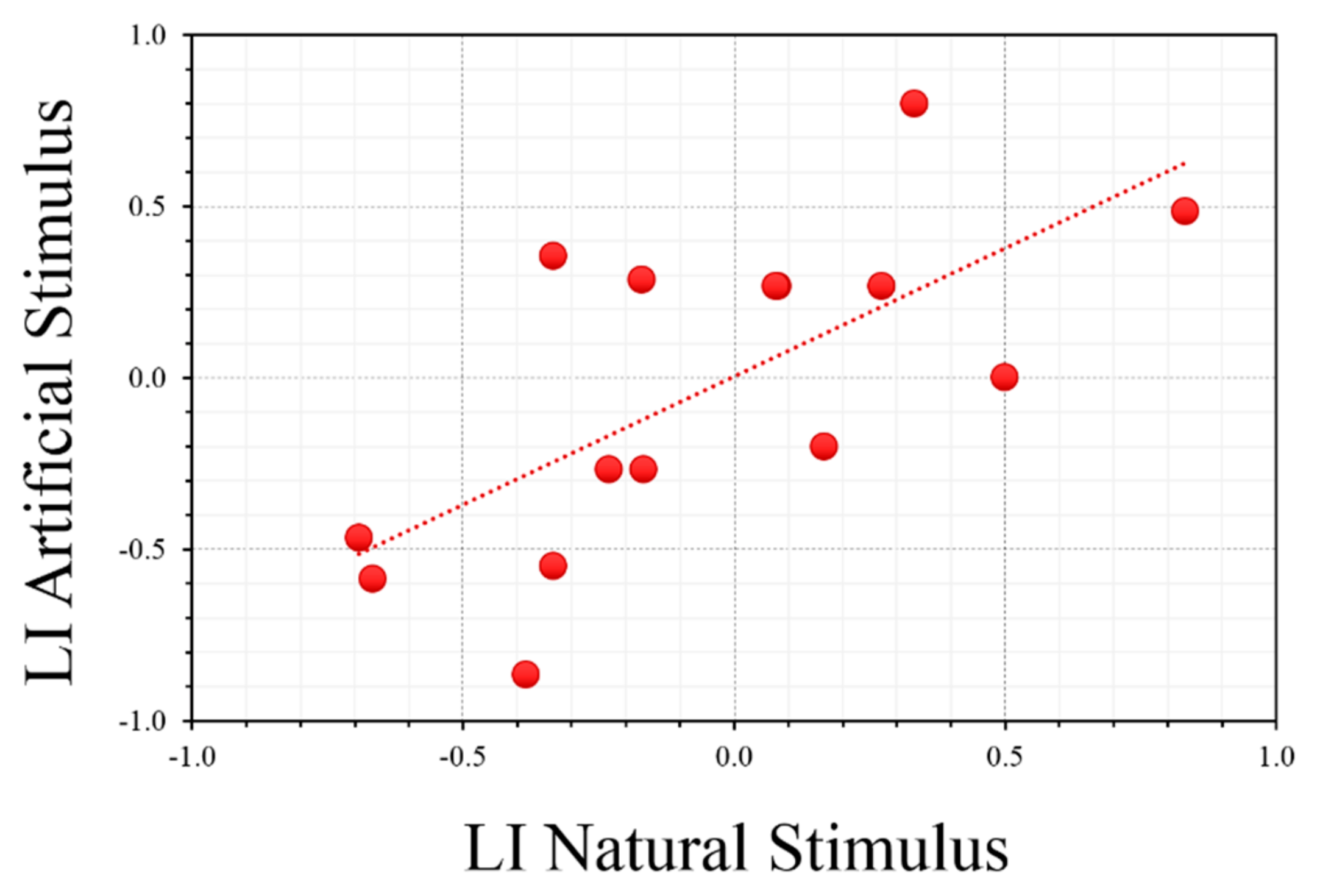
3.1. Asymmetry in Predatory Response: Natural and Artificial Stimuli
3.2. T-Maze
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rogers, L.J.; Vallortigara, G.; Andrew, R.J. Divided Brains: The Biology and Behaviour of Brain Asymmetries; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Rogers, L.J. Asymmetry of brain and behavior in animals: Its development, function, and human relevance. Genesis 2014, 52, 555–571. [Google Scholar] [CrossRef]
- Versace, E.; Vallortigara, G. Forelimb preferences in human beings and other species: Multiple models for testing hypotheses on lateralization. Front. Psychol. 2015, 6, 233. [Google Scholar] [CrossRef] [PubMed]
- Dadda, M.; Koolhaas, W.H.; Domenici, P. Behavioural asymmetry affects escape performance in a teleost fish. Biol. Lett. 2010, 6, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Stancher, G.; Sovrano, V.A.; Vallortigara, G. Chapter 2—Motor asymmetries in fishes, amphibians, and reptiles. In Progress in Brain Research; Forrester, G.S., Hopkins, W.D., Hudry, K., Lindell, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 238, pp. 33–56. [Google Scholar]
- Tommasi, L.; Andrew, R.; Vallortigara, G. Eye use in search is determined by the nature of task in the domestic chick (Gallus gallus). Behav. Brain Res. 2000, 112, 119–126. [Google Scholar] [CrossRef]
- Frasnelli, E.; Vallortigara, G.; Rogers, L.J. Left–right asymmetries of behaviour and nervous system in invertebrates. Neurosci. Biobehav. Rev. 2012, 36, 1273–1291. [Google Scholar] [CrossRef]
- Jereb, P.; Roper, C. Chambered Nautiluses and Sepioids (Nautilidae, Sepiidae, Sepiolidae, Sepiadariidae, Idiosepiidae and Spirulidae); FAO: Rome, Italy, 2005; Volume 1, p. 262. [Google Scholar]
- Jereb, P.; Roper, C. Cephalopods of the World. An Annotated and Illustrated Catalogue of Species Known to Date. Volume 2. Myopsid and Oegopsid Squids; FAO: Rome, Italy, 2010; p. 605. [Google Scholar]
- Jereb, P.; Roper, C.; Norman, M.; Finn, J. Cephalopods of the World. An Annotated and Illustrated Catalogue of Species Known to Date. Volume 3. Octopods and Vampire Squids; FAO: Rome, Italy, 2016; p. 370. [Google Scholar]
