Abstract
The use of polyhydroxyalkanoates (PHA) as slow-release electron donors for environmental remediation represents a novel and appealing application that is attracting considerable attention in the scientific community. In this context, here, the fermentation pattern of different types of PHA-based materials has been investigated in batch and continuous-flow experiments. Along with commercially available materials, produced from axenic microbial cultures, PHA produced at pilot scale by mixed microbial cultures (MMC) using waste feedstock have been also tested. As a main finding, a rapid onset of volatile fatty acids (VFA) production was observed with a low-purity MMC-deriving material, consisting of microbial cells containing 56% (on weight basis) of intracellular PHA. Indeed, with this material a sustained, long-term production of organic acids (i.e., acetic, propionic, and butyric acids) was observed. In addition, the obtained yield of conversion into acids (up to 70% gVFA/gPHA) was higher than that obtained with the other tested materials, made of extracted and purified PHA. These results clearly suggest the possibility to directly use the PHA-rich cells deriving from the MMC production process, with no need of extraction and purification procedures, as a sustainable and effective carbon source bringing remarkable advantages from an economic and environmental point of view.
1. Introduction
Polyhydroxyalkanoates (PHA) are biologically synthesized polyesters that are completely biodegradable in the environment (under both aerobic and anaerobic conditions) and can also be bio-based, i.e., derived from renewable resources. Therefore, they are considered three-time bio-polymers [1,2]. In particular, PHA are not a single polymer, but a family of copolymers featuring a wide array of physical and mechanical properties which depend on the length and composition of the side chains. Among them, the polyhydroxybutyrate (PHB) homopolymer is largely studied and well characterized and the copolymer poly(hydroxybutyrate-hydroxyvalerate) (PHBV) is particularly interesting because it has properties similar to polypropylene. Besides the 3-hydroxybutyrate (3HB) and 3-hydroxyvalerate (3HV) monomers, other constituents, such as the 3-hydroxyhexanoate (3HH), can be englobed in the PHA structure [3]. As a consequence, PHA can be used for a broad portfolio of applications in numerous fields, including the production of packaging materials, disposable utensils, cosmetic containers, as well as medical applications as drug delivery carriers and environmental applications as a source for long-term release of carbon to drive specific reductive reactions [4,5].
However, at present, a broad commercialization of PHA is still limited by their high production costs, mainly linked to the use of pure cultures of selected microorganisms. Therefore, in recent years, more sustainable processes based on the use of mixed microbial cultures (MMC) are being extensively studied, also at pilot scale [6,7]. The use of MMC is a viable strategy to obtain PHA from a wide range of inexpensive feedstock (comprising industrial, agricultural, and urban wastes) by means of a multi-stage process, which allows reducing production costs and in turn contributing to boost a rapid increase in their market in the near future [8,9]. When dealing with MMC, the final PHA composition depends on the operating conditions applied to each stage of the process as well as on the feedstock nature, and it can be ultimately fine-tuned in order to obtain materials with tailored characteristics for specific applications [10,11]. Overall, the reduction of production costs is advantageous also for environmental bioremediation applications, whereby a defined PHA composition is not demanded.
In this context, PHA can be used as sustainable and fully biodegradable materials for long-term organic carbon release to stimulate reductive biological transformations, such as the reductive dechlorination of chlorinated hydrocarbons or the removal of nitrate or other oxidized compounds from contaminated groundwater [12,13]. Potential environmental application scenarios for such materials include the direct introduction in the contaminated aquifer within, for instance, a permeable reactive barrier (in situ application) or the use as a filling material in ex situ bioreactors, and it has been demonstrated that both the nature and availability of carbon sources impact the degradation activity [14,15]. In particular, the use of PHA in environmental applications has already been investigated with main reference to the treatment of chlorinated solvents [16,17]. The latter are among the most frequently encountered groundwater contaminants, which may cause a variety of health and environmental problems due to their prevalent use in industry, recalcitrance in the environment, and toxicity. However, in groundwater ecosystems indigenous microorganisms can effectively reduce the concentration of chlorinated compounds through a biological process often referred to as biological reductive dechlorination (BRD). This is an anaerobic reaction whereby higher chlorinated ethenes or ethanes are transformed, in a stepwise manner, to less chlorinated or eventually harmless non-chlorinated end products such as ethene or ethane [18,19]. In the BRD process, the chlorinated compounds serve as respiratory electron acceptors, while molecular hydrogen or acetate serve as electron donors in the metabolism of anaerobic dechlorinating bacteria [20]. Previous studies have shown that the lack of suitable electron donors is typically a limiting factor in the bioremediation of chlorinated hydrocarbons, in some cases leading to the incomplete reductive dechlorination and, in turn, in the accumulation of lower chlorinated intermediates (e.g., vinyl chloride), which are more toxic than the parent contaminants [21,22]. To overcome this drawback, enhanced processes for in situ anaerobic bioremediation of chlorinated solvents have been developed involving the injection of suitable compounds which provide a sustainable and long-lasting source of electron donors to support reductive dechlorination processes [23,24,25]. In this regard, the attention towards PHA application to sustain in situ BRD processes is recently increasing due to the above-described attractive properties of this material and also because under anaerobic conditions PHA are slowly fermented into volatile fatty acids and H2, thereby resulting in a steady and efficient release of the required electron donors [17]. In particular, the viability of this strategy has been previously verified in research studies conducted at both laboratory and pilot-scale, by using commercial PHA derived from pure cultures [26,27,28].
In view of a perspective advancement in the application of the technology, the main objective of this study was the assessment of the fermentability potential of several PHA-based materials, derived from both pure and mixed microbial cultures and having a distinct composition and purity. The obtained results have been deeply analyzed in order to compare the different materials and identify those showing the greatest potential as slow-release electron donors for environmental applications. Importantly, the MMC-PHA materials used in this study have been produced from urban and agricultural wastes as feedstock and this offers a relevant opportunity to convert a waste into biodegradable inexpensive materials to be exploited for bioremediation applications, hence perfectly fitting with the concept of circular bioeconomy.
2. Materials and Methods
2.1. Experimental Setup
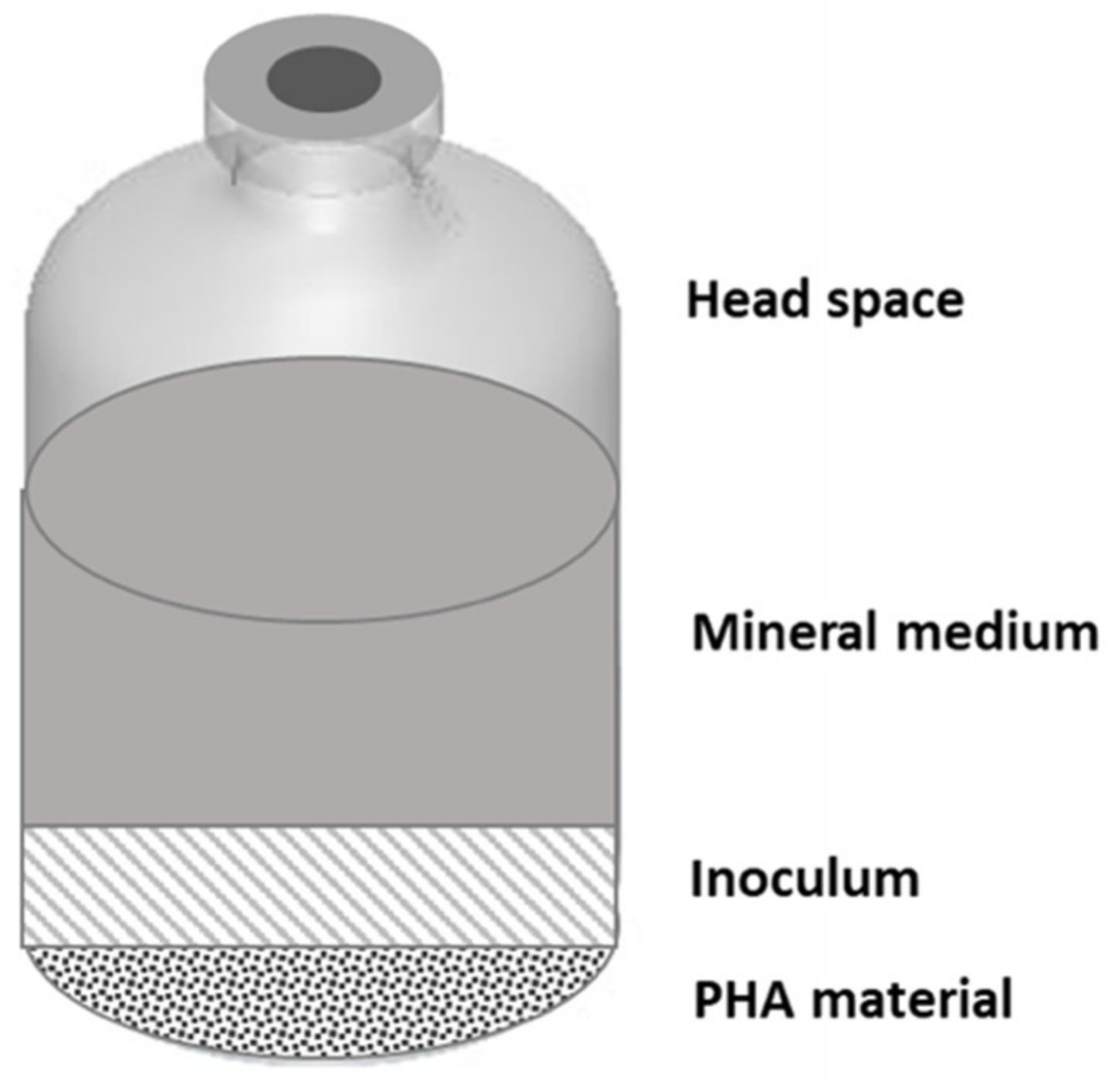
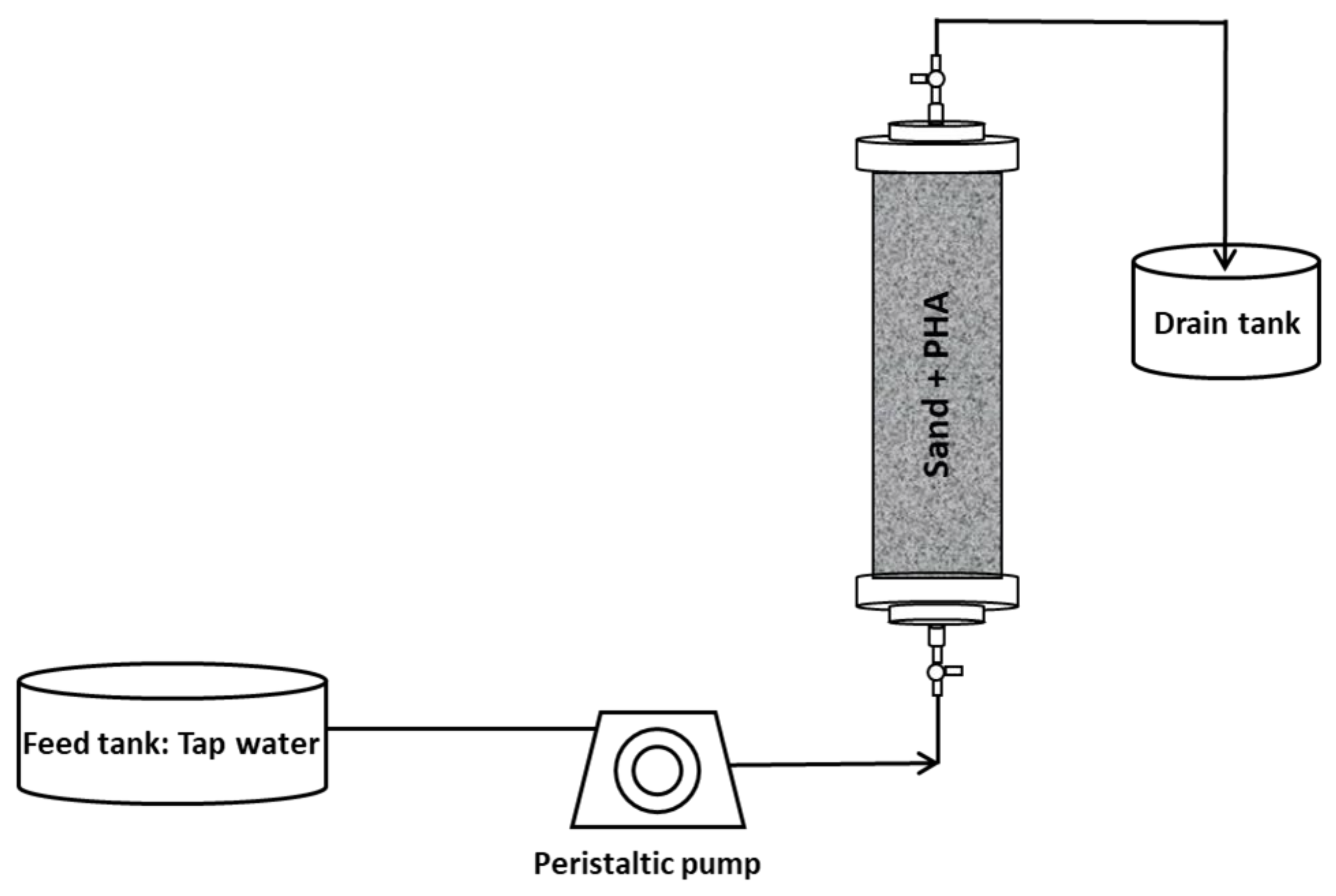
In this study, two experimental setups have been used with the aim to study the anaerobic fermentability of different types of PHA, consisting of mixed microbial cultures (MMC)-based and commercially available (with pure culture origin) materials, both in batch and continuous mode operation. The two setups are represented in Figure 1 and Figure 2.
