Efficiency of Treated Domestic Wastewater to Irrigate Two Rice Cultivars, PK 386 and Basmati 515, under a Hydroponic Culture System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection and Characterisation of Domestic Wastewater
2.2. Source of Alcaligenes Faecalis MT477813 Culture
2.3. Domestic Wastewater Treatment
2.4. Germination of Rice Seedlings for Hydroponic Cultures
2.5. Transfer of Seedlings to Hydroponic Cultures
2.6. Growth of Seedlings and Analysis in Hydroponic Cultures
2.7. Statistical Analyses
3. Results and Discussion
3.1. Physiochemical Characterisation of Treatments
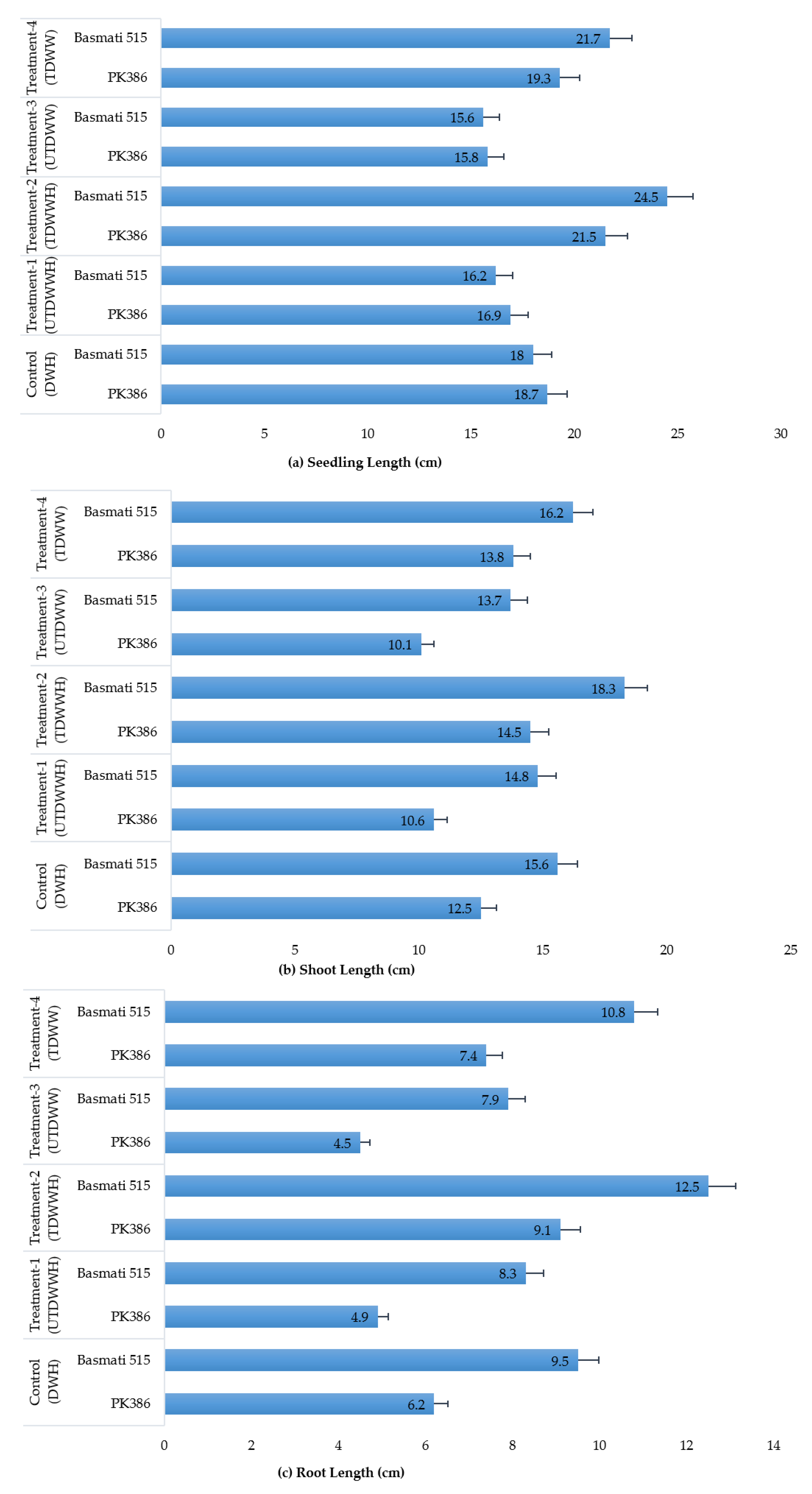
3.2. Growth Parameters of Rice Cultivars in Different Hydroponic Treatments
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khoso, S.; Wagan, F.H.; Tunio, A.H.; Ansari, A.A. An overview on emerging water scarcity in Pakistan, its causes, impacts and remedial measures. J. Appl. Eng. Sci. 2015, 13, 38–40. [Google Scholar] [CrossRef]
- Nabi, G.; Ali, M.; Khan, S.; Kumar, S. The crisis of water shortage and pollution in Pakistan: Risk to public health, biodiversity, and ecosystem. Environ. Sci. Pollut. Res. 2019, 26, 10443–10445. [Google Scholar] [CrossRef] [PubMed]
- Shukla, V. Pakistan’s Alarming Water Crisis: Country to Run out of Clean Water by 2025. 2018. Available online: https://www.valuewalk.com/pakistan-water-crisis-clean-water-2025/ (accessed on 20 July 2023).
- Chung, S.O.; Rodríguez-Díaz, J.A.; Weatherhead, E.K.; Knox, J.W. Climate change impacts on water for irrigating paddy rice in South Korea. Irrig. Drain. 2011, 60, 263–273. [Google Scholar] [CrossRef]
- Zhang, Y.; Shen, Y. Wastewater irrigation: Past, present, and future. Wiley Interdiscip. Rev. Water 2019, 6, 1230–1234. [Google Scholar] [CrossRef]
- Gassama, U.M.; Puteh, A.B.; Abd-Halim, M.R.; Kargbo, B. Influence of municipal wastewater on rice seed germination, seedling performance, nutrient uptake, and chlorophyll content. J. Crop Sci. Biotechnol. 2015, 18, 9–19. [Google Scholar] [CrossRef]
- Singh, P.K.; Deshbhratar, P.B.; Ramteke, D.S. Effects of sewage wastewater irrigation on soil properties, crop yield and environment. Agric. Water Manag. 2012, 103, 100–104. [Google Scholar] [CrossRef]
- Khalil, S.; Kakar, M.K. Agricultural use of untreated urban wastewater in Pakistan. Asian J. Agric. Rural. Dev. 2011, 1, 21–26. [Google Scholar]
