Thermal Polymerisation Synthesis of g-C3N4 for Photocatalytic Degradation of Rhodamine B Dye under Natural Sunlight
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of g-C3N4
2.3. Material Characterisation
2.4. Photocatalytic Performance Measurement
3. Results and Discussions
3.1. Crystal Structure and Functional Group Analysis
3.2. Microstructure and Compositions Analysis
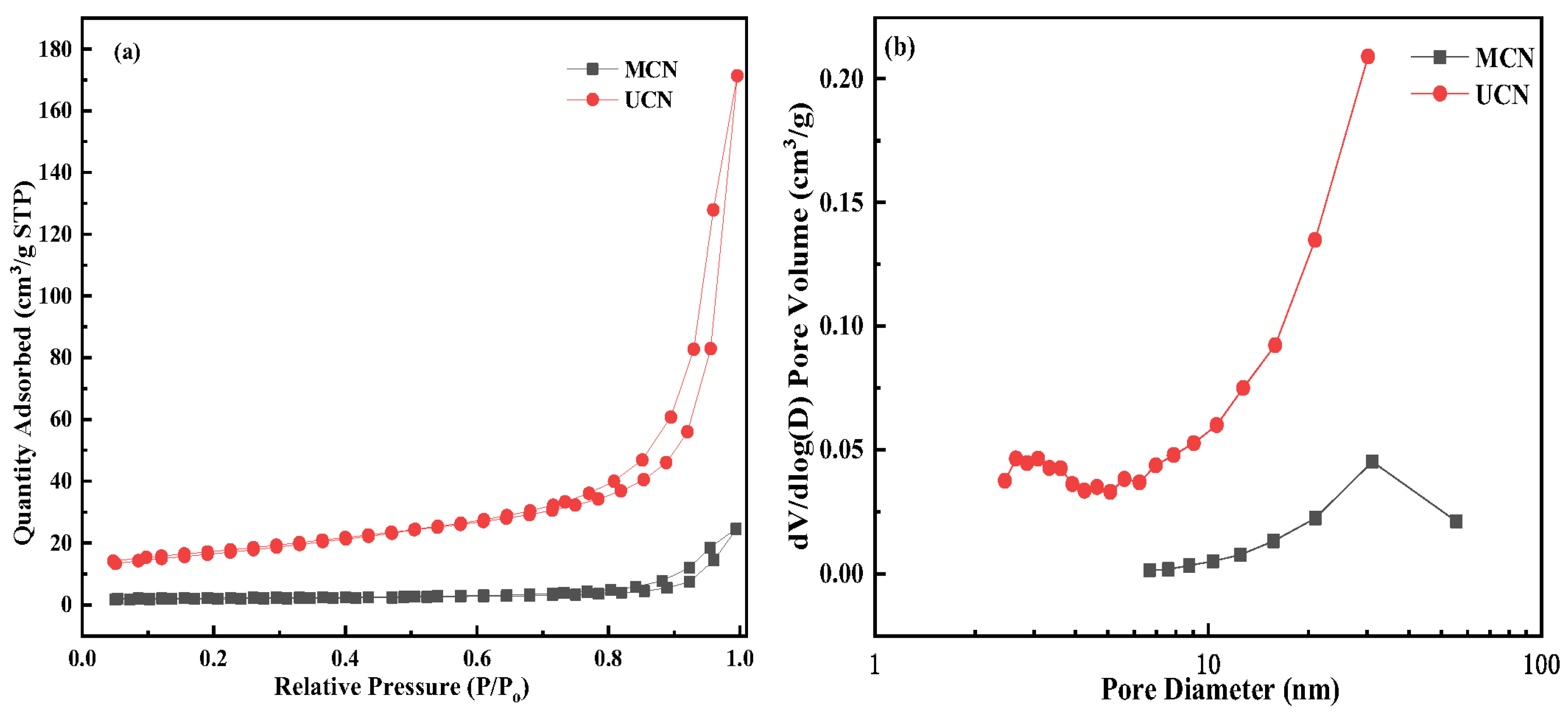
3.3. BET Surface Area and Pore Size Distribution Analysis
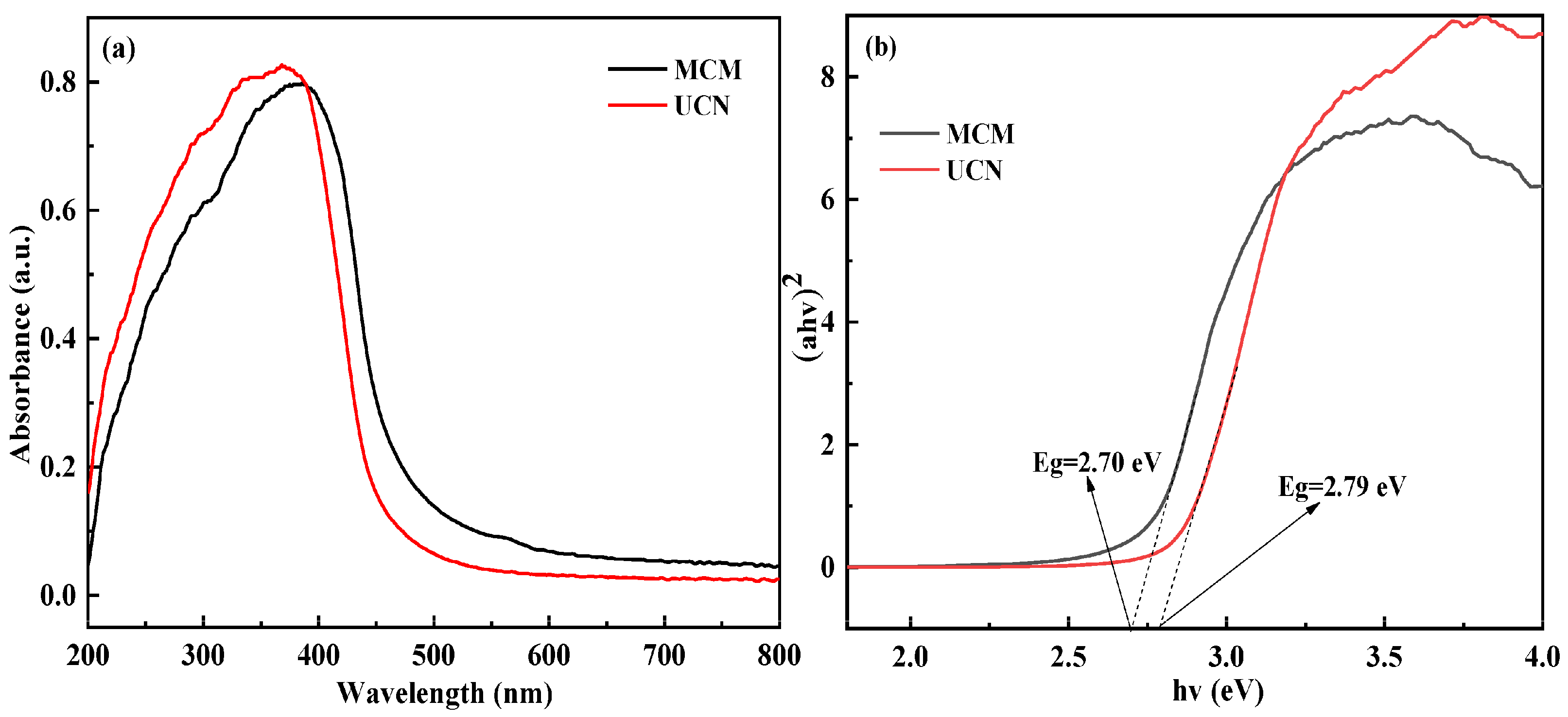
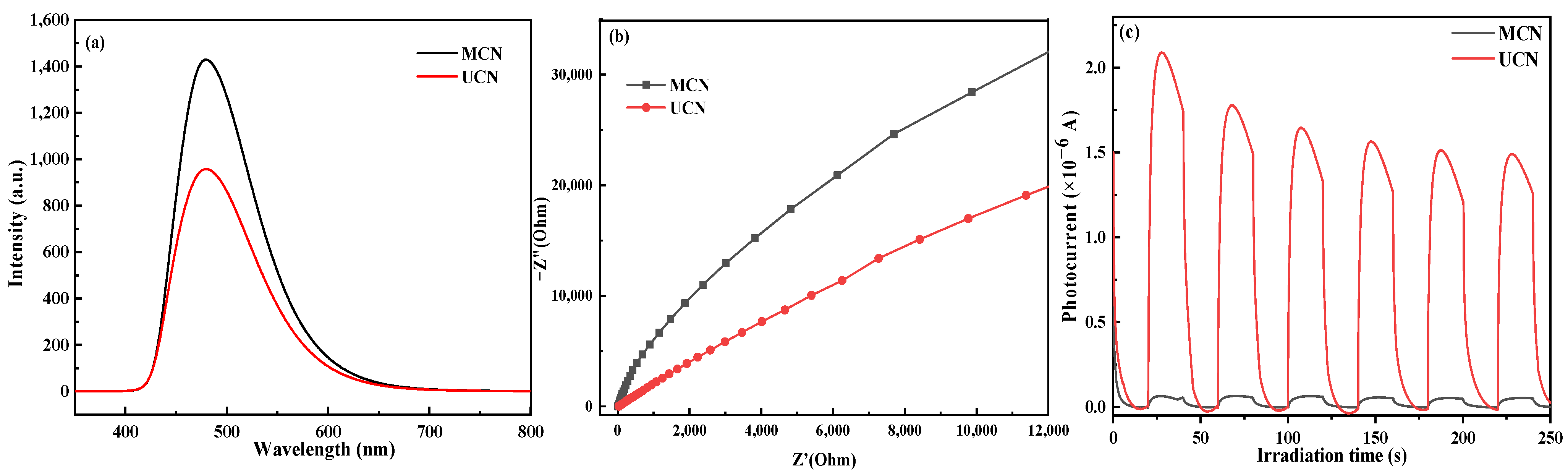
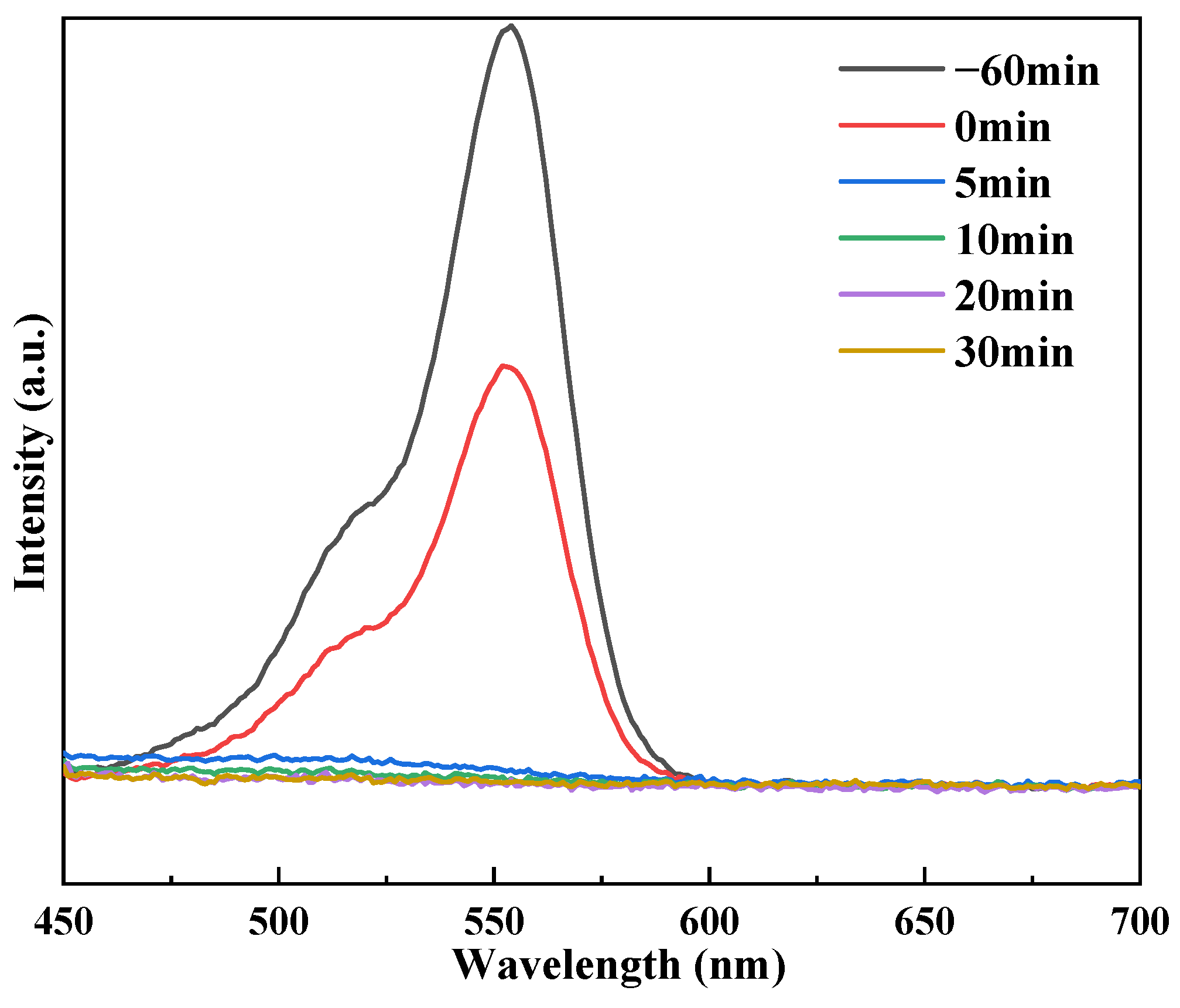
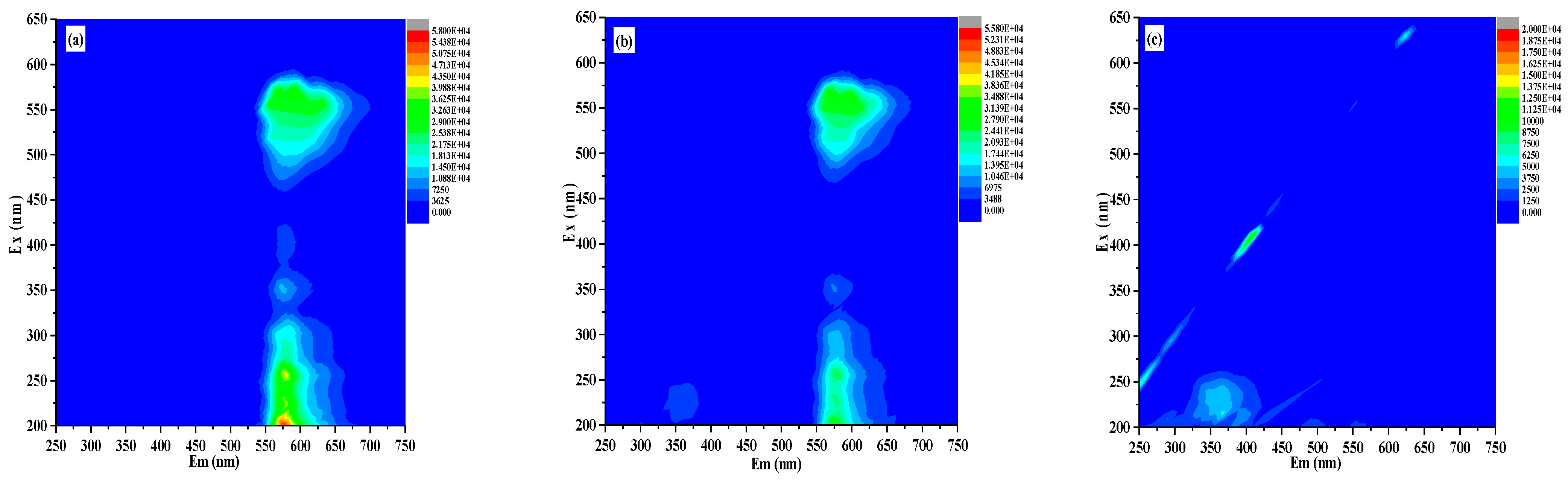
3.4. UV–Vis Spectroscopy and Photochemical Characteristics
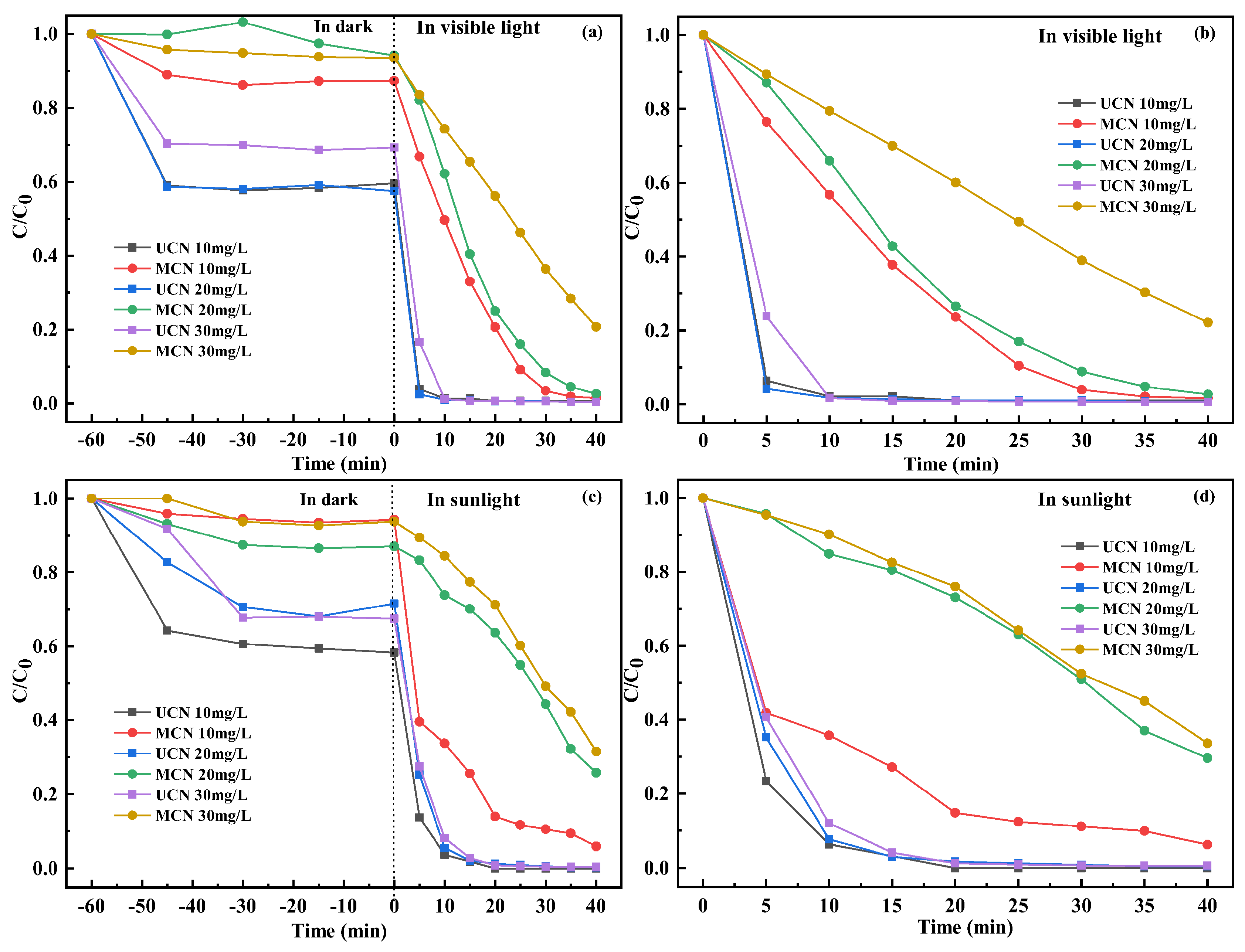
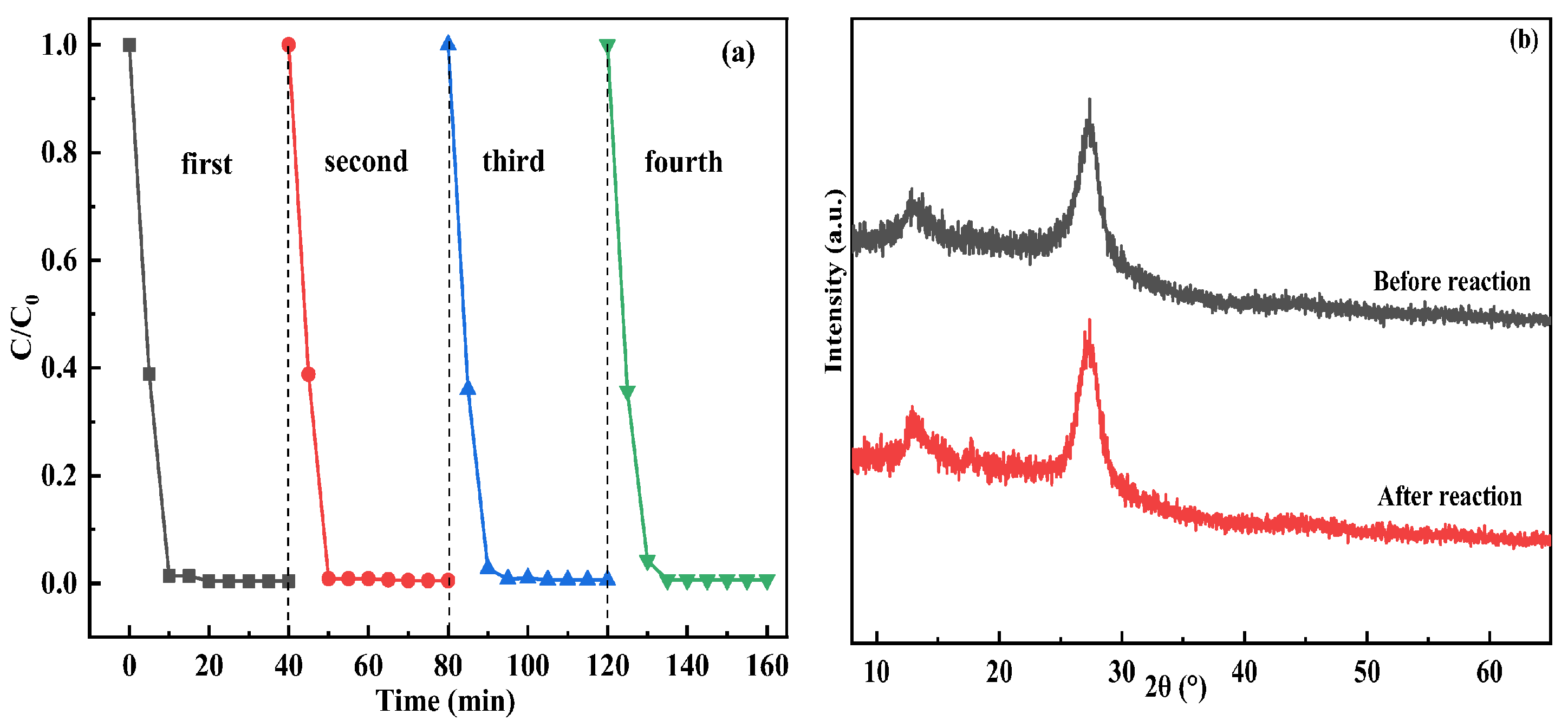
3.5. Photocatalytic Degradation Performance
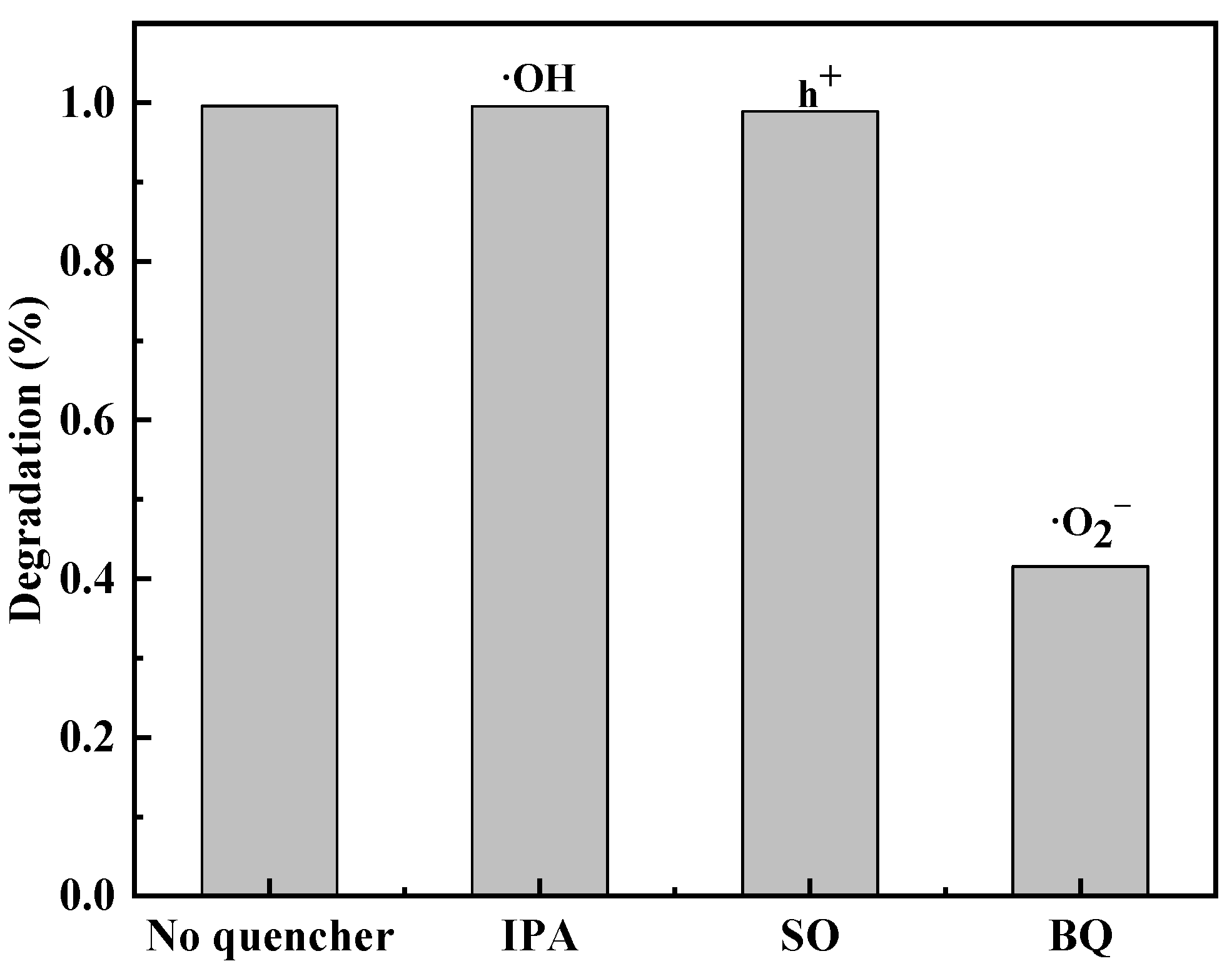
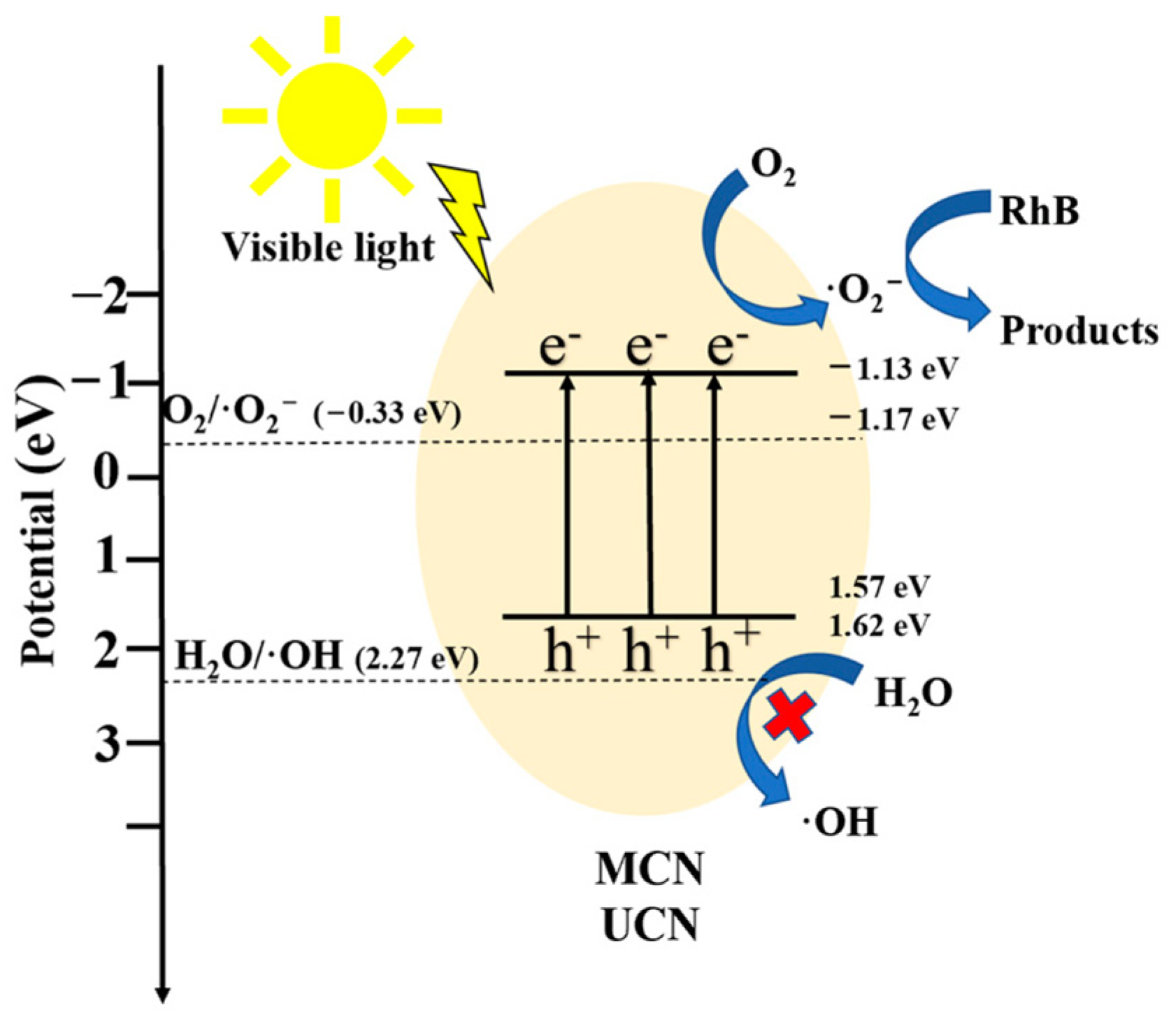
3.6. Photocatalytic Mechanism
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tian, Y.; Zhang, J.; Wang, W.; Liu, J.; Zheng, X.; Li, J.; Guan, X. Facile assembly and excellent elimination behavior of porous BiOBr-g-C3N4 heterojunctions for organic pollutants. Environ. Res. 2022, 209, 112889. [Google Scholar] [CrossRef] [PubMed]
- Mahdieh, V.; Iman, K.; Farzaneh, S.; Parastoo, J. Green synthesis of a novel magnetic Fe3O4@SiO2/TiO2@WO3 nanocomposite for methylene blue removal under UV and visible light irradiations. Res. Chem. Intermed. 2023, 49, 1909–1924. [Google Scholar]
- Zhang, M.; Sun, X.; Wang, C.; Wang, Y.; Tan, Z.; Li, J.; Xi, B. Photocatalytic degradation of rhodamine B using Bi4O5Br2-doped ZSM-5. Mater. Chem. Phys. 2022, 278, 125697. [Google Scholar] [CrossRef]
- Budnyak, T.M.; Onwumere, J.; Pylypchuk, I.V.; Jaworski, A.; Chen, J.; Rokicińska, A.; Lindström, M.E.; Kuśtrowski, P.; Sevastyanova, O.; Slabon, A. LignoPhot: Conversion of hydrolysis lignin into the photoactive hybrid lignin/Bi4O5Br2/BiOBr composite for simultaneous dyes oxidation and Co2+ and Ni2+ recycling. Chemosphere 2021, 279, 130538. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Ma, B.; Zhang, X.; Sang, Y.; Liu, H. One-pot synthesis of BiOCl nanosheets with dual functional carbon for ultra-highly efficient photocatalytic degradation of RhB. Environ. Res. 2020, 182, 109077. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, L.; Wang, D.; Zhang, Q.; Zeng, J.; Zhang, R. Facile synthesis of nitrogen-defective g-C3N4 for superior photocatalytic degradation of rhodamine B. RSC Adv. 2021, 11, 30503–30509. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, W.; Cai, W. The synergistic effect of Ag/AgCl@ZIF-8 modified g-C3N4 composite and peroxymonosulfate for the enhanced visible-light photocatalytic degradation of levofloxacin. Sci. Total Environ. 2019, 696, 133962. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Li, X.; Li, Y.; Zhao, H.; Wang, Y.; Li, N.; Li, X.; Li, G. Synthesis of CdS/m-TiO2 mesoporous spheres and their application in photocatalytic degradation of rhodamine B under visible light. Chem. Res. Chin. Univ. 2017, 33, 436–441. [Google Scholar] [CrossRef]