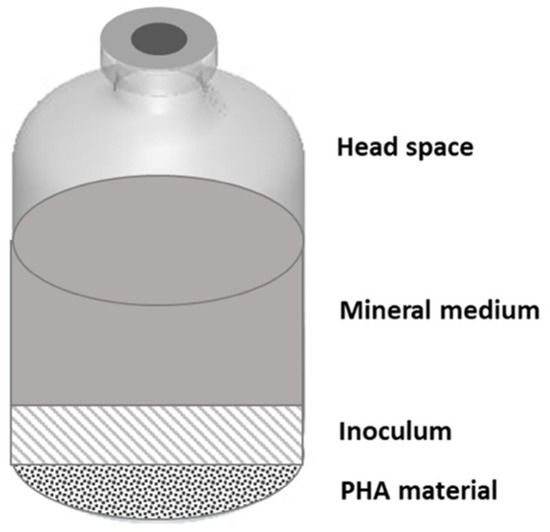
Figure 1.
Experimental setup used for batch experiments with different types of polyhydroxyalkanoates (PHA) materials.
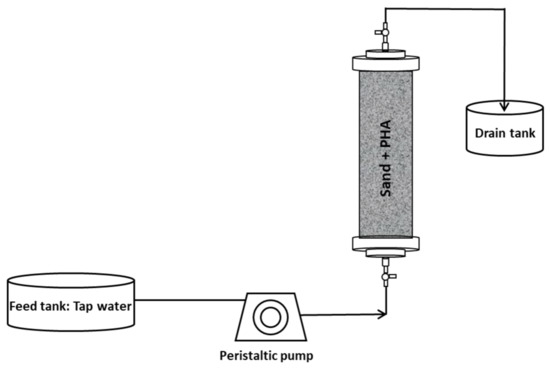
Figure 2.
Experimental setup used for continuous experiments.
2.1.1. Batch Tests
Preliminary batch tests have been carried out in serum bottles by using two MMC-based and three commercial PHA materials. More in detail, MMC-PHA were produced through a multi-stage pilot scale process located at Treviso (northeast of Italy) which used the organic fraction of municipal solid waste (OFMSW) and sewage sludge (SS) as feedstock, as described elsewhere [6]. The PHA-rich biomass directly deriving from the accumulation step of the process is hereafter referred to as “MMC-Raw PHA.” In detail, the microbial suspension was settled by gravity, then centrifuged, and lastly, the pellet was thermally treated (at 145 °C for 15 min and then dried overnight at 60 °C). Finally, the MMC-Raw PHA consisted of a material containing 56% (w/w) of a Poly(3-hydroxybutyrate-co-3-hydroxyvalerate), PHBV, biopolymer with 18% (w/w) of HV monomer. In contrast, the material subjected to purification treatments following the PHA accumulation step is referred to as MMC-Extracted PHA1. In particular, in this case, the PHA-rich microbial cells after thermal stabilization (as previously described) were suspended in an NaClO solution (1.5% active Cl2), stirred, centrifuged, and finally washed three times with distilled water in order to avoid any possible toxicity effect on microbial activity due to NaClO. This procedure allowed to disrupt and eliminate the cellular debris, resulting in a high purity material consisting of 97% (w/w) of PHBV copolymer with 18% (w/w) of HV content. As for the commercial materials, three types of PHA were considered, differing in compositions and particles size. The PHB homopolymer was in powder form, the PHBV copolymer with 3% (w/w) of HV was also in powder form, whereas the PHBH (3-hydroxybutyrate-co-3-hydroxyhexanoate) copolymer having 14% (w/w) of hydroxyhexanoate (HH) was in pellet.
Batch experiments were set up in 240 mL autoclaved serum bottles (Figure 1). Each bottle was filled with the same amount of PHA material (at a concentration of 2 g L−1), anaerobic mineral medium to enhance microbial growth, and was inoculated with anaerobic sludge deriving from the Treviso municipal wastewater treatment plant (at a final concentration of about 1 gVSS L−1; Volatile Suspended Solids), each reaching a working volume of 160 mL. Prior to adding the inoculum, bottles were flushed with a N2/CO2 (70/30, % v/v) gas mixture to establish anaerobic conditions. The mineral medium composition was made of (g L−1): NH4Cl (0.125), MgCl2·6H2O (0.1), K2HPO4 (4), CaCl2·2H2O (0.05), a trace of metal (10 mL L−1), and vitamin (1 mL L−1) solution [29,30].
Fermentability tests were carried out for each of the five types of PHA materials previously described and, in addition, a control test was prepared under the same operating conditions but in absence of PHA. Each test was prepared in duplicate to evaluate reproducibility, accounting for a total of 12 serum bottles. The serum bottles were sealed by a Teflon-faced gray butyl stopper (Wheaton, Millville, NJ, USA) and crimped by an aluminum cap. At the beginning of the fermentability tests, each serum bottle was flushed with a N2/CO2 (70/30, % v/v) gas mixture for about 20 min in order to maintain anaerobic conditions. The tests were performed at room temperature (20 ± 2 °C).
2.1.2. Column Tests
Continuous flow experiments were performed in order to simulate conditions more closely resembling those occurring in actual environmental applications, whereby, for instance, PHA are employed as slow-release electron donors for groundwater bioremediation. The setup consisted of Plexiglas air-tight columns (length 14 cm; internal diameter 2.5 cm) which were filled with a mixture of non-adsorbent silica sand and PHA materials. Four types of PHA materials were tested.
Two of them (MMC-Raw PHA and commercial PHB) were used in the batch experiments. The MMC-Extracted PHA1 consisted of the PHBV copolymer produced at the Treviso pilot plant and acidified by H2SO4 to pH 2.0–2.5, settled under gravity, and then centrifuged; at the end, the wet pellet was stored at 4 °C. Eventually, the acid-stabilized wet biomass was extracted with a mixture of aqueous phase inorganic reagents. After the extraction, the material was dried obtaining a white powder with a high purity grade (94%, w/w) of PHBV copolymer containing 20% (w/w) of HV monomer. Finally, the MMC-Extracted PHA2 consisted of a material produced at a mixed culture pilot scale located in Lisbon (Portugal) and operated by using fermented fruit waste as feedstock. This material underwent the same stabilization and extraction process with inorganic reagents as previously described, in the end obtaining a PHBV copolymer with a high purity grade (96%, w/w) and an HV content of 30% (w/w).
Each column was packed with 100 g of silica sand and 5 g of PHA, while for the column with MMC-Raw PHA, 10 g of biopolymer material was used with 95 g of sand (as 56% of the material consisted of microbial cells). Each of the four columns was equipped with two equal valves with an output side for sampling in the inlet and outlet of columns (Figure 2).
At the beginning of the experiment, the inlet ports of the columns were fed with tap water, by a multichannel peristaltic pump at a flow rate of 0.2 mL min−1 which corresponded to approximately 3.26 h of residence time, while the outlet ports were connected to a drain tank. The use of tap water, instead of mineral medium, was chosen in order to guarantee experimental conditions more representative of those occurring in real environments, such as contaminated aquifers. After 45 days of operation with tap water, each column was inoculated with anaerobic sludge as in the batch experiments, in order to enhance the fermentation process. After inoculation, the columns were operated with the liquid phase being continuously recirculated to avoid an immediate withdrawal of microorganisms and, after 24 h, the columns were again connected to tap water. The continuous experiments lasted 80 days.
