Seasonal Variations in Dissolved Organic Carbon in the Source Region of the Yellow River on the Tibetan Plateau
Abstract
:1. Introduction
2. Data and Methods
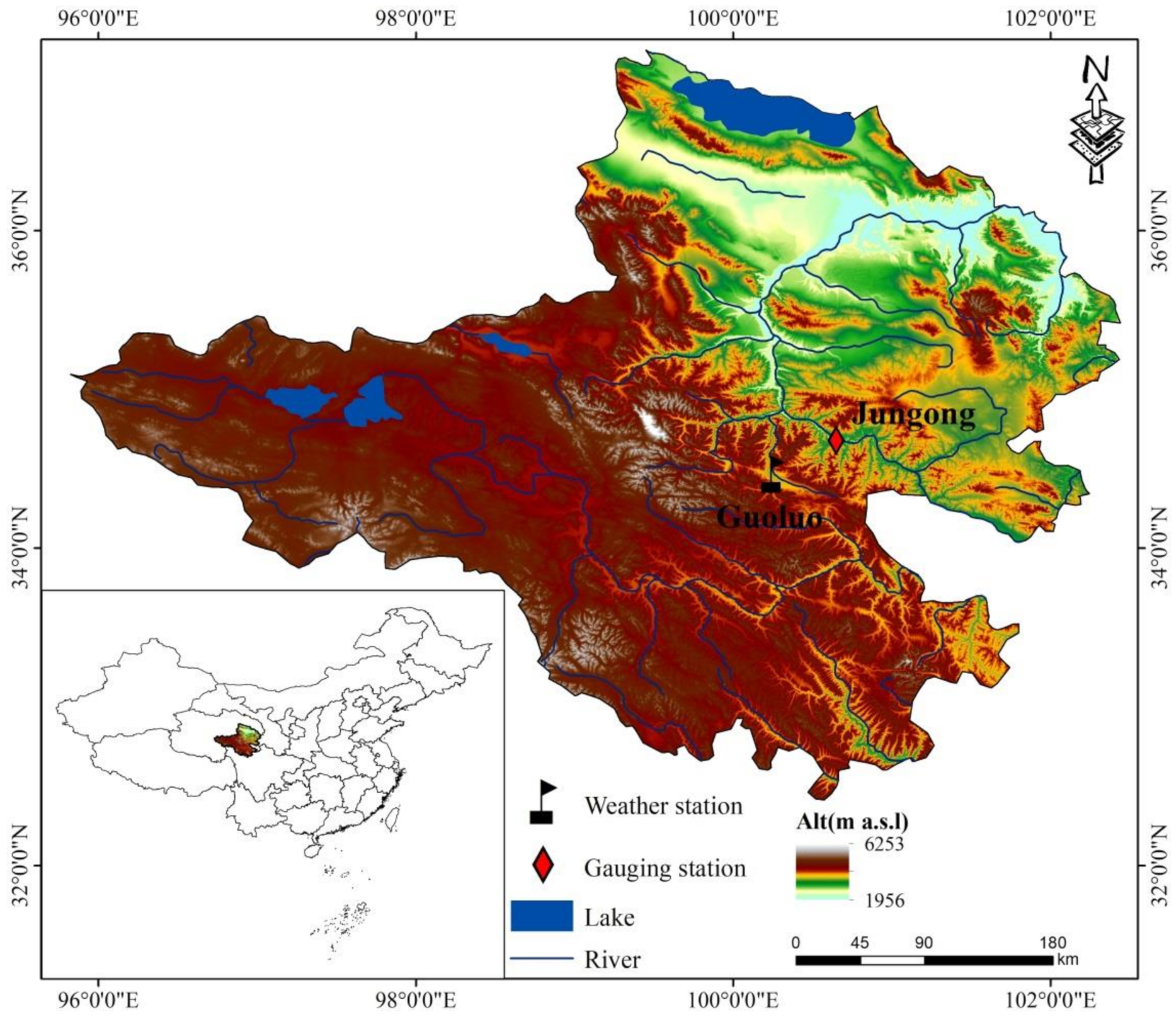
2.1. Study Area
2.2. Sample Collection
2.3. Laboratory Analysis
2.4. Flux Calculation and Statistical Analysis
3. Results and Discussion
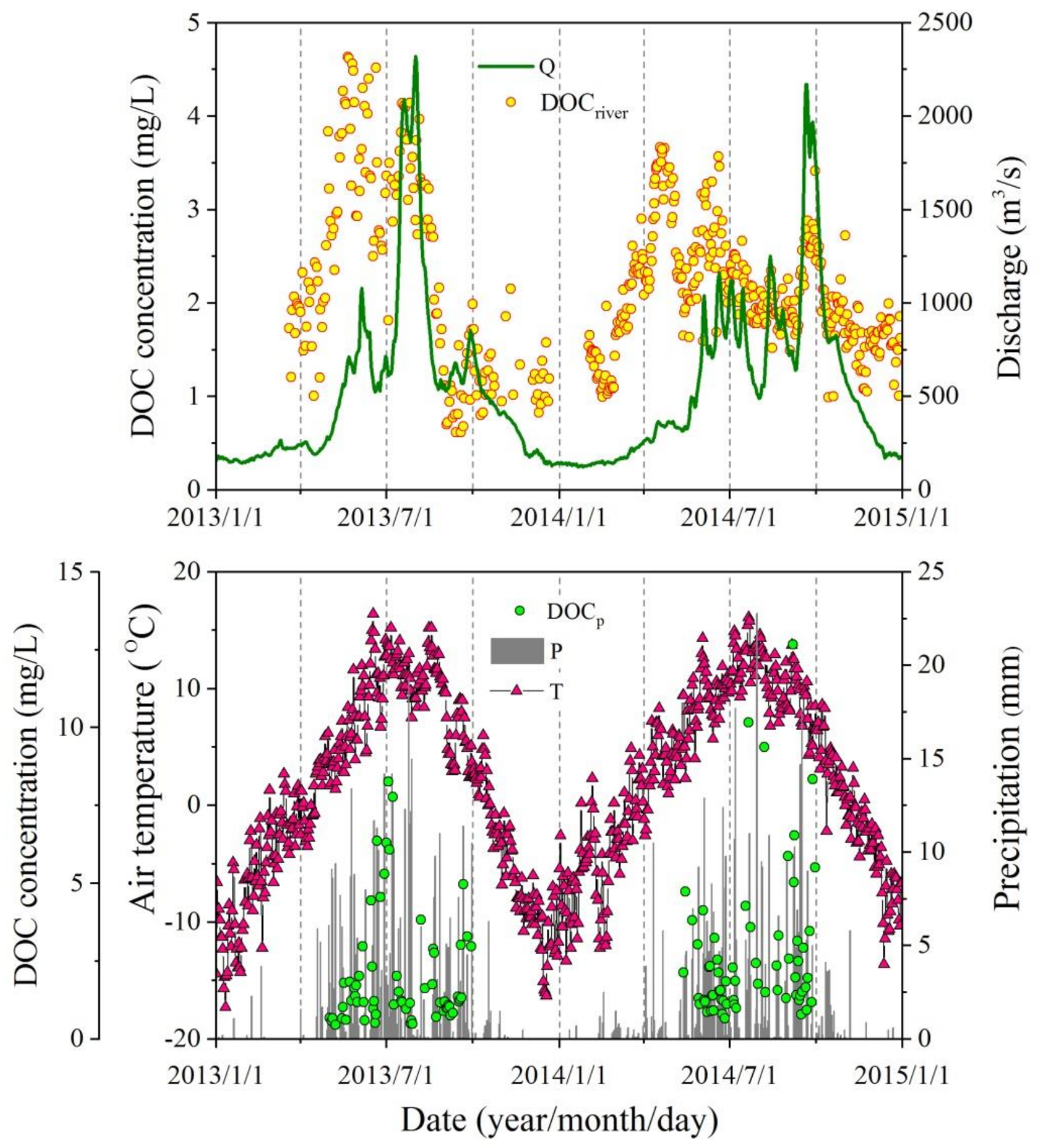
3.1. Seasonal Variations in DOC Concentrations in the River
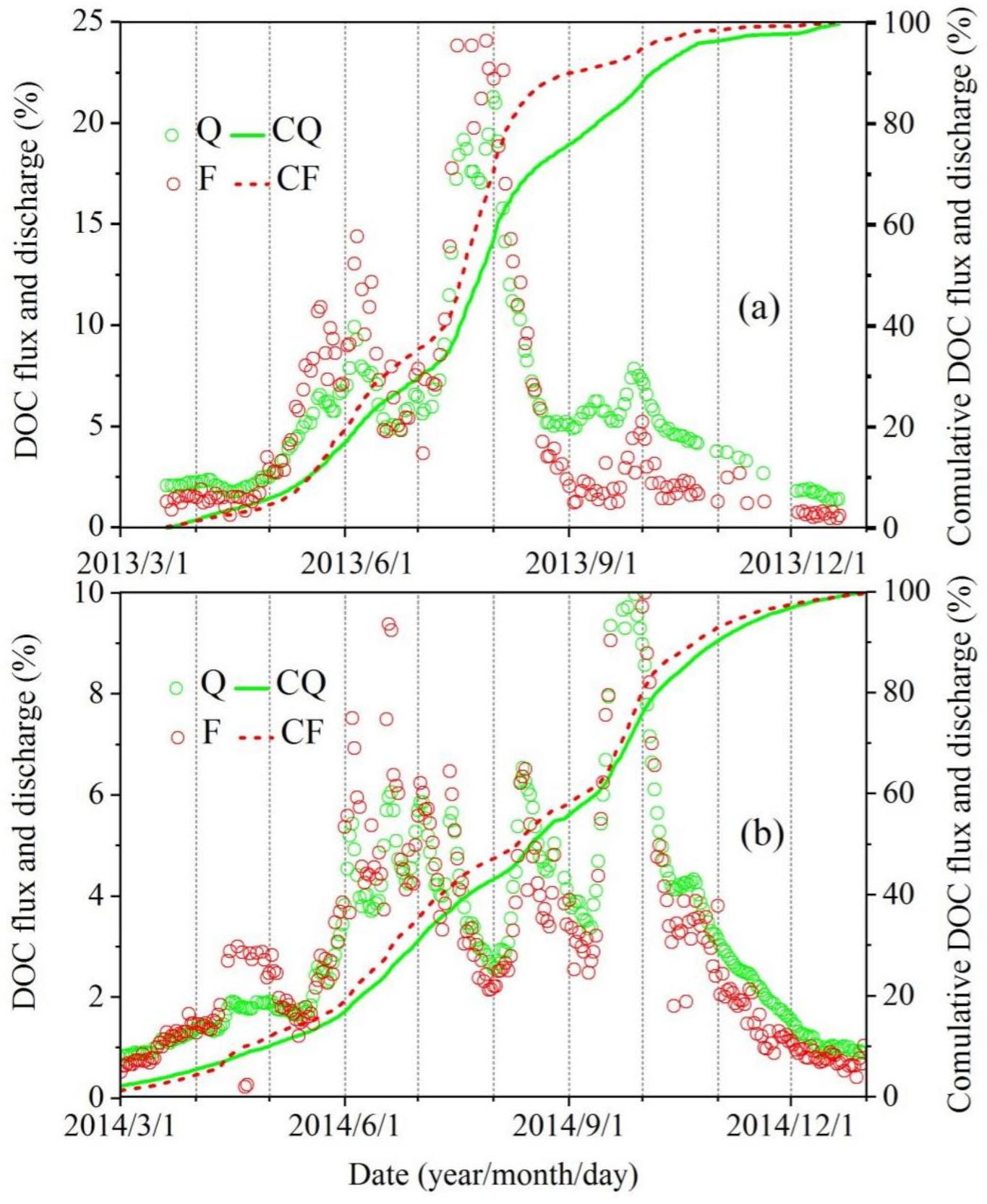
3.2. DOC Fluxes in the River
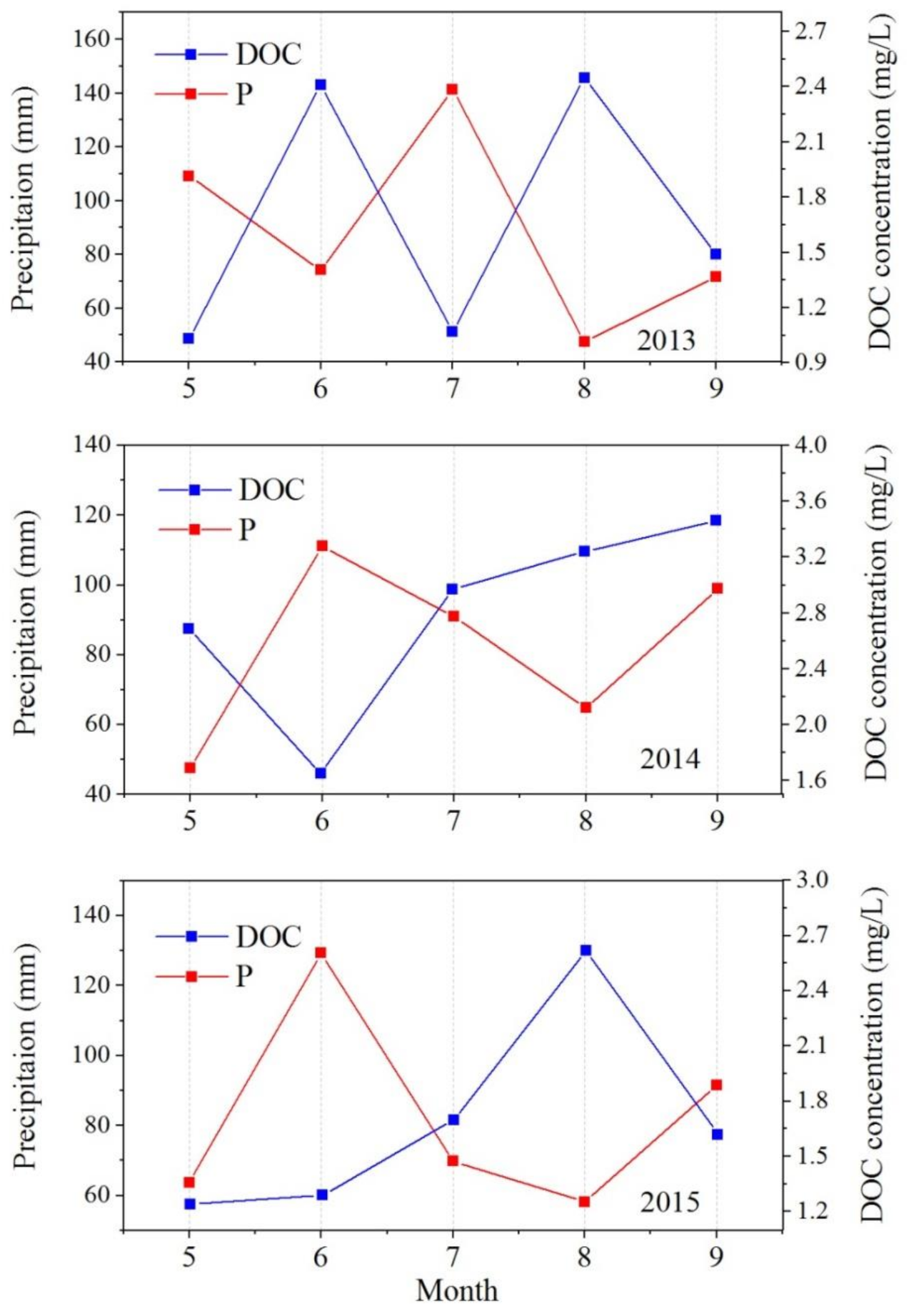
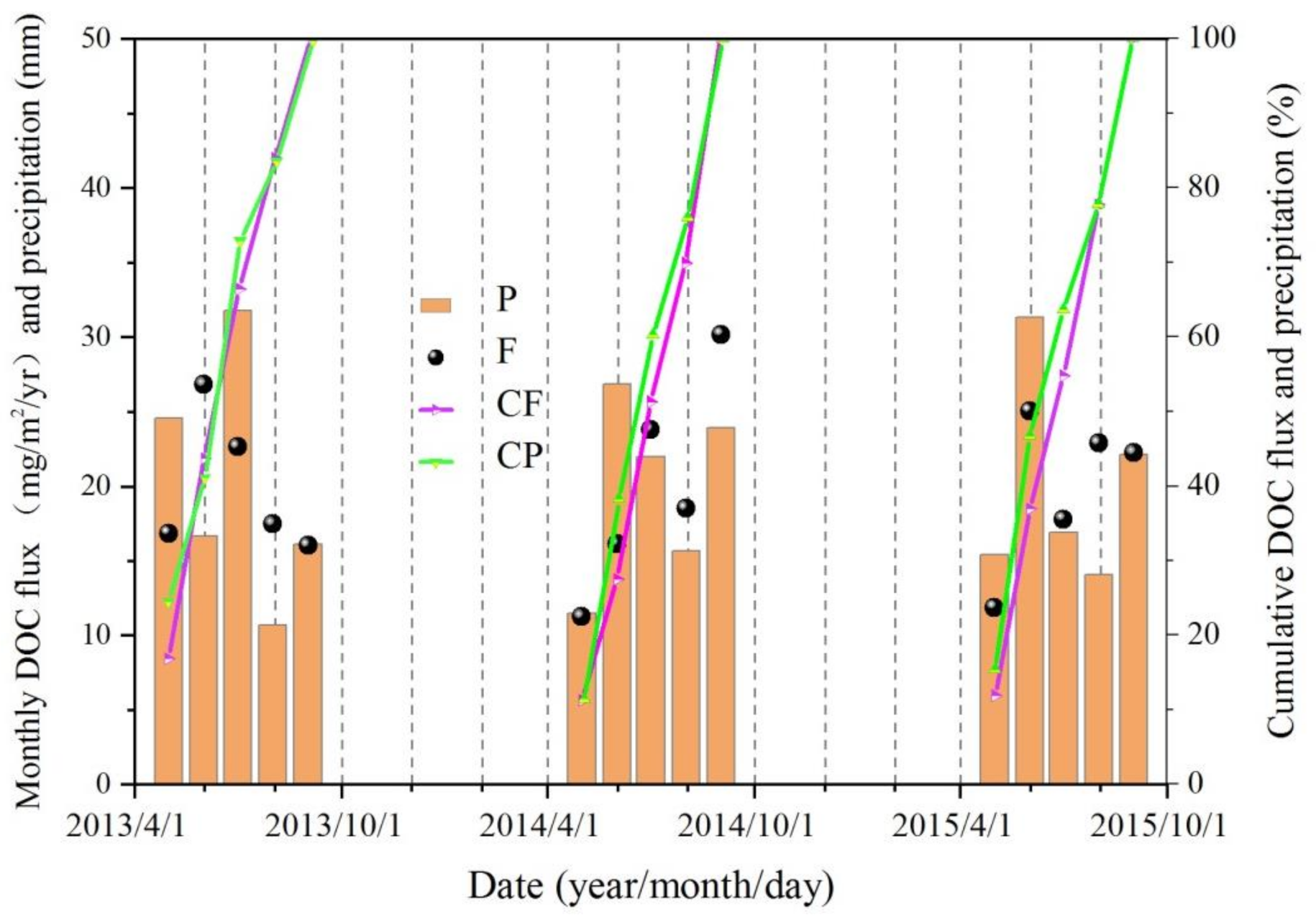
3.3. DOC Concentrations and Fluxes in Precipitation
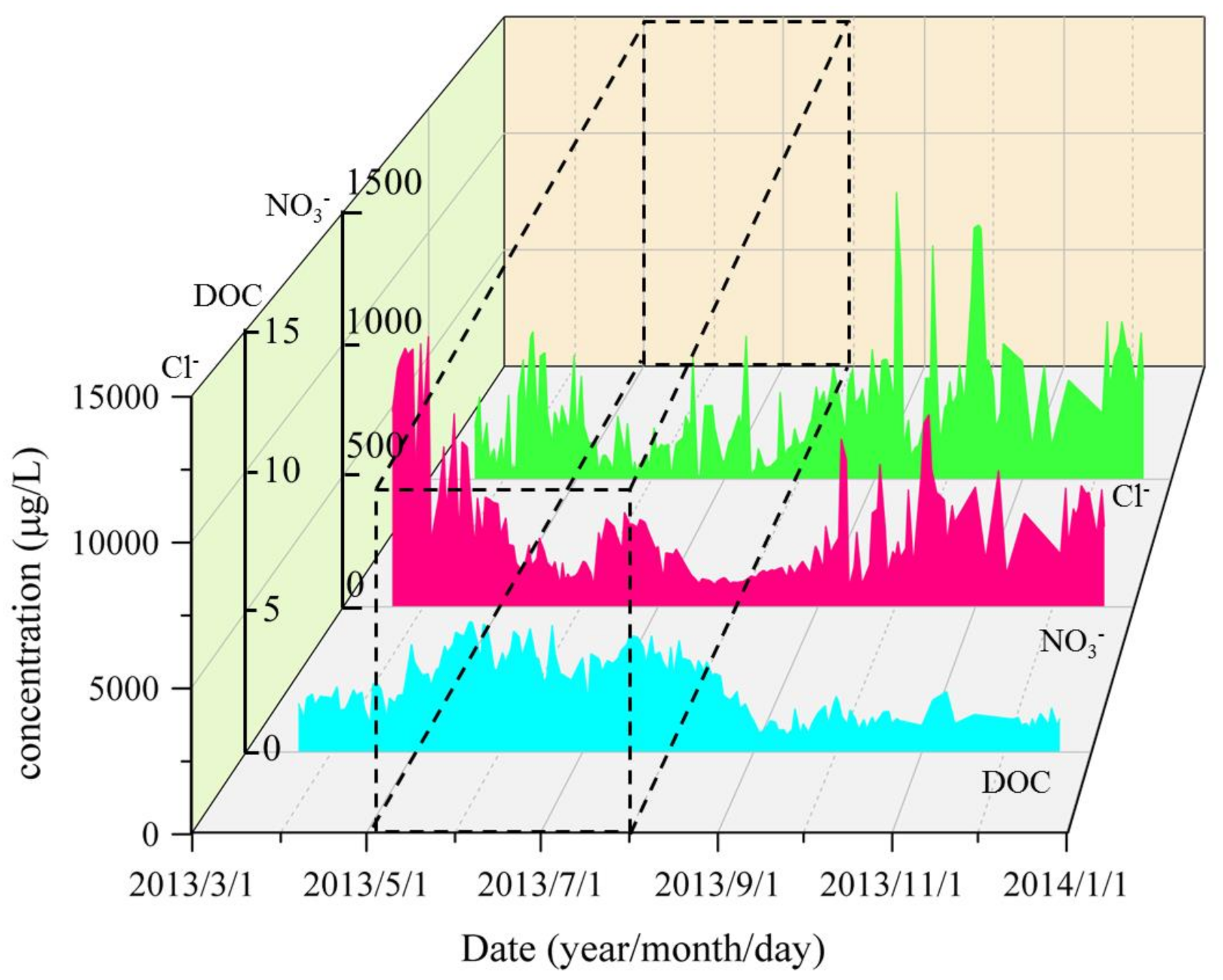
3.4. DOC and Inorganic Ions
4. Conclusions
- Annual mean DOC concentrations in the river during the three years were 2.3 ± 1.1, 2.2 ± 1.4, and 2.0 ± 0.5 mg/L, respectively. Closely related to the local biogeochemical and hydrographic processes, a significant seasonal variation could be observed, with the maximum in summer (2.65 mg/L) and the minimum in winter (1.44 mg/L). Runoff and precipitation, involving glacier and frozen soil meltwater are the main reasons affecting riverine DOC concentration. Concentrations of DOC in rainfall ranged from 0.59 to 12.1 mg/L, with an inverse relationship with precipitation amount, illustrating that heavier precipitation could dilute DOC concentrations.
- Transportation of DOC by the river was estimated as 26,058 tons in 2013, 37,539 tons in 2014, and 24,629 tons in 2015, which was dominated by river discharge. Compared with the rivers in other climatic zones in the world, it is transparent that humid climate, high rate of vegetation coverage, and developed water system can increase the riverine DOC concentration; DOC yield in SRYR is much less than that of rivers in tropical humid climate areas, also less than the cold areas covered by Tundra and Taiga but greater than tropical and temperate dry areas, suggesting that the melting of glaciers and frozen soil is an important factor affecting the DOC yield in SRYR.
- About 86,080, 105,804, and 73,072 tons of DOC during 2013 to 2015 respectively were brought to SRYR by rainfall, which was controlled both by DOC concentrations and precipitations. Although the DOC levels deposited by rainfall were far greater than that of river delivery, only a small part of DOC in precipitation directly entered the river, and most of it fell on the land surface and was distributed under the influence of the local water cycle. Therefore, the main source of riverine DOC was catchment scouring, including both surface runoff and precipitation flushing. The intricate hydraulic connection and complex biological processes also had a potential effect on it.
- The variation trend of DOC concentrations was contrary to that of ions in the river, especially for NO3− and Cl− ions (r = 0.5, p < 0.05 for NO3−; r = 0.4, p < 0.05 for Cl−), which confirms the previous studies indicating that a possible relationship exists between DOC concentration and acid deposition.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McClelland, J.; Townsend-Small, A.; Holmes, R.M.; Pan, F.; Stieglitz, M.; Khosh, M.; Peterson, B.J. River export of nutrients and organic matter from the North Slope of Alaska to the Beaufort Sea. Water Resour. Res. 2014, 50, 1823–1839. [Google Scholar] [CrossRef] [Green Version]
