Effect of Hydrochar Modification on the Adsorption of Methylene Blue from Aqueous Solution: An Experimental Study Followed by Intelligent Modeling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Pristine and Modified Adsorbents
2.3. pHzpc for KOH-Modified Hydrochar
2.4. Adsorption Experiments
2.5. Desorption and Reusability Experiments
2.6. Adsorption Isotherm
2.7. Characterization of Adsorbent
2.8. Neural Network Modeling
3. Results and Discussion
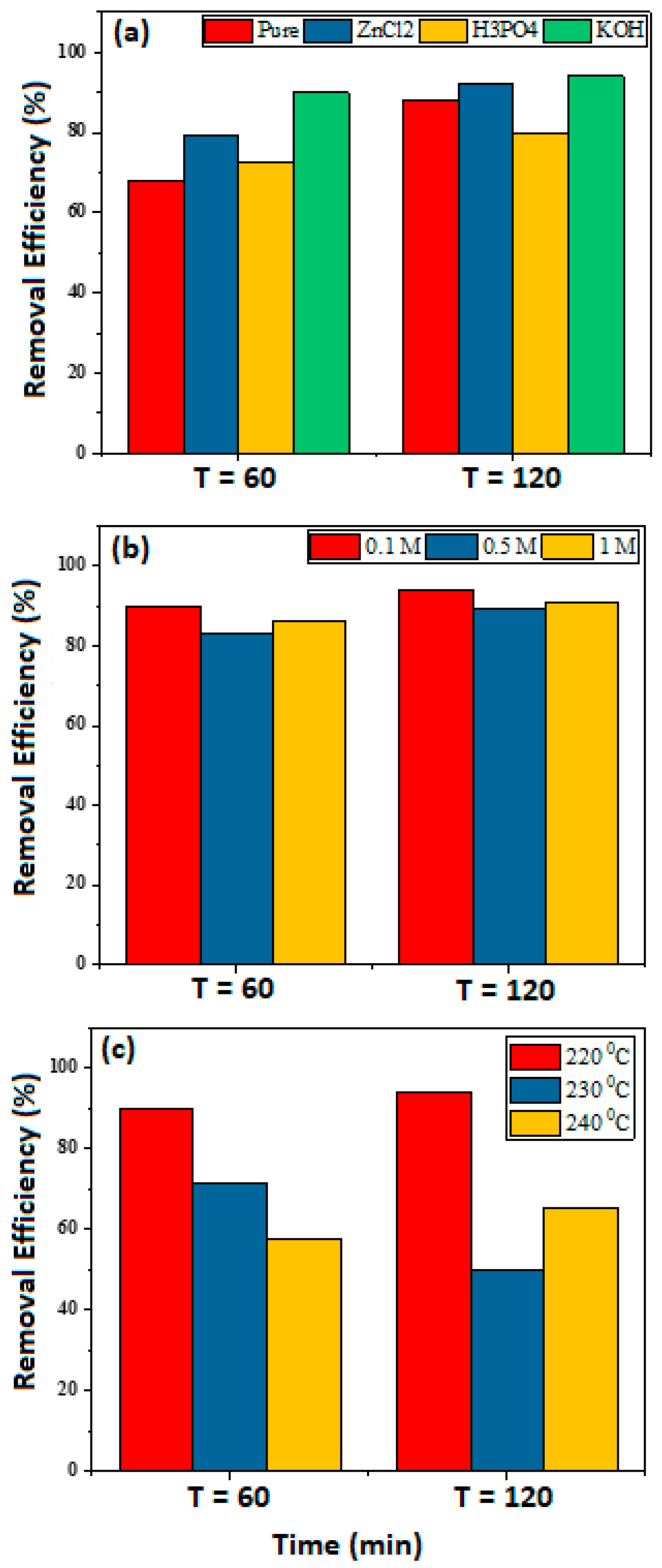
3.1. MB Removal Efficiency with Different Adsorbents
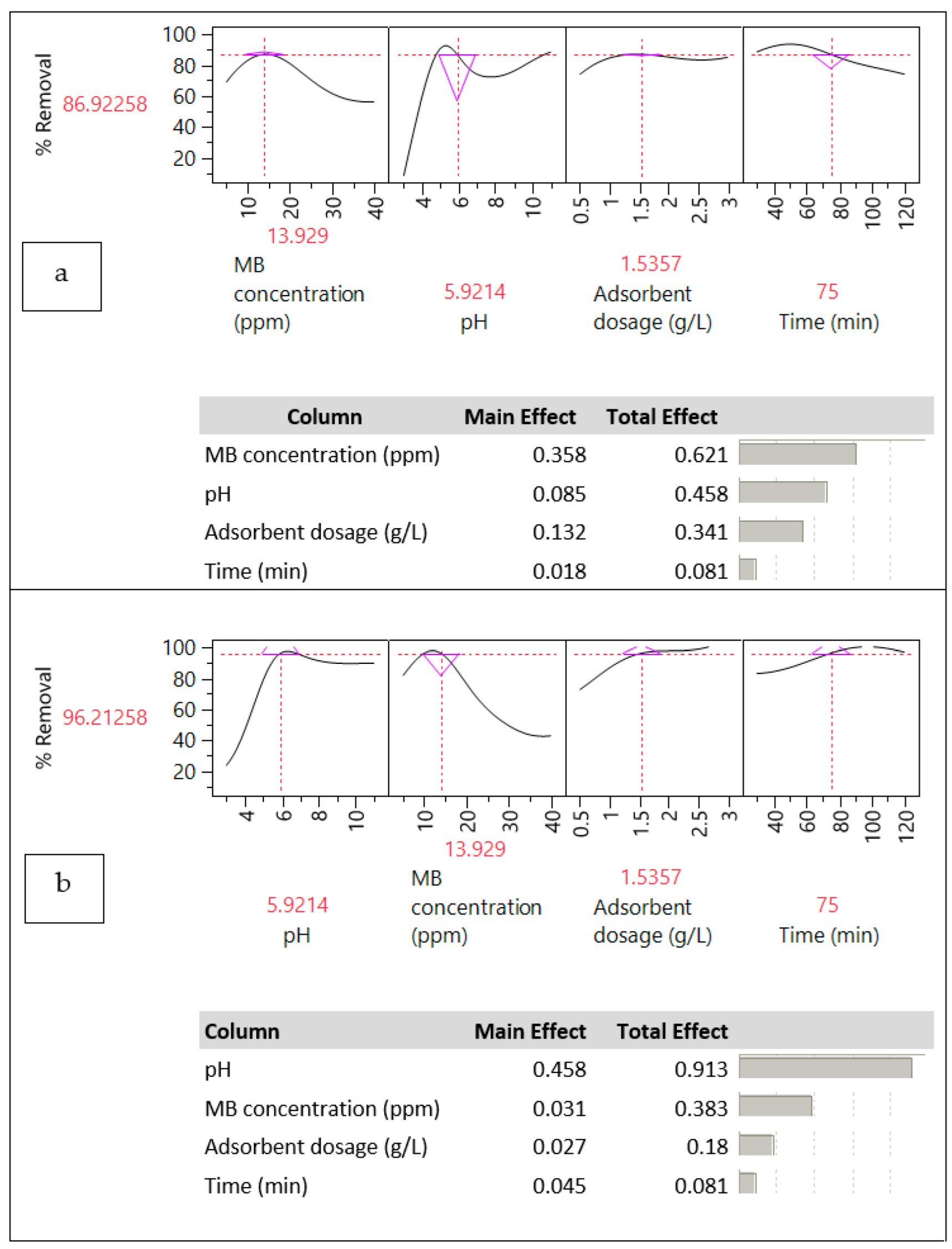
3.2. Effect of Interfering Parameters on MB Removal Efficiency
3.2.1. KOH Modification and Preparing Temperature
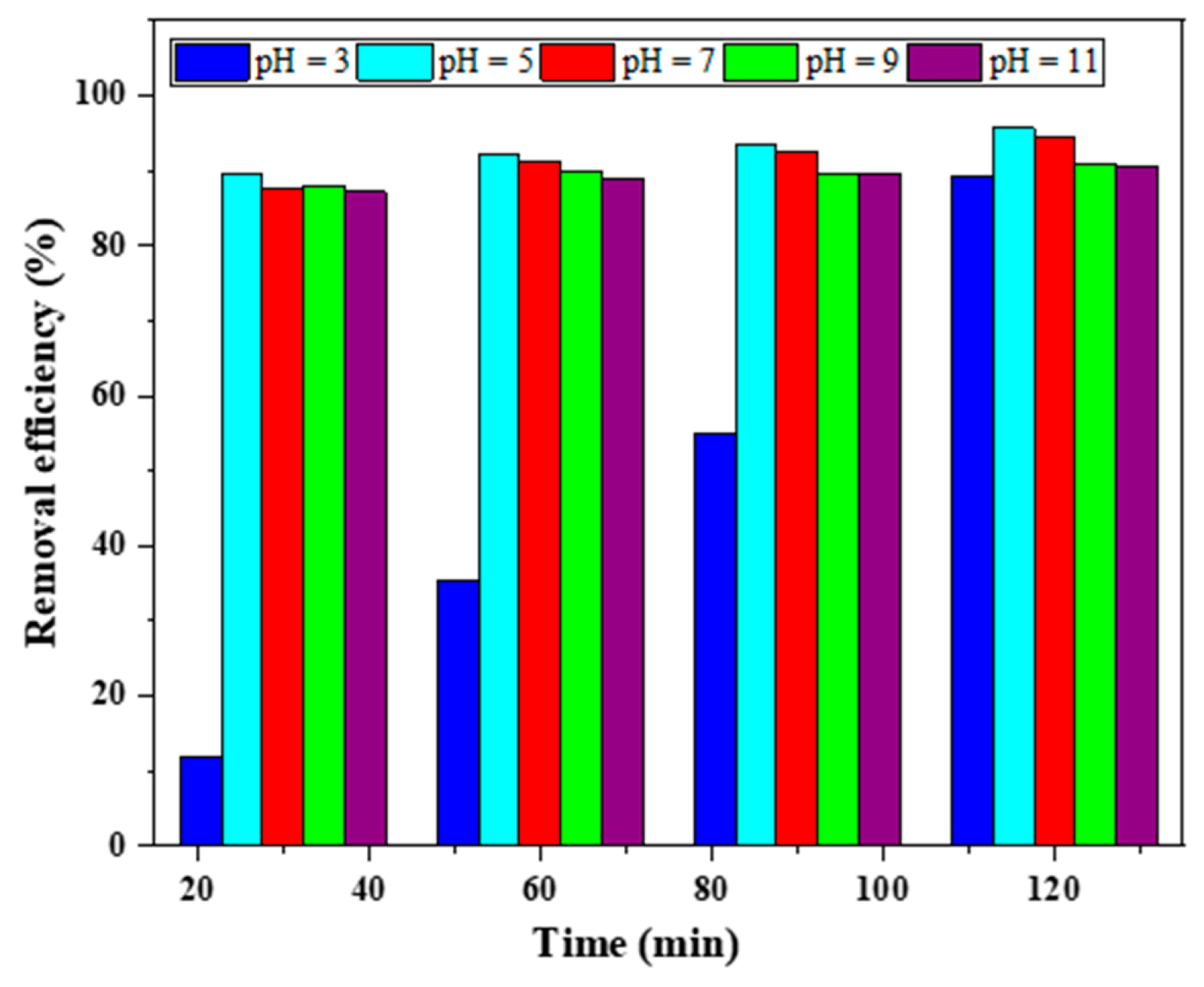
3.2.2. Effect of pH and pHzpc
3.2.3. Effect of Hydrochar Dosage
3.2.4. Effect of MB Concentration
3.2.5. Desorption and Reusability Experiments
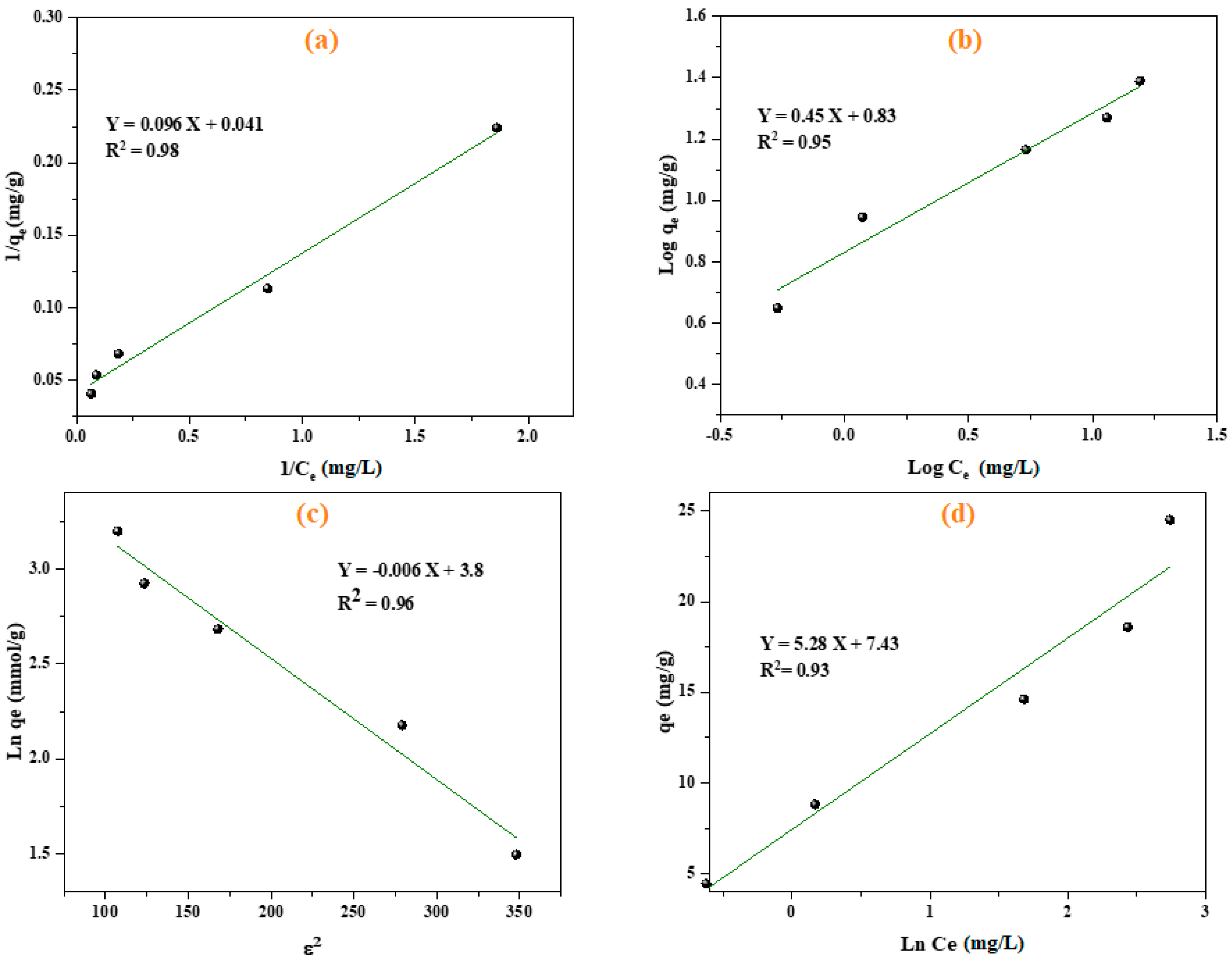
3.3. Adsorption Isotherms
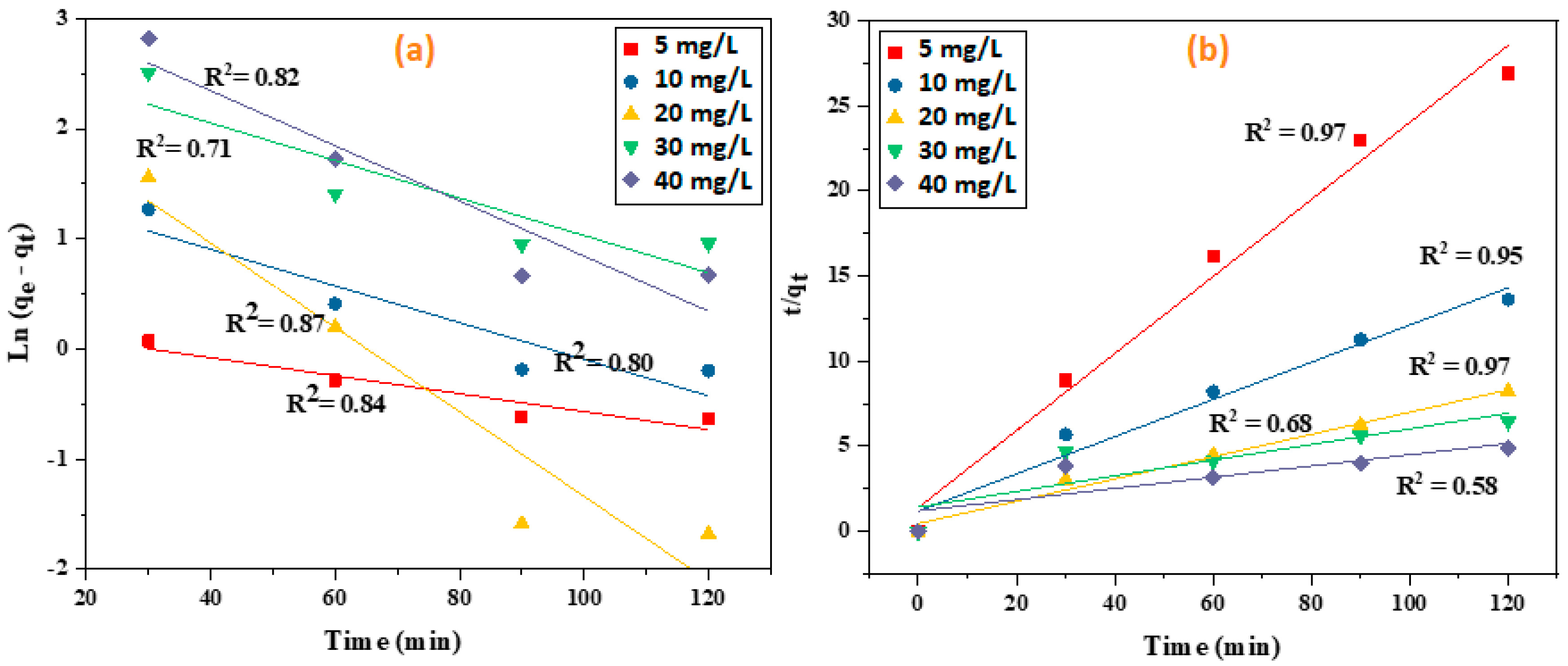
3.4. Kinetic Experiments for the KOH-Modified Hydrochar
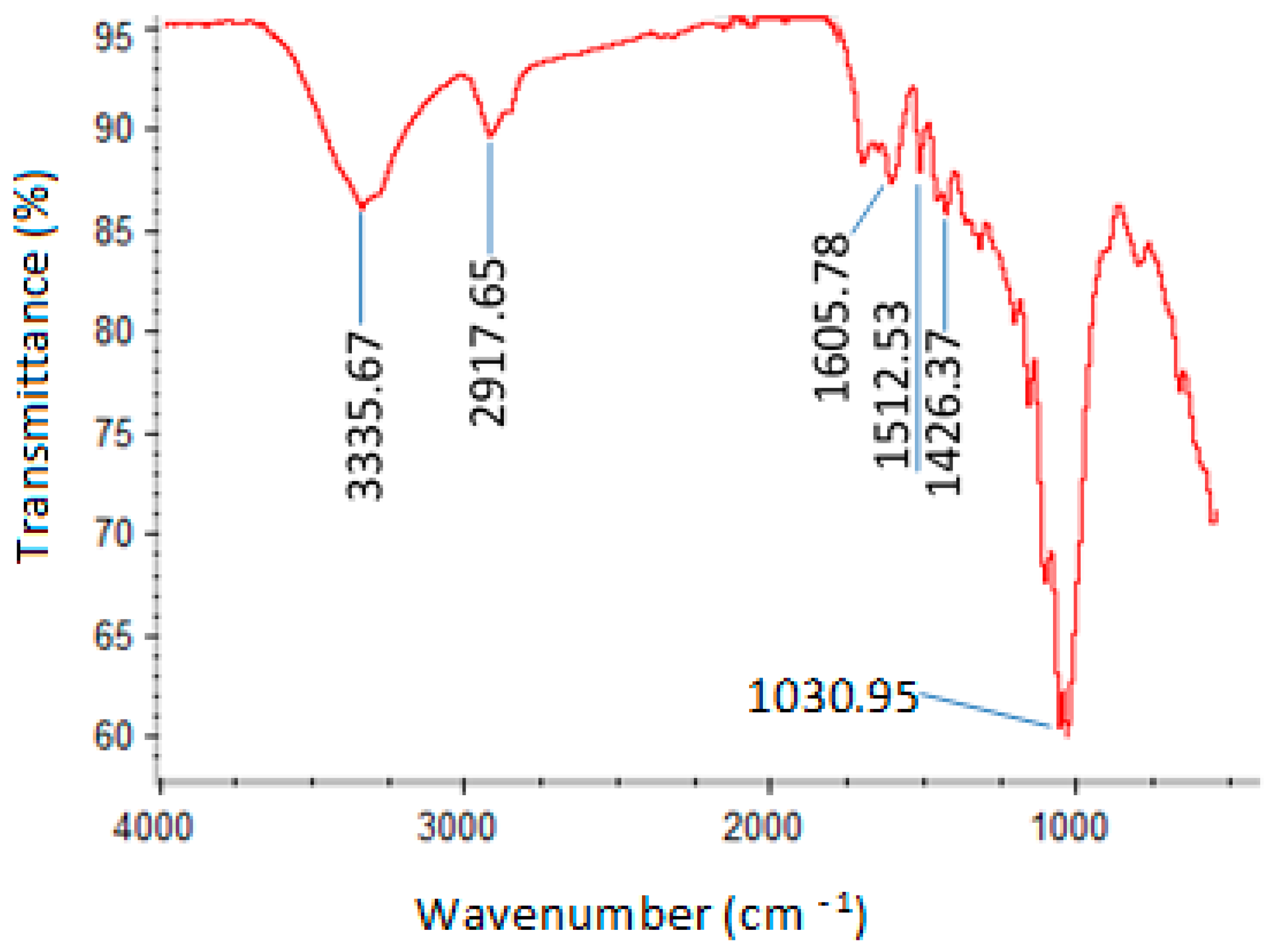
3.5. Characterization of the 0.1 M KOH-Modified Hydrochar
3.6. Neural Network Modeling (NN)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Park, J.-H.; Ok, Y.S.; Kim, S.-H.; Cho, J.-S.; Heo, J.-S.; Delaune, R.D.; Seo, D.-C. Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere 2016, 142, 77–83. [Google Scholar] [CrossRef]
- Jung, K.-W.; Ahn, K.-H. Fabrication of porosity-enhanced MgO/biochar for removal of phosphate from aqueous solution: Application of a novel combined electrochemical modification method. Bioresour. Technol. 2016, 200, 1029–1032. [Google Scholar] [CrossRef] [PubMed]
- Farhan Hanafi, M.; Sapawe, N. A review on the water problem associate with organic pollutants derived from phenol, methyl orange, and remazol brilliant blue dyes. Mater. Today Proc. 2020, 31, A141–A150. [Google Scholar] [CrossRef]
