Identifying Opportunities to Conserve Farm Ponds on Private Lands: Integration of Social, Ecological, and Historical Data
Abstract
1. Introduction
1.1. Balancing Agricultural and Ecological Functions in Farm Ponds
1.2. The Importance of Wildlife Values and Attitudes for Management
1.3. Objectives
2. Materials and Methods
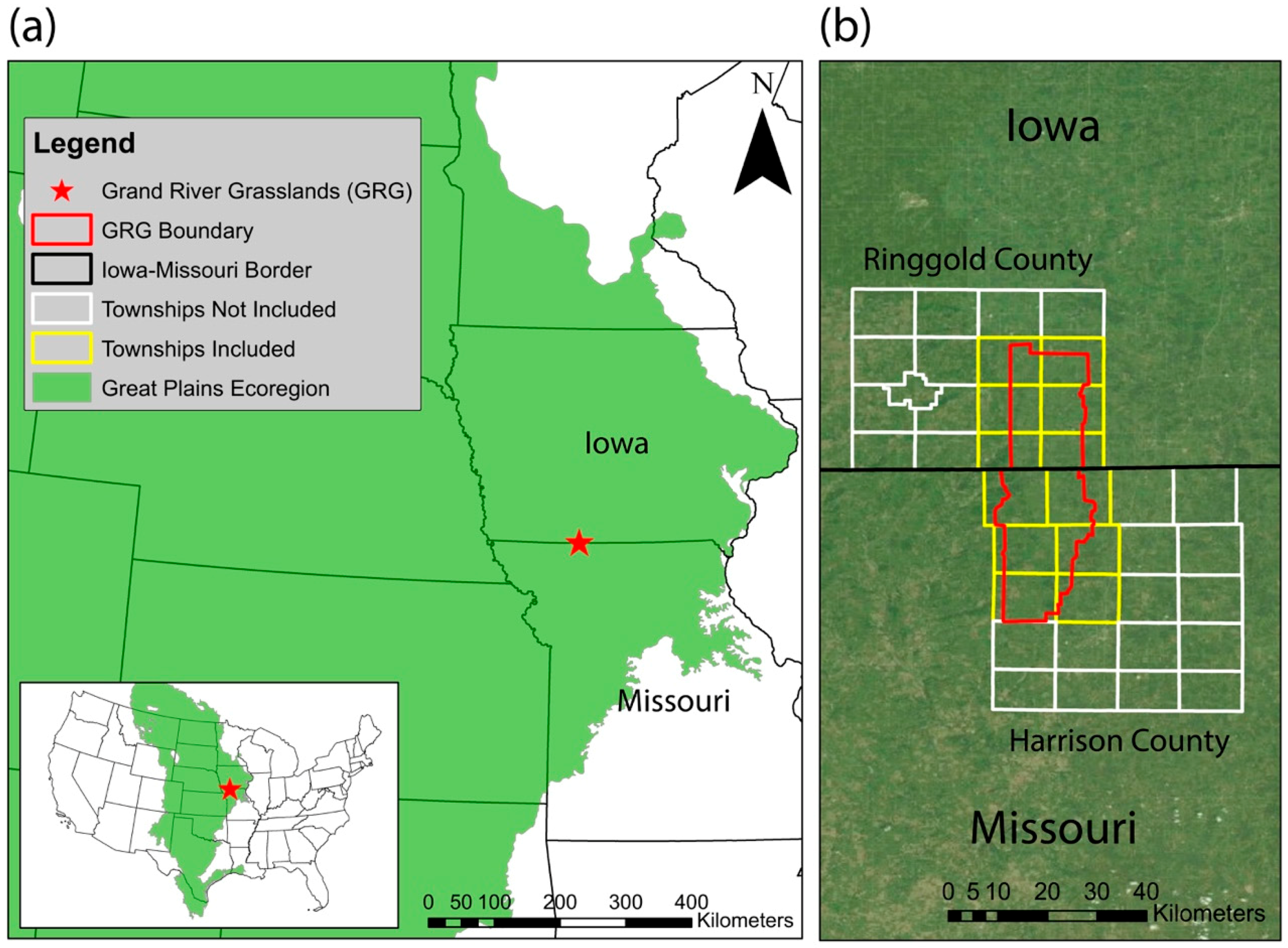
2.1. Study Region
2.2. Landowner Survey Methods
2.3. Imagery Acquisition & Pond Age Estimation
2.4. Pond Management Activity
2.5. Analysis
2.5.1. Determining Bias in Sample, Multiple Imputation of Landowner Results
2.5.2. Regression Analyses of Pond Management Activity
3. Results
3.1. Landowner Survey Respondents
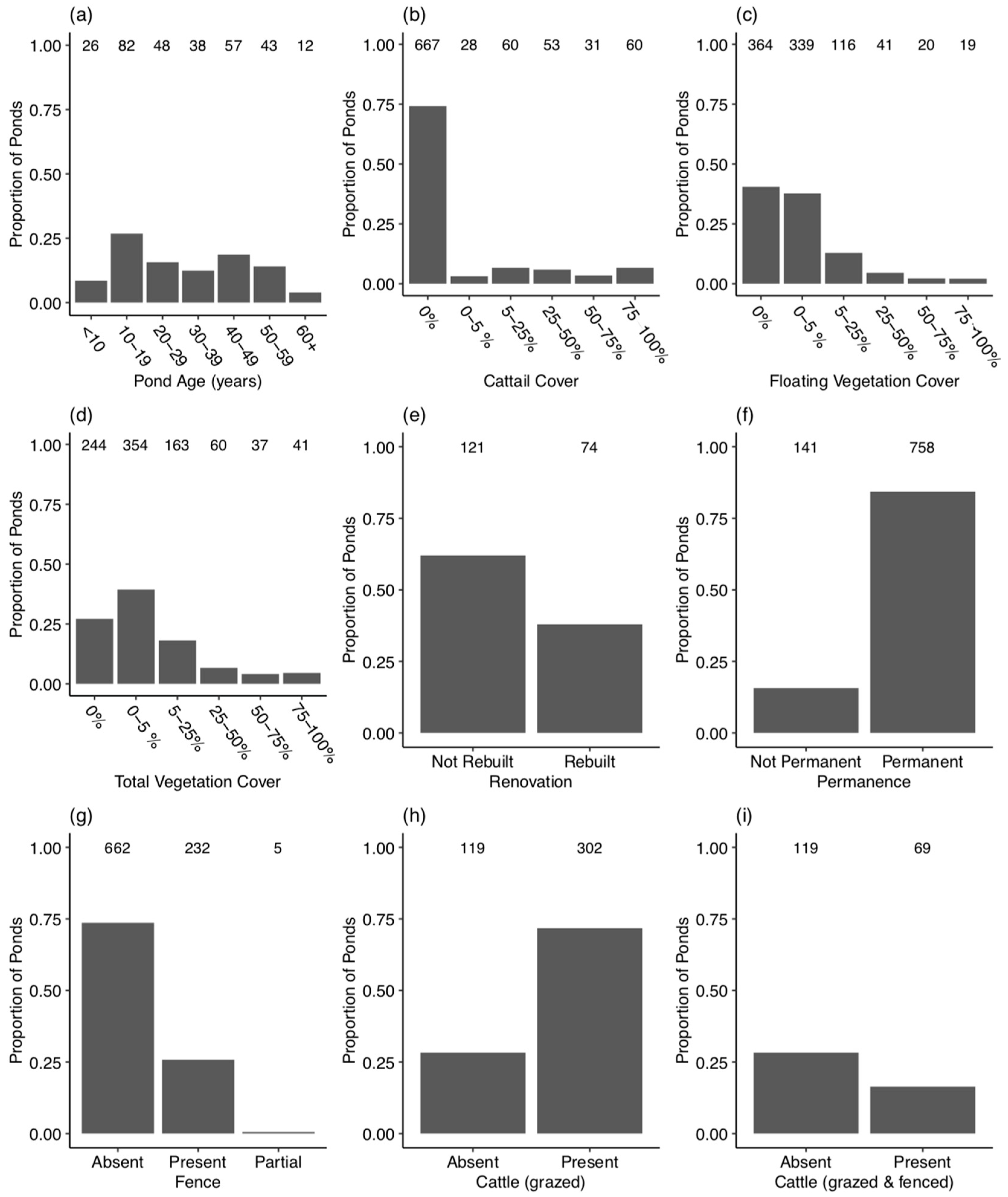
3.2. Pond Habitat and Management Characteristics
3.3. Mixed Effects Regression Models
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tscharntke, T.; Klein, A.M.; Kruess, A.; Steffan-Dewenter, I.; Thies, C. Landscape perspectives on agricultural intensification and biodiversity–ecosystem service management. Ecol. Lett. 2005, 8, 857–874. [Google Scholar] [CrossRef]
- Mora, C.; Sale, P.F. Ongoing global biodiversity loss and the need to move beyond protected areas: A review of the technical and practical shortcomings of protected areas on land and sea. Mar. Ecol. Prog. Ser. 2011, 434, 251–266. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Van Houtan, K.S.; Pimm, S.L.; Sexton, J.O. US protected lands mismatch biodiversity priorities. Proc. Natl. Acad. Sci. USA 2015, 112, 5081–5086. [Google Scholar] [CrossRef] [PubMed]
- Bigelow, D.P.; Borchers, A. Major Uses of Land in the United States. 2012. Available online: https://www.ers.usda.gov/publications/pub-details/?pubid=84879 (accessed on 22 August 2019).
