Contrasting Water Use, Stomatal Regulation, Embolism Resistance, and Drought Responses of Two Co-Occurring Mangroves
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Species and Study Site
2.2. Vulnerability to Cavitation Using the Optical Method
2.3. Soil Water-Withholding Experiment
2.4. Foliar ABA Quantification
2.5. Data Analysis
3. Results
3.1. Photosynthetic Gas Exchange Traits
3.2. Vulnerability to Embolism Formation and Spread
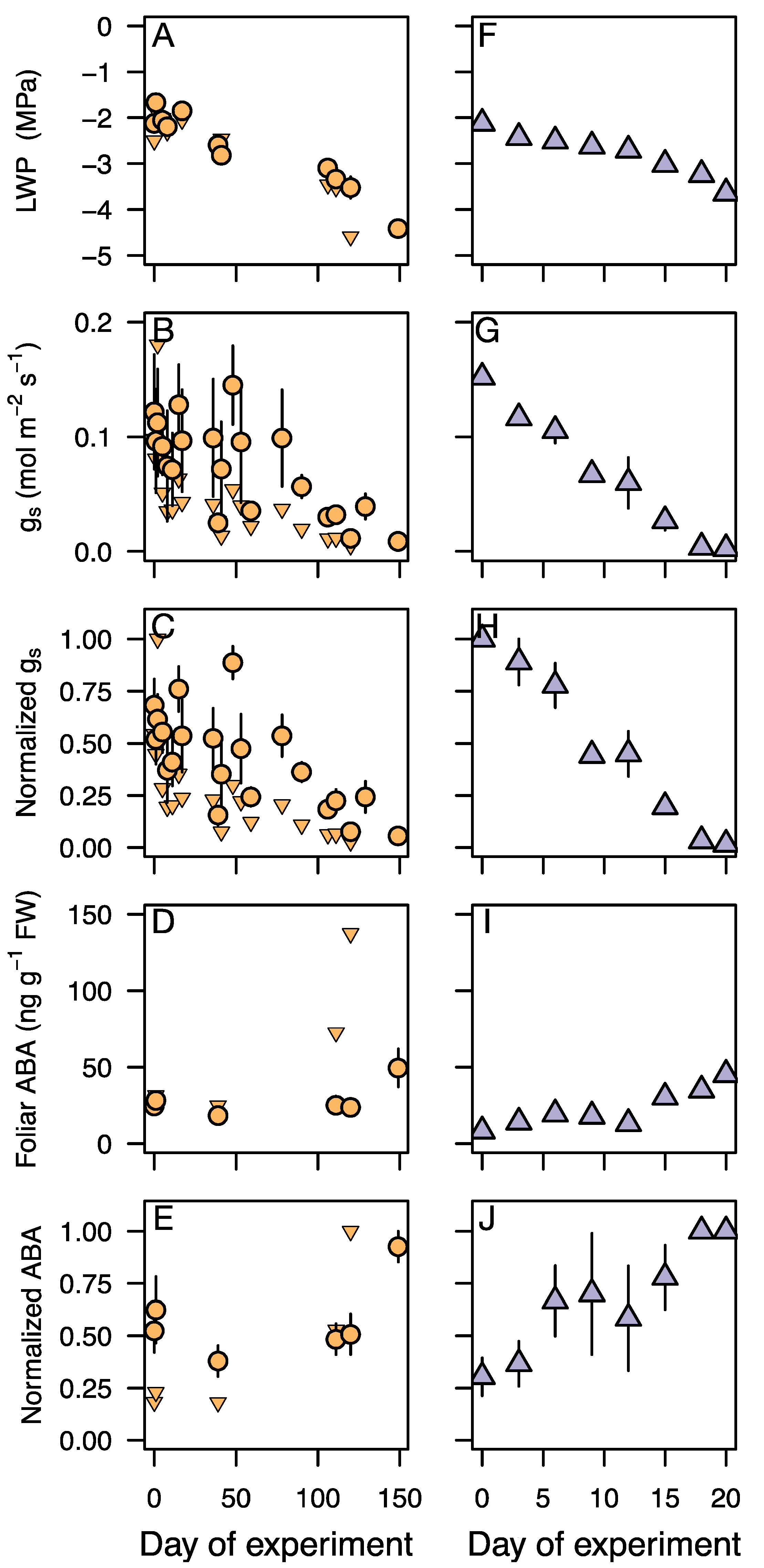
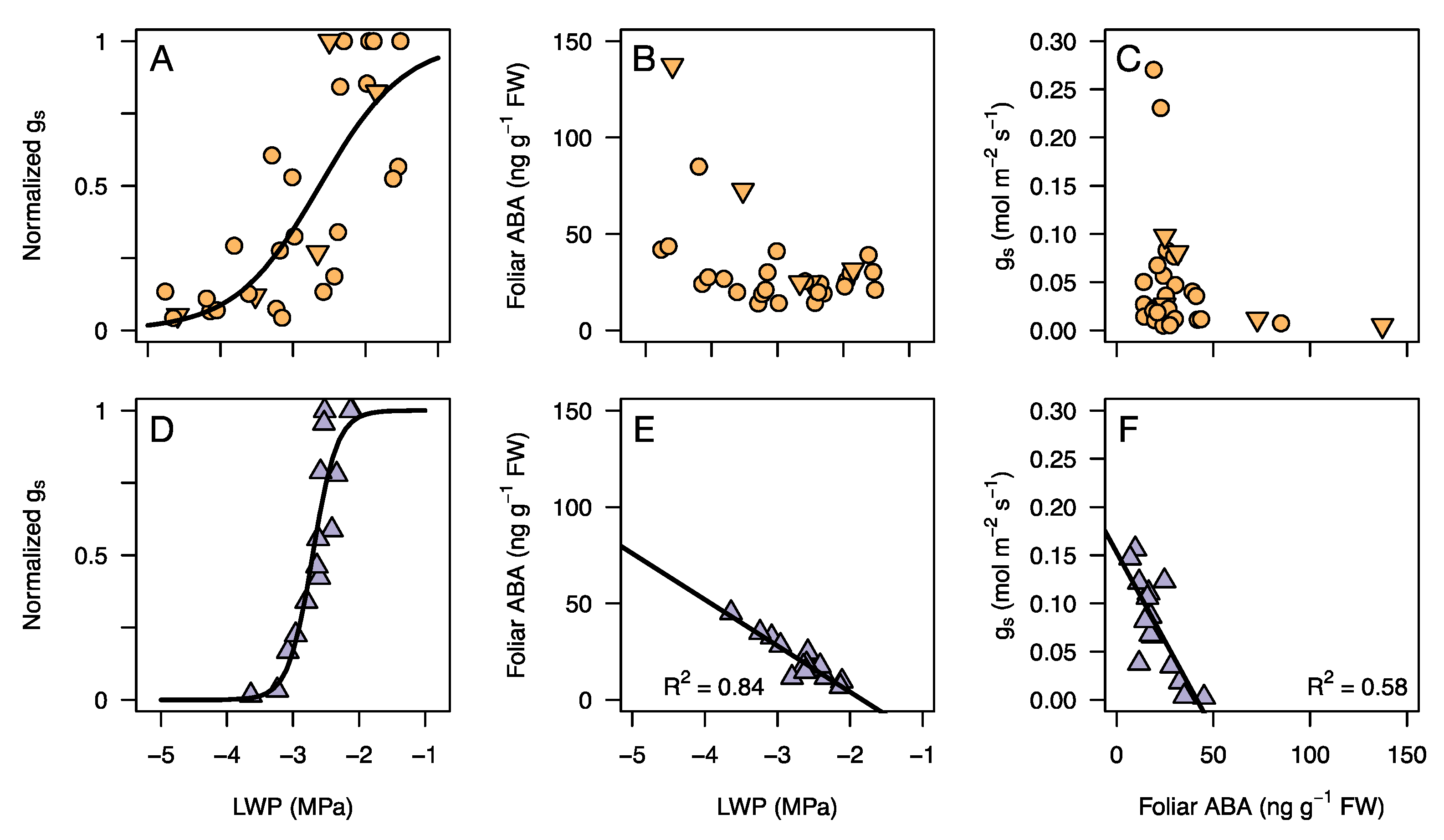
3.3. Response of Leaf Water Potential, gs, and ABA to an Experimental Drought
3.4. Hydraulic Safety Margins
4. Discussion
4.1. Stomatal Regulation during Drought
4.2. Vulnerability to Embolism
4.3. Hydraulic Safety Margins Associated with Different Salt Management Strategies
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brodribb, T.J.; Powers, J.; Cochard, H.; Choat, B. Hanging by a thread? Forests and drought. Science 2020, 368, 261–266. [Google Scholar] [CrossRef]
- Brodribb, T.; Cochard, H.; Rodriguez Dominguez, C. Measuring the pulse of trees; using the vascular system to predict tree mortality in the 21st century. Conserv. Physiol. 2019, 7, coz046. [Google Scholar] [CrossRef]
- Engelbrecht, B.M. Plant ecology: Forests on the brink. Nature 2012, 491, 675–677. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choat, B.; Jansen, S.; Brodribb, T.J.; Cochard, H.; Delzon, S.; Bhaskar, R.; Bucci, S.J.; Feild, T.S.; Gleason, S.M.; Hacke, U.G.; et al. Global convergence in the vulnerability of forests to drought. Nature 2012, 491, 752–755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.T. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef] [Green Version]
- Adams, H.D.; Macalady, A.K.; Breshears, D.D.; Allen, C.D.; Stephenson, N.L.; Saleska, S.R.; Huxman, T.E.; McDowell, N.G. Climate-induced tree mortality: Earth system consequences. Eos Trans. Am. Geophys. Union 2010, 91, 153–154. [Google Scholar] [CrossRef]
- Simonin, K.A.; Roddy, A.B. Genome downsizing, physiological novelty, and the global dominance of flowering plants. PloS Biol. 2018, 16, e2003706. [Google Scholar] [CrossRef] [Green Version]
- Brodribb, T.J.; McAdam, S.A.M. Evolution of the stomatal regulation of plant water content. Plant Physiol 2017, 174, 639–649. [Google Scholar] [CrossRef] [Green Version]
- Sack, L.; Holbrook, N.M. Leaf hydraulics. Annu. Rev. Plant Biol. 2006, 57, 361–381. [Google Scholar] [CrossRef] [Green Version]
- Sperry, J.; Pockman, W. Limitation of transpiration by hydraulic conductance and xylem cavitation in betula occidentalis. Plant Cell Environ. 1993, 16, 279–287. [Google Scholar] [CrossRef]
- Jones, H.; Sutherland, R. Stomatal control of xylem embolism. Plant Cell Environ. 1991, 14, 607–612. [Google Scholar] [CrossRef]
- Meinzer, F.; Grantz, D. Stomatal and hydraulic conductance in growing sugarcane: Stomatal adjustment to water transport capacity. Plant Cell Environ. 1990, 13, 383–388. [Google Scholar] [CrossRef]
