Pollution Biomarkers in the Framework of Marine Biodiversity Conservation: State of Art and Perspectives
Abstract
:1. Introduction
2. Pollution Biomarkers
3. Pollution Biomarkers in Biomonitoring of Marine Protected Areas
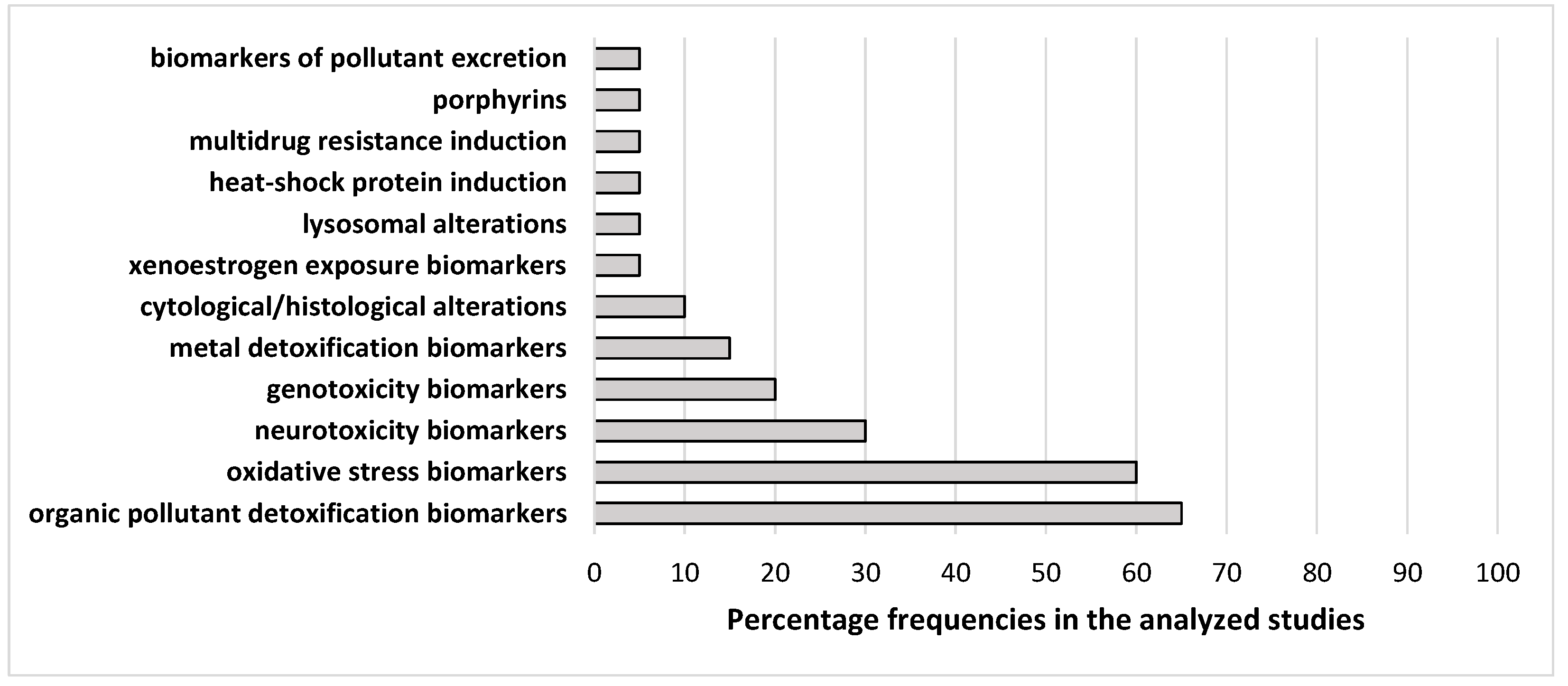
4. Biomarker Analysis in Endangered Species
5. Perspectives for Future Research in the Biomarker Development in the Framework of Biodiversity Conservation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rios, L.M.; Moore, C.; Jones, P.R. Persistent organic pollutants carried by synthetic polymers in the ocean environment. Mar. Pollut. Bull. 2007, 54, 1230–1237. [Google Scholar] [CrossRef]
- Mearns, A.J.; Morrison, A.M.; Arthur, C.; Rutherford, N.; Bissell, M.; Rempel-Hester, M.A. Effect of pollution on marine organisms. Water Environ. Res. 2020, 92, 1510–1532. [Google Scholar] [CrossRef]
- Tornero, V.; Hanke, G. Chemical contaminants entering the marine environment from sea-based sources: A review with a focus on European seas. Mar. Pollut. Bull. 2016, 112, 17–38. [Google Scholar] [CrossRef]
- Lionetto, F.; Corcione, C.E. An Overview of the Sorption Studies of Contaminants on Poly(Ethylene Terephthalate) Microplastics in the Marine Environment. J. Mar. Sci. Eng. 2021, 9, 445. [Google Scholar] [CrossRef]
- Salimi, M.; Esrafili, A.; Gholami, M.; Jafari, A.J.; Kalantari, R.R.; Farzdakia, M.; Kermani, M.; Sobhi, H.R. Contaminants of emerging concern: A review of new approach in AOP technologies. Environ. Monit. Assess. 2017, 189, 414. [Google Scholar] [CrossRef]
- Torres-Padrón, M.E.; Montesdeoca-Esponda, S.; Santana-Viera, S.; Guedes-Alonso, R.; Herrera-Melián, J.A.; Sosa-Ferrera, Z.; Santana-Rodríguez, J.J. An Update of the Occurrence of Organic Contaminants of Emerging Concern in the Canary Islands (Spain). Water 2020, 12, 2548. [Google Scholar] [CrossRef]
- Backhaus, T.; Snape, J.; Lazorchak, J. The impact of chemical pollution on biodiversity and ecosystem services: The need for an improved understanding. Integr. Environ. Assess. Manag. 2012, 8, 575–576. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, P.B.; Rastogi, N. Pollution vis-a-vis Biodiversity. In Environmental Stress: Indication, Mitigation and Eco-Conservation; Yunus, M., Singh, N., de Kok, L.J., Eds.; Kluver Academic Publishers: Amsterdam, The Netherlands, 2000; pp. 73–88. [Google Scholar]
- Fernandez, M.A. Populations Collapses in Marine Invertebrates Due to Endocrine Disruption: A Cause for Concern? Front. Endocrinol. 2019, 10, 721. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Vilella, S.; Trischitta, F.; Cappello, M.S.; Giordano, M.E.; Schettino, T. Effects of CdCl2 on electrophysiological parameters in the intestine of the teleost fish, Anguilla Anguilla. Aquat. Toxicol. 1998, 41, 251–264. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Giordano, M.E.; Vilella, S.; Schettino, T. Inhibition of eel enzymatic activities by cadmium. Aquat. Toxicol. 2000, 48, 561–571. [Google Scholar] [CrossRef]
- Calisi, A.; Lionetto, M.G.; Caricato, R.; Giordano, M.E.; Schettino, T. Morphometric alterations in Mytilus galloprovincialis granulocytes: A new biomarker. Environ. Toxicol. Chem. 2008, 27, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Bolognesi, C.; Cirillo, S. Genotoxicity biomarkers in aquatic bioindicators. Curr. Zool. 2014, 60, 273–284. [Google Scholar] [CrossRef]
- Regoli, F.; Giuliani, M.E. Oxidative pathways of chemical toxicity and oxidative stress biomarkers in marine organisms. Mar. Environ. Res. 2014, 93, 106–117. [Google Scholar] [CrossRef] [PubMed]
- UNEP/POPS/POPRC.8/16/Annex V Guidance for drafters of risk Profiles on consideration of Toxicological Interactions When Evaluating Chemicals Proposed for Listing—Qualitative Literature-BASED approach to Assessing Mixture Toxicity under Annex E. Available online: www.pops.int/TheConvention/POPsReviewCommittee/Guidance/ (accessed on 5 May 2021).
- Rodea-Palomares, I.; Makowski, M.; Gonzalo, S.; González-Pleiter, M.; Leganés, F.; Fernández-Piñas, F. Effect of PFOA/PFOS pre-exposure on the toxicity of the herbicides 2,4-D, Atrazine, Diuron and Paraquat to a model aquatic photosynthetic microorganism. Chemosphere 2015, 139, 65–72. [Google Scholar] [CrossRef]