- Cannavò, P.F. The Working Landscape: Founding, Preservation and the Politics of Place; MIT Press: Cambridge, Massachusetts, USA, 2007; p. 425. [Google Scholar]
- Maresh Nelson, S.B.; Coon, J.J.; Duchardt, C.J.; Miller, J.R.; Debinski, D.M.; Schacht, W.H. Contrasting impacts of invasive plants and human-altered landscape context on nest survival and brood parasitism of a grassland bird. Landsc. Ecol. 2018, 33, 1799–1813. [Google Scholar] [CrossRef]
- Shew, J.J.; Nielsen, C.K.; Sparling, D.W. Finer-scale habitat predicts nest survival in grassland birds more than management and landscape: A multi-scale perspective. J. Appl. Ecol. 2019, 56, 929–945. [Google Scholar] [CrossRef]
- Chumchal, M.M.; Drenner, R.W. An environmental problem hidden in plain sight? Small Human-made ponds, emergent insects, and mercury contamination of biota in the Great Plains. Environ. Toxicol. Chem. 2015, 34, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Compton, L.V. Farm and ranch ponds. J. Wildl. Manag. 1952, 16, 238–242. [Google Scholar] [CrossRef]
- Gallant, A.L.; Sadinski, W.; Roth, M.F.; Rewa, C.A. Changes in historical Iowa land cover as context for assessing the environmental benefits of current and future conservation efforts on agricultural lands. J. Soil Water Conserv. 2011, 66, 67A–77A. [Google Scholar] [CrossRef]
- Chumchal, M.M.; Drenner, R.W.; Adams, K.J. Abundance and size distribution of permanent and temporary farm ponds in the southeastern Great Plains. Inland Waters 2016, 6, 258–264. [Google Scholar] [CrossRef]
- Renwick, W.H.; Sleezer, R.O.; Buddemeier, R.W.; Smith, S.V. Small artificial ponds in the United States: Impacts on sedimentation and carbon budget. In Proceedings of the Eighth Federal Interagency Sedimentation Conference, Reno, NV, USA, 2–6 April 2006; pp. 73–744. [Google Scholar]
- Knutson, M.G.; Richardson, W.B.; Reineke, D.M.; Gray, B.R.; Parmelee, J.R.; Weick, S.E. Agricultural ponds support amphibian populations. Ecol. Appl. 2004, 14, 669–684. [Google Scholar] [CrossRef]
- Shulse, C.D.; Semlitsch, R.D.; Trauth, K.M.; Williams, A.D. Influences of design and landscape placement parameters on amphibian abundance in constructed wetlands. Wetlands 2010, 30, 915–928. [Google Scholar] [CrossRef]
- Swartz, T.M.; Miller, J.R. Managing farm ponds as breeding sites for amphibians: Key trade-offs in agricultural function and habitat conservation. Ecol. Appl. 2019. [Google Scholar] [CrossRef] [PubMed]
- Trimble, S.W.; Mendel, A.C. The cow as a geomorphic agent—A critical review. Geomorphology 1995, 13, 233–253. [Google Scholar] [CrossRef]
- Trimble, S.W. Erosional effects of cattle on streambanks in Tennessee, USA. Earth Surf. Process. Landf. 1994, 19, 451–464. [Google Scholar] [CrossRef]
- Giuliano, W.M. Should I fence the streams, ponds and wetlands in my pastures? Fencing pasture streams, ponds and wetlands can improve fish and wildlife habitat and benefit agricultural landowners. Rangelands 2006, 28, 29–31. [Google Scholar] [CrossRef]
- Reinartz, J.A.; Warne, E.L. Development of vegetation in small created wetlands in southeastern Wisconsin. Wetlands 1993, 13, 153–164. [Google Scholar] [CrossRef]
- Berg, M.D.; Popescu, S.C.; Wilcox, B.P.; Angerer, J.P.; Rhodes, E.C.; McAlister, J.; Fox, W.E. Small farm ponds: Overlooked features with important impacts on watershed sediment transport. J. Am. Water Resour. Assoc. 2016, 52, 67–76. [Google Scholar] [CrossRef]
- Schonrock, A.E. An Internet Survey of Private Pond Owners and Managers in Texas; Texas A&M: College Station, TX, USA, 2005. [Google Scholar]
- Goldberg, C.S.; Waits, L.P. Using habitat models to determine conservation priorities for pond-breeding amphibians in a privately-owned landscape of northern Idaho, USA. Biol. Conserv. 2009, 142, 1096–1104. [Google Scholar] [CrossRef]
