Adsorption of Sunscreen Compounds from Wastewater Using Commercial Activated Carbon: Detailed Kinetic and Thermodynamic Analyses
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. AC Material Characterization
2.3. Batch Adsorption Study
2.4. Adsorption Isotherms
2.5. Kinetic Study
2.6. Thermodynamic Study
2.7. Sunscreen Analysis
2.8. QA and QC
3. Results
3.1. Adsorbent Material Characterization
3.2. Optimization of Adsorption Parameters
3.3. Adsorption Isotherms
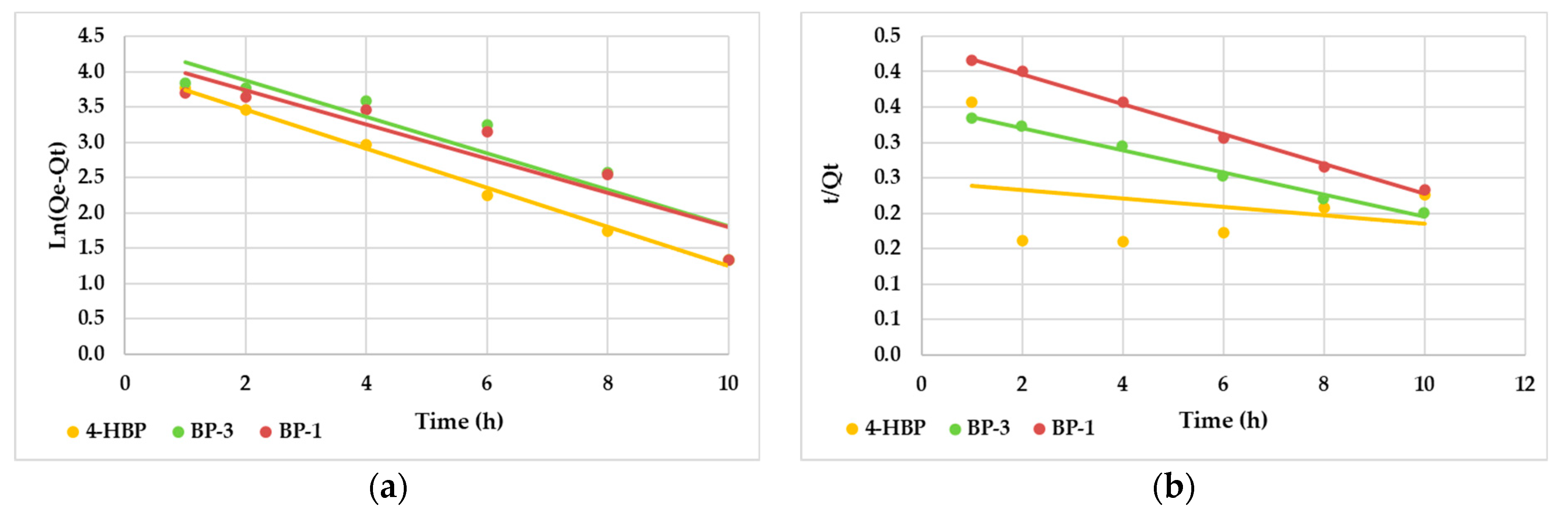
3.4. Adsorption Kinetic Studies
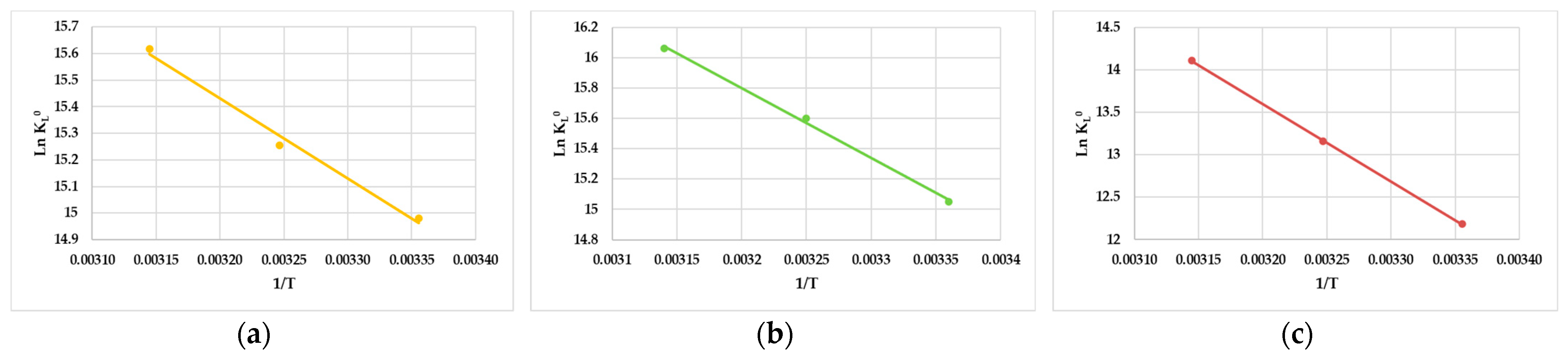
3.5. Adsorption Thermodynamic Studies
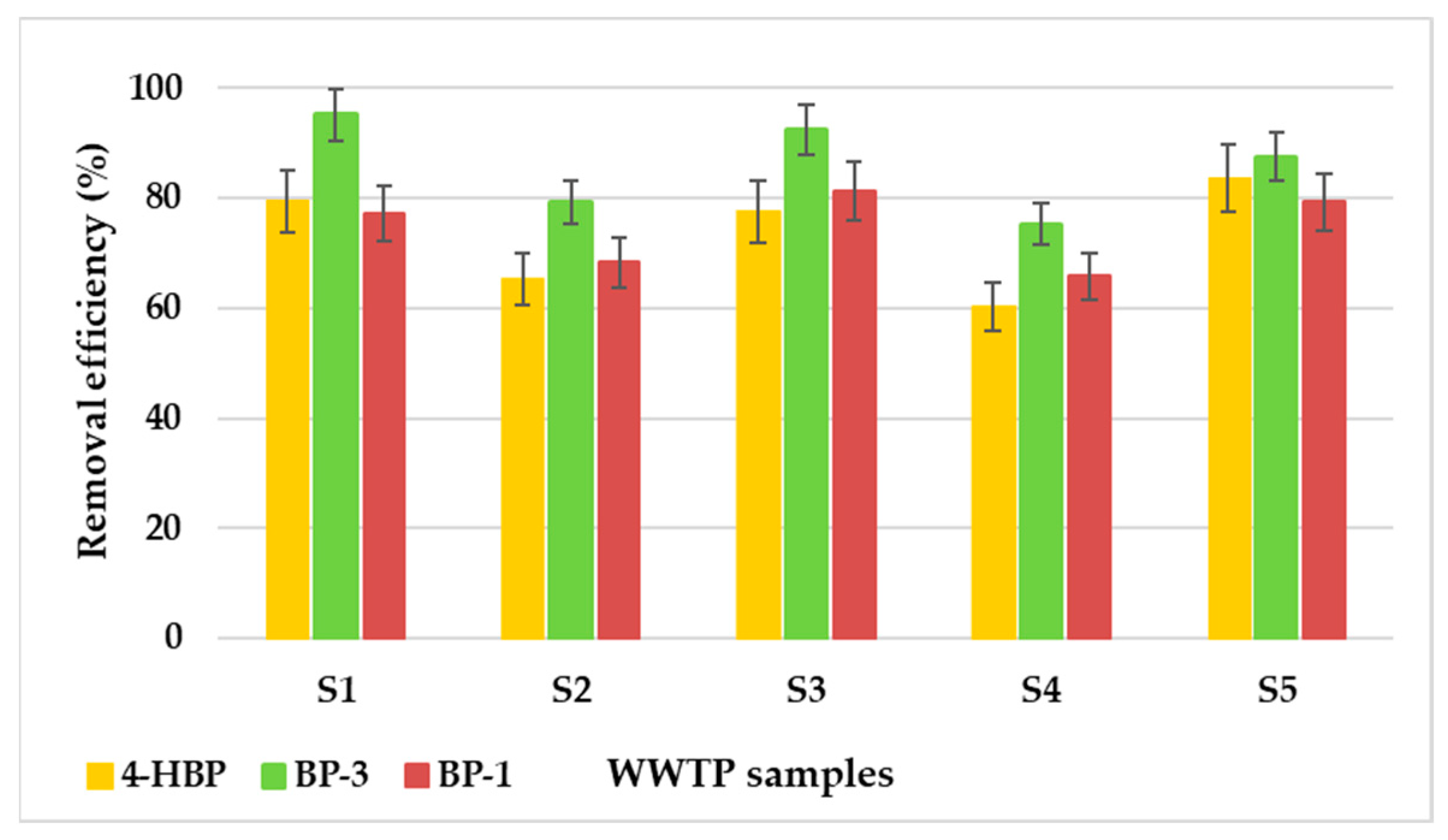
3.6. Elimination of Targeted Sunscreens from Actual Wastewater Samples
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peinado, F.M.; Ocón-Hernández, O.; Iribarne-Durán, L.M.; Vela-Soria, F.; Ubiña, A.; Padilla, C.; Mora, J.C.; Cardona, J.; León, J.; Fernández, M.; et al. Cosmetic and personal care product use, urinary levels of parabens and benzophenones, and risk of endometriosis: Results from the EndEA study. Environ. Res. 2021, 196, 110342. [Google Scholar] [CrossRef]
- Song, J.; Na, J.; An, D.; Jung, J. Role of benzophenone-3 additive in chronic toxicity of polyethylene microplastic fragments to Daphnia magna. Sci. Total. Environ. 2021, 800, 149638. [Google Scholar] [CrossRef]
- Kim, S.; Choi, K. Occurrences, toxicities, and ecological risks of benzophenone-3, a common component of organic sunscreen products: A mini-review. Environ. Int. 2014, 70, 143–157. [Google Scholar] [CrossRef]
- Hu, L.-X.; Cheng, Y.-X.; Wu, D.; Fan, L.; Zhao, J.-H.; Xiong, Q.; Chen, Q.-L.; Liu, Y.-S.; Ying, G.-G. Continuous input of organic ultraviolet filters and benzothiazoles threatens the surface water and sediment of two major rivers in the Pearl River Basin. Sci. Total. Environ. 2021, 798, 149299. [Google Scholar] [CrossRef]
- Tsui, M.M.P.; Leung, H.W.; Wai, T.C.; Yamashita, N.; Taniyasu, S.; Liu, W.; Lam, P.K.S.; Murphy, M.B. Occurrence, distribution and ecological risk assessment of multiple classes of UV filters in surface waters from different countries. Water Res. 2014, 67, 55–65. [Google Scholar] [CrossRef]
