Adsorption Studies on Magnetic Nanoparticles Functionalized with Silver to Remove Nitrates from Waters
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Fe3O4@AgNPs
2.3. Nitrates Adsorption Procedure
3. Results and Discussion
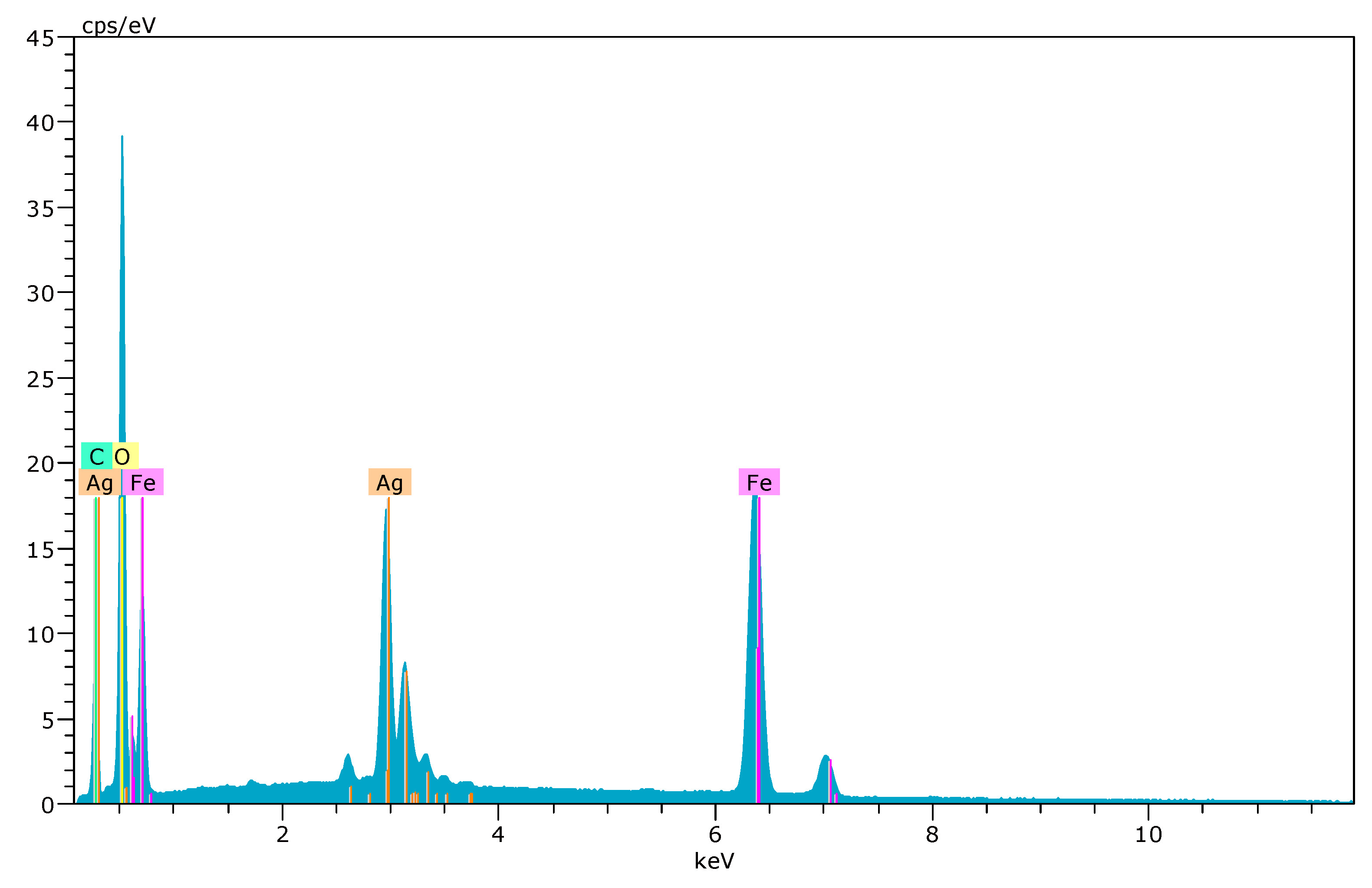
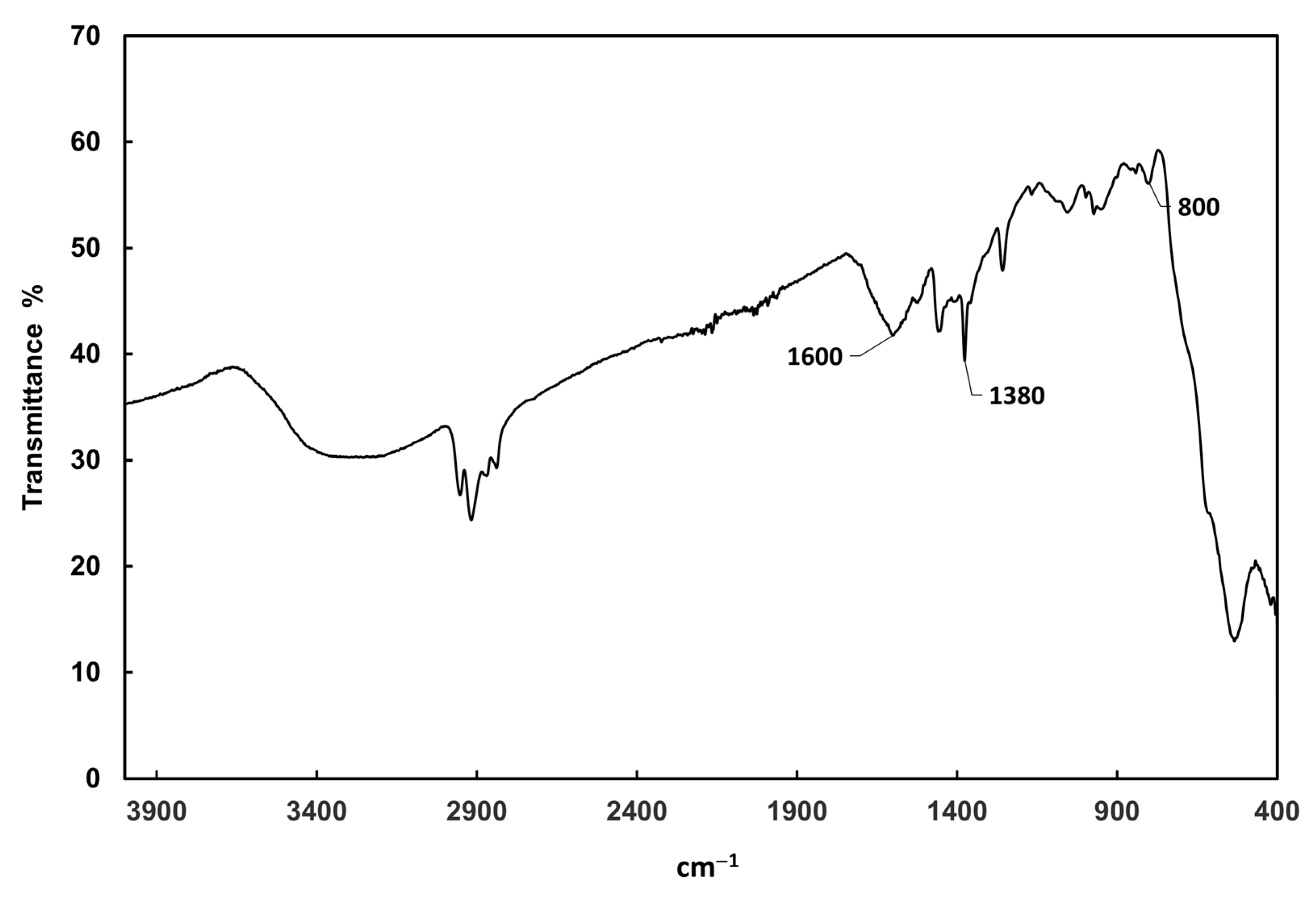
3.1. Characterization of Fe3O4@AgNPs
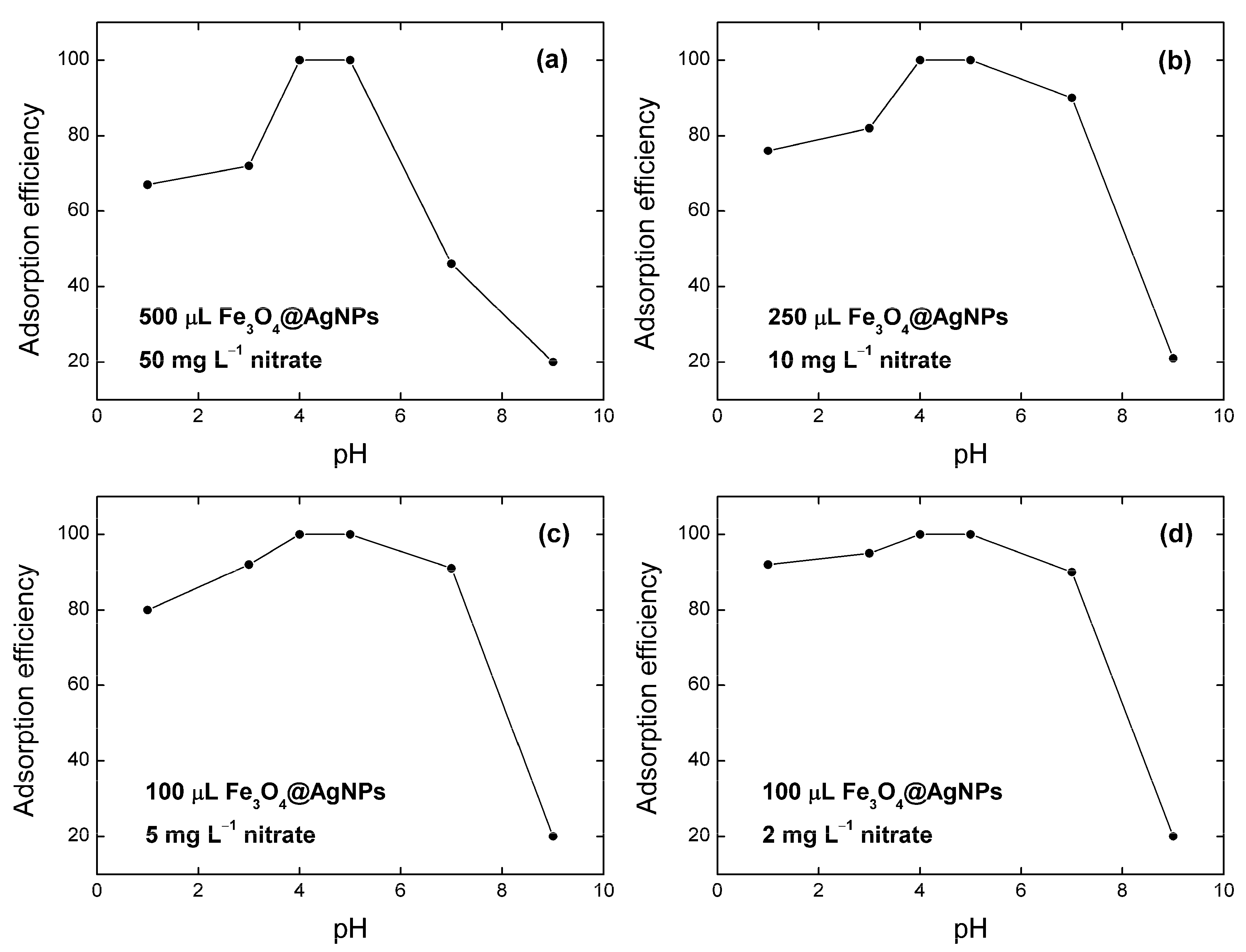
3.2. Effect of pH on Nitrate Removal by Fe3O4@AgNPs
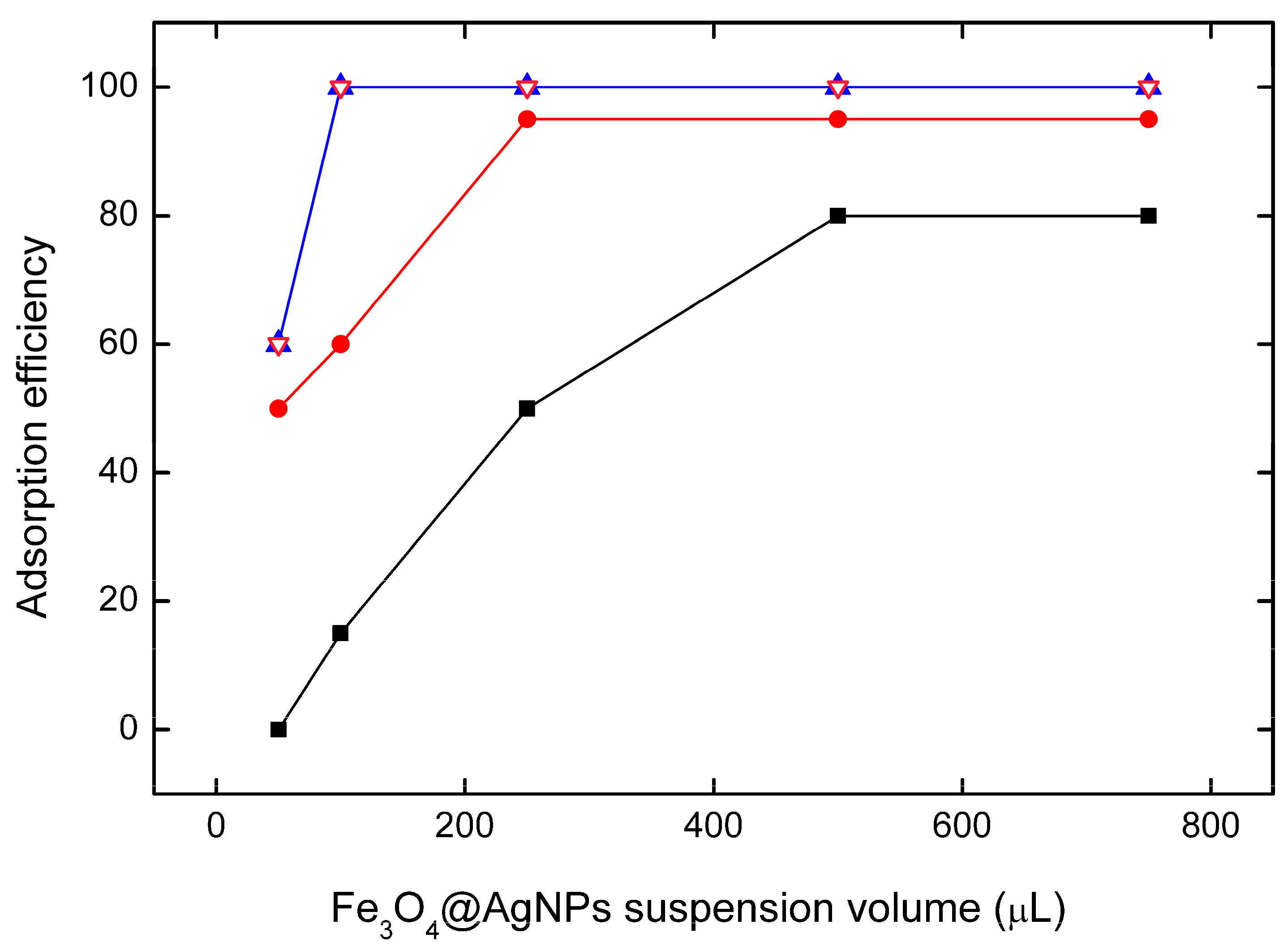
3.3. Effect of Adsorbent Dose and Performance Metrics
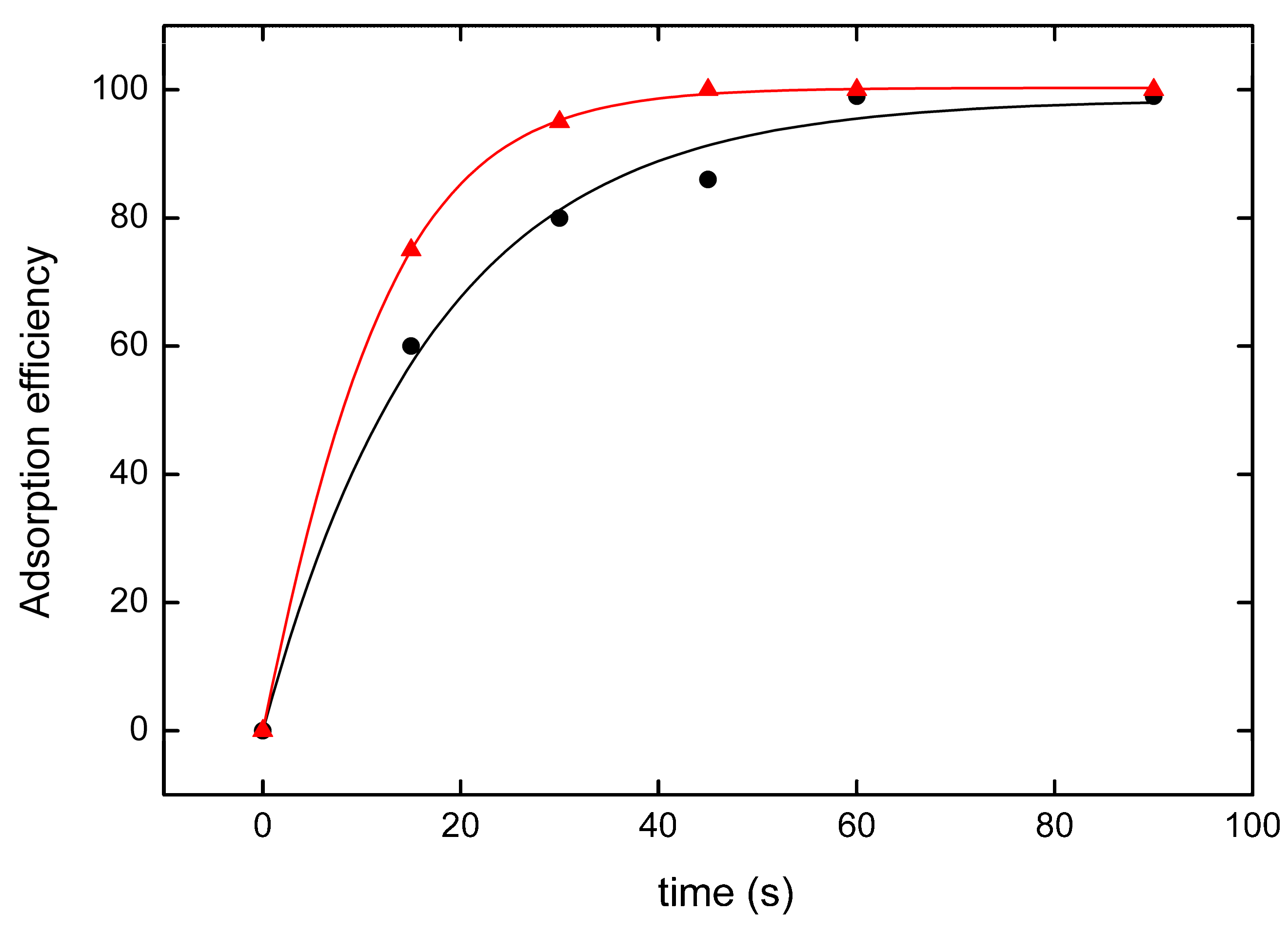
3.4. Contact Time Effect
3.5. Effect of Coexisting Anions on Nitrate Removal
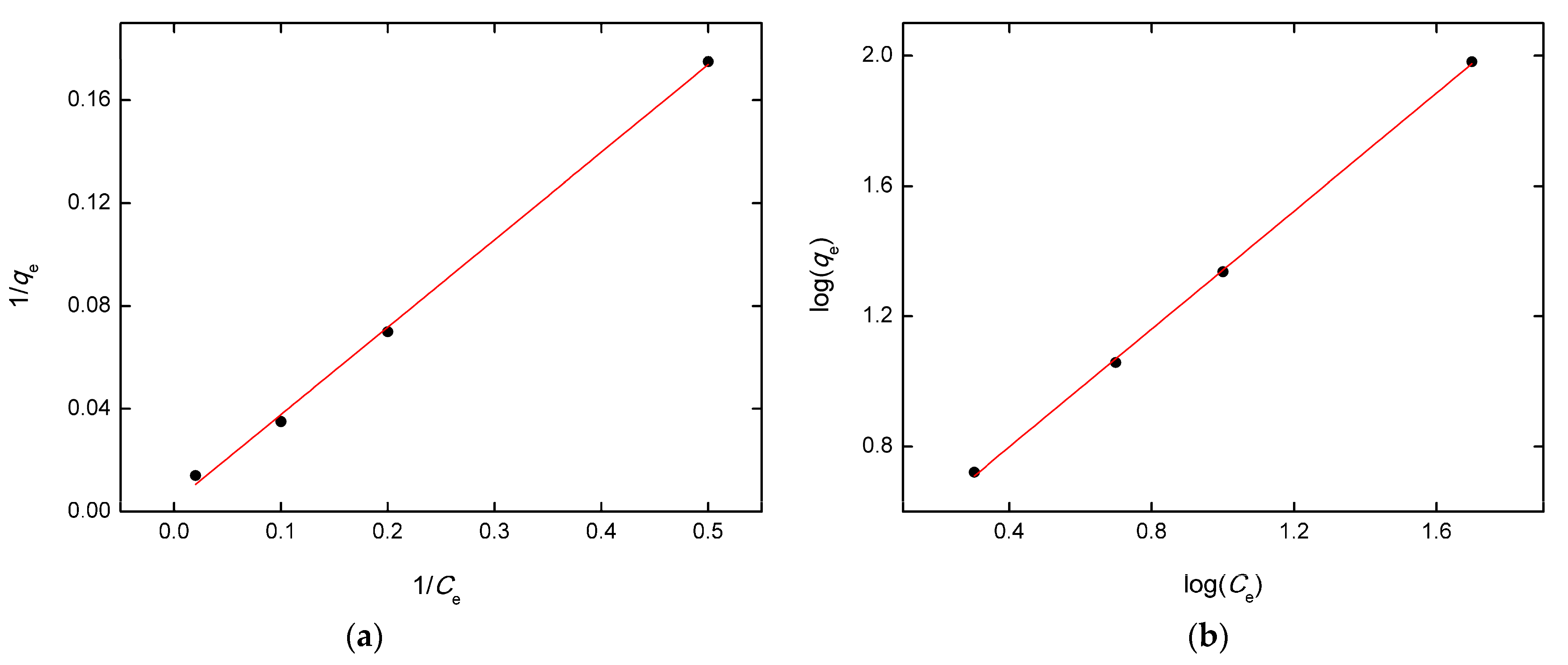
3.6. Adsorption Isotherms
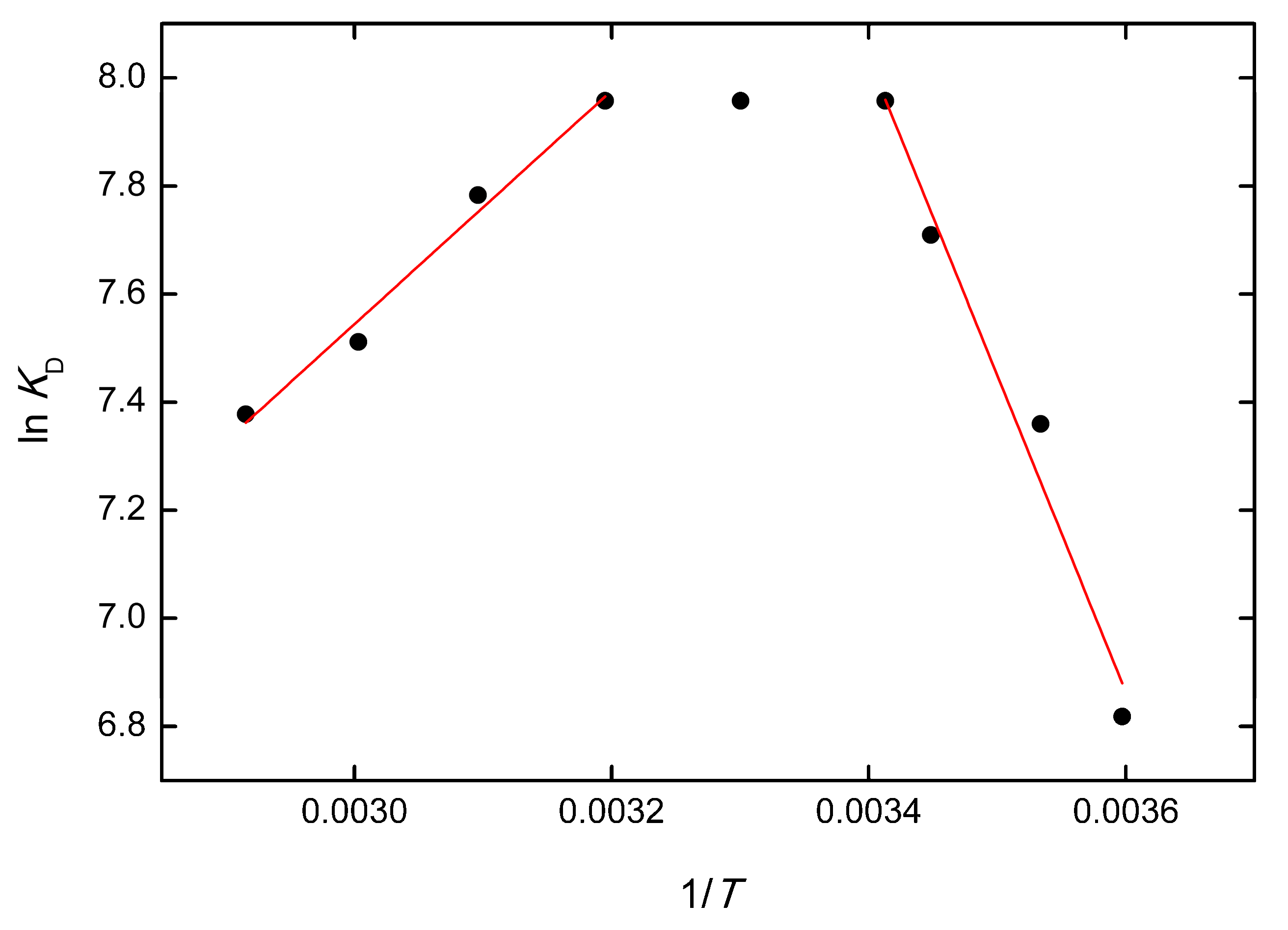
3.7. Thermodynamic Analysis
3.8. Desorption and Recycling Nanoparticles
3.9. Adsorption of Nitrates onto Fe3O4@AgNPs in Real Seawater Samples
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Żołnierczyk, M.; Barbusiński, K. Physicochemical methods of nitrates removal from waste water. Archit. Civ. Eng. Environ. 2019, 12, 153–159. [Google Scholar] [CrossRef] [Green Version]
- Payen, S.; Falconer, S.; Carlson, B.; Yang, W.; Ledgard, S. Eutrophication and climate change impacts of a case study of New Zealand beef to the European market. Sci. Total Environ. 2020, 710. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Chen, Y.; Xia, W.; Qu, X.; Yuan, H.; Xie, S.; Lin, L.S. Eutrophication and heavy metal pollution patterns in the water suppling lakes of China’s south-to-north water diversion project. Sci. Total Environ. 2020, 711, 134543. [Google Scholar] [CrossRef]
- Zhang, Y.; Liang, J.; Zeng, G.; Tang, W.; Lu, Y.; Luo, Y.; Xing, W.; Tang, N.; Ye, S.; Li, X.; et al. How climate change and eutrophication interact with microplastic pollution and sediment resuspension in shallow lakes: A review. Sci. Total Environ. 2020, 705, 135979. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, L.; Zhou, Y.; Mao, X. Eutrophication control strategies for highly anthropogenic influenced coastal waters. Sci. Total Environ. 2020, 705, 135760. [Google Scholar] [CrossRef]