- Greenland-Smith, S.; Brazner, J.; Sherren, K. Farmer perceptions of wetlands and waterbodies: Using social metrics as an alternative to ecosystem service valuation. Ecol. Econ. 2016, 126, 58–69. [Google Scholar] [CrossRef]
- Bennett, N.J.; Roth, R.; Klain, S.C.; Chan, K.; Christie, P.; Clark, D.A.; Cullman, G.; Curran, D.; Durbin, T.J.; Epstein, G. Conservation social science: Understanding and integrating human dimensions to improve conservation. Biol. Conserv. 2017, 205, 93–108. [Google Scholar] [CrossRef]
- Teel, T.L.; Manfredo, M.J.; Jensen, F.S.; Buijs, A.E.; Fischer, A.; Riepe, C.; Arlinghaus, R.; Jacobs, M.H. Understanding the cognitive basis for human-wildlife relationships as a key to successful protected-area management. Int. J. Sociol. 2010, 40, 104–123. [Google Scholar] [CrossRef]
- Heberlein, T.A. Navigating environmental attitudes. Conserv. Biol. 2012, 26, 583–585. [Google Scholar] [CrossRef] [PubMed]
- Ajzen, I. The theory of planned behavior. Organ. Behav. Hum. Decis. Process. 1991, 50, 179–211. [Google Scholar] [CrossRef]
- Ferranto, S.; Huntsinger, L.; Getz, C.; Lahiff, M.; Stewart, W.; Nakamura, G.; Kelly, M. Management without borders? A survey of landowner practices and attitudes toward cross-boundary cooperation. Soc. Nat. Resour. 2013, 26, 1082–1100. [Google Scholar] [CrossRef]
- Stedman, R.C. Is it really just a social construction? The contribution of the physical environment to sense of place. Soc. Nat. Resour. 2003, 16, 671–685. [Google Scholar] [CrossRef]
- Treves, A.; Naughton-Treves, L.; Shelley, V. Longitudinal analysis of attitudes toward wolves. Conserv. Biol. 2013, 27, 315–323. [Google Scholar] [CrossRef]
- Doney, E.; Bath, A.; Vaske, J. Understanding conflict and consensus regarding wood bison management in Alaska, USA. Wildl. Res. 2018, 45, 229–236. [Google Scholar] [CrossRef]
- Teel, T.L.; Manfredo, M.J. Understanding the diversity of public interests in wildlife conservation. Conserv. Biol. 2010, 24, 128–139. [Google Scholar] [CrossRef]
- Lokocz, E.; Ryan, R.L.; Sadler, A.J. Motivations for land protection and stewardship: Exploring place attachment and rural landscape character in Massachusetts. Landsc. Urban. Plan. 2011, 99, 65–76. [Google Scholar] [CrossRef]
- Reimer, A.P.; Thompson, A.W.; Prokopy, L.S. The multi-dimensional nature of environmental attitudes among farmers in Indiana: Implications for conservation adoption. Agric. Hum. Values 2012, 29, 29–40. [Google Scholar] [CrossRef]
- Klain, S.C.; Olmsted, P.; Chan, K.M.; Satterfield, T. Relational values resonate broadly and differently than intrinsic or instrumental values, or the New Ecological Paradigm. PLoS ONE 2017, 12, e0183962. [Google Scholar] [CrossRef]
- Swartz, T.M. Farm Ponds of the Eastern Great Plains: Key Challenges and Opportunities for Conserving Amphibians in These Novel Ecosystems; University of Illinois at Urbana-Champaign: Urbana, IL, USA, 2018. [Google Scholar]
- Miller, J.R.; Morton, L.W.; Engle, D.M.; Debinski, D.M.; Harr, R.N. Nature reserves as catalysts for landscape change. Front. Ecol. Environ. 2012, 10, 144–152. [Google Scholar] [CrossRef]
- Morton, L.W.; Regen, E.; Engle, D.M.; Miller, J.R.; Harr, R.N. Perceptions of landowners concerning conservation, grazing, fire, and eastern redcedar management in tallgrass prairie. Rangel. Ecol. Manag. 2010, 63, 645–654. [Google Scholar] [CrossRef]
- Raynor, E.J.J.C.J.; Swartz, T.M.; Morton, L.W.; Schacht, W.H.; MIller, J.R. Shifting cattle producer beliefs on stocking and invasive forage: Implications for grassland conservation. Rangel. Ecol. Manag. (In Review).
