A Weight of Evidence (WOE) Approach to Assess Environmental Hazard of Marine Sediments from Adriatic Offshore Platform Area
Abstract
1. Introduction
2. Materials and Methods
2.1. Strategy of Sampling and Experimental Design
2.2. Chemical Characterization of Sediments
2.3. Hediste Diversicolor Exposure
2.3.1. Bioaccumulation Analyses
2.3.2. Biomarkers Analyses
2.4. Bioassays
2.5. WOE Elaboration
3. Results
3.1. Chemical Characterization of Sediments
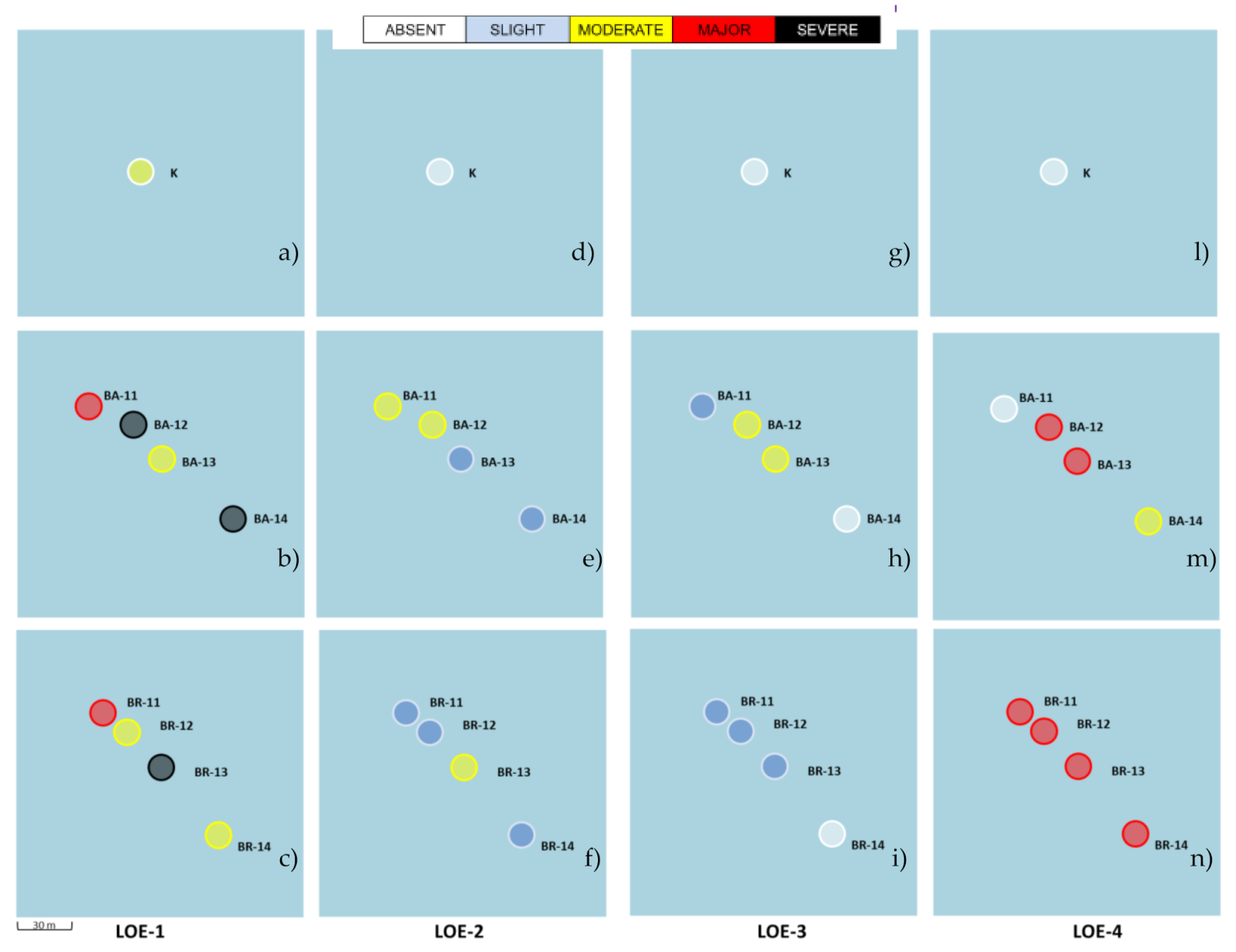
3.2. WOE Approach: Sediqualsoft Elaboration
3.2.1. LOE1—Level of Sediment Chemical Hazard
3.2.2. LOE2—Level of Hazard for Bioaccumulation
3.2.3. LOE3—Level of Hazard for Biomarkers
3.2.4. LOE4—Level of Hazard for Bioassays
3.2.5. WOE Index-Integration of LOEs
4. Discussion and Conclusions
- inclusion of concentrations measured in the control area among the chemical reference values of the Sediqualsoft model;
- routine application of this model to process multidisciplinary data related to environmental monitoring plans of offshore platform impacts, including those concerning the Produced Water discharge into the sea;
- promotion of further LOEs to assess the integrated risk associated with investigated impacts.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Italian Legislative Decree n. 172/2015 “Attuazione Della Direttiva 2013/39/UE, che Modifica le Direttive 2000/60/CE per Quanto Riguarda le Sostanze Prioritarie Nel Settore Della Politica Delle Acque”. Available online: https://pdc.minambiente.it/sites/default/files/allegati/dlgs_172_del_13_ottobre_2015.pdf (accessed on 28 February 2021).
- Long, E.R.; Macdonald, D.D.; Smith, S.L.; Calder, F.D. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ. Manag. 1995, 19, 81–97. [Google Scholar] [CrossRef]
- Macdonald, D.D.; Carr, R.S.; Calder, F.D.; Long, E.R.; Ingersoll, C.G. Development and evaluation of sediment quality guidelines for Florida coastal waters. Ecotoxicology 1996, 5, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Vethaak, A.D.; Davies, I.M.; Thain, J.E.; Gubbins, M.J.; Martínez-Gómez, C.; Robinson, C.D.; Moffat, C.F.; Burgeot, T.; Maes, T.; Wosniok, W.; et al. Integrated indicator framework and methodology for monitoring and assessment of hazardous substances and their effects in the marine environment. Mar. Environ. Res. 2017, 124, 11–20. [Google Scholar] [CrossRef]
- Chapman, P.M. Ecological Risk and Weight of Evidence Assessments. In Marine Ecotoxicology; Blasco, J., Chapman, P.M., Campana, O., Hampel, M., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 257–271. [Google Scholar]
- Mouillot, D.; Villéger, S.; Scherer-Lorenzen, M.; Mason, N.W.H. Functional Structure of Biological Communities Predicts Ecosystem Multifunctionality. PLoS ONE 2011, 6, e17476. [Google Scholar] [CrossRef]
- Environment Canada. Federal Contaminated Sites Action Plan (FCSAP): Ecological Risk Assessment Guidance. Prepared by Azimuth Consulting Group, Vancouver, BC, Canada. 2012. Available online: http://publications.gc.ca/collections/collection_2021/eccc/En14-92-6-2020-eng.pdf (accessed on 15 January 2021).
