Adsorption Kinetics and Isotherm Study of Basic Red 5 on Synthesized Silica Monolith Particles
Abstract
:1. Introduction
2. Material and Methods
2.1. Chemicals and Instrumentation
2.2. Synthesis of Silica Monolith Particles
2.3. Adsorption Studies
2.3.1. Kinetic Studies
2.3.2. Isothermal Studies
2.3.3. Effect of Temperature
3. Results and Discussion
3.1. Characterization
3.1.1. Scanning Electron Microscopy (SEM)
3.1.2. Electron Dispersive X-rays (EDX)
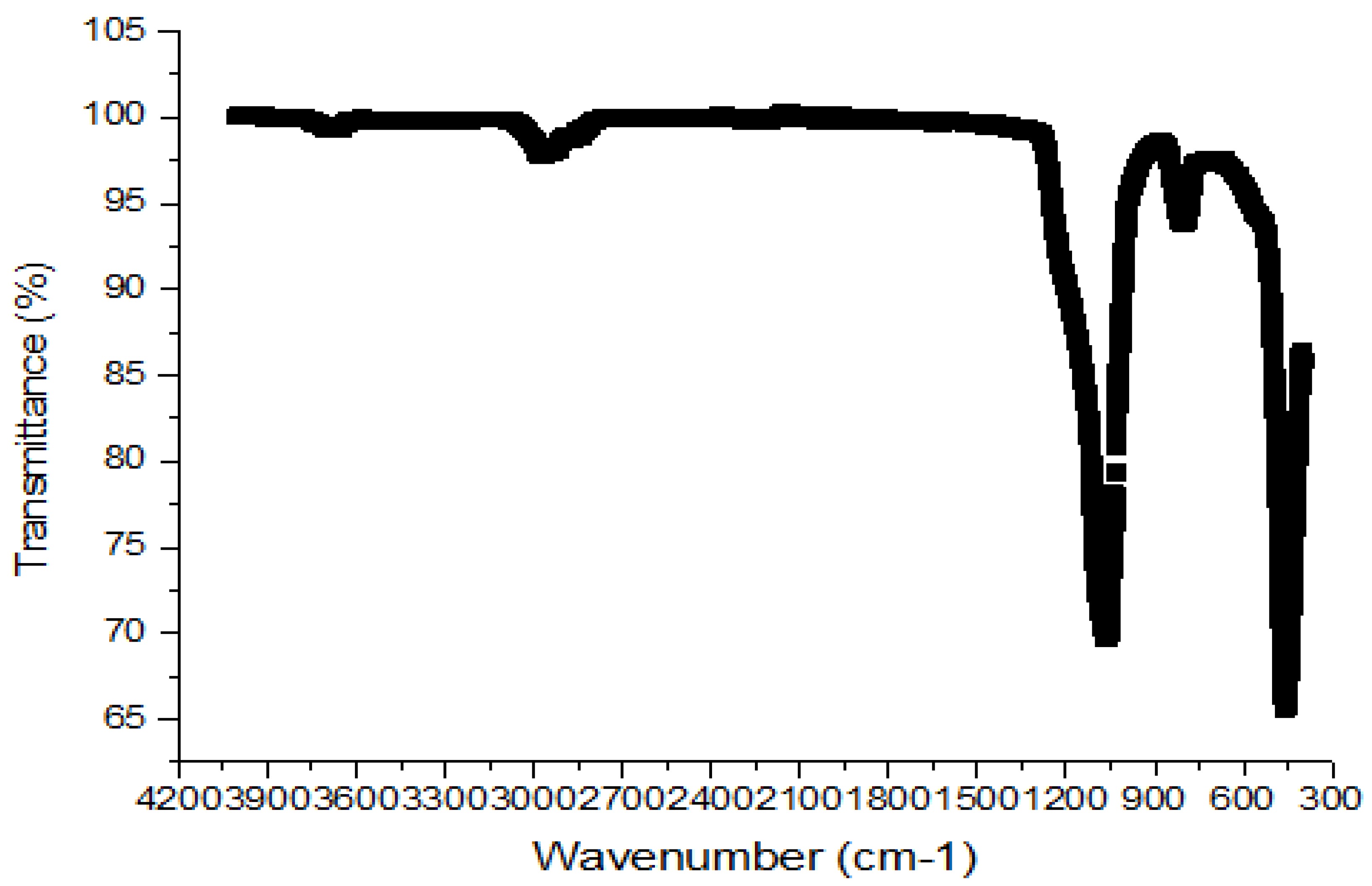
3.1.3. Fourier-Transform Infrared Spectroscopy (FTIR)
3.1.4. Surface Area of SMP
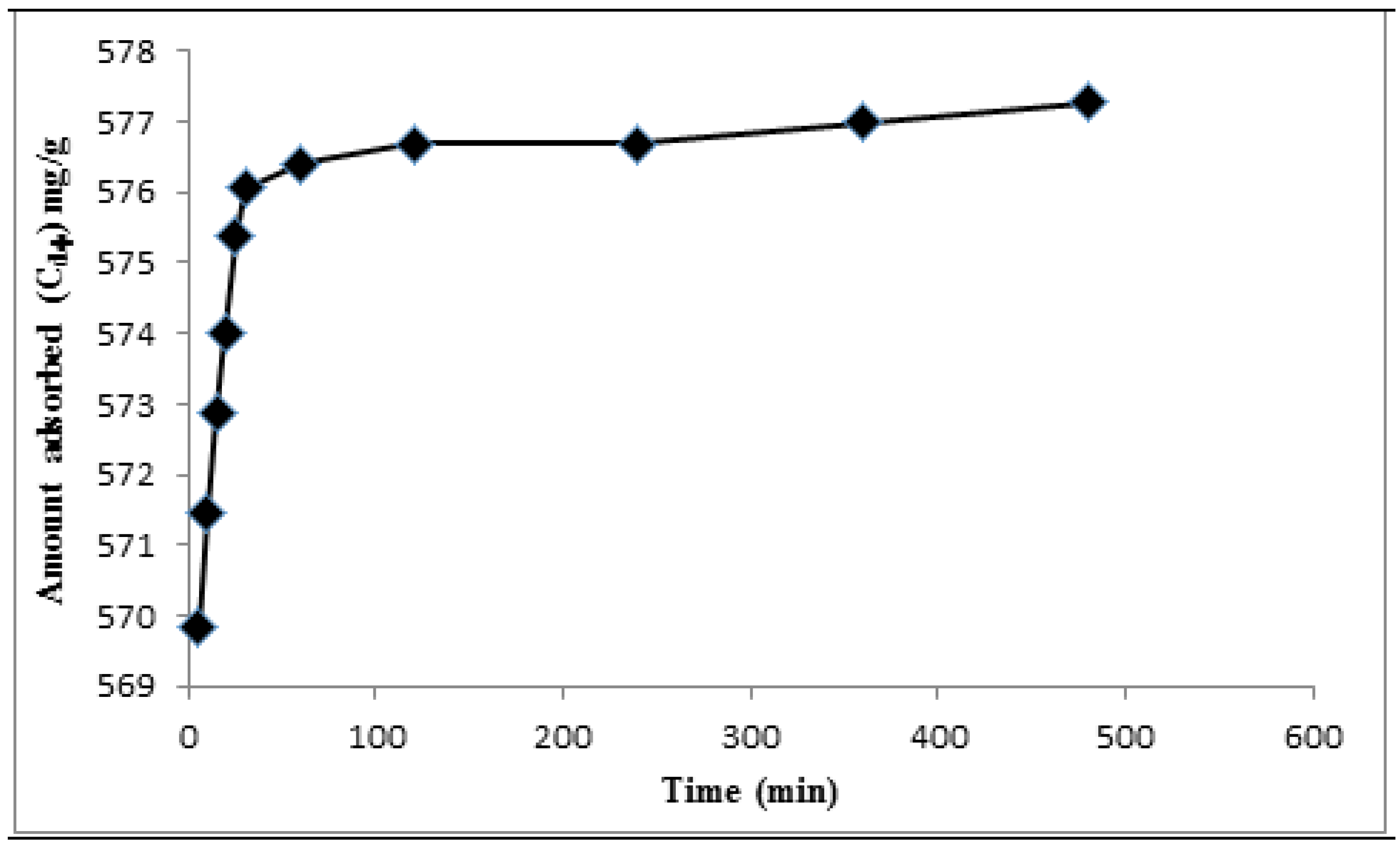
3.2. Adsorption Kinetics Study
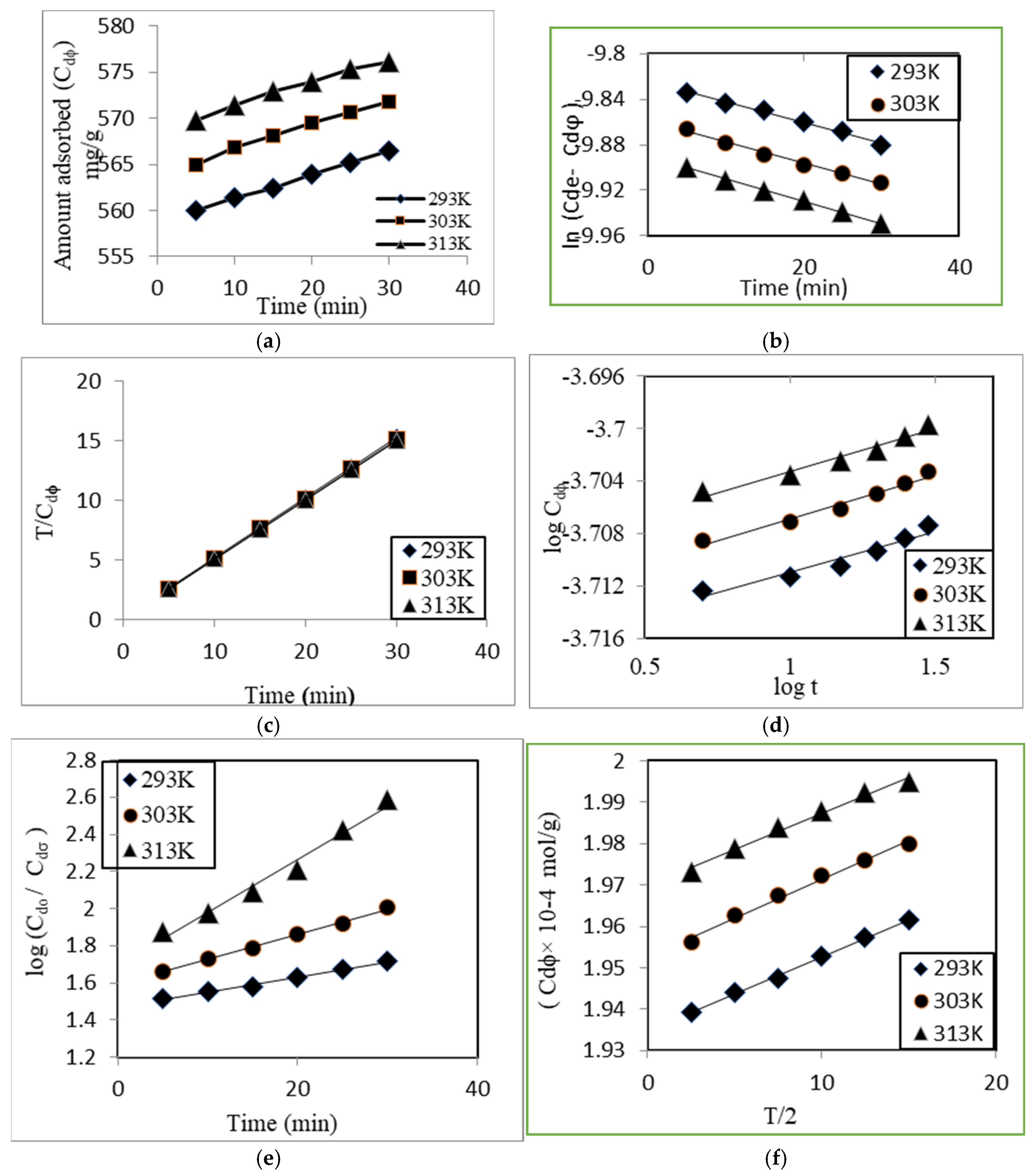
3.2.1. Pseudo 1st Order Kinetics
3.2.2. Pseudo 2nd Order Kinetics
3.2.3. Intraparticle Diffusion Model
3.2.4. Power Function Model
3.2.5. Natarajan and Khalaf Model
3.3. Isotherm Models
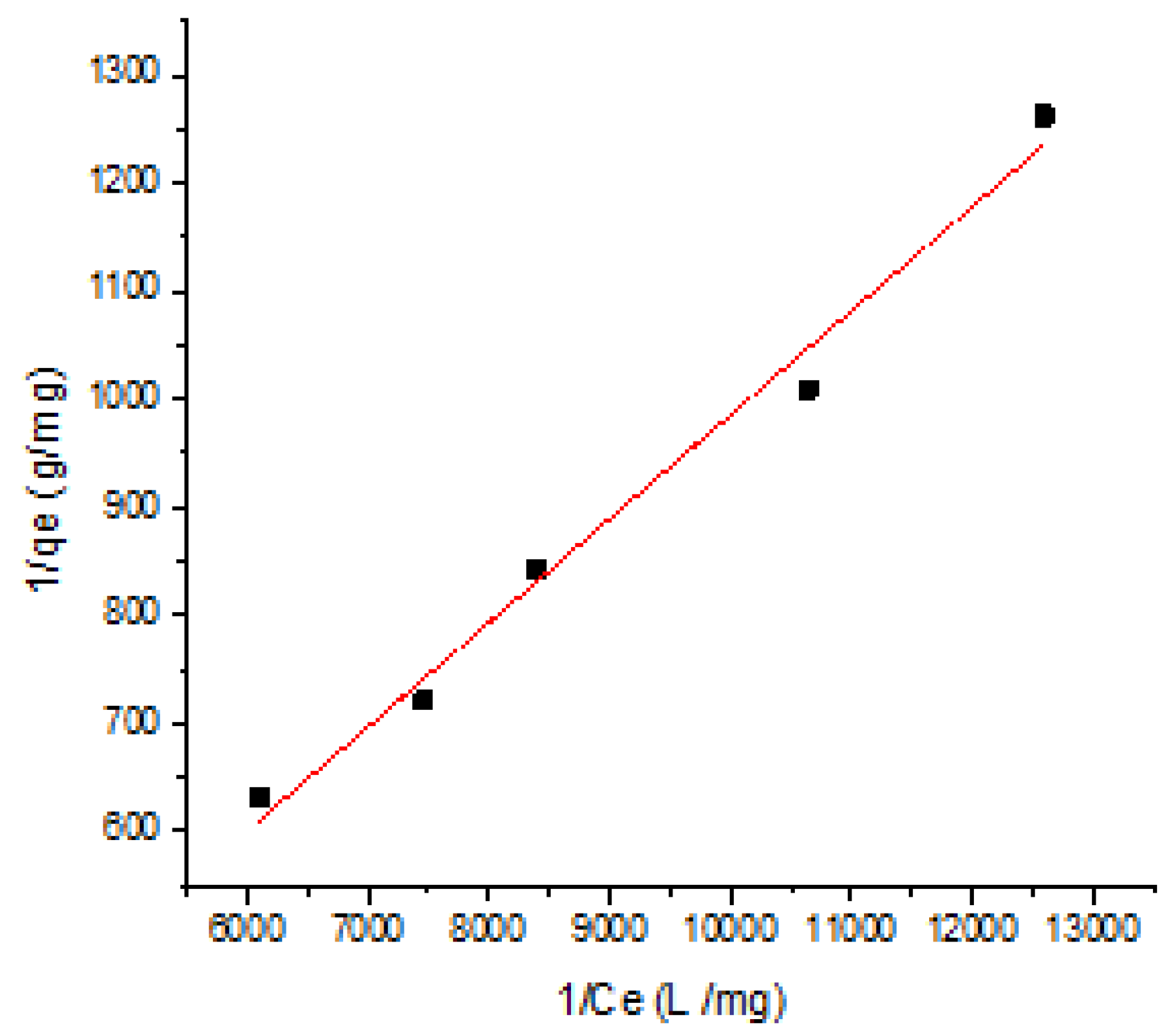
3.3.1. Langmuir Isotherm Model
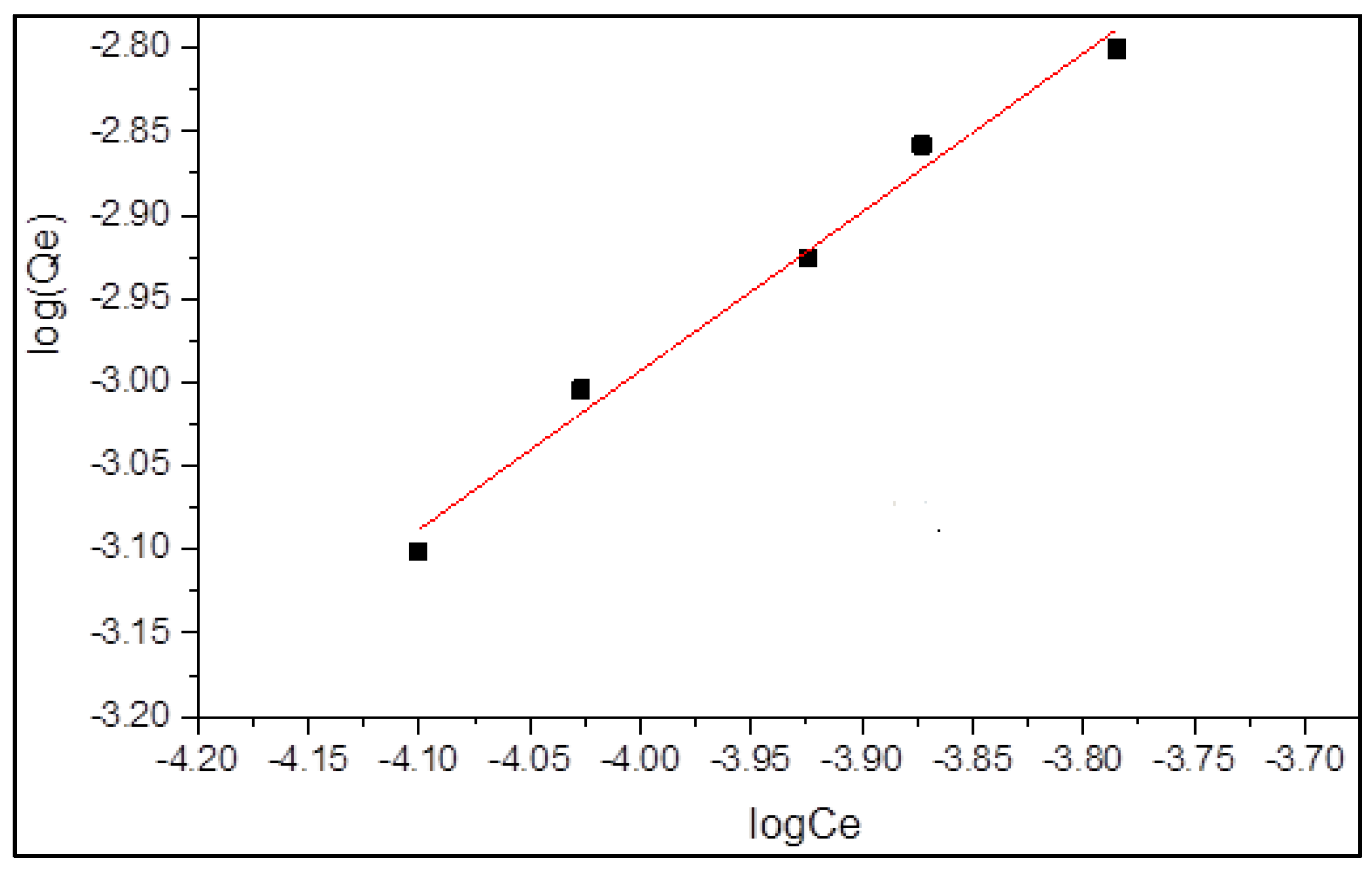
3.3.2. Freundlich Isotherm Model
3.4. Thermodynamic Parameters Study
3.5. Regeneration
3.6. Comparison of SMPs Adsorption Capacity with Other Adsorbents
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vivero-Escoto, J. Silica Monolith Particles: Preparation, Properties and Uses; Nova Science Publishers, Incorporated: Hauppauge, NY, USA, 2011. [Google Scholar]