- Coon, J.J.; Morton, L.W.; Miller, J.R. A Survey of Landowners in the Grand River Grasslands: Managing Wildlife, Cattle and Non-Native Plants.; University of Illinois Department of Natural Resources and Environmental Sciences: Urbana, IL, USA, 2018. [Google Scholar]
- Dillman, D.A.; Smyth, J.D.; Christian, L.M. Internet, Phone, Mail and Mixed-Mode Surveys: The Tailored Design Method, 4th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2014; p. 528. [Google Scholar]
- Lannoo, M. A fish fry: the role of exotic species and aquacultural practices in producing amphibian declines in the upper Midwest. In Minnesota’s Amphibians and Reptiles: Conservation and Status; Moriarty, J.J., Jones, D., Eds.; Serpent’s Tale Press: Excelsior, MN, USA, 1996; pp. 25–27. [Google Scholar]
- Perry, K.; Jayne, D.; Smith, M.; Branson, A. Missouri Pond Handbook; Missouri Department of Conservation: Jefferson City, MO, USA, 2015; p. 72. [Google Scholar]
- Chadde, S.W. A Great Lakes Wetland Flora: A Complete Guide to the Wetland and Aquatic Plants of the Midwest, 4th ed.; CreateSpace Independent Publishing Platform: Scotts Valley, CA, USA, 2012; p. 690. [Google Scholar]
- Miklovic, S. Typha Angustifolia Management: Implications for glacial marsh restoration. Restor. Reclam. Rev. 2000, 6, 1–11. [Google Scholar]
- Stevens, M.; Hoag, C. Plant. Guide: Broad-Leaved Cattail, Typha Latifolia; U.S. Department of Agriculture Natural Resources Conservation Service: Davis, CA, USA, 2003; pp. 1–2.
- Stevens, M.; Hoag, C. Plant. Guide: Narrowleaf Cattail, Typha angustifolia L; U.S. Department of Agriculture Natural Resources Conservation Service: Davis, CA, USA, 2006; pp. 1–4.
- Weller, M. Studies of cattail in relation to management for marsh wildlife. Iowa State J. Res. 1975, 49, 383–412. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Model. Selection and Multimodel Inference: A Practical Information-Theoretical Approach, 2nd ed.; Springer: New York, NY, USA, 2002; p. 488. [Google Scholar]
- Arnold, T.W. Uninformative parameters and model selection using Akaike’s Information Criterion. J. Wild. Manag. 2010, 74, 1175–1178. [Google Scholar] [CrossRef]
- R Core Team. R: A language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Mazerolle, M.J. AICcmodavg: Model. Selection and Multimodel Inference Based on (Q)AIC(c). R package version 2.1-1. Available online: https://cran.r-project.org/package=AICcmodavg (accessed on 22 August 2019).
