The Effects of Temperature, Light, and Feeding on the Physiology of Pocillopora damicornis, Stylophora pistillata, and Turbinaria reniformis Corals
Abstract
1. Introduction
2. Materials and Methods
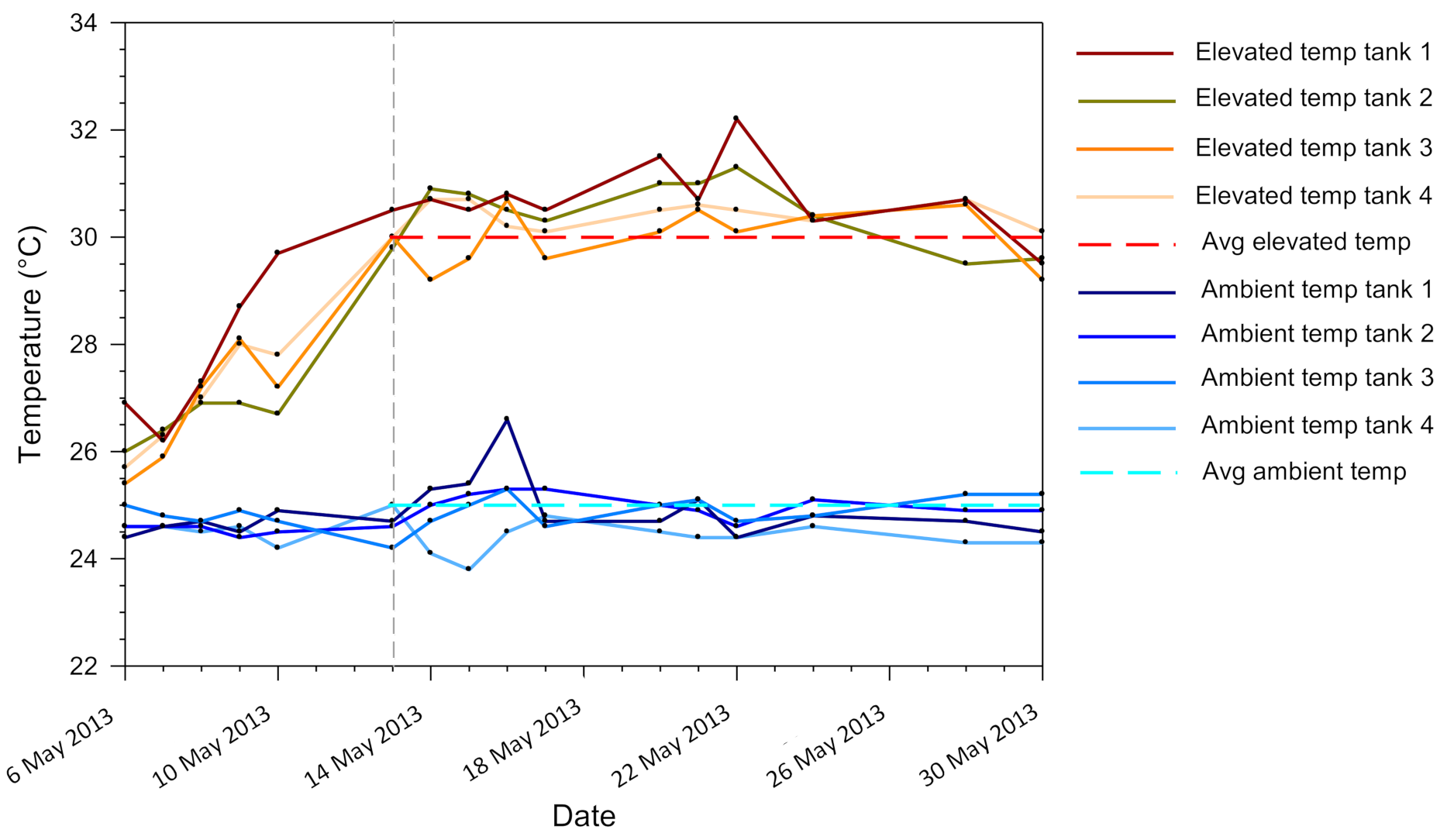
2.1. Experimental Design
2.2. Host and Symbiont Physiology
2.3. Statistical Analyses
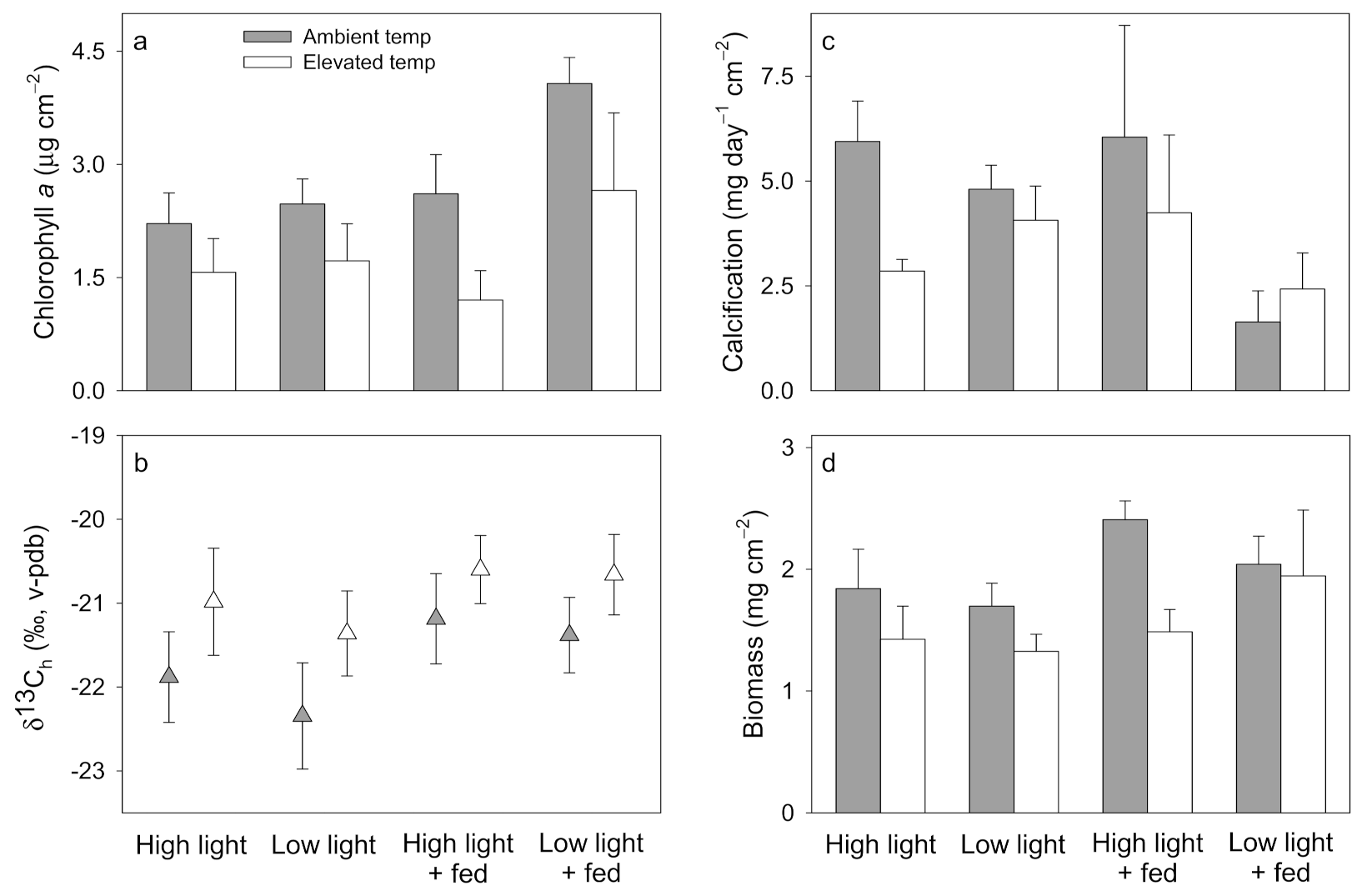
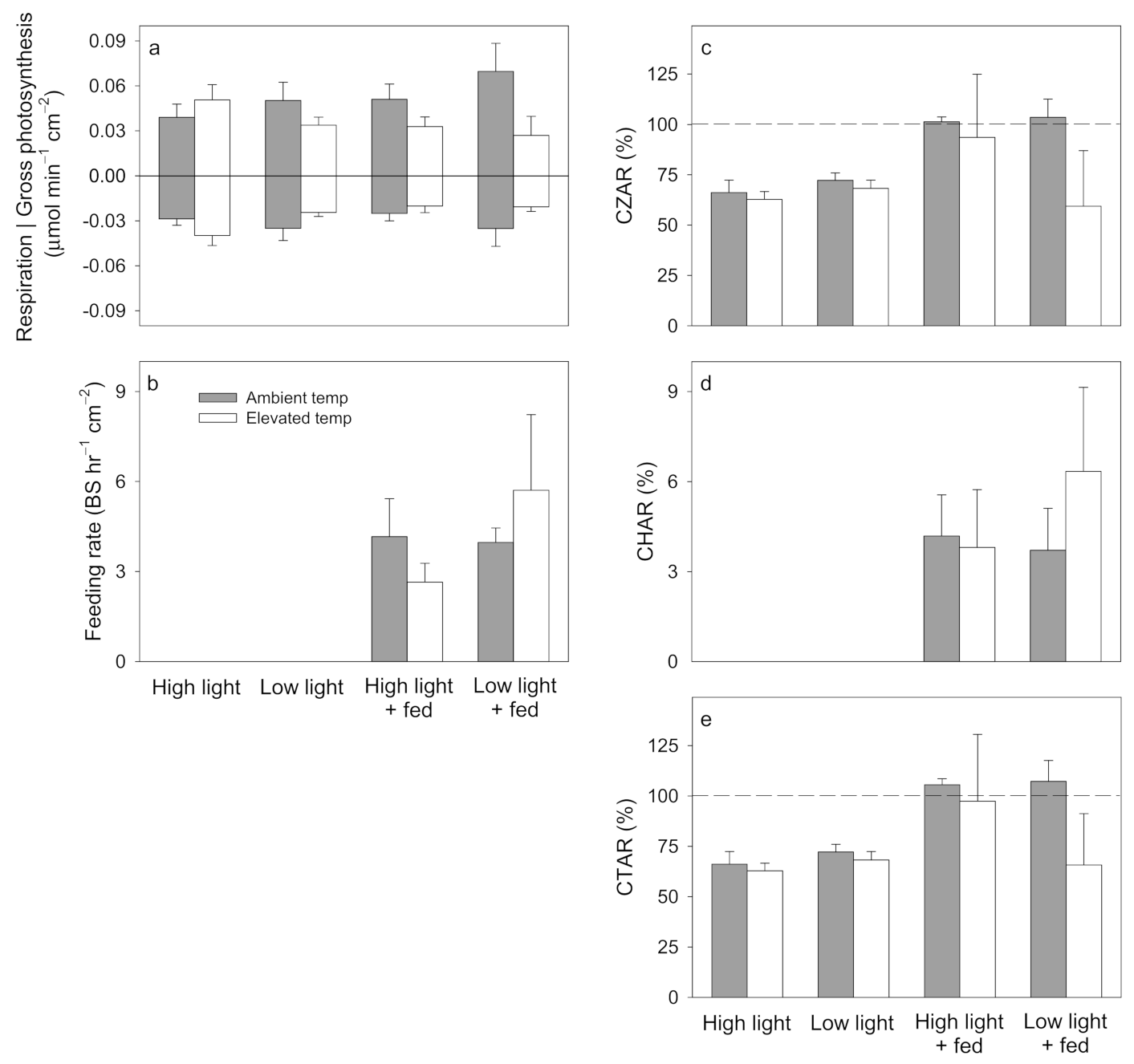
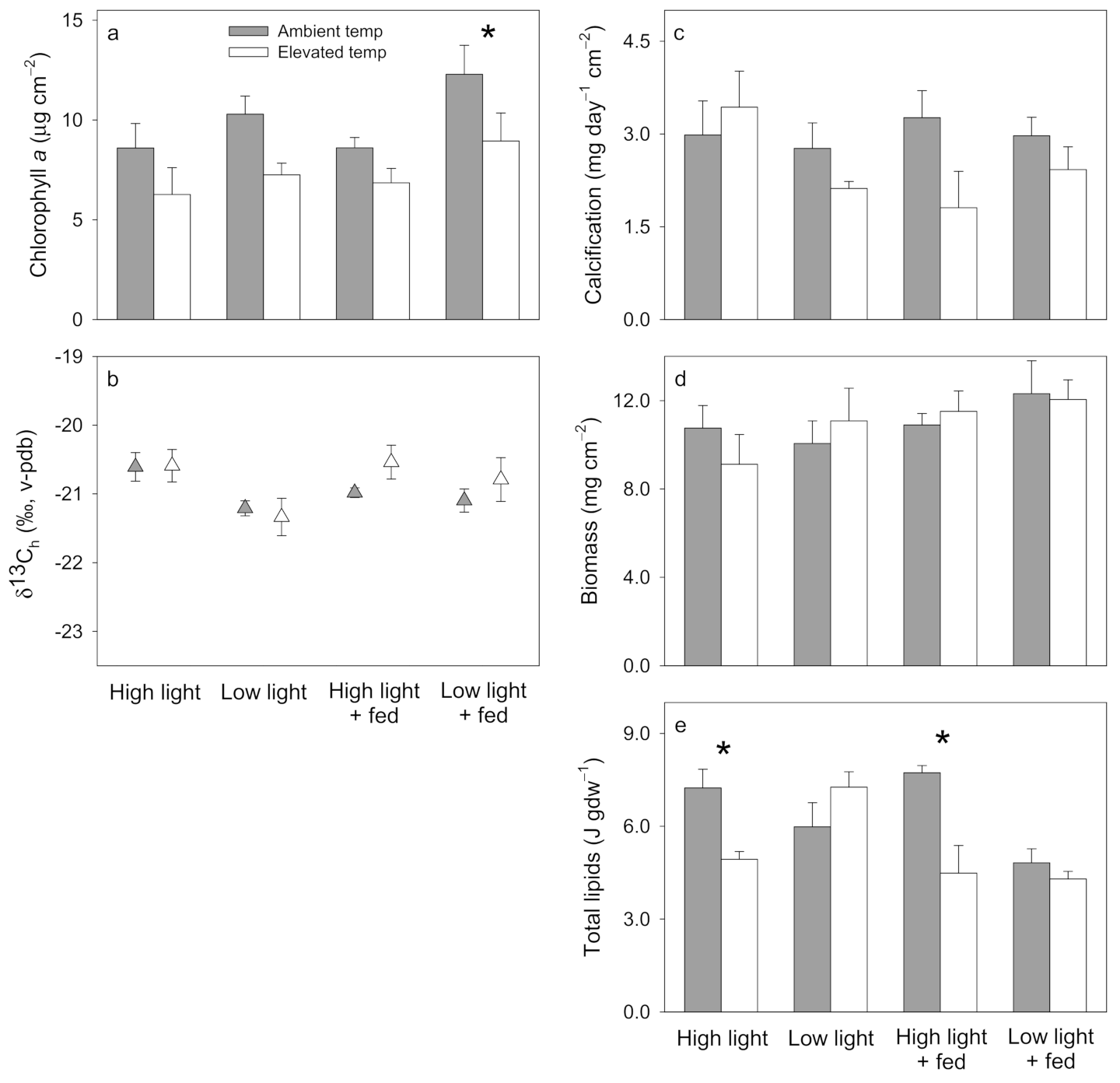
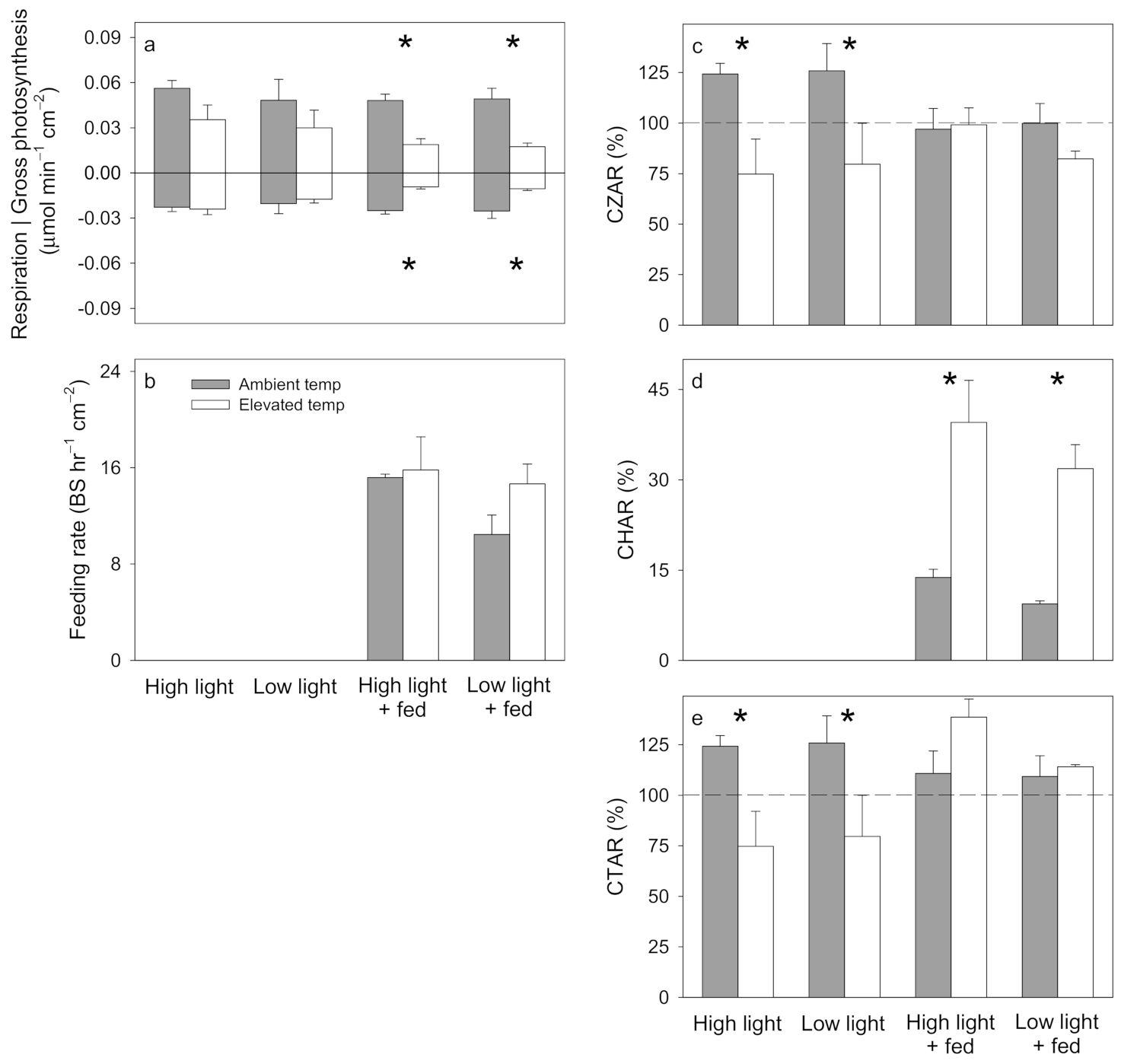
3. Results
3.1. Pocillopora damicornis
3.2. Stylophora pistillata
3.3. Turbinaria reniformis
4. Discussion
4.1. Pocillopora damicornis
4.2. Stylophora pistillata
4.3. Turbinaria reniformis
4.4. Implications
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pachauri, R.K.; Allen, M.R.; Barros, V.R.; Broome, J.; Cramer, W.; Christ, R.; Church, J.A.; Clarke, L.; Dahe, Q.; Dasgupta, P.; et al. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II, and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; The Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; p. 151. [Google Scholar]
- Donner, S.D.; Skirving, W.J.; Little, C.M.; Oppenheimer, M.; Hoegh-Guldberg, O. Global assessment of coral bleaching and required rates of adaptation under climate change. Glob. Chang. Biol. 2005, 11, 2251–2265. [Google Scholar] [CrossRef]
- Hughes, T.P.; Anderson, K.D.; Connolly, S.R.; Heron, S.F.; Kerry, J.T.; Lough, J.M.; Baird, A.H.; Baum, J.K.; Berumen, M.L.; Bridge, T.C.; et al. Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 2018, 359, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.E. Coral bleaching: Causes and consequences. Coral Reefs 1997, 16, S129–S138. [Google Scholar] [CrossRef]
- Gates, R.D.; Baghdasarian, G.; Muscatine, L. Temperature stress causes host-cell detachment in symbiotic Cnidarians—Implications for coral bleaching. Biol. Bull. 1992, 182, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Glynn, P.W. Coral reef bleaching: Facts, hypotheses and implications. Glob. Chang. Biol. 1996, 2, 495–509. [Google Scholar] [CrossRef]
- Grottoli, A.G.; Rodrigues, L.J.; Palardy, J.E. Heterotrophic plasticity and resilience in bleached corals. Nature 2006, 440, 1186–1189. [Google Scholar] [CrossRef]
- Muscatine, L.; McCloskey, L.R.; Marian, R.E. Estimating the daily contribution of carbon from zooxanthellae to coral animal respiration. Limnol. Oceanogr. 1981, 26, 601–611. [Google Scholar] [CrossRef]
- Tremblay, P.; Grover, R.; Maguer, J.F.; Legendre, L.; Ferrier-Pages, C. Autotrophic carbon budget in coral tissue: A new C-13-based model of photosynthate translocation. J. Exp. Biol. 2012, 215, 1384–1393. [Google Scholar] [CrossRef]
- Ferrier-Pages, C.; Hoogenboom, M.; Houlbreque, F. The role of plankton in coral trophodynamics. In Coral Reefs: An Ecosystem in Transition; Dubinsky, Z., Stambler, N., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 215–229. [Google Scholar] [CrossRef]
- Palardy, J.E.; Rodrigues, L.J.; Grottoli, A.G. The importance of zooplankton to the daily metabolic carbon requirements of healthy and bleached corals at two depths. J. Exp. Mar. Biol. Ecol. 2008, 367, 180–188. [Google Scholar] [CrossRef]