- Constantinou, C.L.; Costa, C.N.; Efstathiou, A.M. Catalytic removal of nitrates from waters. Catal. Today 2010, 151, 190–194. [Google Scholar] [CrossRef]
- Theologides, C.P.; Savva, P.G.; Costa, C.N. Catalytic removal of nitrates from waters in a continuous flow process: The remarkable effect of liquid flow rate and gas feed composition. Appl. Catal. B Environ. 2011, 102, 54–61. [Google Scholar] [CrossRef]
- Ruiz-Bevia, F.; Fernandez-Torres, M.J. Comments on “Use of Rh (III)-Heteropolymolybdate as Potential Catalysts for the Removal of Nitrates in Human Drinking Water: Synthesis, Characterisation and Catalytic Performance” by María A. Jaworski et al. (Water Air Soil Pollut (2018) 229: 309). Water Air Soil Pollut. 2019, 230, 1–3. [Google Scholar] [CrossRef]
- Ruiz-Beviá, F.; Fernández-Torres, M.J. Effective catalytic removal of nitrates from drinking water: An unresolved problem? J. Clean. Prod. 2019, 217, 398–408. [Google Scholar] [CrossRef]
- Lacasa, E.; Cañizares, P.; Sáez, C.; Fernández, F.J.; Rodrigo, M.A. Removal of nitrates from groundwater by electrocoagulation. Chem. Eng. J. 2011, 171, 1012–1017. [Google Scholar] [CrossRef]
- Abdel-Aziz, M.H.; El-Ashtoukhy, E.S.Z.; Zoromba, M.S.; Bassyouni, M.; Sedahmed, G.H. Removal of nitrates from water by electrocoagulation using a cell with horizontally oriented Al serpentine tube anode. J. Ind. Eng. Chem. 2020, 82, 105–112. [Google Scholar] [CrossRef]
- Acharya, S.; Sharma, S.K.; Chauhan, G.; Shree, D. Statistical Optimization of Electrocoagulation Process for Removal of Nitrates Using Response Surface Methodology. Indian Chem. Eng. 2018, 60, 269–284. [Google Scholar] [CrossRef]
- Anderson, J.A. Metal-promoted titania photocatalysis for destruction of nitrates and organics from aqueous environments. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2018, 376. [Google Scholar] [CrossRef] [Green Version]