- Zhou, F.; Li, K.; Hang, F.; Zhang, Z.; Chen, P.; Wei, L.; Xie, C. Efficient removal of methylene blue by activated hydrochar prepared by hydrothermal carbonization and NaOH activation of sugarcane bagasse and phosphoric acid. RSC Adv. 2022, 12, 1885–1896. [Google Scholar] [CrossRef]
- Tkaczyk, A.; Mitrowska, K.; Posyniak, A. Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems: A review. Sci. Total Environ. 2020, 717, 137222. [Google Scholar] [CrossRef] [PubMed]
- Santoso, E.; Ediati, R.; Kusumawati, Y.; Bahruji, H.; Sulistiono, D.O.; Prasetyoko, D. Review on recent advances of carbon based adsorbent for methylene blue removal from waste water. Mater. Today Chem. 2020, 16, 100233. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Lichtfouse, E.; Fourmentin, M.; Ribeiro, A.R.L.; Noutsopoulos, C.; Mapelli, F.; Fenyvesi, É.; Vieira, M.G.A.; Picos-Corrales, L.A.; Moreno-Piraján, J.C.; et al. Removal of emerging contaminants from wastewater using advanced treatments. A review. Environ. Chem. Lett. 2022, 20, 1333–1375. [Google Scholar] [CrossRef]
- Söğüta, E.G.; Ergana, E.; Kılıçb, N.Ç.; Dönmezb, H.; Akbaşb, E. Methylene blue adsorption from aqueous solution by functionalized perlites: An experimental and computational chemistry study. Desalin. Water Treat. 2021, 217, 391–410. [Google Scholar] [CrossRef]
- Deng, J.; Liu, Y.; Liu, S.; Zeng, G.; Tan, X.; Huang, B.; Tang, X.; Wang, S.; Yan, Z. Competitive adsorption of Pb (II), Cd (II) and Cu (II) onto chitosan-pyromellitic dianhydride modified biochar. J. Colloid Interface Sci. 2017, 506, 355–364. [Google Scholar] [CrossRef]
- Tan, X.; Liu, Y.; Zeng, G.; Wang, X.; Hu, X.; Gu, Y.; Yang, Z. Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 2015, 125, 70–85. [Google Scholar] [CrossRef]
