Distribution and Removal of Nonylphenol Ethoxylates and Nonylphenol from Textile Wastewater—A Comparison of a Cotton and a Synthetic Fiber Factory in Vietnam
Abstract
:1. Introduction
- To investigate the distribution and removal of NPEOs and NP across two typical textile wastewater treatment processes.
- To assess the influence of hydraulic retention time (HRT), nitrifying conditions, solids retention time (SRT), mixed liquor suspended solids (MLSS), and water temperature on the NPEO and NP removal.
2. Treatment Processes and Factors Influencing Nonylphenol and Nonylphenol Ethoxylate Removal
2.1. Removal by the Coagulation Process
2.2. Removal by Anaerobic Processes
2.3. Removal by Anoxic Processes
2.4. Removal by Aerobic Activated Processes
3. Materials and Methods
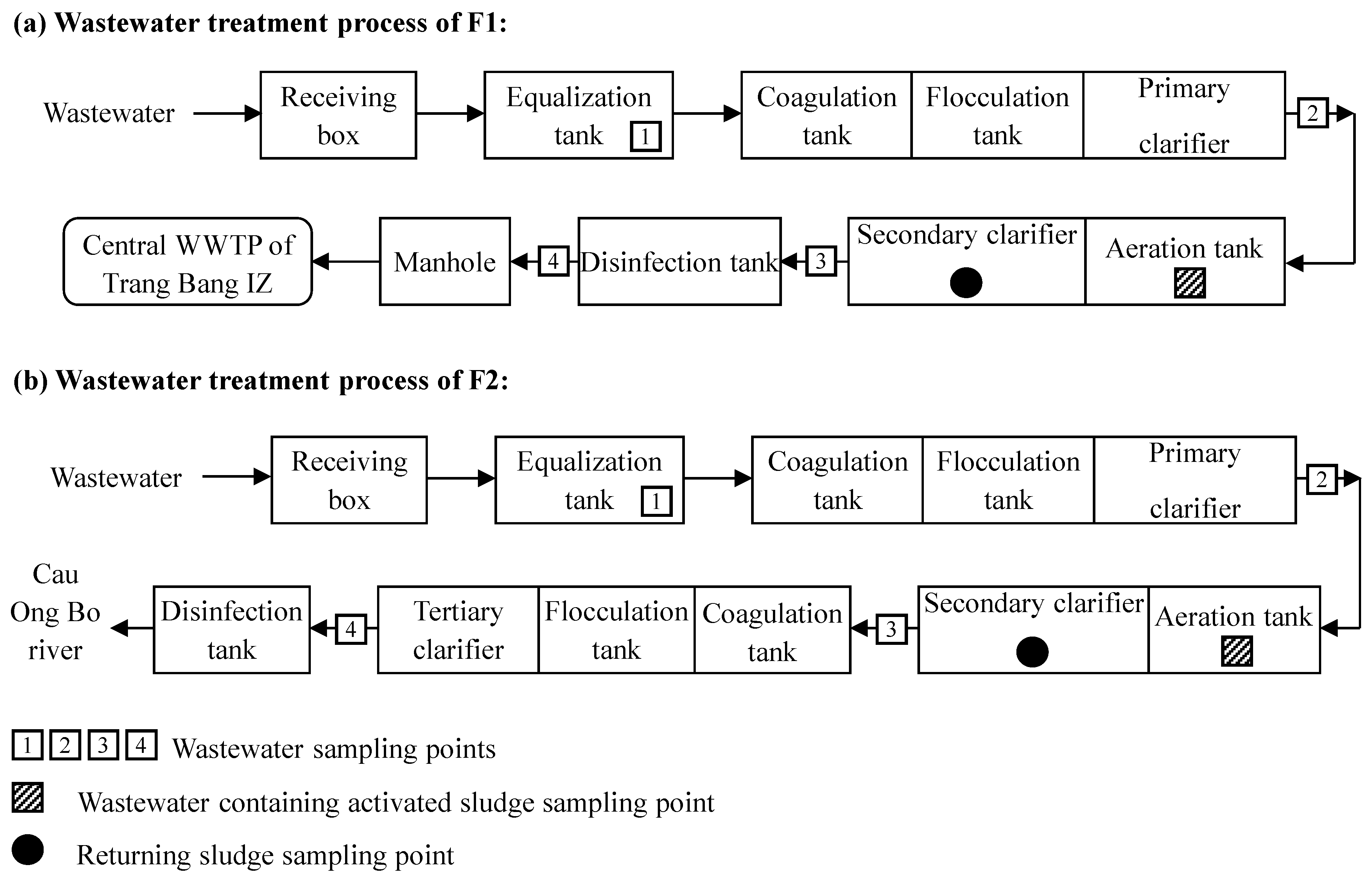
3.1. Selection of Wastewater Treatment Processes
3.2. Field Survey and Sampling
3.3. Method of Analysis
4. Results and Discussion
4.1. Design Figures and Operating Conditions of the Factories during the Campaign
4.2. Textile Wastewater Compositions
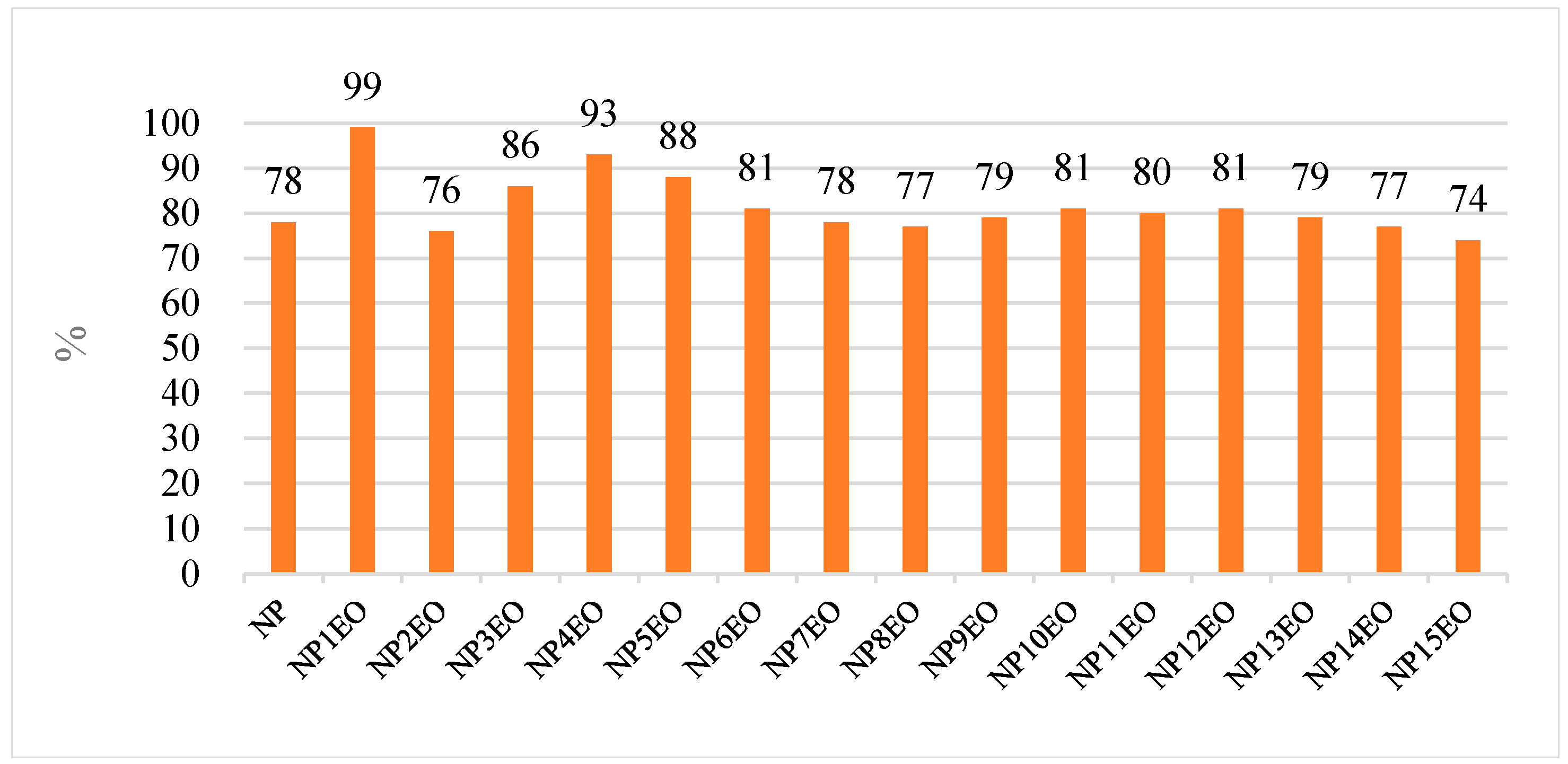
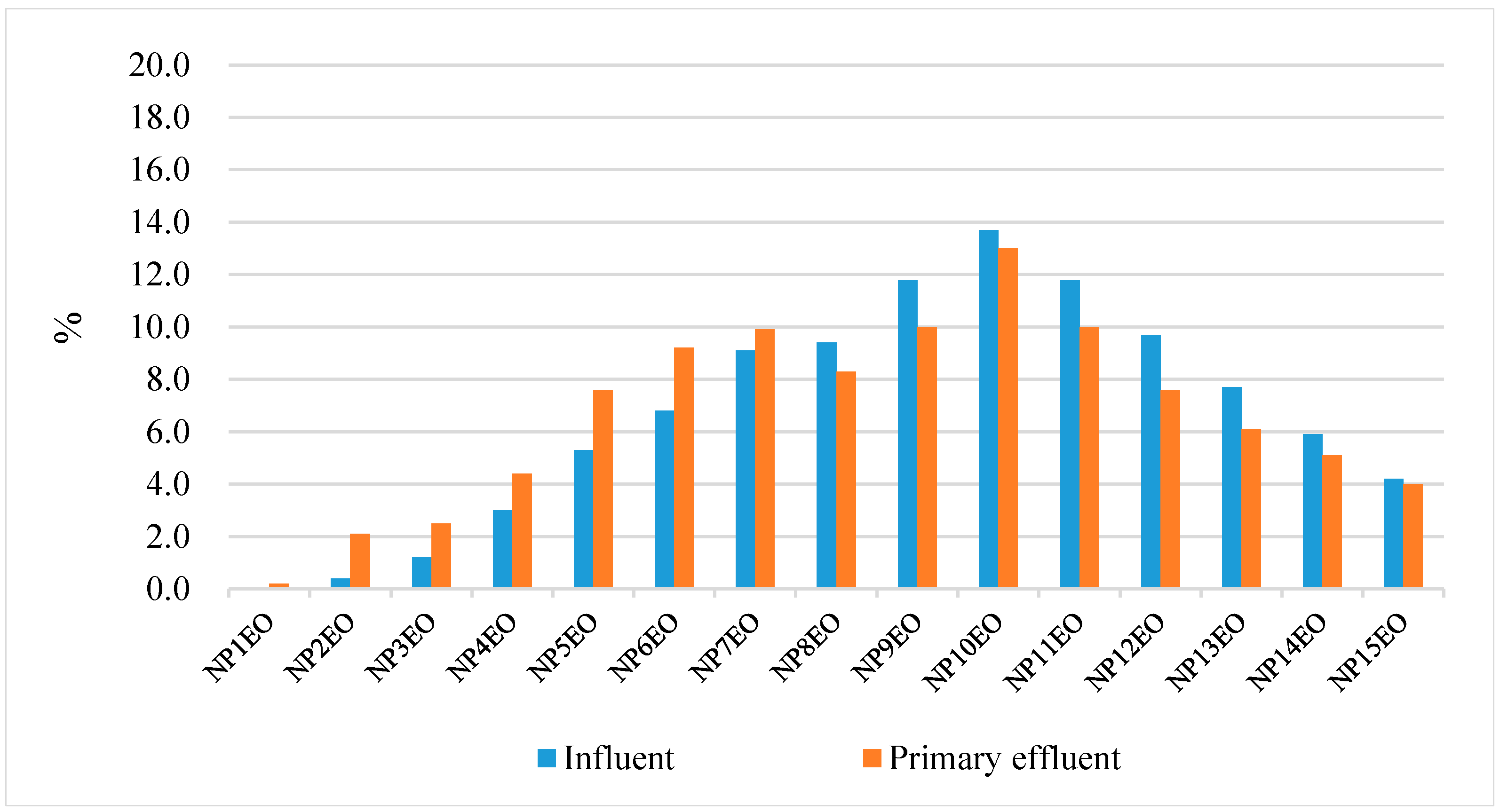
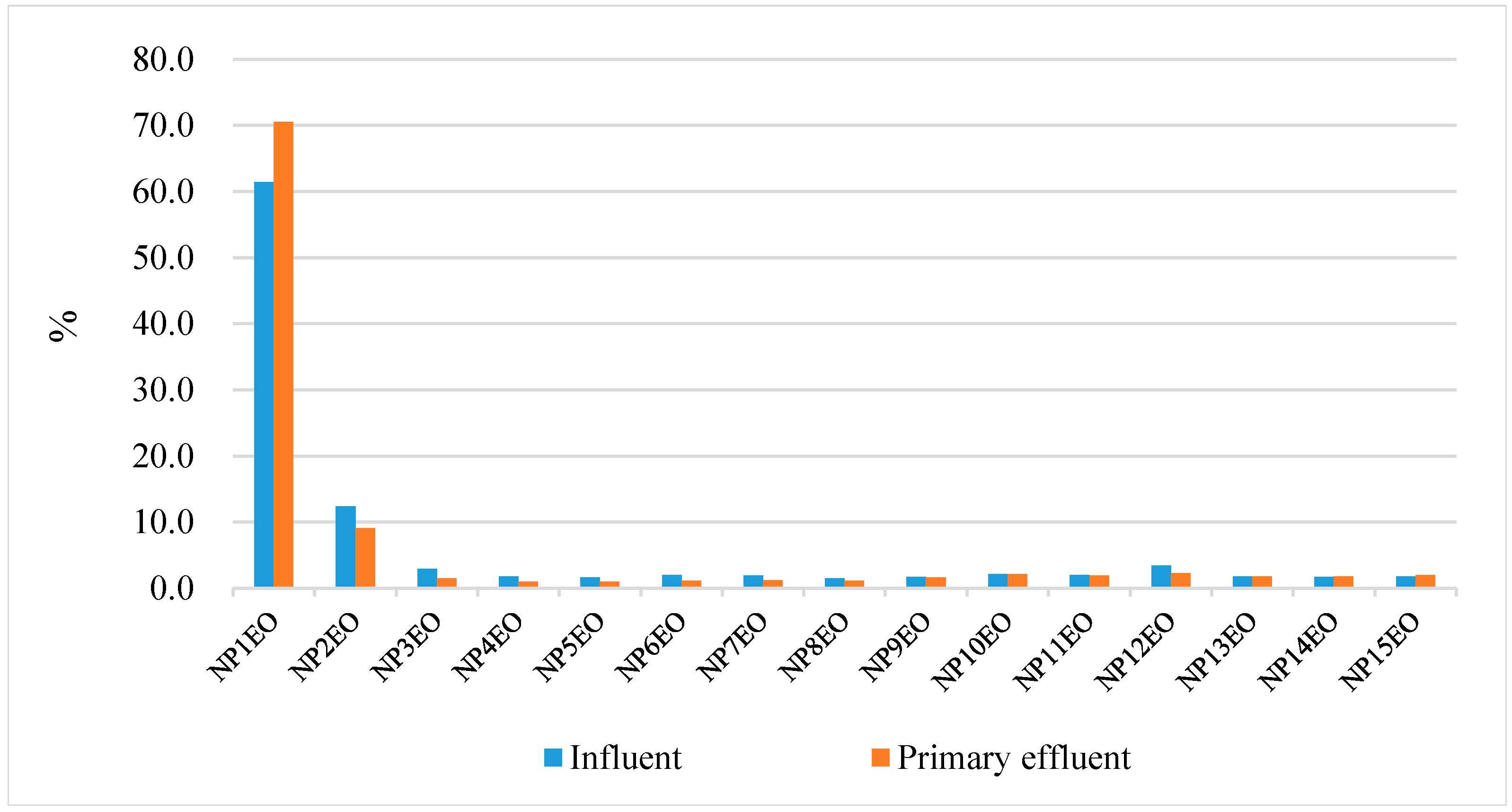
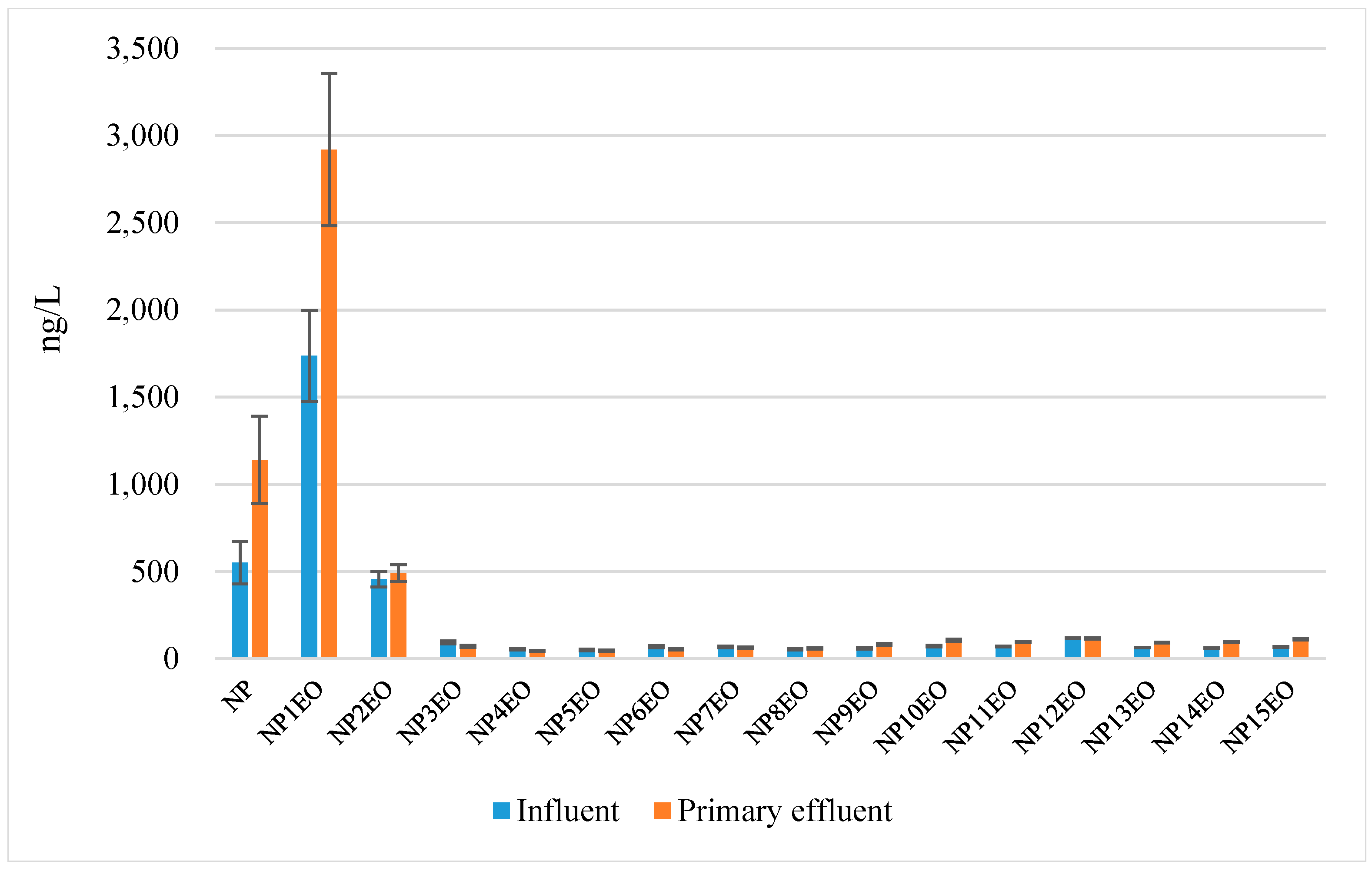
4.3. Removal of NPEO and NP in the Two Textile Factories of Investigation
4.3.1. The Role of Primary Coagulation