- Khawla, K.; Besma, K.; Enrique, M.; Mohamed, H. Accumulation of trace elements by corn (Zea mays) under irrigation with treated domestic wastewater using different irrigation methods. Ecotoxicol. Environ. Saf. 2019, 170, 530–537. [Google Scholar] [CrossRef]
- Khan, A.; Bruce, K. How residues of deinstitutionalised practices persist over time: World Bank boundary work in development projects in Pakistan from the 1970s to the mid-2000s. Bus. Hist. 2021, 4, 1–29. [Google Scholar] [CrossRef]
- Contreras, J.D.; Meza, R.; Siebe, C.; Rodríguez-Dozal, S.; López-Vidal, Y.A.; Castillo-Rojas, G.; Amieva, R.I.; Solano-Gálvez, S.G.; Mazari-Hiriart, M.; Silva-Magaña, M.A.; et al. Health risks from exposure to untreated domestic wastewater used for irrigation in the Mezquital Valley, Mexico: A 25-year update. Water Res. 2017, 123, 834–850. [Google Scholar] [CrossRef]
- Keraita, B.; Jimenez, B.; Drechsel, P. Extent and implications of agricultural reuse of untreated, partly treated and diluted wastewater in developing countries. CABI Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2008, 3, 1–15. [Google Scholar] [CrossRef]
- Hamilton, A.J.; Stagnitti, F.; Xiong, X.; Kreidl, S.L.; Benke, K.K.; Maher, P. Wastewater irrigation: The state of play. Vadose Zone J. 2007, 6, 823–840. [Google Scholar] [CrossRef]
- Rashid, A.; Mirza, S.A.; Keating, C.; Ali, S.; Campos, L.C. Hospital wastewater treated with a novel bacterial consortium (Alcaligenes faecalis and Bacillus paramycoides spp.) for phytotoxicity reduction in Berseem clover and tomato crops. Water Sci. Technol. 2021, 83, 1764–1780. [Google Scholar] [CrossRef] [PubMed]
- Rashid, A.; Mirza, S.A.; Keating, C.; Ali, S.; Campos, L.C. Indigenous Bacillus paramycoides spp. and Alcaligenes faecalis: Sustainable solution for bioremediation of hospital wastewater. Environ. Technol. 2020, 43, 1903–1916. [Google Scholar] [CrossRef]
- Sonune, N.A.; Garode, A.M. Bioremediation potential of bacterial isolates for municipal wastewater treatment. Curr. World Environ. 2015, 10, 619–625. [Google Scholar] [CrossRef]
- Díaz, E. Genomic insights into the aerobic pathways for degradation of organic pollutants. In Microbial Biodegradation: Genomics and Molecular Biology, 1st ed.; McLeod, M.P., Eltis, L.D., Eds.; Caister Academic Press: Wymondham, UK, 2008; pp. 5–24. [Google Scholar]
- Nirmala, B. Rice production in Asia: Key to global food security. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2018, 88, 1323–1328. [Google Scholar]