- Shan, D.; Deng, S.; Zhao, T.; Wang, B.; Wang, Y.; Huang, J.; Yu, G.; Winglee, J.; Wiesner, M.R. Preparation of ultrafine magnetic biochar and activated carbon for pharmaceutical adsorption and subsequent degradation by ball milling. J. Hazard. Mater. 2016, 305, 156–163. [Google Scholar] [CrossRef]
- Maniscalco, M.P.; Volpe, M.; Messineo, A. Hydrothermal carbonization as a valuable tool for energy and environmental applications: A review. Energies 2020, 13, 4098. [Google Scholar] [CrossRef]
- Pauline, A.L.; Joseph, K. Hydrothermal carbonization of organic wastes to carbonaceous solid fuel—A review of mechanisms and process parameters. Fuel 2020, 279, 118472. [Google Scholar] [CrossRef]
- Wang, T.; Zhai, Y.; Zhu, Y.; Li, C.; Zeng, G. A review of the hydrothermal carbonization of biomass waste for hydrochar formation: Process conditions, fundamentals, and physicochemical properties. Renew. Sustain. Energy Rev. 2018, 90, 223–247. [Google Scholar] [CrossRef]
- Juela, D.M. Promising adsorptive materials derived from agricultural and industrial wastes for antibiotic removal: A comprehensive review. Sep. Purif. Technol. 2022, 284, 120286. [Google Scholar]
- Liu, Z.; Quek, A.; Balasubramanian, R. Preparation and characterization of fuel pellets from woody biomass, agro-residues and their corresponding hydrochars. Appl. Energy 2014, 113, 1315–1322. [Google Scholar] [CrossRef]
- Qian, W.-C.; Luo, X.-P.; Wang, X.; Guo, M.; Li, B. Removal of methylene blue from aqueous solution by modified bamboo hydrochar. Ecotoxicol. Environ. Saf. 2018, 157, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wan, Y.; Zheng, Y.; He, F.; Yu, Z.; Huang, J.; Wang, H.; Ok, Y.S.; Jiang, Y.; Gao, B. Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: A critical review. Chem. Eng. J. 2019, 366, 608–621. [Google Scholar] [CrossRef] [PubMed]