- Zeng, X.; Chen, X.; Zhuang, J. The positive relationship between ocean acidification and pollution. Mar. Pollut. Bull. 2015, 91, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Owen, R.; Galloway, T.S.; Hagger, J.A.; Jones, M.B.; Depledge, M.H. Biomarkers and environmental risk assessment: Guiding principles from the human health field. Mar. Pollut. Bull. 2008, 56, 613–619. [Google Scholar] [CrossRef]
- Hook, S.H.; Gallagher, E.P.; Batley, G.E. The role of biomarkers in the assessment of aquatic ecosystem health. Integr. Environ. Assess. Manag. 2014, 10, 327–341. [Google Scholar] [CrossRef] [Green Version]
- Schettino, T.; Caricato, R.; Calisi, A.; Giordano, M.E.; Lionetto, M.G. Biomarker Approach in Marine Monitoring and Assessment: New Insights and Perspectives. Open Environ. Sci. 2012, 6, 20–27. [Google Scholar] [CrossRef] [Green Version]
- Marigómez, I.; Garmendia, L.; Soto, M.; Orbea, A.; Izagirre, U.; Cajaraville, M.P. Marine ecosystem health status assessment through integrative biomarker indices: A comparative study after the Prestige oil spill “mussel watch”. Ecotoxicology 2013, 22, 486–505. [Google Scholar] [CrossRef] [Green Version]
- Hagger, J.A.; Jones, M.B.; Leonard, D.R.P.; Owen, R.; Galloway, T.S. Biomarkers and integrated environmental risk assessment: Are there more questions than answers? Integr. Environ. Assess. Manag. 2009, 2, 312–329. [Google Scholar] [CrossRef]
- Wang, W.C.; Mao, H.; Ma, D.D.; Yang, W.X. Characteristics, functions, and applications of metallothionein in aquatic vertebrates. Front. Mar. Sci. 2014, 1, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Lionetto, M.G.; Giordano, M.E.; Caricato, R.; Pascariello, M.F.; Marinosci, L.; Schettino, T. Biomonitoring of heavy metal contamination along Salento coast (Italy) by metallothionein evaluation in Mytilus galloprovincialis and Mullus barbatus. Aquat. Conserv. 2001, 11, 305–310. [Google Scholar] [CrossRef]
- Lacorn, M.; Lahrssen, A.; Rotzoll, N.; Simat, T.J.; Steinhart, H. Quantification of metallothionein isoforms in fish liver and its implications for biomonitoring. Environ. Toxicol. Chem. 2001, 20, 140–145. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Caricato, R.; Giordano, M.E.; Schettino, T. Biomarker application for the study of chemical contamination risk on marine organisms in the Taranto marine coastal area. Chem. Ecol. 2004, 20, S333–S343. [Google Scholar] [CrossRef]
- Sen, A.; Ulutas, O.K.; Tutuncu, B.; Ertas, N.; Cok, I. Determination of 7- ethoxyresorufin-o-deethylase (EROD) induction in leaping mullet (Liza saliens) from the highly contaminated Aliaga Bay, Turkey. Environ. Monit. Assess. 2010, 165, 87–96. [Google Scholar] [CrossRef]
- Houtman, C.J.; Booij, P.; van der Valk, K.M.; van Bodegom, P.M.; van den Ende, F.; Gerritsen, A.A.M.; Lamoree, M.H.; Legler, A.B.; Brouwer, A. Biomonitoring of estrogenic exposure and identification of responsible compounds in bream from Dutch surface waters. Environ. Toxicol. Chem. 2007, 26, 898–907. [Google Scholar] [CrossRef]
- Leomanni, A.; Schettino, T.; Calisi, A.; Gorbi, S.; Mezzelani, M.; Regoli, F.; Lionetto, M.G. Antioxidant and oxidative stress related responses in the Mediterranean land snail Cantareus apertus exposed to the carbamate pesticide Carbaryl. Comp. Biochem. Physiol. C 2015, 168, 20–27. [Google Scholar] [CrossRef]
- Parra, S.; Varandas, S.; Santos, D.; Félix, L.; Fernandes, L.; Cabecinha, E.; Gago, J.; Monteiro, S.M. Multi-biomarker responses of Asian clam Corbicula fluminea (bivalvia, corbiculidea) to cadmium and microplastics pollutants. Water 2021, 13, 394. [Google Scholar] [CrossRef]
- Köhler, A.; Wahl, E.; Söffker, K. Functional and morphological changes of lysosomes as prognostic biomarkers of toxic liver injury in a marine flatfish (Platichthysf lesus (L.)). Environ. Toxicol. Chem. 2002, 21, 2434–2444. [Google Scholar] [CrossRef]
- Moore, M.N.; Allen, J.I.; McVeigh, A. Environmental Prognostics: An integrated model supporting lysosomal stress responses as predictive biomarkers of animal health status. Mar. Environ. Res. 2006, 61, 278–304. [Google Scholar] [CrossRef] [PubMed]
