Evaluation of the Ecological Potential of Microorganisms for Purifying Water with High Iron Content
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description and Isolation of Microorganisms
2.2. Assessment of Individual Level of Microbial Metal Resistance
2.3. Determination of Iron-Oxidizing Activity and Growth Indicators of Microorganisms
2.4. Identification of the Bacterial Isolates
2.5. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Su, C.; Jiang, L.; Zhang, W. A review on heavy metal contamination in the soil worldwide: Situation, impact and remediation techniques. Environ. Skept. Crit. 2014, 3, 24–38. [Google Scholar]
- Riabova, E.G. Content of heavy metals in urban surface water bodies. Teor. Prikl. Ekol. 2019, 1, 36–40. [Google Scholar] [CrossRef]
- Kovalevskaya, N.P. Biogeochemical potential of microbiological barriers in water quality formation in Kizel coal field rivers. Fundam. Res. 2015, 2, 4203–4208. (In Russian) [Google Scholar]
- Savichev, O.G. Background concentration of substances in river waters of the taiga zone of Western Siberia. Tomsk. State Univ. Bull. 2010, 334, 169–175. (In Russian) [Google Scholar]
- Ministry of Health and Social Development of the Russian Federation. SanPiN 2.1.4.1074-01. Drinking Water. Hygienic Requirements for Water Quality of Centralized Drinking Water Supply Systems. Quality Control; Ministry of Health and Social Development of the Russian Federation: Moscow, Russia, 2001. (In Russian) [Google Scholar]
- Pietrangelo, A. Mechanism of Iron Toxicity. In Iron Chelation Therapy; Hershko, C., Ed.; Springer: Boston, MA, USA, 2002; pp. 19–43. [Google Scholar]
- Islam, M.F.; Islam, M.S.; Ahmed, S.; Miah, M.Y. Removal of chromium from effluent of tannery industries in Bangladesh using rice husks as natural adsorbent. Soil Environ. 2020, 39, 165–172. [Google Scholar] [CrossRef]
- Ou, M.-Y.; Ting, Y.; Ch’Ng, B.-L.; Chen, C.; Cheng, Y.-H.; Chang, T.-C.; Hsi, H.-C. Using Mixed Active Capping to Remediate Multiple Potential Toxic Metal Contaminated Sediment for Reducing Environmental Risk. Water 2020, 12, 1886. [Google Scholar] [CrossRef]
- Ahalya, N.; Ramachandran, T.V.; Kanamadi, R.D. Biosorption of Heavy Metals. Res. J. Chem. Environ. 2003, 7, 71–79. [Google Scholar]
- Kumar, V.; Singh, S.; Kashyap, N.; Singla, S.; Bhadrecha, P.; Kaur, P.; Datta, S.; Kalia, A.; Singh, J. Bioremediation of heavy metals by employing resistant microbial isolates from agricultural soil irrigated with Industrial waste water. Orient. J. Chem. 2015, 31, 357–361. [Google Scholar] [CrossRef]
- Dixit, R.; Malaviya, D.; Pandiyan, K.; Singh, U.B.; Sahu, A.; Shukla, R.; Singh, B.P.; Rai, J.P.; Sharma, P.K.; Lade, H.; et al. Bioremediation of Heavy Metals from Soil and Aquatic Environment: An Overview of Principles and Criteria of Fundamental Processes. Sustainability 2015, 7, 2189–2212. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C. Biosorption of heavy metals by Saccharomyces cerevisiae: A review. Biotechnol. Adv. 2006, 24, 427–451. [Google Scholar] [CrossRef]
- Mosa, K.A.; Saadoun, I.; Kumar, K.; Helmy, M.; Dhankher, O.P. Potential Biotechnological Strategies for the Cleanup of Heavy Metals and Metalloids. Front. Plant Sci. 2016, 7, 303. [Google Scholar] [CrossRef] [PubMed]