- Guerra, Á. Functional Anatomy: Macroscopic Anatomy and Post-mortem Examination. In Handbook of Pathogens and Diseases in Cephalopods; Springer: Berlin, Germany, 2019; pp. 11–38. [Google Scholar]
- Watanuki, N.; Kawamura, G.; Kaneuchi, S.; Iwashita, T. Role of vision in behavior, visual field, and visual acuity of cuttlefish Sepia esculenta. Fish. Sci. 2000, 66, 417–423. [Google Scholar] [CrossRef]
- Williamson, R. A sensory basis for orientation in cephalopods. J. Mar. Biol. Assoc. U.K. 1995, 75, 83–92. [Google Scholar] [CrossRef]
- Packard, A. The behaviour of Octopus vulgaris. Bulletin de l’Institut océanographique 1963, Numéro spécial 1D. 35–49. [Google Scholar]
- Borrelli, L. Testing the Contribution of Relative Brain Size and Learning Capabilities on the Evolution of Octopus Vulgaris and Other Cephalopods. Ph.D. Thesis, Stazione Zoologica Anton Dohrn, Italy & Open University, Rome, Italy, 2007. [Google Scholar]
- Borrelli, L.; Gherardi, F.; Fiorito, G. A Catalogue of Body Patterning in Cephalopoda; Stazione Zoologica A. Dohrn; Firenze University Press: Napoli, Italy, 2006. [Google Scholar]
- Hanlon, R.T.; Messenger, J.B. Cephalopod Behaviour, 2nd ed.; Cambridge University Press: Cambridge, UK, 2018. [Google Scholar]
- Duval, P.; Chichery, M.-P.; Chichery, R. Prey capture by the cuttlefish (Sepia officinalis L): An experimental study of two strategies. Behav. Process. 1984, 9, 13–21. [Google Scholar] [CrossRef]
- Messenger, J.B. Learning in the cuttlefish, Sepia. Anim. Behav. 1973, 21, 801–826. [Google Scholar] [CrossRef]
- Messenger, J.B. The visual attack of the cuttlefish, Sepia officinalis. Anim. Behav. 1968, 16, 342–357. [Google Scholar] [CrossRef]
- Schnell, A.K.; Jozet-Alves, C.; Hall, K.C.; Radday, L.; Hanlon, R.T. Fighting and mating success in giant Australian cuttlefish is influenced by behavioural lateralization. Proc. R. Soc. B 2019, 286, 20182507. [Google Scholar] [CrossRef] [PubMed]
- Schnell, A.K.; Hanlon, R.T.; Benkada, A.; Jozet-Alves, C. Lateralization of eye use in cuttlefish: Opposite direction for anti-predatory and predatory behaviors. Front. Physiol. 2016, 7, 620. [Google Scholar] [CrossRef] [PubMed]