- Calisi, A.; Grimaldi, A.; Leomanni, A.; Lionetto, M.G.; Dondero, F.; Schettino, T. Multibiomarker response in the earthworm Eisenia foetida as tool for assessing multiwalled carbon nanotube ecotoxicity. Ecotoxicology 2016, 25, 677–687. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Caricato, R.; Giordano, M.E.; Pascariello, M.F.; Marinosci, L.; Schettino, T. Integrated use of biomarkers (acetylcholinesterase and antioxidant enzymatic activities) in Mytilus galloprovincialis and Mullus barbatus in an Italian coastal marine area. Mar. Pollut. Bull. 2003, 46, 324–330. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Caricato, R.; Erroi, E.; Giordano, M.E.; Schettino, T. Potential application of carbonic anhydrase activity in bioassay and biomarker studies. Chem. Ecol. 2006, 22, S119–S125. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Caricato, R.; Giordano, M.E.; Erroi, E.; Schettino, T. Carbonic anhydrase as pollution biomarker: An ancient enzyme with a new use. Int. J. Environ. Res. Public Health 2012, 9, 3965–3977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lionetto, M.G.; Caricato, R.; Giordano, M.E.; Schettino, T. The complex relationship between metals and carbonic anhydrase: New insights and perspectives. Int. J. Mol. Sci. 2016, 17, 127. [Google Scholar] [CrossRef] [Green Version]
- Caricato, R.; Giordano, M.E.; Schettino, T.; Lionetto, M.G. Functional involvement of carbonic anhydrase in the lysosomal response to cadmium exposure in Mytilus galloprovincialis digestive gland. Front. Physiol. 2018, 9, 319. [Google Scholar] [CrossRef] [PubMed]
- Caricato, R.; Giordano, M.E.; Schettino, T.; Maisano, M.; Mauceri, A.; Giannetto, A.; Cappello, T.; Parrino, V.; Ancora, S.; Caliani, I.; et al. Carbonic anhydrase integrated into a multimarker approach for the detection of the stress status induced by pollution exposure in Mytilus galloprovincialis: A field case study. Sci. Total Environ. 2019, 690, 140–150. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Caricato, R.; Giordano, M.E. Carbonic Anhydrase Sensitivity to Pesticides: Perspectives for Biomarker Development. Int. J. Mol. Sci. 2020, 21, 3562. [Google Scholar] [CrossRef]
- Zebral, Y.D.; da Silva Fonseca, J.; Marques, J.A.; Bianchini, A. Carbonic Anhydrase as a Biomarker of Global and Local Impacts: Insights from Calcifying Animals. Int. J. Mol. Sci. 2019, 20, 3092. [Google Scholar] [CrossRef] [Green Version]
- Schlenk, D. Necessity of defining biomarkers for use in ecological risk assessments. Mar. Pollut. Bull. 1999, 39, 48–53. [Google Scholar] [CrossRef]
- Klein, C.; Brown, C.; Halpern, B.; Segan, D.B.; McGowan, J.; Beger, M.; Watson, J.E.M. Shortfalls in the global protected area network at representing marine biodiversity. Sci. Rep. 2015, 5, 17539. [Google Scholar] [CrossRef] [Green Version]
- O’Leary, B.C.; Winther-Janson, M.; Bainbridge, J.M.; Aitken, J.; Hawkins, J.P.; Roberts, C.M. Effective Coverage Targets for Ocean Protection. Conserv. Lett. 2016, 9, 398–404. [Google Scholar] [CrossRef]
- Selig, E.R.; Turner, W.R.; Troëng, S.; Wallace, B.P.; Halpern, B.S.; Kaschner, K.; Lascelles, B.G.; Carpenter, K.E.; Mittermeier, R.A. Global Priorities for Marine Biodiversity Conservation. PLoS ONE 2014, 9, e82898. [Google Scholar] [CrossRef] [PubMed]
- Edgar, G.; Stuart-Smith, R.; Willis, T.; Kininmonth, S.; Baker, S.C.; Banks, S.; Barrett, N.S.; Becerro, M.A.; Bernard, A.T.F.; Berkhout, J.; et al. Global conservation outcomes depend on marine protected areas with five key features. Nature 2014, 506, 216–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abessa, D.M.S.; Albuquerque, H.C.; Morais, L.G.; Araújo, G.S.; Fonseca, T.G.; Cruz, A.C.F.; Campos, B.G.; Camargo, J.B.D.A.; Gusso-Choueri, P.K.; Perina, F.C.; et al. Pollution status of marine protected areas worldwide and the consequent toxic effects are unknown. Environ. Pollut. 2018, 243, 1450–1459. [Google Scholar] [CrossRef] [PubMed]
- Pozo, K.; Lazzerini, D.; Perra, G.; Volpi, V.; Corsolini, S.; Focardi, S. Levels and spatial distribution of polychlorinated biphenyls (PCBs) in superficial sediment from 15 Italian Marine Protected Areas (MPA). Mar. Pollut. Bull. 2009, 58, 773–776. [Google Scholar] [CrossRef] [PubMed]
- Chatwin, A. Priorities for Coastal and Marine Conservation in South America; The Nature Conservancy: Arlington, TX, USA, 2007. [Google Scholar]
- Bonacci, S.; Iacocca, A.; Fossi, S.; Lancini, L.; Caruso, T.; Corsi, I.; Focardi, S. Biomonitoring Aquatic Environmental Quality in a Marine Protected Area: A Biomarker Approach. AMBIO 2007, 36, 308–315. [Google Scholar] [CrossRef]
