Heavy Metal Contamination in Surface Water of Harike Wetland, India: Source and Health Risk Assessment
Abstract
1. Introduction
2. Materials and Methods
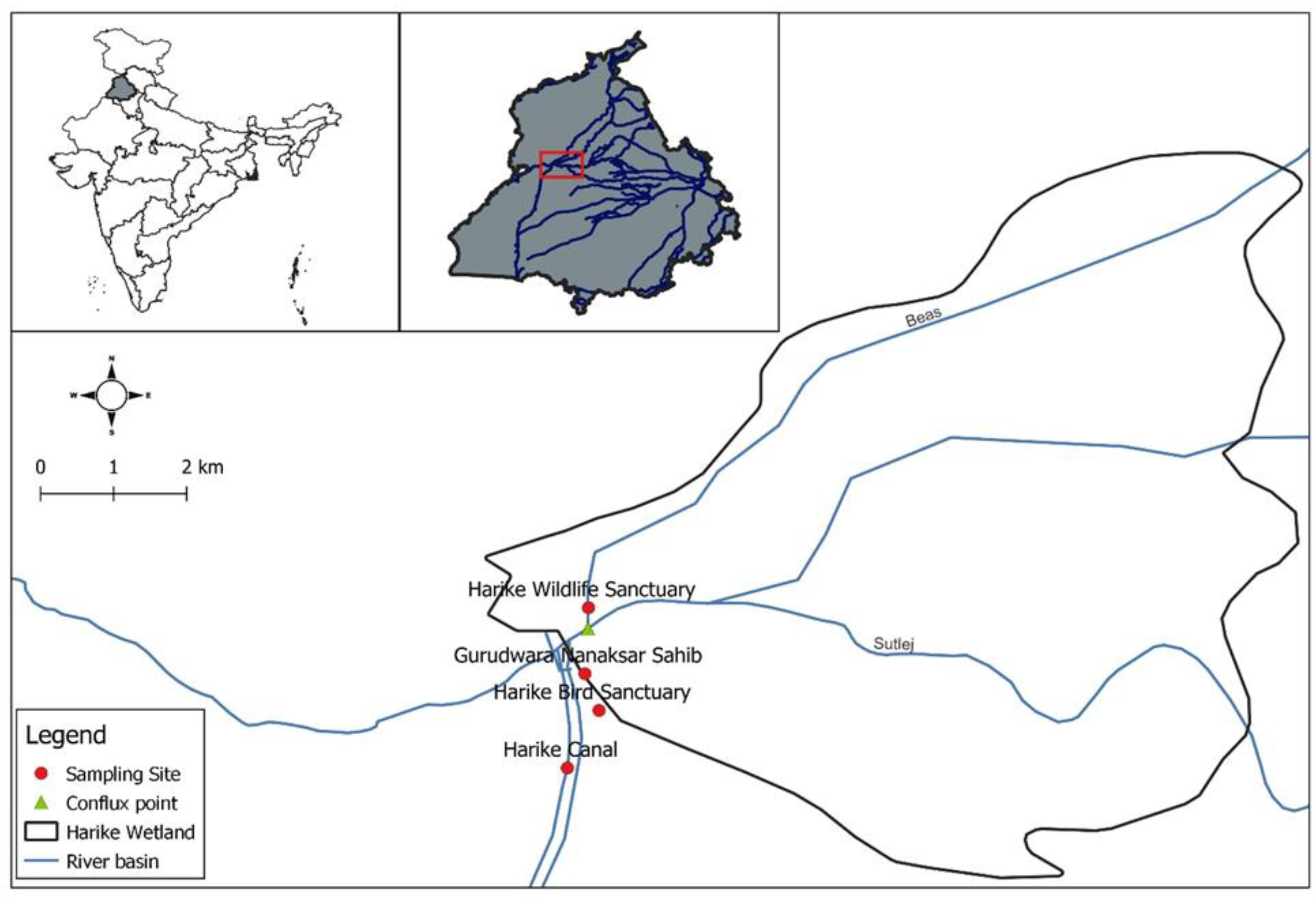
2.1. Site Description
2.2. Sampling
2.3. Chemical Analysis
2.4. Statistical Analysis
2.4.1. Single-Factor Pollution Index
2.4.2. Nemerow Comprehensive Pollution Index
2.4.3. Health Risk Assessment
3. Results
3.1. Descriptive Statistical Analysis
3.2. Multivariate Statistical Analysis
3.2.1. Correlation Analysis
3.2.2. Principal Component Analysis
3.2.3. Heavy Metal Pollution Indices
3.2.4. Health Risk Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arya, S.; Syriac, E.K. Wetlands: The living waters-A review. Agric. Rev. 2018, 39, 122–129. [Google Scholar] [CrossRef]
- Home Page|The Convention on Wetlands. Available online: https://www.ramsar.org/ (accessed on 21 August 2023).
