Conversion of Whey into Value-Added Products through Fermentation and Membrane Fractionation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Microorganisms
2.2. Analytical Techniques
2.3. Whey Fermentations
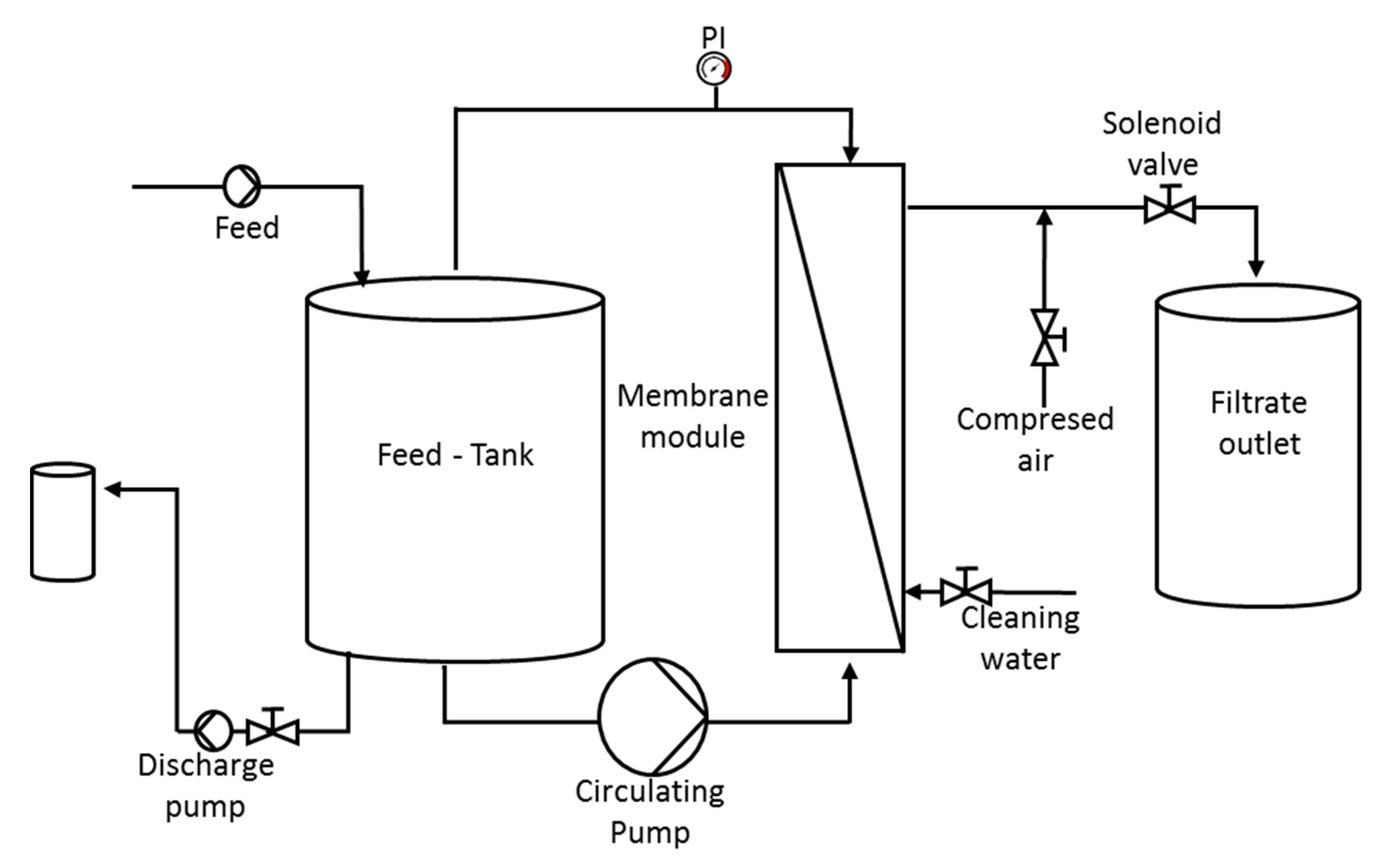