- Wang, P.; Zakeeruddin, S.M.; Comte, P.; Exnar, I.; Grätzel, M. Gelation of ionic liquid-based electrolytes with silica monolith particles for quasi-solid-state dye-sensitized solar cells. J. Am. Chem. Soc. 2003, 125, 1166–1167. [Google Scholar] [CrossRef]
- Slowing, I.I.; Vivero-Escoto, J.L.; Wu, C.-W.; Lin, V.S.-Y. Mesoporous silica monolith particles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev. 2008, 60, 1278–1288. [Google Scholar] [CrossRef]
- Torney, F.; Trewyn, B.G.; Lin, V.S.-Y.; Wang, K. Mesoporous silica monolith particles deliver DNA and chemicals into plants. Nat. Nanotechnol. 2007, 2, 295. [Google Scholar] [CrossRef]
- Lu, J.; Liong, M.; Zink, J.I.; Tamanoi, F. Mesoporous silica monolith particles as a delivery system for hydrophobic anticancer drugs. Small 2007, 3, 1341–1346. [Google Scholar] [CrossRef]
- Chiron, N.; Guilet, R.; Deydier, E. Adsorption of Cu (II) and Pb (II) onto a grafted silica: Isotherms and kinetic models. Water Res. 2003, 37, 3079–3086. [Google Scholar] [CrossRef]
- Vansant, E.F.; van der Voort, P.; Vrancken, K.C. Characterization and Chemical Modification of the Silica Surface; Elsevier: Amsterdam, The Netherlands, 1995; Volume 93. [Google Scholar]
- Kresge, C.; Leonowicz, M.; Roth, W.J.; Vartuli, J.; Beck, J. Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 1992, 359, 710. [Google Scholar] [CrossRef]
- Cue, B.W.; Zhang, J. Green process chemistry in the pharmaceutical industry. Green Chem. Lett. Rev. 2009, 2, 193–211. [Google Scholar] [CrossRef]