- Jain, S.K.; Sarkar, A.; Garg, V. Impact of Declining Trend of Flow on Harike Wetland, India. Water Resour. Manag. 2007, 22, 409–421. [Google Scholar] [CrossRef]
- Jangu, S.; Kumar, D.; Brraich, O.S.; Baek, K.-H.; Mishra, A.K. Alterations in ultra-structures and elemental composition of ovaries of Labeo rohita (Hamilton-Buchanan) as a pollution indicator in Harike Wetland (Ramsar Site), India. J. Histotechnol. 2020, 44, 62–69. [Google Scholar] [CrossRef]
- Kaur, A.; Datta, S.N.; Tewari, G. Study on biometrics and biology of Rohu, Labeo rohita from Harike wetland-Ramsar site. J. Entomol. Zool. Stud. 2018, 6, 496–500. [Google Scholar]
- Kaur, J.; Kaur, V.; Pakade, Y.B.; Katnoria, J.K. A study on water quality monitoring of Buddha Nullah, Ludhiana, Punjab (India). Environ. Geochem. Health 2021, 43, 2699–2722. [Google Scholar] [CrossRef] [PubMed]
- Duncan, A.E.; de Vries, N.; Nyarko, K.B. Assessment of heavy metal pollution in the sediments of the River Pra and its tributaries. Water Air Soil Pollut. 2018, 229, 272. [Google Scholar] [CrossRef] [PubMed]
- Strzebońska, M.; Jarosz-Krzemińska, E.; Adamiec, E. Assessing historical mining and smelting effects on heavy metal pollution of river systems over span of two decades. Water Air Soil Pollut. 2017, 228, 141. [Google Scholar] [CrossRef]
- Singh, V.; Singh, N.; Rai, S.N.; Kumar, A.; Singh, A.K.; Singh, M.P.; Sahoo, A.; Shekhar, S.; Vamanu, E.; Mishra, V. Heavy Metal Contamination in the Aquatic Ecosystem: Toxicity and Its Remediation Using Eco-Friendly Approaches. Toxics 2023, 11, 147. [Google Scholar] [CrossRef] [PubMed]
- Duncan, A.E.; de Vries, N.; Nyarko, K. Microplastics in the surface sediments from the Beijiang River littoral zone: Composition, abundance, surface textures and interaction with heavy metals. Chemosphere 2017, 171, 248–258. [Google Scholar]
- Kang, M.; Tian, Y.; Zhang, H.; Lan, Q. Distribution, ecological risk assessment, and source identification of heavy metals in river sediments from Hai River and its tributaries, Tianjin, China. Water Air Soil Pollut. 2020, 231, 38. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E. Bioaccumulation of non-essential hazardous heavy metals and metalloids in freshwater fish. Risk to human health. Environ. Chem. Lett. 2018, 16, 903–917. [Google Scholar] [CrossRef]
- Dukes, A.D.; Eklund, R.T.; Morgan, Z.D.; Layland, R.C. Heavy metal concentration in the water and sediment of the lake greenwood Watershed. Water Air Soil Pollut. 2020, 231, 11. [Google Scholar] [CrossRef]
- Wen, B.; Jin, S.-R.; Chen, Z.-Z.; Gao, J.-Z.; Liu, Y.-N.; Liu, J.-H.; Feng, X.-S. Single and combined effects of microplastics and cadmium on the cadmium accumulation, antioxidant defence and innate immunity of the discus fish (Symphysodon aequifasciatus). Environ. Pollut. 2018, 243, 462–471. [Google Scholar] [CrossRef]