- Jiang, G.F.; Goodale, U.M.; Liu, Y.Y.; Hao, G.Y.; Cao, K.F. Salt management strategy defines the stem and leaf hydraulic characteristics of six mangrove tree species. Tree Physiol. 2017, 37, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Hao, G.-Y.; Jones, T.J.; Luton, C.; Zhang, Y.-J.; Manzane, E.; Scholz, F.G.; Bucci, S.J.; Cao, K.-F.; Goldstein, G. Hydraulic redistribution in dwarf rhizophora mangle trees driven by interstitial soil water salinity gradients: Impacts on hydraulic architecture and gas exchange. Tree Physiol. 2009, 29, 697–705. [Google Scholar] [CrossRef] [Green Version]
- Brodersen, C.R.; Roddy, A.B.; Wason, J.W.; McElrone, A.J. Functional status of xylem through time. Annu. Rev. Plant Biol. 2019, 70, 407–433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tyree, M.T.; Sperry, J.S. Vulnerability of xylem cavitation and embolism. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 19–38. [Google Scholar] [CrossRef]
- Sperry, J.S.; Tyree, M.T. Mechanism of water stress-induced xylem embolism. Plant Physiol. 1988, 88, 581–587. [Google Scholar] [CrossRef] [Green Version]
- Adams, H.D.; Zeppel, M.J.B.; Anderegg, W.R.L.; Hartmann, H.; Landhausser, S.M.; Tissue, D.T.; Huxman, T.E.; Hudson, P.J.; Franz, T.E.; Allen, C.D.; et al. A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nat. Ecol. Evol. 2017, 1, 1285–1291. [Google Scholar] [CrossRef] [PubMed]
- Brodribb, T.J.; Cochard, H. Hydraulic failure defines the recovery and point of death in water-stressed conifers. Plant Physiol. 2009, 149, 575–584. [Google Scholar] [CrossRef] [Green Version]
- Lens, F.; Tixier, A.; Cochard, H.; Sperry, J.S.; Jansen, S.; Herbette, S. Embolism resistance as a key mechanism to understand adaptive plant strategies. Curr. Opin. Plant Biol. 2013, 16, 287–292. [Google Scholar] [CrossRef] [Green Version]
- Choat, B.; Sack, L.; Holbrook, N.M. Diversity of hydraulic traits in nine cordia species growing in tropical forests with contrasting precipitation. New Phytol. 2007, 175, 686–698. [Google Scholar] [CrossRef]
- Brodribb, T.J.; McAdam, S.A.; Jordan, G.J.; Martins, S.C. Conifer species adapt to low-rainfall climates by following one of two divergent pathways. Proc. Natl. Acad. Sci. USA 2014, 111, 14489–14493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinya, R.; Malhi, Y.; Fisher, J.B.; Brown, N.; Brodribb, T.J.; Aragao, L.E. Xylem cavitation vulnerability influences tree species’ habitat preferences in miombo woodlands. Oecologia 2013, 173, 711–720. [Google Scholar] [CrossRef]
- Oliveira, R.S. Can hydraulic traits be used to predict sensitivity of drought-prone forests to crown decline and tree mortality? Plant Soil 2013, 364, 1–3. [Google Scholar] [CrossRef]
- Pockman, W.T.; Sperry, J.S. Vulnerability to xylem cavitation and the distribution of sonoran desert vegetation. Am. J. Bot. 2000, 87, 1287–1299. [Google Scholar] [CrossRef] [Green Version]
- Brodribb, T.; Hill, R.S. The importance of xylem constraints in the distribution of conifer species. New Phytol. 1999, 143, 365–372. [Google Scholar] [CrossRef]
- Tyree, M.T.; Davis, S.D.; Cochard, H. Biophysical perspectives of xylem evolution: Is there a tradeoff of hydraulic effichîncy for vulnerability to dysfunction? IAWA J. 1994, 15, 335–360. [Google Scholar] [CrossRef] [Green Version]