- Iglesias-Prieto, R.; Matta, J.L.; Robins, W.A.; Trench, R.K. Photosynthetic response to elevated temperature in the symbiotic dinoflagellate Symbiodinum microadriaticum in culture. Proc. Natl. Acad. Sci. USA 1992, 89, 10302–10305. [Google Scholar] [CrossRef]
- Levas, S.J.; Grottoli, A.G.; Hughes, A.; Osburn, C.L.; Matsui, Y. Physiological and biogeochemical traits of bleaching and recovery in the mounding species of coral Porites lobata: Implications for resilience in mouding corals. PLoS ONE 2013, 8, e63267. [Google Scholar] [CrossRef]
- Rodrigues, L.J.; Grottoli, A.G. Energy reserves and metabolism as indicators of coral recovery from bleaching. Limnol. Oceanogr. 2007, 52, 1874–1882. [Google Scholar] [CrossRef]
- Connolly, S.R.; Lopez-Yglesias, M.A.; Anthony, K.R.N. Food availability promotes rapid recovery from thermal stress in a scleractinian coral. Coral Reefs 2012, 31, 951–960. [Google Scholar] [CrossRef]
- Ferrier-Pages, C.; Rottier, C.; Beraud, E.; Levy, O. Experimental assessment of the feeding effort of three scleractinian coral species during a thermal stress: Effect on the rates of photosynthesis. J. Exp. Mar. Biol. Ecol. 2010, 390, 118–124. [Google Scholar] [CrossRef]
- Hughes, A.D.; Grottoli, A.G. Heterotrophic compensation: A possible mechanism for resilience of coral reefs to global warming or a sign of prolonged stress? PLoS ONE 2013, 8, e81172. [Google Scholar] [CrossRef]
- Grottoli, A.G.; Rodrigues, L.J. Bleached Porites compressa and Montipora capitata corals catabolize delta C-13-enriched lipids. Coral Reefs 2011, 30, 687–692. [Google Scholar] [CrossRef]
- Tolosa, I.; Treignier, C.; Grover, R.; Ferrier-Pages, C. Impact of feeding and short-term temperature stress on the content and isotopic signature of fatty acids, sterols, and alcohols in the scleractinian coral Turbinaria reniformis. Coral Reefs 2011, 30, 763. [Google Scholar] [CrossRef]
- Borell, E.M.; Yuliantri, A.R.; Bischof, K.; Richter, C. The effect of heterotrophy on photosynthesis and tissue composition of two scleractinian corals under elevated temperature. J. Exp. Mar. Biol. Ecol. 2008, 364, 116–123. [Google Scholar] [CrossRef]
- Levas, S. Biogeochemistry and Physiology of Bleached and Recovering Hawaiian and Caribbean Corals. Ph.D. Thesis, The Ohio State University, Columbus, OH, USA, 2012. [Google Scholar]
- Cohen, A.L.; Holcomb, M. Why corals care about ocean acidification: Uncovering the mechanism. Oceanography 2009, 22, 118–127. [Google Scholar] [CrossRef]
- Grottoli, A.G.; Warner, M.E.; Levas, S.J.; Aschaffenburg, M.D.; Schoepf, V.; McGinley, M.; Baumann, J.; Matsui, Y. The cumulative impact of annual coral bleaching can turn some coral species winners into losers. Glob. Chang. Biol. 2014, 20, 3823–3833. [Google Scholar] [CrossRef]
- Schoepf, V.; Grottoli, A.G.; Levas, S.J.; Aschaffenburg, M.D.; Baumann, J.H.; Matsui, Y.; Warner, M.E. Annual coral bleaching and the long-term recovery capacity of coral. Proc. R. Soc. B Biol. Sci. 2015, 282, 20151887. [Google Scholar] [CrossRef]