2.2. PHA-Based Materials: Origin and Composition
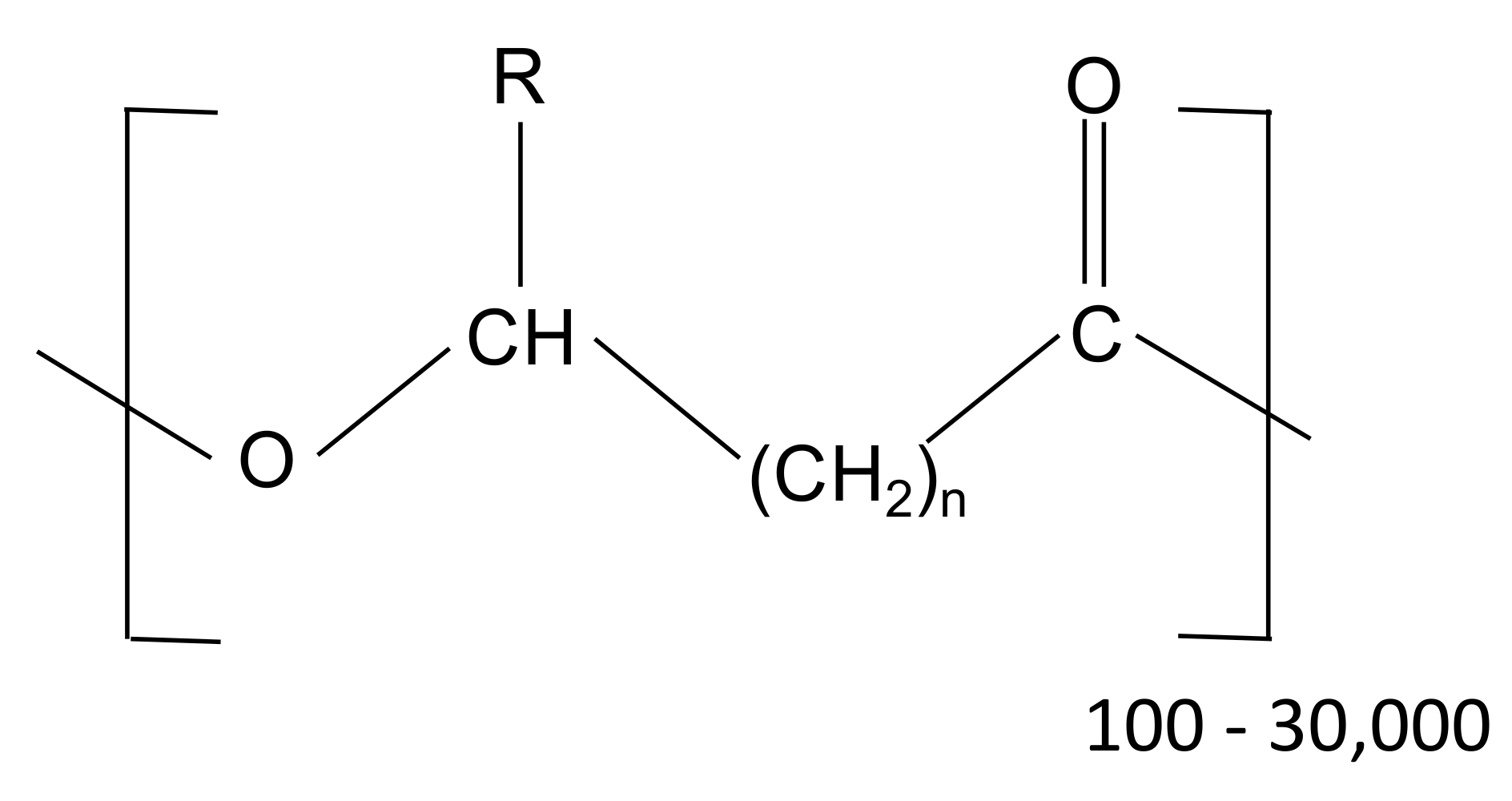
Different PHA-based materials have been used in this study, including commercial materials (produced by axenic cultures) as well as materials produced at pilot scale by using mixed microbial cultures. The general structure of PHA is represented in Figure 3, whereby the number of carbon atoms and the composition of the side chain (R) identify the monomeric unit. As an example, with n = 1, the corresponding monomeric unit is referred to as 3-hydroxybutyrate (3HB), 3-hydroxyvalerate (3HV) or 3-hydroxyhexanoate (3HH), depending on whether the R group is a methyl, ethyl, or propyl, respectively.
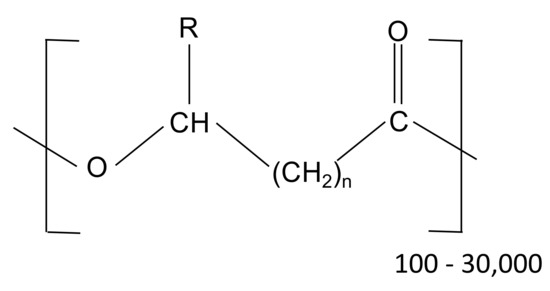
Figure 3.
General chemical structure of PHA (R represents the variable alkyl side chain, n generally varies from 1 to 4).
Here, the three commercially available PHA materials were purchased from three different companies. More in detail, the PHB homopolymer was obtained from Biomer (Schwalbach am Taunus, Germany), the PHBV copolymer (with HV equal to 3%, w/w) from TianAn (Ningbo, China), and the PHBH copolymer (with HH equal to 14%, w/w) from Kaneka (Westerlo-Oevel, Belgium). The latter, differently form the other two materials, was available in pellet form and it was tested as received.
As for the MMC-based materials, these were produced at two pilot scale plants fed with OFMSW and SS or fruit waste and located in Treviso (Italy) or Lisbon (Portugal), respectively. These materials consisted of PHBV copolymers with a different HV content. The exact composition, as well as the purity (i.e., the percentage of PHA content relative to materials dried weight), were determined by means of gas-chromatography analysis, as reported in Valentino et al., 2019 [6] and Lorini et al., 2021 [31].
2.3. Analytical Methods
Liquid and gaseous samples were periodically taken from both batch and continuous experiments in order to monitor the fermentation process in terms of the production of main components. More in detail, acids production was determined by using a Dani Master gas-chromatograph (GC) equipped with a Flame Ionization Detector (FID) and a glass column (2 m × 2 mm) packed with Carbopack B 60/80 mesh. Helium (He) was used as carrier gas at a flow rate of 25 mL min−1, the oven temperature was set at 175 °C and the FID temperature at 200 °C. For each analysis, 1 mL of filtered (a 0.45 μm) sample was acidified with 100 μL of a solution of oxalic acid (0.33 M) and injected (1 μL) into the gas-chromatograph. Liquid samples were also analyzed for pH measurements by using a Crison GLP 22 pH-meter with glass microelectrode, ceramic diaphragm and encapsulated Ag/AgCl reference electrode.
Gas phase analysis from headspace of the serum bottles containing the gaseous products of anaerobic fermentation (CO2, CH4, and H2) and possibly other compounds such as CO and O2 (unwanted products for the anaerobic environment) was performed only for qualitative purposes to determine the composition of the gaseous mixture occupying the headspace. The analysis was carried out by injecting 500 μL of gaseous sample by a gas-tight Hamilton syringe into a “Dani Master” gas-chromatograph (stainless steel column packed with molecular sieve; He carrier gas 18 mL min−1; oven temperature 180 °C; thermal-conductivity detector (TCD) temperature 200 °C).
The concentration of microorganisms in the anaerobic sludge used as inoculum was determined as volatile suspended solids (VSS), according to the standard methods [32].
3. Results and Discussions
3.1. Batch Fermentability Tests
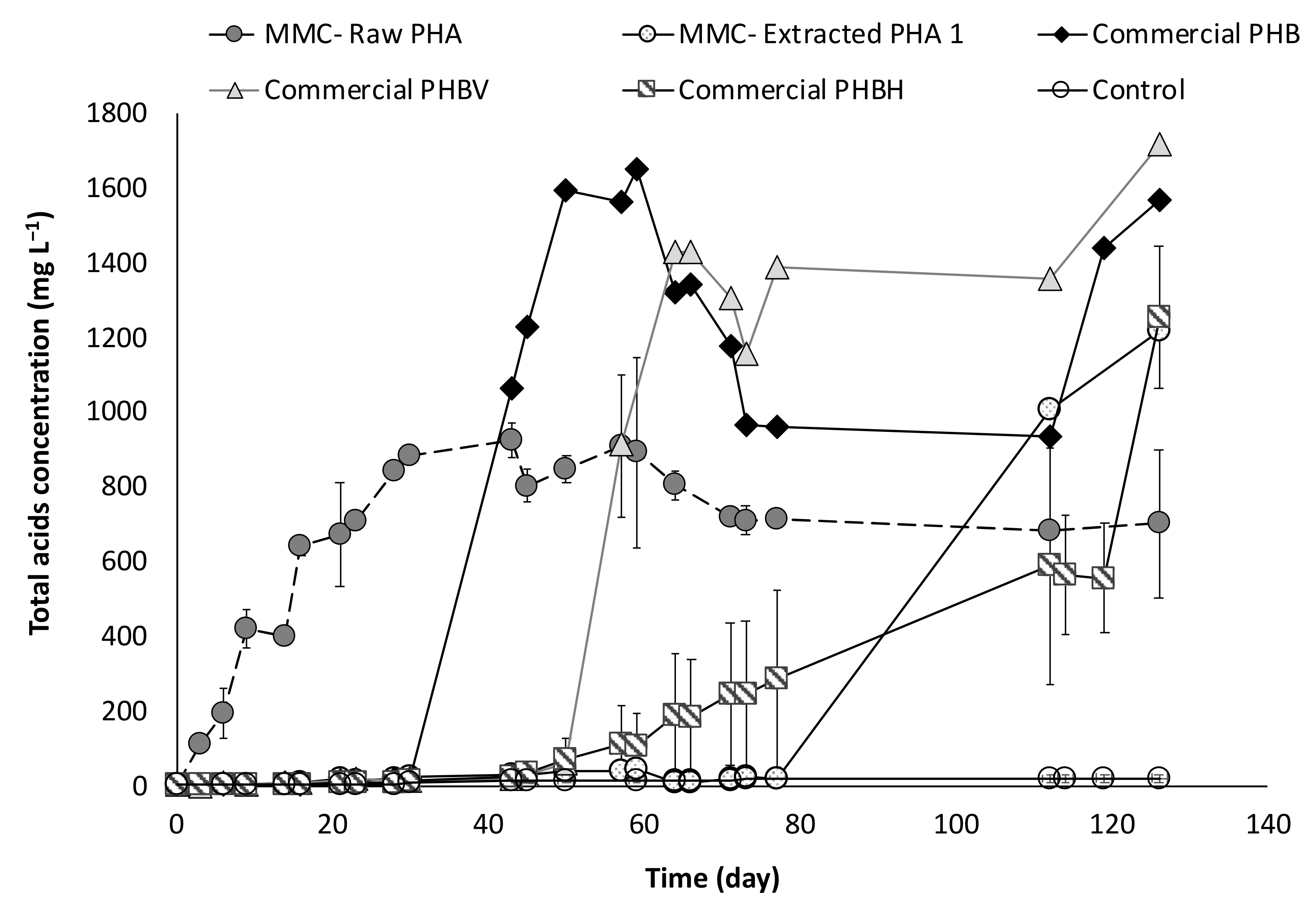
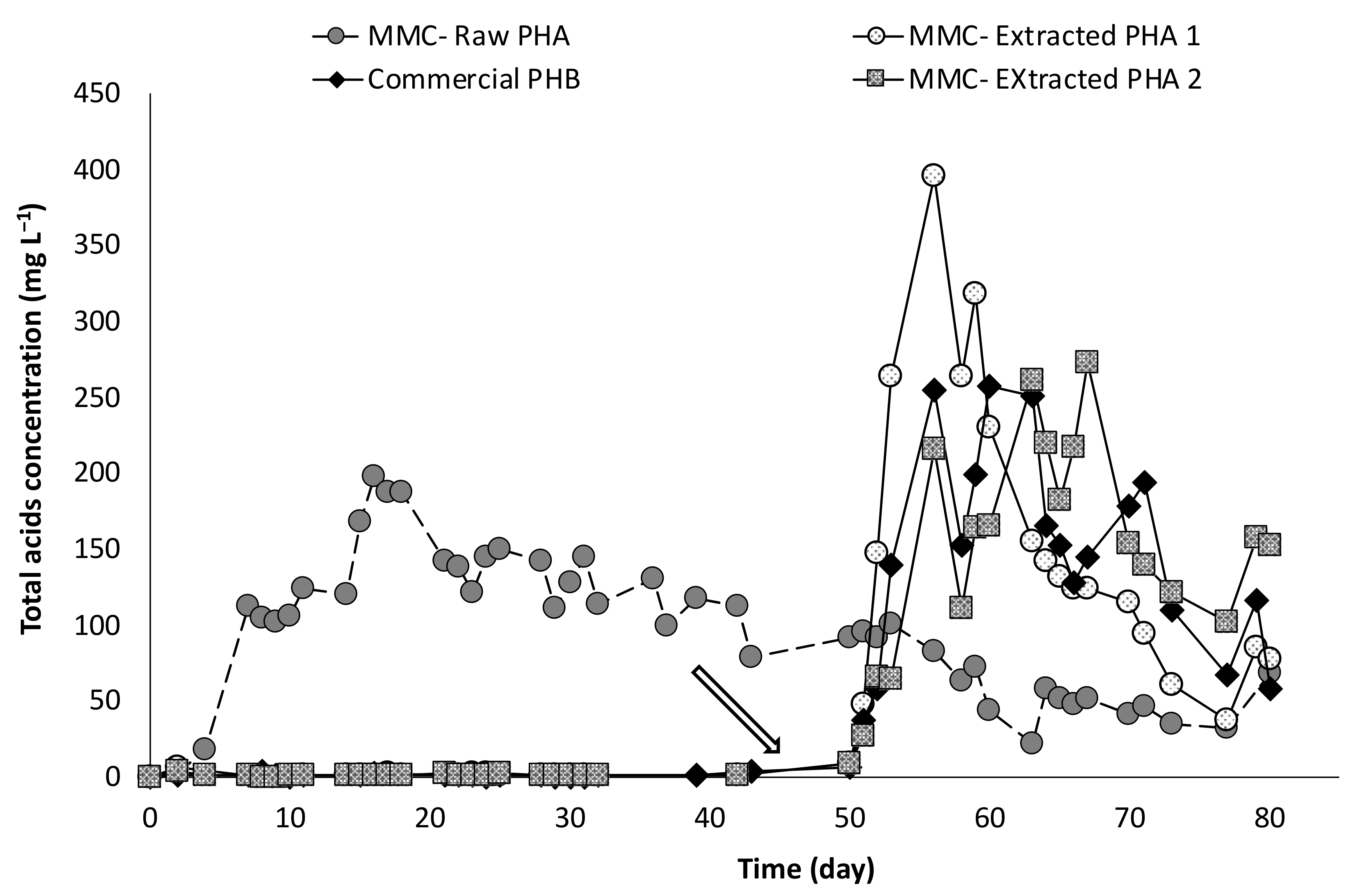
The study of PHA fermentability through batch experiments was carried out with anaerobic sludge as inoculum, in the presence of five types of materials. Specifically, two of them were produced through a multi-stage pilot plant process involving mixed microbial cultures (MMC) and wastes of urban origin as feedstock [6]. The PHA-rich biomass directly deriving from the accumulation stage of the process is hereafter referred to as “MMC-Raw PHA,” while the polymer extracted from the microbial cells after the accumulation stage is referred to as “MMC-Extracted PHA1.” The other three materials consisted of commercial polymers having different composition, and are indicated as “Commercial PHB,” “Commercial PHBV,” and “Commercial PHBH” throughout the manuscript. Finally, a control test, in absence of PHA, was also performed in order to characterize the microbial activity of the anaerobic sludge in terms of endogenous metabolism. Overall, the batch experiments lasted more than 130 days, during which the production of volatile fatty acids (VFA) was regularly monitored, and the obtained trends are reported in Figure 4 as the mean values of the duplicate experiments. Except for the control test, VFA were produced from all the used PHA-based materials.
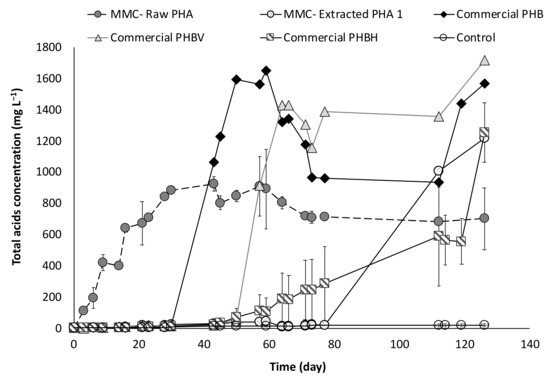
Figure 4.