- Rehman, A.; Jingdong, L.; Chandio, A.A.; Shabbir, M.; Hussain, I. Economic outlook of rice crops in Pakistan: A time series analysis (1970–2015). Financ. Innov. 2017, 3, 13. [Google Scholar] [CrossRef]
- Verma, A.K.; Dash, R.R.; Bhunia, P. A review on chemical coagulation/flocculation technologies for removal of colour from textile wastewaters. J. Environ. Manag. 2012, 93, 154–168. [Google Scholar] [CrossRef]
- Hai, F.I.; Yamamoto, K.; Fukushi, K. Hybrid treatment systems for dye wastewater. Crit. Rev. Environ. Sci. Technol. 2007, 37, 315–377. [Google Scholar] [CrossRef]
- Dash, A.K. Impact of domestic waste water on seed germination and physiological parameters of rice and wheat. Int. J. Recent Res. Appl. Stud. 2012, 12, 280–286. [Google Scholar]
- Li, L.; Yang, Y.; Tam, N.F.; Yang, L.; Mei, X.Q.; Yang, F.J. Growth characteristics of six wetland plants and their influences on domestic wastewater treatment efficiency. Ecol. Eng. 2013, 60, 382–392. [Google Scholar] [CrossRef]
- Singh, R.P.; Singh, P.; Kothari, R.; Gautam, K.; Singh, A. Application of Wastewater in Irrigation and Its Regulation with Special Reference to Agriculture Residues. In Water Pollution and Management Practices; Springer: Singapore, 2021; pp. 177–199. [Google Scholar]
- Tran, L.D.; Phung, L.D.; Pham, D.V.; Pham, D.D.; Nishiyama, M.; Sasaki, A.; Watanabe, T. High yield and nutritional quality of rice for animal feed achieved by continuous irrigation with treated municipal wastewater. Paddy Water Environ. 2019, 17, 507–513. [Google Scholar] [CrossRef]
- Fendri, I.; SBen, R.; Khemakhem, B.; Ben, H.N.; Gdoura, R.; Abdelkafi, S. Effect of treated and untreated domestic wastewater on seed germination, seedling growth and amylase and lipase activities in Avena sativa L. J. Sci. Food Agric. 2013, 93, 74–156. [Google Scholar] [CrossRef] [PubMed]
- Huy, V.; Iwai, C.B. Utilisation of wastewater on seed germination and physiological parameters of rice (Oryza sativa L.). IOP Conf. Ser. Mater. Sci. Eng. 2018, 334, 012036. [Google Scholar] [CrossRef]
- WWF. ‘Study: Growing More Rice with Less Water’. Available online: https://www.bing.com/search?q=rice+need+more+water+an+issue+to+water+scarce+countries&qs=n&form=QBRE&sp=-1&lq=0&pq=&sc=0-0&sk=&cvid=39E27064BE174AD092C4A449E8AA3B89&ghsh=0&ghacc=0&ghpl= (accessed on 28 August 2023).