- Virpiranta, H.; Banasik, M.; Taskila, S.; Leiviskä, T.; Halttu, M.; Sotaniemi, V.-H.; Tanskanen, J. Isolation of Efficient Metal-Binding Bacteria from Boreal Peat Soils and Development of Microbial Biosorbents for Improved Nickel Scavenging. Water 2020, 12, 2000. [Google Scholar] [CrossRef]
- Fortin, D.; Langley, S. Formation and occurrence of biogenic iron-rich minerals. Earth-Sci. Rev. 2005, 72, 1–19. [Google Scholar] [CrossRef]
- Rawlings, D.E. Characteristics and adaptability of iron- and sulfur-oxidizing microorganisms used for the recovery of metals from minerals and their concentrates. Microb. Cell Factories 2005, 4, 13. [Google Scholar] [CrossRef] [PubMed]
- Bellenberg, S.; Barthen, R.; Boretska, M.; Zhang, R.; Sand, W.; Vera, M. Manipulation of pyrite colonization and leaching by iron-oxidizing Acidithiobacillus species. Appl. Microbiol. Biotechnol. 2014, 99, 1435–1449. [Google Scholar] [CrossRef] [PubMed]
- McLean, R.J.C.; Fortin, D.; Brown, D.A. Microbial metal-binding mechanisms and their relation to nuclear waste disposal. Can. J. Microbiol. 1996, 42, 392–400. [Google Scholar] [CrossRef]
- Hedrich, S.; Schlömann, M.; Johnson, D.B. The iron-oxidizing proteobacteria. Microbiology 2011, 157, 1551–1564. [Google Scholar] [CrossRef]
- Dubinina, G.A.; Sorokina, A.Y. Neutrophilic Lithotrophic Iron-Oxidizing Prokaryotes and Their Role in the Biogeochemical Processes of the Iron Cycle. Microbiology 2014, 83, 127–142. (In Russian) [Google Scholar] [CrossRef]
- Neubauer, S.C.; Emerson, D.; Megonigal, J.P. Life at the Energetic Edge: Kinetics of Circumneutral Iron Oxidation by Lithotrophic Iron-Oxidizing Bacteria Isolated from the Wetland-Plant Rhizosphere. Appl. Environ. Microbiol. 2002, 68, 3988–3995. [Google Scholar] [CrossRef]
- Pleshakova, Y.V.; Reshetnikov, M.V.; Ngun, C.T.; Shuvalova, Y.P. Microbiological and Biochemical Indication of Soils of the City Mednogorsk. Agric. Chem. 2016, 1, 66–73. (In Russian) [Google Scholar]
- Zhang, C.; Qiao, Q.; Piper, J.D.; Huang, B. Assessment of heavy metal pollution from a Fe-smelting plant in urban river sediments using environmental magnetic and geochemical methods. Environ. Pollut. 2011, 159, 3057–3070. [Google Scholar] [CrossRef]
- Granina, L.Z.; Parfenova, V.V.; Zemskaya, T.I.; Zakharova, Y.R.; Golobokova, L.P. On Iron and Manganese Oxidizing Microorganisms in Sedimentary Redox Cycling in Lake Baikal. Berl. Palaobiol. Abh. 2003, 4, 121–128. [Google Scholar]
- Summers, A.O.; Silver, S. Mercury Resistance in a Plasmid-Bearing Strain of Escherichia coli. J. Bacteriol. 1972, 112, 1228–1236. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.; Ahmad, M. Seasonal variation in bacterial flora of the wastewater and soil in the vicinity of industrial area. Environ. Monit. Assess. 2002, 73, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Pirt, S.J. Principles of Microbe and Cell Cultivation; Blackwell Scientific Publications: London, UK, 1975. [Google Scholar]
- GOST 4011-72. Drinking Water. Methods for Measuring the Mass Concentration of Total Iron, Moscow, Russia, 1974. (In Russian). Available online: http://docs.cntd.ru/document/1200008210 (accessed on 24 March 2021).