- Wang, C.; Sun, X.; Zhang, M.; Wang, Y.; Tan, Z.; Li, J.; Xi, B. Ultrasound-assisted room-temperature in situ precipitation synthesis of BC doped Bi4O5Br2 for enhanced photocatalytic activity in pollutants degradation under visible light. J. Alloys Compd. 2021, 889, 161609. [Google Scholar] [CrossRef]
- Zhang, G.-Q.; Chang, N.; Han, D.-Q.; Zhou, A.-Q.; Xu, X.-H. The enhanced visible light photocatalytic activity of nanosheet-like Bi2WO6 obtained by acid treatment for the degradation of rhodamine B. Mater. Lett. 2010, 64, 2135–2137. [Google Scholar] [CrossRef]
- Hu, C.; Xu, M.; Zhang, J.; Zhou, Y.; Hu, B.; Yu, G. Recyclable MoO3 nanobelts for photocatalytic degradation of Rhodamine B by near infrared irradiation. Int. J. Chem. Kinet. 2018, 51, 3–13. [Google Scholar] [CrossRef]
- Nguyen, L.T.T.; Vo, D.-V.N.; Nguyen, L.T.H.; Duong, A.T.T.; Nguyen, H.Q.; Chu, N.M.; Nguyen, D.T.C.; Van Tran, T. Synthesis, characterization, and application of ZnFe2O4@ZnO nanoparticles for photocatalytic degradation of Rhodamine B under visible-light illumination. Environ. Technol. Innov. 2021, 25, 102130. [Google Scholar] [CrossRef]
- Lin, X.; Li, Y. Preparation of TiO2/Ag[BMIM]Cl Composites and Their Visible Light Photocatalytic Properties for the Degradation of Rhodamine B. Catalysts 2021, 11, 661. [Google Scholar] [CrossRef]
- Liu, W.; Li, Y.; Liu, F.; Jiang, W.; Zhang, D.; Liang, J. Visible-light-driven photocatalytic degradation of diclofenac by carbon quantum dots modified porous g-C3N4: Mechanisms, degradation pathway and DFT calculation. Water Res. 2018, 151, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Prabavathi, S.L.; Saravanakumar, K.; Mamba, G.; Muthuraj, V. 1D/2D MnWO4 nanorods anchored on g-C3N4 nanosheets for enhanced photocatalytic degradation ofloxacin under visible light irradiation. Colloids Surf. A. Physicochem. Eng. Asp. 2019, 581, 123845. [Google Scholar]
- Song, Y.; Gu, J.; Xia, K.; Yi, J.; Che, H.; She, X.; Chen, Z.; Ding, C.; Li, H.; Xu, H. Construction of 2D SnS2/g-C3N4 Z-scheme composite with superior visible-light photocatalytic performance. Appl. Surf. Sci. 2019, 467/468, 56–64. [Google Scholar] [CrossRef]