- Carstensen, L.; Zippel, R.; Fiskal, R.; Börnick, H.; Schmalz, V.; Schubert, S.; Schaffer, M.; Jungmann, D.; Stolte, S. Trace analysis of benzophenone-type UV filters in water and their effects on human estrogen and androgen receptors. J. Hazard. Mater. 2023, 456, 131617. [Google Scholar] [CrossRef]
- Carstensen, L.; Beil, S.; Börnick, H.; Stolte, S. Structure-related endocrine-disrupting potential of environmental transformation products of benzophenone-type UV filters: A review. J. Hazard. Mater. 2022, 430, 128495. [Google Scholar] [CrossRef]
- Moradi, N.; Amin, M.M.; Fatehizadeh, A.; Ghasemi, Z. Degradation of UV-filter Benzophenon-3 in aqueous solution using TiO2 coated on quartz tubes. J. Environ. Health Sci. Eng. 2018, 16, 213–228. [Google Scholar] [CrossRef]
- Saif, S.; Adil, S.F.; Khan, M.; Hatshan, M.R.; Khan, M.; Bashir, F. Adsorption Studies of Arsenic(V) by CuO Nanoparticles Synthesized by Phyllanthus emblica Leaf-Extract-Fueled Solution Combustion Synthesis. Sustainability 2021, 13, 2017. [Google Scholar] [CrossRef]
- Sabzehmeidani, M.M.; Mahnaee, S.; Ghaedi, M.; Heidari, H.; Roy, V.A.L. Carbon based materials: A review of adsorbents for inorganic and organic compounds. Mater. Adv. 2021, 2, 598–627. [Google Scholar] [CrossRef]
- Du, E.; Li, J.; Zhou, S.; Li, M.; Liu, X.; Li, H. Insight into the Degradation of Two Benzophenone-Type UV Filters by the UV/H2O2 Advanced Oxidation Process. Water 2018, 10, 1238. [Google Scholar] [CrossRef]
- Gong, P.; Yuan, H.; Zhai, P.; Xue, Y.; Li, H.; Dong, W.; Mailhot, G. Investigation on the degradation of benzophenone-3 by UV/H2O2 in aqueous solution. Chem. Eng. J. 2015, 277, 97–103. [Google Scholar] [CrossRef]
- Hopkins, Z.R.; Snowberger, S.; Blaney, L. Ozonation of the oxybenzone, octinoxate, and octocrylene UV-filters: Reaction kinetics, absorbance characteristics, and transformation products. J. Hazard. Mater. 2017, 338, 23–32. [Google Scholar] [CrossRef]