- Akbal, F. Adsorption of basic dyes from aqueous solution onto pumice powder. J. Colloid Interface Sci. 2005, 286, 455–458. [Google Scholar] [CrossRef]
- Margaret, M.A.; Cullen, J.; Giles, C.; Jain, S.; Woods, J. Adsorption of dyes and related compounds by Silica J. Appl. Chem. 1958, 8, 108–116. [Google Scholar]
- Donia, A.M.; Atia, A.A.; Al-amrani, W.A.; El-Nahas, A.M. Effect of structural properties of acid dyes on their adsorption behaviour from aqueous solutions by amine modified silica. J. Hazard. Mater. 2009, 161, 1544–1550. [Google Scholar] [CrossRef]
- Ahmed, M.; Ram, R. Removal of basic dye from waste-water using silica as adsorbent. Environ. Pollut. 1992, 77, 79–86. [Google Scholar] [CrossRef]
- Mckay, G. The adsorption of basic dye onto silica from aqueous solution-solid diffusion model. Chem. Eng. Sci. 1984, 39, 129–138. [Google Scholar] [CrossRef]
- Ali, A.; Ali, F.; Cheong, W.J. Sedimentation assisted preparation of ground particles of silica monolith and their C18 modification resulting in a chromatographic phase of improved separation efficiency. J. Chromatogr. A 2017, 1525, 79–86. [Google Scholar] [CrossRef]
- Gelb, L.D.; Gubbins, K.E. Characterization of porous glasses: Simulation models, adsorption isotherms, and the Brunauer− Emmett− Teller analysis method. Langmuir 1998, 14, 2097–2111. [Google Scholar] [CrossRef]