- Smith, H.J.; Foster, R.A.; McKnight, D.M.; Lisle, J.T.; Littmann, S.; Kuypers, M.M.M.; Foreman, C.M. Microbial formation of labile organic carbon in Antarctic glacial environments. Nat. Geosci. 2017, 10, 356–359. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, K.; Kuwae, T. How organic carbon derived from multiple sources contributes to carbon sequestration processes in a shallow coastal system? Glob. Chang. Biol. 2015, 21, 2612–2623. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Peng, C.; Zhou, X.; Yang, Y.; Guo, Y.; Shi, G.; Zhu, Q. Modeling Global Riverine DOC Flux Dynamics from 1951 to 2015. J. Adv. Model. Earth Syst. 2019, 11, 514–530. [Google Scholar] [CrossRef] [Green Version]
- Mayorga, E.; Aufdenkampe, A.; Masiello, C.; Krusche, A.; Hedges, J.I.; Quay, P.D.; Richey, J.E.; Brown, T. Young organic matter as a source of carbon dioxide outgassing from Amazonian rivers. Nature 2005, 436, 538–541. [Google Scholar] [CrossRef]
- Mora, A.; Alfonso, J.A.; Baquero, J.C.; Balza, L.; Pisapia, D. Caura River basin: Weathering rates, CO2 consumption, and chemistry of major and trace elements in an Orinoco River tributary coming from the Precambrian Guayana Shield, Venezuela. Geochem. Geophy. Geosy. 2010, 11. [Google Scholar] [CrossRef]
- Mora, A.; Laraque, A.; Moreira-Turcq, P.; Alfonso, J.A. Temporal variation and fluxes of dissolved and particulate organic carbon in the Apure, Caura and Orinoco rivers, Venezuela. J. South. Am. Earth Sci. 2014, 54, 47–56. [Google Scholar] [CrossRef]
- Musilova, M.; Tranter, M.; Wadham, J.; Telling, J.; Tedstone, A.; Anesio, A.M. Microbially driven export of labile organic carbon from the Greenland ice sheet. Nat. Geosci. 2017, 10, 360–365. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.L.; He, W.; Wu, R.L.; Xing, B.; Xu, F.L. A general-applicable model for estimating the binding coefficient of organic pollutants with dissolved organic matter. Sci. Total Environ. 2019, 670, 226–235. [Google Scholar] [CrossRef]
- Blair, N.E.; Aller, R.C. The Fate of Terrestrial Organic Carbon in the Marine Environment. Annu. Rev. Mar. Sci. 2012, 4, 401–423. [Google Scholar] [CrossRef]
- Aufdenkampe, A.K.; Mayorga, E.; Raymond, P.A.; Melack, J.M.; Doney, S.; Alin, S.; Aalto, R.E.; Yoo, K. Riverine coupling of biogeochemical cycles between land, oceans, and atmosphere. Front. Ecol. Environ. 2011, 9, 53–60. [Google Scholar] [CrossRef] [Green Version]
- Spencer, R.G.M.; Aiken, G.R.; Wickland, K.; Striegl, R.G.; Hernes, P.J. Seasonal and spatial variability in dissolved organic matter quantity and composition from the Yukon River basin, Alaska. Glob. Biogeochem. Cycles 2008, 22. [Google Scholar] [CrossRef]
- Griffin, C.G.; Finlay, J.; Brezonik, P.L.; Olmanson, L.; Hozalski, R. Limitations on using CDOM as a proxy for DOC in temperate lakes. Water Res. 2018, 144, 719–727. [Google Scholar] [CrossRef]
- Galy, V.; Peucker-Ehrenbrink, B.; Eglinton, T. Global carbon export from the terrestrial biosphere controlled by erosion. Nature 2015, 521, 204–207. [Google Scholar] [CrossRef]