- Lee, Y.M.; Lee, G.; Kim, M.K.; Zoh, K.D. Kinetics and degradation mechanism of Benzophenone-3 in chlorination and UV/chlorination reactions. Chem. Eng. J. 2020, 393, 124780. [Google Scholar] [CrossRef]
- Bhadra, B.N.; Yoo, D.K.; Jhung, S.H. Carbon-derived from metal-organic framework MOF-74: A remarkable adsorbent to remove a wide range of contaminants of emerging concern from water. Appl. Surf. Sci. 2020, 504, 144348. [Google Scholar] [CrossRef]
- Wang, T.; He, J.; Lu, J.; Zhou, Y.; Wang, Z.; Zhou, Y. Adsorptive removal of PPCPs from aqueous solution using carbon-based composites: A review. Chin. Chem. Lett. 2022, 33, 3585–3593. [Google Scholar] [CrossRef]
- Zhu, X.; He, M.; Sun, Y.; Xu, Z.; Wan, Z.; Hou, D.; Alessi, D.S.; Tsang, D.C.W. Insights into the adsorption of pharmaceuticals and personal care products (PPCPs) on biochar and activated carbon with the aid of machine learning. J. Hazard. Mater. 2022, 423 Pt B, 127060. [Google Scholar] [CrossRef]
- Elias, K.D.; Ejidike, I.P.; Mtunzi, F.M.; Pakade, V.E. Endocrine Disruptors-(estrone and β-estradiol) removal from water by Nutshell activated carbon: Kinetic, Isotherms and Thermodynamic studies. Chem. Thermodyn. Therm. Anal. 2021, 3–4, 100013. [Google Scholar] [CrossRef]
- Pirvu, F.; Covaliu-Mierla, C.I.; Paun, I.; Paraschiv, G.; Iancu, V. Treatment of Wastewater Containing Nonsteroidal Anti-Inflammatory Drugs Using Activated Carbon Material. Materials 2022, 15, 559. [Google Scholar] [CrossRef]
- Serban, G.V.; Iancu, V.I.; Dinu, C.; Tenea, A.; Vasilache, N.; Cristea, I.; Niculescu, M.; Ionescu, I.; Chiriac, F.L. Removal Efficiency and Adsorption Kinetics of Methyl Orange from Wastewater by Commercial Activated Carbon. Sustainability 2023, 15, 12939. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, X.; Xiang, Y.; Wang, P.; Zhang, J.; Zhang, F.; Tang, L. Modification of biochar derived from sawdust and its application in removal of tetracycline and copper from aqueous solution: Adsorption mechanism and modelling. Bioresour. Technol. 