Concentration of total acids produced during batch fermentability experiments.
In particular, a faster initial production was observed in presence of the MMC-Raw PHA, whereby VFA were detected just after three days from the beginning of the experiments and this material was the only one to be fermented during the first 30 days of operation. The maximum acids concentration was rapidly reached (at around day 28) and accounted for about 843 ± 5 mg/L, with the production stabilizing at a slightly lower value during the remainder period of the experiment. A different trend was observed with the other PHA material originating from mixed cultures. Indeed, with the MMC-Extracted PHA1, the acids production initiated only after a long lag-phase of about 80 days and reached a concentration of approximately 1200 mg/L, in correspondence to the end of the experiment.
The reason of this discrepancy between the two MMC-PHA-based materials is likely related to the different treatment they underwent after production, which also resulted in a different polymer content. In the case of Raw-PHA, no extraction procedure was performed after the accumulation stage of the PHA production process and the material consisted of microbial cells with an intracellular polymer content of about 56% (w/w). As for the MMC-Extracted PHA, the microbial cells deriving from the accumulation stage were treated with an NaClO solution in order to have a material with a higher purity grade in which the PHA content reached a value of approximately 97% (w/w). In both cases, the PHA consisted of a PHBV copolymer with an HV content of about 18% (w/w). Therefore, by also considering that the same inoculum was used in all batch experiments, the more rapid onset of the fermentation process in presence of Raw-PHA can possibly be due to the co-existence of microbial cells (44%, w/w) along with the polymer in this material. In other words, this cellular fraction could release upon lysis micro- and macronutrients (e.g., trace elements and organic cofactors) able to stimulate the anaerobic metabolism and, in turn, to shorten the lag-phase characterizing the fermentative processes. Nonetheless, it cannot be excluded that the production of VFA also resulted from the fermentation of soluble microbial products released during the lysis of microbial cells accompanying the PHA-based material. Taken as a whole, these findings are in very good agreement with those reported in a previous study, whereby aerobically grown biomass rapidly stimulated the reductive dechlorination process with negligible lag-phase [33].
By using the obtained results, it is possible to calculate the conversion yield, which represents the percentage of PHA-based material that is converted into VFA. With reference to the Raw-PHA, a value of 37% (w/w) was reached just after around 30 days from the beginning of the experiments, when no acids were detected in the other conditions, and this value slightly increased to 39% (w/w) only in correspondence to day 43. Notably, if the yield is strictly calculated referring to the real amount of PHA in the Raw material, the value is significantly higher and corresponds to about 70% (w/w). The conversion yield in the presence of the Extracted-PHA reached its maximum value (39%, w/w) on day 126, and therefore, at the end of the test, and this value is almost unchanged (40%, w/w) if referred to the effective amount of PHA contained in the material. With reference to the commercial materials, two of them showed a similar trend with a different temporally onset in the VFA production, which is likely correlated to the different composition of the used materials. Indeed, the commercial material consisting of a PHB monomer started to be fermented after about 30 days from the beginning of the experiments with the acids concentration rapidly reaching a value as high as 1600 mg/L at day 59, which corresponds to a conversion yield into VFA of more than 60%. When the commercial material made of PHBV copolymer was used, the production of VFA started later, in correspondence of day 50 of operation and quickly reached a value of about 1440 mg/L (on day 64), which represents a yield of the material conversion into acids of approximately 55%. Differently, in presence of the commercial PHBH material, a very slow and gradual fermentation process was observed which started on day 40 and showed a clear surge in the acid production after 120 days of operation. As a result, a lower fraction of the material was fermented into acids with a yield of 40% at the end of the test. Besides the difference in PHA composition, this could be attributed to the fact that the commercial PHB and commercial PHBV were used in the form of fine powder, whereas the PHBH material was utilized in the form of granular pellet, thereby offering a lower surface area for microbial degradation with respect to the other materials. Along with acids measurements, pH was also regularly monitored in each test, and a strict correlation between these two parameters was noted (Figure 5).