- Naqash, N.; Prakash, S.; Kapoor, D.; Singh, R. Interaction of freshwater microplastics with biota and heavy metals: A review. Environ. Chem. Lett. 2020, 18, 1813–1824. [Google Scholar] [CrossRef]
- Brraich, O.S.; Jangu, S. Evaluation of water quality pollution indices for heavy metal contamination monitoring in the water of Harike Wetland (Ramsar Site), India. Int. J. Sci. Res. Publ. 2015, 5, 1–6. [Google Scholar]
- Kaur, J.; Walia, H.; Mabwoga, S.O.; Arora, S. Water quality monitoring of an international wetland at Harike, Punjab and its impact on biological systems. Appl. Water Sci. 2017, 7, 1107–1115. [Google Scholar] [CrossRef][Green Version]
- Nisari, A.; Sujatha, C. Assessment of trace metal contamination in the Kol wetland, a Ramsar site, Southwest coast of India. Reg. Stud. Mar. Sci. 2021, 47, 101953. [Google Scholar] [CrossRef]
- Sharma, S.; Nagpal, A.K.; Kaur, I. Appraisal of heavy metal contents in groundwater and associated health hazards posed to human population of Ropar wetland, Punjab, India and its environs. Chemosphere 2019, 227, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Sfakianakis, D.; Renieri, E.; Kentouri, M.; Tsatsakis, A. Effect of heavy metals on fish larvae deformities: A review. Environ. Res. 2015, 137, 246–255. [Google Scholar] [CrossRef]
- Wang, H.; Wu, Z.; Chen, B.; He, M.; Hu, B. Chip-based array magnetic solid phase microextraction on-line coupled with inductively coupled plasma mass spectrometry for the determination of trace heavy metals in cells. Anal. 2015, 140, 5619–5626. [Google Scholar] [CrossRef]
- Known and Probable Human Carcinogens. Available online: https://www.cancer.org/cancer/risk-prevention/understanding-cancer-risk/known-and-probable-human-carcinogens.html (accessed on 21 August 2023).
- Singh, V.; Singh, N.; Verma, M.; Kamal, R.; Tiwari, R.; Chivate, M.S.; Rai, S.N.; Kumar, A.; Singh, A.; Singh, M.P.; et al. Hexavalent-Chromium-Induced Oxidative Stress and the Protective Role of Antioxidants against Cellular Toxicity. Antioxidants 2022, 11, 2375. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Singh, J.; Mishra, V. Sorption kinetics of an eco-friendly and sustainable Cr (VI) ion scavenger in a batch reactor. J. Environ. Chem. Eng. 2021, 9, 105125. [Google Scholar] [CrossRef]
- Kumar, G.; Singh, K.K. Mapping and monitoring the selected wetlands of Punjab, India, using geospatial techniques. J. Indian Soc. Remote Sens. 2020, 48, 615–625. [Google Scholar] [CrossRef]
- Chopra, R.; Verma, V.K.; Sharma, P.K. Mapping, monitoring and conservation of Harike wetland ecosystem, Punjab, India, through remote sensing. Int. J. Remote. Sens. 2001, 22, 89–98. [Google Scholar] [CrossRef]
- Ağca, N.; Karanlık, S.; Ödemiş, B. Assessment of ammonium, nitrate, phosphate, and heavy metal pollution in groundwater from Amik Plain, southern Turkey. Environ. Monit. Assess. 2014, 186, 5921–5934. [Google Scholar] [CrossRef] [PubMed]