- Hydroponics Rice Paddy Nursery: An Innovative Twist on Growing Rice in India, Rice Today. Available online: https://ricetoday.irri.org/hydroponics-rice-paddy-nursery-an-innovative-twist-on-growing-rice-in-india/#:~:text=The%20other%20advantages%20of%20hydroponics,Uses%2095%25%20less%20water (accessed on 28 August 2023).
- Johnson, L. Why Use Hydroponics? Available online: https://ezgrogarden.com/hydroponics/why-use-hydroponics/#:~:text=Hydroponics%20eliminates%20soil%20and%20soil,and%20streams%2C%20harming%20fish%20populations (accessed on 28 August 2023).
- Motuzas, J.; Drobek, M.; Martens, D.L.; Vallicari, C.; Julbe, A.; Diniz da Costa, J.C. Environmental mineralization of caffeine micro-pollutant by Fe-MFI zeolites. Environ. Sci. Pollut. Res. 2017, 25, 3628–3635. [Google Scholar] [CrossRef] [PubMed]
- Gushit, J.S.; Ekanem, E.O.; Adamu, H.M.; Chindo, I.Y. Analysis of herbicide residues and organic priority pollutants in selected root and leafy vegetable crops in Plateau state, Nigeria. World J. Anal. Chem. 2013, 1, 23–28. [Google Scholar]
- Wojcieszynska, D.; Domaradzka, D.; Hupert-Kocurek, K.; Guzik, U. Bacterial degradation of naproxen—Undisclosed pollutant in the environment. J. Environ. Manag. 2014, 145, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Al Hamedi, F.; Karthishwaran, K.; Alyafei, M.A.M. Hydroponic wheat production using fresh water and treated domestic wastewater under the semi-arid region. Emir. J. Food Agric. 2021, 33, 178–186. [Google Scholar] [CrossRef]
- Ranjitha, K.M.; Biradar, S.S.; Desai, S.A.; Naik, V.R.; Bhat, S.; Satisha, T.N.; Hiremath, G.; Kumar, K.J.Y.; Chethana, C.K.; Venkatesh, K. Media standardization for hydroponic culture to screen wheat genotypes for nitrogen use efficiency. Int. J. Curr. Microbiol. App. Sci. 2017, 6, 2814–2820. [Google Scholar] [CrossRef]
- Mackowiak, C.L.; Owens, L.P.; Hinkie, C.R.; Kennedy Space Center, Florida. Continuous Hydroponic Wheat Production Using a Recirculating System; NASA John F. Kennedy Space Center: Merritt Island, FL, USA, 1989. [Google Scholar]
- Prazeres, A.R.; Albuquerque, A.; Luz, S.; Jeronimo, E.; Carvalho, F. Food Biosynthesis, In Hydroponic System: A Promising Biotechnology for Food Production and Wastewater Treatment; Elsevier Inc.: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Shah, A.H.; Munir, S.U.; Amin, N.U.; Shah, S.H. Evaluation of two nutrient solutions for growing tomatoes in a non-circulating hydroponics system. Sarhad J. Agric. 2011, 27, 557–567. [Google Scholar]
- Jesse, S.D. Treatment of Post Hydrothermal Liquefaction Wastewater and Suitability for Hydroponic Lettuce Production. Master’s Thesis, University of Illinois Urbana-Champaign, Champaign, IL, USA, 2019. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2002. [Google Scholar]
- National Environment Quality Standards (NEQS). The Gazette of Pakistan. Ministry of Environment, Local Government and Rural Development; Government of Pakistan: Islamabad, Pakistan, 2000; pp. 1289–1294.