- Brodribb, T.J.; Buckley, T.N. Leaf water transport: A core system in the evolution and physiology of photosynthesis. In The Leaf: A Platform for Performing Photosynthesis, Advances in Photosynthesis and Respiration; Adams, W.W., III, Terashima, I., Eds.; Springer International Publishing AG: Berlin/Heidelberg, Germany, 2018; pp. 81–92. [Google Scholar]
- Anderegg, W.; Wolf, A.; Arangovelez, A.; Choat, B.; Chmura, D.J.; Jansen, S.; Kolb, T.; Li, S.; Meinzer, F.C.; Pita, P. Woody plants optimise stomatal behaviour relative to hydraulic risk. Ecol. Lett. 2018, 21, 968–977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meinzer, F.C. Co-ordination of vapour and liquid phase water transport properties in plants. Plant Cell Environ. 2002, 25, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Zuccarini, P.; Ciurli, A.; Alpi, A.; Hegedüšová, K. Hydraulic and chemical mechanisms in the response of pinus pinaster ait. To conditions of water stress. Ekol. Bratisl. 2011, 30, 422–437. [Google Scholar]
- Mittelheuser, C.J.; van Stevenick, R.F.M. Stomatal closure and the inhibition of transpiration by (rs)-abscisic acid. Nature 1969, 221, 281–282. [Google Scholar] [CrossRef]
- Franks, P.J.; Berry, J.A.; Lombardozzi, D.L.; Bonan, G.B. Stomatal function across temporal and spatial scales: Deep-time trends, land-atmosphere coupling and global models. Plant Physiol. 2017, 174, 583–602. [Google Scholar] [CrossRef] [Green Version]
- Santelia, D.; Lawson, T. Rethinking guard cell metabolism. Plant Physiol. 2016, 172, 1371–1392. [Google Scholar] [CrossRef] [Green Version]
- Brodribb, T.J.; McAdam, S.A. Passive origins of stomatal control in vascular plants. Science 2011, 331, 582–585. [Google Scholar] [CrossRef]
- Martin-StPaul, N.; Delzon, S.; Cochard, H. Plant resistance to drought depends on timely stomatal closure. Ecol. Lett. 2017, 20, 1437–1447. [Google Scholar] [CrossRef] [PubMed]
- Powers, J.S.; Vargas-G, G.; Brodribb, T.J.; Schwartz, N.B.; Perez-Aviles, D.; Smith-Martin, C.M.; Becknell, J.M.; Aureli, F.; Blanco, R.; Calderón-Morales, E.; et al. A catastrophic tropical drought kills hydraulically vulnerable tree species. Glob. Chang. Biol. 2020, 26, 3122–3133. [Google Scholar] [CrossRef]
- Creek, D.; Lamarque, L.J.; Torres-Ruiz, J.M.; Parise, C.; Burlett, R.; Tissue, D.T.; Delzon, S. Xylem embolism in leaves does not occur with open stomata: Evidence from direct observations using the optical visualization technique. J. Exp. Bot. 2020, 71, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, C.; Coste, S.; Stahl, C.; Delzon, S.; Levionnois, S.; Cazal, J.; Cochard, H.; Esquivel-Muelbert, A.; Goret, J.-Y.; Heuret, P. Large hydraulic safety margins protect neotropical canopy rainforest tree species against hydraulic failure during drought. Ann. For. Sci. 2019, 76, 115. [Google Scholar] [CrossRef] [Green Version]
- Choat, B.; Brodribb, T.J.; Brodersen, C.R.; Duursma, R.A.; López, R.; Medlyn, B.E. Triggers of tree mortality under drought. Nature 2018, 558, 531–539. [Google Scholar] [CrossRef]