- Baly, E.C.C.; Heilbron, I.M.; Hudson, D.P. CXXX.—Photocatalysis. Part II. The photosynthesis of nitrogen compounds from nitrates and carbon dioxide. J. Chem. Soc. Trans. 1922, 121, 1078–1088. [Google Scholar] [CrossRef]
- Richard, Y.; Thebault, P. Biological removal of nitrates—Report on 7 years of operation and progress. Water Supply 1992, 10, 151–160. [Google Scholar]
- Janda, V.; Rudovsky, J. Removal of nitrate from drinking water by biological denitrificationp. ChemInform 1993, 87, 179–186. [Google Scholar]
- Wąsik, E.; Bohdziewicz, J.; Blaszczyk, M. Removal of nitrates from ground water by a hybrid process of biological denitrification and microfiltration membrane. Process Biochem. 2001, 37, 57–64. [Google Scholar] [CrossRef]
- Marecik, R.; Dembczyński, R.; Juzwa, W.; Chrzanowski, Ł.; Cyplik, P. Removal of nitrates from processing wastewater by cryoconcentration combined with biological denitrification. Desalin. Water Treat. 2015, 54, 1903–1911. [Google Scholar] [CrossRef]
- Ketcha, M.J.; Manga, N.H.; Daouda, K.; Tchoua, N.P. Kinetic and equilibrium studies of the adsorption of nitrates ions in aqueous solutions by activated carbons and zeolite. Res. J. Chem. Environ. 2007, 11, 47–51. [Google Scholar]
- Chabani, M.; Amrane, A.; Bensmaili, A. Kinetics of nitrates adsorption on Amberlite IRA 400 resin. Desalination 2007, 206, 560–567. [Google Scholar] [CrossRef]
- Chabani, M.; Amrane, A.; Bensmaili, A. Kinetic modelling of the adsorption of nitrates by ion exchange resin. Chem. Eng. J. 2006, 125, 111–117. [Google Scholar] [CrossRef]
- Milmile, S.N.; Pande, J.V.; Karmakar, S.; Bansiwal, A.; Chakrabarti, T.; Biniwale, R.B. Equilibrium isotherm and kinetic modeling of the adsorption of nitrates by anion exchange Indion NSSR resin. Desalination 2011, 276, 38–44. [Google Scholar] [CrossRef]