2017, 245, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Zietzschmann, F.; Stutzer, C.; Jekel, M. Granular activated carbon adsorption of organic micro-pollutants in drinking water and treated wastewater—Aligning breakthrough curves and capacities. Water Res. 2016, 92, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yue, Q.; Gao, B.; Gao, Y.; Xu, X.; Li, Q.; Wang, Y. Adsorption and cosorption of ciprofloxacin and Ni(II) on activated carbon-mechanism study. J. Taiwan Inst. Chem. Eng. 2014, 45, 681–688. [Google Scholar] [CrossRef]
- Boudrahem, N.; Delpeux-Ouldriane, S.; Khenniche, L.; Boudrahem, F.; Aissani-Benissad, F.; Gineys, M. Single and mixture adsorption of clofibric acid, tetracycline and paracetamol onto Activated carbon developed from cotton cloth residue. Process. Saf. Environ. Prot. 2017, 111, 544–559. [Google Scholar] [CrossRef]
- Sbardella, L.; Comas, J.; Fenu, A.; Rodriguez-Roda, I.; Weemaes, M. Advanced biological activated carbon filter for removing pharmaceutically active compounds from treated wastewater. Sci. Total. Environ. 2018, 636, 519–529. [Google Scholar] [CrossRef]
- Teixeira, S.; Delerue-Matos, C.; Santos, L. Application of experimental design methodology to optimize antibiotics removal by walnut shell based activated carbon. Sci. Total. Environ. 2019, 646, 168–176. [Google Scholar] [CrossRef]
- Charaabi, S.; Tchara, L.; Marminon, C.; Bouaziz, Z.; Holtzinger, G.; Pensé-Lhéritier, A.-M.; Le Borgne, M.; Issa, S. A comparative adsorption study of benzophenone-3 onto synthesized lipophilic organosilicate, Laponite and montmorillonite. Appl. Clay Sci. 2019, 170, 114–124. [Google Scholar] [CrossRef]
- Rossner, A.; Snyder, S.A.; Knappe, D.R.U. Removal of emerging contaminants of concern by alternative adsorbents. Water Res. 2009, 43, 3787–3796. [Google Scholar] [CrossRef]
- Tran, T.; Dang, B.T.; Thuy, L.T.T.; Hoang, H.G.; Bui, X.T.; Le, V.G.; Lin, C.; Nguyen, M.K.; Nguyen, K.Q.; Nguyen, P.T.; et al. Advanced Treatment Technologies for the Removal of Organic Chemical Sunscreens from Wastewater: A Review. Curr. Pollut. Rep. 2022, 8, 288–302. [Google Scholar] [CrossRef]
