Abstract
The purpose of this study is to exploit the euryhaline nature of commercially attractive species for their cultivation in freshwater aquaponic systems. This approach may increase the profitability of aquaponic production in coastal countries where the consumption of marine fish is traditional and of commercial relevance. For this purpose, juvenile European sea bass (Dicentrarchus labrax) were reared in an aquaponic freshwater (AFW) system and an aquaponic saltwater (ASW) system (salinity 20 ppt), in combination with chard (Beta vulgaris var. cicla) seedlings, a salt tolerant plant. At the end of the trial, nitrate and phosphate concentration in water significantly increased in the ASW system, suggesting that the ability of B. vulgaris to absorb these substances was limited by salinity. Total reflection X-ray fluorescence spectrometry revealed that the concentration of some oligoelements such as Fe remained lower with respect to the concentration in the freshwater hydroponic solution, in both AFW and ASW. FTIR-Fourier transform infrared spectroscopy on plants showed that growth at high salinity affected their lipid content. In the case of fish, freshwater had no effects on mono- and poly-unsaturated fatty acid profiles, although saturated fatty acids were significantly decreased in D. labrax reared in AFW. Our results demonstrates that it is possible to increase aquaponic profitability by farming D. labrax juveniles in an aquaponic freshwater system together with Beta vulgaris, obtaining good quality products.
1. Introduction
Aquaponics combine the principal advantages of the Recirculating Aquaculture System (RAS) and hydroponics. Aquaponics recirculates the water, accumulating waste such as nitrogen and phosphate that derive from urine and feces produced by fish. The wastewater is pumped into a plant grow bed where plant roots absorb and remove nitrogen and phosphate, principal causes of eutrophication when released in the natural environment. This process purifies the water that becomes ready to be recirculated in the fish tank. Thanks to this process, aquaponics allows for water to be used efficiently [1]. Moreover, water savings connected to landless cultivation permits the use of non-cultivated soil and requalification of disused areas.
Most of the research on aquaponics focuses on freshwater systems [2,3]. However, this mode of cultivation may also be of interest for growing species that live in the sea, for example in the Mediterranean area, where the consumption of marine fish is traditional and of commercial relevance. Moreover, only salt or brackish water is available in some zones, as in Negev desert where Kotzen and Appelbaum [4] conducted a study on the aquaponic rearing of tilapia in brackish water.
The main disadvantage of the farming of marine fish using aquaponics lies in the limited variety of plants capable to grow at high salinity. In fact, the vast majority of plants are negatively affected by high salinity and only halophile or salt tolerant species are amenable to be reared in salt or brackish water [5]. Moreover, salinity affects some water parameters. For instance, the presence of dissolved salts makes pH more basic (8.0–8.4) than in freshwater (6.0–7.0), influencing the availability of nutrients. Consequently, plants cultivated in marine aquaponics must tolerate extreme pH and salinity conditions in order to grow healthily. However it is possible to acclimate some marine fish species to tolerate lower salinity. For instance, European sea bass (Dicentrarchus labrax), one of the most important commercial species in the Mediterranean area, is a euryhaline fish that has the potential to be reared in freshwater aquaponic systems together with a number of commune plant species [6,7]. This approach may increase the profitability of the aquaponic production in the coastal countries.
This research aims at broadening the knowledge on marine aquaponics by investigating either the quality of reared species or approaches to cope with the common disadvantages such as the reduced number of suitable plant species and costs. The study evaluation of Dicentrarchus labrax meats and Beta vulgaris var. cicla vegetable quality in freshwater and saltwater aquaponic systems was the major aim of the study. Furthermore, given that in some cases the concentration of some nutrients (particularly iron, potassium and calcium) is insufficient to ensure correct plant development in freshwater aquaponics [8], analysis on water parameters was also conducted.
2. Materials and Methods
2.1. Experiment Setup
Aquaponic filled media systems were made by Aquaguide s.a.s. (Falconara Marittima, IT) and were composed of a 500 L fish tank and a 2 m2 plant growth bed filled with expanded clay (LECA, Milan, Italy). Expanded clay operates either as mechanical filter by removing solid waste or as a biological filter by sustaining the population of nitrifying bacteria that oxidize ammonium—which is highly toxic for fish [9,10]—to nitrate. According to “ebb and flow technology” [11], the water flow was activated at 15 min/h by a pump at a rate of 600 L/h. A siphon regulated the water excess in the plant growth bed, draining it into the fish tank. European sea bass juveniles (Dicentrarchus labrax, L., 1758) weighing 20 ± 0.5 g were divided into two groups: one reared in an aquaponic freshwater (AFW) system (0 ppt); and the other reared in an aquaponic saltwater (ASW) system, (20 ppt). In each system, the plant growth bed hosted 100 chard (Beta vulgaris var. cicla) seedlings. Over the 65 days of the experiment, fish were fed commercial pellet (1.5% bw). In ASW, saltwater was prepared using synthetic “RedSea” salt. Addition of salt was not required in the AFW tank.