- Setia, R.; Dhaliwal, S.S.; Kumar, V.; Singh, R.; Kukal, S.S.; Pateriya, B. Impact assessment of metal contamination in surface water of Sutlej River (India) on human health risks. Environ. Pollut. 2020, 265, 114907. [Google Scholar] [CrossRef]
- Dung, T.T.T.; Cappuyns, V.; Swennen, R.; Phung, N.K. From geochemical background determination to pollution assessment of heavy metals in sediments and soils. Rev. Environ. Sci. Biotechnol. 2013, 12, 335–353. [Google Scholar] [CrossRef]
- Zhaoyong, Z.; Xiaodong, Y.; Shengtian, Y. Heavy metal pollution assessment, source identification, and health risk evaluation in Aibi Lake of northwest China. Environ. Monit. Assess. 2018, 190, 69. [Google Scholar] [CrossRef]
- Singh, U.K.; Kumar, B. Pathways of heavy metals contamination and associated human health risk in Ajay River basin, India. Chemosphere 2017, 174, 183–199. [Google Scholar] [CrossRef]
- Giri, S.; Singh, A.K. Human health risk assessment via drinking water pathway due to metal contamination in the groundwater of Subarnarekha River Basin, India. Environ. Monit. Assess. 2015, 187, 63. [Google Scholar] [CrossRef]
- Mitra, S.; Sarkar, S.K.; Raja, P.; Biswas, J.K.; Murugan, K. Dissolved trace elements in Hooghly (Ganges) River Estuary, India: Risk assessment and implications for management. Mar. Pollut. Bull. 2018, 133, 402–414. [Google Scholar] [CrossRef]
- Prasad, S.; Saluja, R.; Joshi, V.; Garg, J.K. Heavy metal pollution in surface water of the Upper Ganga River, India: Human health risk assessment. Environ. Monit. Assess. 2020, 192, 742. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Zhao, D.Y.; Jia, H.Y.; Zhang, Y.; Zhang, X.X.; Cheng, S.P. Preliminary Risk Assessment of Trace Metal Pollution in Surface Water from Yangtze River in Nanjing Section, China. Bull. Environ. Contam. Toxicol. 2009, 82, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Abuduwaili, J.; Zhang, Z.Y.; Jiang, F.Q. Assessment of the Distribution, Sources and Potential Ecological Risk of Heavy Metals in the Dry Surface Sediment of Aibi Lake in Northwest China. PLoS ONE 2015, 10, e0120001. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Sharma, A.; Kumar, R.; Bhardwaj, R.; Kumar Thukral, A.; Rodrigo-Comino, J. Assessment of heavy-metal pollution in three different Indian water bodies by combination of multivariate analysis and water pollution indices. Hum. Ecol. Risk Assess. Int. J. 2018, 26, 1–16. [Google Scholar] [CrossRef]
- Minhas, P.S.; Saha, J.K.; Dotaniya, M.; Sarkar, A.; Saha, M. Wastewater irrigation in India: Current status, impacts and response options. Sci. Total Environ. 2022, 808, 152001. [Google Scholar] [CrossRef]
- Verma, V.K.; Sur, K.; Prakash, C. Ecotope-Based Diversity Monitoring of Wetland Using Infused Machine Learning Technique. Water Conserv. Sci. Eng. 2023, 8, 38. [Google Scholar] [CrossRef]
- Walia, G.; Handa, D.; Kaur, H.; Kalotra, R. Erythrocyte abnormalities in a freshwater fish, Labeo rohita exposed to tan-nery industry effluent. Int. J. Pharm. Biol. Sci. 2013, 3, 287–295. [Google Scholar]