- Spencer, R.G.; Hernes, P.J.; Aufdenkampe, A.K.; Baker, A.; Gulliver, P.; Stubbins, A.; Aiken, G.R.; Dyda, R.Y.; Butler, K.D.; Mwamba, V.L.; et al. An initial investigation into the organic matter biogeochemistry of the Congo River. Geochim. Cosmochim. Acta 2012, 84, 614–627. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, J.; Liu, S.; Zhang, Z.; Yao, Q.; Hong, G.; Cooper, L. Sources and distribution of carbon within the Yangtze River system. Estuar. Coast. Shelf Sci. 2007, 71, 13–25. [Google Scholar] [CrossRef]
- Yan, F.; Kang, S.; Li, C.; Zhang, Y.; Qin, X.; Li, Y.; Zhang, X.; Hu, Z.; Chen, P.; Li, X.; et al. Concentration, sources and light absorption characteristics of dissolved organic carbon on a medium-sized valley glacier, northern Tibetan Plateau. Cryosphere 2016, 10, 2611–2621. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Xue, M.; Wang, M.; Cai, W.-J.; Wang, L.; Yu, Z. The spatiotemporal distribution of dissolved inorganic and organic carbon in the main stem of the Changjiang (Yangtze) River and the effect of the Three Gorges Reservoir. J. Geophys. Res. Biogeosci. 2014, 119, 741–757. [Google Scholar] [CrossRef] [Green Version]
- Raymond, P.A.; Oh, N.H. An empirical study of climatic controls on riverine C export from three major U.S. watersheds. Glob. Biogeochem. Cycles 2007, 21, GB2022. [Google Scholar] [CrossRef] [Green Version]
- Li, X.Y.; Ding, Y.J.; Han, T.D.; Xu, J.Z.; Kang, S.C.; Wu, Q.B.; Sillanpää, M.; Yu, Z.B.; Yu, C.R. Seasonal variations of organic carbon and nitrogen in the upper basins of Yangtze and Yellow Rivers. J. Mt. Sci. 2017, 14, 1577–1590. [Google Scholar] [CrossRef]
- Worrall, F.; Harriman, R.; Evans, C.D.; Watts, C.D.; Adamson, J.; Neal, C.; Tipping, E.; Burt, T.P.; Grieve, I.; Monteith, D.; et al. Trends in Dissolved Organic Carbon in UK Rivers and Lakes. Biogeochemistry 2004, 70, 369–402. [Google Scholar] [CrossRef]
- Clark, J.; Bottrell, S.; Evans, C.; Monteith, D.; Bartlett, R.; Rose, R.; Newton, R.; Chapman, P. The importance of the relationship between scale and process in understanding long-term DOC dynamics. Sci. Total Environ. 2010, 408, 2768–2775. [Google Scholar] [CrossRef] [PubMed]
- Monteith, D.T.; Stoddard, J.; Evans, C.; de Wit, H.A.; Forsius, M.; Høgåsen, T.; Wilander, A.; Skjelkvåle, B.L.; Jeffries, D.S.; Vuorenmaa, J.; et al. Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 2007, 450, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Findlay, S.E. Increased carbon transport in the Hudson River: Unexpected consequence of nitrogen deposition? Front. Ecol. Environ. 2005, 3, 133–137. [Google Scholar] [CrossRef]
- Garnett, M.H.; Ineson, P.; Stevenson, A.C. Effects of burning and grazing on carbon sequestration in a Pennine blanket bog, UK. Holocene 2000, 10, 729–736. [Google Scholar] [CrossRef] [Green Version]
- Evans, C.D.; Chapman, P.J.; Clark, J.M.; Monteith, D.T.; Cresser, M.S. Alternative explanations for rising dissolved organic carbon export from organic soils. Glob. Change Biol. 2006, 12, 2044–2053. [Google Scholar] [CrossRef]
- Zhang, S.; Bai, Y.; Wen, X.; Ding, A.; Zhi, J. Seasonal and downstream alterations of dissolved organic matter and dissolved inorganic ions in a human-impacted mountainous tributary of the Yellow River, China. Environ. Sci. Pollut. Res. 2018, 25, 17967–17979. [Google Scholar] [CrossRef]
- Schuur, E.A.G.; McGuire, A.D.; Schadel, C.; Grosse, G.; Harden, J.W.; Hayes, D.; Hugelius, G.; Koven, C.; Kuhry, P.; Lawrence, D.; et al. Climate change and the permafrost carbon feedback. Nature 2015, 520, 171–179. [Google Scholar] [CrossRef]