- Orooji, Y.; Ghanbari, M.; Amiri, O.; Salavati-Niasari, M. Facile fabrication of silver iodide/graphitic carbon nitride nanocomposites by notable photo-catalytic performance through sunlight and antimicrobial activity. J. Hazard. Mater. 2020, 389, 122079. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, Z.; Lu, Y.; Liu, S.; Chen, X.; Wei, G.; Ye, G.; Chen, J. Microplasma electrochemistry (MIPEC) methods for improving the photocatalytic performance of g-C3N4 in degradation of RhB. Appl. Surf. Sci. 2020, 531, 147307. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, Y.; Schnepp, Z. Soft and hard templating of graphitic carbon nitride. J. Mater. Chem. A 2015, 3, 14081–14092. [Google Scholar] [CrossRef]
- Wang, W.; Li, G.; An, T.; Chan, D.K.L.; Yu, J.C.; Wong, P.K. Photocatalytic hydrogen evolution and bacterial inactivation utilizing sonochemical-synthesized g-C3N4/red phosphorus hybrid nanosheets as a wide-spectral-responsive photocatalyst: The role of type I band alignment. Appl. Catal. B Environ. 2018, 238, 126–135. [Google Scholar] [CrossRef]
- Zhong, S.; Zhou, H.; Shen, M.; Yao, Y.; Gao, Q. Rationally designed a g-C3N4/BiOI/Bi2O2CO3 composite with promoted photocatalytic activity. J. Alloys Compd. 2021, 853, 157307. [Google Scholar] [CrossRef]
- Cao, C.; Wang, H. Carbon nitride films deposited from organic solutions by electrodeposition. Diam. Relat. Mater. 1999, 8, 1786–1789. [Google Scholar] [CrossRef]
- Viehland, J.; Lin, S.; Feldman, B.J.; Kilgore, K.; Jones, M.T. Search for the nitrogen dangling bond in amorphous hydrogenated carbon nitride. Solid State Commun. 1991, 80, 597–599. [Google Scholar] [CrossRef]
- Wei, H.; McMaster, W.A.; Tan, J.Z.Y.; Cao, L.; Chen, D.; Caruso, R.A. Mesoporous TiO2/g-C3N4 Microspheres with Enhanced Visible-Light Photocatalytic Activity. J. Phys. Chem. C. Nanomater. Interfaces 2017, 121, 22114–22122. [Google Scholar] [CrossRef]
- Bai, Y.-J.; Lü, B.; Liu, Z.-G.; Li, L.; Cui, D.-L.; Xu, X.-G.; Wang, Q.-L. Solvothermal preparation of graphite-like C3N4 nanocrystals. J. Cryst. Growth 2003, 247, 505–508. [Google Scholar] [CrossRef]
- Niu, P.; Zhang, L.; Liu, G.; Cheng, H.-M. Graphene-Like Carbon Nitride Nanosheets for Improved Photocatalytic Activities. Adv. Funct. Mater. 2012, 22, 4763–4770. [Google Scholar] [CrossRef]
- Andreyev, A.; Akaishi, M.; Golberg, D. Sodium flux-assisted low-temperature high-pressure synthesis of carbon nitride with high nitrogen content. Chem. Phys. Lett. 2003, 372, 635–639. [Google Scholar] [CrossRef]
- Goettmann, F.; Fischer, A.; Antonietti, M.; Thomas, A. Chemical Synthesis of Mesoporous Carbon Nitrides Using Hard Templates and Their Use as a Metal-Free Catalyst for Friedel-Crafts Reaction of Benzene. Angew. Chem. 2006, 45, 4467–4471. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guo, F.; Wang, X. An Optimized and General Synthetic Strategy for Fabrication of Polymeric Carbon Nitride Nanoarchitectures. Adv. Funct. Mater. 2013, 23, 3008–3014. [Google Scholar] [CrossRef]