- Holt, J.G.; Krieg, N.R.; Sneath, P.H.A. (Eds.) Bergey’s Manual of Determinative Bacteriology; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 1993; Translated under the Title Opredelitel’ Bakterii Berdzhi; Mir: Moscow, Russia, 1997. [Google Scholar]
- Okpokwasili, G.C.; Nweke, C.O. Microbial growth and substrate utilization kinetics. Afr. J. Biotechnol. 2005, 5, 305–317. [Google Scholar]
- Allen, D.A.; Austin, B.; Colwell, R.R. Numerical Taxonomy of Bacterial Isolates Associated with a Freshwater Fishery. Microbiology 1983, 129, 2043–2062. [Google Scholar] [CrossRef][Green Version]
- Singh, Y.; Lal, N. Investigations on the heavy metal resistant bacterial isolates in vitro from industrial effluents. WJPPS 2015, 4, 343–350. [Google Scholar]
- Yu, Q.; Rao, R.; Xu, X.; Ganji, S.; Jacob, M.R.; Yu, B.; Li, X. Isolation and synthesis of novel antimicrobial chromene derivatives from Psorothamnus fremontii. Planta Med. 2015, 81. [Google Scholar] [CrossRef]
- Mishra, R.R.; Prajapati, S.; Das, J.; Dangar, T.K.; Das, N.; Thatoi, H. Reduction of selenite to red elemental selenium by moderately halotolerant Bacillus megaterium strains isolated from Bhitarkanika mangrove soil and characterization of reduced product. Chemosphere 2011, 84, 1231–1237. [Google Scholar] [CrossRef]
- Salehizadeh, H.; Shojaosadati, S. Removal of metal ions from aqueous solution by polysaccharide produced from Bacillus firmus. Water Res. 2003, 37, 4231–4235. [Google Scholar] [CrossRef]
- Kalaimurugan, D.; Balamuralikrishnan, B.; Durairaj, K.; Vasudhevan, P.; Shivakumar, M.S.; Kaul, T.; Chang, S.W.; Ravindran, B.; Venkatesan, S. Isolation and characterization of heavy-metal-resistant bacteria and their applications in environmental bioremediation. Int. J. Environ. Sci. Technol. 2020, 17, 1455–1462. [Google Scholar] [CrossRef]