- Vadivelan, V.; Kumar, K.V. Equilibrium, kinetics, mechinisium, and process design for the sorption of methyleneblue onto rise husk. Colloid Interf. Sci. 2005, 286, 90–100. [Google Scholar] [CrossRef]
- Nidheesh, P.V.; Gandhimathi, R.; Ramesh, S.T.; Singh, T.S.A. Kinetic analysis of crystal violet adsorption on to bottom ash. Turkish J. Eng. Env. Sci. 2012, 36, 249–262. [Google Scholar]
- Luo, J.; Yu, D.; Hristovski, K.D.; Fu, K.; Shen, Y.; Westerhoff, P.; Crittenden, J.C. Critical review of advances in engineering nanomaterial adsorbents for metal removal and recovery from water: Mechanism identification and engineering design. Env. Sci. Tech. 2021, 55, 4287–4304. [Google Scholar] [CrossRef]
- Hensen, E.J.M.; Dejongand, A.M.; van Santen, R.A. Adsorption and Diffusion; Karge, H.G., Weitkamp, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 277–328. [Google Scholar]
- Rahman, N.U.; Ullah, I.; Alam, S.; Khan, M.S.; Shah, L.A.; Zekker, I.; Burlakovs, J.; Kallistova, A.; Pimenov, N.; Vincevica-Gaile, Z.; et al. Activated Ailanthus altissima Sawdust as Adsorbent for Removal of Acid Yellow 29 from Wastewater: Kinetics Approach. Water 2021, 13, 2136. [Google Scholar] [CrossRef]
- Alam, S.; Khan, M.S.; Bibi, W.; Zekker, I.; Burlakovs, J.; Ghangrekar, M.M.; Bhowmick, G.D.; Kallistova, A.; Pimenov, N.; Zahoor, M. Preparation of Activated Carbon from the Wood of Paulownia tomentosa as an Efficient Adsorbent for the Removal of Acid Red 4 and Methylene Blue Present in Wastewater. Water 2021, 13, 1453. [Google Scholar] [CrossRef]