- Pinsino, A.; Della Torre, C.; Sammarini, V.; Bonaventura, R.; Amato, E.; Matranga, V. Sea urchin coelomocytes as a novel cellular biosensor of environmental stress: A field study in the Tremiti Island Marine Protected Area, Southern Adriatic Sea, Italy. Cell Biol. Toxicol. 2008, 24, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Moschino, V.; Schintu, M.; Marrucci, A.; Marras, B.; Nesto, N.; Da Ros, L. An ecotoxicological approach to evaluate the effects of tourism impacts in the Marine Protected Area of La Maddalena (Sardinia, Italy). Mar. Pollut. Bull. 2017, 122, 306–315. [Google Scholar] [CrossRef]
- Caliani, I.; Rodríguez, L.P.; Casini, S.; Granata, A.; Zagami, G.; Pansera, M.; Querci, G.; Minutoli, R. Biochemical and genotoxic biomarkers in Atherina boyeri to evaluate the status of aquatic ecosystems. Reg. Stud. Mar. Sci. 2019, 28, 100566. [Google Scholar] [CrossRef] [Green Version]
- Fossi, M.C.; Borsani, J.F.; Di Mento, R.; Marsili, L.; Casini, S.; Neri, G.; Mori, G.; Ancora, S.; Leonzio, C.; Minutoli, R.; et al. Multi-trial biomarker approach in Meganyctiphanes norvegica: A potential early indicator of health status of the Mediterranean “whale sanctuary”. Mar. Environ. Res. 2002, 54, 761–767. [Google Scholar] [CrossRef]
- Fossi, M.C.; Panti, C.; Marsili, L.; Maltese, S.; Spinsanti, G.; Casini, S.; Caliani, I.; Gaspari, S.; Muñoz-Arnanz, J.; Jimenez, B.; et al. The Pelagos Sanctuary for Mediterranean marine mammals: Marine Protected Area (MPA) or marine polluted area? The case study of the striped dolphin (Stenella coeruleoalba). Mar. Pollut. Bull. 2013, 70, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Fossi, M.C.; Marsili, L.; Baini, M.; Giannetti, M.; Coppola, D.; Guerranti, C.; Caliani, I.; Minutoli, R.; Lauriano, G.; Finoia, M.G.; et al. Fin whales and microplastics: The Mediterranean Sea and the Sea of Cortez scenarios. Environ. Pollut. 2016, 209, 68–78. [Google Scholar] [CrossRef]
- Marangi, M.; Airoldi, S.; Beneduce, L.; Zaccone, C. Wild whale faecal samples as a proxy of anthropogenic impact. Sci. Rep. 2021, 11, 5822. [Google Scholar] [CrossRef]
- Downs, C.A.; Fauth, J.E.; Robinson, C.E.; Curry, R.; Lanzendorf, B.; Halas, J.C.; Halas, J.; Woodley, C.M. Cellular diagnostics and coral health: Declining coral health in the Florida Keys. Mar. Pollut. Bull. 2005, 51, 558–569. [Google Scholar] [CrossRef]
- Gold-Bouchot, G.; Rubio-Piña, J.; Montero-Muñoz, J.; Ramirez-Miss, N.; Echeverría-García, A.; Patiño-Suarez, V.; Puch-Hau, C.A.; Zapata-Pérez, O. Pollutants and biomarker responses in two reef fish species (Haemulon aurolineatum and Ocyurus chrysurus) in the Southern Gulf of Mexico. Mar. Pollut. Bull. 2017, 116, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, C.; Leija, A.; Torres, M.; Mendoza, R. Assessment of Environmental Quality in the Tamaulipas Laguna Madre, Gulf of Mexico, by Integrated Biomarker Response Using the Cross-Barred Venus Clam Chione elevata. Water Air Soil Pollut. 2019, 230, 27. [Google Scholar] [CrossRef]
- Ramos, R.; Bastidas, C.; Debrot, D.; García, E. Phase I and II biotransformation and antioxidant enzymes in the coral Siderastrea siderea act as biomarkers for reproductive condition and habitat quality. Mar. Biol. Res. 2011, 7, 398–406. [Google Scholar]
- De Freitas Prazeres, M.; Eslava Martins, S.; Bianchini, A. Assessment of water quality in coastal waters of Fernando de Noronha, Brazil: Biomarker analyses in Amphistegina lesson. J. Foramin. Res. 2012, 42, 56–65. [Google Scholar] [CrossRef]
- De Oliveira Ribeiro, C.A.; Katsumiti, A.; França, P.; Maschio, J.; Zandoná, E.; Cestari, M.M.; Vicari, T.; Roche, H.; de Assis, H.C.S.; Neto, F.F. Biomarkers responses in fish (Atherinella brasiliensis) of Paraganuá Bay, Southern Brazil, for assessment of pollution effects. Braz. J. Oceanogr. 2013, 61, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Gusso-Choueri, P.K.; Choueri, R.B.; de Araújo, G.S.; Cruz, A.C.F.; Stremel, T.; Campos, S.; de Sousa Abessa, D.M.; Oliveira Ribeiro, C.A. Assessing pollution in marine protected areas: The role of a multi-biomarker and multi-organ approach. Environ. Sci. Pollut. Res. 2015, 22, 18047–18065. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gusso-Choueri, P.K.; Choueri, R.B.; Santos, G.S.; de Araújo, G.S.; Cruz, A.C.F.; Stremel, T.; de Campos, S.X.; Cestari, M.M.; Oliveira Ribeiro, C.A.; de Sousa Abessa, D.M. Assessing genotoxic effects in fish from a marine protected area influenced by former mining activities and other stressors. Mar. Pollut. Bull. 2016, 104, 229–239. [Google Scholar] [CrossRef] [Green Version]