- Pishchik, V.N.; Vorob’Ev, N.I.; Provorov, N.A.; Khomyakov, Y.V. Mechanisms of plant and microbial adaptation to heavy metals in plant–microbial systems. Microbiology 2016, 85, 257–271. [Google Scholar] [CrossRef]
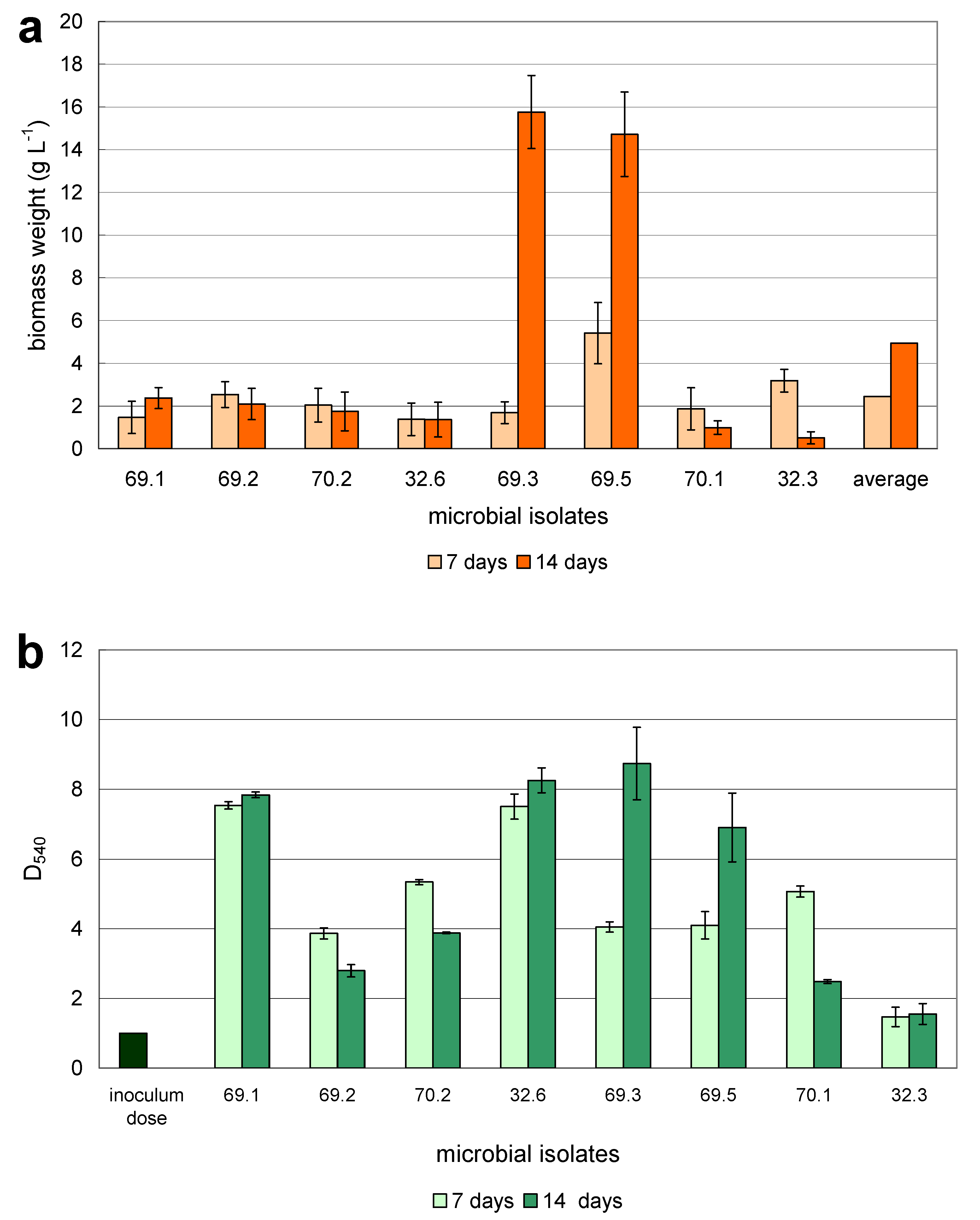
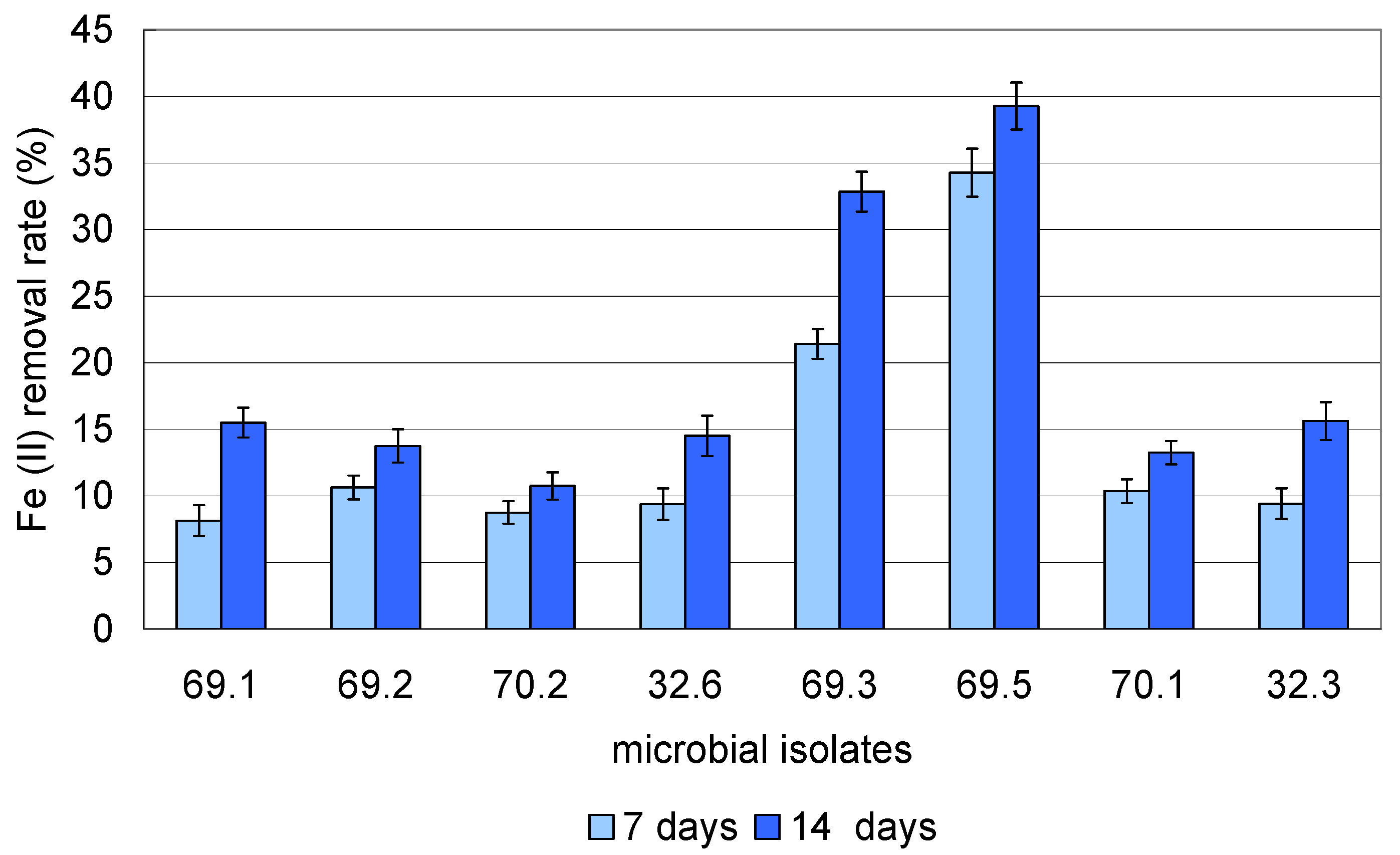
| Isolate No. | Specific Growth Rate, h−1 Based on the Method: | |||
|---|---|---|---|---|
| Weight Method | Photometric Method | |||
| 7 days | 14 days | 7 days | 14 days | |
| 69.1 | 0.0023 | −0.0006 | 0.0120 | −0.0071 |
| 69.2 | 0.0055 | DP | 0.0080 | DP |
| 70.2 | 0.0042 | DP | 0.0100 | DP |
| 32.6 | 0.0019 | DP | 0.0120 | −0.0017 |
| 69.3 | 0.0031 | 0.0157 | 0.0083 | 0.0092 |
| 69.5 | 0.0100 | 0.0133 | 0.0084 | 0.0061 |
| 70.1 | 0.0037 | DP | 0.0096 | DP |
| 32.3 | 0.0069 | DP | 0.0023 | DP |
| Characteristic | Bacteria | |
|---|---|---|
| 69.3 | 69.5 | |
| Motility | Motile | Motile |
| Characteristics of Colony | Circular, opaque, cream, with a slightly convex smooth shiny surface and uneven edges, Ø ≈ 2 mm | Circular, opaque, cream, with a slightly convex smooth shiny surface and uneven edges, Ø ≈ 1 mm |
| Shape of Bacterium | Rod, arranged singly and in pairs | Rod, arranged singly and in pairs |
| Gram Stain | + | + |
| Spore Characteristics | Elliptical, central position | Elliptical, central position |
| Growth at 42 °C | − | − |
| Growth at 10 °C | ± | − |
| Acid-resistant | − | − |
| Oxidase | − | − |
| Catalase | + | + |
| Lecithinase | + | − |
| Lipase | − | − |
| Urease | − | + |
| Arginine Dihydrolase | − | − |
| Phenylalanine Deaminase | − | + |