- Benito Garzón, M.; González Muñoz, N.; Wigneron, J.P.; Moisy, C.; Fernández-Manjarrés, J.; Delzon, S. The legacy of water deficit on populations having experienced negative hydraulic safety margin. Glob. Ecol. Biogeogr. 2018, 27, 346–356. [Google Scholar] [CrossRef]
- Skelton, R.P.; West, A.G.; Dawson, T.E. Predicting plant vulnerability to drought in biodiverse regions using functional traits. Proc. Natl. Acad. Sci. USA 2015, 112, 5744–5749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, X.; Choat, B.; Zhang, Y.-J.; Guan, X.-Y.; Shi, W.; Cao, K.-F. Variation in xylem hydraulic structure and function of two mangrove species across a latitudinal gradient in eastern australia. Water 2021, 13, 850. [Google Scholar] [CrossRef]
- Sperry, J.S.; Tyree, M.T.; Donnelly, J.R. Vulnerability of xylem to embolism in a mangrove vs an inland species of rhizophoraceae. Physiol. Plant. 1988, 74, 276–283. [Google Scholar] [CrossRef]
- Ball, M.C. Ecophysiology of mangroves. Trees 1988, 2, 129–142. [Google Scholar] [CrossRef]
- Scholander, P.F.; Hammel, H.T.; Hemmingsen, E.A.; Bradstreet, E.D. Hydrostatic pressure and osmotic potential in leaves of mangroves and some other plants. Proc. Natl. Acad. Sci. USA 1964, 52, 119–125. [Google Scholar] [CrossRef] [Green Version]
- Ewers, F.W.; Lopez-Portillo, J.; Angeles, G.; Fisher, J.B. Hydraulic conductivity and embolism in the mangrove tree laguncularia racemosa. Tree Physiol. 2004, 24, 1057–1062. [Google Scholar] [CrossRef] [Green Version]
- Melcher, P.J.; Goldstein, G.; Meinzer, F.C.; Yount, D.E.; Jones, T.J.; Holbrook, N.M.; Huang, C. Water relations of coastal and estuarine rhizophora mangle: Xylem pressure potential and dynamics of embolism formation and repair. Oecologia 2001, 126, 182–192. [Google Scholar] [CrossRef]
- Reef, R.; Lovelock, C.E. Regulation of water balance in mangroves. Ann. Bot. 2015, 115, 385–395. [Google Scholar] [CrossRef] [Green Version]
- Krauss, K.W.; Lovelock, C.E.; Mckee, K.L.; Lopez-Hoffman, L.; Ewe, S.M.L.; Sousa, W.P. Environmental drivers in mangrove establishment and early development: A review. Aquat. Bot. 2008, 89, 105–127. [Google Scholar] [CrossRef]
- Duke, N.C. Mangrove floristics and biogeography. In Tropical Mangrove Ecosystems; Robertson, A.I., Alongi, D.M., Eds.; American Geophysiocal Union: Washington, DC, USA, 1992; Volume 41, pp. 63–100. [Google Scholar]
- Nguyen, H.T.; Meir, P.; Sack, L.; Evans, J.R.; Oliveira, R.S.; Ball, M.C. Leaf water storage increases with salinity and aridity in the mangrove avicennia marina: Integration of leaf structure, osmotic adjustment, and access to multiple water sources. Plant Cell Environ. 2017, 40, 1576–1591. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, H.T.; Meir, P.; Wolfe, J.; Mencuccini, M.; Ball, M.C. Plumbing the depths: Extracellular water storage in specialized leaf structures and its functional expression in a three-domain pressure-volume relationship. Plant Cell Environ. 2016, 40, 1021–1038. [Google Scholar] [CrossRef] [Green Version]