- Adamu, H.; Shand, M.; Taylor, R.S.F.; Manyar, H.G.; Anderson, J.A. Use of carbon-based composites to enhance performance of TiO2 for the simultaneous removal of nitrates and organics from aqueous environments. Environ. Sci. Pollut. Res. 2018, 25, 32001–32014. [Google Scholar] [CrossRef] [Green Version]
- Ferrando, L.; Matamoros, V. Attenuation of nitrates, antibiotics and pesticides from groundwater using immobilised microalgae-based systems. Sci. Total Environ. 2020, 703. [Google Scholar] [CrossRef]
- Valiyeva, G.G.; Bavasso, I.; Di Palma, L.; Hajiyeva, S.R.; Ramazanov, M.A.; Hajiyeva, F.V. Synthesis of fe/ni bimetallic nanoparticles and application to the catalytic removal of nitrates from water. Nanomaterials 2019, 9, 1130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dharmagunawardhane, D.S.; De Silva, N.L.; Gunatilake, U.B.; Yan, C.F.; Bandara, J. Removal of groundwater nitrates by heterogeneous supramolecular complexes-like photocatalytic system based on in-situ generated and highly active Ti3+/Ti2+ states in the reduced TiO2. Mol. Catal. 2019, 470, 89–96. [Google Scholar] [CrossRef]
- Galan, C.R.; Silva, M.F.; Mantovani, D.; Bergamasco, R.; Vieira, M.F. Green synthesis of copper oxide nanoparticles impregnated on activated carbon using Moringa oleifera leaves extract for the removal of nitrates from water. Can. J. Chem. Eng. 2018, 96, 2378–2386. [Google Scholar] [CrossRef]
- García-Fernández, M.J.; Sancho-Querol, S.; Pastor-Blas, M.M.; Sepúlveda-Escribano, A. Surfactant-assisted synthesis of conducting polymers. Application to the removal of nitrates from water. J. Colloid Interface Sci. 2017, 494, 98–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chianese, A.; Di Palma, L.; Petrucci, E.; Stoller, M.; Muradova, G.G.; Gadjieva, S.R.; Di Palma, L.; Vilardi, G. Nitrates Removal by Bimetallic Nanoparticles in Water. Chem. Eng. Trans. 2016, 47, 205–210. [Google Scholar]
- Vicente-Martínez, Y.; Caravaca, M.; Soto-Meca, A.; Solana-González, R. Magnetic core-modified silver nanoparticles for ibuprofen removal: An emerging pollutant in waters. Sci. Rep. 2020, 10, 18288. [Google Scholar] [CrossRef]