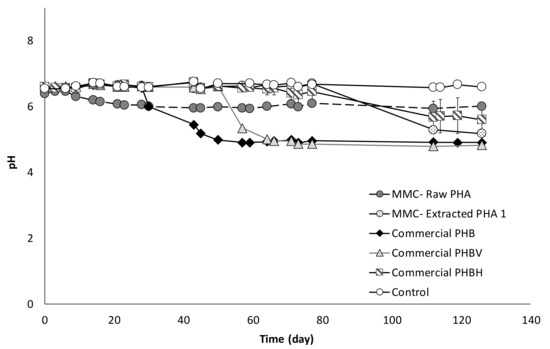
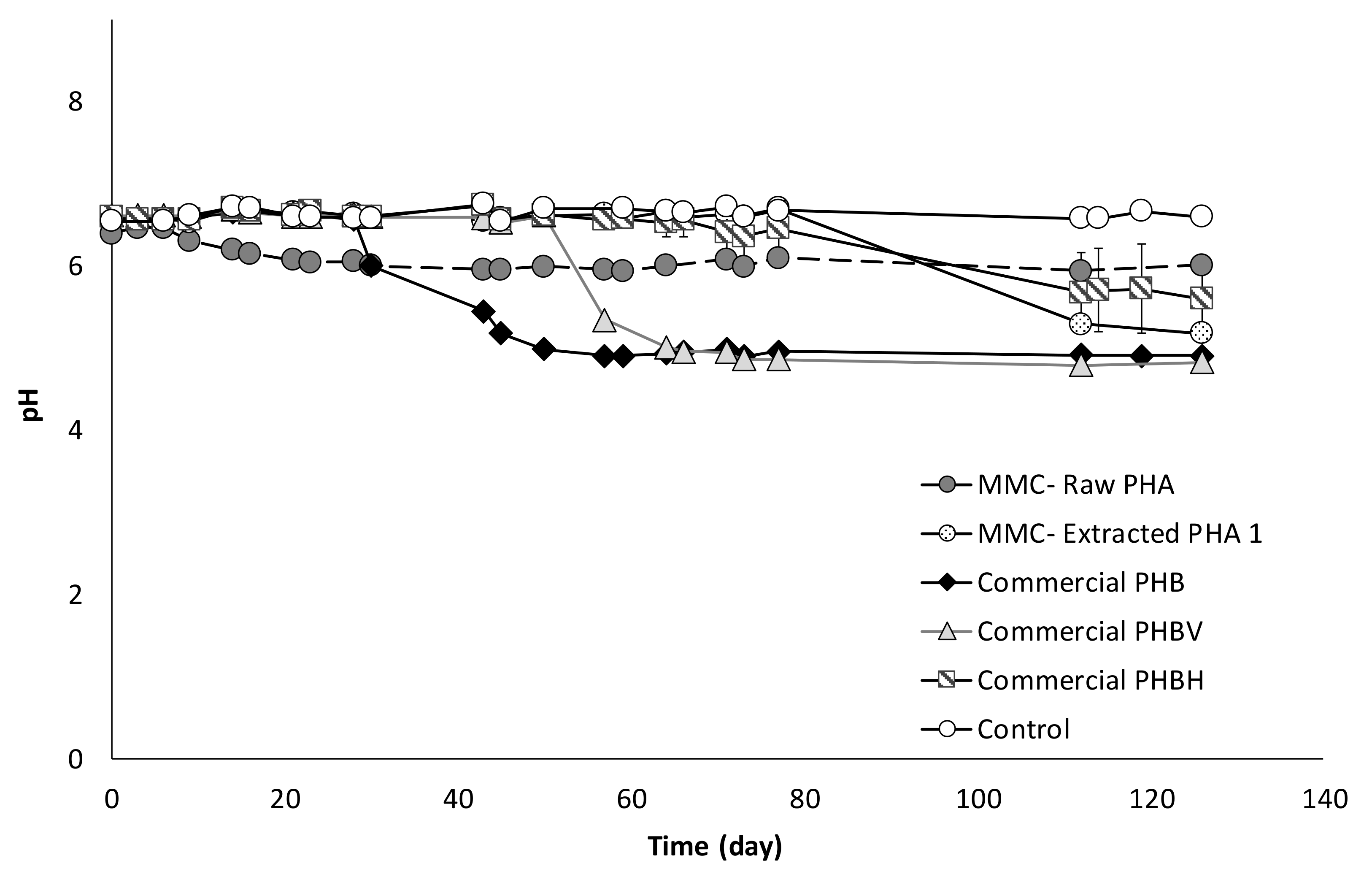
Figure 5.
Trend of pH in all batch experiments.
Indeed, the pH was initially the same in all experiments and the slightly acid value (circa 6.5) was due to the use of a CO2-containing gas mixture to establish anaerobic conditions. However, during the tests, changes in pH values were observed, whereby a lower value of pH corresponded to a higher VFA production, as shown in Figure 5.
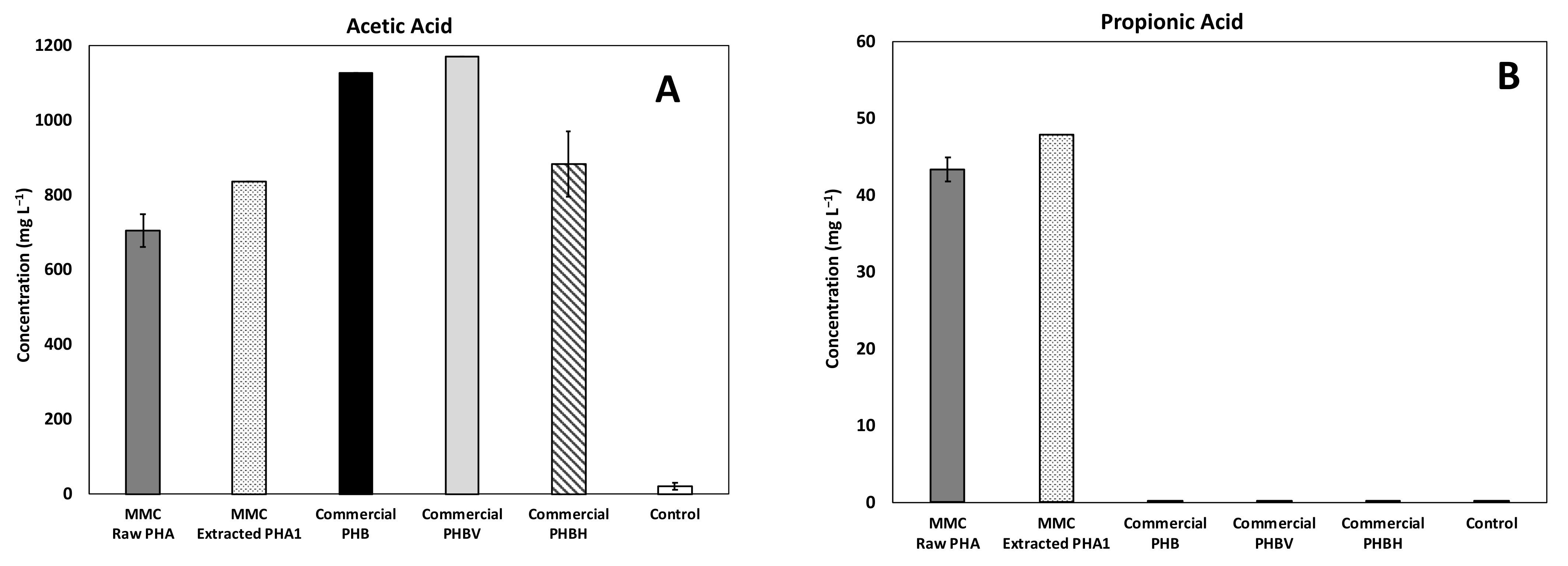
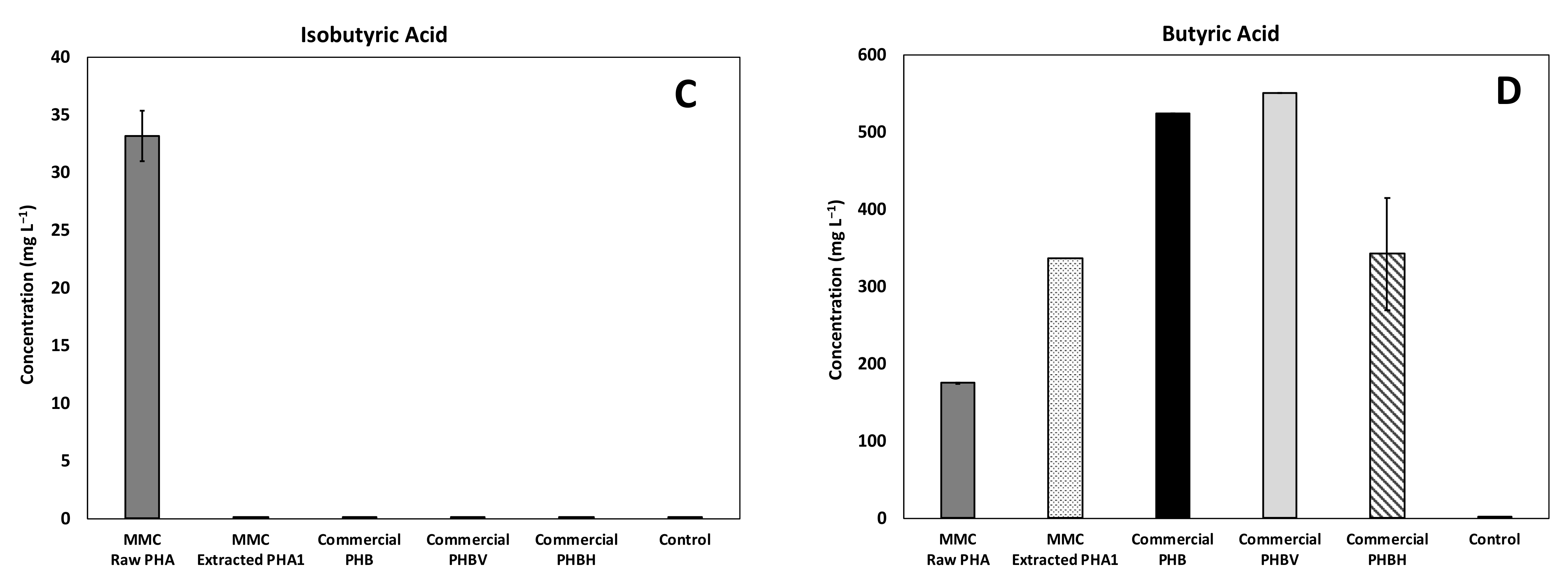
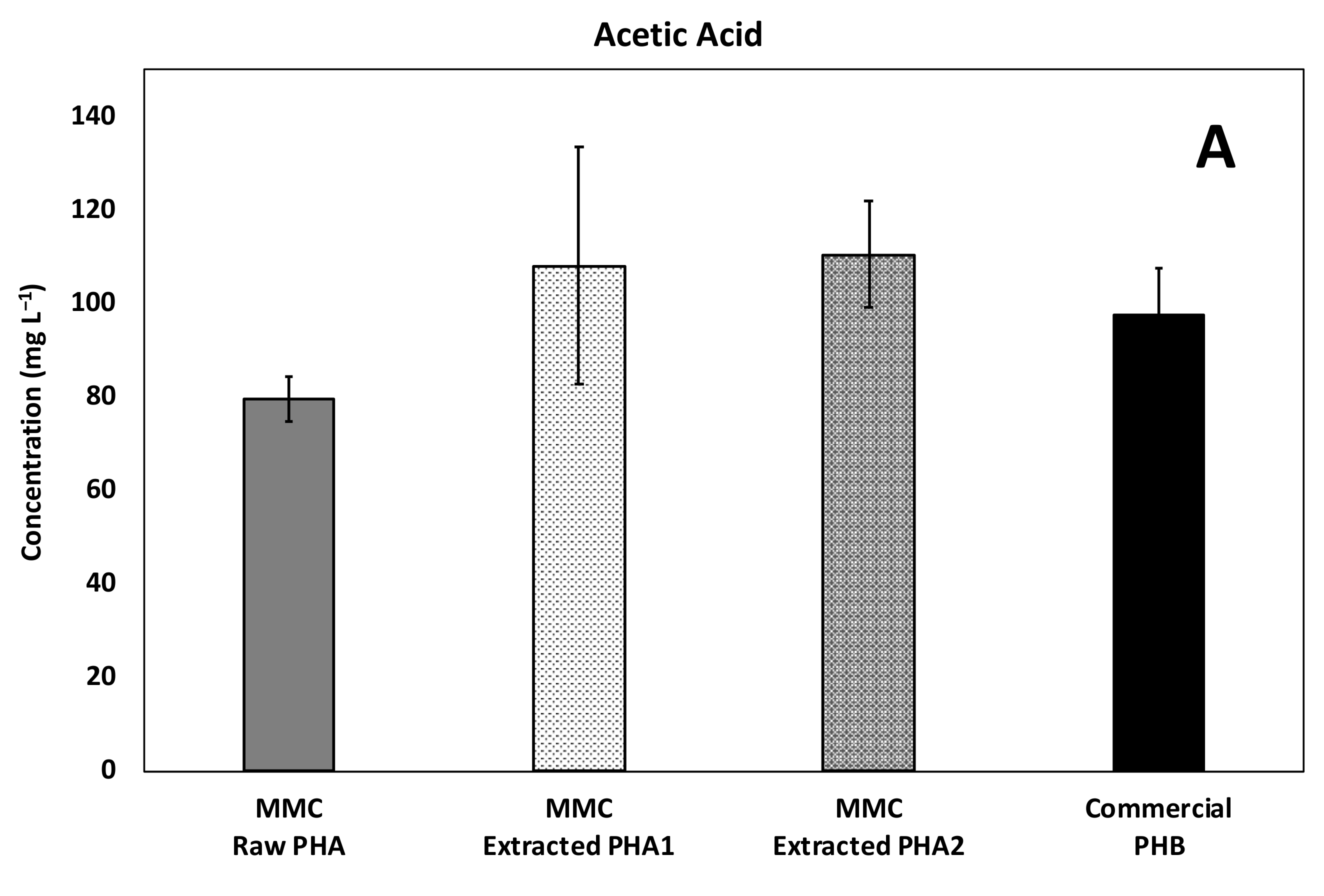
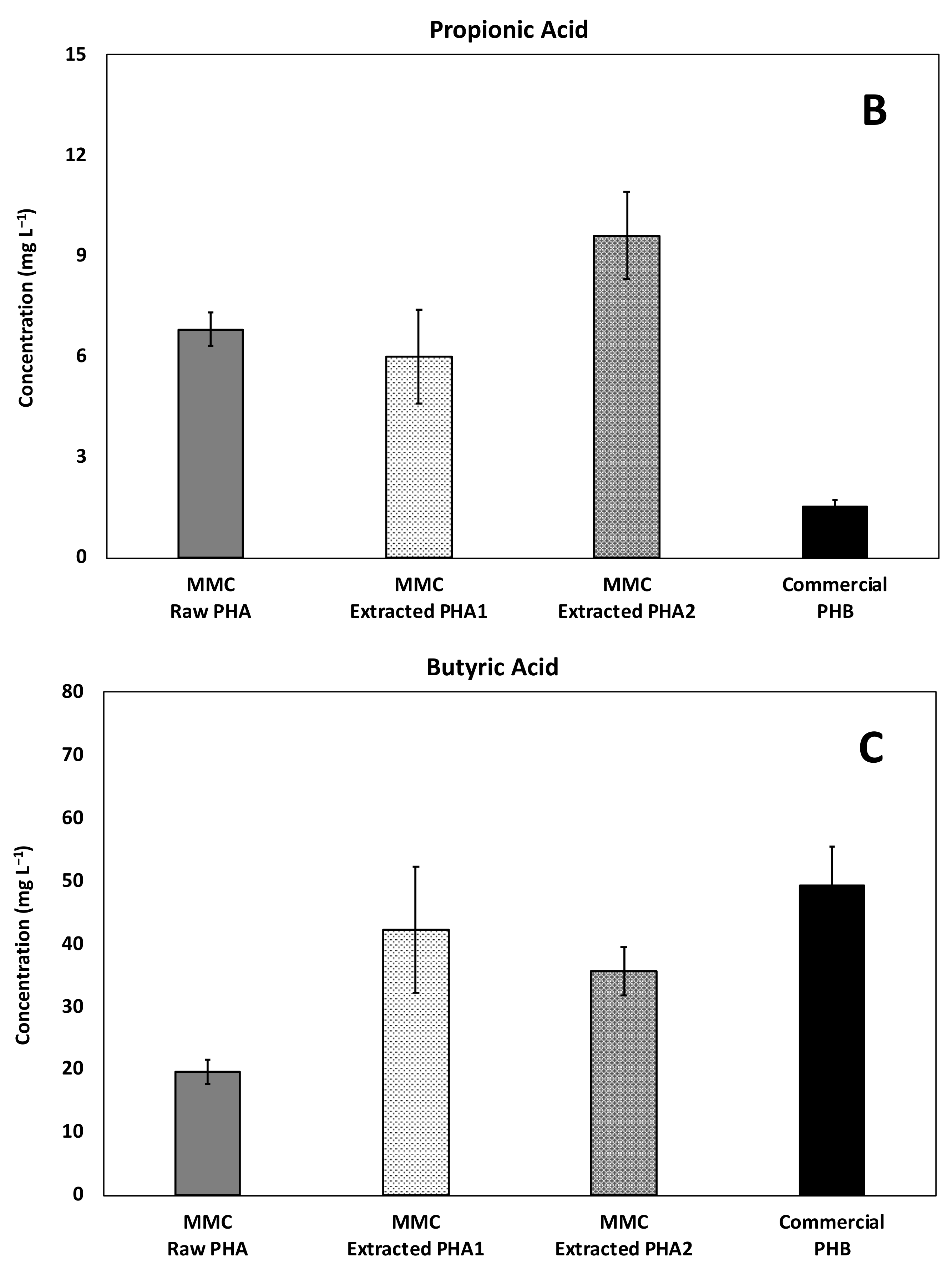
The composition of acids produced in all experiments is reported in Figure 6, with as a main reference the maximum obtained concentration. In particular, a mixture of acetic and butyric acid was detected in all the tested conditions with the predominance of acetic acid, while a relevant presence of propionic acid was detected only in the experiments performed with MMC-PHA materials. This aspect perfectly reflects the fact that acetic and butyric acids are the precursors for the biological synthesis of the HB monomer and propionic acid is the precursor for the HV monomer synthesis [34,35].
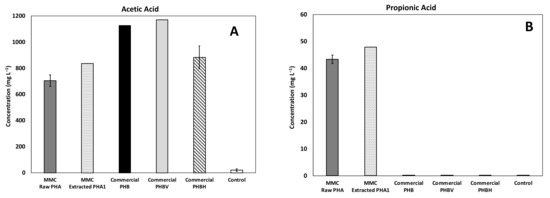
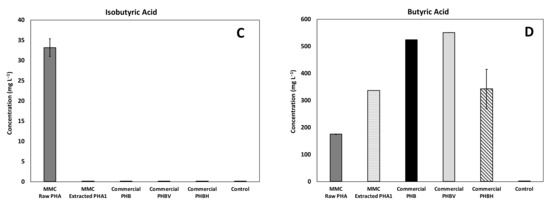
Figure 6.
Maximum concentration of the main acids produced during batch fermentability experiments: (A) Acetic, (B) Propionic, (C) Isobutyric, and (D) Butyric acids.
However, the commercial PHBV material showed a performance comparable to that obtained with the commercial PHB material, both in terms of composition and quantity of produced acids, and this could be due to the fact that the commercial copolymer contained a much lower (3%, w/w) percentage of HV than MMC based PHA (18%, w/w). Additionally, more interesting evidence to highlight is the presence of a low concentration (33 ± 2 mg/L) of isobutyric acid only in the test with the MMC-Raw PHA. This finding requires further investigations, though it could possibly be due to the lower purity of this material with respect to the others. Finally, the analysis of the headspace of all batch experiments revealed a negligible presence of methane, in agreement with the fact that upon production, VFA were not further consumed to a substantial extent. This finding was likely related to both the acidification of the medium consequent to PHA degradation and to the temperature of the tests, which remained at 20 ± 2 °C. As for hydrogen gas, it was not detected throughout the study and its absence is consistent with the fact that, during the anaerobic fermentation process, the hydrogen partial pressure is expected to remain below a threshold value (between 10−4 and 10−5 atm), and therefore, well below the instrumental detection limit of the GC-TCD.
3.2. Column Fermentability Tests