- Mahmood, A.; Malik, R.N. Human health risk assessment of heavy metals via consumption of contaminated vegetables collected from different irrigation sources in Lahore, Pakistan. Arab. J. Chem. 2014, 7, 91–99. [Google Scholar] [CrossRef]
- Ajaz, M.; Rehman, A.; Khan, Z.; Nisar, M.A.; Hussain, S. Degradation of azo dyes by Alcaligenes aquatilis 3C and its potential use in the wastewater treatment. AMB Express 2019, 9, 64. [Google Scholar] [CrossRef] [PubMed]
- Rice Research Institute, Kala Shah Kaku. Ayub Agricultural Research Institute, Faisalabad. Available online: https://aari.punjab.gov.pk/crop_varities_rice (accessed on 24 July 2023).
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Circular. Calif. Agric. Exp. Stn. 1950, 347, 117–139. [Google Scholar]
- Dwivedi, P.; Tomar, R.S. Bioremediation of textile effluent for degradation and decolourisation of synthetic dyes: A review. Int. J. Curr. Res. Life Sci. 2018, 7, 1948–1951. [Google Scholar]
- Yokoyama, S.I.; Adachi, Y.; Asakura, S.; Kohyama, E. Characterization of Alcaligenes faecalis strain AD15 indicating biocontrol activity against plant pathogens. J. Gen. Appl. Microbiol. 2013, 59, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Rodil, R.; Quintana, J.B.; Concha-Grana, E.; Lopez-Mahia, P.; Muniategui-Lorenzo, S.; Prada-Rodriguez, D. Emerging pollutants in sewage, surface and drinking water in Galicia (NW Spain). Chemosphere 2012, 86, 1040–1049. [Google Scholar] [CrossRef]
- Meerbergen, K.; Willems, K.A.; Dewil, R.; Van Impe, J.; Appels, L.; Lievens, B. Isolation and screening of bacterial isolates from wastewater treatment plants to decolourise azo dyes. J. Biosci. Bioeng. 2018, 125, 448–456. [Google Scholar] [CrossRef]
- Spehia, R.S.; Devi, M.; Singh, J.; Sharma, S.; Negi, A.; Singh, S.; Chauhan, N.; Sharma, D.; Sharma, J.C. Lettuce growth and yield in hoagland solution with an organic concoction. Int. J. Veg. Sci. 2018, 24, 557–566. [Google Scholar] [CrossRef]
- Delden, S.H.; Nazarideljou, M.J.; Marcells, L.F.M. Nutrient solutions for Arabidopsis thaliana: A study on nutrient solution composition in hydroponics systems. Plant Methods 2020, 16, 72. [Google Scholar] [CrossRef]
- Kane, C.D. Influence of Nutrient Solution and Solution pH on Onion Growth and Mineral Content. Ph.D. Thesis, Texas Tech University, Lubbock, TX, USA, 2003. [Google Scholar]
- Shi, X.; Sun, H.; Pan, H.; Chen, Y.; Jiang, Z.; Liu, J.; Wang, S. Growth and efficiency of nutrient removal by Salix jiangsuensis J172 for phytoremediation of urban wastewater. Environ. Sci. Pollut. Res. 2016, 23, 2715–2723. [Google Scholar] [CrossRef]