- Chapman, P.M. Determining when contamination is pollution—Weight of evidence determinations for sediments and effluents. Environ. Int. 2007, 33, 492–501. [Google Scholar] [CrossRef]
- Regoli, F.; d’Errico, G.; Nardi, A.; Mezzelani, M.; Fattorini, D.; Benedetti, M.; Di Carlo, M.; Pellegrini, D.; Gorbi, S. Application of a Weight of Evidence Approach for Monitoring Complex Environmental Scenarios: The Case-Study of Off-Shore Platforms. Front. Mar. Sci. 2019, 6, 377. [Google Scholar] [CrossRef]
- Depledge, M.H. The Rational Basis for the Use of Biomarkers as Ecotoxicological Tools. In Nondestructive Biomarkers in Vertebrates; Leonzio, C., Ed.; CRC: London, UK, 1994; pp. 271–295. [Google Scholar]
- Regoli, F.; Frenzilli, G.; Bocchetti, R.; Annarumma, F.; Scarcelli, V.; Fattorini, D.; Nigro, M. Time-course variations of oxyradical metabolism. DNA integrity and lysosomal stability in mussels. Mytilus galloprovincialis during a fiel translocation experiment. Aquat. Toxicol. 2004, 68, 167–178. [Google Scholar] [CrossRef]
- Morroni, L.; d’Errico, G.; Sacchi, M.; Molisso, F.; Armiento, G.; Chiavarini, S.; Rimauro, J.; Guida, M.; Siciliano, A.; Ceparano, M.; et al. Integrated characterization and risk management of marine sediments: The case study of the industrialized Bagnoli area (Naples, Italy). Mar. Environ. Res. 2020, 160, 104984. [Google Scholar] [CrossRef]
- Piva, F.; Ciaprini, F.; Onorati, F.; Benedetti, M.; Fattorini, D.; Ausili, A.; Regoli, F. Assessing sediment hazard through a weight of evi-dence approach with bioindicator organisms: A practical model to elaborate data from sediment chemistry bioavailability, biomarkers and ecotoxico-logical bioassays. Chemosphere 2011, 83, 475–485. [Google Scholar] [CrossRef]
- Khosrovyan, A.; Rodriguez-Romero, A.; Ramos, M.A.; Del Valls, T.Á.; Riba, I. Comparative analysis of two weight-of-evidence methodologies for integrated sediment quality assessment. Chemosphere 2015, 120, 138–144. [Google Scholar] [CrossRef]
- Sany, S.B.T.; Hashim, R.; Rezayi, M.; Salleh, A.; Safari, O. A review of strategies to monitor water and sediment quality for a sustainability assessment of marine environment. Environ. Sci. Pollut. Res. 2014, 21, 813–833. [Google Scholar] [CrossRef]
- Benedetti, M.; Ciaprini, F.; Piva, F.; Onorati, F.; Fattorini, D.; Notti, A.; Ausili, A.; Regoli, F. A multidisciplinary weight of evidence approach for classifying polluted sediments: Integrating sediment chemistry, bioavailability, biomarkers responses and bioassays. Environ. Int. 2012, 38, 17–28. [Google Scholar] [CrossRef]
- Beyer, J.; Goksøyr, A.; Hjermann, D.Ø.; Klungsøyr, J. Environmental effects of offshore produced water discharges: A review focused on the Norwegian continental shelf. Mar. Environ. Res. 2020, 162, 105155. [Google Scholar] [CrossRef]
- Barrick, A.; Marion, J.-M.; Perrein-Ettajani, H.; Chatel, A.; Mouneyrac, C. Baseline levels of biochemical biomarkers in the endobenthic ragworm Hediste diversicolor as useful tools in biological monitoring of estuaries under anthropogenic pressure. Mar. Pollut. Bull. 2018, 129, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Durou, C.; Poirier, L.; Amiard, J.C.; Budzinski, H.; Gnassia-Barelli, M.; Lemenach, K.; Peluhet, L.; Mouneyrac, C.; Roméo, M.; Amiard-Triquet, C. Biomonitoring in a clean and multi-contaminated estuary based on biomarkers and chemical analyses in the endobenthic worm Nereis di-versicolor. Environ. Poll. 2007, 148, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Moreira, S.M.; Lima, I.; Ribeiro, R.; Guilhermino, L. Effects of estuarine sediment contamination on feeding and on key physiological functions of the polychaete Hediste diversicolor: Laboratory and in situ assays. Aquat. Toxicol. 2006, 78, 186–201. [Google Scholar] [CrossRef] [PubMed]
- Pérez, E.; Blasco, J.; Sole, M. Biomarker responses to pollution in two invertebrate species: Scrobicularia plana and Nereis diversicolor from the Cádiz bay (SW Spain). Mar. Environ. Res. 2004, 58, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Gomiero, A.; Strafella, P.; Pellini, G.; Salvalaggio, V.; Fabi, G. Comparative effects of ingested PVC micro particles with and without adsorbed Benzo(a)pyrene vs. Spiked sediments on the cellular and subcellular processes of the benthic organism Hediste diversicolor. Front. Mar. Sci. 2018, 5, 1–12. [Google Scholar] [CrossRef]
- Benedetti, M.; Gorbi, S.; Fattorini, D.; d’Errico, G.; Piva, F.; Pacitti, D.; Regoli, F. Environmental hazards from natural hydrocarbons seepage: Integrated classification of risk from sediment chemistry, bioavailability and biomarkers responses in sentinel species. Environ. Pollut. 2014, 185, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Regoli, F.; Pellegrini, D.; Cicero, A.M.; Nigro, M.; Benedetti, M.; Gorbi, S.; Fattorini, D.; d’Errico, G.; Di Carlo, M.; Nardi, A.; et al. A multidisciplinary weight of evidence approach for environmental risk asses-ment at the Costa Concordia wreck: Integrative indices from Mussel Watch. Mar. Environ. Res. 2014, 96, 92–104. [Google Scholar] [CrossRef]
- Bebianno, M.; Pereira, C.; Rey, F.; Cravo, A.; Duarte, D.; d’Errico, G.; Regoli, F. Integrated approach to assess ecosystem health in harbor areas. Sci. Total Environ. 2015, 514, 92–107. [Google Scholar] [CrossRef]
- Mezzelani, M.; Gorbi, S.; Da Ros, Z.; Fattorini, D.; d’Errico, G.; Milan, M.; Bargelloni, L.; Regoli, F. Ecotoxicological potential of non-steroidal anti-inflammatory drugs (NSAIDs) in marine organisms: Bioavailability, biomarkers and natural occurrence in Mytilus galloprovincialis. Mar. Environ. Res. 2016, 121, 31–39. [Google Scholar] [CrossRef]
- Mestre, N.C.; Rocha, T.L.; Canals, M.; Cardoso, C.; Danovaro, R.; Dell’Anno, A.; Gambi, C.; Regoli, F.; Sanchez-Vidal, A.; Bebianno, M.J. Environmental hazard assessment of a marine mine tailings deposit site and potential implications for deep-sea mining. Environ. Pollut. 2017, 228, 169–178. [Google Scholar] [CrossRef]
- Nardi, A.; Mincarelli, L.F.; Benedetti, M.; Fattorini, D.; d’Errico, G.; Regoli, F. Indirect effects of climate changes on cadmium bioavailability and biological effects in the Mediterranean mussel Mytilus galloprovincialis. Chemosphere 2017, 169, 493–502. [Google Scholar] [CrossRef]
- Pittura, L.; Avio, C.G.; Giuliani, M.E.; d’Errico, G.; Keiter, S.H.; Cormier, B.; Gorbi, S.; Regoli, F. Microplastics as Vehicles of Environmental PAHs to Marine Organisms: Combined Chemical and Physical Hazards to the Mediterranean Mussels, Mytilus galloprovincialis. Front. Mar. Sci. 2018, 5. [Google Scholar] [CrossRef]
- UNI EN ISO 16703:2011. Soil Quality—Determination of Content of Hydrocarbon in the Range C10 to C40 by Gas Chromatography; ISO: Geneva, Switzerland, 2011. [Google Scholar]
- EPA. Method 5035A: Closed-System Purge-and-Trap and Extraction for Volatile Organics in Soil and Waste Samples. Revision 1, Washington, DC, USA; 2002. Available online: https://www.epa.gov/sites/production/files/2015-07/documents/epa-5035a.pdf (accessed on 14 November 2020).