- Bates, D.; Maechler, M.; Bolker, B.M.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Coon, J.J.; van Riper, C.J.; Morton, L.W.; Miller, J.R. Assessing non-response bias in a survey conducted in the rural Midwest. (In review)
- Cowardin, L.M.; Myers, V.I. Remote sensing for identification and classification of wetland vegetation. J. Wildl. Manag. 1974, 38, 308–314. [Google Scholar] [CrossRef]
- Bradbury, R.B.; Hill, R.A.; Mason, D.C.; Hinsley, S.A.; Wilson, J.D.; Balzter, H.; Anderson, G.Q.; Whittingham, M.J.; Davenport, I.J.; Bellamy, P.E. Modelling relationships between birds and vegetation structure using airborne LiDAR data: A review with case studies from agricultural and woodland environments. Ibis 2005, 147, 443–452. [Google Scholar] [CrossRef]
- Barrett, W. Frogging in Iowa. Ann. Iowa 1964, 37, 362–365. [Google Scholar] [CrossRef]
- Prokopy, L.S.; Floress, K.; Klotthor-Weinkauf, D.; Baumgart-Getz, A. Determinants of agricultural best management practice adoption: Evidence from the literature. J. Soil Water Conserv. 2008, 63, 300–311. [Google Scholar] [CrossRef]
- Lutter, S.H.; Dayer, A.A.; Rodewald, A.D.; McNeil, D.J.; Larkin, J.L. Early successional forest management on private lands as a coupled human and natural system. Forests 2019, 10, 499. [Google Scholar] [CrossRef]
- Porej, D.; Hetherington, T.E. Designing wetlands for amphibians: The importance of predatory fish and shallow littoral zones in structuring of amphibian communities. Wetl. Ecol. Manag. 2005, 13, 445–455. [Google Scholar] [CrossRef]
- Shulse, C.D.; Semlitsch, R.D.; Trauth, K.M.; Gardner, J.E. Testing wetland features to increase amphibian reproductive success and species richness for mitigation and restoration. Ecol. Appl. 2012, 22, 1675–1688. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Arano, K.G. Determinants of private forest management decisions: A study on West Virginia NIPF landowners. For. Policy Econ. 2009, 11, 118–125. [Google Scholar] [CrossRef]
- Langpap, C. Conservation incentives programs for endangered species: An analysis of landowner participation. Land Econ. 2004, 80, 375–388. [Google Scholar] [CrossRef]
- Skelly, D.K. Pond drying, predators, and the distribution of Pseudacris tadpoles. Copeia 1996, 599–605. [Google Scholar] [CrossRef]
- Pechmann, J.H.; Scott, D.E.; Gibbons, J.W.; Semlitsch, R.D. Influence of wetland hydroperiod on diversity and abundance of metamorphosing juvenile amphibians. Wetl. Ecol. Manag. 1989, 1, 3–11. [Google Scholar] [CrossRef]
- NRCS. Farm Pond Ecosystems; United States Department of Agriculture: Washington, DC, USA, 2006; p. 12.
- Huggins, D.; Kastens, J.; Baker, D.; Freeman, C. Conversion of Existing Farm Ponds to Wetlands in Agricultural Landscapes for Mitigation, Land Use Treatment and Conservation with a Perspective Toward Climate Change, No. 189; University of Kansas: Lawrence, KS, USA, June 2017; p. 91. [Google Scholar]
- Ribeiro, P.F.; Santos, J.L.; Santana, J.; Reino, L.; Beja, P.; Moreira, F. An applied farming systems approach to infer conservation-relevant agricultural practices for agri-environment policy design. Land Use Policy 2016, 58, 165–172. [Google Scholar] [CrossRef]




| Dependent Variable | Description | N |
|---|---|---|
| Pond Age | Age of the pond calculated from dates of historical imagery | 306 |
| Cattails | Cover class of cattails along perimeter | 899 |
| Floating | Cover class of floating vegetation on pond surface | 899 |
| Total Vegetation | Cover class of total vegetation on pond surface | 899 |