- Madrid, E.N.; Armitage, A.R.; Lopez-Portillo, J. Avicennia germinans (black mangrove) vessel architecture is linked to chilling and salinity tolerance in the gulf of mexico. Front. Plant Sci. 2014, 5, 503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmitz, N.; Egerton, J.; Lovelock, C.; Ball, M. Light-dependent maintenance of hydraulic function in mangrove branches: Do xylary chloroplasts play a role in embolism repair? New Phytol. 2012, 195, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Cardona-Olarte, P.; Krauss, K.W.; Twilley, R.R. Leaf gas exchange and nutrient use efficiency help explain the distribution of two neotropical mangroves under contrasting flooding and salinity. Int. J. For. Res. 2013, 2013, 524–625. [Google Scholar] [CrossRef] [Green Version]
- Lovelock, C.E.; Ball, M.C.; Choat, B.; Engelbrecht, B.M.; Holbrook, N.M.; Feller, I.C. Linking physiological processes with mangrove forest structure: Phosphorus deficiency limits canopy development, hydraulic conductivity and photosynthetic carbon gain in dwarf rhizophora mangle. Plant Cell Environ. 2006, 29, 793–802. [Google Scholar] [CrossRef] [Green Version]
- Sobrado, M. Effect of drought on leaf gland secretion of the mangrove avicennia germinans L. Trees 2002, 16, 1–4. [Google Scholar] [CrossRef]
- Sobrado, M. Effect of high external nacl concentration on the osmolality of xylem sap, leaf tissue and leaf glands secretion of the mangrove avicennia germinans (l.) L. Flora 2001, 196, 63–70. [Google Scholar] [CrossRef]
- Farnsworth, E.J.; Farrant, J.M. Reductions in abscisic acid are linked with viviparous reproduction in mangroves. Am. J. Bot. 1998, 85, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Saintilan, N.; Khan, N.S.; Ashe, E.; Kelleway, J.J.; Rogers, K.; Woodroffe, C.D.; Horton, B.P. Thresholds of mangrove survival under rapid sea level rise. Science 2020, 368, 1118–1121. [Google Scholar] [CrossRef] [PubMed]
- Lovelock, C.E. Blue carbon from the past forecasts the future. Science 2020, 368, 1050–1052. [Google Scholar] [CrossRef]
- Mafigholami, D.; Mahmoudi, B.; Zenner, E.K. An analysis of the relationship between drought events and mangrove changes along the northern coasts of the persian gulf and oman sea. Estuar. Coast. Shelf Sci. 2017, 199, 141–151. [Google Scholar] [CrossRef]
- Duke, N.C.; Kovacs, J.M.; Griffiths, A.D.; Preece, L.; Hill, D.J.E.; Oosterzee, P.V.; Mackenzie, J.; Morning, H.S.; Burrows, D. Large-scale dieback of mangroves in australia’s gulf of carpentaria: A severe ecosystem response, coincidental with an unusually extreme weather event. Mar. Freshw. Res. 2017, 68, 1816–1829. [Google Scholar] [CrossRef]
- Matthijs, S.; Tack, J.; Speybroeck, D.V.; Koedam, N. Mangrove species zonation and soil redox state, sulphide concentration and salinity in gazi bay (kenya), a preliminary study. Mangroves Salt Marshes 1999, 3, 243–249. [Google Scholar] [CrossRef]
- Clough, B.F. Primary productivity and growth of mangrove forests. In Tropical Mangrove Ecosystems; Robertson, A.I., Alongi, D.M., Eds.; American Geophysical Union: Washington, DC, USA, 1992; Volume 41, pp. 225–249. [Google Scholar]
- Tomlinson, P.B. The Botany of Mangroves; Cambridge University Press: Cambridge, UK; New York, NY, USA, 1986; p. xii. 413p. [Google Scholar]
- Odum, W.E.; McIvor, C.C.; Smith III, T.J. The Ecology of the Mangroves of South Florida: A Community Profile; DTIC Document 5137473; DTIC: Charlottesville, VA, USA, 1982. [Google Scholar]