- Chiriac, F.L.; Paun, I.; Pirvu, F.; Iancu, V.; Galaon, T. Distribution, removal efficiencies and environmental risk assessment of benzophenone and salicylate UV filters in WWTPs and surface waters from Romania. New J. Chem. 2021, 45, 2478. [Google Scholar] [CrossRef]
- Abril, D.; Ferrer, V.; Mirabal-Gallardo, Y.; Cabrera-Barjas, G.; Segura, C.; Marican, A.; Pereira, A.; Durán-Lara, E.F.; Valdés, O. Comparative Study of Three Dyes’ Adsorption onto Activated Carbon from Chenopodium quinoa Willd and Quillaja saponaria. Materials 2022, 15, 4898. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.; Kumar, A.; Dhodapkar, R.; Pal, S. Adsorption of five emerging contaminants on activated carbon from aqueous medium: Kinetic characteristics and computational modeling for plausible mechanism. Environ. Sci. Pollut. Res. 2021, 28, 21347–21358. [Google Scholar] [CrossRef] [PubMed]
- Chiriac, F.L.; Stoica, C.; Paun, I.; Pirvu, F.; Galaon, T.; Nita-Lazar, M. Biodegradation of two organic ultraviolet-filters by single bacterial strains. Int. J. Environ. Sci. Technol. 2023, 20, 9065–9076. [Google Scholar] [CrossRef]
- Chiriac, F.L.; Lucaciu, I.E.; Paun, I.; Pirvu, F.; Gheorghe, S. In Vivo Bioconcentration, Distribution and Metabolization of Benzophenone-3 (BP-3) by Cyprinus carpio (European Carp). Foods 2022, 11, 1627. [Google Scholar] [CrossRef] [PubMed]
- Scapin, E.; Maciel, G.P.d.S.; Polidoro, A.D.S.; Lazzari, E.; Benvenutti, E.V.; Falcade, T.; Jacques, R.A. Activated Carbon from Rice Husk Biochar with High Surface Area. Biointerface Res. Appl. Chem. 2021, 11, 10265–10277. [Google Scholar] [CrossRef]
- Kumar, A.; Jena, H.M. High surface area microporous activated carbons prepared from Fox nut (Euryale ferox) shell by zinc chloride activation. Appl. Surf. Sci. 2015, 356, 753–761. [Google Scholar] [CrossRef]
- Phele, M.J.; Ejidike, I.P.; Mtunzi, F.M. Adsorption efficiency of activated macadamia nutshell for the removal Organochlorine pesticides: Endrin and 4,4-DDT from aqueous solution. J. Pharm. Sci. Res. 2019, 11, 258–262. [Google Scholar]