- Giarratano, E.; Gil, M.N.; Marinho, C.H.; Malanga, G. Metals from mine waste as potential cause of oxidative stress in burrowing crab Neohelice granulata from San Antonio bay. Ecotoxicol. Environ. Saf. 2016, 132, 68–76. [Google Scholar] [CrossRef]
- Araujo, G.S.; Gusso-Choueri, P.K.; Favaro, D.I.T.; Rocha, R.C.C.; Saint’Pierre, T.D.; Hauser-Davis, R.A.; Braz, B.; Santelli, R.E.; Freire, A.S.; Machado, W.T.V.; et al. Metal-Associated Biomarker Responses in Crabs from a Marine Protected Area in Southeastern Brazil. Arch. Environ. Contam. Toxicol. 2020, 78, 463–477. [Google Scholar] [CrossRef]
- Salgado, L.D.; Meister Luz Marques, A.E.; Kramer, R.D.; de Oliveira, F.G.; Moretto, S.L.; de Lima, B.A.; Prodocimo, M.M.; Cestari, M.M.; de Azevedo, J.C.R.; de Assis, H.C.S. Sediment contamination and toxic effects on Violet Goby fish (Gobioides broussonnetii—Gobiidae) from a marine protected area in South Atlantic. Environ. Res. 2021, 195, 110308. [Google Scholar] [CrossRef]
- Klumpp, D.; Humphrey, C.; Codi King, S. Biomarker Responses in Coral Trout (Plectropomus leopardus) as an Indicator of Exposure to Contaminants in a Coral Reef Environment. Australas J. Ecotoxicol. 2007, 13, 9–17. [Google Scholar]
- Jin, Y.K.; Lundgren, P.; Lutz, A.; Raina, J.-B.; Howells, E.J.; Paley, A.S.; van Oppen, M.J.H. Genetic markers for antioxidant capacity in a reef-building coral. Sci. Adv. 2016, 2, e1500842. [Google Scholar] [CrossRef] [Green Version]
- Bacchetta, C.; Rossi, A.; Aleb, A.; Campana, M.; Parma, M.J. Combined toxicological effects of pesticides: A fish multi-biomarker approach. Ecol. Indic. 2014, 36, 532–538. [Google Scholar] [CrossRef]
- Burkina, V.; Zlabek, V.; Zamaratskaia, G. Effects of pharmaceuticals present in aquatic environment on Phase I metabolism in fish. Environ. Toxicol. Pharmacol. 2015, 40, 430–444. [Google Scholar] [CrossRef]
- Maisano, M.; Cappello, T.; Natalotto, A.; Vitale, V.; Parrino, V.; Giannetto, A.; Oliva, S.; Mancini, G.; Cappello, S.; Mauceri, A.; et al. Effects of petrochemical contamination on caged marine mussels using a multi-biomarker approach: Histological changes, neurotoxicity and hypoxic stress. Mar. Environ. Res. 2017, 128, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Micheletti, C.; Critto, A.; Marcomini, A. Assessment of ecological risk from bioaccumulation of PCDD/Fs and dioxin-like PCBs in a coastal lagoon. Environ. Int. 2007, 33, 45–55. [Google Scholar] [CrossRef]
- Ricciardi, F.; Matozzo, V.; Binelli, A.; Marin, M.G. Biomarker responses and contamination levels in crabs (Carcinus aestuarii) from the Lagoon of Venice: An integrated approach in biomonitoring estuarine environments. Water Res. 2010, 44, 1725–1736. [Google Scholar] [CrossRef]
- Finlayson, K.A.; Leusch, F.D.L.; van de Merwe, J.P. The current state and future directions of marine turtle toxicology research. Environ. Int. 2016, 94, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Richardson, K.L.; Lopez Castro, M.; Gardner, S.C.; Schlenk, D. Polychlorinated Biphenyls and Biotransformation Enzymes in Three Species of Sea Turtles from the Baja California Peninsula of Mexico. Arch. Environ. Contam. Toxicol. 2010, 58, 183–193. [Google Scholar] [CrossRef] [Green Version]
- Labrada-Martagón, V.; Rodríguez, P.A.T.; Méndez-Rodríguez, L.C.; Zenteno-Savín, T. Oxidative stress indicators and chemical contaminants in East Pacific green turtles (Chelonia mydas) inhabiting two foraging coastal lagoons in the Baja California peninsula. Comp Biochem. Physiol. C 2011, 154, 65–75. [Google Scholar] [CrossRef]
- Keller, J.M. Occurrence and Effects of Organochlorine Contaminants in Sea Turtles; Umi Microforum 3092875; ProQuest Information and Learning Company: Ann Arbor, MI, USA, 2003. [Google Scholar]
- Caliani, I.; Campani, T.; Giannetti, M.; Marsili, L.; Casini, S.; Fossi, M.C. First application of comet assay in blood cells of Mediterranean loggerhead sea turtle (Caretta caretta). Mar. Environ. Res. 2018, 96, 68–72. [Google Scholar]