- EPA. Method 8260D: Volatile Organic Compounds by Gas Chromatography/Mass Spectrometry (GC/MS). Revision 4 June 2018; 2006. Available online: https://www.epa.gov/sites/production/files/2017-04/documents/method_8260d_update_vi_final_03-13-2017.pdf (accessed on 14 November 2020).
- EPA. Method 3545A (SW-846): Pressurized Fluid Extraction (PFE). 2007. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/3545a.pdf (accessed on 14 November 2020).
- EPA. Method 8310 (SW-846): Polynuclear Aromatic Hydrocarbons. 1986. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/8310.pdf (accessed on 14 November 2020).
- EPA. Method 6010C (SW-846): Inductively Coupled Plasma—Atomic Emission Spectrometry. 2010. Available online: http://gwmc.ou.edu/files/metals_EPA6010c.pdf (accessed on 14 November 2020).
- EPA. Method 7060A: Arsenic (Atomic Absorption, Furnace Technique). 1994. Available online: https://settek.com/documents/EPA-Methods/PDF/EPA-Method-7060A.pdf (accessed on 14 November 2020).
- EPA. Method 7131: Cadmium (Atomic Absorption, Furnace Technique). 1994. Available online: https://settek.com/documents/EPA-Methods/PDF/EPA-Method-7131-A.pdf (accessed on 14 November 2020).
- EPA. Method 7473: Mercury in Solids and Solutions by Thermal Decomposition, Amalgamation, and Atomic Absorption Spectro-photometry. 2007. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/7473.pdf (accessed on 14 November 2020).
- EPA. Method 7421: Lead (Atomic Absorption, Furnace Technique). 1986. Available online: http://ctntes.arpa.piemonte.it/Raccolta%20Metodi%202003/pdf/Metodi%20EPA/7421.pdf (accessed on 14 November 2020).
- UNEP/RAMOGE. Manual on the Biomarkers Recommended for the MED POL Biomonitoring Programme; UNEP: Athens, GA, USA, 1999; 39p. [Google Scholar]
- Cong, Y.; Banta, G.T.; Selck, H.; Berhanu, D.; Valsami-Jones, E.; Forbes, V.E. Toxic effects and bioaccumulation of nano-, micron- and ionic-Ag in the polychaete, Nereis diversicolor. Aquat. Toxicol. 2011, 105, 403–411. [Google Scholar] [CrossRef]
- Gorbi, S.; Lamberti, C.V.; Notti, A.; Benedetti, M.; Fattorini, D.; Moltedo, G.; Regoli, F. An ecotoxicological protocol with caged mussels, Mytilus galloprovincialis, for monitoring the impact of an offshore platform in the Adriatic sea. Mar. Environ. Res. 2008, 65, 34–49. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Courtney, K.D.; Andres, V., Jr.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–90. [Google Scholar] [CrossRef]
- Regoli, F.; Winston, G.W. Quantification of Total Oxidant Scavenging Capacity of Antioxidants for Peroxynitrite, Peroxyl Radicals, and Hydroxyl Radicals. Toxicol. Appl. Pharmacol. 1999, 156, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Akerboom, T.P.; Sies, H. Assay of glutathione, glutathione disulfide, and glutathione mixed disulfides in biological samples. Methods Enzymol. 1981, 77, 373–382. [Google Scholar] [CrossRef]
- ISO 11348-3, 2019. Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Vibrio Fischeri (Luminescent Bacteria Test)—Part 3: Method Using Freeze-Dried Bacteria; ISO: Geneva, Switzerland, 2007. [Google Scholar]
- ISO 10253:2016. Water Quality—Marine Algal Growth Inhibition Test with Skeletonema sp. and Phaeodactylum Tricornutum; ISO: Geneva, Switzerland, 2016. [Google Scholar]
- UNICHIM. Qualità Dell’Acqua–Determinazione Della Tossicità Le-Tale a 24 h, 48 h e 96 h di Esposizione con Naupli di Tigriopus fulvus (Fischer, 1860) (Crustacea: Copepoda); Method no. M.U. 2396:14; UNICHIM: Milano, Italy, 2014. [Google Scholar]
- EPA. Method 3051A: Microwave Assisted Acid Digestion of Sediments, Sludges, Soils, and Oils. 2007. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/3051a.pdf (accessed on 16 November 2020).
- Bryan, G.; Hummerstone, L. Adaptation of the polychaete Nereis diversicolor to estuarine sediments containing high concentrations of zinc and cadmium. J. Mar. Biol. Assoc. UK. 1973, 53, 839–857. [Google Scholar] [CrossRef]
- ASTM. E1611-00: Standard Guide for Conducting Sediment Toxicity Tests with Polychaetous Annelids; ASTM International: West Conshohocken, PA, USA, 2007. [Google Scholar]
- EPA. Method 3052: Microwave Assisted Acid Digestion of Siliceous and Organically Based Matrices. 1996. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/3052.pdf (accessed on 16 November 2020).