4.3.2. The Role of the Secondary Activated Sludge Process
4.4. Limitations of This Study
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nguyet, V.A. Report on the Textile Industry of Vietnam; VietinBankSc.: Hanoi, Vietnam, 2015. [Google Scholar]
- Tot, H.V. Report on the Textile Industry; FPT: Hochiminh, Vietnam, 2014. [Google Scholar]
- Loan, N.T.P. Greening Textile Industry in Vietnam. Ph.D. Thesis, Wageningen University and Research Centre, Wageningen, The Netherlands, 2011. [Google Scholar]
- Zhang, X. Fundamentals of Fiber Science; DEStech Publications, Inc: Lancaster, PA, USA, 2014. [Google Scholar]
- Hasanbeigi, A.; Price, L. A technical review of emerging technologies for energy and water efficiency and pollution reduction in the textile industry. J. Clean. Prod. 2015, 95, 30–44. [Google Scholar] [CrossRef]
- Antal, B.; Kuki, Á.; Nagy, L.; Nagy, T.; Zsuga, M.; Kéki, S. Rapid detection of hazardous chemicals in textiles by direct analysis in real-time mass spectrometry (dart-ms). Anal. Bioanal. Chem. 2016, 408, 5189–5198. [Google Scholar] [CrossRef] [PubMed]
- Loos, R.; Hanke, G.; Umlauf, G.; Eisenreich, S.J. LC–MS–MS analysis and occurrence of octyl-and nonylphenol, their ethoxylates and their carboxylates in belgian and italian textile industry, waste water treatment plant effluents and surface waters. Chemosphere 2007, 66, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Cobbing, M.; Ruffinengo, E.; Corre, M.