- Saleem, A.; Zulfiqar, A.; Ali, B.; Naseeb, M.A.; Almasaudi, A.S.; Harakeh, S. Iron Sulfate (FeSO4) Improved physiological attributes and antioxidant capacity by reducing oxidative stress of Oryza sativa L. cultivars in alkaline soil. Sustainability 2022, 14, 16845. [Google Scholar] [CrossRef]
- Singh, R.P.; Agrawal, M. Variations in heavy metal accumulation, growth and yield of rice plants grown at different sewage sludge amendment rates. Ecotoxicol. Environ. Saf. 2010, 73, 632–641. [Google Scholar] [CrossRef]
- Kang, J.G.; Iersel, M.W. Nutrient Solution Concentration Affects Shoot: Root Ratio, Leaf Area Ratio, and Growth of Subirrigated Salvia (Salvia splendens). HortScience 2004, 39, 49–54. [Google Scholar] [CrossRef]
- Trejo-Téllez, L.I.; Gómez-Merino, F.C. Hydroponics—A Standard Methodology for Plant Biological Researches, In Nutrient Solutions for Hydroponic Systems; IntechOpen: London, UK, 2012. [Google Scholar]
- Amedi, J.; Rasheed, R.; Ibrahim, D. Responce of Physico-Chemical Soil Properties to Wastewater Application and Subsequent Effects on Three Woody Species Plants. J. Duhok Univ. 2021, 24, 106–125. [Google Scholar] [CrossRef]
- Rana, S.; Kumar, K. Study of phytotoxic effect of textile wastewater on seed germination and seedling growth of Triticum aestivum. Int. J. BioSci. Technol. 2017, 10, 58–66. [Google Scholar]
- Magwaza, S.T.; Magwaza, L.S.; Odindo, A.O.; Mditshwa, A. Hydroponic technology as decentralized system for domestic wastewater treatment and vegetable production in urban agriculture: A review. Sci. Total Environ. 2020, 698, 134154. [Google Scholar] [CrossRef]
- Xu, B.; Wang, F.; Zhang, Q.; Lan, Q.; Liu, C.; Guo, X.; Cai, Q.; Chen, Y.; Wang, G.; Ding, J. Influence of iron plaque on the uptake and accumulation of chromium by rice (Oryza sativa L.) seedlings: Insights from hydroponic and soil cultivation. Ecotoxicol. Environ. Saf. 2018, 162, 51–58. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | PK386 | Basmati 515 |
|---|---|---|
| Type | Fine | Fine Basmati |
| Class | Long grain | Extra long grain |
| Aroma | Yes | Yes |
| Chalkiness | Absent | Absent |
| Height of plant (cm) | 117 | 130 |
| Grain length (mm) | 6.85 | 7.56 |
| Grain width (mm) | 1.78 | 1.64 |
| Grain thickness (mm) | 1.56 | 1.52 |
| Nitrogen (N) (mg/L) | 22 | 24 |
| Phosphorus (P) (mg/L) | 115 | 119 |
| Potassium (K) (mg/L) | 115 | 123 |
| Iodine (I) (mg/L) | Nd | Nd |
| Zinc (Zn) (mg/L) | 1.1 | 1.3 |
| Manganese (Mn) (mg/L) | 2 | 2.4 |
| Iron (Fe) (mg/L) | 0.8 | 1.1 |
| Treatment Type | Composition of Treatment | Water + Hoagland Concentration |
|---|---|---|
| Control | Distilled water with Hoagland Solution (DWH) | 400 mL distilled wastewater + 100 mL Hoagland solution |
| Treatment 1 | Untreated Domestic wastewater with Hoagland Solution (UTDWWH) | 400 mL untreated domestic wastewater + 100 mL Hoagland solution |