- Scholander, P.; Hammel, H.; Hemmingsen, E.; Garey, W. Salt balance in mangroves. Plant Physiol. 1962, 37, 722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brodribb, T.J.; Carriqui, M.; Delzon, S.; Lucani, C. Optical measurement of stem xylem vulnerability. Plant Physiol. 2017, 174, 2054–2061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brodribb, T.J.; Bienaime, D.; Marmottant, P. Revealing catastrophic failure of leaf networks under stress. Proc. Natl. Acad. Sci. USA 2016, 113, 4865–4869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McAdam, S. Physicochemical quantification of abscisic acid levels in plant tissues with an added internal standard by ultra-performance liquid chromatography. Bio-Protocol 2015, 5, 1–13. [Google Scholar] [CrossRef]
- Vilaró, F.; Canela-Xandri, A.; Canela, R. Quantification of abscisic acid in grapevine leaf (vitis vinifera) by isotope-dilution liquid chromatography-mass spectrometry. Anal. Bioanal. Chem. 2006, 386, 306–312. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. Stat. Comput. 2015, 067, 133–199. [Google Scholar]
- Duursma, R.; Choat, B. Fitplc—An r package to fit hydraulic vulnerability curves. J. Plant Hydraul. 2017, 4, e002. [Google Scholar] [CrossRef]
- Paliyavuth, C.; Clough, B.; Patanaponpaiboon, P. Salt uptake and shoot water relations in mangroves. Aquat. Bot. 2004, 78, 349–360. [Google Scholar] [CrossRef]
- Waisel, Y.; Eshel, A.; Agami, M. Salt balance of leaves of the mangrove avicennia marina. Physiol. Plant. 1986, 67, 67–72. [Google Scholar] [CrossRef]
- Moon, G.J.; Clough, B.F.; Peterson, C.A.; Allaway, W.G. Apoplastic and symplastic pathways in avicennia marina (forsk.) vierh. Roots revealed by fluorescent tracer dyes. Funct. Plant Biol. 1986, 13, 637–648. [Google Scholar] [CrossRef]
- Oren, R.; Sperry, J.; Katul, G.; Pataki, D.; Ewers, B.; Phillips, N.; Schäfer, K. Survey and synthesis of intra-and interspecific variation in stomatal sensitivity to vapour pressure deficit. Plant Cell Environ. 1999, 22, 1515–1526. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Chan, Z.; Gao, J.; Xing, L.; Cao, M.; Yu, C.; Hu, Y.; You, J.; Shi, H.; Zhu, Y. Aba receptor pyl9 promotes drought resistance and leaf senescence. Proc. Natl. Acad. Sci. USA 2016, 113, 1949–1954. [Google Scholar] [CrossRef] [Green Version]
- López-Portillo, J.; Ewers, F.W.; Angeles, G. Sap salinity effects on xylem conductivity in two mangrove species. Plant Cell Environ. 2005, 28, 1285–1292. [Google Scholar] [CrossRef]
- Hammond, W.M.; Yu, K.; Wilson, L.A.; Will, R.E.; Anderegg, W.R.L.; Adams, H.D. Dead or dying? Quantifying the point of no return from hydraulic failure in drought-induced tree mortality. New Phytol. 2019, 223, 1834–1843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jantsch, A.; Melo Júnior, J.C.F.d.; Amorim, M.W.; Larcher, L.; Soffiatti, P. Wood anatomy of laguncularia racemosa (combretaceae) in mangrove and transitional forest, southern brazil. Rev. De Biol. Trop. 2018, 66, 647–657. [Google Scholar] [CrossRef] [Green Version]