- Borell, E.M.; Bischof, K. Feeding sustains photosynthetic quantum yield of a scleractinian coral during thermal stress. Oecologia 2008, 157, 593–601. [Google Scholar] [CrossRef]
- Tremblay, P.; Gori, A.; Maguer, J.F.; Hoogenboom, M.; Ferrier-Pages, C. Heterotrophy promotes the re-establishment of photosynthate translocation in a symbiotic coral after heat stress. Sci. Rep. 2016, 6, 38112. [Google Scholar] [CrossRef]
- Baumann, J.; Grottoli, A.G.; Hughes, A.D.; Matsui, Y. Photoautotrophic and heterotrophic carbon in bleached and non-bleached coral lipid acquisition and storage. J. Exp. Mar. Biol. Ecol. 2014, 461, 469–478. [Google Scholar] [CrossRef]
- Hughes, A.D.; Grottoli, A.G.; Pease, T.K.; Matsui, Y. Acquisition and assimilation of carbon in non-bleached and bleached corals. Mar. Ecol. Prog. Ser. 2010, 420, 91–101. [Google Scholar] [CrossRef]
- Brown, B.E.; Ambarsari, I.; Warner, M.E.; Fitt, W.K.; Dunne, R.P.; Gibb, S.W.; Cummings, D.G. Diurnal changes in photochemical efficiency and xanthophyll concentrations in shallow water reef corals: Evidence for photoinhibition and photoprotection. Coral Reefs 1999, 18, 99–105. [Google Scholar]
- Bhagooli, R.; Hidaka, M. Photoinhibition, bleaching susceptibility and mortality in two scleractinian corals, Platygyra ryukyuensis and Stylophora pistillata, in response to thermal and light stresses. Comp. Biochem. Physiol. A-Mol. Integr. Physiol. 2004, 137, 547–555. [Google Scholar] [CrossRef]
- Coles, S.L.; Jokiel, P.L. Synergistic effects of temperature, salinity and light on hermatypic coral Montipora verrucosa. Mar. Biol. 1978, 49, 187–195. [Google Scholar] [CrossRef]
- Lesser, M.P.; Stochaj, W.R.; Tapley, D.W.; Shick, J.M. Bleaching in coral reef Anthozoans—Effects of irradiance, ultraviolet-radiation, and temperature on the activities of protective enzymens against active oxygen. Coral Reefs 1990, 8, 225–232. [Google Scholar] [CrossRef]
- Browne, N.; Braoun, C.; McIlwain, J.; Nagarajan, R.; Zinke, J. Borneo coral reefs subject to high sediment loads show evidence of resilience to various environmental stressors. PeerJ 2019, 7, e7382. [Google Scholar] [CrossRef]
- Cacciapaglia, C.; van Woesik, R. Climate-change refugia: Shading reef corals by turbidity. Glob. Chang. Biol. 2016, 22, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Morgan, K.M.; Perry, C.T.; Johnson, J.A.; Smithers, S.G. Nearshore turbid-zone corals exhibit high bleaching tolerance on the Great Barrier Reef following the 2016 ocean warming event. Front. Mar. Sci. 2017, 4, 224. [Google Scholar] [CrossRef]
- Nitschke, M.R.; Gardner, S.G.; Goyen, S.; Fujise, L.; Camp, E.F.; Ralph, P.J.; Suggett, D.J. Utility of photochemical traits as diagnostics of thermal tolerance amongst Great Barrier Reef corals. Front. Mar. Sci. 2018, 5, 460. [Google Scholar] [CrossRef]
- Sully, S.; van Woesik, R. Turbid reefs moderate coral bleaching under climate-related temperature stress. Glob. Chang. Biol. 2020, 26, 1367–1373. [Google Scholar] [CrossRef]