- Moltedo, G.; Martuccio, G.; Catalano, B.; Gastaldi, L.; Maggi, C.; Virno-Lamberti, C.; Cicero, A.M. Biological responses of the polychaete Hediste diversicolor (O.F.Müller, 1776) to inorganic mercury exposure: A multimarker approach. Chemosphere 2019, 219, 989–996. [Google Scholar] [CrossRef]
- Homa, J.; Olchawa, E.; Sturzenbaum, S.R.; Morgan, A.J.; Plytycz, B. Early-phase immunodetection of metallothionein and heat shock proteins in extruded earthworm coelomocytes after dermal exposure to metal ions. Environ. Pollut. 2005, 135, 275–280. [Google Scholar] [CrossRef]
- Catalano, B.; Moltedo, G.; Martuccio, G.; Gastaldi, L.; Virno-Lamberti, C.; Lauria, A.; Ausili, A. Can Hediste diversicolor (Nereidae, Polychaete) be considered a good candidate in evaluating PAH contamination? A multimarker approach. Chemosphere 2012, 86, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Manfra, L.; Moltedo, G.; Lamberti, C.V.; Maggi, C.; Finoia, M.G.; Giuliani, S.; Onorati, F.; Gabellini, M.; Di Mento, R.; Cicero, A.M. Metal Content and Toxicity of Produced Formation Water (PFW): Study of the Possible Effects of the Discharge on Marine Environment. Arch. Environ. Contam. Toxicol. 2007, 53, 183–190. [Google Scholar] [CrossRef] [PubMed]
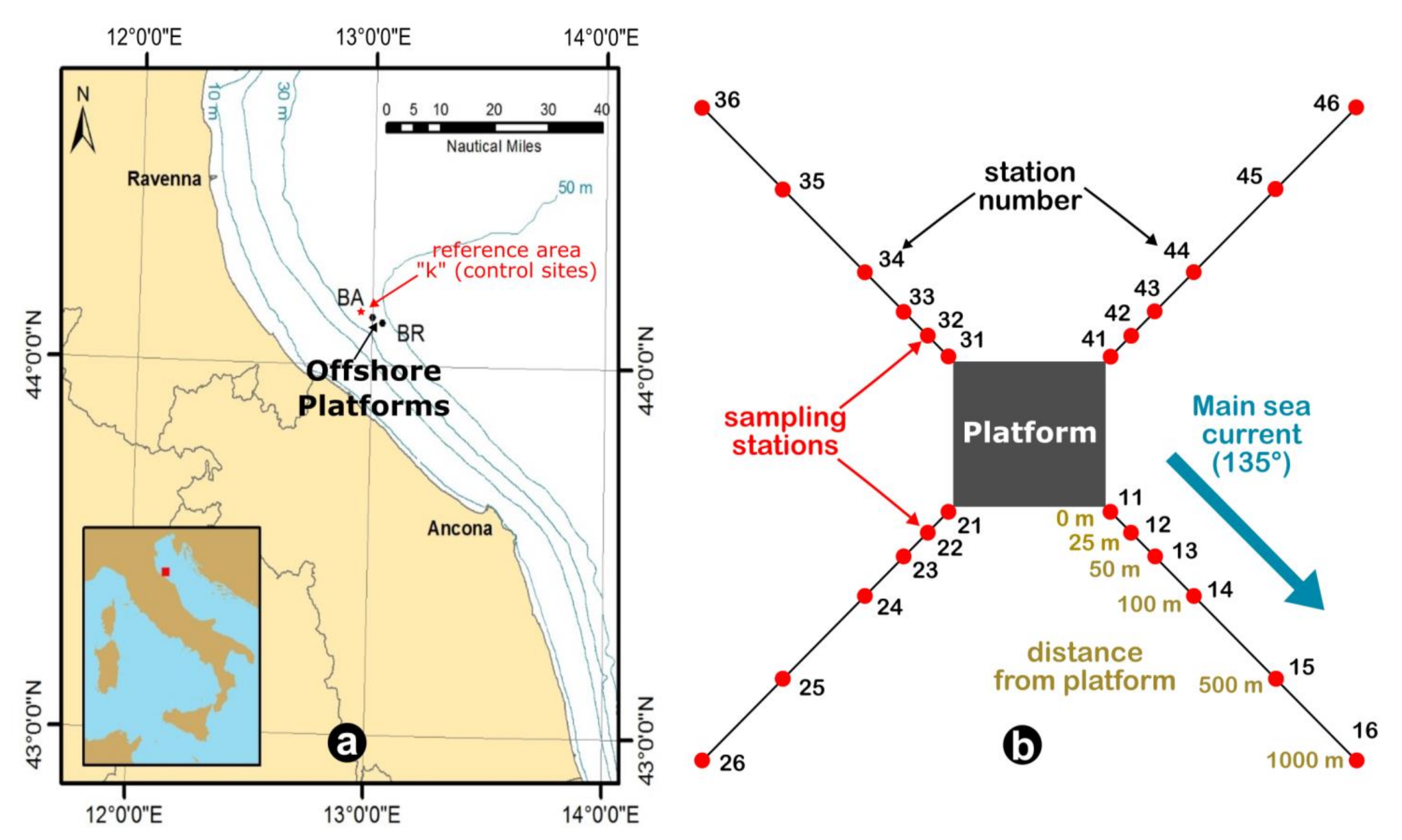
| LOE | Evidence Line | Analysis | Method |
|---|---|---|---|
| 1 | Sediment chemical characterization | Total hydrocarbons (C10–C40) Volatile hydrocarbons (BTEX and nC6–C10) PAHs Metals: Ba, Crtot, Cu, Fe, Mn, Ni, Pb, Zn As Cd Hg | UNI EN ISO 16703, 2011 [30] EPA 5035A, 2002 [31] EPA 8260D, 2006 [32] EPA 3545A, 2007 [33] EPA 8310, 1986 [34] EPA 6010C, 2010 [35] EPA 7060, 1994 [36] EPA 7131, 1994 [37] EPA 7473, 2007 [38] |
| 2 | Bioaccumulation Hediste diversicolor (polychaete) | PAHs Metals: Ba, Crtot, Cu, Fe, Mn, Ni, Zn As Cd Pb Hg | EPA 3545A, 2007 [33] EPA 8310, 1986 [34] EPA 6010C, 2010 [35] EPA 7060, 1994 [36] EPA 7131, 1994 [37] EPA 7421, 1986 [39] EPA 7473, 2007 [38] |
| 3 | Biomarkers Hediste diversicolor (polychaete) | LMS-ESO (whole tissue) COMET (coelomocytes) MN (coelomocytes) AChE (whole tissue) MT/MTLPs (whole tissue) CAT (whole tissue) TOSCA HO and ROO (whole tissue) GSTs (whole tissue) tGSH (whole tissue) | UNEP_Ramoge, 1999 [40] Cong et al., 2011 [41] Gorbi et al., 2008 [42] Ellman et al., 1961 [43] UNEP_Ramoge, 1999 [40] Regoli et al., 2004 [11] Regoli and Winston, 1999 [44] Regoli et al., 2004 [11] Akerboom and Sies, 1981 [45] |
| 4 | Bioassays | Bacterial bioluminescence test (Aliivibrio fischeri) Algal growth assay (Dunaliella tertiolecta) Copepod mortality test (Tigriopus fulvus) | ISO 11348-3, 2019 [46] ISO 10253, 2016 (modified) [47] UNICHIM 2396, 2014 [48] |
| Control Sites | BA Platform Stations | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Contaminants | 11 | 12 | 13 | 14 | ||||||
| mean | s.d. | mean | s.d. | mean | s.d. | mean | s.d. | mean | s.d. | |
| Polycyclic aromatic hydrocarbons PAH (ng/g) | ||||||||||
| Naphthalene | 3.18 | 2.87 | 21.89 | 4.59 | 93.20 | 6.49 | 4.31 | 2.68 | 9.44 | 2.87 |
| Acenaphthene | 0.59 | 0.14 | 0.37 | 0.21 | 7.42 | 3.44 | 0.41 | 0.27 | <0.5 | |
| Fluorene | 1.13 | 1.05 | 3.52 | 0.55 | 4.34 | 2.24 | 2.36 | 0.22 | 2.74 | 0.69 |
| Phenanthrene | 22.01 | 1.66 | 42.89 | 13.04 | 72.44 | 23.25 | 32.64 | 5.02 | 28.47 | 6.51 |
| Anthracene | 0.66 | 0.04 | 1.36 | 0.16 | 3.93 | 0.86 | 1.00 | 0.07 | 1.23 | 0.21 |
| Fluoranthene | 1.29 | 0.19 | 3.41 | 1.81 | 12.74 | 2.36 | 3.06 | 1.19 | 3.45 | 1.41 |
| Pyrene | 1.59 | 0.13 | 5.05 | 1.92 | 26.15 | 6.68 | 3.03 | 0.84 | 10.25 | 2.38 |
| Benzo(a)Anthracene | 0.75 | 0.19 | 0.44 | 0.17 | 2.11 | 0.28 | 0.67 | 0.13 | 0.45 | 0.35 |
| Chrysene | 0.65 | 0.38 | <0.5 | 10.89 | 1.56 | 0.63 | 0.38 | 2.60 | 0.42 | |
| Benzo(b)Fluoranthene | 2.56 | 0.38 | 0.67 | 0.39 | 4.02 | 0.73 | 2.30 | 1.21 | 2.88 | 0.35 |
| Benzo(k)Fluoranthene | 0.86 | 0.06 | 0.59 | 0.13 | 1.09 | 0.09 | 0.66 | 0.06 | 0.64 | 0.09 |
| Benzo(a)Pyrene | 0.77 | 0.21 | 0.47 | 0.19 | 1.07 | 0.18 | 0.72 | 0.02 | 0.99 | 0.35 |
| Dibenzo(a,h)Anthracene | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | |||||
| Benzo(g,h,i)Perylene | 0.74 | 0.61 | 0.38 | 0.23 | 1.10 | 0.32 | 0.75 | 0.16 | 1.69 | 0.35 |
| Indeno(1,2,3,c,d)Pyrene | 0.74 | 0.02 | 0.36 | 0.19 | 0.63 | 0.33 | 0.41 | 0.28 | 0.35 | 0.17 |
| Total PAHs | 37.20 | 3.10 | 80.65 | 22.51 | 241.05 | 26.82 | 52.57 | 9.93 | 64.85 | 11.24 |
| Trace metals (mg/kg) | ||||||||||