- Alam, S.; Khan, M.S.; Umar, A.; Khattak, R.; Rahman Nu Zekker, I.; Burlakovs, J.; Rubin SSd Ghangrekar, M.M.; Bhowmick, G.D.; Kallistova, A.; Pimenov, N.; et al. Preparation of Pd–Ni Nanoparticles Supported on Activated Carbon for Efficient Removal of Basic Blue 3 from Water. Water 2021, 13, 1211. [Google Scholar] [CrossRef]
- Umar, A.; Khan, M.S.; Alam, S.; Zekker, I.; Burlakovs, J.; Rubin, S.D.; Bhowmick, G.; Kallistova, A.; Pimenov, N.; Zahoor, M. Synthesis and Characterization of Pd-Ni Bimetallic Nanoparticles as Efficient Adsorbent for the Removal of Acid Orange 8 Present in Wastewater. Water 2021, 13, 1095. [Google Scholar] [CrossRef]
- Do, D.D. Adsorption Analysis: Equilibria and Kinetics; Imperial College Press: London, UK, 1998. [Google Scholar]
- Suzuki, M. Adsorption Engineering; Kodansha Ltd.: Tokyo, Japan; Elsevier Science Publishers: Amsterdam, The Netherlands, 1990. [Google Scholar]
- Anirudha, T.S.; Radhakrishana, P.G. Thermodynamics and kinetics of adsorption of Cu(II) from aqueous solutions onto a new cation exchanger derived from tamarind fruit shell. J. Chem. Themodyn. 2008, 40, 702–709. [Google Scholar] [CrossRef]
- Chowdhury, S.; Mishra, R.R.; Saha, P.; Kushwaha, P. Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk. Desalination 2011, 265, 159–168. [Google Scholar] [CrossRef]