By taking into account the results collected with batch tests, column reactors were set up in order to study the PHA fermentability under a continuous-flow mode of operation. To accomplish this objective, four types of materials have been considered including three MMC-based and one commercial PHA. The latter consisted of a PHB homopolymer material, which was the commercial material displaying the best performance among the three investigated in the batch experiments. As for the other materials, two of them (i.e., MMC-Raw PHA and MMC-Extracted PHA1) were produced from the pilot scale plant fed with urban wastes, and an additional one was derived from a pilot scale PHA-producing process fed with fermented fruit waste. Likewise, the MMC-Extracted PHA1 underwent an extraction step from microbial cells, as described in paragraph 2.1.2, and it is hereafter referred to as “MMC-Extracted PHA2.” The Raw PHA material was the same as in the batch tests. Initially, the continuous-flow experiments were not inoculated with anaerobic sludge, which was supplied after 45 days of operation to verify whether the fermentative activity was limited by the lack of an appropriate inoculum.
Overall, the experiments lasted 80 days, and no fermentation products were detected during the first 50 days of operation, except for the column containing the MMC-Raw PHA (Figure 7), in agreement with the results of the batch experiments. In this column, the start of the fermentation process was almost immediate and acids production reached a maximum concentration (approximately 200 mg/L) just after 17 days of operation.
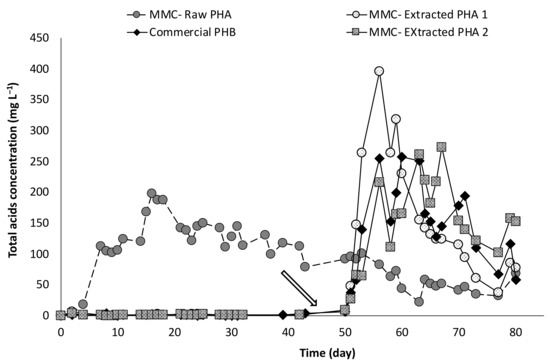
Figure 7.
Concentration of total acids produced throughout all the operation, before and after inoculation, of continuous-flow experiments. The arrow indicates the inoculation.
As a consequence, more than 25% of the PHA contained in the Raw material was converted into acids already before columns inoculation. This finding provides an additional line of evidence that this material enables a more rapid microbial colonization (e.g., by the microorganisms contained in the tap water used as the feeding solution), and accordingly, a much shorter startup of the fermentation process. In this context, the addition of anaerobic microorganisms did not further intensify the fermentation activity in the test with MMC-Raw PHA, and about 40% of the polymer contained in this material was transformed into VFA in correspondence with the end of the experimentation. However, as shown in Figure 7, the microbial inoculation significantly affected the performance of tests with the other materials triggering a rapid VFA production.
Initially, a higher acids concentration (up to 395 mg/L) was reached with the MMC-Extracted PHA1 material than with the other materials, but all of them showed a similar trend and, by the end of the tests, a comparable conversion of PHA into VFA was obtained (from 27% with commercial PHB to 28% for MMC-Extracted PHA2 and 29% for MMC-Extracted PHA1).