| Ba | 0.48 | 0.28 | 27.64 | 41.54 | 9.61 | 6.17 | 1.69 | 0.68 | 15.29 | 21.19 |
| Cr | 0.83 | 0.18 | 1.03 | 0.57 | 0.56 | 0.23 | 0.58 | 0.20 | 0.65 | 0.25 |
| Cu | 7.77 | 0.39 | 6.40 | 0.63 | 7.23 | 0.38 | 6.84 | 0.08 | 9.18 | 0.39 |
| Fe | 1496.59 | 366.50 | 1860.34 | 1126.92 | 1322.17 | 4.99 | 1270.40 | 113.29 | 1462.09 | 218.43 |
| Mn | 8.75 | 2.39 | 10.61 | 5.37 | 7.76 | 0.14 | 7.52 | 0.21 | 7.99 | 1.35 |
| Ni | 2.73 | 0.07 | 2.95 | 0.34 | 2.78 | 0.12 | 2.79 | 0.32 | 3.27 | 0.48 |
| Pb | 0.23 | 0.04 | 0.29 | 0.12 | 0.31 | 0.08 | 0.17 | 0.01 | 0.29 | 0.10 |
| Zn | 56.78 | 5.98 | 61.75 | 7.84 | 67.39 | 4.29 | 57.97 | 0.58 | 60.52 | 6.76 |
| As | 14.21 | 0.62 | 12.80 | 0.94 | 13.68 | 0.46 | 15.12 | 1.04 | 14.87 | 0.26 |
| Cd | 0.04 | 0.00 | 0.06 | 0.00 | 0.07 | 0.01 | 0.07 | 0.02 | 0.05 | 0.01 |
| V | 1.16 | 0.09 | 1.26 | 0.51 | 0.93 | 0.02 | 0.94 | 0.08 | 1.11 | 0.16 |
| Control Sites | BR Platform Stations | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Contaminants | 11 | 12 | 13 | 14 | ||||||
| mean | s.d. | mean | s.d. | mean | s.d. | mean | s.d. | mean | s.d. | |
| Polycyclic aromatic hydrocarbons PAH (ng/g) | ||||||||||
| Naphthalene | 0.84 | 0.52 | 6.11 | 2.11 | 7.03 | 1.97 | 4.92 | 2.63 | 5.32 | 3.54 |
| Acenaphthene | 0.78 | 0.91 | 2.01 | 0.73 | 2.21 | 0.06 | 1.03 | 0.71 | 1.51 | 0.29 |
| Fluorene | 2.11 | 0.67 | 5.63 | 0.50 | 4.63 | 0.56 | 3.84 | 0.60 | 4.38 | 0.64 |
| Phenanthrene | 34.96 | 8.55 | 69.81 | 6.68 | 63.83 | 21.69 | 49.62 | 9.22 | 45.01 | 4.12 |
| Anthracene | 1.50 | 0.20 | 2.78 | 0.26 | 2.41 | 0.92 | 1.91 | 0.70 | 1.67 | 0.20 |
| Fluoranthene | 4.59 | 0.58 | 8.82 | 1.02 | 7.10 | 2.19 | 6.17 | 0.85 | 4.83 | 1.31 |
| Pyrene | 1.97 | 0.27 | 6.10 | 0.94 | 5.78 | 1.88 | 4.94 | 1.59 | 4.12 | 0.66 |
| Benzo(a)Anthracene | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | |||||
| Chrysene | <0.5 | <0.5 | 0.40 | 0.26 | 0.50 | 0.23 | <0.5 | |||
| Benzo(b)Fluoranthene | 1.27 | 0.49 | 2.35 | 1.18 | 2.40 | 1.10 | 1.89 | 0.23 | 2.31 | 0.80 |
| Benzo(k)Fluoranthene | 0.54 | 0.27 | 0.77 | 0.16 | 0.46 | 0.18 | 0.66 | 0.15 | 0.71 | 0.21 |
| Benzo(a)Pyrene | <0.5 | 0.33 | 0.14 | 0.51 | 0.22 | 0.45 | 0.17 | 0.38 | 0.22 | |
| Dibenzo(a,.h)Anthracene | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | |||||
| Benzo(g,h,i)Perylene | <0.5 | <0.5 | 0.52 | 0.25 | <0.5 | <0.5 | ||||
| Indeno(1,2,3,c,d)Pyrene | 0.62 | 0.35 | 0.56 | 0.53 | 0.67 | 0.21 | 0.55 | 0.28 | 0.51 | 0.23 |
| Total PAHs | 48.78 | 9.28 | 104.93 | 6.59 | 97.52 | 29.01 | 76.15 | 14.55 | 70.49 | 7.05 |
| Trace metals (mg/kg) | ||||||||||
| Ba | 0.29 | 0.07 | 1.90 | 2.78 | 1.40 | 1.47 | 24.88 | 25.54 | 14.49 | 15.04 |
| Cr | 0.44 | 0.13 | 0.73 | 0.63 | 0.48 | 0.09 | 0.88 | 0.47 | 0.87 | 0.55 |
| Cu | 9.07 | 1.30 | 6.09 | 0.99 | 6.92 | 0.83 | 8.05 | 1.04 | 9.08 | 0.39 |
| Fe | 2929.52 | 144.50 | 2903.30 | 1545.63 | 3156.19 | 1297.85 | 2913.22 | 449.07 | 3160.75 | 1256.85 |
| Mn | 10.77 | 0.44 | 9.92 | 1.28 | 10.01 | 0.77 | 13.10 | 3.92 | 12.78 | 6.37 |
| Ni | 2.04 | 0.33 | 2.03 | 0.25 | 2.09 | 0.12 | 2.62 | 0.31 | 2.79 | 0.10 |
| Pb | 0.25 | 0.06 | 0.16 | 0.08 | 0.14 | 0.03 | 0.22 | 0.04 | 0.23 | 0.01 |
| Zn | 55.92 | 2.56 | 52.55 | 3.11 | 52.40 | 4.51 | 53.32 | 2.43 | 51.83 | 3.95 |
| As | 11.95 | 0.77 | 10.17 | 0.80 | 11.58 | 1.31 | 11.24 | 1.32 | 10.40 | 0.66 |
| Cd | 0.01 | 0.00 | 0.02 | 0.00 | 0.01 | 0.01 | 0.02 | 0.00 | 0.02 | 0.00 |
| V | 0.85 | 0.02 | 0.82 | 0.47 | 0.63 | 0.03 | 1.02 | 0.30 | 1.06 | 0.52 |
| LMS-ESO | AChE | CAT | COMET | MN | tGSH | GSTs | MT/ MTLPs | TOSCA HO | TOSCA ROO | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| st | mean | s.d. | mean | s.d. | mean | s.d. | mean | s.d. | mean | s.e.m. | mean | s.d. | mean | s.d. | mean | s.d. | mean | s.d. | mean | s.d. |
| K | 22.46 | 7.40 | 71.19 | 16.20 | 9.59 | 3.31 | 25.70 | 6.77 | 0.35 | 0.15 | 0.57 | 0.18 | 47.87 | 14.61 | 0.37 | 0.20 | 574.68 | 147.72 | 807.32 | 209.73 |
| BA | ||||||||||||||||||||
| 11 | 20.25 | 14.17 | 88.86 | 17.76 | 7.92 | 2.45 | 34.20 | 5.62 | 0.60 | 0.10 | 0.46 | 0.13 | 48.84 | 9.22 | 0.42 | 0.11 | 494.86 | 71.87 | 669.86 | 170.30 |
| 12 | 19.89 | 9.47 | 73.64 | 15.45 | 10.89 | 3.73 | 34.70 | 6.58 | 1.10 | 0.24 | 0.36 | 0.04 | 48.27 | 4.15 | 0.43 | 0.13 | 668.93 | 127.78 | 878.56 | 169.02 |
| 13 | 22.76 | 5.80 | 77.72 | 15.55 | 11.02 | 4.11 | 41.73 | 9.39 | 1.10 | 0.37 | 0.44 | 0.06 | 52.24 | 9.03 | 0.45 | 0.21 | 675.31 | 147.63 | 792.54 | 37.62 |