- Sheen, S.; Pan, T.; Liu, X.; Yuan, L.; Zhag, Y.; Wang, J.; Guo, Z. Adsorption of Pd(II) complexes from chloride solutions obtained by leaching chlorinated spent automotive catalysts on ion exchangers in Diaion. J. Colloid Interface Sci. 2010, 345, 12–18. [Google Scholar] [CrossRef]
- Mane, V.S.; Babu, P.V. Studies on the adsorption of Brilliant Green dye from aqueous solution onto low-cost NaOH treated saw dust. Desalination 2011, 273, 321–329. [Google Scholar] [CrossRef]
- Deniz, F.; Karaman, S. Removal of Basic Red 46 dye from aqueous solution by pine tree leaves. Chem. Eng. J. 2011, 170, 67–74. [Google Scholar] [CrossRef]
- Aichour, A.; Zaghouane-Boudiaf, H.; Iborra, C.V.; Polo, M.S. Bioadsorbent beads prepared from activated biomass/alginate for enhanced removal of cationic dye from water medium: Kinetics, equilibrium and thermodynamic studies. J. Mol. Liq. 2018, 256, 533–540. [Google Scholar] [CrossRef]
- Dutta, S.; Gupta, B.; Srivastava, S.K.; Gupta, A.K. Recent advances on the removal of dyes from wastewater using various adsorbents: A critical review. Environ. Pollut. 2019, 252, 352–365. [Google Scholar]
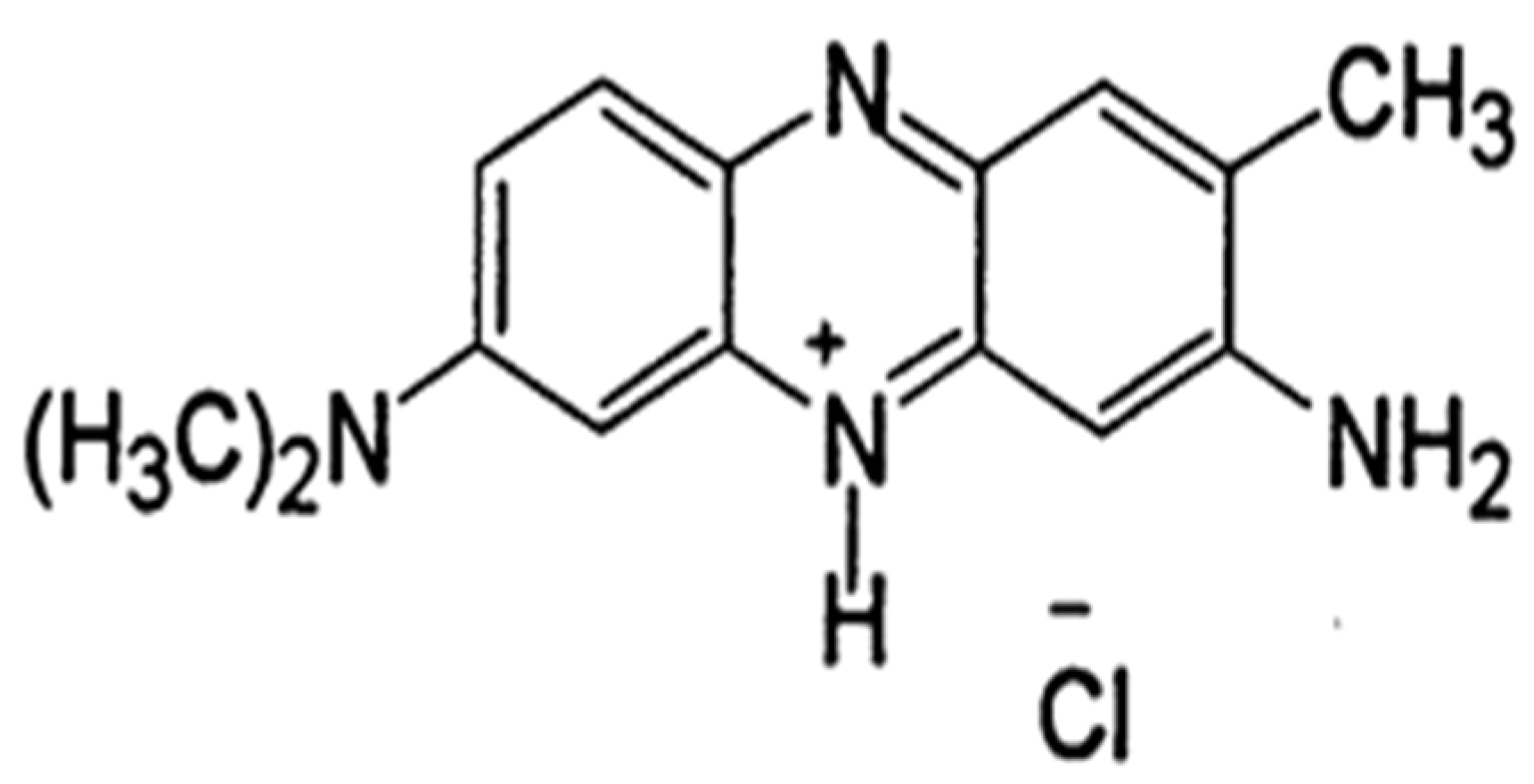

| Name of Dye | Basic Red 5 |
|---|---|
| (M.F) Molecular Formula | C15H17ClN4 |
| (M.W) Molecular Weight | 288.78 g/mol |
| UV- visible λmax | 540 nm |
| Dye content | >90% |
| Water solubility | Soluble in water |
| Element | Weight% | Atomic% |
|---|---|---|
| O | 47.87 | 61.72 |
| Si | 52.13 | 38.28 |
| Total | 100 | 100 |
| Parameters | Adsorption Temperature | |||||||