- Ghanim, B.; O’dwyer, T.F.; Leahy, J.J.; Willquist, K.; Courtney, R.; Pembroke, J.T.; Murnane, J.G. Application of KOH modified seaweed hydrochar as a biosorbent of Vanadium from aqueous solution: Characterisations, mechanisms and regeneration capacity. J. Environ. Chem. Eng. 2020, 8, 104176. [Google Scholar] [CrossRef]
- Yudha, S.P.; Tekasakul, S.; Phoungthong, K.; Chuenchom, L. Green synthesis of low-cost and eco-friendly adsorbent for dye and pharmaceutical adsorption: Kinetic, isotherm, thermodynamic and regeneration studies. Mater. Res. Express 2019, 6, 125526. [Google Scholar] [CrossRef]
- Kim, U.-J.; Kimura, S.; Wada, M. Highly enhanced adsorption of Congo red onto dialdehyde cellulose-crosslinked cellulose-chitosan foam. Carbohydr. Polym. 2019, 214, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Agbovi, H.K.; Wilson, L.D. Adsorption processes in biopolymer systems: Fundamentals to practical applications. Nat. Polym. Based Green Adsorbents Water Treat. 2021, 1, 1–51. [Google Scholar]
- Akrawi, H.S.Y.; Al-Obaidi, M.A.; Abdulrahman, C.H. Evaluation of Langmuir and Frendlich isotherm equation for Zinc Adsorption in some calcareous soil of Erbil province north of Iraq. IOP Conf. Ser. Earth Environ. Sci. 2021, 761, 012017. [Google Scholar] [CrossRef]
- Dawood, S.; Sen, T.K. Removal of anionic dye Congo red from aqueous solution by raw pine and acid-treated pine cone powder as adsorbent: Equilibrium, thermodynamic, kinetics, mechanism and process design. Water Res. 2012, 46, 1933–1946. [Google Scholar] [CrossRef]
- Muinde, V.M.; Onyari, J.M.; Wamalwa, B.; Wabomba, J.N. Adsorption of malachite green dye from aqueous solutions using mesoporous chitosan–zinc oxide composite material. Environ. Chem. Ecotoxicol. 2020, 2, 115–125. [Google Scholar] [CrossRef]
- Sumanjit; Rani, S.; Mahajan, R. Equilibrium, kinetics and thermodynamic parameters for adsorptive removal of dye Basic Blue 9 by ground nut shells and Eichhornia. Arab. J. Chem. 2016, 9, S1464–S1477. [Google Scholar] [CrossRef]