| Treatment 2 | Treated domestic wastewater with Hoagland Solution (TDWWH) | 400 mL treated domestic wastewater + 100 mL Hoagland solution |
| Treatment 3 | Untreated domestic wastewater (UTDWW) | 500 mL untreated domestic wastewater |
| Treatment 4 | Treated domestic wastewater (TDWW) | 500 mL treated domestic wastewater |
| Characters | Units | NEQS [41] | Control (DWH) | Treatment-1 (UTDWWH) | Treatment-2 (TDWWH) | Treatment-3 (UTDWW) | Treament-4 (TDWW) |
|---|---|---|---|---|---|---|---|
| pH | 6.6–8.5 | 7.6 | 7.8 **** | 7.2 **** | 8.5 **** | 8.2 **** | |
| EC | µs/cm | - | 170 | 413.2 *** | 215.4 *** | 973 *** | 345.9 **** |
| TDS | mg/L | 1000 | 298.3 | 500 **** | 221.3 ** | 730 **** | 330 **** |
| TSS | mg/L | <500 | 200 | 930 **** | 298 **** | 383 *** | 300 **** |
| Salinity | ppt | - | 0.02 | 0.3 **** | 0.12 **** | 0.42 **** | 0.2 **** |
| Turbidity | NTU | 5 | 2.5 | 13.5 **** | 4.2 *** | 38.5 **** | 18.3 **** |
| COD | mg/L | 150–400 | 200 | 273.2 **** | 140.5 **** | 412.8 **** | 266 **** |
| BOD | mg/L | 80–250 | 140 | 266.9 **** | 171.5 *** | 295.7 **** | 190 **** |
| Nitrogen (N) | mg/L | - | 210 | 140.7 **** | 86 **** | 40.7 **** | 16 **** |
| Phosphorous (P) | mg/L | - | 31 | 47.5 **** | 27.3 **** | 17.5 **** | 7.3 **** |
| Potassium (K) | mg/L | - | 235 | 260 **** | 83.6 **** | 60 **** | 22.6 **** |
| Zinc (Zn) | mg/L | 5 | 0.023 | 2.099 **** | 1.83 **** | 2 **** | 1.3 **** |
| Manganese (Mn) | mg/L | 1.5 | 0.11 | 0.064 **** | 0.094 **** | 0.044 **** | 0.004 **** |
| Iron (Fe) | mg/L | 2 | 1 | 3.3 **** | 1.36 **** | 2.3 **** | 0.36 **** |
| Morphological Parameters | Categories | Types of Hydroponic Cultures | ||||
|---|---|---|---|---|---|---|
| Control (DWH) | Treatment-1 (UTDWWH) | Treatment-2 (TDWWH) | Treatment-3 (UTDWW) | Treament-4 (TDWW) | ||
| Wet weight (g) | Shoot | 0.248 (±0.034, 1.71 × 10−3) | 0.282 (±0.016, 3.86 × 10−4) * | 0.278 (±0.026, 1.15 × 10−3) * | 0.343 (±0.032, 1.56 × 10−3) * | 0.282 (±0.026, 8.86 × 10−4) * |
| Root | 0.124 (±0.017, 8.56 × 10−4) | 0.141 (±0.08, 1.93 × 10−4) * | 0.139 (±0.013, 5.76 × 10−4) ** | 0.217 (±0.0161, 7.83 × 10−4) ** | 0.141 (±0.013, 4.43 × 10−4) * | |
| Seedling | 0.372 (±0.051, 2.57 × 10−3) | 0.412 (±0.024, 5.8 × 10−4) * | 0.416 (±0.039, 1.73 × 10−3) * | 0.38 (±0.0485, 2.35 × 10−3) * | 0.424 (±0.039, 1.33 × 10−3) * | |
| Dry weight (g) | Shoot | 0.0128 (±0.002, 1.05 × 10−5) | 0.013 (±0.0014, 3.33 × 10−6) * | 0.0014 (±0.002, 1.35 × 10−5) * | 0.0114 (±0.0022, 6.6 × 10−6) * | 0.018 (±0.003, 1.82 × 10−5) * |
| Root | 0.0064 (±0.001, 5.26 × 10−6) | 0.0063 (±0.0007, 1.66 × 10−6) ** | 0.007 (±0.001, 6.76 × 10−6) * | 0.0057 (±0.0011, 3.3 × 10−6) * | 0.009 (±0.0015, 9.1 × 10−6) * | |