-F.; Rehnby, W.; Brigden, K.M.; Saarinen, K.; Emissions, A.; Vaton, L. Textiles: Stop the Chemical Overdose. Available online: http://www.wecf.eu/english/articles/2013/10/textiles-chemicals.php (accessed on 30 May 2017).
- Munn, K. The Chemicals in Products Project: Case Study of the Textiles Sector; United Nations Environment Program Dtie/Chemicals Branch: Geneva, Switzerland, 2011. [Google Scholar]
- Brigden, K.; Casper, K.; Cobbing, M.; Crawford, T.; Dawe, A.; Erwood, S.; Wüthrich, M. Toxic Threads: The Big Fashion Stitch-Up; Greenpeace: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Brigden, K.; Hetherington, S.; Wang, M.; Santillo, D.; Johnston, P. Hazardous Chemicals in Branded Textile Products on Sale in 25 Countries/Regions during 2013; Greenpeace: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Brigden, K.; Santillo, D.; Johnston, P. Nonylphenol Ethoxylates (Npes) in Textile Products, and their Release through Laundering; Greenpeace: Amsterdam, The Netherlands, 2012. [Google Scholar]
- European Communities. European Union Risk Assessment Report: 4-Nonylphenol (Branched) and Nonylphenol; European Communities: Merseyside, UK, 2002. [Google Scholar]
- USEPA. Rm-1 Document for Para-Nonylphenol; USEPA: Washington, DC, USA, 1996.