Before starting the experiment, juveniles in AFW were acclimated for one month by gradually decreasing the salinity by adding freshwater to saltwater. The following protocol was used:
- first week: acclimation from 25 to 15 ppt; salinity was decreased 5 ppt every 48 h
- second week: acclimation from 15 to 10 ppt; salinity was decreased 2 or 3 ppt every 48 h
- third and fourth week: acclimation from 10 to 0 ppt; salinity was decreased 1 or 2 points every 48 h.
Chard plants in AFW were acclimated to grow at 20 ppt with a similar protocol (in reverse). The temperature in the fish tank was set a ~25 ± 0.5 °C, oxygen concentration was at 80% saturation. Plants were illuminated with a fluorescent lamps (58 W) suspended 50 cm above the plant bed, with a 12L/12D photoperiod. Freshwater was added to replace losses due to the evaporation. At the end of the trial, the color, fillet total lipids and fatty acid composition of all fish were assessed. Muscles were dissected and Fourier transform infrared spectroscopy (FTIR) and C, N, and S (CNS) analysis was performed. Chard were harvested and leaves, stems, taproot and lateral roots were sampled for FTIR and CNS analyses.
The handling procedure and sampling methods involving fish used in the trial followed the guidelines of the European Union directive 2010/63/EU on the protection of animal for scientific purposes.
2.2. Water Parameters
Water samples were collected in the fish tank three times per week for the entire duration of the experiment. All parameters were measured using a Hanna HI 83203 Multiparameter Photometer for aquaculture. The reagents used were: Cadmium Reduction Method (HI 93728) for nitrate; diazotization method (HI 93707) for nitrite LR; Nessler Method (HI 93715A) for ammonia MR; Amino Acid method for phosphate HR (HI 93717). The pH was recorded continuously during the experiment.
2.3. Total Reflection X-ray Fluorescence (TXRF) Spectrometry (Metals in Water)
At the end of the trial, water samples were collected and the concentrations of Na, Cl, Ca, Mg, K, Fe, Zn, S and Cu were measured using total reflection X-ray fluorescence (TXRF) spectrometry, following the protocol described by Fanesi et al. [12]. An internal standard (gallium) was added to the samples in order to obtain a final concentration of 5 g·mL−1, for absolute quantification. An aliquot of 10 μL of sample was deposited on a quartz sample carrier (Bruker AXS Microanalysis GmbH, Berlin, Germany) and the X-ray fluorescence due to the various elements was measured with a Picofox S2 spectrometer (Bruker AXS Microanalysis GmbH, Berlin, Germany). The spectra were acquired and analyzed with the software SPECTRA 5.3 (Bruker AXS Microanalysis GmbH, Berlin, Germany).
2.4. Analyses on Fish
2.4.1. Color
A Spectro-color®116 colorimeter (Bell Technology Ltd., Auckland, New Zealand), equipped with the Spectral qc 3.6 software, was utilized for colorimetric measurement in the CIELab [13] system. In this system, lightness (L*) is expressed on a 0–100 scale from black to white; redness index (a*) ranges from red (+60) to green (−60) and yellowness index (b*) ranges from yellow (+60) to blue (−60). Color was measured in three different dorsal and ventral sites of the left side of the skin of each fish, and in three different sites of epaxial part of the left and right fillet of each fish. The values of the dorsal and ventral sites for skin and the values from right and left fillets of each specimen were considered for the mean in the inferential analysis.
2.4.2. Total Lipids
Total lipid extraction was performed following a modification of the method by Folch et al. [14]. Freeze-dried samples, reconstituted by the addition of distilled water, were homogenized with a 2:1 chloroform-methanol (v/v) solution and then filtered. The filter was washed several times, and distilled water with 0.88% KCl was added to the filtrate so that the [Choloroform:Methanol]:water ratio was 4:1. After stirring and standing overnight, a biphasic system was obtained. The lower phase, containing lipids dissolved in chloroform, was siphoned off and collected. The total lipid content was determined gravimetrically; after complete vacuum evaporation of the chloroform, lipids were resu spended in a known volume of chloroform (5 mL) and weighed in a crucible (gross weight minus tare).