- López-García, I.; Vicente-Martínez, Y.; Hernández-Córdoba, M. Determination of ultratraces of mercury species using separation with magnetic core-modified silver nanoparticles and electrothermal atomic absorption spectrometry. J. Anal. At. Spectrom. 2015, 30, 1980–1987. [Google Scholar] [CrossRef]
- Vicente-Martínez, Y.; Caravaca, M.; Soto-Meca, A. Total removal of Hg (II) from wastewater using magnetic nanoparticles coated with nanometric Ag and functionalized with sodium 2-mercaptoethane sulfonate. Environ. Chem. Lett. 2020, 18, 975–981. [Google Scholar] [CrossRef]
- Vicente-Martínez, Y.; Caravaca, M.; Soto-Meca, A.; De Francisco-Ortiz, O.; Gimeno, F. Graphene oxide and graphene oxide functionalized with silver nanoparticles as adsorbents of phosphates in waters. A comparative study. Sci. Total Environ. 2020, 709, 136111. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Wang, Q.; Chen, W.; Pang, Y. One-step synthesis of water-dispersible cysteine functionalized magnetic Fe3O4 nanoparticles for mercury(II) removal from aqueous solutions. Appl. Surf. Sci. 2014, 317, 1028–1034. [Google Scholar] [CrossRef]
- Cohen, H.; Gedanken, A.; Zhong, Z. One-step synthesis and characterization of ultrastable and amorphous Fe3O4 colloids capped with cysteine molecules. J. Phys. Chem. C 2008, 112, 15429–15438. [Google Scholar] [CrossRef]
- Zhang, Y.R.; Shen, S.L.; Wang, S.Q.; Huang, J.; Su, P.; Wang, Q.R.; Zhao, B.X. A dual function magnetic nanomaterial modified with lysine for removal of organic dyes from water solution. Chem. Eng. J. 2014, 239, 250–256. [Google Scholar] [CrossRef]
- Trivedi, M.K.; Dahryn Trivedi, A.B. Spectroscopic Characterization of Disodium Hydrogen Orthophosphate and Sodium Nitrate after Biofield Treatment. Chromatogr. Sep. Tech. 2015, 6. [Google Scholar] [CrossRef] [Green Version]