| 14 | 30.46 | 9.55 | 98.75 | 20.87 | 9.07 | 3.24 | 22.30 | 1.21 | 0.40 | 0.19 | 0.43 | 0.07 | 50.59 | 13.27 | 0.45 | 0.20 | 573.47 | 171.05 | 664.13 | 238.23 |
| BR | ||||||||||||||||||||
| 11 | 34.72 | 14.98 | 70.36 | 12.38 | 8.38 | 2.51 | 33.95 | 3.47 | 0.40 | 0.10 | 0.62 | 0.09 | 47.06 | 16.99 | 0.63 | 0.19 | 432.06 | 188.41 | 767.00 | 256.33 |
| 12 | 36.38 | 9.56 | 69.51 | 17.53 | 7.15 | 2.19 | 30.20 | 3.35 | 0.50 | 0.27 | 0.66 | 0.11 | 40.99 | 3.36 | 0.32 | 0.19 | 459.96 | 183.92 | 850.40 | 238.88 |
| 13 | 33.88 | 8.65 | 71.08 | 14.89 | 11.24 | 4.33 | 24.59 | 1.53 | 0.40 | 0.19 | 0.64 | 0.21 | 35.50 | 8.61 | 0.55 | 0.12 | 617.25 | 197.41 | 908.23 | 225.94 |
| 14 | 36.25 | 10.22 | 68.10 | 16.77 | 8.49 | 3.49 | 25.51 | 5.78 | 0.30 | 0.12 | 0.73 | 0.25 | 36.83 | 3.08 | 0.45 | 0.19 | 522.72 | 135.92 | 618.67 | 212.85 |
| Aliivibrio fischeri | Dunaliella tertiolecta | Tigriopus fulvus | ||||
|---|---|---|---|---|---|---|
| st | Mean Effect Bioluminescence Inhibition (%) | s.d. | Mean Effect Growth Rate (%) | s.d. | Mean Effect Mortality Rate (%) | s.d. |
| K1 | 15.04 | 0.36 | −69.40 | 10.84 | 2.5 | 5.0 |
| K2 | 16.09 | 1.48 | −78.12 | 4.79 | 2.5 | 5.0 |
| K3 | −19.11 | 1.27 | −70.44 | 6.35 | 10.00 | 11.55 |
| BA | ||||||
| 11 | 7.50 | 0.65 | −136.11 | 11.32 | 0.0 | 0.0 |
| 12 | 12.56 | 4.35 | −115.17 | 7.21 | 82.5 | 22.17 |
| 13 | 7.17 | 1.54 | −106.35 | 18.08 | 65.0 | 5.77 |
| 14 | 8.61 | 1.98 | −68.81 | 10.78 | 50.0 | 14.14 |
| BR | ||||||
| 11 | 7.71 | 1.44 | −111.62 | 14.62 | 95.0 | 5.77 |
| 12 | 7.30 | 0.58 | −104.33 | 7.01 | 87.5 | 15.0 |
| 13 | 5.45 | 1.00 | −93.53 | 2.99 | 100.0 | 0.0 |
| 14 | 8.68 | 1.57 | −82.13 | 14.24 | 72.5 | 17.08 |
| Station | LOE1 (Chemical Hazard Against SQA) | LOE2 (Hazard for Bioavailability) | LOE3 (Hazard for Biomarkers) | LOE4 (Hazard for Bioassays) | WOE Index (Integrated Risk) | |
|---|---|---|---|---|---|---|
| K | MODERATE 66.8% | ABSENT 11.7% | ABSENT 9.8% | ABSENT 11.7% | SLIGHT |  |
| BA | ||||||
| 11 | MAJOR 49.6% | MODERATE 29.2% | SLIGHT 15.1% | ABSENT 6.1% | MODERATE |  |
| 12 | SEVERE 38.2% | MODERATE 17.7% | MODERATE 16.0% | MAJOR 28.1% | MAJOR |  |
| 13 | MODERATE 29.7% | SLIGHT 14.8% | MODERATE 22.2% | MAJOR 33.3% | MODERATE | 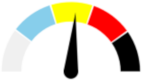 |
| 14 | SEVERE 48.4% | SLIGHT 17.4% | ABSENT 4.3% | MODERATE 29.8% | MODERATE | 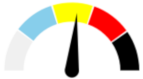 |
| BR | ||||||
| 11 | MAJOR 34.8% | SLIGHT 13.1% | SLIGHT 13.6% | MAJOR 38.6% | MODERATE |  |
| 12 | MODERATE 36.3% | SLIGHT 13.5% | SLIGHT 10.0% | MAJOR 40.2% | MODERATE | 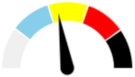 |
| 13 | SEVERE 37.9% | MODERATE 20.7% | SLIGHT 7.9% | MAJOR 33.5% | MAJOR | 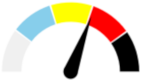 |
| 14 | MODERATE 33.8% | SLIGHT 21.0% | ABSENT 5.0% | MAJOR 40.2% | MODERATE | 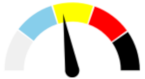 |
| Area | LOE1 (Chemical Hazard Against K) | LOE2 (Hazard for Bioavailability) | LOE3 (Hazard for Biomarkers) | LOE4 (Hazard for Bioassays) | WOE Index (Integrated Risk) | |
|---|---|---|---|---|---|---|
| K | ABSENT 25.0% | ABSENT 25.0% | ABSENT 25.0% | ABSENT 25.0% | ABSENT | 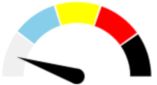 |
| BA | ||||||
| 11 | SEVERE 50.2% | MODERATE 28.8% | SLIGHT 15% | ABSENT 6% | MODERATE | 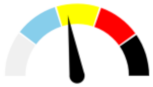 |
| 12 | SEVERE 34.8% | MODERATE 18.6% | MODERATE 16.9% | MAJOR 29.7% | MAJOR |  |
| 13 | SEVERE 38.1% | SLIGHT 13.0% | MODERATE 19.6% | MAJOR 29.3% | MODERATE | 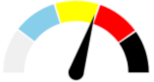 |
| 14 | SEVERE 45.5% | SLIGHT 18.4% | ABSENT 4.6% | MODERATE 31.5% | MODERATE |  |
| BR | ||||||
| 11 | SEVERE 39.8% | SLIGHT 12.1% | SLIGHT 12.5% | MAJOR 35.6% | MODERATE | 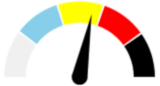 |
| 12 | SEVERE 42.2% | SLIGHT 12.3% | SLIGHT 9.1% | MAJOR 36.4% | MODERATE |  |
| 13 | SEVERE 37.3% | MODERATE 20.8% | SLIGHT 8.0% | MAJOR 33.8% | MAJOR | 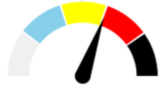 |
| 14 | SEVERE 43.2% | SLIGHT 18.0% | ABSENT 4.3% | MAJOR 34.5% | MODERATE | 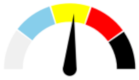 |
| Area of Application (Location) | LOE1 —Chemistry (Matrix) | LOE2 —Bioavailability (Tissues) (sp.) (Exposure Type) | LOE 3 —Biomarkers (Tissues) (sp.) | LOE 4 —Bioassays (sp.) (Matrix) | LOE5— Benthic Community | Further (Statistical) Analysis | Chemical Reference Value/Threshold/SQG | References |
|---|---|---|---|---|---|---|---|---|