2.4.3. Determination of Fatty Acid (FA) Composition
FAME (Fatty Acid Methyl Ester) analysis was performed according to Morrison and Smith [15]. Lipids were saponified with 0.5 M KOH in methanol, and FAs were hydrolyzed by adding 2 N HCl. Methyl esters were prepared by transmethylation, using a 14% boron fluoride-methanol solution. Methylated FAs were dissolved in petroleum ether, dried, and finally resuspended in 1 mL of hexane. FA composition was determined by liquid gas chromatography (LGC). A GC Varian 430 gas chromatograph (Varian Inc., Palo Alto, CA, USA), equipped with a flame ionization detector (FID) and a Supelco Omegawax™ 320 capillary column (30 m × 0.32 mm internal diameter, 0.25 μm film and polyethylene glycol bonded phase; Supelco, Bellefonte, PA, USA) was utilized. The oven temperature was kept at 100 °C for 2 min, increased to 160 °C over 4 min at a rate of 12 °C/min, increased to 220 °C over 14 min at the rate of 3 °C/min, and kept at 220 °C for 25 min. Temperatures of injector and detector were set at 220 °C and 300 °C, respectively. One microliter of sample in hexane was injected into the column with the carrier gas (helium), at a constant flow of 1.5 mL/min. The split ratio was 1:20.
The chromatograms were recorded with computing integrator software (Galaxie Chromatography Data System 1.9.302.952; Varian Inc., Palo Alto, CA, USA). FAs were identified by comparing the retention time of the FAMEs in the samples with those of FAME in the standard Supelco 37 FAME mix (Supelco, Bellefonte, PA, USA). FAs were quantified through calibration curves, using tricosanoic acid (C23:0) (Supelco, Bellefonte, PA, USA) as internal standard.
2.5. Plants Biometric Data
At the sampling time, 20 chards from each experimental group were harvested to record the shoots length. A total of 20 pictures of the same samples were shot to evaluate the roots area with Image J ver. 1.49 (Bethesda, MD, USA).
2.6. FTIR-Fourier Transform Infrared Spectroscopy: Fish and Plants
Preliminary trials were performed to determine the correct tissue amount necessary to produce spectra with a good signal-to-noise ratio without band saturation. Plant tissues (leaf, taproot and lateral root) were sampled and stored at −20 °C until the analysis. Tissues were homogenized with milli-Q water using a polytron; 50 μL of sample were deposited on silicon windows, in triplicate. Samples were dried at +80 °C until completely dry. Spectra were collected with a Tensor 27 FTIR spectrometer (Bruker Optics, Ettlingen, Germany). The Bruker system was controlled by an IBM-compatible PC running the software OPUS version 6.5 (Bruker Optik GmbH, Ettlingen, Germany). Bands were attributed to cellular pools according to Giordano et al. [16]. Relative ratios of carbohydrates, lipids and proteins were calculated as described by Domenighini and Giordano [17] and Palmucci et al. [18].
The same protocol was applied to evaluate relative ratios of carbohydrates, lipids and proteins in muscles of D. labrax.
2.7. Carbon to Nitrogen Ratio (CNS Analysis)
Leaf, stem, taproot and lateral root were dried at 80 °C and cellular abundances of C, N and S were determined using an elemental analyzer (EA1108; Carlo Erba Instruments, Milan, Italy), following the method described by Ratti et al. [19]. Data acquisition and analysis were performed with the software EASCLARITY (DataApex Ltd. 2006, Prague, Czech Republic). The same protocol was applied to evaluate carbon to nitrogen ratio in muscles of D. labrax.
2.8. Statistical Analysis
Data were analyzed performing Shapiro-Wilk normality test followed by t-test, with the statistical software package Prism5 (GraphPad Software, La Jolla, San Diego, CA, USA) Significance level was set at p < 0.05. The data of fish analyses were submitted to one-way ANOVA (aquaponic system, with two levels: ASW or AFW), with the PROC GLM of SAS® Software (Cary, NC, USA).
3. Results
3.1. Water Quality
During the experiments, pH varied between 8.0 and 8.4. At the end of the experiment, the values of nitrate (NO3) and phosphate (PO4) were significantly higher (p < 0.05) in ASW than in AFW (Figure 1). During the trial, the values of NH4 and NO2 ranged between 0.05 mg/L and 0.01 mg/L and between 0.03 mg/L and 0.01 mg/L, respectively. At the end of the experiment, the concentrations of Na, Cl, Mg, K, Zn, S and Cu resulted significantly higher in AFW than in ASW, while the concentrations of Ca and Fe were not statistically different (Figure 2) between the two groups.
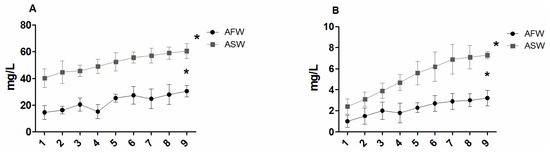
Figure 1.
Trend of NO3 (A) and PO4 (B) measured during the trial in two aquaponic systems (aquaponic freshwater, AFW and aquaponic saltwater, ASW). Knots represent the means of triplicate groups; bars indicate SD. Statistical significance is labelled with asterisks (p < 0.05).