- Das, A.; Pal, A.; Saha, S.; Maji, S.K. Behaviour of fixed-bed column for the adsorption of malachite green on surfactant-modified alumina. J. Environ. Sci. Health Part A 2009, 44, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Günay, A.; Arslankaya, E.; Tosun, I. Lead removal from aqueous solution by natural and pretreated clinoptilolite: Adsorption equilibrium and kinetics. J. Hazard. Mater. 2007, 146, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Marzban, N.; Libra, J.A.; Hosseini, S.H.; Fischer, M.G.; Rotter, V.S. Experimental evaluation and application of genetic programming to develop predictive correlations for hydrochar higher heating value and yield to optimize the energy content. J. Environ. Chem. Eng. 2022, 10, 108880. [Google Scholar] [CrossRef]
- Tkachenko, V.; Marzban, N.; Vogl, S.; Filonenko, S.; Antonietti, M. Chemical insights into the base-tuned hydrothermal treatment of side stream biomasses. Sustain. Energy Fuels 2023, 7, 769–777. [Google Scholar] [CrossRef]
- Marzban, N.; Moheb, A.; Filonenko, S.; Hosseini, S.H.; Nouri, M.J.; Libra, J.A.; Farru, G. Intelligent modeling and experimental study on methylene blue adsorption by sodium alginate-kaolin beads. Int. J. Biol. Macromol. 2021, 186, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Hunsom, M.; Autthanit, C. Adsorptive purification of crude glycerol by sewage sludge-derived activated carbon prepared by chemical activation with H3PO4, K2CO3 and KOH. Chem. Eng. J. 2013, 229, 334–343. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Wnętrzak, R.; Leahy, J.; Hayes, M.; Kwapiński, W.; Hubicki, Z. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Muinde, V.M.; Onyari, J.M.; Wamalwa, B.; Wabomba, J.; Nthumbi, R.M. Adsorption of Malachite Green from Aqueous Solutions onto Rice Husks: Kinetic and Equilibrium Studies. J. Environ. Prot. 2017, 8, 215–230. [Google Scholar] [CrossRef]