- Robert, E.M.; Koedam, N.; Beeckman, H.; Schmitz, N. A safe hydraulic architecture as wood anatomical explanation for the difference in distribution of the mangroves avicennia and rhizophora. Funct. Ecol. 2009, 23, 649–657. [Google Scholar] [CrossRef]
- Schmitz, N.; Robert, E.M.R.; Verheyden, A.; Kairo, J.G.; Beeckman, H.; Koedam, N. A patchy growth via successive and simultaneous cambia: Key to success of the most widespread mangrove species avicennia marina? Ann. Bot. 2008, 101, 49–58. [Google Scholar] [CrossRef]
- Cochard, H.; Damour, G.; Bodet, C.; Tharwat, I.; Poirier, M.; Améglio, T. Evaluation of a new centrifuge technique for rapid generation of xylem vulnerability curves. Physiol. Plant. 2005, 124, 410–418. [Google Scholar] [CrossRef]
- Sperry, J.S.; Donnelly, J.R.; Tyree, M.T. A method for measuring hydraulic conductivity and embolism in xylem. Plant Cell Environ. 1988, 11, 35–40. [Google Scholar] [CrossRef]
- Wheeler, J.K.; Huggett, B.A.; Tofte, A.N.; Rockwell, F.E.; Holbrook, N.M. Cutting xylem under tension or supersaturated with gas can generate plc and the appearance of rapid recovery from embolism. Plant Cell Environ. 2013, 36, 1938–1949. [Google Scholar] [CrossRef] [PubMed]
- Sperry, J.; Saliendra, N. Intra-and inter-plant variation in xylem cavitation in betula occidentalis. Plant Cell Environ. 1994, 17, 1233–1241. [Google Scholar] [CrossRef]
- Rockwell, F.E.; Wheeler, J.K.; Holbrook, N.M. Cavitation and its discontents: Opportunities for resolving current controversies. Plant Physiol. 2014, 164, 1649–1660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.J.; Maenpuen, P.; Zhang, Y.J.; Barai, K.; Katabuchi, M.; Gao, H.; Kaewkamol, S.; Tao, L.B.; Zhang, J.L. Quantifying vulnerability to embolism in tropical trees and lianas using five methods: Can discrepancies be explained by xylem structural traits? New Phytol. 2021, 229, 805–819. [Google Scholar] [CrossRef]
- Skelton, R.; Diaz, J. Quantifying losses of plant hydraulic function: Seeing the forest, the trees and the xylem. Tree Physiol. 2020, 40, 285–289. [Google Scholar] [CrossRef]
- Brodribb, T.; Carriquí, M.; Delzon, S.; McAdam, S.; Holbrook, N. Advanced vascular function discovered in a widespread moss. Nat. Plants 2020, 6, 273–279. [Google Scholar] [CrossRef]
- Skelton, R.P.; Brodribb, T.J.; Choat, B. Casting light on xylem vulnerability in an herbaceous species reveals a lack of segmentation. New Phytol. 2017, 214, 561–569. [Google Scholar] [CrossRef] [Green Version]
- Venturas, M.D.; Pratt, R.B.; Jacobsen, A.L.; Castro, V.; Fickle, J.C.; Hacke, U.G. Direct comparison of four methods to construct xylem vulnerability curves: Differences among techniques are linked to vessel network characteristics. Plant Cell Environ. 2019, 42, 2422–2436. [Google Scholar] [CrossRef]