- Campeau, A.; Soerensen, A.L.; Martma, T.; Kerblom, S.; Zdanowicz, C. Controls on the 14C content of dissolved and particulate organic carbon mobilized across the mackenzie river basin, Canada. Glob. Biogeochem. Cycles 2020, 34, e2020GB006671. [Google Scholar] [CrossRef]
- Zou, D.; Zhao, L.; Sheng, Y.; Chen, J.; Hu, G.; Wu, T.; Wu, J.; Xie, C.; Wu, X.; Pang, Q.; et al. A new map of permafrost distribution on the Tibetan Plateau. Cryosphere 2017, 11, 2527–2542. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.; Wang, F.; Morgenstern, U.; Zhang, Y.; Grigholm, B.; Kaspari, S.; Schwikowski, M.; Ren, J.; Yao, T.; Qin, D.; et al. Dramatic loss of glacier accumulation area on the Tibetan Plateau revealed by ice core tritium and mercury records. Cryosphere 2015, 9, 1213–1222. [Google Scholar] [CrossRef] [Green Version]
- Wu, T.; Zhao, L.; Li, R.; Wang, Q.; Xie, C.; Pang, Q. Recent ground surface warming and its effects on permafrost on the central Qinghai-Tibet Plateau. Int. J. Clim. 2012, 33, 920–930. [Google Scholar] [CrossRef]
- Wang, X.; Ma, H.; Li, R.; Song, Z.; Wu, J. Seasonal fluxes and source variation of organic carbon transported by two major Chinese Rivers: The Yellow River and Changjiang (Yangtze) River. Glob. Biogeochem. Cycles 2012, 26. [Google Scholar] [CrossRef]
- Mu, C.; Zhang, T.; Wu, Q.; Peng, X.; Cao, B.; Zhang, X.; Cheng, G. Organic carbon pools in permafrost regions on the Qinghai–Xizang (Tibetan) Plateau. Cryosphere 2015, 9, 479–486. [Google Scholar] [CrossRef] [Green Version]
- Köhler, S.; Buffam, I.; Seibert, J.; Bishop, K.; Laudon, H. Dynamics of stream water TOC concentrations in a boreal headwater catchment: Controlling factors and implications for climate scenarios. J. Hydrol. 2009, 373, 44–56. [Google Scholar] [CrossRef]
- Liu, D.; Pan, D.; Bai, Y.; He, X.; Wang, D.; Zhang, L. Variation of dissolved organic carbon transported by two Chinese rivers: The Changjiang River and Yellow River. Mar. Pollut. Bull. 2015, 100, 60–69. [Google Scholar] [CrossRef]
- Ran, L.; Lu, X.X.; Sun, H.; Han, J.; Li, R.; Zhang, J. Spatial and seasonal variability of organic carbon transport in the Yellow River, China. J. Hydrol. 2013, 498, 76–88. [Google Scholar] [CrossRef]
- Xia, X.; Dong, J.; Wang, M.; Xie, H.; Xia, N.; Li, H.; Zhang, X.; Mou, X.; Wen, J.; Bao, Y. Effect of water-sediment regulation of the Xiaolangdi reservoir on the concentrations, characteristics, and fluxes of suspended sediment and organic carbon in the Yellow River. Sci. Total Environ. 2016, 571, 487–497. [Google Scholar] [CrossRef] [Green Version]
- Wen, Z.; Song, K.; Shang, Y.; Lyu, L.; Tao, H.; Liu, G. Natural and anthropogenic impacts on the DOC characteristics in the Yellow River continuum. Environ. Pollut. 2021, 287, 117231. [Google Scholar] [CrossRef]
- Lan, Y.; Zhao, G.; Zhang, Y.; Wen, J.; Liu, J.; Hu, X. Response of runoff in the source region of the Yellow River to climate warming. Quat. Int. 2010, 226, 60–65. [Google Scholar] [CrossRef]
- Li, X.; Ding, Y.; Han, T.; Sillanpää, M.; Jing, Z.; You, X.; Liu, S.; Yang, C.; Yu, C.; Li, G. Seasonal and interannual changes of river chemistry in the source region of Yellow River, Tibetan Plateau. Appl. Geochem. 2020, 119, 104638. [Google Scholar] [CrossRef]