- Yan, S.C.; Li, Z.S.; Zou, Z.G. Photodegradation Performance of g-C3N4 Fabricated by Directly Heating Melamine. Langmuir 2009, 25, 10397–10401. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, T.; Wang, Z. Simple pyrolysis of urea into graphitic carbon nitride with recyclable adsorption and photocatalytic activity. J. Mater. Chem. A 2011, 21, 14398–14401. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, E.; Hu, X.; Tang, C.; Wan, J.; Li, J.; Fan, J. simple process to prepare few-layer g-C3N4 nanosheets with enhanced photocatalytic activities. Appl. Surf. Sci. 2015, 358, 246–251. [Google Scholar] [CrossRef]
- Dong, L.; Chunling, L.; Jiayu, G.; Congyue, Z.; Qian, Z.; Fengquan, Z.; Tianjun, N.; Weidong, W. 3D interconnected g-C3N4 hybridized with 2D Ti3C2 MXene nanosheets for enhancing visible light photocatalytic hydrogen evolution and dye contaminant elimination. Appl. Surf. Sci. 2021, 579, 152180. [Google Scholar]
- Xu, F.; Cheng, G.; Song, S.; Wei, Y.; Chen, R. Insights into Promoted Adsorption Capability of Layered BiOCl Nanostructures Decorated with TiO2 Nanoparticles. ACS Sustain. Chem. Eng. 2016, 4, 12. [Google Scholar] [CrossRef]
- Xiong, J.; Gan, Y.; Zhu, J.; Li, W.; Gao, C.; Wei, Y.; Cheng, G.; Li, Z.; Dou, S. Insights into the structure-induced catalysis dependence of simply engineered one-dimensional zinc oxide nanocrystals towards photocatalytic water purification. Inorg. Chem. Front. 2017, 4, 2075–2087. [Google Scholar] [CrossRef]
- Zhang, M.; Xiong, J.; Yang, H.; Wen, Z.; Chen, R.; Cheng, G. Surface Potential/Wettability and Interface Charge Transfer Engineering of Copper-Oxide (Cu–MOx, M = W, Ti, and Ce) Hybrids for Efficient Wastewater Treatment through Adsorption–Photocatalysis Synergy. Ind. Eng. Chem. Res. 2020, 59, 35. [Google Scholar] [CrossRef]
- Luo, Y.; Wei, X.; Gao, B.; Zou, W.; Zheng, Y.; Yang, Y.; Zhang, Y.; Tong, Q.; Dong, L. Synergistic adsorption-photocatalysis processes of graphitic carbon nitrate (g-C3N4) for contaminant removal: Kinetics, models, and mechanisms. Chem. Eng. J. 2019, 375, 122019. [Google Scholar] [CrossRef]
- Yu, Y.; Chen, D.; Xu, W.; Fang, J.; Sun, J.; Liu, Z.; Chen, Y.; Liang, Y.; Fang, Z. Synergistic adsorption-photocatalytic degradation of different antibiotics in seawater by a porous g-C3N4/calcined-LDH and its application in synthetic mariculture wastewater. J. Hazard. Mater. 2021, 416, 126183. [Google Scholar] [CrossRef]