- Grigg, N.; Ahmad, M.-D.; Imran, S.; Podger, G.; Kirby, M.; Colloff, M. Water Quality in the Ravi and Sutlej Rivers, Pakistan: A System View; CSIRO: Canberra, Australia. [CrossRef]
- Ali, S.A.; Baloch, M.; Ahmed, N.; Ali, A.A.; Iqbal, A. The outbreak of Coronavirus Disease 2019 (COVID-19)—An emerging global health threat. J. Infect. Public Heal. 2020, 13, 644–646. [Google Scholar] [CrossRef]
- Dutta, V.; Dubey, D.; Kumar, S. Cleaning the River Ganga: Impact of lockdown on water quality and future implications on river rejuvenation strategies. Sci. Total. Environ. 2020, 743, 140756. [Google Scholar] [CrossRef]
- Khan, R.; Saxena, A.; Shukla, S.; Sekar, S.; Goel, P. Effect of COVID-19 lockdown on the water quality index of River Gomti, India, with potential hazard of faecal-oral transmission. Environ. Sci. Pollut. Res. 2021, 28, 33021–33029. [Google Scholar] [CrossRef] [PubMed]
- Gagan, M.; D.P., U.; K., C.S.; Sunil, K.; Gaurav, P.; Avinash, K.; Anjali, N.; Pawan, K. Impact of Pandemic COVID19 on Air and Water Quality in India: A Systematic Review. Int. J. Eng. Adv. Technol. 2022, 11, 149–167. [Google Scholar] [CrossRef]





| Heavy Metal | Sampling Season | |||
|---|---|---|---|---|
| Winter | Summer | Monsoon | Post-Monsoon | |
| Station I | ||||
| As | 3.65 | 1.22 | 2.03 | 2.67 |
| Cd | 7.84 | 3.76 | 2.42 | 6.47 |
| Cr | 11.09 | 6.64 | 8.98 | 7.38 |
| Pb | 286.33 | 3.76 | 15.44 | 6.75 |
| Zn | 257.54 | 186.95 | 197.53 | 234.75 |
| Station II | ||||
| As | 5.04 | 1.21 | 1.68 | 0.71 |
| Cd | 10.65 | 1.67 | 6.89 | 2.31 |
| Cr | 4.76 | 3.49 | 3.47 | 3.76 |
| Pb | 280.53 | 3.69 | 5.8 | 3.61 |
| Zn | 221.16 | 209.45 | 186.83 | 190.65 |
| Station III | ||||
| As | 5.74 | 20.13 | 3.91 | 0.25 |
| Cd | 6.32 | 1.06 | 6.92 | 1.9 |
| Cr | 5.99 | 1.86 | 2.56 | 1.79 |
| Pb | 189.36 | 6.23 | 18.44 | 5.33 |
| Zn | 132.86 | 157.84 | 182.72 | 181.67 |
| Station IV | ||||
| As | 3.08 | 1.72 | 2.19 | 1.63 |
| Cd | 3.47 | 0.73 | 1.6 | 1.87 |
| Cr | 12.48 | 9.19 | 1.28 | 5.73 |
| Pb | 357.79 | 9.75 | 18.64 | 22.19 |
| Zn | 469.33 | 326.85 | 198.92 | 194.91 |
| Parameters | As | Cd | Cr | Pb | Zn |
| Winter | |||||
| Mean (µg/L) | 4.37 | 7.07 | 8.58 | 278.5 | 270.22 |
| Minimum (µg/L) | 2.98 | 3.35 | 4.58 | 179.89 | 128.96 |
| Maximum (µg/L) | 5.88 | 11.05 | 13.1 | 374.98 | 484.5 |
| SD | 1.11 | 2.71 | 3.43 | 63.54 | 129.28 |
| CV | 1.24 | 7.39 | 11.8 | 4038.41 | 16,715.69 |
| Skewness | 0.06 | 0.009 | 0.05 | −0.21 | 0.79 |
| Kurtosis | −1.84 | −1.06 | −2.12 | −0.75 | −0.71 |
| Summer | |||||
| Mean (µg/L) | 6.07 | 1.80 | 5.29 | 5.85 | 220.27 |
| Minimum (µg/L) | 1.18 | .7 | 1.77 | 3.51 | 142.94 |
| Maximum (µg/L) | 20.63 | 3.94 | 9.45 | 10.22 | 343.19 |
| SD | 8.48 | 1.23 | 2.96 | 2.59 | 68.14 |
| CV | 71.99 | 1.52 | 8.77 | 6.71 | 4643.65 |
| Skewness | 1.32 | 1.03 | 0.20 | 0.77 | 0.96 |
| Kurtosis | −0.31 | −0.62 | −1.64 | −1.03 | −0.52 |