| Lysine Decarboxylase | − | − |
| Ornithine Decarboxylase | − | − |
| Nitrate Reductase | + | − |
| Gelatinase | + | + |
| Starch Hydrolysis | + | + |
| Casein Hydrolysis | + | ± |
| Indole Production | − | − |
| Hydrogen Sulfide Production | − | + |
| Ammonia Production | − | − |
| Citrate on Simmons Medium Utilization | − | − |
| Fermentation and Acid and Gas Production from Glucose, Lactose, Sucrose, Fructose, Maltose, Mannitol | + | + |
| Fermentation Arabinose | + | + |
| Acid and Gas Production from Xylose, Arabinose | − | + |
| Acid and Gas Production from Xylose | − | + |
| Vogues-Proskauer Test | − | − |
| Genus | Bacillus | Bacillus |
| Species | megaterium | megaterium |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pleshakova, E.; Ngun, C.; Reshetnikov, M.; Larionov, M.V. Evaluation of the Ecological Potential of Microorganisms for Purifying Water with High Iron Content. Water 2021, 13, 901. https://doi.org/10.3390/w13070901
Pleshakova E, Ngun C, Reshetnikov M, Larionov MV. Evaluation of the Ecological Potential of Microorganisms for Purifying Water with High Iron Content. Water. 2021; 13(7):901. https://doi.org/10.3390/w13070901
Chicago/Turabian StylePleshakova, Ekaterina, Clement Ngun, Mikhail Reshetnikov, and Maxim Viktorovich Larionov. 2021. "Evaluation of the Ecological Potential of Microorganisms for Purifying Water with High Iron Content" Water 13, no. 7: 901. https://doi.org/10.3390/w13070901
APA StylePleshakova, E., Ngun, C., Reshetnikov, M., & Larionov, M. V. (2021). Evaluation of the Ecological Potential of Microorganisms for Purifying Water with High Iron Content. Water, 13(7), 901. https://doi.org/10.3390/w13070901