- Ni, T.; Zhang, H.; Yang, Z.; Zhou, L.; Pan, L.; Li, C.; Yang, Z.; Liu, D. Enhanced adsorption and catalytic degradation of antibiotics by porous 0D/3D Co3O4/g-C3N4 activated peroxymonosulfate: An experimental and mechanistic study. J. Colloid Interface Sci. 2022, 625, 466–478. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Li, C.; Zhao, C.; Zhao, Q.; Niu, T.; Pan, L.; Xu, P.; Zhang, F.; Wu, W.; Ni, T. Facile synthesis of three-dimensional hollow porous carbon doped polymeric carbon nitride with highly efficient photocatalytic performance. Chem. Eng. J. 2022, 438, 135623. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, Y.; Liu, Y.; Yang, P. Effect of nonmetal element dopants on photo- and electro-chemistry performance of ultrathin g-C3N4 nanosheets. Int. J. Hydrogen Energy 2020, 45, 16519–16527. [Google Scholar] [CrossRef]
- Di, X.; Shuo, Z.; Tianyu, W.; Mei, H.; Yiyan, C.; Panpan, Z.; Fangchao, C. Efficient RhB degradation using MnFe2O4/g-C3N4 composites under visible light irradiation. Opt. Mater. 2022, 124, 111965. [Google Scholar]
- Zhang, M.; Xing, M.; Dong, B.; Sun, X.; Zhang, H.; Wang, C.; Zhu, H. Preparation of BiVO4/CO32−-Bi2O2CO3 heterojunctions for enhanced photocatalytic activity in the degradation of levofloxacin under visible light. J. Alloys Compd. 2023, 965, 171471. [Google Scholar] [CrossRef]
- Minjie, Z.; Aiyue, J.; Yujuan, C.; Mengyu, T.; Yulian, W.; Zhigang, T. Synergistic effects of Au/g-C3N4/montmorillonite composite nanocatalysts for enhanced photocatalytic performance with visible light illumination. J. Nanoparticle Res. 2023, 25, 87. [Google Scholar]
- Li, D.; Huang, J.; Li, R.; Chen, P.; Chen, D.; Cai, M.; Liu, H.; Feng, Y.; Lv, W.; Liu, G. Synthesis of a carbon dots modified g-C3N4/SnO2 Z-scheme photocatalyst with superior photocatalytic activity for PPCPs degradation under visible light irradiation. J. Hazard. Mater. 2021, 401, 123257.1–123257.13. [Google Scholar] [CrossRef] [PubMed]
- Van, K.N.; Huu, H.T.; Nguyen Thi, V.N.; Le Thi, T.L.; Truong, D.H.; Truong, T.T.; Dao, N.N.; Vo, V.; Tran, D.L.; Vasseghian, Y. Facile construction of S-scheme SnO2/ g-C3N4 photocatalyst for improved photoactivity. Chemosphere 2021, 289, 133120. [Google Scholar] [CrossRef]
- Ghafuri, H.; Rashidizadeh, A.; Zaron, T.M. Facile preparation of CuS-g-C3N4/Ag nanocomposite with improved photocatalytic activity for the degradation of rhodamine B. Polyhedron 2020, 179, 114368. [Google Scholar] [CrossRef]
- Ma, W.; Wang, N.; Guo, Y.; Yang, L.; Lv, M.; Tang, X.; Li, S. Enhanced photoreduction CO2 activity on g-C3N4: By synergistic effect of nitrogen defective-enriched and porous structure, and mechanism insights. Chem. Eng. J. 2020, 388, 124288. [Google Scholar] [CrossRef]
- Zhu, Z.; Fan, W.; Liu, Z.; Yu, Y.; Dong, H.; Huo, P.; Yan, Y. Fabrication of the metal-free biochar-based graphitic carbon nitride for improved 2-Mercaptobenzothiazole degradation activity. J. Photochem. Photobiol. A Chem. 2018, 358, 284–293. [Google Scholar] [CrossRef]