| Monsoon | |||||
| Mean (µg/L) | 2.45 | 4.45 | 4.07 | 14.58 | 191.5 |
| Minimum (µg/L) | 1.6 | 1.58 | 1.22 | 5.51 | 170.72 |
| Maximum (µg/L) | 4.05 | 7.22 | 9.41 | 19.57 | 208.87 |
| SD | 0.90 | 2.58 | 3.07 | 5.49 | 12.91 |
| CV | 0.81 | 6.65 | 9.45 | 30.19 | 166.71 |
| Skewness | 1.15 | −0.03 | 1.06 | −1.06 | −0.15 |
| Kurtosis | −00.38 | −2.33 | −0.51 | −0.62 | −1.14 |
| Post−monsoon | |||||
| Mean (µg/L) | 1.31 | 3.13 | 4.66 | 9.47 | 200.49 |
| Minimum (µg/L) | 0.24 | 1.82 | 1.71 | 3.46 | 165.88 |
| Maximum (µg/L) | 2.8 | 6.72 | 7.74 | 23.2 | 246.49 |
| SD | 0.96 | 2.02 | 2.19 | 7.77 | 24.07 |
| CV | 0.94 | 4.08 | 4.83 | 60.42 | 579.42 |
| Skewness | 0.40 | 1.3 | −0.08 | 1.24 | 0.55 |
| Kurtosis | −1.39 | −0.319 | −1.43 | −0.35 | −0.27 |
| Element | BIS (mg/L) | WHO (mg/L) | US-EPA (mg/L) | FAO (mg/L) |
|---|---|---|---|---|
| Arsenic (As) | 0.05 | 0.01 | 0.34 | 0.10 |
| Cadmium (Cd) | 0.003 | 0.003 | 0.002 | 0.01 |
| Chromium (Cr) | 2 | 0.05 | 0.016 | 0.10 |
| Lead (Pb) | 0.1 | 0.01 | 0.065 | 5.0 |
| Zinc (Zn) | 15 | 3 | 0.12 | 2.0 |
| Seasons | Pollution Index | As | Cd | Cr | Pb | Zn |
|---|---|---|---|---|---|---|
| Winter | pi | 0.087 | 2.356 | 0.004 | 2.785 | 0.018 |
| Pi | 0.072 | 2.130 | 0.003 | 2.267 | 0.018 | |
| Summer | pi | 0.121 | 0.601 | 0.002 | 0.058 | 0.014 |
| Pi | 0.210 | 0.695 | 0.002 | 0.056 | 0.013 | |
| Monsoon | pi | 0.049 | 1.485 | 0.002 | 0.145 | 0.012 |
| Pi | 0.046 | 1.371 | 0.002 | 0.118 | 0.009 | |
| Post-monsoon | pi | 0.026 | 1.045 | 0.002 | 0.094 | 0.013 |
| Pi | 0.029 | 1.198 | 0.002 | 0.120 | 0.010 |
| Heavy Metal | CDIingestion | CDIdermal | HQingestion | HQdermal | ||||
|---|---|---|---|---|---|---|---|---|
| Winter Season | ||||||||
| Adult | Child | Adult | Child | Adult | Child | Adult | Child | |
| As | 1.56339E−07 | 2.2763E−07 | 4.49132E−12 | 2.09595E−11 | 5.21131E−07 | 7.58767E−07 | 3.65148E−10 | 1.70402E−09 |
| Cd | 2.525E−07 | 3.6764E−07 | 7.25382E−12 | 3.38512E−11 | 0.000000505 | 7.3528E−07 | 1.45076E−09 | 6.77023E−09 |
| Cr | 3.06429E−07 | 4.4616E−07 | 8.80308E−12 | 4.11E−11 | 1.02143E−07 | 1.4872E−07 | 5.86872E−10 | 2.73874E−09 |
| Pb | 9.94652E−06 | 1.44821E−05 | 2.85744E−10 | 1.33347E−09 | 7.10466E−06 | 1.03444E−05 | 6.80342E−10 | 3.17493E−09 |
| Zn | 9.6508E−06 | 1.40516E−05 | 2.77248E−10 | 1.29383E−09 | 3.21693E−08 | 4.68386E−08 | 4.6208E−12 | 2.15638E−11 |
| Summer Season | ||||||||
| Adult | Child | Adult | Child | Adult | Child | Adult | Child | |
| As | 2.16786E−07 | 3.1564E−07 | 6.22782E−12 | 2.90632E−11 | 7.22619E−07 | 1.05213E−06 | 5.06327E−10 | 2.36286E−09 |
| Cd | 6.44643E−08 | 9.386E−08 | 1.85193E−12 | 8.64234E−12 | 1.28929E−07 | 1.8772E−07 | 3.70386E−10 | 1.72847E−09 |
| Cr | 1.89107E−07 | 2.7534E−07 | 5.43267E−12 | 2.54E−11 | 6.30357E−08 | 9.178E−08 | 3.62178E−10 | 1.69016E−09 |
| Pb | 2.09196E−07 | 3.0459E−07 | 6..0098E−12 | 2.80457E−11 | 1.49426E−07 | 2.17564E−07 | 1.4309E−11 | 6.67755E−11 |