- Aljeboree, A.M.; Alshirifi, A.N.; Alkaim, A.F. Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon. Arab. J. Chem. 2017, 10, S3381–S3393. [Google Scholar] [CrossRef]
- Rincon-Silva, N.G.; Moreno-Pirajan, J.C.; Giraldo, L. Equilibrium, kinetics and thermodynamics study of phenols adsorption onto activated carbon obtained from lignocellulosic material (Eucalyptus Globulus labill seed). Adsorption 2016, 22, 33–48. [Google Scholar] [CrossRef]
- Machedi, S.; Ejidike, I.P.; Mtunzi, F.M.; Pakade, V.E.; Klink, M.J. Chlorinated Phenols Sorption Performance by Macadamia Activated Carbon and Grafted Macadamia Activated Carbon: Characterization, Kinetics, and Thermodynamic studies. Orient. J. Chem. 2019, 35, 1469–1479. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. Part I. Solids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- AlOthman, Z.A.; Habila, M.A.; Ali, R.; Ghafar, A.A.; Hassouna, M.S.E. Valorization of two waste streams into activated carbon and studying its adsorption kinetics, equilibrium isotherms and thermodynamics for methylene blue removal. Arab. J. Chem. 2023, 2, 1148–1158. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Über die Adsorption in Lösungen. Z. Phys. Chem. 1906, 57U, 385–470. [Google Scholar] [CrossRef]
- Archana, G.; Dhodapkar, R.; Kumar, A. Ecotoxicological risk assessment and seasonal variation of some pharmaceuticals and personal care products in the sewage treatment plant and surface water bodies (lakes). Environ. Monit. Assess. 2017, 189, 446. [Google Scholar] [CrossRef] [PubMed]
- Altıntıg, E.; Yenigun, M.; Sarı, A.; Altundag, H.; Tuzen, M.; Saleh, T.A. Facile synthesis of zinc oxide nanoparticles loaded activated carbon as an eco-friendly adsorbent for ultra-removal of malachite green from water. Environ. Technol. Innov. 2021, 21, 101305. [Google Scholar] [CrossRef]
- Tengfei, C.H.U.; Dongdong, L.I.; Erdeng, D.U.; Wenhai, C.H.U.; Yingqing, G.U.O.; Naiyun, G.A.O. Adsorption characteristics and adsorption thermodynamics of typical UV sunscreen oxybenzone(BP-3)at trace level in water by carbon-based adsorption materials. Acta Sci. Circumstantiae 2016, 3, 865–872. [Google Scholar] [CrossRef]