- Xu, Z.; Zhang, J.; Zhong, J.; Wu, J.; Li, M. Reductively-induced carbon vacancies facilitate visible light-driven hydrogen evolution enhancement of g-C3N4. Mater. Sci. Semicond. Process. 2023, 161, 107459. [Google Scholar] [CrossRef]
- Shi, J.; Chen, T.; Guo, C.; Liu, Z.; Feng, S.; Li, Y.; Hu, J. The bifunctional composites of AC restrain the stack of g-C3N4 with the excellent adsorption-photocatalytic performance for the removal of RhB. Colloids Surf. A. Physicochem. Eng. Asp. 2019, 580, 123701. [Google Scholar] [CrossRef]
- Bai, J.; Yin, C.; Xu, H.; Chen, G.; Ni, Z.; Wang, Z.; Li, Y.; Kang, S.; Zheng, Z.; Li, X. Facile urea-assisted precursor pre-treatment to fabricate porous g-C3N4 nanosheets for remarkably enhanced visible-light-driven hydrogen evolution. J. Colloid Interface Sci. 2018, 532, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Xiong, Y.; Mu, X.; Wan, J.; Li, T.; Jin, Y.; Li, R. Cobalt-Doped g-C3N4 Nanosheets for One-Pot Synthesis of Imines under Mild Conditions. ACS Appl. Nano Mater. 2023, 6, 7. [Google Scholar] [CrossRef]
- Guo, Y.; Li, C.; Guo, Y.; Wang, X.; Li, X. Ultrasonic-assisted synthesis of mesoporous g-C3N4/Na-bentonite composites and its application for efficient photocatalytic simultaneous removal of Cr(VI) and RhB. Colloids Surf. A. Physicochem. Eng. Asp. 2019, 578, 123624. [Google Scholar] [CrossRef]
- Liu, X.; Pang, F.; He, M.; Ge, J. Confined reaction inside nanotubes: New approach to mesoporous g-C3N4 photocatalysts. Nano Res. 2017, 10, 3638–3647. [Google Scholar] [CrossRef]
- Liqun, Y.; Xiaoli, J.; Chuan, L.; Chenghua, D.; Haiquan, X.; Ka Him, C.; Po Keung, W. Thickness-ultrathin and bismuth-rich strategies for BiOBr to enhance photoreduction of CO2 into solar fuels. Appl. Catal. B Environ. 2016, 187, 281–290. [Google Scholar]
- Feng, W.; Fang, J.; Zhou, G.; Zhang, L.; Lu, S.; Wu, S.; Chen, Y.; Ling, Y.; Fang, Z. Rationally designed Bi@BiOCl/g-C3N4 heterostructure with exceptional solar-driven photocatalytic activity. Mol. Catal. 2017, 434, 69–79. [Google Scholar] [CrossRef]
- Hong, Y.; Li, C.; Zhang, G.; Meng, Y.; Yin, B.; Zhao, Y.; Shi, W. Efficient and stable Nb2O5 modified g-C3N4 photocatalyst for removal of antibiotic pollutant. Chem. Eng. J. 2016, 299, 74–84. [Google Scholar] [CrossRef]
- Wang, J.; Yang, Z.; Gao, X.; Yao, W.; Wei, W.; Chen, X.; Zong, R.; Zhu, Y. Core-shell g-C3N4@ZnO composites as photoanodes with double synergistic effects for enhanced visible-light photoelectrocatalytic activities. Appl. Catal. B 2017, 217, 169–180. [Google Scholar] [CrossRef]