| Zn | 7.86688E−06 | 1.14542E−05 | 2.26E−10 | 1.05466E−09 | 2.62229E−08 | 3.81806E−08 | 3.76666E−12 | 1.75777E−11 |
| Monsoon Season | ||||||||
| Adult | Child | Adult | Child | Adult | Child | Adult | Child | |
| As | 8.75893E−08 | 1.2753E−07 | 2.51627E−12 | 1.17426E−11 | 2.91964E−07 | 4.251E−07 | 2.04574E−10 | 9.5468E−10 |
| Cd | 1.59196E−07 | 2.3179E−07 | 4.5734E−12 | 2.13425E−11 | 3.18393E−07 | 4.6358E−07 | 9.14679E−10 | 4.2685E−09 |
| Cr | 1.45446E−07 | 2.1177E−07 | 4.17839E−12 | 1.95E−11 | 4.84821E−08 | 7.059E−08 | 2.78559E−10 | 1.29994E−09 |
| Pb | 5.20714E−07 | 7.5816E−07 | 1.49591E−11 | 6.9809E−11 | 3.71939E−07 | 5.41543E−07 | 3.56169E−11 | 1.66212E−10 |
| Zn | 6.83929E−06 | 0.000009958 | 1.96479E−10 | 9.16902E−10 | 2.27976E−08 | 3.31933E−08 | 3.27465E−12 | 1.52817E−11 |
| Post-monsoon Season | ||||||||
| Adult | Child | Adult | Child | Adult | Child | Adult | Child | |
| As | 4.69643E−08 | 6.838E−08 | 1.34919E−12 | 6.29622E−12 | 1.56548E−07 | 2.27933E−07 | 1.0969E−10 | 5.11888E−10 |
| Cd | 1.12054E−07 | 1.6315E−07 | 3.21908E−12 | 1.50224E−11 | 2.24107E−07 | 3.263E−07 | 6.43815E−10 | 3.00447E−09 |
| Cr | 1.66607E−07 | 2.4258E−07 | 4.78629E−12 | 2.23E−11 | 5.55357E−08 | 8.086E−08 | 3.19086E−10 | 1.48907E−09 |
| Pb | 3.38214E−07 | 4.9244E−07 | 9.71622E−12 | 4.53424E−11 | 2.41582E−07 | 3.51743E−07 | 2.31339E−11 | 1.07958E−10 |
| Zn | 7.86688E−06 | 1.04257E−05 | 2.05708E−10 | 9.5997E−10 | 2.38685E−08 | 3.47525E−08 | 3.42846E−12 | 1.59995E−11 |
| Heavy Metal | (Brraich and Jangu 2015) [16] | (Kaur et al., 2017) [17] | (Kumar et al., 2018) [37] | Present Study | |||
|---|---|---|---|---|---|---|---|
| Winter | Summer | Monsoon | Post-Monsoon | ||||
| As | - | - | - | 0.0043 | 0.0060 | 0.0024 | 0.0013 |
| Cr | 0.12 | 0.121 | 0.09 | 0.0085 | 0.0052 | 0.0040 | 0.0046 |
| Cd | 0.01 | - | 0.02 | 0.0070 | 0.0018 | 0.0044 | 0.0031 |
| Pb | 0.53 | 0.704 | 0.72 | 0.2785 | 0.0058 | 0.0145 | 0.0094 |
| Zn | 0.69 | 2.589 | 0.55 | 0.2702 | 0.2202 | 0.1915 | 0.2004 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naqash, N.; Jamal, M.T.; Singh, R. Heavy Metal Contamination in Surface Water of Harike Wetland, India: Source and Health Risk Assessment. Water 2023, 15, 3287. https://doi.org/10.3390/w15183287
Naqash N, Jamal MT, Singh R. Heavy Metal Contamination in Surface Water of Harike Wetland, India: Source and Health Risk Assessment. Water. 2023; 15(18):3287. https://doi.org/10.3390/w15183287
Chicago/Turabian StyleNaqash, Nafiaah, Mamdoh T. Jamal, and Rahul Singh. 2023. "Heavy Metal Contamination in Surface Water of Harike Wetland, India: Source and Health Risk Assessment" Water 15, no. 18: 3287. https://doi.org/10.3390/w15183287
APA StyleNaqash, N., Jamal, M. T., & Singh, R. (2023). Heavy Metal Contamination in Surface Water of Harike Wetland, India: Source and Health Risk Assessment. Water, 15(18), 3287. https://doi.org/10.3390/w15183287