- Schnell, A.K.; Bellanger, C.; Vallortigara, G.; Jozet-Alves, C. Visual asymmetries in cuttlefish during brightness matching for camouflage. Curr. Biol. 2018, 28, R925–R926. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.; Chichery, R.; Boal, J.G.; Dickel, L. Orientation in the cuttlefish Sepia officinalis: Response versus place learning. Anim. Cogn. 2007, 10, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, H. The positive learning process in Octopus vulgaris. Z. Vgl. Physiol. 1963, 47, 191–214. [Google Scholar] [CrossRef]
- Maldonado, H. The visual attack learning system in Octopus vulgaris. J. Theor. Biol. 1963, 5, 470–488. [Google Scholar] [CrossRef]
- Byrne, R.A.; Kuba, M.; Griebel, U. Lateral asymmetry of eye use in Octopus vulgaris. Anim. Behav. 2002, 64, 461–468. [Google Scholar] [CrossRef]
- Byrne, R.A.; Kuba, M.J.; Meisel, D.V. Lateralized eye use in Octopus vulgaris shows antisymmetrical distribution. Anim. Behav. 2004, 68, 1107–1114. [Google Scholar] [CrossRef]
- Fiorito, G.; von Planta, C.; Scotto, P. Problem solving ability of Octopus vulgaris Lamarck (Mollusca, Cephalopoda). Behav. Neural Biol. 1990, 53, 217–230. [Google Scholar] [CrossRef]
- Moriyama, T.; Gunji, Y.P. Autonomous learning in maze solution by Octopus. Ethology 1997, 103, 499–513. [Google Scholar] [CrossRef]
- Bierens de Haan, J.A. Versuche ueber den Farbensinn und das psychische Leben von Octopus vulgaris. Z. Vgl. Physiol. 1926, 4, 766–796. [Google Scholar] [CrossRef]
- Boycott, B.B. Learning in Octopus vulgaris and other cephalopods. Pubbl. Staz. Zool. Napoli. 1954, 25, 67–93. [Google Scholar]
- Buytendijk, F.J.J. Das Verhalten von Octopus nach teilweiser zerstörung des “Gehims”. Arch. Neert. Physiol. 1933, 18, 24–70. [Google Scholar]
- Wells, M.J. Learning and movement in octopuses. Anim. Behav 1965, (Suppl. 1), 115–128. [Google Scholar]
- Hvorecny, L.M.; Grudowski, J.L.; Blakeslee, C.J.; Simmons, T.L.; Roy, P.R.; Brooks, J.A.; Hanner, R.M.; Beigel, M.E.; Karson, M.A.; Nichols, R.H.; et al. Octopuses (Octopus bimaculoides) and cuttlefishes (Sepia pharaonis, S. officinalis) can conditionally discriminate. Anim. Cogn. 2007, 10, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Boal, J.G.; Dunham, A.W.; Williams, K.T.; Hanlon, R.T. Experimental evidence for spatial learning in octopuses (Octopus bimaculoides). J. Comp. Psychol. 2000, 114, 246. [Google Scholar] [CrossRef]