- Pei, Q.; Mengjie, L.; Gang, C.; Wei, L.; Lijun, L.; Jinyan, X. Co-implantation of oxygen vacancy and well-dispersed Cu cocatalyst into TiO2 nanoparticles for promoting solar-to-hydrogen evolution. Int. J. Hydrogen Energy 2022, 48, 933–942. [Google Scholar]
- Chen, X.; Yao, L.; He, J.; Li, J.; Xu, S.; Li, N.; Zhu, Y.; Chen, X.; Zhu, R. Enhanced degradation of tetracycline under natural sunlight through the synergistic effect of Ag3PO4/MIL-101(Fe) photocatalysis and Fenton catalysis: Mechanism, pathway, and toxicity assessment. J. Hazard. Mater. 2023, 449, 131024. [Google Scholar] [CrossRef]
- Guo, H.; Niu, C.-G.; Wen, X.-J.; Zhang, L.; Liang, C.; Zhang, X.-G.; Guan, D.-L.; Tang, N.; Zeng, G.-M. Construction of highly efficient and stable ternary AgBr/Ag/PbBiO2Br Z-scheme photocatalyst under visible light irradiation: Performance and mechanism insight. J. Colloid Interface Sci. 2017, 513, 852–865. [Google Scholar] [CrossRef]
- Zhang, J.; Li, J.; Liu, X. Ternary nanocomposite ZnO- g-C3N4–Go for enhanced photocatalytic degradation of RhB. Opt. Mater. 2021, 119, 111351. [Google Scholar] [CrossRef]
- Yi, F.; Ma, J.; Lin, C.; Wang, L.; Zhang, H.; Qian, Y.; Zhang, K. Insights into the enhanced adsorption/photocatalysis mechanism of a Bi4O5Br2/g-C3N4 nanosheet. J. Alloys Compd. 2020, 821, 153557. [Google Scholar] [CrossRef]
- Fatemeh, Y.; Qahtan, A.Y.; Mojgan, G.; Masoud, S.-N. Fabrication of TlSnI3/C3N4 nanocomposites for enhanced photodegradation of toxic contaminants below visible light and investigation of kinetic and mechanism of photocatalytic reaction. J. Mol. Liq. 2022, 349, 118443. [Google Scholar]
- Jiang, Z.; Zhu, C.; Wan, W.; Qian, K.; Xie, J. Constructing graphite-like carbon nitride modified hierarchical yolk–shell TiO2 spheres for water pollution treatment and hydrogen production. J. Mater. Chem. A 2015, 4, 1806–1818. [Google Scholar] [CrossRef]
- Danish, M.; Muneer, M. Facile synthesis of highly efficient Co@ZnSQDs/g-C3N4/MWCNT nanocomposites and their photocatalytic potential for degradation of RhB dye: Efficiency, degradation kinetics, and mechanism pathway. Ceram. Int. 2021, 47, 13043–13056. [Google Scholar] [CrossRef]
- Renji, R.; Shanmugam, V.; Asokan, S.; Palanisamy, P.; Sanjeevamuthu, S.; Vairamuthu, R.; Jeyaperumal Kalyana, S.; Manickam, S.; Mohd, S.; AlFaify, S. Investigation on novel Cu2O modified g-C3N4/ZnO heterostructures for efficient photocatalytic dye degradation performance under visible-light exposure. Colloid Interface Sci. Commun. 2021, 44, 100480. [Google Scholar]