- Andreani, G.; Santoro, M.; Cottignoli, S.; Fabbri, M.; Carpenè, M.; Isani, G. Metal distribution and metallothionein in loggerhead (Caretta caretta) and green (Chelonia mydas) sea turtles. Sci. Total Environ. 2008, 390, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Casini, S.; Caliani, I.; Giannetti, M.; Marsili, L.; Maltese, S.; Coppola, D.; Bianchi, N.; Campani, T.; Ancora, S.; Caruso, C.; et al. First ecotoxicological assessment of Caretta caretta (Linnaeus, 1758) in the Mediterranean Sea using an integrated nondestructive protocol. Sci. Total Environ. 2018, 631–632, 1221–1233. [Google Scholar] [CrossRef] [PubMed]
- Foltz, K.M.; Baird, R.W.; Ylitalo, G.M.; Jensen, B.A. Cytochrome P4501A1 expression in blubber biopsies of endangered false killer whales (Pseudorca crassidens) and nine other odontocete species from Hawai’i. Ecotoxicology 2014, 23, 1607–1618. [Google Scholar] [CrossRef] [PubMed]
- Baini, M.; Panti, C.; Fossi, M.C.; Tepsich, P.; Jiménez, B.; Coomber, F.; Bartalini, A.; Muñoz-Arnanz, J.; Moulins, A.; Rosso, M. First assessment of POPs and cytochrome P450 expression in Cuvier’s beaked whales (Ziphius cavirostris) skin biopsies from the Mediterranean Sea. Sci. Rep. 2020, 10, 21891. [Google Scholar] [CrossRef]
- Mancia, A.; Abelli, L.; Fossi, M.C.; Panti, C. Skin distress associated with xenobiotics exposure: An epigenetic study in the Mediterranean fin whale (Balaenoptera physalus). Mar. Genom 2021, 57, 100822. [Google Scholar] [CrossRef]
- Consales, G.; Marsili, L. Assessment of the conservation status of Chondrichthyans: Underestimation of the pollution threat. Eur. Zool. J. 2021, 1, 165–180. [Google Scholar] [CrossRef]
- Barrera-García, A.; O’Hara, T.; Galván-Magaña, F.; Méndez-Rodríguez, L.C.; Castellini, J.M.; Zenteno-Savín, T. Oxidative stress indicators and trace elements in the blue shark (Prionace glauca) off the east coast of the Mexican Pacific Ocean. Comp. Biochem. Physiol. Part. C 2012, 156, 59–66. [Google Scholar] [CrossRef]
- Barrera-García, A.; O’Hara, T.; Galván-Magaña, F.; Méndez-Rodríguez, L.C.; Castellini, J.M.; Zenteno-Savín, T. Trace elements and oxidative stress indicators in the liver and kidney of the blue shark (Prionace glauca). Comp. Biochem. Physiol. Part. A 2013, 165, 483–490. [Google Scholar] [CrossRef]
- Vélez-Alavez, M.; Labrada-Martagón, V.; Méndez-Rodriguez, L.C.; Galván-Magaña, F.; Zenteno-Savín, T. Oxidative stress indicators and trace element concentrations in tissues of mako shark (Isurus oxyrinchus). Comp. Biochem. Physiol. 2013, 165, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Alves, L.M.; Nunes, M.; Marchand, P.; Le Bizec, B.; Mendes, S.; Correia, J.P.; Lemos, M.F.L.; Novais, S.C. Blue sharks (Prionace glauca) as bioindicators of pollution and health in the Atlantic Ocean: Contamination levels and biochemical stress responses. Sci. Total Environ. 2019, 563–564, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Marsili, L.; Coppola, D.; Giannetti, M.; Casini, S.; Fossi, M.C.; van Wyk, J.H.; Sperone, E.; Tripepi, S.; Micarelli, P.; Rizzuto, S. Skin biopsies as a sensitive non-lethal technique for the ecotoxicological studies of great white shark (Carcharodon carcharias) sampled in South Africa. Expert Opin. Environ. Biol. J. 2016, 4, 2. [Google Scholar]
- Fossi, M.C.; Baini, M.; Panti, C.; Galli, M.; Jiménez, B.; Muñoz-Arnanz, J.; Marsili, L.; Finoia, M.G.; Ramírez-Macías, D. Are whale sharks exposed to persistent organic pollutants and plastic pollution in the Gulf of California (Mexico)? First ecotoxicological investigation using skin biopsies. Comp. Biochem. Physiol. C 2017, 199, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Cullen, J.A.; Marshall, C.D.; Hala, D. Integration of multi-tissue PAH and PCB burdens with biomarker activity in three coastal shark species from the northwestern Gulf of Mexico. Sci. Total Environ. 2019, 650, 1158–1172. [Google Scholar] [CrossRef]
- Sureda, A.; Tejada, S.; Box, A.; Deudero, S. Polycyclic aromatic hydrocarbon levels and measures of oxidative stress in the Mediterranean endemic bivalve Pinna nobilis exposed to the Don Pedro oil spill. Mar. Pollut. Bull. 2013, 71, 69–73. [Google Scholar] [CrossRef]