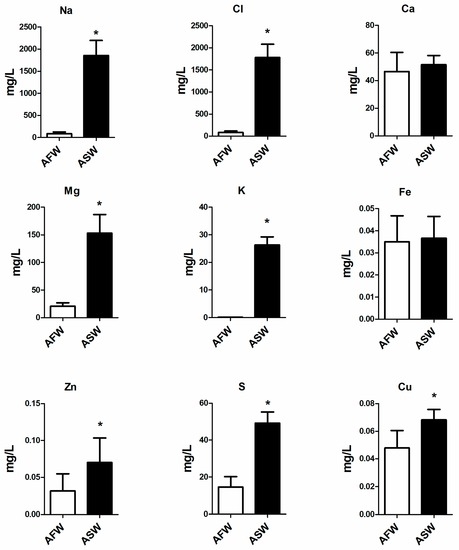
Figure 2.
Concentration of Na, Cl, Ca, Mg, K, Fe, Zn, S, Cu in AFW and ASW groups, measured at the end of the trial. Statistical significance is labelled with asterisks (p < 0.05), bars indicate SD.
3.2. Plant Quality
At the end of the trial, the chard shoots grown in ASW were significantly shorter (p < 0.05) with respect to AFW (31.97 ± 2.5 cm and 40.49 ± 2.69 cm respectively). In addition, plants sampled in ASW showed greater development of lateral roots with respect to chard cultivated in AFW (Figure 3). This was confirmed by the comparison of the area occupied by roots (lateral + taproot) conducted with Image J program that revealed significant differences (p < 0.05) between groups (46.16 ± 16.31 cm2 and 126.9 ± 72.36 cm2, respectively).

Figure 3.
Different phases of sampling. (A) Experimental marine aquaponic systems; (B) Chard cultivated in AFW; (C) Chard cultivated in ASW; (D) Chard root grown in AFW; (E) Chard root grown in ASW.
The analysis of leaf spectra revealed statistically significant differences between groups (p < 0.05); more specifically, a higher lipid to protein ratio and carbohydrate to protein ratio and a lower carbohydrate to lipid ratio were observed in chard cultivated in ASW than in those grown in AFW (Figure 4A). In taproot, saltwater significantly decreased the lipid to protein ratio and increased the carbohydrate to lipid ratio, while it did not affect the carbohydrate to protein ratio as shown in Figure 4B. No significant differences between groups were found in the organic composition of lateral roots (Figure 4C).
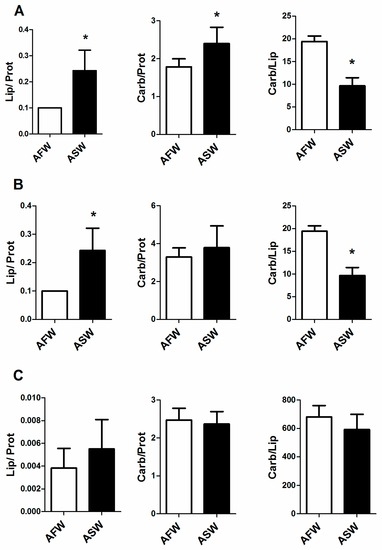
Figure 4.
Lipid to protein, carbohydrate to protein and carbohydrate to lipid ratio in leaf (A); taproot (B) and lateral root (C) of B. vulgaris cultivated in AFW and ASW. Statistical significance is labelled with asterisks (p < 0.05), bars indicate SD.
The ratio carbon to nitrogen in leaf, stem, taproot and lateral root of B. vulgaris grown in AFW and ASW are illustrated in Figure 5; no significant differences were recorded between the groups.
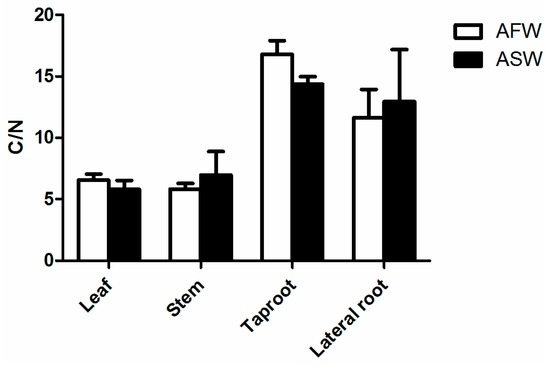
Figure 5.
Carbon to nitrogen ratio (C/N) in leaf, stem taproot and lateral root of Beta vulgaris var. cicla cultivated in AFW and ASW. Bars indicate SD.