- Courtial, L.; Roberty, S.; Shick, J.M.; Houlbreque, F.; Ferrier-Pages, C. Interactive effects of ultraviolet radiation and thermal stress on two reef-building corals. Limnol. Oceanogr. 2017, 62, 1000–1013. [Google Scholar] [CrossRef]
- Lesser, M.P. Phylogenetic signature of light and thermal stress for the endosymbiotic dinoflagellates of corals (Family Symbiodiniaceae). Limnol. Oceanogr. 2019, 64, 1852–1863. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Dubinsky, Z. Light-shade adaptation of Stylophora pistillata, a hermatypic coral from the Gulf of Eilat. Nature 1981, 289, 172–174. [Google Scholar] [CrossRef]
- Porter, J.W.; Muscatine, L.; Dubinsky, Z.; Falkowski, P.G. Primary production and photoadaptation in light-adapted and shade-adapted colonies of the symbiotic coral, Stylophora pistillata. Proc. R. Soc. Ser. B Biol. Sci. 1984, 222, 161–180. [Google Scholar] [CrossRef]
- Titlyanov, E.A.; Titlyanova, T.V.; Yamazato, K.; van Woesik, R. Photo-acclimation dynamics of the coral Stylophora pistillata to low and extremely low light. J. Exp. Mar. Biol. Ecol. 2001, 263, 211–225. [Google Scholar] [CrossRef]
- Anthony, K.R.N.; Fabricius, K.E. Shifting roles of heterotrophy and autotrophy in coral energetics under varying turbidity. J. Exp. Mar. Biol. Ecol. 2000, 252, 221–253. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Dubinsky, Z.; Muscatine, L.; Porter, J.W. Light and the bioenergetics of a symbiotic coral. Bioscience 1984, 34, 705–709. [Google Scholar] [CrossRef]
- Levy, O.; Karako-Lampert, S.; Ben-Asher, H.W.; Zoccola, D.; Pages, G.; Ferrier-Pages, C. Molecular assessment of the effect of light and heterotrophy in the scleractinian coral Stylophora pistillata. Proc. R. Soc. B Biol. Sci. 2016, 283, 20153025. [Google Scholar] [CrossRef]
- Treignier, C.; Grover, R.; Ferrier-Pages, C.; Tolosa, I. Effect of light and feeding on the fatty acid and sterol composition of zooxanthellae and host tissue isolated from the scleractinian coral Turbinaria reniformis. Limnol. Oceanogr. 2008, 53, 2702–2710. [Google Scholar] [CrossRef]
- Tilstra, A.; Wijgerde, T.; Dini-Andreote, F.; Eriksson, B.K.; Salles, J.F.; Pen, I.; Osinga, R.; Wild, C. Light induced intraspecific variability in response to thermal stress in the hard coral Stylophora pistillata. PeerJ 2017, 5, e3802. [Google Scholar] [CrossRef]
- Tremblay, P.; Grover, R.; Maguer, J.F.; Hoogenboom, M.; Ferrier-Pages, C. Carbon translocation from symbiont to host depends on irradiance and food availability in the tropical coral Stylophora pistillata. Coral Reefs 2014, 33, 1–13. [Google Scholar] [CrossRef]
- Hoogenboom, M.O.; Campbell, D.A.; Beraud, E.; DeZeeuw, K.; Ferrier-Pages, C. Effects of light, food availability and temperature stress on the function of photosystem II and photosystem I of coral symbionts. PLoS ONE 2012, 7, e30167. [Google Scholar] [CrossRef]
- Ferrier-Pages, C.; Gattuso, J.P.; Dallot, S.; Jaubert, J. Effect of nutrient enrichment on growth and photosynthesis of the zooxanthellate coral Stylophora pistillata. Coral Reefs 2000, 19, 103–113. [Google Scholar] [CrossRef]