- Chaousis, S.; Leusch, F.D.L.; van de Merwe, J.P. Charting a path towards non-destructive biomarkers in threatened wildlife: A systematic quantitative literature review. Environ. Pollut. 2018, 234, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, D.; Almunia, C.; Cogne, Y.; Pible, O.; Degli-Esposti, D.; Salvador, A.; Cristobal, S.; Sheehan, D.; Chaumot, A.; Geffard, O.; et al. Ecotoxicoproteomics: A decade of progress in our understanding of anthropogenic impact on the environment. J. Proteomics 2019, 198, 66–77. [Google Scholar] [CrossRef]
- Zhang, X.; Xia, P.; Wang, P.; Yang, J.; Baird, D.J. Omics Advances in Ecotoxicology. Environ. Sci. Technol. 2018, 52, 3842–3851. [Google Scholar] [CrossRef] [PubMed]
- Martins, C.; Dreij, K.; Costa, P.M. The State of the Art of Environmental Toxicogenomics: Challenges and Perspectives of “Omics” Approaches Directed to Toxicant Mixtures. Int. J. Environ. Res. Public Health 2019, 6, 4718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monsinjon, T.; Knigge, T. Proteomic applications in ecotoxicology. Proteomics 2007, 7, 2997–3009. [Google Scholar] [CrossRef]
- Tomanek, L. Environmental Proteomics: Changes in the proteome of marine organisms in response to environmental stress, pollutants, infection, symbiosis, and development. Annu. Rev. Mar. Sci. 2011, 3, 373–399. [Google Scholar] [CrossRef] [Green Version]
- Trapp, J.; Armengaud, J.; Salvador, A.; Chaumot, A.; Geffard, O. Next-generation Proteom: Toward customized biomarkers for environmental biomonitoring. Environ. Sci. Technol. 2014, 48, 13560–13572. [Google Scholar] [CrossRef]
- Armengaud, J.; Trapp, J.; Pible, O.; Geffard, O.; Chaumot, A.; Hartmann, E.M. Non-model organisms, a species endangered by proteogenomics. J. Proteom. 2014, 105, 5–18. [Google Scholar] [CrossRef]
| Protected Areas | Bioindicator Species | Bioindicator Class | Endpoint | Biomarkers Analyzed | Ref. |
|---|---|---|---|---|---|
| Europe | |||||
| Egadi Islands Marine Protected Area (Italy) | Coris julis, Patella caerulea, Paracentrotus lividus | Osteichthyes Gastropoda Echinoidea | Detoxification of organic pollutants | EthoxyresorufinO-deethylase, BaPMO, NADH ferry red, and NADH cyt c | [50] |
| Tremiti Islands Marine Protected Area (Italy) | Paracentrotus lividus | Echinoidea | Coelomocytes alterations | Coelomocytes subpopulations ratio, heat-shock protein 70 | [51] |
| National Park of La Maddalena Arcipelago (Italy) | Mytilus galloprovincialis | Bivalvia | Lysosomal alterations | Lysosomal membrane stability, lipofuscin content, neutral lipid contents, lysosomal structural changes | [52] |
| Capo Peloro Natural Reserve (Italy) | Atherina boyeri | Osteichthyes | Detoxification of organic pollutants, neurotoxicity, genotoxicity | Acetylcholinesterase, benzo(a)pyrene-monooxygenase, polycyclic aromatic hydrocarbons metabolites in bile, erythrocytic nuclear abnormalities assay | [53] |
| The Pelagos Sanctuary (International Sanctuary for the Protection of Mediterranean Marine Mammals) (Italy, France) | Meganyctiphanes norvegica | Malacostraca | Detoxification of organic pollutants, neurotoxicity, response to xenoestrogens | Cytochrome P450, BaPMO activity, NADPH cytochromec reductase, NADH-ferricyanide reductase, esterases, porphyrins, vitellogenin, zona radiata proteins, acetylcholinesterase | [54] |
| Stenella coeruleoalba | Mammalia | Detoxification of organic pollutants, oxidative stress | Cytochrome P4501A, cytochrome P4502B, catalase | [55] | |
| Balaenoptera physalus | Mammalia | Detoxification of organic chemical pollutants, oxidative stress | Cytochrome P4501A, cytochrome P4502B, lipoperoxidation | [56] | |
| Balaenoptera physalus, Physeter macrocephalus | Mammalia | Metal excretion | Metals in the fecal material | [57] | |
| North America | |||||
| Florida Keys National Marine Sanctuary (U.S.A.) | Montastraea annularis |
Anthozoa | Oxidative stress, stress protein multidrug resistance induction | Superoxide dismutase, glutathione peroxidase, glutathione-s-transferase, heat-shock proteins, metabolic condition, multixenobiotic resistance proteins | [58] |
| Veracruz Coral Reef System National Park (Mexico) | Haemulon Aurolineatum, Ocyurus chrysurus | Osteichthyes | Detoxification of organic chemical pollutants, response to xenoestrogens | Cytochrome P4501A, vitellogenin, glutathione-S-transferase, PAH metabolites in fish bile | [59] |