- Pratt, R.B.; Castro, V.; Fickle, J.C.; Jacobsen, A.L. Embolism resistance of different aged stems of a california oak species (quercus douglasii): Optical and microct methods differ from the benchtop-dehydration standard. Tree Physiol. 2019, 40, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Simonin, K.A.; Limm, E.B.; Dawson, T.E. Hydraulic conductance of leaves correlates with leaf lifespan: Implications for lifetime carbon gain. New Phytol. 2012, 193, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Tyree, M.T.; Zimmermann, M.H. Xylem Structure and the Ascent of Sap; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Tyree, M.T.; Ewers, F.W. The hydraulic architecture of trees and other woody plants. New Phytol. 1991, 119, 345–360. [Google Scholar] [CrossRef]
- Zimmermann, M.H. Hydraulic architecture of some diffuse-porous trees. Can. J. Bot. 1978, 56, 2286–2295. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, E.K.A.; Abdel-Hamid, A. Zonation pattern of avicennia marina and rhizophora mucronata along the red sea coast, egypt. World Appl. Sci. J. 2007, 2, 283–288. [Google Scholar]
- Coopman, R.E.; Nguyen, H.T.; Mencuccini, M.; Oliveira, R.S.; Sack, L.; Lovelock, C.E.; Ball, M.C. Harvesting water from unsaturated atmospheres: Deliquescence of salt secreted onto leaf surfaces drives reverse sap flow in a dominant arid climate mangrove, avicennia marina. New Phytol. 2021. [CrossRef]
- Fuenzalida, T.I.; Bryant, C.J.; Ovington, L.I.; Yoon, H.-J.; Oliveira, R.S.; Sack, L.; Ball, M.C. Shoot surface water uptake enables leaf hydraulic recovery in avicennia marina. New Phytol. 2019, 2019, 1504–1511. [Google Scholar] [CrossRef] [PubMed]

| Species | A ** (μmol m−2 s−1) | gs ** (mol m−2 s−1) | Ci (μmol mol−1) | A/gs (μmol mol−1) | Tr * (μmol m−2 s−1) |
|---|---|---|---|---|---|
| Avicennia MaRina | 13.43 ± 2.61 | 0.21 ± 0.03 | 296.3 ± 16.1 | 63.2 ± 9.7 | 4.26 ± 0.53 |
| Bruguiera Gymnorrhiza | 8.25 ± 1.52 | 0.15 ± 0.01 | 317.7 ± 17.7 | 56.3 ± 12.7 | 3.28 ± 0.27 |
| Trait * | Avicennia marina | Bruguiera gymnorrhiza | t-Test |
|---|---|---|---|
| P88_seedlings (MPa) | −9.56 ± 0.46 | −4.53 ± 0.35 | p < 0.0001 |
| P88_mature (MPa) | −10.75 ± 0.90 | −3.02 ± 0.07 | p < 0.0001 |
| P50_seedlings (MPa) | −7.55 ± 0.29 | −4.13 ± 0.33 | p < 0.0001 |
| P50_mature (MPa) | −8.30 ± 0.17 | −2.83 ± 0.10 | p < 0.0001 |
| P12_seedlings (MPa) | −5.39 ± 0.44 | −3.60 ± 0.32 | p < 0.0001 |
| P12_mature (MPa) | −5.71 ± 0.48 | −2.56 ± 0.15 | p < 0.0001 |
| Pg12_seedlings (MPa) | −3.80 | −3.08 | NA |
| Pg50_seedlings (MPa) | −2.63 | −2.70 | NA |
| Pg88_seedlings (MPa) | −1.47 | −2.33 | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, G.-F.; Brodribb, T.J.; Roddy, A.B.; Lei, J.-Y.; Si, H.-T.; Pahadi, P.; Zhang, Y.-J.; Cao, K.-F. Contrasting Water Use, Stomatal Regulation, Embolism Resistance, and Drought Responses of Two Co-Occurring Mangroves. Water 2021, 13, 1945. https://doi.org/10.3390/w13141945
Jiang G-F, Brodribb TJ, Roddy AB, Lei J-Y, Si H-T, Pahadi P, Zhang Y-J, Cao K-F. Contrasting Water Use, Stomatal Regulation, Embolism Resistance, and Drought Responses of Two Co-Occurring Mangroves. Water. 2021; 13(14):1945. https://doi.org/10.3390/w13141945
Chicago/Turabian StyleJiang, Guo-Feng, Timothy J. Brodribb, Adam B. Roddy, Jin-Yan Lei, Huai-Tong Si, Pratima Pahadi, Yong-Jiang Zhang, and Kun-Fang Cao. 2021. "Contrasting Water Use, Stomatal Regulation, Embolism Resistance, and Drought Responses of Two Co-Occurring Mangroves" Water 13, no. 14: 1945. https://doi.org/10.3390/w13141945