3.3. Fish Quality
At the end of the treatment, no significant differences were recorded in body characteristics (fillet, skin, fins, skeletal residual, weight and incidence on total body weight) (Table 1). Regarding skin color, the only significant difference (p < 0.05) between AFW and ASW was found in the yellowness (b*) measured in ventral site (Table 2), whilst more differences were found for color of left and right fillet, as is shown in Table 3.

Table 1.
Body characteristics of European sea bass reared in AFW and ASW.

Table 2.
Skin color at dorsal and ventral sites, in European sea bass reared in AFW and ASW.

Table 3.
Color of the left and right fillet of European sea bass reared in AFW and ASW.
FTIR analysis on muscle revealed a significant decrease in the carbohydrate to protein ratio in muscle of D. labrax reared in ASW whilst no differences were recorded in lipid to protein ratio and in carbohydrate to lipid ratio (Figure 6). Similarly, no differences between groups were found in carbon to nitrogen ratio measured on muscle, as shown in Figure 7.
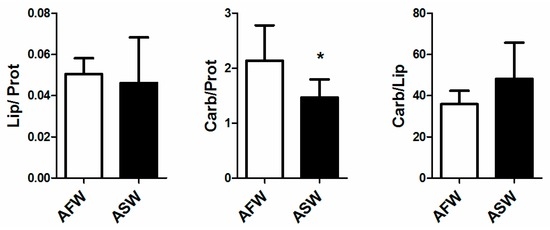
Figure 6.
Lipid to protein, carbohydrate to protein and carbohydrate to lipid ratio in European sea bass muscle, reared in AFW and ASW. Statistical significance is labelled with asterisk (p < 0.05), bars indicate SD.
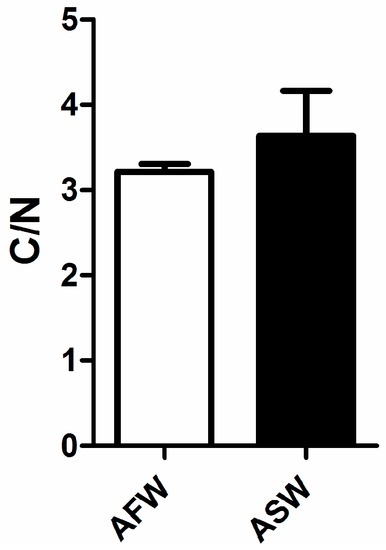
Figure 7.
Carbon to nitrogen ratio in the muscle of European sea bass in the two groups (AFW and ASW). Bars indicate SD.
Total lipids, monounsaturated fatty acids (MUFAs) and polyunsaturated fatty acids (PUFAs) were not statistically different, while saturated fatty acids (SFAs) reported a significant decrease (p < 0.05) in D. labrax reared in AFW with respect to fish farmed in ASW (Table 4). Also, arachidonic acid (AA, 20:4n-6) and docosapentaenoic acid (DPA, 22:5n-3) showed a significant difference between groups. Table 5 reports the fatty acid profile, in percentages, of the European sea bass exposed to freshwater (AFW) and saltwater (ASW).

Table 4.
Groups of fatty acids (in % of total fatty acids) of the fillet in European sea bass reared in AFW and ASW.

Table 5.
Fatty acid profile (in % of total fatty acids) of the fillet in European sea bass muscle, reared in AFW and ASW.
4. Discussion
4.1. Water
The addition of synthetic salt to the ASW system in order to obtain a 20 ppt of salinity substantially increased the ion concentration in the growth medium as compared to AFW. However, although Red Sea salt supplied Fe at a dose of 0.001 g/L (0.5 g in 500 L tank) in ASW, no significant differences were found between groups for this element and its concentration was below the threshold value indicated in hydroponics [20]. These results indicate that the shortage of Fe, which may limit plant growth, also occurs in marine aquaponics aside from freshwater, although the Fe requirement is highly species-specific. NO3 and PO4 in the water were more concentrated in ASW than in AFW, suggesting that a salinity value of 20 ppt may affect the ability of B. vulgaris to assimilate these substances. This result is in accordance with the study by Grattan and Grieve [21] showing that the major ions that usually compose the saline water (as sodium and chlorine) inhibit the phosphate and nitrate uptake.
4.2. Plants
Plant biometric data evidenced salinity induced morphological modification in the plant cultured in ASW, stimulating a significant development of B. vulgaris lateral roots (Figure 3D,E). This may be due to the fact that the increased external salinity (i.e., more negative water potential) diminishes the flow of water into the plant; plants may respond to this by stimulating root growth [22,23,24]. However, FTIR and CN analyses did not reveal significant differences in the allocation of macromolecules in lateral roots. Also, 20 ppt salinity attenuated shoot development, probably by inhibiting the uptake of nutrient such nitrate and phosphate that tend to accumulate into the water (see Section 4.1).