| Natural protected area of Laguna Madre in the Gulf of Mexico (Mexico) | Chione elevata | Bivalvia | Neurotoxic effects, oxidative stress, metabolic alterations | Acetylcholinesterase, butyrylcholinesterase, carboxylesterase, alkaline phosphatase, glutathione s-transferase, oxygen radical absorbance capacity | [60] |
| South America | |||||
| Morrocoy National Park (Venezuela) | Siderastrea sidereal | Anthozoa | Detoxification of organic chemical pollutants, oxidative stress | Cytochrome P450 I, cytochrome P450 II, NADPH reductase, glutathione S-transferase, catalase, superoxide dismutase | [61] |
| Parque Nacional Archipielago Los Roques (Venezuela) | Siderastrea sidereal | Anthozoa | Detoxification of organic chemical pollutants, oxidative stress | Cytochrome P450 I, cytochrome P450 II, NADPH reductase, glutathione S-transferase, catalase, superoxide dismutase | [61] |
| Fernando de Noronha Archipelago protected area (Brazil) | Amphistegina lessonii | Foraminifera | Oxidative stress, metal detoxification | Antioxidant capacity against peroxyl radicals, lipid peroxidation, protein carbonylation, metallothionein-like proteins | [62] |
| Paranaguá Bay protected areas (Brazil) | Atherinella brasiliensis | Osteichthyes | Neurotoxicity, detoxification of organic chemical pollutants, oxidative stress | Cholinesterase, ethoxyresorufinO-deethylase, glutathione S-transferase, catalase | [63] |
| Cananéia–Iguape–Peruíbe Environmental Protected Area (Brazil) | Cathorops spixii | Osteichthyes | Detoxification of organic pollutants, oxidative stress, genotoxicity, metal detoxification | Glutathione S-transferase, glutathione peroxidase, GSH levels, lipid peroxidation, DNA strand breaks, metallothonein | [64] |
| Cathorops spixii | Osteichthyes | Genotoxicity | Comet assay, micronucleus test (MN), and nuclear abnormalities test (NA) in peripheral blood | [65] | |
| Natural Protected Area San Antonio Bay (Argentina) | Neohelice granulata | Malacostraca | Detoxification of organic pollutants, oxidative stress, metal detoxification | Catalase, lipid radical content, lipid peroxidation, α-tocopherol, catalase, glutathione-S-transferases, metallothioneins | [66] |
| Cananéia–Iguape–Peruíbe Protected Area (Brazil) | Callinectes danae | Malacostraca | Genotoxicity, detoxification of organic pollutants, oxidative stress, metal detoxification, neurotoxicity | Glutathione S-transferase, glutathione peroxidase, intracellular glutathione, acetylcholinesterase, lipid peroxidation, metallothionein, DNA strand breaks | [67] |
| Estuarine Lagoon Complex of Iguape–Cananéia (Brazil) | Gobioides broussonnetii | Osteichthyes | Oxidative stress, genotoxicity, metal detoxification, histopathological alterations | Superoxide dismutase, catalase, glutathione peroxidase activity, glutathione S-transferase, glutathione, metallothionein, lipoperoxidation, micronuclei, histological alterations | [68] |
| Australia | |||||
| Great Barrier Reef (Australia) | Plectropomus leopardus | Osteichthyes | Detoxification of organic chemical pollutants, neurotoxicity | EROD, cholinesterase | [69] |
| Acropora millepora | Anthozoa | Oxidative stress | Genetic loci involved in environmental stress tolerance and antioxidant capacity | [70] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lionetto, M.G.; Caricato, R.; Giordano, M.E. Pollution Biomarkers in the Framework of Marine Biodiversity Conservation: State of Art and Perspectives. Water 2021, 13, 1847. https://doi.org/10.3390/w13131847
Lionetto MG, Caricato R, Giordano ME. Pollution Biomarkers in the Framework of Marine Biodiversity Conservation: State of Art and Perspectives. Water. 2021; 13(13):1847. https://doi.org/10.3390/w13131847
Chicago/Turabian StyleLionetto, Maria Giulia, Roberto Caricato, and Maria Elena Giordano. 2021. "Pollution Biomarkers in the Framework of Marine Biodiversity Conservation: State of Art and Perspectives" Water 13, no. 13: 1847. https://doi.org/10.3390/w13131847
APA StyleLionetto, M. G., Caricato, R., & Giordano, M. E. (2021). Pollution Biomarkers in the Framework of Marine Biodiversity Conservation: State of Art and Perspectives. Water, 13(13), 1847. https://doi.org/10.3390/w13131847