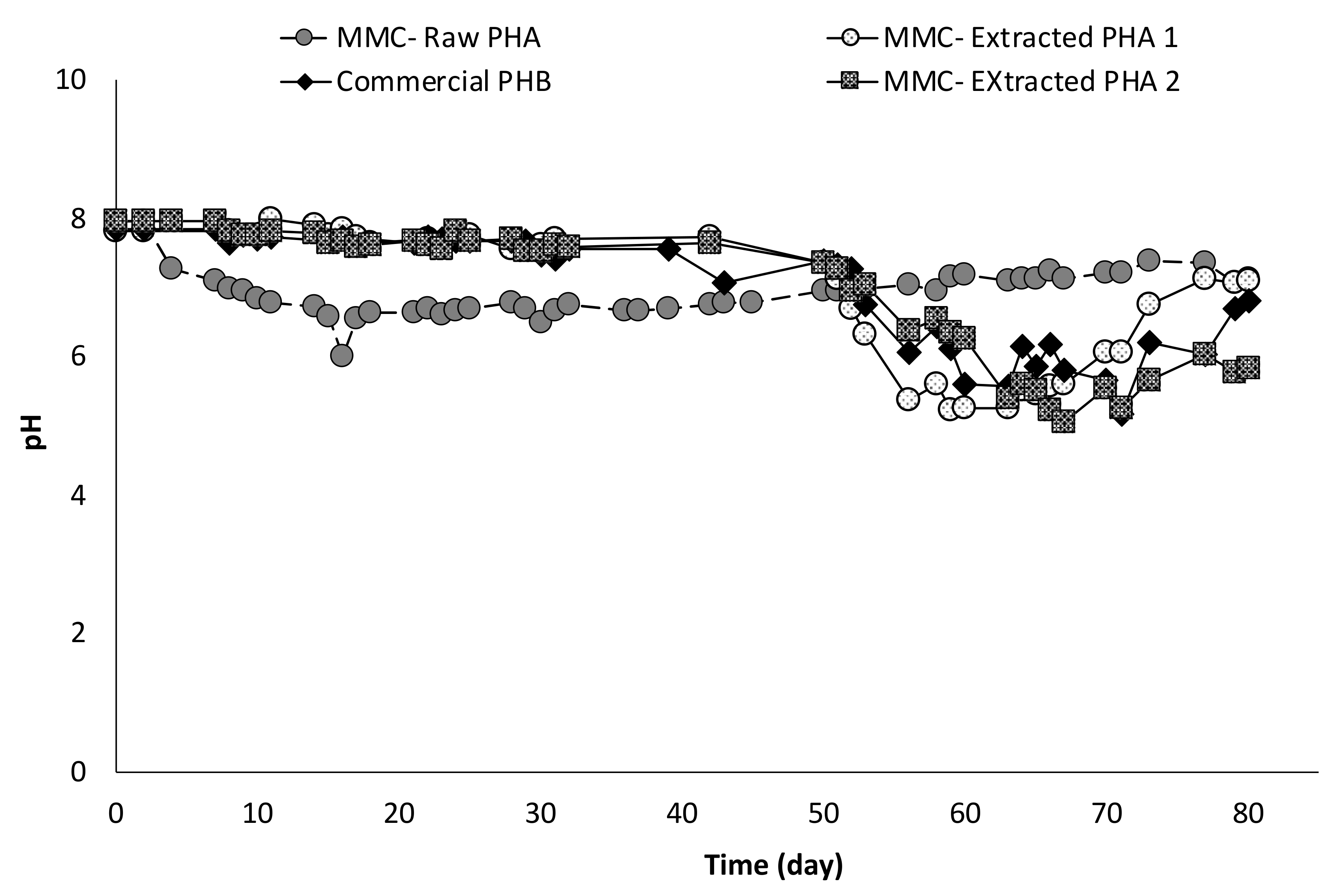
Correspondingly, with the onset of the fermentation activity, the pH of the columns suddenly decreased from around 8 to values between 5 and 6 to further rise again when acids production slowed down, as reported in Figure 8. The correlation between pH and acids concentration is also evident from the trend of pH values in the column with Raw PHA, whereby the continuous production of VFA resulted in an almost constant pH (6.9 ± 0.5).
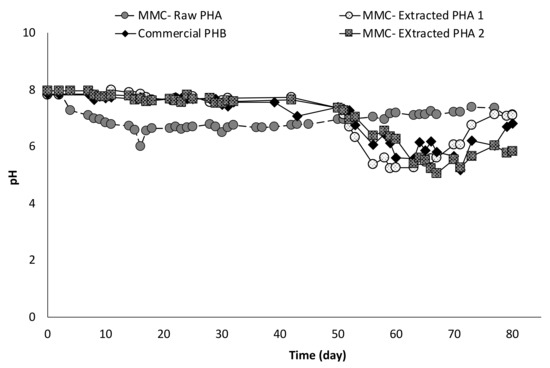
Figure 8.
Trend of pH in all continuous experiments.
Notably, in column tests, the initial pH of all experiments was higher than the initial pH value measured in batch tests. This, as previously discussed, was due to the use of a gas mixture containing CO2 to establish anaerobic conditions in the batch experiments, whereas this procedure was not applied in the column experiments. However, in the latter case, the use of air-tight reactors along with the excess of carbon source likely contributed to the spontaneous establishment of an anaerobic environment, as also confirmed by the production of organic acids. In fact, these are intermediate metabolites in the anaerobic degradation of PHA. The acids’ composition, in terms of main components, is reported in Figure 9.
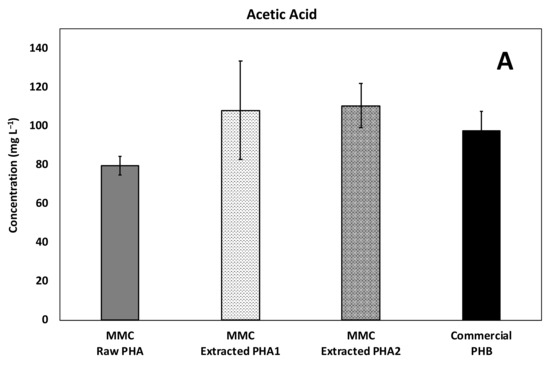
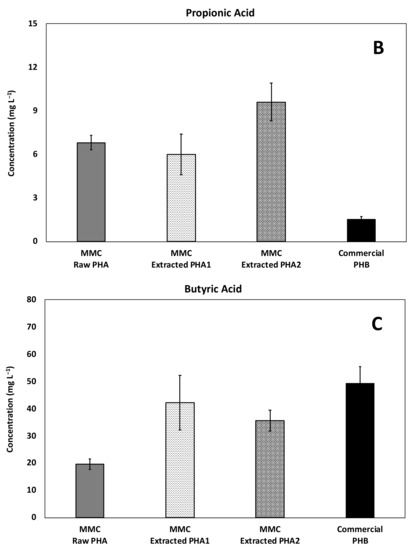
Figure 9.
Average concentration of the main acids produced during continuous fermentability experiments: (A) Acetic, (B) Propionic, and (C) Butyric acids.
Acetic acid was the principal fermentation product in all the continuous experiments, and on average, its concentration reached a value of about 80 ± 5 mg/L with the Raw PHA, whereas higher and comparable values were obtained with the other materials (Figure 9A).
Propionic acid was also produced (Figure 9B) and, as expected, a very low concentration was found in the column with the commercial material made of PHB, while a very similar production was reached with the two materials with a similar HV content (i.e., the MMC-Raw PHA and the MMC-Extracted PHA1) and, accordingly, the MMC-Extracted PHA2 having a higher HV content resulted in an even higher concentration. This is an extremely relevant finding, since propionate is commonly regarded as an ideal electron donor for environmental applications due to its high yield of hydrogen (i.e., the ultimate electron donor in the reductive dechlorination process) generation during fermentation (i.e., 3 mol of H2 and 1 mol of acetate) and due to the fact that it is degraded slowly and only under low H2 partial pressures, with both conditions known to favor, as an example, the reductive dechlorination process over competing metabolisms [36,37].
On the contrary, a higher average production of butyric acid (Figure 9C) was observed with the commercial PHB than with the MMC-based PHA materials and, in general, its concentration was intermediate between the acetic and propionic acid concentration. As for isobutyric acid, its production was negligible in all the continuous-flow experiments. Taken as a whole, these results clearly indicate that several factors (including material origin, composition, purity, and morphology) can affect PHA conversion into organic acids and, in turn, their potential degradation in subsurface environments [38].
4. Conclusions
This study examined the fermentation pattern of different PHA-based materials in the context of their possible use as electron donors for environmental remediation applications. In particular, three types of MMC-PHA materials (produced through pilot scale processes using waste feedstock) have been tested and compared to commercially available materials by means of batch and continuous-flow experiments. Under both conditions, the MMC-Raw PHA material displayed a more rapid fermentability, leading to a steady production of VFA (primarily consisting of acetic, propionic, and butyric acid). Remarkably, this material consisted of PHA-rich microbial cells directly deriving from the accumulation stage of the MMC-PHA production process, which were subjected to a thermal treatment without any following physico-chemical steps, and it showed a higher yield of conversion into acids (up to 70% gVFA/gPHA) compared to all other tested materials, made of extracted and purified PHA. Results of this study clearly demonstrated that the presence of microbial cells accompanying the MMC-Raw PHA, and accounting for approximately 44% of the material on a weight basis, likely provides soluble substrates (such as micro- and macronutrients released upon the lysis of the cellular fraction), which stimulate the anaerobic metabolism, thereby enhancing the yield of VFA production relative to other used PHA-based materials. Additionally, this resulted in a reduced or absent lag phase for the onset of the fermentation process with the Raw PHA. However, further research is needed to clarify the mechanisms, at a chemical and biochemical level, triggering this interesting behavior.
In terms of acids production, higher amounts of propionic acid were detected in the experiments carried out using PHA materials obtained from MMC-based processes, containing a significant amount of HV, reflecting the correlation between products deriving from PHA fermentation and its composition. In conclusion, this study pinpoints the advantageous potential of not-purified PHA as a suitable and inexpensive slow-release carbon source for environmental applications and future research will be focused to verify its applicability in the specific field of groundwater bioremediation. Being completely biodegradable in the environment and obtained from waste organic streams by means of an MMC-based bioprocess, without the need for expensive and typically polluting extraction procedures, this material brings both economic and environmental advantages in the context of subsurface bioremediation and, as a consequence, could have new marketable applications which could represent important drivers toward the implementation of circular bioeconomy principles. Further studies, including ecotoxicity tests, are, however, still warranted to evaluate whether this material complies with all regulatory standards needed for environmental applications.
Author Contributions
Conceptualization, N.A. and M.P.P.; formal analysis, N.A., M.P.P., and M.V.; investigation, N.A. and M.M.R.; resources, M.P.P. and F.V.; data curation, N.A., M.M.R., and M.V.; writing—original draft preparation, N.A., M.P.P., and M.V.; visualization, N.A., B.M., and M.V.; supervision, M.P.P.; project administration, M.P.P.; funding acquisition, M.P.P. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the RES URBIS (RESources from URban BIo-waSte) Project (GA No 730349) in the frame of the European Horizon 2020 (Call CIRC-05-2016: Unlocking the potential of urban organic waste) program.
Data Availability Statement
Not applicable.