- Amodio, P.; Andrews, P.; Salemme, M.; Ponte, G.; Fiorito, G. The Use of Artificial Crabs for Testing Predatory Behavior and Health in the Octopus. Altex-Altern. Anim. Exp. 2014, 31, 494–499. [Google Scholar]
- Fiorito, G.; Scotto, P. Observational Learning in Octopus vulgaris. Science 1992, 256, 545–547. [Google Scholar] [CrossRef]
- Forrester, G.S.; Leavens, D.A.; Quaresmini, C.; Vallortigara, G. Target animacy influences gorilla handedness. Anim. Cogn. 2011, 14, 903–907. [Google Scholar] [CrossRef][Green Version]
- Forrester, G.S.; Quaresmini, C.; Leavens, D.A.; Spiezio, C.; Vallortigara, G. Target animacy influences chimpanzee handedness. Anim. Cogn. 2012, 15, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Bisazza, A.; Facchin, L.; Pignatti, R.; Vallortigara, G. Lateralization of detour behaviour in poeciliid fish: The effect of species, gender and sexual motivation. Behav. Brain Res. 1998, 91, 157–164. [Google Scholar] [CrossRef]
- Bisazza, A.; Pignatti, R.; Vallortigara, G. Detour tests reveal task-and stimulus-specific behavioural lateralization in mosquitofish (Gambusia holbrooki). Behav. Brain Res. 1997, 89, 237–242. [Google Scholar] [CrossRef]
- Ghirlanda, S.; Vallortigara, G. The evolution of brain lateralization: A game-theoretical analysis of population structure. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2004, 271, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Ghirlanda, S.; Frasnelli, E.; Vallortigara, G. Intraspecific competition and coordination in the evolution of lateralization. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.; Frasnelli, E.; Versace, E. Lateralized antennal control of aggression and sex differences in red mason bees, Osmia bicornis. Sci. Rep. 2016, 6, 29411. [Google Scholar] [CrossRef]
- Jozet-Alves, C.; Viblanc, V.A.; Romagny, S.; Dacher, M.; Healy, S.D.; Dickel, L. Visual lateralization is task and age dependent in cuttlefish, Sepia officinalis. Anim. Behav. 2012, 83, 1313–1318. [Google Scholar] [CrossRef]
- Jozet-Alves, C.; Romagny, S.; Bellanger, C.; Dickel, L. Cerebral correlates of visual lateralization in Sepia. Behav. Brain Res. 2012, 234, 20–25. [Google Scholar] [CrossRef]
- Frasnelli, E.; Vallortigara, G. Individual-level and population-level lateralization: Two sides of the same coin. Symmetry 2018, 10, 739. [Google Scholar] [CrossRef]