| Seedling | 0.0194 (±0.0039, 1.58 × 10−5) | 0.019 (±0.002, 5.0 × 10−6) * | 0.022 (±0.004, 2.03 × 10−5) * | 0.017 (±0.0032, 1.0 × 10−5) * | 0.028 (±0.0045, 2.73 × 10−5) * | |
| F-value | 0.0244 | 0.0437 | 0.0028 | 0.091 | 0.058 | |
| Morphological Parameters | Categories | Types of Hydroponic Cultures | ||||
|---|---|---|---|---|---|---|
| Control (DWH) | Treatment-1 (UTDWWH) | Treatment-2 (TDWWH) | Treatment-3 (UTDWW) | Treament-4 (TDWW) | ||
| Wet weight (g) | Shoot | 0.138 (±0.072, 6.86 × 10−4) | 0.172 (±0.021, 7.13 × 10−4) *** | 0.204 (±0.014, 1.53 × 10−4) *** | 0.198 (±0.0264, 2.86 × 10−4) *** | 0.242 (±0.022, 1.11 × 10−3) *** |
| Root | 0.069 (±0.036, 3.43 × 10−4) | 0.086 (±0.010, 3.56 × 10−4) *** | 0.102 (±0.007, 7.66 × 10−5) *** | 0.099 (±0.013, 1.43 × 10−4) *** | 0.121 (±0.011, 5.56 × 10−4) *** | |
| Seedling | 0.208 (±0.107, 1.03 × 10−3) | 0.258 (±0.031, 1.07 × 10−3) *** | 0.306 (±0.021, 2.3 × 10−4) *** | 0.296 (±0.0397, 4.3 × 10−4) *** | 0.364 (±0.034, 1.67 × 10−3) *** | |
| Dry weight (g) | Shoot | 0.012 (±0.0028, 1.21 × 10−5) | 0.012 (±0.0016, 5.95 × 10−3) *** | 0.015 (±0.002, 1.18 × 10−5) *** | 0.094 (±0.021, 1.58 × 10−5) *** | 0.016 (±0.002, 4.1 × 10−3) *** |
| Root | 0.006 (±0.0014, 6.06 × 10−6) | 0.0061 (±0.0008, 2.97 × 10−3) *** | 0.007 (±0.001, 5.9 × 10−6) *** | 0.047 (±0.0103, 7.93 × 10−6) *** | 0.008 (±0.0.001, 2.5 × 10−3) *** | |
| Seedling | 0.019 (±0.0043, 1.82 × 10−5) | 0.0184 (±0.0023, 8.93 × 10−3) *** | 0.022 (±0.004, 1.77 × 10−5) *** | 0.0142 (±0.031, 2.38 × 10−5) *** | 0.024 (±0.0029, 6.15 × 10−3) *** | |
| F-value | 0.0042 | 0.0392 | 0.0494 | 0.0589 | 0.0658 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aslam, T.; Mirza, S.A.; Rashid, A.; Javed, M.A.; Campos, L.C. Efficiency of Treated Domestic Wastewater to Irrigate Two Rice Cultivars, PK 386 and Basmati 515, under a Hydroponic Culture System. Water 2023, 15, 3149. https://doi.org/10.3390/w15173149
Aslam T, Mirza SA, Rashid A, Javed MA, Campos LC. Efficiency of Treated Domestic Wastewater to Irrigate Two Rice Cultivars, PK 386 and Basmati 515, under a Hydroponic Culture System. Water. 2023; 15(17):3149. https://doi.org/10.3390/w15173149
Chicago/Turabian StyleAslam, Tahira, Safdar A. Mirza, Aneeba Rashid, Muhammad Arshad Javed, and Luiza C. Campos. 2023. "Efficiency of Treated Domestic Wastewater to Irrigate Two Rice Cultivars, PK 386 and Basmati 515, under a Hydroponic Culture System" Water 15, no. 17: 3149. https://doi.org/10.3390/w15173149
APA StyleAslam, T., Mirza, S. A., Rashid, A., Javed, M. A., & Campos, L. C. (2023). Efficiency of Treated Domestic Wastewater to Irrigate Two Rice Cultivars, PK 386 and Basmati 515, under a Hydroponic Culture System. Water, 15(17), 3149. https://doi.org/10.3390/w15173149