- Karthikeyan, P.; Banu, H.A.T.; Meenakshi, S. Synthesis and characterization of metal loaded chitosan-alginate biopolymeric hybrid beads for the efficient removal of phosphate and nitrate ions from aqueous solution. Int. J. Biol. Macromol. 2019, 130, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Battas, A.; El Gaidoumi, A.; Ksakas, A.; Kherbeche, A. Adsorption study for the removal of nitrate from water using local clay. Sci. World J. 2019, 2019. [Google Scholar] [CrossRef] [Green Version]
- Azari, A.; Kalantary, R.R.; Ghanizadeh, G.; Kakavandi, B.; Farzadkia, M.; Ahmadi, E. Iron-silver oxide nanoadsorbent synthesized by co-precipitation process for fluoride removal from aqueous solution and its adsorption mechanism. RSC Adv. 2015, 5, 87377–87391. [Google Scholar] [CrossRef]
- López-Ramón, V.; Moreno-Castilla, C.; Rivera-Utrilla, J.; Radovic, L.R. Ionic strength effects in aqueous phase adsorption of metal ions on activated carbons. Carbon 2003, 41, 2020–2022. [Google Scholar] [CrossRef]
- Moreno-Castilla, C. Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon 2004, 42, 83–94. [Google Scholar] [CrossRef] [Green Version]
- Adar, R.M.; Safran, S.A.; Diamant, H.; Andelman, D. Screening length for finite-size ions in concentrated electrolytes. Phys. Rev. E 2019, 100, 042615. [Google Scholar] [CrossRef] [Green Version]
- Barton, B.I.; Lique, C.; Lenn, Y.D. Water Mass Properties Derived From Satellite Observations in the Barents Sea. J. Geophys. Res. Ocean. 2020, 125, e2019JC015449. [Google Scholar] [CrossRef]
- Rodriguez, J.A. The adsorption of nitrogen dioxide, nitrate and sulfate on Ag(110): A quantum-chemical study. Surf. Sci. 1990, 230, 335–349. [Google Scholar] [CrossRef]
- Al-Wabel, M.; Elfaki, J.; Usman, A.; Hussain, Q.; Ok, Y.S. Performance of dry water- and porous carbon-based sorbents for carbon dioxide capture. Environ. Res. 2019, 174, 69–79. [Google Scholar] [CrossRef]
- Islam, A.; Khan, M.R.; Mozumder, S.I. Adsorption equilibrium and adsorption kinetics: A unified approach. Chem. Eng. Technol. 2004, 27, 1095–1098. [Google Scholar] [CrossRef]