- Liu, S.; Yao, X.; Guo, W.; Xu, J.; Shangguan, D.; Wei, J.; Bao, W.; Wu, L. The contemporary glaciers in China based on the Second Chinese Glacier Inventory. Acta Geogr. Sin.-Chin. Ed. 2015, 70, 3–16. [Google Scholar]
- Li, X.; Ding, Y.; Xu, J.; He, X.; Han, T.; Kang, S.; Wu, Q.; Mika, S.; Yu, Z.; Li, Q. Importance of Mountain Glaciers as a Source of Dissolved Organic Carbon. J. Geophys. Res. Earth Surf. 2018, 123, 2123–2134. [Google Scholar] [CrossRef]
- Li, X.; Ding, Y.; Han, T.; Kang, S.; Yu, Z.; Jing, Z. Seasonal controls of meltwater runoff chemistry and chemical weathering at Urumqi Glacier No.1 in central Asia. Hydrol. Process. 2019, 33, 3258–3281. [Google Scholar] [CrossRef]
- Hood, E.W.; Battin, T.J.; Fellman, J.B.; O’Neel, S.; Spencer, R.G.M. Storage and release of organic carbon from glaciers and ice sheets. Nat. Geosci. 2015, 8, 91–96. [Google Scholar] [CrossRef]
- Bhatia, M.P.; Das, S.B.; Xu, L.; Charette, M.A.; Wadham, J.L.; Kujawinski, E.B. Organic carbon export from the Greenland ice sheet. Geochim. Cosmochim. Acta 2013, 109, 329–344. [Google Scholar] [CrossRef]
- Dittmar, T.; Kattner, G. The biogeochemistry of the river and shelf ecosystem of the Arctic Ocean: A review. Mar. Chem. 2003, 83, 103–120. [Google Scholar] [CrossRef]
- Dai, M.; Yin, Z.; Meng, F.; Liu, Q.; Cai, W.J. Spatial distribution of riverine DOC inputs to the ocean: An updated global synthesis. Curr. Opin. Environ. Sustain. 2012, 4, 170–178. [Google Scholar] [CrossRef]
- Qu, B.; Sillanpää, M.; Li, C.; Kang, S.; Stubbins, A.; Yan, F.; Aho, K.S.; Zhou, F.; Raymond, P.A. Aged dissolved organic carbon exported from rivers of the Tibetan Plateau. PLoS ONE 2017, 12, e0181295. [Google Scholar] [CrossRef]
- Mann, P.; Davydova, A.; Zimov, N.; Spencer, R.G.; Davydov, S.; Bulygina, E.; Zimov, S.; Holmes, R. Controls on the composition and lability of dissolved organic matter in Siberia’s Kolyma River basin. J. Geophys. Res. Biogeosci. 2012, 117. [Google Scholar] [CrossRef] [Green Version]
- Bhandari, R.; Routh, J.; Sharma, S.; Joshi, R. Contrasting lipid biomarkers in mountain rivers in the Nepal Himalayas: Organic matter characteristics and contribution to the fluvial carbon pool. Geosci. Front. 2021, 12, 101231. [Google Scholar] [CrossRef]
- Zeng, Y.; Feng, Z.; Cao, G.; Xue, L. The soil organic carbon storage and its spatial distribution of alpine grassland in the source 529 region of the Yellow River. Acta Geogr. Sin.-Chin. Ed. 2004, 59, 497–504. [Google Scholar]
- Petrone, K.C.; Jones, J.B.; Hinzman, L.D.; Boone, R.D. Seasonal export of carbon, nitrogen, and major solutes from Alaskan catchments with discontinuous permafrost. J. Geophys. Res. Biogeosci. 2006, 111, G2. [Google Scholar] [CrossRef]
- Nkounkou, R.R.; Probst, J.L. Hydrology and geochemistry of the Congo river system. SCOPE/UNEP-Sonderband 1987, 64, 483–508. [Google Scholar]
- Probst, J.; Mortatti, J.; Tardy, Y. Carbon river fluxes and weathering CO2 consumption in the Congo and Amazon river basins. Appl. Geochem. 1994, 9, 1–13. [Google Scholar] [CrossRef]
- Richey, J.E.; Hedges, J.I.; Devol, A.H.; Quay, P.D.; Victoria, R.; Martinelli, L.; Forsberg, B.R. Biogeochemistry of carbon in the Amazon River. Limnol. Oceanogr. 1990, 35, 352–371. [Google Scholar] [CrossRef]