| Petrol-chemical site (Adriatic Sea, Italy) Harbour site (Adriatic Sea, Italy) | PAHs, Aliphatic hydrocarbons, trace metals (As, Cd, Cr, Cu, Fe, Mn, Ni, Hg, Pb, V) (sediments) | PAHs, Aliphatic hydrocarbons, trace metals (eel liver and gills) (Anguilla anguilla) (field, lab) | EROD, MTs, AOX CAT, GSTs, GR, GPx-H2O2, GPx-CHP, tGSH, TOSCA ROO, TOSCA HO, LLP (liver) PAH metabolites (bile) AChE (brain) MN (gills) (A. anguilla) | growth test (Phaedactylum tricornutum) (elutriate) growth test (Dunaliella tertiolecta) (elutriate) Bioluminescence test (Aliivibrio fischeri) (elutriate, centrifuged sediment), mortality test (Tigriopus fulvus) (elutriate) | N.A. | N.A. | (Italian legislative Decree n. 152/2006) | Piva et al., 2011 [13] |
| Venice lagoon (Adriatic Sea, Italy) | PAHs, trace metals (As, Cd, Cr, Cu, Hg, Ni, Pb, V, Zn) (sediments) | PAHs, trace metals (As, Cd, Cr, Cu, Fe Hg, Mn, Ni, Pb, V, Zn) (eel liver) (A. anguilla) (lab.) | EROD, MTs, AOX, CAT, GSTs, GR, GPx-H2O2, GPx-CHP, tGSH, TOSCA ROO, TOSCA HO, LLP, LP, MDA (liver) PAH metabolites (bile) AChE (brain) Comet assay (blood) MN (gills) (A. anguilla) | Bioluminescence test (A. fischeri) (solid phase), embryo development test (Crassostrea Gigas) (elutriate) mortality/growth test (T. fulvus) (elutriate) larval development test (Acartia tonsa) (elutriate), fertilization test (Paracentrotus lividus) (elutriate) Mortality test (Corophium orientale) (whole sediment) | N.A. | N.A. | normative limits (Venice Protocol. 1993) | Benedetti et al., 2012 [16] |
| Offshore platform and seepage area (Adriatic Sea, Italy) | PAHs, Aliphatic hydrocarbons, VHs, trace metals (As, Cd, Cr, Cu, Fe, Hg, Mn, Ni, Pb, V, Zn) (sediments) | PAHs, Aliphatic hydrocarbons, VHs, trace metals (eel gills and liver, mussel whole tissues) (A. anguilla, Mytilus galloprovincialis) | EROD, AOX, CAT, GSTs, GR, GPx-CHP, TOSCA ROO, TOSCA HO, LLP (liver) PAH metabolites (bile) Comet Assay (blood) MN (gills) (A. anguilla) MTs, AOX, CAT, GSTs, GR, GPx-H2O2, GPx-CHP, tGSH, TOSCA ROO, TOSCA HO, LP, MDA (dig, gland) NRRT, Comet assay, MN, AChE (haemolymph) (M. galloprovincialis) | N.A. | N.A. | N.A. | TEL/PEL (Macdonald et al., 1996); (Italian legislative Decree n. 152/2006) | Benedetti et al., 2014 [23] |
| Harbour site (Portimão harbor, Atlantic Ocean, Portugal) | trace metals (Cd, Cr, Cu, Pb, Ni, Zn), PAHs, PCBs, HCB (sediment) | trace metals (mussel whole soft tissues) (M. galloprovincialis) (field) | SOD, CAT, GPx-H2O2, MDA, 4-HNE, MTs (dig. gland and gills). AChE (gills) ALP (gonad) ALAD (whole soft tissue) (M. galloprovincialis) | Mortality test (Corophium insidiosum) (whole sediment) Bioluminescence test (A. fischeri) (solid phase) | N.A. | N.A. | ERL/ERM (Long et al., 1995); TEL/PEL (Macdonald et al., 1996); Portuguese legislation (Portaria n°1450) French. Spanish. Uk. Italian normative limits for dredged sediments (Arrêté du 14/06/00; CEDEX. 1994; OSPAR. 2004; Italian D.Lvo 152/2006) | Bebianno et al., 2015 [25] |
| Non steroidal anti-inflammatory drugs (NSAIDs) exposure Portonovo Bay (Adriatic Sea, Italy) | N.A. | Not included | NRRT, G/H ratio, PA, Comet assay, MN (haemolymph) LP, NL, AOX, CAT, GSTs, GPx-H2O2, GPx-CHP, GR, tGSH, TOSCA ROO, TOSCA HO (dig. gland) AChE (haemolymph and gills) (M. galloprovincialis) | N.A. | N.A. | Multivariate PCA analysis of biomarker responses | N.A. | Mezzelani et al., 2016 [26] |
| Costa Concordia shipwreck (Giglio Island, Tirrenian Sea, Italy) | N.A. | VHs, Aliphatic hydrocarbons, PAHs, PCBs, OCPs, OSn, BFRs, trace metals (As, Cd, Cr, Cu, Hg, Ni, Pb, V, Zn), TASs (mussel whole soft tissues) (M. galloprovincialis) (field) | AChE (haemolymph and gills) MTs, AOX, CAT, GSTs, GPx-H2O2, GPx-CHP, GR, tGSH, MDA, TOSCA ROO, TOSCA HO, LP, NL, LLP (dig. gland) Comet assay, MN (gills) (M. galloprovincialis) | N.A. | N.A. | non-metric Multidimensional Scaling (nMDS) on bioaccumulation and biomarker responses | Regoli et al., 2014 [24] | |
| Mine tailing disposal (Portman Bay, Spain) | Trace metals (Ag, As, Au, Cd, Cr, Cu, Fe, Hg, Ni, Pb, Sb, Zn) (sediments) | Trace metals (mussel gills, digestive gland and mantle) (M. galloprovincialis) (field) | SOD, CAT, GPx, GSTs, MTs, MDA, 4-HNE (dig. gland, gills and mantle) (M. galloprovincialis) | Bioluminescence test (A. fischeri) (solid phase) | N.A. | N.A. | Spanish limits for dredged sediments (CIEM. 2015) | Mestre et al., 2017 [27] |