- Yoshimura, K. Biodegradation and fish toxicity of nonionic surfactants. J. Am. Oil Chem. Soc. 1986, 63, 1590–1596. [Google Scholar] [CrossRef]
- Manzano, M.A.; Perales, J.A.; Sales, D.; Quiroga, J.M. The effect of temperature on the biodegradation of a nonylphenol polyethoxylate in river water. Water Res. 1999, 33, 2593–2600. [Google Scholar] [CrossRef]
- Ejlertsson, J.; Nilsson, M.-L.; Kylin, H.; Bergman, Å.; Karlson, L.; Öquist, M.; Svensson, B.H. Anaerobic degradation of nonylphenol mono-and diethoxylates in digestor sludge, landfilled municipal solid waste, and landfilled sludge. Environ. Sci. Technol. 1999, 33, 301–306. [Google Scholar] [CrossRef]
- Ahel, M.; Giger, W.; Koch, M. Behaviour of alkylphenol polyethoxylate surfactants in the aquatic environment—i. Occurrence and transformation in sewage treatment. Water Res. 1994, 28, 1131–1142. [Google Scholar] [CrossRef]
- Ahel, M.; Giger, W.; Schaffner, C. Behaviour of alkylphenol polyethoxylate surfactants in the aquatic environment—ii. Occurrence and transformation in rivers. Water Res. 1994, 28, 1143–1152. [Google Scholar] [CrossRef]
- Ademollo, N.; Ferrara, F.; Delise, M.; Fabietti, F.; Funari, E. Nonylphenol and octylphenol in human breast milk. Environ. Int. 2008, 34, 984–987. [Google Scholar] [CrossRef] [PubMed]
- Ahel, M.; McEvoy, J.; Giger, W. Bioaccumulation of the lipophilic metabolites of nonionic surfactants in freshwater organisms. Environ. Pollut. 1993, 79, 243–248. [Google Scholar] [CrossRef]
- Ying, G.-G. Fate, behavior and effects of surfactants and their degradation products in the environment. Environ. Int. 2006, 32, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Casatta, N.; Mascolo, G.; Roscioli, C.; Viganò, L. Tracing endocrine disrupting chemicals in a coastal lagoon (Sacca Di Goro, Italy): Sediment contamination and bioaccumulation in manila clams. Sci. Total Environ. 2015, 511, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Casatta, N.; Stefani, F.; Pozzoni, F.; Guzzella, L.; Marziali, L.; Mascolo, G.; Viganò, L. Endocrine-disrupting chemicals in coastal lagoons of the po river delta: Sediment contamination, bioaccumulation and effects on manila clams. Environ. Sci. Pollut. Res. 2016, 23, 10477–10493. [Google Scholar] [CrossRef] [PubMed]
- Diehl, J.; Johnson, S.E.; Xia, K.; West, A.; Tomanek, L. The distribution of 4-nonylphenol in marine organisms of north american pacific coast estuaries. Chemosphere 2012, 87, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Yu, J.; Hu, X.; Yin, D. Characteristics of the alkylphenol and bisphenol a distributions in marine organisms and implications for human health: A case study of the East China Sea. Sci. Total Environ. 2016, 539, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Jin, F.; Wan, Y.; Yang, M.; An, L.; An, W.; Tao, S. Trophodynamic behavior of 4-nonylphenol and nonylphenol polyethoxylate in a marine aquatic food web from Bohai Bay, North China: Comparison to ddts. Environ. Sci. Technol. 2005, 39, 4801–4807. [Google Scholar] [CrossRef] [PubMed]
- Isobe, T.; Takada, H.; Kanai, M.; Tsutsumi, S.; Isobe, K.O.; Boonyatumanond, R.; Zakaria, M.P. Distribution of polycyclic aromatic hydrocarbons (PAHs) and phenolic endocrine disrupting chemicals in South and Southeast Asian Mussels. Environ. Monit. Assess. 2007, 135, 423–440. [Google Scholar] [CrossRef] [PubMed]
- Korsman, J.C.; Schipper, A.M.; de Vos, M.G.; van den Heuvel-Greve, M.J.; Vethaak, A.D.; de Voogt, P.; Hendriks, A.J. Modeling bioaccumulation and biomagnification of nonylphenol and its ethoxylates in estuarine-marine food chains. Chemosphere 2015, 138, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Kidd, K.A.; Becher, G.; Bergman, Å.; Muir, D.C.; Woodruff, T.J. Human and wildlife exposures to edcs. In State of the Science of Endocrine Disrupting Chemicals-2012; WHO-UNEP: Geneva, Switzerland, 2012; pp. 1–261. [Google Scholar]
- Chen, G.-W.; Ding, W.-H.; Ku, H.-Y.; Chao, H.-R.; Chen, H.-Y.; Huang, M.-C.; Wang, S.-L. Alkylphenols in human milk and their relations to dietary habits in Central Taiwan. Food Chem. Toxicol. 2010, 48, 1939–1944. [Google Scholar] [CrossRef] [PubMed]
- Guenther, K.; Heinke, V.; Thiele, B.; Kleist, E.; Prast, H.; Raecker, T. Endocrine disrupting nonylphenols are ubiquitous in food. Environ. Sci. Technol. 2002, 36, 1676–1680. [Google Scholar] [CrossRef] [PubMed]
- Gyllenhammar, I.; Glynn, A.; Darnerud, P.O.; Lignell, S.; van Delft, R.; Aune, M. 4-nonylphenol and bisphenol a in swedish food and exposure in Swedish nursing women. Environ. Int. 2012, 43, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.-Y.; Chen, M.-L.; Sung, F.-C.; Paulus Shyi-Gang, W.; Mao, I.F. Daily intake of 4-nonylphenol in Taiwanese. Environ. Int. 2007, 33, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Shao, B.; Hu, J.; Yang, M.; An, W.; Tao, S. Nonylphenol and nonylphenol ethoxylates in river water, drinking water, and fish tissues in the area of Chongqing, China. Arch. Environ. Contam. Toxicol. 2005, 48, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Espinosa, M.J.; Freire, C.; Arrebola, J.P.; Navea, N.; Taoufiki, J.; Fernandez, M.F.; Ballesteros, O.; Prada, R.; Olea, N. Nonylphenol and octylphenol in adipose tissue of women in Southern Spain. Chemosphere 2009, 76, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Calafat, A.M.; Kuklenyik, Z.; Reidy, J.A.; Caudill, S.P.; Ekong, J.; Needham, L.L. Urinary concentrations of bisphenol a and 4-nonylphenol in a human reference population. Environ. Health Perspect. 2005, 113, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Shekhar, S.; Sood, S.; Showkat, S.; Lite, C.; Chandrasekhar, A.; Vairamani, M.; Barathi, S.; Santosh, W. Detection of phenolic endocrine disrupting chemicals (EDCs) from maternal blood plasma and amniotic fluid in Indian population. Gen. Comp. Endocrinol. 2017, 241, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Azzouz, A.; Rascón, A.J.; Ballesteros, E. Simultaneous determination of parabens, alkylphenols, phenylphenols, bisphenol a and triclosan in human urine, blood and breast milk by continuous solid-phase extraction and gas chromatography-mass spectrometry. J. Pharm. Biomed. Anal. 2016, 119, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Fan, Z.; Zhao, F.; Gao, F.; Mu, D.; Zhou, Y.; Shen, H.; Hu, J. Occurrence and maternal transfer of chlorinated bisphenol a and nonylphenol in pregnant women and their matching embryos. Environ. Sci. Technol. 2016, 50, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Cosnefroy, A.; Brion, F.; Guillet, B.; Laville, N.; Porcher, J.M.; Balaguer, P.; Aït-Aïssa, S. A stable fish reporter cell line to study estrogen receptor transactivation by environmental (xeno)estrogens. Toxicol. Vitro 2009, 23, 1450–1454. [Google Scholar] [CrossRef] [PubMed]
- Ghisari, M.; Bonefeld-Jorgensen, E.C. Impact of environmental chemicals on the thyroid hormone function in pituitary rat gh3 cells. Mol. Cell. Endocrinol. 2005, 244, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Jie, X.; Yang, W.; Jie, Y.; Fan, Q.-Y.; Liu, X.-Y.; Yan, L.; Huang, H.-J. Immune effects of nonylphenol on offspring of rats exposed during pregnancy. Hum. Ecol. Risk Assess. Int. J. 2010, 16, 444–452. [Google Scholar] [CrossRef]
- Jobling, S.; Sumpter, J.P.; Sheahan, D.; Osborne, J.A.; Matthiessen, P. Inhibition of testicular growth in Rainbow Trout (Oncorhynchus Mykiss) exposed to estrogenic alkylphenolic chemicals. Environ. Toxicol. Chem. 1996, 15, 194–202. [Google Scholar] [CrossRef]
- Kim, S.K.; Kim, B.K.; Shim, J.H.; Gil, J.E.; Yoon, Y.D.; Kim, J.H. Nonylphenol and octylphenol-induced apoptosis in human embryonic stem cells is related to fas-fas ligand pathway. Toxicol. Sci. 2006, 94, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; Zheng, Y.L.; Zhang, Y.Q. Behavioral impairment and oxidative damage induced by chronic application of nonylphenol. Int. J. Mol. Sci. 2010, 12, 114–127. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, K.; Ohno, Y. Modulation by estrogens and xenoestrogens of recombinant human neuronal nicotinic receptors. Eur. J. Pharmacol. 2001, 430, 175–183. [Google Scholar] [CrossRef]