- Holmes, R.M.; McClelland, J.; Peterson, B.J.; Tank, S.; Bulygina, E.; Eglinton, T.I.; Gordeev, V.V.; Gurtovaya, T.Y.; Raymond, P.A.; Repeta, D.J.; et al. Seasonal and Annual Fluxes of Nutrients and Organic Matter from Large Rivers to the Arctic Ocean and Surrounding Seas. Chesap. Sci. 2011, 35, 369–382. [Google Scholar] [CrossRef]
- Kempe, S.; Depetris, P.J. Factors controlling the concentration of particulate carbohydrates and amino acids in the Paraná River. Hydrobiologia 1992, 242, 175–183. [Google Scholar] [CrossRef]
- Ludwig, W.; Probst, J.L.; Kempe, S. Predicting the oceanic input of organic carbon by continental erosion. Glob. Biogeochem. Cycles 1996, 10, 23–41. [Google Scholar] [CrossRef] [Green Version]
- Khosh, M.S.; McClelland, J.W.; Jacobson, A.D.; Douglas, T.A.; Barker, A.J.; Lehn, G.O. Seasonality of dissolved nitrogen from spring melt to fall freeze up in Alaskan Arctic tundra and mountain streams. J. Geophys. Res. Biogeosci. 2017, 122, 1718–1737. [Google Scholar] [CrossRef]
- Hugelius, G.; Strauss, J.; Zubrzycki, S.; Harden, J.W.; Schuur, E.A.G.; Ping, C.-L.; Schirrmeister, L.; Grosse, G.; Michaelson, G.J.; Koven, C.D.; et al. Estimated stocks of circumpolar permafrost carbon with quantified uncertainty ranges and identified data gaps. Biogeosciences 2014, 11, 6573–6593. [Google Scholar] [CrossRef] [Green Version]
- Immerzeel, W.W.; van Beek, L.P.H.; Bierkens, M.F.P. Climate Change Will Affect the Asian Water Towers. Science 2010, 328, 1382–1385. [Google Scholar] [CrossRef]
- Yang, X.; Yao, T.; Joswiak, D.; Yao, P. Integration of Tibetan Plateau ice-core temperature records and the influence of atmospheric circulation on isotopic signals in the past century. Quat. Res. 2014, 81, 520–530. [Google Scholar] [CrossRef]
- Jin, H.; He, R.; Cheng, G.; Wu, Q.; Wang, S.; Chang, X. Changes in frozen ground in the source area of the yellow river on the Qinghai-Tibet Plateau, China, and their eco-environmental impacts. Environ. Res. Lett. 2009, 4, 045206. [Google Scholar] [CrossRef]
- Pohl, E.; Gloaguen, R.; Andermann, C.; Knoche, M. Glacier melt buffers river runoff in the Pamir Mountains. Water Resour. Res. 2017, 53, 2467–2489. [Google Scholar] [CrossRef]
- Kraaijenbrink, P.; Bierkens, M.F.; Lutz, A.; Immerzeel, W. Impact of a global temperature rise of 1.5 degrees Celsius on Asia’s glaciers. Nature 2017, 549, 257–260. [Google Scholar] [CrossRef]
- Lutz, A.; Immerzeel, W.; Shrestha, A.B.; Bierkens, M.F. Consistent increase in High Asia’s runoff due to increasing glacier melt and precipitation. Nat. Clim. Chang. 2014, 4, 587–592. [Google Scholar] [CrossRef] [Green Version]
- Pritchard, H.D. Asia’s glaciers are a regionally important buffer against drought. Nature 2017, 545, 169–174. [Google Scholar] [CrossRef]
- Romanovsky, V.E.; Drozdov, D.S.; Oberman, N.G.; Malkova, G.V.; Kholodov, A.L.; Marchenko, S.; Moskalenko, N.G.; Sergeev, D.O.; Ukraintseva, N.G.; Abramov, A.; et al. Thermal state of permafrost in Russia. Permafr. Periglac. Process. 2010, 21, 136–155. [Google Scholar] [CrossRef]
- Frey, K.E.; McClelland, J.W. Impacts of permafrost degradation on arctic river biogeochemistry. Hydrol. Process. 2008, 23, 169–182. [Google Scholar] [CrossRef]
- Zhang, S.; Gan, W.B.; Ittekkot, V. Organic matter in large turbid rivers: The Huanghe and its estuary. Mar. Chem. 1992, 38, 53–68. [Google Scholar] [CrossRef]