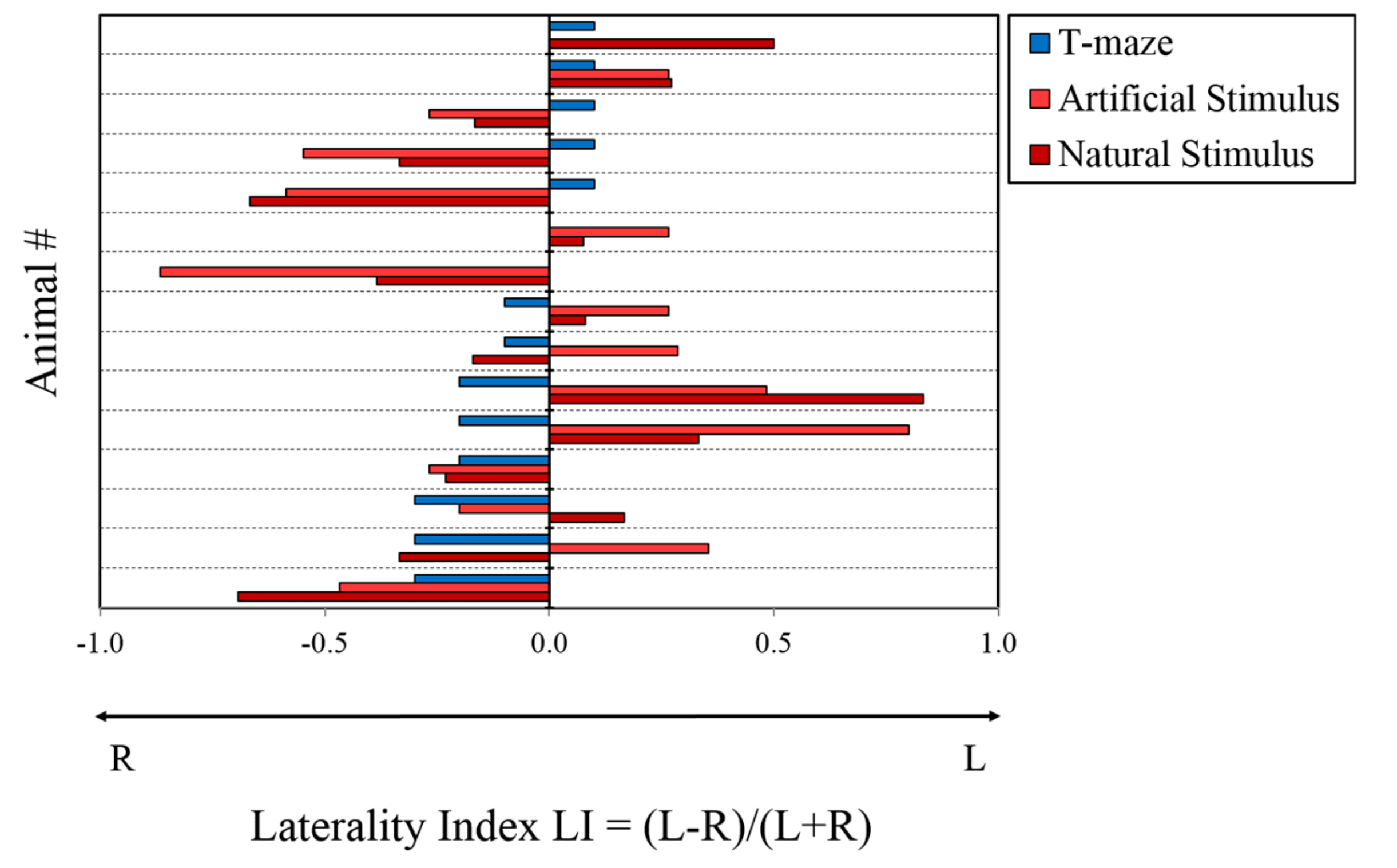
| Animal # | Natural Stimulus | Artificial Stimulus | T-Maze | ||||||
|---|---|---|---|---|---|---|---|---|---|
| LI | Z-Score | p | LI | Z-Score | p | LI | Z-score | p | |
| 1 | 0.50 | 1.44 | 0.150 | 0.00 | 0.00 | 1.000 | 0.10 | 0.22 | 0.820 |
| 2 | 0.17 | 0.29 | 0.770 | −0.20 | −0.91 | 0.360 | −0.30 | −1.12 | 0.260 |
| 3 | 0.27 | 0.60 | 0.550 | 0.27 | 1.28 | 0.200 | 0.10 | 0.22 | 0.820 |
| 4 | 0.83 | 2.60 | 0.010 * | 0.48 | 2.51 | 0.010 * | −0.20 | −0.67 | 0.500 |
| 5 | −0.33 | −0.87 | 0.390 | 0.35 | 1.80 | 0.070 | −0.30 | −1.12 | 0.260 |
| 6 | −0.33 | −0.87 | 0.390 | −0.55 | −2.87 | 0.003 ** | 0.10 | 0.22 | 0.820 |
| 7 | 0.33 | 0.87 | 0.390 | 0.80 | 4.20 | >0.001 *** | −0.20 | −0.67 | 0.500 |
| 8 | 0.08 | 0.00 | 1.000 | 0.27 | 1.28 | 0.200 | −0.10 | −0.22 | 0.820 |
| 9 | −0.17 | 0.29 | 0.770 | 0.29 | 1.32 | 0.180 | −0.10 | −0.22 | 0.820 |
| 10 | −0.67 | 2.02 | 0.040 * | −0.59 | −2.97 | 0.002 ** | 0.10 | 0.22 | 0.820 |
| 11 | 0.08 | 0.00 | 1.000 | 0.27 | 1.28 | 0.200 | 0.00 | 0.00 | 1.000 |
| 12 | −0.69 | 2.22 | 0.020 * | −0.47 | −2.37 | 0.020 | −0.30 | −1.12 | 0.260 |
| 13 | −0.38 | 1.11 | 0.270 | −0.87 | −4.56 | >0.001 *** | 0.00 | 0.00 | 1.000 |
| 14 | −0.23 | 0.55 | 0.580 | −0.27 | −1.28 | 0.200 | −0.20 | −0.67 | 0.500 |
| 15 | −0.17 | 0.29 | 0.770 | −0.27 | −1.28 | 0.200 | 0.10 | 0.22 | 0.820 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frasnelli, E.; Ponte, G.; Vallortigara, G.; Fiorito, G. Visual Lateralization in the Cephalopod Mollusk Octopus vulgaris. Symmetry 2019, 11, 1121. https://doi.org/10.3390/sym11091121
Frasnelli E, Ponte G, Vallortigara G, Fiorito G. Visual Lateralization in the Cephalopod Mollusk Octopus vulgaris. Symmetry. 2019; 11(9):1121. https://doi.org/10.3390/sym11091121
Chicago/Turabian StyleFrasnelli, Elisa, Giovanna Ponte, Giorgio Vallortigara, and Graziano Fiorito. 2019. "Visual Lateralization in the Cephalopod Mollusk Octopus vulgaris" Symmetry 11, no. 9: 1121. https://doi.org/10.3390/sym11091121
APA StyleFrasnelli, E., Ponte, G., Vallortigara, G., & Fiorito, G. (2019). Visual Lateralization in the Cephalopod Mollusk Octopus vulgaris. Symmetry, 11(9), 1121. https://doi.org/10.3390/sym11091121