- Tumpa, N.F.; Jeong, Y.-K. Adsorptive Removal of a Pharmaceutical and Personal Care Product Oxybenzone from Water with “Metal-Organic Frameworks”. Asian Rev. Environ. Earth Sci. 2018, 5, 1–7. [Google Scholar] [CrossRef]

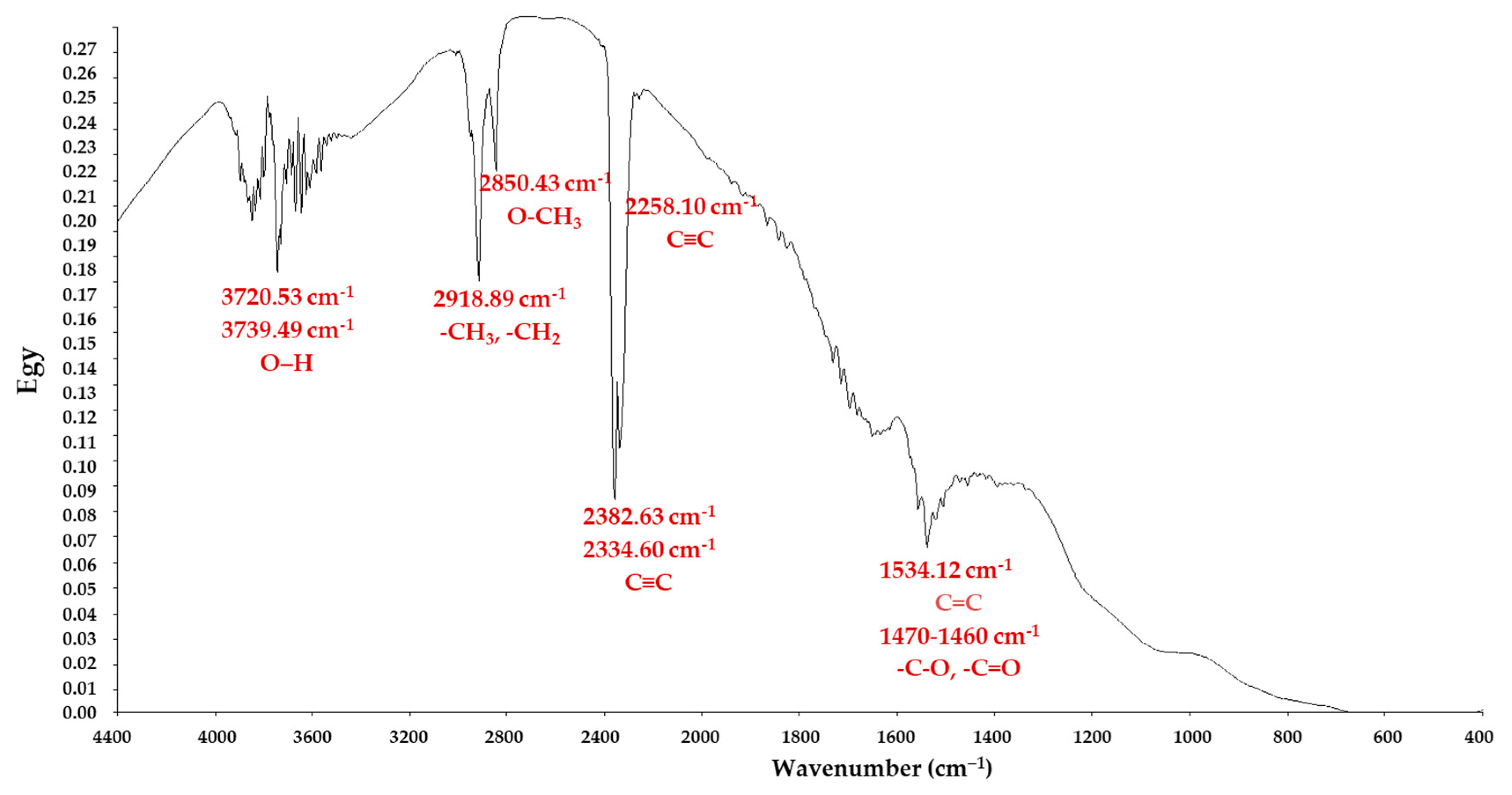
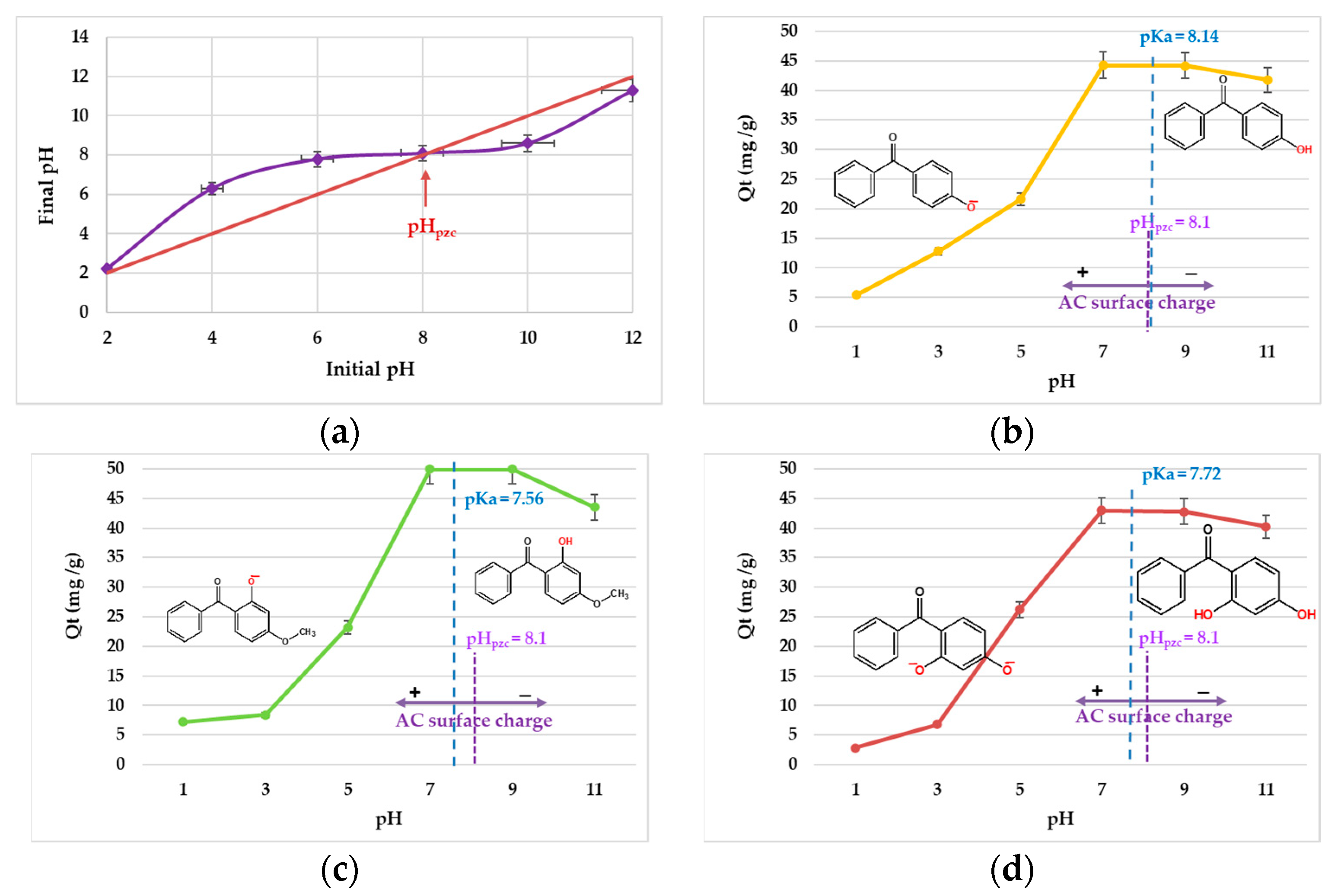

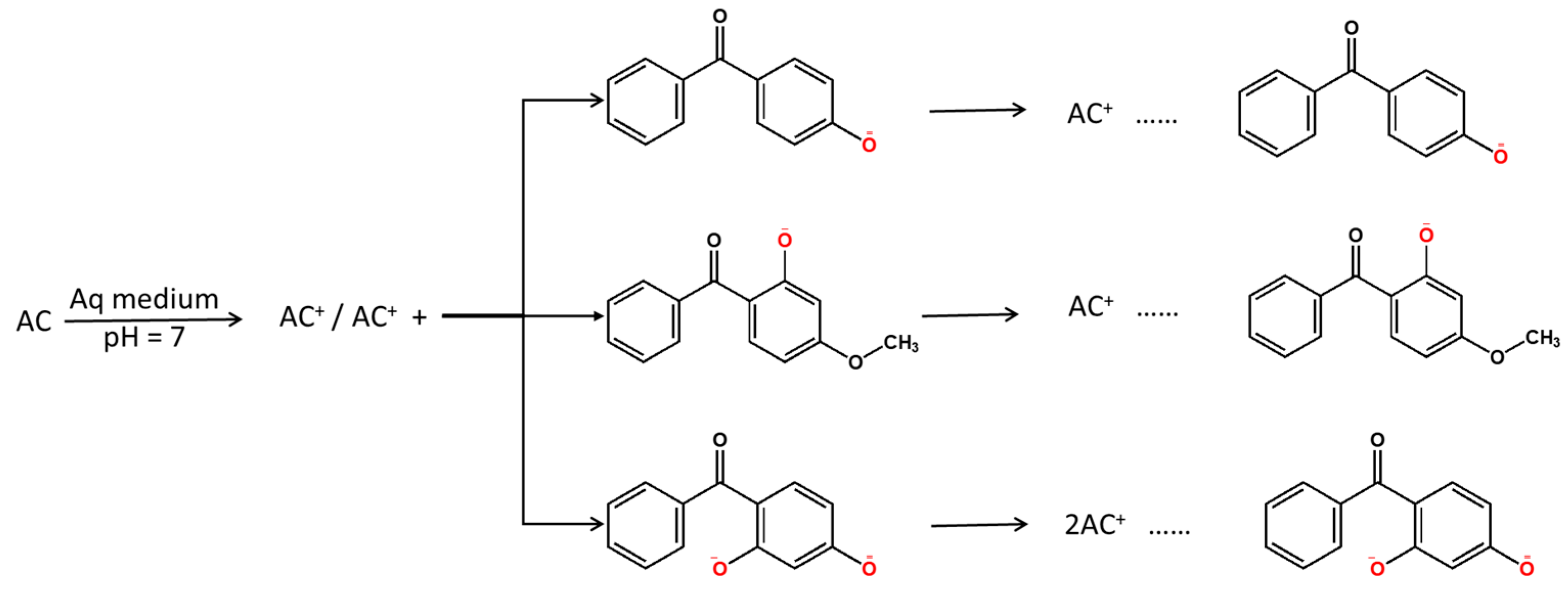
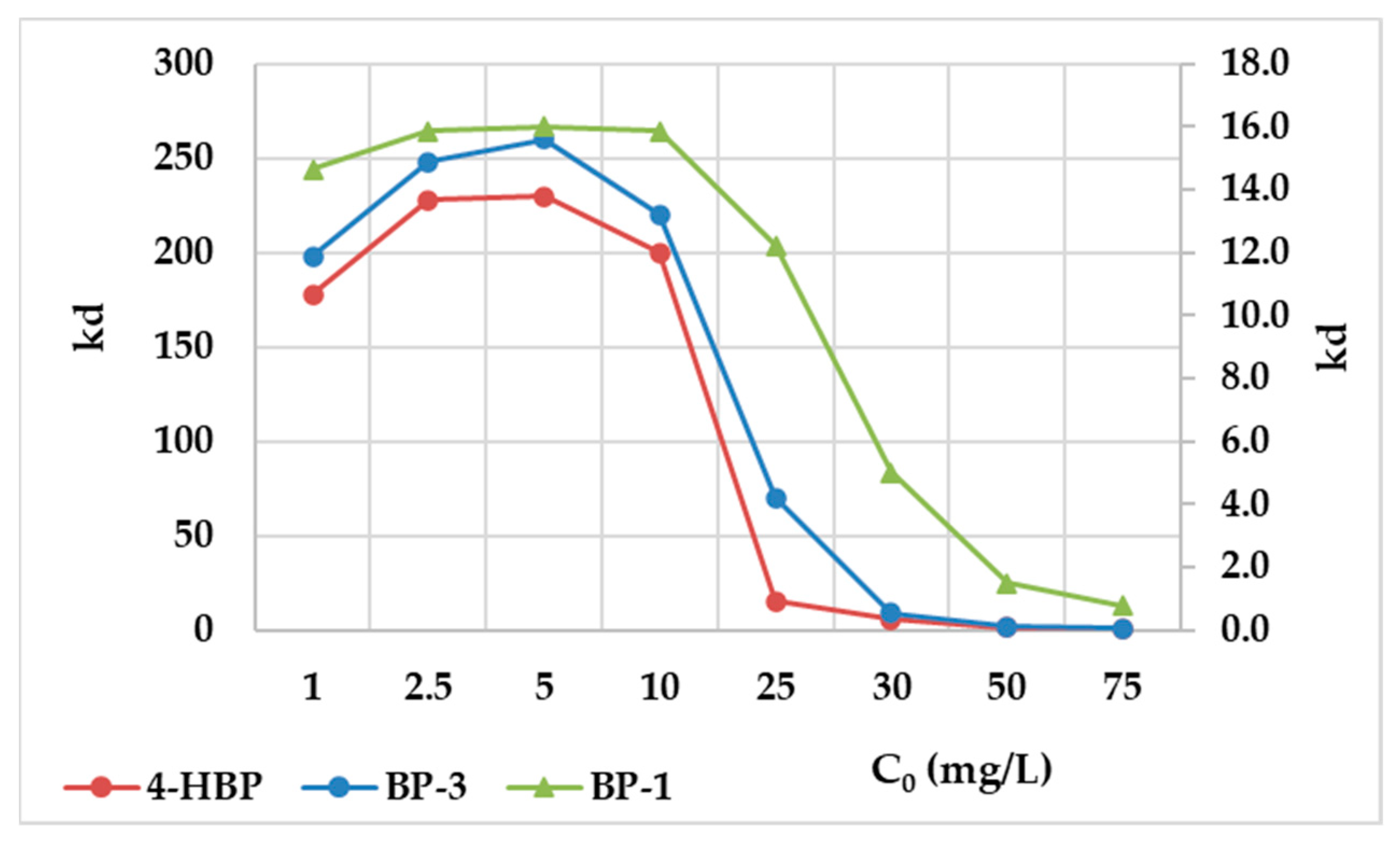

| Compound | Abbreviation | Molecular Formula | Molecular Weight | Solubility (mg/L) | Log Know a | pKa | Chemical Structure |
|---|---|---|---|---|---|---|---|
| 4-hydroxybenzophenone | 4-HBP | C13H10O2 | 198.2 | 405.8 | 3.07 | 8.14 | 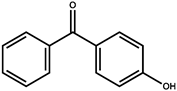 |
| 2,4-dihydroxybenzophenone | BP-1 | C13H10O3 | 214.2 | 413.4 | 2.96 | 7.72 |  |
| 2-hydroxy-4-methoxy-benzophenone (oxybenzone) | BP-3 | C14H12O3 | 228.2 | 68.56 | 3.52 | 7.56 | 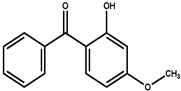 |
| Sunscreen Compound | Retention Time (min) | MRM Transition | FV (V) | CE (V) | CAV (V) | Dwell Time (ms) | ESI Mode |
|---|---|---|---|---|---|---|---|
| BP-3 | 3.75 | 229→151 | 135 | 20 | 1 | 250 | Positive |
| 4-HBP | 7.12 | 197→92.0 | 150 | 45 | 5 | 250 | Negative |
| BP-1 | 9.74 | 213→135 | 130 | 20 | 4 | 250 | Negative |
| Isotherm Model | 4-HBP | BP-3 | BP-1 |
|---|---|---|---|
| Langmuir isotherm model | |||
| Qe (mg/g) | 43.8 | 48.8 | 41.1 |
| Qm (mg/g) | 44 | 49 | 44 |
| KL (L/mg) | 9.81 | 10.5 | 0.57 |
| RL | 0.0041 | 0.0038 | 0.0659 |
| R2 | 0.9999 | 0.9999 | 0.9964 |
| Freundlich isotherm model | |||
| KF (mg/g) | 22 | 28 | 20 |
| 1/n | 0.3450 | 0.2625 | 0.5171 |
| R2 | 0.8119 | 0.4204 | 0.8209 |
| Kinetic Model | 4-HBP | BP-3 | BP-1 |
|---|---|---|---|
| Pseudo-first-order kinetic model | |||
| Qe (mg/g) experimental | 53.9 | 80.4 | 68.1 |
| Q (mg/g) theoretical | 44.3 | 49.9 | 42.9 |
| K1 (L/mg) | 0.028 | 0.026 | 0.024 |
| R2 | 0.9922 | 0.8177 | 0.8627 |
| Pseudo-second-order kinetic model | |||
| Qe (mg/g) experimental | 163.9 | 52.6 | 47.2 |
| K2 | 0.0002 | 0.0007 | 0.0010 |
| R2 | 0.0798 | 0.9908 | 0.9961 |