Several studies demonstrated that salinity affects lipid quality and quantity in glycophyte as well as in halophile plants [25,26,27]; this phenomenon was attributed to the fact that lipids are involved in responses to osmotic stress via their role in maintaining membrane, integrity and fluidity [28].
In this study, B. vulgaris grown at 20 ppt of salinity showed a decreased relative lipid content in taproots, and an increased relative lipid content in leaves. Similar results were obtained by Chalbi et al. [23], who studied three different plant species with different levels of tolerance to salinity; these authors discovered that concentration of 200 mM NaCl affected the lipid content both in leaves and roots, as well as in halophilic species. This value is markedly lower than the concentration of Na and Cl measured at the end of the trial in ASW (1854 ± 341.4 mg/L and 1782 ± 299.1 mg/L, respectively), indicating that the osmotic stress affects lipid content. The lipid to protein ratio and carbohydrate to lipid ratio recorded in leaves and in taproot of chards showed the inverse trend between groups; these results suggest that 20 ppt of salinity not only affected the lipid content, but also the other main organic pools in chard. However, the above mentioned data were not correlated with the carbon to nitrogen ratio measured in leaves and taproots.
4.3. Fish
European sea bass, one of the most relevant commercially attractive species in the Mediterranean area, is usually farmed in sea cages or raceways at ~35 ppt. Changes in rearing parameters may result in changes of standard parameters that are indicator of fish quality. In this study, fish acclimated to freshwater maintained body characteristics equal to those of fish reared in saltwater, in accordance with results obtained by Eroldoǧan and Kumlu [29] who studied the same species. However, freshwater seemed to influence the fillet yellowness, resulting significantly lower in fish reared in AFW compared to ASW.
Other than pigmentation, flesh quality is recognized by the analysis of macromolecule composition, and in particular, the fatty acid profile. Literature data on the effects of salinity on fatty acid composition are dissimilar; Xu et al. [30] found a upsurge in muscle PUFA and MUFA of Japanese sea bass (Lateolabrax japonicus) acclimated to saltwater compared to those grown in freshwater. In contrast, according to Eroldoǧan et al. [31], freshwater induced an increase in total lipids respect to saltwater D. labrax. However, in the present research, D. labrax farmed in AFW showed similar total lipids, MUFA and PUFA profiles as well as fish reared in ASW; these results match with the findings of Haliloǧlu et al. [32], who tested the same parameters on rainbow trout (Oncorhynchus mykiss) living in seawater and freshwater. In particular, within PUFAs no differences in n-3 were recorded. These are mostly contained in seafood and play a crucial importance to human health. In fact n-3 PUFAs are considered to have beneficial effects on inflammatory and cardiovascular diseases, neural development and even a protective effect against some kinds of cancers [33]. More specifically, eicosapentaenoic acid (EPA, 20:5n-3) and docosahexaenoic acid (DHA, 22:6n-3) are the most important n-3 PUFAs responsible for fish meat quality and human healthiness [33].
Despite no differences being found in total n-6 PUFAs, European sea bass reared in ASW showed a significant increase in the percentage of arachidonic acid (AA, 20:4n-6). Van Anholt et al. [34] found an increase in the AA content in tilapia (Oreochromis mossambicus) acclimated to seawater, suggesting a possible role of AA in the osmoregulation. However, the rise of AA was more evident in tissues involved in osmoregulation as gills and kidney respect to muscle.
Lastly, results obtained in the present study suggest that freshwater affected the incidence of SFAs, which resulted significantly lower in fillet of European sea bass raised in AFW compared to ASW. As suggested by Hunt et al. [35] who studied the same species, it is possible that fish farmed in freshwater prefer to use SFAs as principal source of energy.
Results obtained by C/N excluded the influence of freshwater in the pool of carbon and nitrogen. However, FTIR revealed a higher carbohydrate to protein ratio in AFW respect to ASW, suggesting a modification in macromolecule allocation.
5. Conclusions
Development of aquaponics is important in countries with markets strictly dependent on marine species and/or limited sources of freshwater. This study broadened the knowledge on marine aquaponics by testing a marine aquaponics system that provides good quality fish meat while reducing growth performance of chards and modifying macromolecule allocation. However, rearing marine species in aquaponics requires a saltwater source or, alternatively, synthetic salt, which is expensive. A further requirement is the need for halophile plants which diminishes the number of species suitable for rearing. This research exploits commercially interesting euryhaline fish in freshwater environment in order to overcome the above-mentioned disadvantages, obtaining fish meat quality comparable to fish reared in saltwater system and chards with good growth performance. In addition, exploiting euryhaline species allows for an increase in profitability by cultivating commercially interesting common plants.
Acknowledgments
The authors wish to thank Marche region (Italy) for the financial support for fellowship (EUREKA program).