- Maor-Landaw, K.; Karako-Lampert, S.; Ben-Asher, H.W.; Goffredo, S.; Falini, G.; Dubinsky, Z.; Levy, O. Gene expression profiles during short-term heat stress in the red sea coral Stylophora pistillata. Glob. Chang. Biol. 2014, 20, 3026–3035. [Google Scholar] [CrossRef]
- Reynaud, S.; Ferrier-Pages, C.; Meibom, A.; Mostefaoui, S.; Mortlock, R.; Fairbanks, R.; Allemand, D. Light and temperature effects on Sr/Ca and Mg/Ca ratios in the scleractinian coral Acropora sp. Geochim. Cosmochim. Acta 2007, 71, 354–362. [Google Scholar] [CrossRef]
- Reynaud, S.; Martinez, P.; Houlbreque, F.; Billy, I.; Allemand, D.; Ferrier-Pages, C. Effect of light and feeding on the nitrogen isotopic composition of a zooxanthellate coral: Role of nitrogen recycling. Mar. Ecol. Prog. Ser. 2009, 392, 103–110. [Google Scholar] [CrossRef]
- Reynaud-Vaganay, S.; Ferrier, C.; Sambrotto, R.; Juillet-Leclerc, A.; Gattuso, J.P. Effect of feeding on the carbon isotopic composition of the zooxanthellate coral Stylophora pistillata. Geochim. Cosmochim. Acta 2002, 66, A636. [Google Scholar]
- Hoogenboom, M.; Rottier, C.; Sikorski, S.; Ferrier-Pages, C. Among-species variation in the energy budgets of reef-building corals: Scaling from coral polyps to communities. J. Exp. Biol. 2015, 218, 3866–3877. [Google Scholar] [CrossRef] [PubMed]
- Grottoli, A.G.; Toonen, R.J.; van Woesik, R.; Vega Thurber, R.; Warner, M.E.; McLachlan, R.H.; Price, J.T.; Bahr, K.D.; Baums, I.B.; Castillo, K.; et al. Increasing comparability among coral bleaching experiments. Ecol. Appl. 2020, 31, e02262. [Google Scholar] [CrossRef]
- Fine, M.; Gildor, H.; Genin, A. A coral reef refuge in the Red Sea. Glob. Chang. Biol. 2013, 19, 3640–3647. [Google Scholar] [CrossRef]
- Grottoli, A.G.; Tchernov, D.; Winters, G. Physiological and biogeochemical responses of super-corals to thermal stress from the Northern Gulf of Aqaba, Red Sea. Front. Mar. Sci. 2017, 4, 215. [Google Scholar] [CrossRef]
- Osman, E.O.; Smith, D.J.; Ziegler, M.; Kurten, B.; Conrad, C.; El-Haddad, K.M.; Voolstra, C.R.; Suggett, D.J. Thermal refugia against coral bleaching throughout the northern Red Sea. Glob. Chang. Biol. 2018, 24, E474–E484. [Google Scholar] [CrossRef]
- Lesser, M.P. Oxidative stress causes coral bleaching during exposure to elevated temperatures. Coral Reefs 1997, 16, 187–192. [Google Scholar] [CrossRef]
- Marubini, F.; Barnett, H.; Langdon, C.; Atkinson, M.J. Dependence of calcification on light and carbonate ion concentration for the hermatypic coral Porites compressa. Mar. Ecol. Prog. Ser. 2001, 220, 153–162. [Google Scholar] [CrossRef]
- Nakamura, T.; Yamasaki, H. Flicker light effects on photosynthesis of symbiotic algae in the reef-building coral Acropora digitifera (Cnidaria: Anthozoa: Scleractinia). Pac. Sci. 2008, 62, 341–350. [Google Scholar] [CrossRef]
- Schutter, M.; Kranenbarg, S.; Wijffels, R.H.; Verreth, J.; Osinga, R. Modification of light utilization for skeletal growth by water flow in the scleractinian coral Galaxea fascicularis. Mar. Biol. 2011, 158, 769–777. [Google Scholar] [CrossRef]