| Sunscreen | Temperature (K) | KL (L/mg) | KL0 (Dimensionless) | ΔG0 (kJ/mol) | ΔH0 (kJ/mol) | ΔS0 (J/molK) |
|---|---|---|---|---|---|---|
| 4-HBP | 298 | 9.80 | 32.1 | −10.8 | −3209 | 25.7 |
| 308 | 12.9 | 42.2 | −11.1 | |||
| 318 | 18.5 | 60.6 | −11.4 | |||
| BP-3 | 298 | 10.5 | 34.4 | −13.5 | −4590 | 30.5 |
| 308 | 18.2 | 59.6 | −13.8 | |||
| 318 | 28.9 | 94.6 | −14.1 | |||
| BP-1 | 298 | 0.6 | 1.96 | −22.7 | −9092 | 42.7 |
| 308 | 1.58 | 5.17 | −23.1 | |||
| 318 | 4.09 | 13.39 | −23.5 |
| Sample | 4-HBP | BP-3 | BP-1 |
|---|---|---|---|
| (mg/L ± RSD) | |||
| S1 | 5.15 ± 0.12 | 1.20 ± 0.03 | 5.70 ± 0.15 |
| S2 | 8.65 ± 0.20 | 5.20 ± 0.14 | 7.93 ± 0.21 |
| S3 | 5.63 ± 0.13 | 1.90 ± 0.05 | 4.68 ± 0.13 |
| S4 | 9.93 ± 0.23 | 6.20 ± 0.16 | 8.55 ± 0.23 |
| S5 | 4.10 ± 0.09 | 3.13 ± 0.08 | 5.15 ± 0.14 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gheorghe, S.; Iancu, V.I.; Ionescu, I.A.; Pirvu, F.; Paun, I.C.; Pascu, L.F.; Chiriac, F.L. Adsorption of Sunscreen Compounds from Wastewater Using Commercial Activated Carbon: Detailed Kinetic and Thermodynamic Analyses. Water 2023, 15, 4190. https://doi.org/10.3390/w15234190
Gheorghe S, Iancu VI, Ionescu IA, Pirvu F, Paun IC, Pascu LF, Chiriac FL. Adsorption of Sunscreen Compounds from Wastewater Using Commercial Activated Carbon: Detailed Kinetic and Thermodynamic Analyses. Water. 2023; 15(23):4190. https://doi.org/10.3390/w15234190
Chicago/Turabian StyleGheorghe, Stefania, Vasile Ion Iancu, Ioana Alexandra Ionescu, Florinela Pirvu, Iuliana Claudia Paun, Luoana Florentina Pascu, and Florentina Laura Chiriac. 2023. "Adsorption of Sunscreen Compounds from Wastewater Using Commercial Activated Carbon: Detailed Kinetic and Thermodynamic Analyses" Water 15, no. 23: 4190. https://doi.org/10.3390/w15234190
APA StyleGheorghe, S., Iancu, V. I., Ionescu, I. A., Pirvu, F., Paun, I. C., Pascu, L. F., & Chiriac, F. L. (2023). Adsorption of Sunscreen Compounds from Wastewater Using Commercial Activated Carbon: Detailed Kinetic and Thermodynamic Analyses. Water, 15(23), 4190. https://doi.org/10.3390/w15234190