- Luo, J.; Zhou, X.; Ma, L.; Xu, X. Enhancing visible-light photocatalytic activity of g-C3N4 by doping phosphorus and coupling with CeO2 for the degradation of methyl orange under visible light irradiation. RSC Adv. 2015, 5, 68728–68735. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, G.; Xiong, Z.; Tang, H.; Jiang, C. Fabrication of flower-like direct Z-scheme beta-Bi2O3/g-C3N4 photocatalyst with enhanced visible light photoactivity for Rhodamine B degradation. Appl. Surf. Sci. 2018, 436, 162–171. [Google Scholar] [CrossRef]
- Huang, L.; Liu, J.; Li, P.; Li, Y.; Wang, C.; Shu, S.; Song, Y. CQDs modulating Z-scheme g-C3N4/BiOBr heterostructure for photocatalytic removing RhB, BPA and TC and E. coli by LED light. J. Alloys Compd. 2022, 895, 162637. [Google Scholar] [CrossRef]



| Samples | C (%) | N (%) | C/N |
|---|---|---|---|
| MCN | 33.93 | 60.97 | 0.56 |
| UCN | 34.73 | 57.32 | 0.61 |
| Samples | BET Surface Area (m2·g−1) | Pore Volume (cm3·g−1) | Average Pore Size (nm) |
|---|---|---|---|
| MCN | 7.4408 | 0.002278 | 20.4142 |
| UCN | 60.0322 | 0.005151 | 17.6493 |
| Photocatalyst (Dosages) | RhB (mg/L) | Time (min) | Rate (%) | Light Source | Refs. |
|---|---|---|---|---|---|
| g-C3N4 (50 mg) | 10 20 30 | 20 | 99.36 99.39 99.35 | 300 W Xe lamp | This paper |
| g-C3N4 (50 mg) | 10 20 30 | 20 | 100 98.78 99.15 | Direct sunlight | This paper |
| ZnO-g-C3N4 (40%)-Go (15%) (15 mg) | 10 | 100 | 98 | 350 W Xe lamp | [63] |
| g-C3N4/Bi4O5Br2-75 (50 mg) | 10 | 10 | 92 | 72 W LED lamp | [64] |
| TlSnI3/g-C3N4 (100 mg) | 5 | 120 | 84.60 | 150 W Osram bulb | [65] |
| TiO2/g-C3N4 (50 mg) | 10 | 150 | 99.30 | 350 W Xe lamp | [66] |
| Co@ZnSQDs/g-C3N4/MWCNT (220 mg) | 10 | 75 | 96 | 500 W halogen lamp | [67] |
| g-C3N4/ZnO/Cu2O (50 mg) | 30 | 100 | 91.4 | 500 W halogen lamp | [68] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, M.; Xing, M.; Dong, B.; Zhang, H.; Sun, X.; Li, Q.; Lu, X.; Mo, J.; Zhu, H. Thermal Polymerisation Synthesis of g-C3N4 for Photocatalytic Degradation of Rhodamine B Dye under Natural Sunlight. Water 2023, 15, 2903. https://doi.org/10.3390/w15162903
Zhang M, Xing M, Dong B, Zhang H, Sun X, Li Q, Lu X, Mo J, Zhu H. Thermal Polymerisation Synthesis of g-C3N4 for Photocatalytic Degradation of Rhodamine B Dye under Natural Sunlight. Water. 2023; 15(16):2903. https://doi.org/10.3390/w15162903
Chicago/Turabian StyleZhang, Muxi, Meiyan Xing, Bin Dong, Hongxia Zhang, Xiaojie Sun, Qiuhong Li, Xueshuang Lu, Jingjing Mo, and Hongxiang Zhu. 2023. "Thermal Polymerisation Synthesis of g-C3N4 for Photocatalytic Degradation of Rhodamine B Dye under Natural Sunlight" Water 15, no. 16: 2903. https://doi.org/10.3390/w15162903
APA StyleZhang, M., Xing, M., Dong, B., Zhang, H., Sun, X., Li, Q., Lu, X., Mo, J., & Zhu, H. (2023). Thermal Polymerisation Synthesis of g-C3N4 for Photocatalytic Degradation of Rhodamine B Dye under Natural Sunlight. Water, 15(16), 2903. https://doi.org/10.3390/w15162903