- Ulstrup, K.E.; Ralph, P.J.; Larkum, A.W.D.; Kuhl, M. Intra-colonial variability in light acclimation of zooxanthellae in coral tissues of Pocillopora damicornis. Mar. Biol. 2006, 149, 1325–1335. [Google Scholar] [CrossRef]
- Houlbreque, F.; Tambutte, E.; Allemand, D.; Ferrier-Pages, C. Interactions between zooplankton feeding, photosynthesis and skeletal growth in the scleractinian coral Stylophora pistillata. J. Exp. Biol. 2004, 207, 1461–1469. [Google Scholar] [CrossRef]
- Levas, S.; Grottoli, A.G.; Schoepf, V.; Aschaffenburg, M.; Baumann, J.; Bauer, J.E.; Warner, M.E. Can heterotrophic uptake of dissolved organic carbon and zooplankton mitigate carbon budget deficits in annually bleached corals? Coral Reefs 2016, 35, 495–506. [Google Scholar] [CrossRef]
- Ferrier-Pages, C.; Peirano, A.; Abbate, M.; Cocito, S.; Negri, A.; Rottier, C.; Riera, P.; Rodolfo-Metalpa, R.; Reynaud, S. Summer autotrophy and winter heterotrophy in the temperate symbiotic coral Cladocora caespitosa. Limnol. Oceanogr. 2011, 56, 1429–1438. [Google Scholar] [CrossRef]
- Leal, M.C.; Ferrier-Pages, C.; Calado, R.; Brandes, J.A.; Frischer, M.E.; Nejstgaard, J.C. Trophic ecology of the facultative symbiotic coral Oculina arbuscula. Mar. Ecol. Prog. Ser. 2014, 504, 171–179. [Google Scholar] [CrossRef]
- Tremblay, P.; Naumann, M.S.; Sikorski, S.; Grover, R.; Ferrier-Pages, C. Experimental assessment of organic carbon fluxes in the scleractinian coral Stylophora pistillata during a thermal and photo stress event. Mar. Ecol. Prog. Ser. 2012, 453, 63–77. [Google Scholar] [CrossRef]
- Levas, S.; Grottoli, A.G.; Warner, M.E.; Cai, W.-J.; Bauer, J.; Schoepf, V.; Baumann, J.H.; Matsui, Y.; Gearing, C.; Melman, T.F.; et al. Organic carbon fluxes mediated by corals at elevated pCO2 and temperature. Mar. Ecol. Prog. Ser. 2015, 519, 153–164. [Google Scholar] [CrossRef]
- Jokiel, P.L.; Maragos, J.E.; Franzisket, L. Coral growth: Buoyant weight technique. In Coral Reef: Research Methods; Stoddart, D., Johannes, R.E., Eds.; UNESCO: Paris, France, 1978; Volume 78, pp. 379–396. [Google Scholar]
- Ferrier-Pages, C.; Gevaert, F.; Reynaud, S.; Beraud, E.; Menu, D.; Janquin, M.A.; Cocito, S.; Peirano, A. In situ assessment of the daily primary production of the temperate symbiotic coral Cladocora caespitosa. Limnol. Oceanogr. 2013, 58, 1409–1418. [Google Scholar] [CrossRef]
- Veal, C.J.; Carmi, M.; Fine, M.; Hoegh-Guldberg, O. Increasing the accuracy of surface area estimation using single wax dipping of coral fragments. Coral Reefs 2010, 29, 893–897. [Google Scholar] [CrossRef]
- Marsh, J.A. Primary productivity of reef building calcareous red algae. Ecology 1970, 51, 255–263. [Google Scholar] [CrossRef]
- Jeffrey, S.W.; Humphrey, G.F. New spectrophotometric equations for determining chlorophylls a, b, c1, and c2 in higher-plants, algae and natural phytoplankton. Biochem. Und Physiol. Der Pflanz. 1975, 167, 191–194. [Google Scholar] [CrossRef]
- McLachlan, R.H.; Dobson, K.L.; Grottoli, A.G. Quantification of Total Biomass in Ground Coral Samples. Available online: http://www.protocols.io/view/quantification-of-biomass-in-ground-coral-samples-bdyai7se (accessed on 23 April 2020).