Acknowledgments
Maria Reis (Faculty of Sciences and Technology, Universidade NOVA de Lisboa) and Mauro Majone (Department of Chemistry, Sapienza University of Rome) are gratefully acknowledged for providing some of the PHA samples tested in this study and for supporting and coordinating the RES URBIS Project, respectively. The authors also want to acknowledge Biotrend SA. for extracting some of the MMC-PHA materials.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sabapathy, P.C.; Devaraj, S.; Meixner, K.; Anburajan, P.; Kathirvel, P.; Ravikumar, Y.; Zabed, H.M.; Qi, X. Recent developments in Polyhydroxyalkanoates (PHAs) production—A review. Bioresour. Technol. 2020, 306, 123132. [Google Scholar] [CrossRef]
- Valentino, F.; Morgan-Sagastume, F.; Campanari, S.; Villano, M.; Werker, A.; Majone, M. Carbon recovery from wastewater through bioconversion into biodegradable polymers. New Biotechnol. 2017, 37, 9–23. [Google Scholar] [CrossRef]
- Kumar, M.; Rathour, R.; Singh, R.; Sun, Y.; Pandey, A.; Gnansounou, E.; Lin, K.Y.A.; Tsang, D.C.W.; Thakur, I.S. Bacterial polyhydroxyalkanoates: Opportunities, challenges, and prospects. J. Clean. Prod. 2020, 263, 121500. [Google Scholar] [CrossRef]
- Rodriguez-Contreras, A. Recent advances in the use of polyhydroxyalkanoates in biomedicine. Bioengineering 2019, 6, 82. [Google Scholar] [CrossRef]
- Koller, M. Biodegradable and biocompatible polyhydroxy-alkanoates (PHA): Auspicious microbial macromolecules for pharmaceutical and therapeutic applications. Molecules 2018, 23, 362. [Google Scholar] [CrossRef]
- Valentino, F.; Moretto, G.; Lorini, L.; Bolzonella, D.; Pavan, P.; Majone, M. Pilot-Scale Polyhydroxyalkanoate Production from Combined Treatment of Organic Fraction of Municipal Solid Waste and Sewage Sludge. Ind. Eng. Chem. Res. 2019, 58, 12149–12158. [Google Scholar] [CrossRef]
- Bengtsson, S.; Karlsson, A.; Alexandersson, T.; Quadri, L.; Hjort, M.; Johansson, P.; Morgan-Sagastume, F.; Anterrieu, S.; Arcos-Hernandez, M.; Karabegovic, L.; et al. A process for polyhydroxyalkanoate (PHA) production from municipal wastewater treatment with biological carbon and nitrogen removal demonstrated at pilot-scale. New Biotechnol. 2017, 35, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.; Albuquerque, M.; Villano, M.; Majone, M. Mixed Culture Processes for Polyhydroxyalkanoate Production from Agro-Industrial Surplus/Wastes as Feedstocks. In Comprehensive Biotechnology, 2nd ed.; Moo-Young, M., Ed.; Academic Press: Cambridge, MA, USA, 2011; Volume 6, pp. 669–683. [Google Scholar]
- Rodriguez-Perez, S.; Serrano, A.; Pantión, A.A.; Alonso-Fariñas, B. Challenges of scaling-up PHA production from waste streams—A review. J. Environ. Manag. 2018, 205, 215–230. [Google Scholar] [CrossRef]
- Luo, Z.; Wu, Y.; Li, Z.; Loh, X.J. Recent Progress in Polyhydroxyalkanoates-Based Copolymers for Biomedical Applications. Biotechnol. J. 2019, 14, 1900283. [Google Scholar] [CrossRef] [PubMed]
- Bugnicourt, E.; Cinelli, P.; Lazzeri, A.; Alvarez, V. Polyhydroxyalkanoate (PHA): Review of synthesis, characteristics, processing and potential applications in packaging. Express Polym. Lett. 2014, 8, 791–808. [Google Scholar] [CrossRef]
- Zhang, W.; Ruan, X.; Bai, Y.; Yin, L. The characteristics and performance of sustainable-releasing compound carbon source material applied on groundwater nitrate in-situ remediation. Chemosphere 2018, 205, 635–642. [Google Scholar] [CrossRef]
- Majone, M.; Verdini, R.; Aulenta, F.; Rossetti, S.; Tandoi, V.; Kalogerakis, N.; Agathos, S.; Puig, S.; Zanaroli, G.; Fava, F. In situ groundwater and sediment bioremediation: Barriers and perspectives at European contaminated sites. New Biotechnol. 2015, 32, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Zhang, F.; Shi, Y.; Kong, X.; Xie, Y.; Du, X.; Li, Y.; Zhang, R. Diversity of bacteria and archaea in the groundwater contaminated by chlorinated solvents undergoing natural attenuation. Environ. Res. 2020, 185, 109457. [Google Scholar] [CrossRef] [PubMed]
- Borden, R.C. Natural bioremediation of hydrocarbon-contaminated ground water. In Handbook of Bioremediation; CRC Press: Boca Raton, FL, USA, 2017; pp. 177–199. [Google Scholar]
- Baric, M.; Majone, M.; Beccari, M.; Petrangeli Papini, M. Coupling of polyhydroxybutyrate (PHB) and zero valent iron (ZVI) for enhanced treatment of chlorinated ethanes in permeable reactive barriers (PRBs). Chem. Eng. J. 2012, 195–196, 22–30. [Google Scholar] [CrossRef]
- Aulenta, F.; Fuoco, M.; Canosa, A.; Petrangeli Papini, M.; Majone, M. Use of poly-β-hydroxy-butyrate as a slow-release electron donor for the microbial reductive dechlorination of TCE. Water Sci. Technol. 2008, 57, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Borden, R.C.; Richardson, S.D.; Bodour, A.A. Enhanced reductive dechlorination of trichloroethene in an acidic DNAPL impacted aquifer. Environ. Manag. 2019, 237, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Maier, R.M.; Gentry, T.J. Microorganisms and Organic Pollutants. In Environmental Microbiology, 3rd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Aulenta, F.; Majone, M.; Tandoi, V. Enhanced anaerobic bioremediation of chlorinated solvents: Environmental factors influencing microbial activity and their relevance under field conditions. Chem. Technol. Biotechnol. 2006, 81, 1463–1474. [Google Scholar] [CrossRef]
- Blázquez-Pallí, N.; Rosell, M.; Varias, J.; Bosch, M.; Soler, A.; Vicent, T.; Marco-Urrea, E. Integrative isotopic and molecular approach for the diagnosis and implementation of an efficient in-situ enhanced biological reductive dechlorination of chlorinated ethenes. Water Res. 2019, 167, 115106. [Google Scholar] [CrossRef]
- Bradley, P.M. Microbial degradation of chloroethenes in groundwater systems. Hydrogeol. J. 2000, 8, 104–111. [Google Scholar] [CrossRef]
- Petrangeli Papini, M.; Majone, M.; Arjmand, F.; Silvestri, D.; Sagliaschi, M.; Sucato, S.; Alesi, E.; Barstch, E.; Pierro, L. First pilot test on the integration of GCW (groundwater circulation well) with ENA (enhanced natural attenuation) for chlorinated solvents source remediation. Chem. Eng. Trans. 2016, 49, 91–96. [Google Scholar] [CrossRef]
- Mannino, P.; Ceccarelli, V. Poly-hydroxybutyrate-co-hydroxyvalerate as solid slow-releasing source of electron donors for the reductive dechlorination of 1,2-dichloroethane in-situ. Int. Biodeterior. Biodegrad. 2014, 86, 278–285. [Google Scholar] [CrossRef]
- Aulenta, F.; Gossett, J.M.; Petrangeli Papini, M.; Rossetti, S.; Majone, M. Comparative study of methanol, butyrate, and hydrogen as electron donors for long-term dechlorination of tetrachloroethene in mixed anerobic cultures. Biotechnol. Bioeng. 2005, 91, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Matturro, B.; Pierro, L.; Frascadore, E.; Petrangeli Papini, M.; Rossetti, S. Microbial community changes in a chlorinated solvents polluted aquifer over the field scale treatment with poly-3-hydroxybutyrate as amendment. Front. Microbiol. 2018, 9, 1664. [Google Scholar] [CrossRef] [PubMed]
- Pierro, L.; Matturro, B.; Rossetti, S.; Sagliaschi, M.; Sucato, S.; Alesi, E.; Bartsch, E.; Arjmand, F.; Petrangeli Papini, M. Polyhydroxyalkanoate as a slow-release carbon source for in situ bioremediation of contaminated aquifers: From laboratory investigation to pilot-scale testing in the field. New Biotechnol. 2017, 37, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Baric, M.; Pierro, L.; Pietrangeli, B.; Petrangeli Papini, M. Polyhydroxyalkanoate (PHB) as a slow-release electron donor for advanced in situ bioremediation of chlorinated solvent-contaminated aquifers. New Biotechnol. 2014, 31, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Zeppilli, M.; Dell’Armi, E.; Cristiani, L.; Petrangeli Papini, M.; Majone, M. Reductive/Oxidative Sequential Bioelectrochemical Process for Perchloroethylene Removal. Water 2019, 11, 2579. [Google Scholar] [CrossRef]
- Aulenta, F.; Fina, A.; Potalivo, M.; Petrangeli Papini, M.; Rossetti, S.; Majone, M. Anaerobic transformation of tetrachloroethane, perchloroethylene, and their mixtures by mixed-cultures enriched from contaminated soils and sediments. Water Sci. Technol. 2005, 52, 357–362. [Google Scholar] [CrossRef]
- Lorini, L.; Martinelli, A.; Capuani, G.; Frison, N.; Reis, M.; Ferreira, B.S.; Villano, M.; Majone, M.; Valentino, F. Characterization of Polyhydroxyalkanoates Produced at Pilot Scale From Different Organic Wastes. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association; American Water Works Association; Water Environment Federation: Washington, DC, USA, 2005. [Google Scholar]
- Yang, Y.; McCarty, P.L. Biomass, oleate, and other possible substrates for chloroethene reductive dehalogenation. Bioremediat. J. 2000, 4, 125–133. [Google Scholar] [CrossRef]
- Montano-Herrera, L.; Laycock, B.; Werker, A.; Pratt, S. The evolution of polymer composition during PHA accumulation: The significance of reducing equivalents. Bioengineering 2017, 4, 20. [Google Scholar] [CrossRef]
- Dionisi, D.; Majone, M.; Papa, V.; Beccari, M. Biodegradable Polymers from Organic Acids by Using Activated Sludge Enriched by Aerobic Periodic Feeding. Biotechnol. Bioeng. 2004, 85, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Ciampi, P.; Esposito, C.; Petrangeli Papini, M. Hydrogeochemical Model Supporting the Remediation Strategy of a Highly Contaminated Industrial Site. Water 2019, 11, 1371. [Google Scholar] [CrossRef]
- Dong, X.; Plugge, C.M.; Stams, A.J.M. Anaerobic Degradation of Propionate by a Mesophilic Acetogenic Bacterium in Coculture and Triculture with Different Methanogens. Appl. Environ. Microbiol. 1994, 60, 2834–2838. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.; Salvador, A.; Alves, M.M.; Vicente, A.A. Factors affecting polyhydroxyalkanoates biodegradation in soil. Polym. Degrad. Stab. 2020, 182, 109408. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).