- Pedersen, S.N.; Christiansen, L.B.; Pedersen, K.L.; Korsgaard, B.; Bjerregaard, P. In vivo estrogenic activity of branched and linear alkylphenols in Rainbow Trout (Oncorhynchus Mykiss). Sci. Total Environ. 1999, 233, 89–96. [Google Scholar] [CrossRef]
- Razia, S.; Maegawa, Y.; Tamotsu, S.; Oishi, T. Histological changes in immune and endocrine organs of quail embryos: Exposure to estrogen and nonylphenol. Ecotoxicol. Environ. Saf. 2006, 65, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Soto, A.M.; Justicia, H.; Wray, J.W.; Sonnenschein, C. P-nonyl-phenol: An estrogenic xenobiotic released from “modified” polystyrene. Environ. Health Perspect. 1991, 92, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Vosges, M.; Kah, O.; Hinfray, N.; Chadili, E.; Le Page, Y.; Combarnous, Y.; Porcher, J.-M.; Brion, F. 17α-ethinylestradiol and nonylphenol affect the development of forebrain gnrh neurons through an estrogen receptors-dependent pathway. Reprod. Toxicol. 2012, 33, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Couderc, M.; Gandar, A.; Kamari, A.; Allain, Y.; Zalouk-Vergnoux, A.; Herrenknecht, C.; Le Bizec, B.; Mouneyrac, C.; Poirier, L. Neurodevelopmental and behavioral effects of nonylphenol exposure during gestational and breastfeeding period on f1 rats. Neurotoxicology 2014, 44, 237–249. [Google Scholar] [CrossRef] [PubMed]
- WHO-UNEP. State of the Science of Endocrine Disrupting Chemicals-2012; WHO-UNEP: Geneva, Switzerland, 2013. [Google Scholar]
- Wu, F.; Khan, S.; Wu, Q.; Barhoumi, R.; Burghardt, R.; Safe, S. Ligand structure-dependent activation of estrogen receptor α/sp by estrogens and xenoestrogens. J. Steroid Biochem. Mol. Biol. 2008, 110, 104–115. [Google Scholar] [CrossRef] [PubMed]
- Forte, M.; Di Lorenzo, M.; Carrizzo, A.; Valiante, S.; Vecchione, C.; Laforgia, V.; De Falco, M. Nonylphenol effects on human prostate non tumorigenic cells. Toxicology 2016, 357–358, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Nam, K.H.; Hwang, K.A.; Choi, K.C. Influence of hexabromocyclododecane and 4-nonylphenol on the regulation of cell growth, apoptosis and migration in prostatic cancer cells. Toxicol. Vitro 2016, 32, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Lepretti, M.; Paolella, G.; Giordano, D.; Marabotti, A.; Gay, F.; Capaldo, A.; Esposito, C.; Caputo, I. 4-nonylphenol reduces cell viability and induces apoptosis and er-stress in a human epithelial intestinal cell line. Toxicol. Vitro 2015, 29, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Pillon, A.; Boussioux, A.-M.; Escande, A.; Aït-Aïssa, S.; Gomez, E.; Fenet, H.; Ruff, M.; Moras, D.; Vignon, F.; Duchesne, M.J.; et al. Binding of estrogenic compounds to recombinant estrogen receptor-α: Application to environmental analysis. Environ. Health Perspect. 2005, 113, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Mnif, W.; Pascussi, J.-M.; Pillon, A.; Escande, A.; Bartegi, A.; Nicolas, J.-C.; Cavaillès, V.; Duchesne, M.-J.; Balaguer, P. Estrogens and antiestrogens activate hpxr. Toxicol. Lett. 2007, 170, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, G.G.J.M.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.-A.K. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor β. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef] [PubMed]
- Preuss, T.G.; Gurer-Orhan, H.; Meerman, J.; Ratte, H.T. Some nonylphenol isomers show antiestrogenic potency in the Mvln Cell Assay. Toxicol. Vitro 2010, 24, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, P.J.; Schomburg, L.; Köhrle, J. Interference of endocrine disrupters with thyroid hormone receptor-dependent transactivation. Toxicol. Sci. 2009, 110, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; Dong, J. Binding and activation of the seven-transmembrane estrogen receptor GPR30 by environmental estrogens: A potential novel mechanism of endocrine disruption. J. Steroid Biochem. Mol. Biol. 2006, 102, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, S.; Safe, S. Activation of kinase pathways in MCF-7 cells by 17β-estradiol and structurally diverse estrogenic compounds. J. Steroid Biochem. Mol. Biol. 2006, 98, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Bulayeva, N.N.; Watson, C.S. Xenoestrogen-induced ERK-1 and ERK-2 activation via multiple membrane-initiated signaling pathways. Environ. Health Perspect. 2004, 112, 1481–1487. [Google Scholar] [CrossRef] [PubMed]
- OSPAR. Ospar Strategy with Regards Towards Hazardous Substances; OSPAR: London, UK, 1998. [Google Scholar]
- Hanh, D.T.; Kadomami, K.; Matsuura, N.; Trung, N.Q. Screening analysis of a thousand micro-pollutants in vietnamese rivers. In Proceedings of the 10th International Symposium on Southeast Asian Water Environment, Hanoi, Vietnam, 8–10 November 2012. [Google Scholar]
- Chau, H.T.C.; Kadokami, K.; Duong, H.T.; Kong, L.; Nguyen, T.T.; Nguyen, T.Q.; Ito, Y. Occurrence of 1153 organic micropollutants in the aquatic environment of Vietnam. Environ. Sci. Pollut. Res. 2015, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Brigden, K.; Labunska, I.; Santillo, D.; Wang, M.; Johnston, P. Organic Chemical and Heavy Metal Contaminants in Wastewaters Discharged from Two Textile Manufacturing Facilities in Indonesia; Greenpeace: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Brigden, K.; Labunska, I.; Pearson, M.; Santillo, D.; Johnston, P. Dirty Laundry, Unravelling the Corporate Connections to Toxic Water Pollution in China; Greenpeace: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Vietnam Environment Administration (VEA). Technical Document—Guideline for Assessing the Appropriateness of the Wastewater Treatment Processes and Proposing Some Wastewater Treatment Processes for the Fishery, Textile, Paper and Pulping Industries in Vietnam; VEA: Monre, Vietnam, 2011. (In Vietnamese)
- Zhou, H.; Huang, X.; Wang, X.; Zhi, X.; Yang, C.; Wen, X.; Wang, Q.; Tsuno, H.; Tanaka, H. Behaviour of selected endocrine-disrupting chemicals in three sewage treatment plants of Beijing, China. Environ. Monit. Assess. 2009, 161, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Ahel, M.; Hršak, D.; Giger, W. Aerobic transformation of short-chain alkylphenol polyethoxylates by mixed bacterial cultures. Arch. Environ. Contam. Toxicol. 1994, 26, 540–548. [Google Scholar] [CrossRef]
- Mann, R.M.; Boddy, M.R. Biodegradation of a nonylphenol ethoxylate by the autochthonous microflora in lake water with observations on the influence of light. Chemosphere 2000, 41, 1361–1369. [Google Scholar] [CrossRef]
- Potter, T.L.; Simmons, K.; Wu, J.; Sanchez-Olvera, M.; Kostecki, P.; Calabrese, E. Static die-away of a nonylphenol ethoxylate surfactant in Estuarine Water Samples. Environ. Sci. Technol. 1999, 33, 113–118. [Google Scholar] [CrossRef]
- Scott, M.J.; Jones, M.N. The biodegradation of surfactants in the environment. Biochim. Biophys. Acta (BBA) Biomembr. 2000, 1508, 235–251. [Google Scholar] [CrossRef]
- Charles, W.; Ho, G.; Cord-Ruwisch, R. Anaerobic bioflocculation of wool scouring effluent: The influence of non-ionic surfactant on efficiency. Water Sci. Technol. 1996, 34, 1–8. [Google Scholar] [CrossRef]
- Di Corcia, A.; Samperi, R.; Marcomini, A. Monitoring aromatic surfactants and their biodegradation intermediates in raw and treated sewages by solid-phase extraction and liquid chromatography. Environ. Sci. Technol. 1994, 28, 850–858. [Google Scholar] [CrossRef] [PubMed]
- Field, J.A.; Reed, R.L. Nonylphenol polyethoxy carboxylate metabolites of nonionic surfactants in U.S. Paper mill effluents, municipal sewage treatment plant effluents, and river waters. Environ. Sci. Technol. 1996, 30, 3544–3550. [Google Scholar] [CrossRef]