|---|---|---|---|---|---|---|---|---|
| 293 K | 303 K | 313 K | ||||||
| Units | value | R2 | value | R2 | value | R2 | ||
| Pseudo 1st order | k1 | Min−1 | 0.00180 | 0.997 | 0.00185 | 0.991 | 0.00195 | 0.988 |
| Pseudo 2nd order | k2 | g.mg−1.min−1 | 29787.5 | 1 | 30568.5 | 1 | 30597.0 | 1 |
| Power function | α × 10−3 | 0.1918 | 0.1936 | 0.1954 | ||||
| β | 0.00760 | 0.00691 | 0.00624 | |||||
| R2 | 0.932 | 0.974 | 0.958 | |||||
| Natrajan and Khalaf | Kn | 0.016121 | 0.029939 | 0.064484 | ||||
| Intercept | 1.472 | 1.589 | 1.691 | |||||
| R2 | 0.992 | 0.997 | 0.982 | |||||
| Intraparticle diffusion | Kip | 0.00179 | 0.00187 | 0.00191 | ||||
| Intercept | 1.9347 | 1.9527 | 1.9698 | |||||
| R2 | 0.997 | 0.990 | 0.987 | |||||
| Langmuir Adsorption Isotherm Model | ||||
| intercept | slope | qmax (mg/g) | KL (g/mg) | R2 |
| 23.52 | 0.096 | 0.0425 | 0.44 | 0.988 |
| Freundlich Adsorption Isotherm Model | ||||
| intercept | slope | 1/n | Kf (mg/g) | R2 |
| 0.804 | 0.949 | 0.949 | 6.37 | 0.982 |
| ∆G° (KJ/mol) | ∆H° (KJ/mol) | ∆S° (KJ/mol·K) | ΔEa (kJ/mol) | ||
|---|---|---|---|---|---|
| 293 K | 303 K | 313 K | 0.995 | −297 | 3.431 |
| 87.983 | 89.976 | 93.739 | |||
| Adsorbents | Dyes | Adsorption Capacity (mg/g) | Reference |
|---|---|---|---|
| NaOH treated saw dust | Brilliant Green | 55.86 | [30] |
| Pine tree leaves | Basic Red 46 | 71.94 | [31] |
| Lemonpeels/sodium alginate (Activated Carbon) | Methylene Blue | 841.37 | [32] |