| Permanent | Presence or absence of water in pond during height of 2012 drought | 899 |
| Renovation | For ponds built before 1990, status of pond as having been renovated or not | 195 |
| Cattle In (grazed) | Presence of cattle in area immediately surrounding the pond on grazed pastures | 421 |
| Cattle Out (grazed and fenced) | Absence of cattle in pond area for fenced ponds on grazed pastures | 188 |
| Fenced | Presence of fence around pond | 899 |
| Fenced (grazed) | Presence of fence around ponds on a grazed pasture | 421 |
| Predictor Variable | Survey Question or Metric |
|---|---|
| Gender | Gender |
| IncomeTotal | Annual household income before taxes |
| Education | What is the highest level of education you have completed? |
| Age | 2017 minus year born |
| PropIncomeLand | What portion of your household income comes from your land? (%) |
| AcresOwned | How many acres do you own, rent, or manage in Southern IA/Northern MO? |
| AcresRented | How many acres do you own, rent, or manage in Southern IA/Northern MO? |
| LiveonLand | Do you currently live on your land? Yes/No. If no, how far do you live from your land? (miles) |
| IncomeAg | How important to you are each of the following when deciding how to manage your land? Income from agriculture |
| Livestock | How important to you are each of the following when deciding how to manage your land? Livestock |
| PropertyVal | How important to you are each of the following when deciding how to manage your land? Property Values |
| EnjoyingLand | How important to you are each of the following when deciding how to manage your land? Quietly enjoying my land |
| Recreation | How important to you are each of the following when deciding how to manage your land? Recreation (hunting/fishing) |
| Crops | How important to you are each of the following when deciding how to manage your land? Row crops |
| Soil | How important to you are each of the following issues? Reducing soil erosion? |
| Biodiversity | How important to you are each of the following issues? Increasing biodiversity? |
| WildlifeHabitat | How important to you are each of the following issues? Protecting wildlife habitat? |
| Ducks | How important is it to you to have the following animals/insects on your land? Ducks (all types) |
| Bullfrogs | How important is it to you to have the following animals/insects on your land? Bullfrogs |
| OtherFrogs | How important is it to you to have the following animals/insects on your land? Other frogs |
| Sportfish | How important is it to you to have the following animals/insects on your land? Sport fish (e.g., bass) |
| ManageDucks | Would you change your land management to benefit them? Ducks (all types) |
| ManageBullfrogs | Would you change your land management to benefit them? Bullfrogs |
| ManageOtherFrogs | Would you change your land management to benefit them? Other frogs |
| ManageSportFish | Would you change your land management to benefit them? Sport fish (e.g., bass) |
| Ponds Owned | Number of ponds found on parcel of the pond’s owner |
| Response Variable | Predictor Variable | K | AIC | ΔAIC | wi | Log-Likelihood | Evidence Ratio |
|---|---|---|---|---|---|---|---|
| Pond Age | IncomeAg | 4 | 2724.36 | 0.00 | 0.09 | −1358.18 | 1.50 |
| Crops | 4 | 2724.87 | 0.52 | 0.07 | −1358.44 | 1.17 | |
| Null | 3 | 2724.93 | 0.58 | 0.06 | −1359.47 | — | |
| Cattails | Crops | 4 | 3317.19 | 0.00 | 0.77 | −1654.60 | 77.00 |
| Age | 4 | 3322.74 | 5.55 | 0.05 | −1657.37 | 5.00 | |
| IncomeAg | 4 | 3323.18 | 5.99 | 0.04 | −1657.59 | 4.00 | |
| IncomeTotal | 4 | 3324.14 | 6.95 | 0.02 | −1658.07 | 2.00 | |