- Jun, B.M.; Heo, J.; Park, C.M.; Yoon, Y. Comprehensive evaluation of the removal mechanism of carbamazepine and ibuprofen by metal organic framework. Chemosphere 2019, 235, 527–537. [Google Scholar] [CrossRef]
- Su, Y.; Yang, W.; Sun, W.; Li, Q.; Shang, J.K. Synthesis of mesoporous cerium-zirconium binary oxide nanoadsorbents by a solvothermal process and their effective adsorption of phosphate from water. Chem. Eng. J. 2015, 268, 270–279. [Google Scholar] [CrossRef]
- Das, J.; Patra, B.S.; Baliarsingh, N.; Parida, K.M. Adsorption of phosphate by layered double hydroxides in aqueous solutions. Appl. Clay Sci. 2006, 32, 252–260. [Google Scholar] [CrossRef]
- López-García, I.; Vicente-Martínez, Y.; Hernández-Córdoba, M. Determination of very low amounts of free copper and nickel ions in beverages and water samples using cloud point extraction assisted by silver nanoparticles. Anal. Methods 2015, 7, 3786–3792. [Google Scholar] [CrossRef]
- Jeppu, G.P.; Clement, T.P. A modified Langmuir-Freundlich isotherm model for simulating pH-dependent adsorption effects. J. Contam. Hydrol. 2012, 129–130, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and Interpretation of Adsorption Isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.J.; Hosseini-Bandegharaei, A.; Chao, H.P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Chen, Z.; Li, Y.; Li, C.; Ahmad, N.M.; Cheema, W.A.; Zhu, S. Removal of Hg(ii) in aqueous solutions through physical and chemical adsorption principles. RSC Adv. 2019, 9, 20941–20953. [Google Scholar] [CrossRef] [Green Version]


| Optimum Conditions (Temperature (°C), pH) | Initial Nitrate Concentration (mg L−1) | Fe3O4@AgNPs Mass (mg) | Maximum Removal Efficiency (%) | Adsorption Capacity (mg g−1) | Partition Coefficient (mg g−1 µM−1) |
|---|---|---|---|---|---|
| 25, 5 | 2 | 9.5 | 100 | 2.11 | - |
| 25, 5 | 2 | 7 | 100 | 2.86 | - |
| 25, 5 | 2 | 3.5 | 100 | 5.71 | - |
| 25, 5 | 2 | 1.4 | 100 | 14.29 | - |