Author Contributions
Valentina Nozzi (v.nozzi@univpm.it): conceived and designed the experiments, performed the experiments and analysis, analyzed the data, wrote the paper and revised. Giuliana Parisi (giuliana.parisi@unifi.it): performed the experiments and analysis, analyzed the data, contributed reagents/materials/analysis tools, wrote the paper and revised. Mario Giordano (m.giordano@univpm.it): analyzed the data, contributed reagents/materials/analysis tools, wrote the paper and revised. Davide Di Crescenzo (davide@quaguide.com): contributed with the infrastructure and the reagents/materials. Oliana Carnevali (o.carnevali@univpm.it): Conceived and designed the experiments, contributed reagents/materials/analysis tools, wrote the paper and revised.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- McMurtry, M.R.; Sanders, D.C.; Cure, J.D.; Hodson, R.G.; Haning, B.C.; Amand, E.C.S. Efficiency of water use of an integrated fish/vegetable co-culture system. J. World Aquac. Soc. 1997, 28, 420–428. [Google Scholar] [CrossRef]
- Seawright, D.E.; Stickney, R.R.; Walker, R.B. Nutrient dynamics in integrated aquaculture-hydroponics systems. Aquaculture 1998, 160, 215–237. [Google Scholar] [CrossRef]
- Lennard, W.A.; Leonard, B.V. A comparison of three different hydroponic sub-systems (gravel bed, floating and nutrient film technique) in an Aquaponic Test System. Aquac. Int. 2006, 14, 539–550. [Google Scholar] [CrossRef]
- Kotzen, B.; Appelbaum, S. An investigation of aquaponics using brackish water resources in the Negev Desert. J. Appl. Aquac. 2010, 22, 297–320. [Google Scholar] [CrossRef]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity effects on plants: A review. Ecotoxicol. Environ. Saf. 2005, 60, 324–349. [Google Scholar] [CrossRef] [PubMed]
- Allegrucci, G.; Caccone, A.; Cataudella, S.; Powell, J.R.; Sbordoni, V. Acclimation of the European sea bass to freshwater: Monitoring genetic changes by RAPD polymerase chain reaction to detect DNA polymorphisms. Mar. Biol. 1995, 121, 591–599. [Google Scholar] [CrossRef]
- Riche, M.A.; Pfeiffer, T.J.; Wills, P.S.; Amberg, J.J.; Sepùlveda, M.S. Inland marine fish culture in low salinity recirculating aquaculture systems. Bull. Fish. Res. Agen. 2012, 35, 65–75. [Google Scholar]
- Rakocy, J.E.; Masser, M.P.; Losordo, T.M. Recirculating aquaculture tank production systems: Aquaponics—Integrating fish and plant culture. SRAC Publ. 2006, 454, 1–16. [Google Scholar]
- Lemarié, G.; Dosdat, A.; Covès, D.; Dutto, G.; Gasset, E.; Person-Le Ruyet, J. Effect of chronic ammonia exposure on growth of European seabass (Dicentrarchus labrax) juveniles. Aquaculture 2004, 229, 479–491. [Google Scholar] [CrossRef]
- Dosdat, A.; Person-Le Ruyet, J.; Covès, D.; Dutto, G.; Gusset, E.; Le Roux, A.; Lemarié, G. Effect of chronic exposure to ammonia on growth, food utilization and metabolism of the European sea bass (Dicentrarchus labrax). Aquat. Living Resour. 2003, 16, 509–520. [Google Scholar] [CrossRef]
- Lennard, W.A.; Leonard, B.V. A comparison of reciprocating flow versus constant flow in an integrated, gravel bed, aquaponic test system. Aquac. Int. 2004, 12, 539–553. [Google Scholar] [CrossRef]
- Fanesi, A.; Raven, J.A.; Giordano, M. Growth rate affects the responses of the green alga Tetraselmis suecica to external perturbations. Plant Cell Environ. 2014, 37, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.R.; Lozano, R.D.; Alman, D.H.; Orchard, S.E.; Keitch, J.A.; Connelly, R.; Graham, L.A.; Acree, W.L. CIE Recommendations on uniform colour space, colour difference equations and metric colour terms. Color Res. Appl. 1997, 2, 5–7. [Google Scholar]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1956, 226, 497–509. [Google Scholar]
- Morrison, W.R.; Smith, L.M. Preparation of fatty acid methyl esters and dimethylacetals from lipids with born fluoride-methanol. J. Lipid Res. 1964, 5, 600–608. [Google Scholar] [PubMed]
- Giordano, M.; Kansiz, M.; Heraud, P.; Beardall, J.; Wood, B.; Mc Naughton, D. Fourier transform infrared spectroscopy as a novel tool to investigate changes in intracellular macromolecular pools in the marine microalga Chaetoceros muellerii (Bacillariophyceae). J. Phycol. 2001, 279, 271–279. [Google Scholar] [CrossRef]