- Staples, C.A.; Williams, J.B.; Blessing, R.L.; Varineau, P.T. Measuring the biodegradability of nonylphenol ether carboxylates, octylphenol ether carboxylates, and nonylphenol. Chemosphere 1999, 38, 2029–2039. [Google Scholar] [CrossRef]
- Cirja, M.; Ivashechkin, P.; Schäffer, A.; Corvini, P.F. Factors affecting the removal of organic micropollutants from wastewater in conventional treatment plants (CTP) and membrane bioreactors (MBR). Rev. Environ. Sci. Bio/Technol. 2008, 7, 61–78. [Google Scholar] [CrossRef]
- Fauser, P.; Sørensen, P.B.; Carlsen, L.; Vikelsøe, J. Phthalates, Nonylphenols and Las in Roskilde Wastewater Treatment Plant: Fate Modelling Based on Measured Concentrations in Wastewater and Sludge; National Environmental Research Institute: Roskilde, Denmark, 2001. [Google Scholar]
- Bertanza, G.; Pedrazzani, R.; Papa, M.; Mazzoleni, G.; Steimberg, N.; Caimi, L.; Montani, C.; Dilorenzo, D. Removal of BPA and NPnEOs from secondary effluents of municipal WWTPs by means of ozonation. Ozone Sci. Eng. 2010, 32, 204–208. [Google Scholar] [CrossRef]
- Bertanza, G.; Papa, M.; Pedrazzani, R.; Repice, C.; Dal Grande, M. Tertiary ozonation of industrial wastewater for the removal of estrogenic compounds (NP and BPA): A full-scale case study. Water Sci. Technol. 2013, 68, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Grau, P. Textile industry wastewaters treatment. Water Sci. Technol. 1991, 24, 97–103. [Google Scholar]
- Castiglioni, S.; Bagnati, R.; Fanelli, R.; Pomati, F.; Calamari, D.; Zuccato, E. Removal of pharmaceuticals in sewage treatment plants in Italy. Environ. Sci. Technol. 2006, 40, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Pothitou, P.; Voutsa, D. Endocrine disrupting compounds in municipal and industrial wastewater treatment plants in Northern Greece. Chemosphere 2008, 73, 1716–1723. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.C.; Aerni, H.R.; Gerritsen, A.; Gibert, M.; Giger, W.; Hylland, K.; Jürgens, M.; Nakari, T.; Pickering, A.; Suter, M.J.F.; et al. Comparing steroid estrogen, and nonylphenol content across a range of european sewage plants with different treatment and management practices. Water Res. 2005, 39, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Ahel, M.; Giger, W. Partitioning of alkylphenols and alkylphenol polyethoxylates between water and organic solvents. Chemosphere 1993, 26, 1471–1478. [Google Scholar] [CrossRef]
- Vogelsang, C.; Grung, M.; Jantsch, T.G.; Tollefsen, K.E.; Liltved, H. Occurrence and removal of selected organic micropollutants at mechanical, chemical and advanced wastewater treatment plants in Norway. Water Res. 2006, 40, 3559–3570. [Google Scholar] [CrossRef] [PubMed]
- Eddy, M. Wastewater Engineering: Treatment and Reuse, 4th ed.; McGraw-Hill: New York, NY, USA, 2004. [Google Scholar]
- Zhou, Z.; Guo, Q.; Xu, Z.; Wang, L.; Cui, K. Distribution and removal of endocrine-disrupting chemicals in industrial wastewater treatment. Environ. Eng. Sci. 2015, 32, 203–211. [Google Scholar] [CrossRef]
- Tan, B.L.L.; Hawker, D.W.; Müller, J.F.; Leusch, F.D.L.; Tremblay, L.A.; Chapman, H.F. Comprehensive study of endocrine disrupting compounds using grab and passive sampling at selected wastewater treatment plants in South East Queensland, Australia. Environ. Int. 2007, 33, 654–669. [Google Scholar] [CrossRef] [PubMed]
- Luppi, L.I.; Hardmeier, I.; Babay, P.A.; Itria, R.F.; Erijman, L. Anaerobic nonylphenol ethoxylate degradation coupled to nitrate reduction in a modified biodegradability batch test. Chemosphere 2007, 68, 2136–2143. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, J.; Li, Y. Removal of bisphenol a and 4-n-nonylphenol coupled to nitrate reduction using acclimated activated sludge under anaerobic conditions. J. Chem. Technol. Biotechnol. 2013, 89, 391–400. [Google Scholar] [CrossRef]
- Lee Ferguson, P.; Brownawell, B.J. Degradation of nonylphenol ethoxylates in estuarine sediment under aerobic and anaerobic conditions. Environ. Toxicol. Chem. 2003, 22, 1189–1199. [Google Scholar] [CrossRef]
- Lu, J.; Jin, Q.; He, Y.; Wu, J.; Zhang, W.; Zhao, J. Biodegradation of nonylphenol polyethoxylates by denitrifying activated sludge. Water Res. 2008, 42, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Zhou, Y.; Li, H.; Wang, F. Fate and removal of selected endocrine-disrupting compounds in sewage using activated sludge treatment. Water Environ. J. 2012, 26, 435–444. [Google Scholar] [CrossRef]
- Nie, Y.; Qiang, Z.; Ben, W.; Liu, J. Removal of endocrine-disrupting chemicals and conventional pollutants in a continuous-operating activated sludge process integrated with ozonation for excess sludge reduction. Chemosphere 2014, 105, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Langford, K.H.; Scrimshaw, M.D.; Birkett, J.W.; Lester, J.N. Degradation of nonylphenolic surfactants in activated sludge batch tests. Water Res. 2005, 39, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Li, Z.; Guan, J.; Li, Y.; Ren, N. Removal of surfactants nonylphenol ethoxylates from municipal sewage-comparison of an a/o process and biological aerated filters. Chemosphere 2014, 97, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Langford, K.H.; Scrimshaw, M.D.; Birkett, J.W.; Lester, J.N. The partitioning of alkylphenolic surfactants and polybrominated diphenyl ether flame retardants in activated sludge batch tests. Chemosphere 2005, 61, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Langford, K.; Scrimshaw, M.; Lester, J. The impact of process variables on the removal of pbdes and npeos during simulated activated sludge treatment. Arch. Environ. Contam. Toxicol. 2007, 53, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Loyo-Rosales, J.E.; Rice, C.P.; Torrents, A. Fate of octyl- and nonylphenol ethoxylates and some carboxylated derivatives in three American wastewater treatment plants. Environ. Sci. Technol. 2007, 41, 6815–6821. [Google Scholar] [CrossRef] [PubMed]
- Scruggs, C.; Hunter, G.; Snyder, E.; Long, B.; Snyder, S. Edcs in wastewater: What’s the next step? Proc. Water Environ. Fed. 2004, 2004, 642–664. [Google Scholar] [CrossRef]
- Ivashechkin, P.; Corvini, P.; Fahrbach, M.; Hollender, J.; Konietzko, M.; Meesters, R.; Schröder, H.F.; Dohmann, M. Comparison of the elimination of endocrine disrupters in conventional wastewater treatment plants and membrane bioreactors. In Proceedings of the 2nd IWA Leading-Edge Conference on Water and Wastewater Treatment Technologies, Prague, Czech Republic, 1–4 June 2004. [Google Scholar]
- Stasinakis, A.S.; Kordoutis, C.I.; Tsiouma, V.C.; Gatidou, G.; Thomaidis, N.S. Removal of selected endocrine disrupters in activated sludge systems: Effect of sludge retention time on their Sorption and Biodegradation. Bioresour. Technol. 2010, 101, 2090–2095. [Google Scholar] [CrossRef] [PubMed]
- Petrie, B.; McAdam, E.J.; Lester, J.N.; Cartmell, E. Assessing potential modifications to the activated sludge process to improve simultaneous removal of a diverse range of micropollutants. Water Res. 2014, 62, 180–192. [Google Scholar] [CrossRef] [PubMed]
- Birkett, J.W.; Lester, J.N. Endocrine Disrupters in Wastewater and Sludge Treatment Processes; IWA Publishing: London, UK, 2003. [Google Scholar]
- Kreuzinger, N.; Clara, M.; Strenn, B.; Kroiss, H. Relevance of the sludge retention time (SRT) as design criteria for wastewater treatment plants for the removal of endocrine disruptors and pharmaceuticals from wastewater. Water Sci. Technol. 2004, 50, 149–156. [Google Scholar] [PubMed]
- Clara, M.; Scharf, S.; Scheffknecht, C.; Gans, O. Occurrence of selected surfactants in untreated and treated Sewage. Water Res. 2007, 41, 4339–4348. [Google Scholar] [CrossRef] [PubMed]
- Center for Environmental Technology and Management (CENTEMA). Investigation and Assessment of the Appropriateness of the Wastewater Treatment Plants in Several Sectors as a Basis for Proposing a List of Encouraging Wastewater Treatment Processes in Vietnam—Textile Industry; VEA: Monre, Vietnam, 2010.