- Zhang, J.; Cai, D.; Zhang, G.; Cai, C.; Zhang, C.; Qiu, G.; Zheng, K.; Wu, Z. Adsorption of methylene blue from aqueous solution onto multiporous palygorskite modified by ion beam bombardment: Effect of contact time, temperature, pH and ionic strength. Appl. Clay Sci. 2013, 83, 137–143. [Google Scholar] [CrossRef]
- Baek, M.H.; Ijagbemi, C.O.; Se-Jin, O.; Kim, D.S. Removal of Malachite Green from aqueous solution using degreased coffee bean. J. Hazard. Mater. 2010, 176, 820–828. [Google Scholar] [CrossRef]
- Kohzadi, S.; Marzban, N.; Libra, J.A.; Bundschuh, M.; Maleki, A. Removal of RhB from water by Fe-modified hydrochar and biochar—An experimental evaluation supported by genetic programming. J. Mol. Liq. 2023, 369, 120971. [Google Scholar] [CrossRef]
- Dada, A.O.; Olalekan, A.P.; Olatunya, A.M.; Dada, O.J. Langmuir, Freundlich, Temkin and Dubinin–Radushkevich isotherms studies of equilibrium sorption of Zn2+ unto phosphoric acid modified rice husk. IOSR J. Appl. Chem. 2012, 3, 38–45. [Google Scholar]
- He, Y.; Lin, X.; Yan, T.; Zhang, X.; Zhou, J.; Chen, Y.; Luo, X. Selective adsorption of uranium from salt lake-simulated solution by phenolic-functionalized hollow sponge-like adsorbent. J. Chem. Technol. Biotechnol. 2019, 94, 455–467. [Google Scholar] [CrossRef]
- He, T.; Zhou, X.; Bao, Z.; Fu, S.; Lin, X. Preparation of hydrochar by one-step hydrothermal carbonization with potassium ferrate-assisted activation for the removal of tetracycline in water. J. Chem. Technol. Biotechnol. 2023, 98, 2039–2050. [Google Scholar] [CrossRef]
- Inyinbor, A.A.; Adekola, F.A.; Olatunji, G.A. Kinetics, isotherms and thermodynamic modeling of liquid phase adsorption of Rhodamine B dye onto Raphia hookerie fruit epicarp. Water Resour. Ind. 2016, 15, 14–27. [Google Scholar] [CrossRef]