- McLachlan, R.H.; Munoz-Garcia, A.; Grottoli, A.G. Extraction of Total Soluble Lipid from Ground Coral Samples. Available online: https://www.protocols.io/view/extraction-of-total-soluble-lipid-from-ground-cora-bc4qiyvw (accessed on 14 March 2020).
- Gnaiger, E.; Bitterlich, G. Proximate biochemical composition and caloric content calculated from elemental CHN analysis—A stochiometric concept. Oecologia 1984, 62, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Anthony, K.R.N. Coral suspension feeding on fine particulate matter. J. Exp. Mar. Biol. Ecol. 1999, 232, 85–106. [Google Scholar] [CrossRef]
- Ferrier-Pages, C.; Allemand, D.; Gattuso, J.P.; Jaubert, J.; Rassoulzadegan, R. Microheterotrophy in the zooxanthellate coral Stylophora pistillata: Effects of light and ciliate density. Limnol. Oceanogr. 1998, 43, 1639–1648. [Google Scholar] [CrossRef]
- Ferrier-Pages, C.; Witting, J.; Tambutte, E.; Sebens, K.P. Effect of natural zooplankton feeding on the tissue and skeletal growth of the scleractinian coral Stylophora pistillata. Coral Reefs 2003, 22, 229–240. [Google Scholar] [CrossRef]
- Sturaro, N.; Hsieh, Y.E.; Chen, Q.; Wang, P.-L.; Denis, V. Toward a standardised protocol for the stable isotope analysis of scleractinian corals. Rapid Commun. Mass Spectrom. 2020, 34, e8663. [Google Scholar] [CrossRef]
- Price, J.; Smith, A.; Dobson, K.D.; Grottoli, A.G. Airbrushed Coral Sample Preparation for Organic Stable Carbon and Nitrogen Isotope Analyses. Available online: https://www.protocols.io/view/airbrushed-coral-sample-preparation-for-organic-st-bgi7juhn (accessed on 10 July 2020).
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carre, G.; Garcia Marquez, J.R.; Gruber, B.; Lafourcade, B.; Leitao, P.J.; et al. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- Clarke, K.; Gorley, R. Primer v6: User Manual/Tutorial; PRIMER-E Ltd.: Plymouth, UK, 2006. [Google Scholar]
- Cook, R.D. Detection of influential observation in linear regression. Technometrics 1977, 19, 15–18. [Google Scholar] [CrossRef]
- Kikvidze, Z.; Moya-Laraño, J. Unexpected failures of recommended tests in basic statistical analyses of ecological data. Web Ecol. 2008, 8, 67–73. [Google Scholar] [CrossRef]
- Grover, R.; Maguer, J.F.; Reynaud-Vaganay, S.; Ferrier-Pages, C. Uptake of ammonium by the scleractinian coral Stylophora pistillata: Effect of feeding, light, and ammonium concentrations. Limnol. Oceanogr. 2002, 47, 782–790. [Google Scholar] [CrossRef]
- Moran, M.D. Arguments for rejecting the sequential Bonferroni in ecological studies. Oikos 2003, 100, 403–405. [Google Scholar] [CrossRef]
- Titlyanov, E.A.; Titlyanova, T.V.; Yamazato, K. Acclimation of symbiotic reef-building corals to extremely low light. Symbiosis 2002, 33, 125–143. [Google Scholar]
- Ziegler, M.; Roder, C.M.; Buechel, C.; Voolstra, C.R. Limits to physiological plasticity of the coral Pocillopora verrucosa from the central Red Sea. Coral Reefs 2014, 33, 1115–1129. [Google Scholar] [CrossRef]
- Cohen, I.; Dubinsky, Z.; Erez, J. Light enhanced calcification in hermatypic corals: New insights from light spectral responses. Front. Mar. Sci. 2016, 2, 122. [Google Scholar] [CrossRef]
- Kawaguti, S.; Sakumoto, D. The effect of light on the calcium deposition of corals. Bull. Oceanogr. Inst. Taiwan 1948, 4, 65–70. [Google Scholar]
- Houlbreque, F.; Tambutte, E.; Ferrier-Pages, C. Effect of zooplankton availability on the rates of photosynthesis, and tissue and skeletal growth in the scleractinian coral Stylophora pistillata. J. Exp. Mar. Biol. Ecol. 2003, 296, 145–166. [Google Scholar] [CrossRef]
- Wellington, G.M. An experimental analysis of the effects of light and zooplankton on coral zonation. Oecologia 1982, 52, 311–320. [Google Scholar] [CrossRef]
- Rodrigues, L.J.; Grottoli, A.G. Calcification rate and the stable carbon, oxygen, and nitrogen isotopes in the skeleton, host tissue, and zooxanthellae of bleached and recovering Hawaiian corals. Geochim. Cosmochim. Acta 2006, 70, 2781–2789. [Google Scholar] [CrossRef]
- Lyndby, N.H.; Holm, J.B.; Wangpraseurt, D.; Grover, R.; Rottier, C.; Kuhl, M.; Ferrier-Pages, C. Effect of temperature and feeding on carbon budgets and O2 dynamics in Pocillopora damicornis. Mar. Ecol. Prog. Ser. 2020, 652, 49–62. [Google Scholar] [CrossRef]
- Mass, T.; Einbinder, S.; Brokovich, E.; Shashar, N.; Vago, R.; Erez, J.; Dubinsky, Z. Photoacclimation of Stylophora pistillata to light extremes: Metabolism and calcification. Mar. Ecol. Prog. Ser. 2007, 334, 93–102. [Google Scholar] [CrossRef]
- Titlyanov, E.A.; Titlyanova, T.V.; Yamazato, K.; van Woesik, R. Photo-acclimation of the hermatypic coral Stylophora pistillata while subjected to either starvation or food provisioning. J. Exp. Mar. Biol. Ecol. 2001, 257, 163–181. [Google Scholar] [CrossRef]
- Loya, Y.; Sakai, K.; Yamazato, K.; Nakano, Y.; Sambali, H.; van Woesik, R. Coral bleaching: The winners and the losers. Ecol. Lett. 2001, 4, 122–131. [Google Scholar] [CrossRef]
- Godinot, C.; Ferrier-Pages, C.; Sikorski, S.; Grover, R. Alkaline phosphatase activity of reef-building corals. Limnol. Oceanogr. 2013, 58, 227–234. [Google Scholar] [CrossRef]
- LaJeunesse, T.C.; Parkinson, J.E.; Gabrielson, P.W.; Jeong, H.J.; Reimer, J.D.; Voolstra, C.R.; Santos, S.R. Systematic revision of Symbiodiniaceae highlights the antiquity and diversity of coral endosymbionts. Curr. Biol. 2018, 28, 2570–2580.e6. [Google Scholar] [CrossRef]
- Tremblay, P.; Maguer, J.F.; Grover, R.; Ferrier-Pages, C. Trophic dynamics of scleractinian corals: Stable isotope evidence. J. Exp. Biol. 2015, 218, 1223–1234. [Google Scholar] [CrossRef]
 , Stylophora pistillata
, Stylophora pistillata  , and Turbinaria reniformis
, and Turbinaria reniformis  were fragmented into eight large and eight small ramets and distributed randomly and evenly among eight treatments of a fully factorial experimental design: two seawater temperatures (ambient temperature of 25 °C, elevated temperature of 30 °C), two light levels (high light of 300 μmol photons m−2 s−1, low light of 150 μmol photons m−2 s−1), and either fed (Artemia nauplii) or unfed.