- Leenheer, J. United States geological survey data information service. In Transport of Carbon and Minerals in Major World Rivers. Part I; Geologisch-Palaontologischen Institut der Universitat Hamburg: Hamburg, Germany, 1982; Volume 1, pp. 355–356. [Google Scholar]
- Hart, R. Carbon transport in the upper Orange River. In Transport of Carbon and Minerals in Major World Rivers. Part I; Geologisch-Palaontologischen Institut der Universitat Hamburg: Hamburg, Germany, 1987; Volume 4, pp. 509–512. [Google Scholar]
- Dahm, C.N.; Gregory, S.V.; Park, P.K. Organic carbon transport in the Columbia River. Estuar. Coast. Shelf Sci. 1981, 13, 645–658. [Google Scholar] [CrossRef]
- Abu El Ella, E. Preliminary studies on the geochemistry of the Nile river basin, Egypt. In Transport of Carbon and Minerals in Major World Rivers; Geologisch-Palaontologischen Institut der Universitat Hamburg: Hamburg, Germany, 1993; Volume 6, pp. 115–135. [Google Scholar]
- Lesack, L.F.W.; Hecky, R.E.; Melack, J.M. Transport of carbon, nitrogen, phosphorus, and major solutes in the Gambia River, West Africa. Limnol. Oceanogr. 1984, 29, 816–830. [Google Scholar] [CrossRef]
- Li, C.; Yan, F.; Kang, S.; Chen, P.; Qu, B.; Hu, Z.; Sillanpää, M. Concentration, sources, and flux of dissolved organic carbon of precipitation at Lhasa city, the Tibetan Plateau. Environ. Sci. Pollut. Res. 2016, 23, 12915–12921. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, Y.; Xin, J.; Tang, G.; Song, T.; Wang, Y.; Li, X.; Wu, F. Study on dissolved organic carbon in precipitation in Northern China. Atmos. Environ. 2010, 44, 2350–2357. [Google Scholar] [CrossRef]
- Huang, J.; Kang, S.; Shen, C.; Cong, Z.; Liu, K.; Wang, W.; Liu, L. Seasonal variations and sources of ambient fossil and biogenic-derived carbonaceous aerosols based on 14C measurements in Lhasa, Tibet. Atmos. Res. 2010, 96, 553–559. [Google Scholar] [CrossRef]
- Yan, G.; Kim, G. Dissolved organic carbon in the precipitation of Seoul, Korea: Implications for global wet depositional flux of fossil-fuel derived organic carbon. Atmos. Environ. 2012, 59, 117–124. [Google Scholar] [CrossRef]
- Coelho, C.H.; Francisco, J.G.; Nogueira, R.P.; Campos, M.L.A.M. Dissolved organic carbon in rainwater from areas heavily impacted by sugar cane burning. Atmos. Environ. 2008, 42, 7115–7121. [Google Scholar] [CrossRef]
- Yde, J.C.; Knudsen, N.T.; Nielsen, O.B. Glacier hydrochemistry, solute provenance, and chemical denudation at a surge-type glacier in Kuannersuit Kuussuat, Disko Island, West Greenland. J. Hydrol. 2005, 300, 172–187. [Google Scholar] [CrossRef]
- Hodgkins, R.; Tranter, M.; Dowdeswell, J.A. Solute provenance, transport and denudation in a high arctic glacierized catchment. Hydrol. Process. 1997, 11, 1813–1832. [Google Scholar] [CrossRef]
- Yu, Z.; Wu, G.; Keys, L.; Li, F.; Yan, N.; Qu, D.; Liu, X. Seasonal variation of chemical weathering and its controlling factors in two alpine catchments, Nam Co basin, central Tibetan Plateau. J. Hydrol. 2019, 576, 381–395. [Google Scholar] [CrossRef]
- Evans, A.; Zelazny, L.W.; Zipper, C. Solution parameters influencing dissolved organic carbon levels in three forest soils. Soil Sci. Soc. Am. J. 1988, 52, 1789–1792. [Google Scholar] [CrossRef]