| 25, 5 | 2 | 0.7 | 60 | 28.57 | 2214.29 |
| 25, 5 | 5 | 9.5 | 100 | 5.26 | - |
| 25, 5 | 5 | 7 | 100 | 4.17 | - |
| 25, 5 | 5 | 3.5 | 100 | 14.29 | - |
| 25, 5 | 5 | 1.4 | 100 | 35.71 | - |
| 25, 5 | 5 | 0.7 | 60 | 71.43 | 163.16 |
| 25, 5 | 10 | 9.5 | 100 | 10.53 | - |
| 25, 5 | 10 | 7 | 100 | 14.29 | - |
| 25, 5 | 10 | 3.5 | 100 | 28.57 | - |
| 25, 5 | 10 | 1.4 | 95 | 71.43 | 8857.14 |
| 25, 5 | 10 | 0.7 | 95 | 142.86 | 17,714.29 |
| 25, 5 | 50 | 9.5 | 99 | 26.32 | 3263.16 |
| 25, 5 | 50 | 7 | 99 | 35.71 | 4428.57 |
| 25, 5 | 50 | 3.5 | 86 | 71.43 | 632.65 |
| 25, 5 | 50 | 1.4 | 80 | 157.57 | 1107.14 |
| 25, 5 | 50 | 0.7 | 80 | 357.14 | 2214.29 |
| Sample | Absorbance before Adsorption | Absorbance after Adsorption | Removal Efficiency (%) |
|---|---|---|---|
| 1 | 0.37 | 0.02 | 94.6 |
| 2 | 0.22 | 0.01 | 95.4 |
| 3 | 0.20 | 0.00 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vicente-Martínez, Y.; Caravaca, M.; Soto-Meca, A.; Martín-Pereira, M.Á.; García-Onsurbe, M.d.C. Adsorption Studies on Magnetic Nanoparticles Functionalized with Silver to Remove Nitrates from Waters. Water 2021, 13, 1757. https://doi.org/10.3390/w13131757
Vicente-Martínez Y, Caravaca M, Soto-Meca A, Martín-Pereira MÁ, García-Onsurbe MdC. Adsorption Studies on Magnetic Nanoparticles Functionalized with Silver to Remove Nitrates from Waters. Water. 2021; 13(13):1757. https://doi.org/10.3390/w13131757
Chicago/Turabian StyleVicente-Martínez, Yesica, Manuel Caravaca, Antonio Soto-Meca, Miguel Ángel Martín-Pereira, and María del Carmen García-Onsurbe. 2021. "Adsorption Studies on Magnetic Nanoparticles Functionalized with Silver to Remove Nitrates from Waters" Water 13, no. 13: 1757. https://doi.org/10.3390/w13131757
APA StyleVicente-Martínez, Y., Caravaca, M., Soto-Meca, A., Martín-Pereira, M. Á., & García-Onsurbe, M. d. C. (2021). Adsorption Studies on Magnetic Nanoparticles Functionalized with Silver to Remove Nitrates from Waters. Water, 13(13), 1757. https://doi.org/10.3390/w13131757