were fragmented into eight large and eight small ramets and distributed randomly and evenly among eight treatments of a fully factorial experimental design: two seawater temperatures (ambient temperature of 25 °C, elevated temperature of 30 °C), two light levels (high light of 300 μmol photons m−2 s−1, low light of 150 μmol photons m−2 s−1), and either fed (Artemia nauplii) or unfed.
 , Stylophora pistillata
, Stylophora pistillata  , and Turbinaria reniformis
, and Turbinaria reniformis  were fragmented into eight large and eight small ramets and distributed randomly and evenly among eight treatments of a fully factorial experimental design: two seawater temperatures (ambient temperature of 25 °C, elevated temperature of 30 °C), two light levels (high light of 300 μmol photons m−2 s−1, low light of 150 μmol photons m−2 s−1), and either fed (Artemia nauplii) or unfed.
were fragmented into eight large and eight small ramets and distributed randomly and evenly among eight treatments of a fully factorial experimental design: two seawater temperatures (ambient temperature of 25 °C, elevated temperature of 30 °C), two light levels (high light of 300 μmol photons m−2 s−1, low light of 150 μmol photons m−2 s−1), and either fed (Artemia nauplii) or unfed.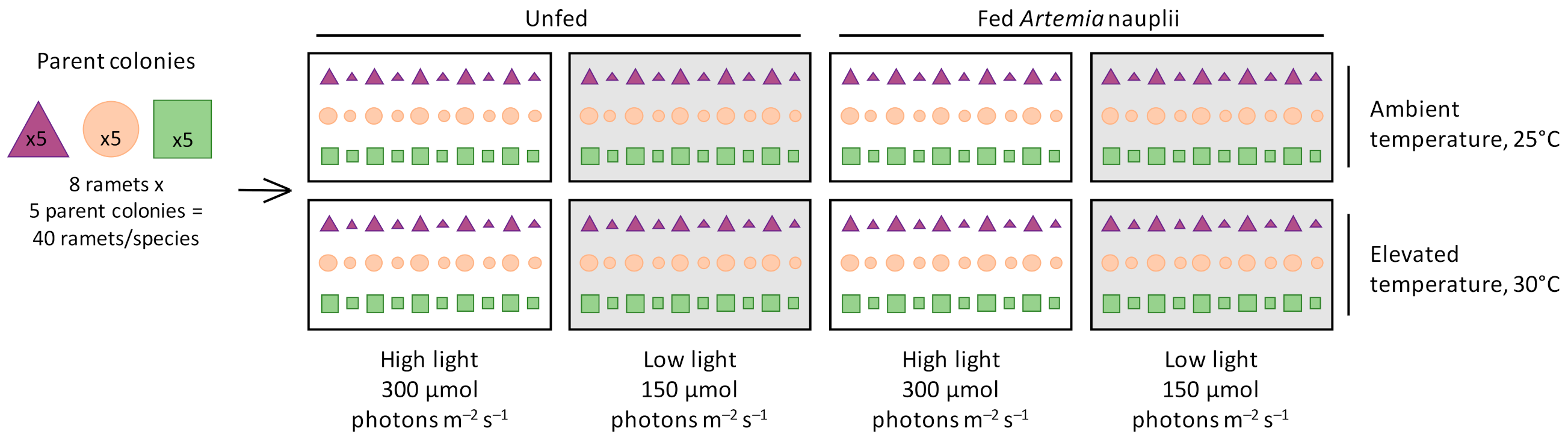


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobson, K.L.; Ferrier-Pagès, C.; Saup, C.M.; Grottoli, A.G. The Effects of Temperature, Light, and Feeding on the Physiology of Pocillopora damicornis, Stylophora pistillata, and Turbinaria reniformis Corals. Water 2021, 13, 2048. https://doi.org/10.3390/w13152048
Dobson KL, Ferrier-Pagès C, Saup CM, Grottoli AG. The Effects of Temperature, Light, and Feeding on the Physiology of Pocillopora damicornis, Stylophora pistillata, and Turbinaria reniformis Corals. Water. 2021; 13(15):2048. https://doi.org/10.3390/w13152048
Chicago/Turabian StyleDobson, Kerri L., Christine Ferrier-Pagès, Casey M. Saup, and Andréa G. Grottoli. 2021. "The Effects of Temperature, Light, and Feeding on the Physiology of Pocillopora damicornis, Stylophora pistillata, and Turbinaria reniformis Corals" Water 13, no. 15: 2048. https://doi.org/10.3390/w13152048
APA StyleDobson, K. L., Ferrier-Pagès, C., Saup, C. M., & Grottoli, A. G. (2021). The Effects of Temperature, Light, and Feeding on the Physiology of Pocillopora damicornis, Stylophora pistillata, and Turbinaria reniformis Corals. Water, 13(15), 2048. https://doi.org/10.3390/w13152048