- Luo, Y.; Guo, W.; Ngo, H.H.; Nghiem, L.D.; Hai, F.I.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473–474, 619–641. [Google Scholar] [CrossRef] [PubMed]
- Ort, C.; Lawrence, M.G.; Rieckermann, J.; Joss, A. Sampling for pharmaceuticals and personal care products (PPCPS) and illicit drugs in wastewater systems: Are your conclusions valid? A critical review. Environ. Sci. Technol. 2010, 44, 6024–6035. [Google Scholar] [CrossRef] [PubMed]
- American Public Health Association (APHA). Standard methods for the examination of water and wastewater. In SPE-8998-PA, 21st ed.; APHA: Washington, DC, USA, 2005. [Google Scholar]
- Tuc Dinh, Q.; Alliot, F.; Moreau-Guigon, E.; Eurin, J.; Chevreuil, M.; Labadie, P. Measurement of trace levels of antibiotics in river water using on-line enrichment and triple-quadrupole Lc–Ms/Ms. Talanta 2011, 85, 1238–1245. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Yang, F.; Fu, Z.; Lei, R. Comparison between a moving bed membrane bioreactor and a conventional membrane bioreactor on organic carbon and nitrogen removal. Bioresour. Technol. 2009, 100, 2369–2374. [Google Scholar] [CrossRef] [PubMed]
- Houweling, D.; Monette, F.; Millette, L.; Comeau, Y. Modelling nitrification of a lagoon effluent in moving-bed biofilm reactors. Water Qual. Res. J. Can. 2007, 42, 284–294. [Google Scholar]
- European Parliament and Council. Directive 2008/105/EC of the European Parliament and of the Council of 16 December 2008 on Environmental Quality Standards in the Field of Water Policy, Amending and Subsequently Repealing Council Directives 82/176/EEC, 83/513/EEC, 84/156/EEC, 84/491/EEC, 86/280/EEC and Amending Directive 2000/60/EC of the European Parliament and of the Council; 2008/105/EC; European Parliament and Council: Strasbourg, France, 2008. [Google Scholar]
- Stackelberg, P.E.; Gibs, J.; Furlong, E.T.; Meyer, M.T.; Zaugg, S.D.; Lippincott, R.L. Efficiency of conventional drinking-water-treatment processes in removal of pharmaceuticals and other organic compounds. Sci. Total Environ. 2007, 377, 255–272. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.W.; Jo, B.I.; Yoon, Y.; Zoh, K.D. Occurrence and removal of selected micropollutants in a water treatment plant. Chemosphere 2014, 95, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Loyo-Rosales, J.E.; Rice, C.P.; Torrents, A. Octyl and nonylphenol ethoxylates and carboxylates in wastewater and sediments by liquid chromatography/tandem mass spectrometry. Chemosphere 2007, 68, 2118–2127. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, G.; Paul, E.; Novais, J.; Pinheiro, H. Studies on activated sludge response to variations in the composition of a synthetic surfactant-containing feed effluent. Waste Sci. Technol. 2000, 42, 135–143. [Google Scholar]
- Nimrod, A.C.; Benson, W.H. Environmental estrogenic effects of alkylphenol ethoxylates. Crit. Rev. Toxicol. 1996, 26, 335–364. [Google Scholar] [CrossRef] [PubMed]
- Maki, H.; Masuda, N.; Fujiwara, Y.; Ike, M.; Fujita, M. Degradation of alkylphenol ethoxylates by pseudomonas sp. Strain TR01. Appl. Environ. Microbiol. 1994, 60, 2265–2271. [Google Scholar] [PubMed]
- Johnson, A.; Darton, R. Removing oestrogenic compounds from sewage effluent. Chem. Eng. 2003, 741. [Google Scholar]
- Cicek, N.; Macomber, J.; Davel, J.; Suidan, M.T.; Audic, J.; Genestet, P. Effect of solids retention time on the performance and biological characteristics of a membrane bioreactor. Water Sci. Technol. 2001, 43, 43–50. [Google Scholar] [PubMed]
- Kim, J.Y.; Ryu, K.; Kim, E.J.; Choe, W.S.; Cha, G.C.; Yoo, I.-K. Degradation of bisphenol a and nonylphenol by nitrifying activated sludge. Process Biochem. 2007, 42, 1470–1474. [Google Scholar] [CrossRef]
- Körner, W.; Spengler, P.; Bolz, U.; Schuller, W.; Hanf, V.; Metzger, J.W. Substances with estrogenic activity in effluents of sewage treatment plants in Southwestern Germany. 2. Biological analysis. Environ. Toxicol. Chem. 2001, 20, 2142–2151. [Google Scholar] [CrossRef] [PubMed]
- Vader, J.S.; van Ginkel, C.G.; Sperling, F.M.G.M.; de Jong, J.; de Boer, W.; de Graaf, J.S.; van der Most, M.; Stokman, P.G.W. Degradation of ethinyl estradiol by nitrifying activated sludge. Chemosphere 2000, 41, 1239–1243. [Google Scholar] [CrossRef]
- McAdam, E.J.; Bagnall, J.P.; Soares, A.; Koh, Y.K.K.; Chiu, T.Y.; Scrimshaw, M.D.; Lester, J.N.; Cartmell, E. Fate of alkylphenolic compounds during activated sludge treatment: Impact of loading and organic composition. Environ. Sci. Technol. 2011, 45, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Gerardi, M.H. Nitrification in the Activated Sludge Process; Wiley Online Library: Weinheim, Germany, 2005. [Google Scholar]
- Hanaki, K.; Wantawin, C.; Ohgaki, S. Effects of the activity of heterotrophs on nitrification in a suspended-growth reactor. Water Res. 1990, 24, 289–296. [Google Scholar] [CrossRef]
- Ito, K.; Matsuo, T. The effect of organic loading on nitrification in RBC wastewater treatment processes. In Proceedings of the First National Symposium on RBC Technology, Champion, PA, USA, 4–6 February 1980; pp. 1165–1176. [Google Scholar]
- Sharma, B.; Ahlert, R.C. Nitrification and nitrogen removal. Water Res. 1977, 11, 897–925. [Google Scholar] [CrossRef]
- Zhu, G.; Peng, Y.; Li, B.; Guo, J.; Yang, Q.; Wang, S. Biological removal of nitrogen from wastewater. In Reviews of Environmental Contamination and Toxicology; Whitacre, D.M., Ed.; Springer: New York, NY, USA, 2008; pp. 159–195. [Google Scholar]
- Van Hulle, S.W.H.; Volcke, E.I.P.; Teruel, J.L.; Donckels, B.; van Loosdrecht, M.C.M.; Vanrolleghem, P.A. Influence of temperature and ph on the kinetics of the sharon nitritation process. J. Chem. Technol. Biotechnol. 2007, 82, 471–480. [Google Scholar] [CrossRef]
- Li, J.; Yu, D.; Zhang, P. Partial nitrification in a sequencing batch reactor treating acrylic fiber wastewater. Biodegradation 2013, 24, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Bae, W.; Chung, J.; Baek, S.-C. Empirical model of the pH dependence of the maximum specific nitrification rate. Process Biochem. 2007, 42, 1671–1676. [Google Scholar] [CrossRef]
- Guo, J.H.; Peng, Y.Z.; Wang, S.Y.; Zheng, Y.N.; Huang, H.J.; Ge, S.J. Effective and robust partial nitrification to nitrite by real-time aeration duration control in an SBR treating domestic wastewater. Process Biochem. 2009, 44, 979–985. [Google Scholar] [CrossRef]