| Climate changes and Cd exposure | N.A. | Trace metals (Cd) (mussel gills, digestive gland) (M. galloprovincialis) (lab) | MTs, CAT, GSTs, GPx-H2O2, GPx-CHP, GR, tGSH, TOSCA ROO, TOSCA HO, MDA (dig. gland and gills) LP. NL (dig. glands) NRRT, PA, G/H ratio, Comet Assay, MN (haemolymph) (M. galloprovincialis) | N.A. | N.A. | N.A. | N.A. | Nardi et al., 2017 [28] |
| Microplastics (LDPE) and PAHs (Benzo-a-pyrene) exposure | N.A. | Benzo-a-pyrene (mussel digestive gland and gills) (M. galloprovincialis) (lab) | NRRT, PA, G/H ratio, Comet assay, MN (haemolymph) AChE (haemolymph and gills) AOX, CAT, GSTs, GPx-H2O2, GPx-CHP; GR, tGSH, TOSCA ROO, TOSCA HO, MDA, NL, (dig. gland) (M. galloprovincialis) | N.A. | N.A. | Multivariate statistical analyses (principal component analysis, PCA) | Pittura et al., 2018 [29] | |
| Offshore platforms (Adriatic Sea, Italy) | trace metals (Al, As, Cd, Cu, Cr, Hg, Ni, Pb, Zn), PAHs, Aliphatic hydrocarbons, (sediments) | trace metals, PAHs, Aliphatic hydrocarbons (native and transplanted mussel whole soft tissues) (M. galloprovincialis) (field) PAHs (ragworm whole tissues) (Hediste diversicolor) (lab) | CAT, GSTs, GPx-H2O2, GPx-CHP, GR, tGSH, TOSCA ROO, TOSCA HO, MDA, MTs (dig. gland) AChE, NRRT, PA, G/H ratio, Comet assay, MN (haemolymph) (M. galloprovincialis) | Bioluminescence test (A. fischeri) (solid phase) larval development test (A. tonsa) (solid phase) growth test (P. tricornutum) (elutriate) embryotoxicity assay (P. lividus) (elutriate) | AMBI index | N.A. | SQG (2000/60/EC) | Regoli et al., 2019 [9] |
| Industrial site (Gulf of Naples, Italy) | OM, trace metals (Al, As, Cd, Cr, Cu, Fe, Hg, Ni, Pb, V, Zn), metalloids, Aliphatic hydrocarbons, PAHs, PCBs, OTC, OCP, PCDDs, PCDFs (sediments) | trace metals, PAHs (mussel whole soft tissues) (M. galloprovincialis) (fish liver) (Mullus barbatus Pagellus erythrinus, Diplodus vulgaris) (field) | NRRT, AChE, MN (haemolymph) MTs (dig. gland) (M. galloprovincialis) PAH metabolites (bile) AChE (brain) EROD (liver) MN (gills) (M. barbatus, P. erythrinus, D. vulgaris) | Bioluminescence test (A. fischeri) (solid phase) growth test (Skeletonema costatum) (elutriate) Embryo test (P. lividus) (elutriate) | AMBI index | N.A. | dredged marine sediments SQG (DM 173/2016) | Morroni et al., 2020 [12] |
| Offshore platforms (Adriatic Sea, Italy) | trace metals (As, Cd, Ba, Mn, Cu, Cr, Fe Hg, Ni, Pb, Zn), PAHs, Aliphatic hydrocarbons, Volatile hydrocarbons, BTEX (sediments) | trace metals, PAHs (ragworm whole tissues) (H. diversicolor) (lab) | LLP (whole tissue) COMET (coelomocytes) MN (coelomocytes) AChE (whole tissue) MT/MTLPs (whole tissue) CAT (whole tissue) TOSCA HO and ROO (whole tissue) GSTs (whole tissue) tGSH (whole tissue) | Bioluminescence test (A. fischeri) (sediment pore water) growth test (D. tertiolecta) (sediment pore water) mortality test (T. fulvus) (sediment pore water) | N.A. | N.A. | EQS, (Italian D.Lvo 152/2006), ERL values, (Long et al., 1995),TEL, (Macdonald et al., 1996) | This Study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manfra, L.; Maggi, C.; d’Errico, G.; Rotini, A.; Catalano, B.; Maltese, S.; Moltedo, G.; Romanelli, G.; Sesta, G.; Granato, G.; et al. A Weight of Evidence (WOE) Approach to Assess Environmental Hazard of Marine Sediments from Adriatic Offshore Platform Area. Water 2021, 13, 1691. https://doi.org/10.3390/w13121691
Manfra L, Maggi C, d’Errico G, Rotini A, Catalano B, Maltese S, Moltedo G, Romanelli G, Sesta G, Granato G, et al. A Weight of Evidence (WOE) Approach to Assess Environmental Hazard of Marine Sediments from Adriatic Offshore Platform Area. Water. 2021; 13(12):1691. https://doi.org/10.3390/w13121691
Chicago/Turabian StyleManfra, Loredana, Chiara Maggi, Giuseppe d’Errico, Alice Rotini, Barbara Catalano, Silvia Maltese, Ginevra Moltedo, Giulia Romanelli, Giulio Sesta, Giuseppe Granato, and et al. 2021. "A Weight of Evidence (WOE) Approach to Assess Environmental Hazard of Marine Sediments from Adriatic Offshore Platform Area" Water 13, no. 12: 1691. https://doi.org/10.3390/w13121691
APA StyleManfra, L., Maggi, C., d’Errico, G., Rotini, A., Catalano, B., Maltese, S., Moltedo, G., Romanelli, G., Sesta, G., Granato, G., Lanera, P., Amici, M., Martuccio, G., Onorati, F., Di Mento, R., Berducci, M. T., Chiaretti, G., Faraponova, O., Regoli, F., & Tornambè, A. (2021). A Weight of Evidence (WOE) Approach to Assess Environmental Hazard of Marine Sediments from Adriatic Offshore Platform Area. Water, 13(12), 1691. https://doi.org/10.3390/w13121691