- Domenighini, A.; Giordano, M. Fourier transform infrared spectroscopy of microalgae as a novel tool for biodiversity studies, species identification, and the assessment of water quality. J. Phycol. 2009, 45, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Palmucci, M.; Ratti, S.; Giordano, M. Ecological and evolutionary implication of carbon allocation in marine phytoplankton as a function of nitrogen availability: A Fourier transform infrared spectroscopy approach. J. Phycol. 2011, 47, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Ratti, S.; Knoll, A.H.; Giordano, M. Did sulphate availability facilitate the evolutionary expansion of chlorophyll a + c phytoplankton in the oceans? Geobiology 2011, 9, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Tyson, R.V.; Treadwel, D.D.; Simonne, E.H. Opportunities and challenges to sustainability in aquaponic systems. Horttechnology 2011, 21, 1–13. [Google Scholar]
- Grattan, S.R.; Grieve, C.M. Salinity-mineral nutrient relations in horticultural crops. Sci. Hortic. Amst. 1999, 78, 127–157. [Google Scholar] [CrossRef]
- Hsiao, T.C.; Xu, L.K. Sensitivity of growth of roots versus leaves to water stress: Biophysical analysis and relation to water transport. J. Exp. Bot. 2000, 51, 1595–1616. [Google Scholar] [CrossRef] [PubMed]
- Chalbi, N.; Martínez-Ballesta, M.C.; Youssef, N.B.; Carvajal, M. Intrinsic stability of Brassicaceae plasma membrane in relation to changes in proteins and lipids as a response to salinity. J. Plant Physiol. 2015, 175, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Stuiver, C.E.E.; Kuiper, P.J.C.; Marschner, H.; Kylin, A. Effects of salinity and replacement of K+ by Na+ on lipid-composition in 2 sugar-beet inbred lines. Physiol. Plant. 1981, 52, 77–82. [Google Scholar] [CrossRef]
- Kuiper, P.J.C. Environmental changes and lipid metabolism of higher plants. Physiol. Plant. 1985, 64, 118–122. [Google Scholar] [CrossRef]
- Wu, J.; Seliskar, D.M.; Gallagher, J.L. The response of plasma membrane lipid composition in callus of the halophyte Spartina patens (Poaceae) to salinity stress. Am. J. Bot. 2005, 92, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.M.F.; Salama, K.H.A.; Allam, H.Y.H. Role of the plasma membrane in saline conditions: Lipids and proteins. Bot. Rev. 2015, 81, 416–451. [Google Scholar] [CrossRef]
- Eroldoǧan, O.T.; Kumlu, M. Growth performance, body traits and fillet composition of the European sea bass (Dicentrarchus labrax) reared in various salinities and fresh water. Turkish J. Vet. Anim. Sci. 2002, 26, 993–1001. [Google Scholar]
- Xu, J.; Yan, B.; Teng, Y.; Lou, G.; Lu, Z. Analysis of nutrient composition and fatty acid profiles of Japanese sea bass Lateolabrax japonicus (Cuvier) reared in seawater and freshwater. J. Food Compos. Anal. 2010, 23, 401–405. [Google Scholar] [CrossRef]
- Eroldoǧan, O.T.; Kumlu, M.; Aktaş, M. Optimum feeding rates for European sea bass Dicentrarchus labrax L. reared in seawater and freshwater. Aquaculture 2004, 231, 501–515. [Google Scholar] [CrossRef]
- Haliloǧlu, İ.H.; Bayır, A.; Sirkecioǧlu, N.A.; Aras, M.N.; Atamanalp, M. Comparison of fatty acid composition in some tissues of rainbow trout (Oncorhynchus mykiss) living in seawater and freshwater. Food Chem. 2004, 86, 55–59. [Google Scholar] [CrossRef]
- Tocher, D.R. Omega-3 long-chain polyunsaturated fatty acids and aquaculture in perspective. Aquaculture 2015, 449, 94–107. [Google Scholar] [CrossRef]
- Van Anholt, R.D.; Spanings, F.A.T.; Nixon, O.; Wendelaar Bonga, S.E.; Koven, W.M. The effects of arachidonic acid on the endocrine and osmoregulatory response of tilapia (Oreochromis mossambicus) acclimated to seawater and subjected to confinement stress. Fish Physiol. Biochem. 2012, 38, 703–713. [Google Scholar] [CrossRef] [PubMed]
- Hunt, A.Ö.; Özkan, F.; Engin, K.; Tekelioğlu, N. The effects of freshwater rearing on the whole body and muscle tissue fatty acid profile of the European sea bass (Dicentrarchus labrax). Aquac. Int. 2010, 19, 51–61. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).