- Terzic, S.; Matosic, M.; Ahel, M.; Mijatovic, I. Elimination of aromatic surfactants from municipal wastewaters: Comparison of conventional activated sludge treatment and membrane biological reactor. Water Sci. Technol. 2005, 51, 447–453. [Google Scholar] [PubMed]
- Brunner, P.H.; Capri, S.; Marcomini, A.; Giger, W. Occurrence and behaviour of linear alkylbenzenesulphonates, nonylphenol, nonylphenol mono- and nonylphenol diethoxylates in sewage and sewage sludge treatment. Water Res. 1988, 22, 1465–1472. [Google Scholar] [CrossRef]
- Liao, B.; Allen, D.; Droppo, I.; Leppard, G.; Liss, S. Surface properties of sludge and their role in bioflocculation and settleability. Water Res. 2001, 35, 339–350. [Google Scholar] [CrossRef]
- Aboobakar, A.; Cartmell, E.; Stephenson, T.; Jones, M.; Vale, P.; Dotro, G. Nitrous oxide emissions and dissolved oxygen profiling in a full-scale nitrifying activated sludge treatment plant. Water Res. 2013, 47, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Morita, M.; Liljestrand, H. Estimated fate of selected endocrine disruptors in the aquatic environment and the biological treatment processes: Sorption by dissolved organic matter and synthetic membrane vesicles. In Proceedings of the Fourth Specialized Conference on Assessment and Control of Hazardous Substances in Water; International Water Association (IWA): Aachen, Germany, 2003; pp. 14–17. [Google Scholar]
- Keller, H.; Xia, K.; Bhandari, A. Occurrence and degradation of estrogenic nonylphenol and its precursors in Northeast Kansas wastewater treatment plants. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2003, 7, 203–213. [Google Scholar] [CrossRef]
- Stechemesser, H.; Dobiáš, B. Coagulation and Flocculation; Taylor & Francis: Boca Raton, FL, USA, 2005. [Google Scholar]

| Substance | Anaerobic Process a/Coagulation Process b | Activated Sludge Process | Conditions | Source |
|---|---|---|---|---|
| NP | 82.5% a | 6.5% | Anaerobic followed by aerobic activated sludge | [93] |
| NP | 42% a | 25% | Anaerobic activated sludge followed by oxidation ditch SRT~16–17 days | [72] |
| NP | Negligible | 93–94% | Anaerobic followed by aerobic activated sludge * HRT *~7.8–9.8 h SRT *~10.5–11.9 days MLSS *~3500–4000 mg/L | [92] |
| NP | - | ~99% | Activated sludge Long SRT~25 days | [106] |
| NP | - | 97–99% | Activated sludge with nitrifying Ammonia removal ~62% to >90% HRT~10 h SRT~3–20 days MLSS~500–3000 mg/L | [107] |
| NP | - | 80% | Activated sludge Temperature~20–30 °C MLSS~1676 mg/L HTR~64 h Relatively low loadings | [87] |
| NP1-2EO | - | >90% | ||
| NP | - | 70% | Activated sludge Long SRT~27 days HRT~16 h | [108] |
| NP1-3EO | 93% | |||
| NP4-12EO | 93% | |||
| NP | - | 37% | Activated sludge High loading/non-nitrifying | [109] |
| NP1EO | −3% (produced) | |||
| NP2EO | −5% (produced) | |||
| NP6EO | 78% | |||
| NP | - | 77% | Activated sludge Low loading/nitrifying | [109] |
| NP1EO | 31% | |||
| NP2EO | 91% | |||
| NP6EO | 98% | |||
| NP and NPEOs | - | >80% | Activated sludge Low loading/nitrifying STR > 10 days | [110] |
| NP and NP1-2EO | - | >90% | Activated sludge Low loading/nitrifying-denitrifying | [111] |
| NP | >90% b | - | Coagulation | [90] |
| NP | 53% b | - | Coagulation | [87] |
| NP1EO | 91% b | |||
| NP2EO | 94% b |
| Factory | Wastewater Flowrate Q (m3/Day) | Aeration Volume VA (m3) | MLSS/MLVSS XA a (mg/L) | Waste Solids Flow Rate QW (m3/d) | Returning Solids Concentration XR a (mg/L) | Effluent Solids Concentration XE a (mg/L) | HRT b (h) | SRT b (Day) | R b | |
|---|---|---|---|---|---|---|---|---|---|---|
| F1 | During the campaign | 700 | 607.2 | 2267/1651 | 0.8 | 9913 | 21 | 20.8 | 60.8 | 0.29 |
| At full capacity b | 800 | 607.2 | - | - | - | - | 18.2 | - | - | |
| F2 | During the campaign | 1500 | 1013.8 | 2657/2141 | 1.6 | 4753 | 22 | 16.2 | 66.3 | 1.25 |
| At full capacity b | 2800 | 1013.8 | - | - | - | - | 8.7 | - | - | |
| Composition | Unit | Hoa Sen Factory—F1 a | Chyang Sheng Factory—F2 a | QCVN 13:2008/BTNMT | |||
|---|---|---|---|---|---|---|---|
| Influent | Disinfection Effluent | Influent | Tertiary Effluent | Class A | Class B | ||
| pH | - | 8.6–10.1 | 7.8–8.0 | 7.9–8.6 | 6.8–7.4 | 6–9 | 5.5–9.0 |
| Temperature | °C | 40 | - | 41 | - | 40 | 40 |
| Color (pH = 7) | Pt-Co | 848 | 31 | 945 | 87 | 20 b/50 c | 150 |
| TSS | mg/L | 139 | 19 | 159 | 19 | 50 | 100 |
| CODtotal | mgO2/L | 317 | - | 541 | - | 50 | 150 |
| CODf | mgO2/L | 247 | 36 | 416 | 101 | - | - |
| BOD5-f | mgO2/L | 163 | 7 | 180 | 57 | 30 d | 50 d |
| Composition | Unit | Hoa Sen Factory a | Chyang Sheng Factory b | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Equalization | Primary Effluent | Secondary Effluent | Disinfection Effluent | Equalization | Primary Effluent | Secondary Effluent | Tertiary Effluent | ||
| pH | - | 10.1 | 6.7 | 7.7 | 7.9 | 8.1 | 7.1 | 7.8 | 7.4 |
| Temperature | °C | 39 | - | 34 c | - | 41 | - | 37 c | - |
| DO | mgO2/L | - | - | 4.8 c | - | - | - | 4.3 c | - |
| Alkalinity | mgCaCO3/L | 700 | 280 | 320 | - | 450 | 300 | 190 | - |
| TSS | mg/L | 46 | 25 | 25 | 18 | 212 | 3 | 15 | 19 |
| CODf | mgO2/L | 265 | 157 | 58 | 66 | 570 | 504 | 138 | 104 |
| BOD5-f | mgO2/L | 165 | 28 | 25 | 8 | 297 | 246 | 53 | 92 |
| N–NH4+ | mg/L | 55 | <MDL | <MDL | - | 85 | 87 | 67 | - |
| N–NO2- | mg/L | 0.4 | 0.2 | 0.0 | - | 0.8 | 0.4 | 8.4 | - |
| N–NO3- | mg/L | 1.0 | 2.2 | 2.1 | - | 3.4 | 2.3 | 1.7 | - |
| NP | ng/L | 474 | 487 | 109 | - | 551 | 1141 | 244 | - |
| ΣNP1-15EO | ng/L | 117,594 | 6469 | <SQL | - | 3099 | 4453 | <SQL | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ho, H.; Watanabe, T. Distribution and Removal of Nonylphenol Ethoxylates and Nonylphenol from Textile Wastewater—A Comparison of a Cotton and a Synthetic Fiber Factory in Vietnam. Water 2017, 9, 386. https://doi.org/10.3390/w9060386
Ho H, Watanabe T. Distribution and Removal of Nonylphenol Ethoxylates and Nonylphenol from Textile Wastewater—A Comparison of a Cotton and a Synthetic Fiber Factory in Vietnam. Water. 2017; 9(6):386. https://doi.org/10.3390/w9060386
Chicago/Turabian StyleHo, Hien, and Tsunemi Watanabe. 2017. "Distribution and Removal of Nonylphenol Ethoxylates and Nonylphenol from Textile Wastewater—A Comparison of a Cotton and a Synthetic Fiber Factory in Vietnam" Water 9, no. 6: 386. https://doi.org/10.3390/w9060386
APA StyleHo, H., & Watanabe, T. (2017). Distribution and Removal of Nonylphenol Ethoxylates and Nonylphenol from Textile Wastewater—A Comparison of a Cotton and a Synthetic Fiber Factory in Vietnam. Water, 9(6), 386. https://doi.org/10.3390/w9060386