- Tipping, E.; Woof, C. The distribution of humic substances between the solid and aqueous phases of acid organic soils; a description based on humic heterogeneity and charge-dependent sorption equilibria. J. Soil Sci. 1991, 42, 437–448. [Google Scholar] [CrossRef]
| 2013 | 2014 | 2015 | |
|---|---|---|---|
| DOC concentration in river | 2.3 ± 1.1 | 2.2 ± 1.3 | 2.0 ± 0.5 |
| DOC concentration in precipitation | 2.0 ± 1.4 | 2.8 ± 2.5 | 1.8 ± 1.2 |
| DOC flux from river | 26,058.2 ± 174.1 | 37,539.6 ± 100.1 | 24,629.3 ± 153.2 |
| DOC deposition flux from precipitation | 86,080.1 ± 451.3 | 105,804.5 ± 887.4 | 73,072.6 ± 364.2 |
| DOC yield | 0.2 | 0.4 | 0.3 |
| C | F | P | Q | T | Na+ | K+ | Mg2+ | Ca2+ | Cl− | SO42− | NO3− | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | 1.00 | 0.72 * | 0.16 | 0.50 * | 0.55 * | −0.01 | −0.16 * | −0.06 | −0.20 * | −0.41 * | −0.19 * | −0.51 * |
| F | 1.00 | 0.03 | 0.94 * | 0.54 * | −0.27 * | −0.10 | −0.12 | −0.03 | −0.49 * | −0.17 * | −0.36 * | |
| P | 1.00 | −0.04 | 0.11 | −0.18 | −0.17 | −0.17 | −0.04 | −0.13 | −0.16 | −0.19 | ||
| Q | 1.00 | 0.57 * | −0.38 * | −0.04 | −0.23 * | −0.01 | −0.52 * | −0.11 | −0.23 * | |||
| T | 1.00 | −0.37 * | −0.05 | −0.32 * | −0.39 * | −0.48 * | −0.01 | −0.29 * | ||||
| Na+ | 1.00 | 0.73 * | 0.90 * | 0.29 * | 0.51 * | 0.17 * | 0.04 | |||||
| K+ | 1.00 | 0.80 * | 0.48 * | 0.17 * | 0.08 | 0.03 | ||||||
| Mg2+ | 1.00 | 0.52 * | 0.37 * | 0.12 | 0.04 | |||||||
| Ca2+ | 1.00 | 0.01 | −0.04 | 0.27 * | ||||||||
| Cl− | 1.00 | 0.71 * | 0.27 * | |||||||||
| SO42− | 1.00 | 0.44 * | ||||||||||
| NO3− | 1.00 |
| Rivers | Area (103 km2) | Flux (103 tons/year) | Yields (tons/km2/year) | Temperature Zone | Source |
|---|---|---|---|---|---|
| Yellow | 823 | 396 | 4.81 | Temperate wet | [71] |
| Changjiang | 1817 | 1580 | 0.87 | Temperate wet | [33] |
| Ob’ | 2990 | 4119 | 1.40 | Tundra and Taiga | [57] |
| Yukon | 830 | 1472 | 1.77 | Tundra and Taiga | [57] |
| Mackenzie | 1780 | 1377 | 0.82 | Tundra and Taiga | [57] |
| Orinoco | 1000 | 3280 | 3.28 | Tropical wet | [7] |
| Amazon | 5903 | 26,333 | 4.46 | Tropical wet | [55] |
| Zaire | 3704 | 9130 | 2.47 | Tropical wet | [54] |
| Mississippi | 3243 | 4278 | 1.32 | Temperate wet | [72] |
| Orange | 716 | 179 | 0.25 | Tropical dry | [73] |
| Brazos | 127 | 31 | 0.24 | Tropical dry | [74] |
| Nile | 1874 | 167 | 0.09 | Tropical dry | [75] |
| SRYR | 122 | 29 | 0.31 | Sub cold zone dry | This study |
| Gambia | 63 | 17 | 0.26 | Tropical dry | [76] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
You, X.; Li, X. Seasonal Variations in Dissolved Organic Carbon in the Source Region of the Yellow River on the Tibetan Plateau. Water 2021, 13, 2901. https://doi.org/10.3390/w13202901
You X, Li X. Seasonal Variations in Dissolved Organic Carbon in the Source Region of the Yellow River on the Tibetan Plateau. Water. 2021; 13(20):2901. https://doi.org/10.3390/w13202901
Chicago/Turabian StyleYou, Xiaoni, and Xiangying Li. 2021. "Seasonal Variations in Dissolved Organic Carbon in the Source Region of the Yellow River on the Tibetan Plateau" Water 13, no. 20: 2901. https://doi.org/10.3390/w13202901
APA StyleYou, X., & Li, X. (2021). Seasonal Variations in Dissolved Organic Carbon in the Source Region of the Yellow River on the Tibetan Plateau. Water, 13(20), 2901. https://doi.org/10.3390/w13202901