- Hu, Q.; Zhang, Z. Application of Dubinin–Radushkevich isotherm model at the solid/solution interface: A theoretical analysis. J. Mol. Liq. 2019, 277, 646–648. [Google Scholar] [CrossRef]
- Rind, I.K.; Memon, N.; Khuhawar, M.Y.; Soomro, W.A.; Lanjwani, M.F. Modeling of cadmium(II) removal in a fixed bed column utilizing hydrochar-derived activated carbon obtained from discarded mango peels. Sci. Rep. 2022, 12, 8001. [Google Scholar] [CrossRef]
- Peng, H.; Cao, J.; Xiong, W.; Yang, Z.; Jia, M.; Sun, S.; Xu, Z.; Zhang, Y.; Cai, H. Two-dimension N-doped nanoporous carbon from KCl thermal exfoliation of Zn-ZIF-L: Efficient adsorption for tetracycline and optimizing of response surface model. J. Hazard. Mater. 2020, 402, 123498. [Google Scholar] [CrossRef] [PubMed]
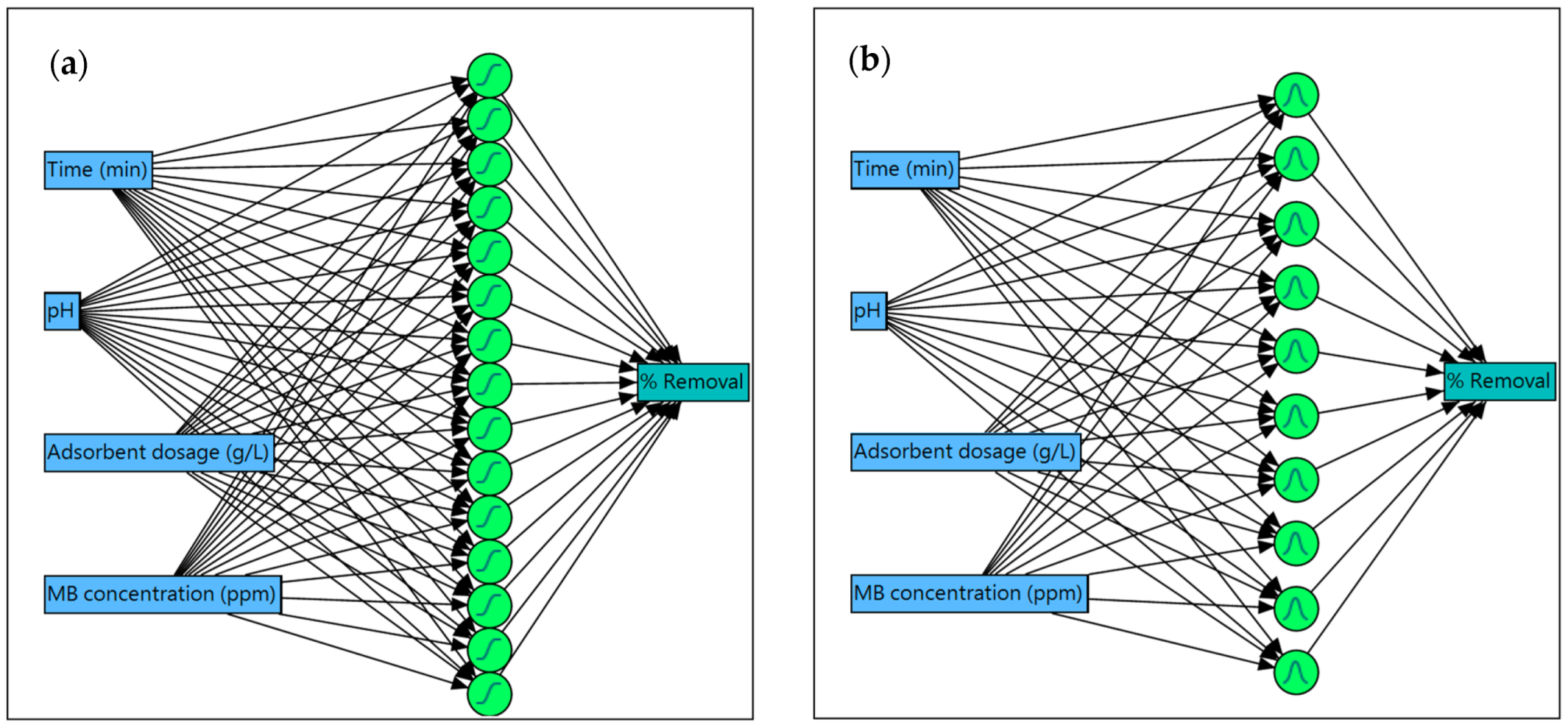




| Group | Sample | TanH | Gaussian | ||||
|---|---|---|---|---|---|---|---|
| Number of Neurons | RMSE | R2 | Number of Neurons | RMSE | R2 | ||
| Training | 36 | 15 | 5.20 | 0.95 | 10 | 5.04 | 0.95 |
| Validation | 18 | 3.15 | 0.96 | 5.40 | 0.88 | ||
| Models | Parameters | Value |
|---|---|---|
| Langmuir | ||
| qmax (mg g−1) | 24.15 | |
| KL (L mg−1) | 0.42 | |
| R2 | 0.98 | |
| RL | 0.05–0.31 | |
| Freundlich model | ||
| KF | 6.77 | |
| n | 2.2 | |
| R2 | 0.96 | |
| D–R model | ||
| Q0 (mmol g−1) | 44.7 | |
| KD-R | 0.006 | |
| R2 | 0.97 | |
| E (KJ mol−1) | 8.83 | |
| Temkin model | ||
| B (J mol−1) | 5.288 | |
| KT (L g−1) | 4.07 | |
| BT | 468.5 | |
| R2 | 0.94 |
| MB Conc. mg L−1 | qe,exp | Pseudo-First Order | Pseudo-Second Order | ||||
|---|---|---|---|---|---|---|---|
| qe,cal mg g−1 | K1 min−1 | R2 | qe,cal mg g−1 | K2 mg g1min−1 | R2 | ||
| 5 | 4.46 | 1.27 | −0.0001 | 0.84 | 4.42 | 0.036 | 0.97 |
| 10 | 8.81 | 4.80 | −0.0004 | 0.80 | 9.14 | 0.009 | 0.95 |
| 20 | 14.62 | 12.06 | −0.0006 | 0.87 | 15.38 | 0.009 | 0.97 |