| Rice husk | Methylene Blue | 1350 | [33] |
| Silica monolith particles | Basic Red-5 | 0.0425 | Present work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, S.; Ullah, B.; Khan, M.S.; Rahman, N.u.; Khan, L.; Shah, L.A.; Zekker, I.; Burlakovs, J.; Kallistova, A.; Pimenov, N.; et al. Adsorption Kinetics and Isotherm Study of Basic Red 5 on Synthesized Silica Monolith Particles. Water 2021, 13, 2803. https://doi.org/10.3390/w13202803
Alam S, Ullah B, Khan MS, Rahman Nu, Khan L, Shah LA, Zekker I, Burlakovs J, Kallistova A, Pimenov N, et al. Adsorption Kinetics and Isotherm Study of Basic Red 5 on Synthesized Silica Monolith Particles. Water. 2021; 13(20):2803. https://doi.org/10.3390/w13202803
Chicago/Turabian StyleAlam, Sultan, Barkat Ullah, Muhammad Sufaid Khan, Najeeb ur Rahman, Luqman Khan, Luqman Ali Shah, Ivar Zekker, Juris Burlakovs, Anna Kallistova, Nikolai Pimenov, and et al. 2021. "Adsorption Kinetics and Isotherm Study of Basic Red 5 on Synthesized Silica Monolith Particles" Water 13, no. 20: 2803. https://doi.org/10.3390/w13202803
APA StyleAlam, S., Ullah, B., Khan, M. S., Rahman, N. u., Khan, L., Shah, L. A., Zekker, I., Burlakovs, J., Kallistova, A., Pimenov, N., Yandri, E., Hendroko Setyobudi, R., Jani, Y., & Zahoor, M. (2021). Adsorption Kinetics and Isotherm Study of Basic Red 5 on Synthesized Silica Monolith Particles. Water, 13(20), 2803. https://doi.org/10.3390/w13202803