| AcresRented | 4 | 3325.25 | 8.05 | 0.01 | −1658.62 | 1.00 | |
| WildlifeHabitat | 4 | 3325.81 | 8.62 | 0.01 | −1658.91 | 1.00 | |
| Livestock | 4 | 3326.20 | 9.01 | 0.01 | −1659.10 | 1.00 | |
| Null | 3 | 3326.31 | 9.12 | 0.01 | −1660.16 | — | |
| TotalVeg | IncomeTotal | 4 | 3025.68 | 0.00 | 0.15 | −1508.84 | 3.00 |
| IncomeAg | 4 | 3026.24 | 0.56 | 0.11 | −1509.12 | 2.20 | |
| Crops | 4 | 3026.78 | 1.10 | 0.09 | −1509.39 | 1.80 | |
| ManageSportFish | 4 | 3027.11 | 1.43 | 0.07 | −1509.55 | 1.40 | |
| Education | 4 | 3027.37 | 1.69 | 0.06 | −1509.68 | 1.20 | |
| Null | 3 | 3028.04 | 2.37 | 0.05 | −1511.02 | — | |
| Floating | Null | 3 | 2756.93 | 0.00 | 0.07 | −1375.47 | — |
| Renovated | ManageBullfrogs | 3 | 260.43 | 0.00 | 0.14 | −127.22 | 2.33 |
| Gender | 3 | 261.17 | 0.74 | 0.10 | −127.59 | 1.67 | |
| Crops | 3 | 262.20 | 1.76 | 0.06 | −128.10 | 1.00 | |
| Null | 2 | 262.29 | 1.85 | 0.06 | −129.14 | — | |
| Cattle In (grazed ponds) | OtherFrogs | 3 | 474.86 | 0.00 | 0.10 | −234.43 | 1.67 |
| Bullfrogs | 3 | 475.33 | 0.47 | 0.08 | −234.66 | 1.33 | |
| Age | 3 | 475.55 | 0.69 | 0.07 | −234.78 | 1.17 | |
| Null | 2 | 475.98 | 1.12 | 0.06 | −235.99 | — | |
| Cattle Out (grazed & fenced ponds) | Livestock | 3 | 239.39 | 0.00 | 0.09 | −116.69 | 1.50 |
| Bullfrogs | 3 | 239.76 | 0.38 | 0.08 | −116.88 | 1.33 | |
| OtherFrogs | 3 | 239.84 | 0.45 | 0.07 | −116.92 | 1.17 | |
| Age | 3 | 240.12 | 0.73 | 0.06 | −117.06 | 1.00 | |
| LiveonLand | 3 | 240.12 | 0.73 | 0.06 | −117.06 | 1.00 | |
| Null | 2 | 240.28 | 0.89 | 0.06 | −118.14 | — | |
| Fenced | Livestock | 3 | 916.15 | 0.00 | 1.00 | −455.08 | — |
| WildlifeHabitat | 3 | 943.29 | 27.14 | 0.00 | −468.65 | — | |
| LiveonLand | 3 | 948.94 | 32.79 | 0.00 | −471.47 | — | |
| Ducks | 3 | 949.74 | 33.59 | 0.00 | −471.87 | — | |
| Recreation | 3 | 950.01 | 33.86 | 0.00 | −472.01 | — | |
| Crops | 3 | 951.04 | 34.89 | 0.00 | −472.52 | — | |
| IncomeAg | 3 | 951.23 | 35.08 | 0.00 | −472.62 | — | |
| PropIncomeLand | 3 | 951.48 | 35.33 | 0.00 | −472.74 | — | |
| Null | 2 | 952.67 | 36.52 | 0.00 | −474.34 | — | |
| Fenced (grazed) | ManageSportFish | 3 | 563.45 | 0.00 | 0.09 | −278.72 | 1.50 |
| Crops | 3 | 563.66 | 0.21 | 0.08 | −278.83 | 1.33 | |
| SportFish | 3 | 564.00 | 0.55 | 0.07 | −279.00 | 1.17 | |
| Null | 2 | 564.31 | 0.87 | 0.06 | −280.16 | — | |
| Permanent | Ponds Owned | 3 | 773.96 | 0.00 | 0.34 | −383.98 | 6.80 |
| Education | 3 | 777.12 | 3.17 | 0.07 | −385.56 | 1.40 | |
| Null | 2 | 777.90 | 3.95 | 0.05 | −386.95 | — |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Swartz, T.M.; Coon, J.J.; Mattes, J.R.; Miller, J.R. Identifying Opportunities to Conserve Farm Ponds on Private Lands: Integration of Social, Ecological, and Historical Data. Land 2019, 8, 127. https://doi.org/10.3390/land8090127
Swartz TM, Coon JJ, Mattes JR, Miller JR. Identifying Opportunities to Conserve Farm Ponds on Private Lands: Integration of Social, Ecological, and Historical Data. Land. 2019; 8(9):127. https://doi.org/10.3390/land8090127
Chicago/Turabian StyleSwartz, Timothy M., Jaime J. Coon, Jenna R. Mattes, and James R. Miller. 2019. "Identifying Opportunities to Conserve Farm Ponds on Private Lands: Integration of Social, Ecological, and Historical Data" Land 8, no. 9: 127. https://doi.org/10.3390/land8090127
APA StyleSwartz, T. M., Coon, J. J., Mattes, J. R., & Miller, J. R. (2019). Identifying Opportunities to Conserve Farm Ponds on Private Lands: Integration of Social, Ecological, and Historical Data. Land, 8(9), 127. https://doi.org/10.3390/land8090127