| 30 | 18.60 | 15.33 | −0.0002 | 0.71 | 21.45 | 0.001 | 0.68 |
| 40 | 24.51 | 28.21 | 0.0004 | 0.82 | 30.30 | 0.0009 | 0.58 |
| Name of Compound | Concentration (g/kg DM) | |
|---|---|---|
| Sugars | Glucose | 0.10 |
| Sucrose | 0.03 | |
| Fructose | 0.01 | |
| Acids | Acetic acid | 1.44 |
| Lactic acid | 3.81 | |
| Formic acid | 1.27 | |
| Aromatics | HMF | 4.70 |
| Furfural | 0.45 | |
| Phenol | - | |
| Catechol | - | |
| Guaiacol | 0.14 | |
| Cresol | - |
| pH | C% | H% | N% | S% | O% | Ash% | H/C | O/C | HHV (MJ/kg) | BET (m2/g) |
|---|---|---|---|---|---|---|---|---|---|---|
| 4.06 | 52.19 | 3.37 | 0.1 | 0.15 | 35.66 | 8.50 | 0.78 | 0.51 | 21.37 | 11.54 |
| Element | Concentration mg/kg DM | Element | Concentration mg/kg DM |
|---|---|---|---|
| Al | 430.16 | Mg | 176.10 |
| Ca | 875.72 | Mn | 7.87 |
| Cd | 0.16 | P | 38.49 |
| Co | 0.14 | Pb | 2.43 |
| Cr | 39.76 | S | 365.11 |
| Cu | 1.13 | Sn | 16.38 |
| Fe | 357.57 | V | 1.39 |
| K | 295.35 | Zn | 11.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kohzadi, S.; Marzban, N.; Godini, K.; Amini, N.; Maleki, A. Effect of Hydrochar Modification on the Adsorption of Methylene Blue from Aqueous Solution: An Experimental Study Followed by Intelligent Modeling. Water 2023, 15, 3220. https://doi.org/10.3390/w15183220
Kohzadi S, Marzban N, Godini K, Amini N, Maleki A. Effect of Hydrochar Modification on the Adsorption of Methylene Blue from Aqueous Solution: An Experimental Study Followed by Intelligent Modeling. Water. 2023; 15(18):3220. https://doi.org/10.3390/w15183220
Chicago/Turabian StyleKohzadi, Shadi, Nader Marzban, Kazem Godini, Nader Amini, and Afshin Maleki. 2023. "Effect of Hydrochar Modification on the Adsorption of Methylene Blue from Aqueous Solution: An Experimental Study Followed by Intelligent Modeling" Water 15, no. 18: 3220. https://doi.org/10.3390/w15183220