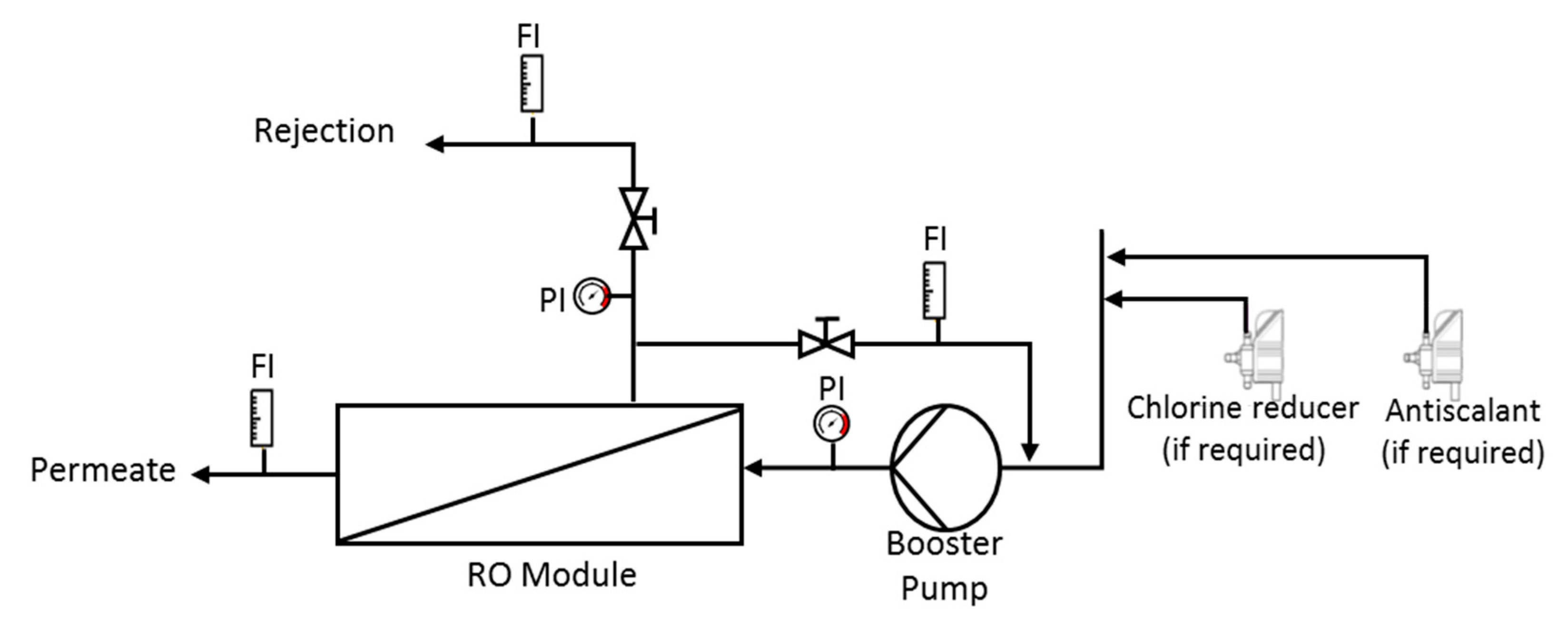
2.4. Filtration Test
- (1)
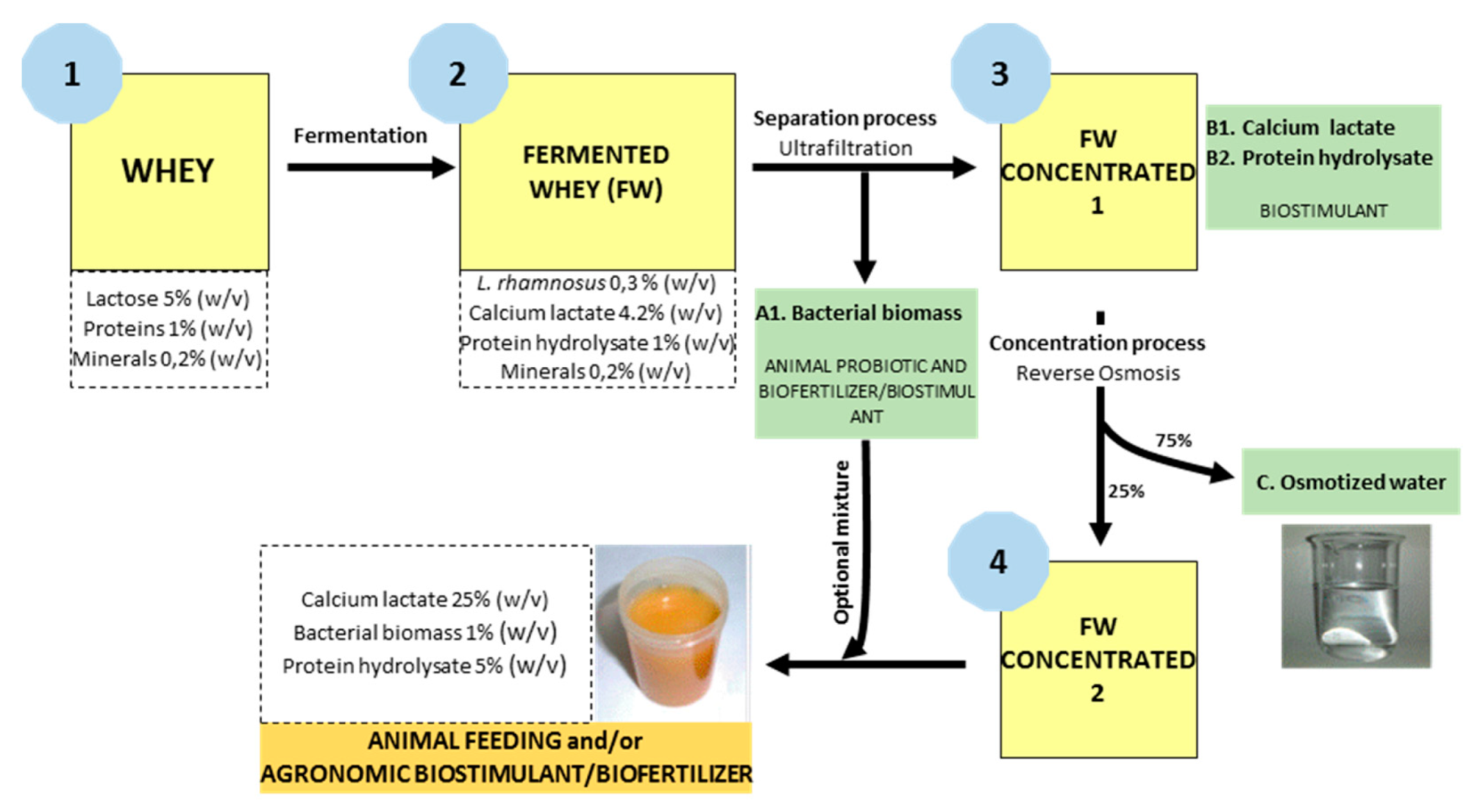
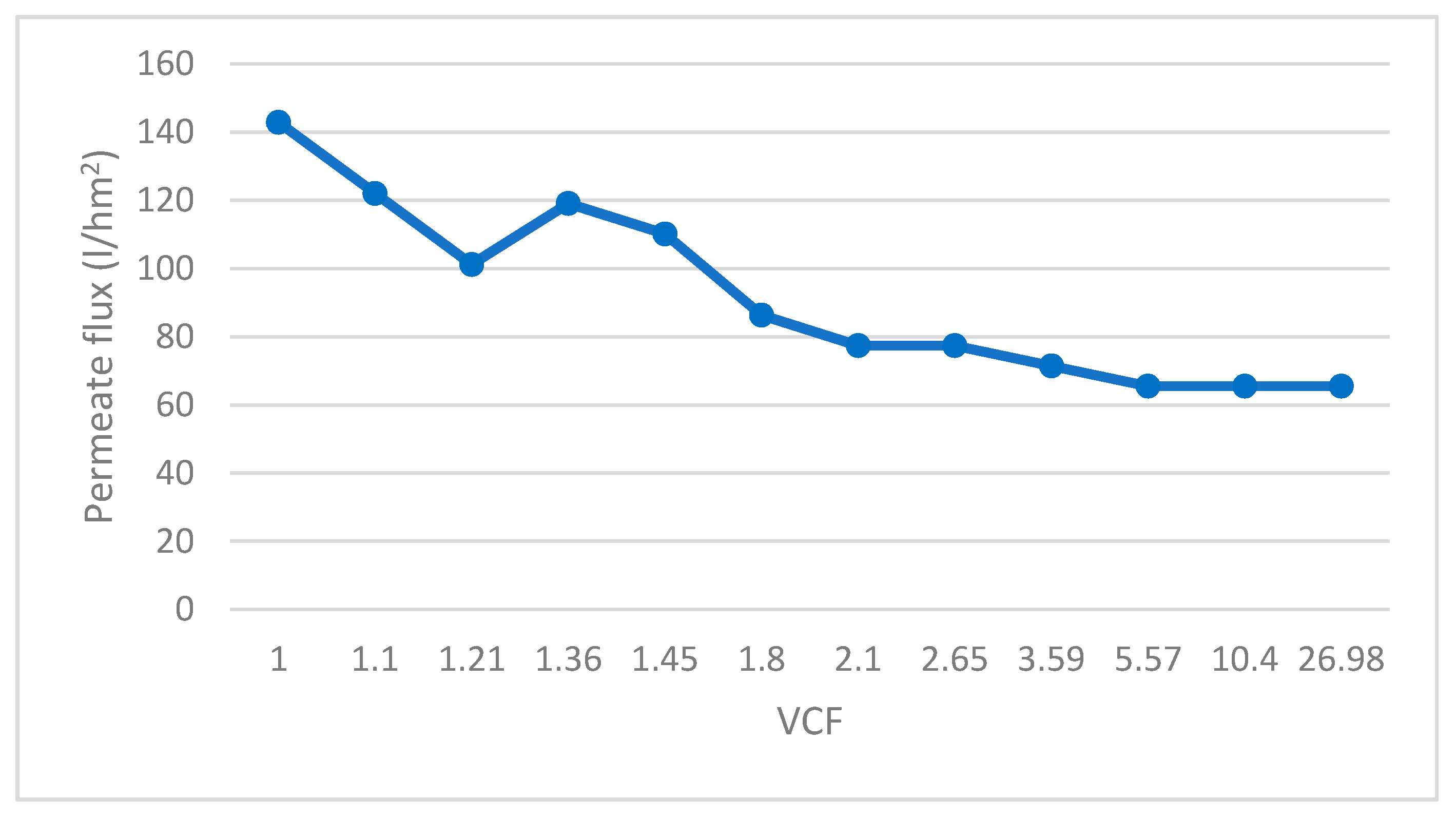
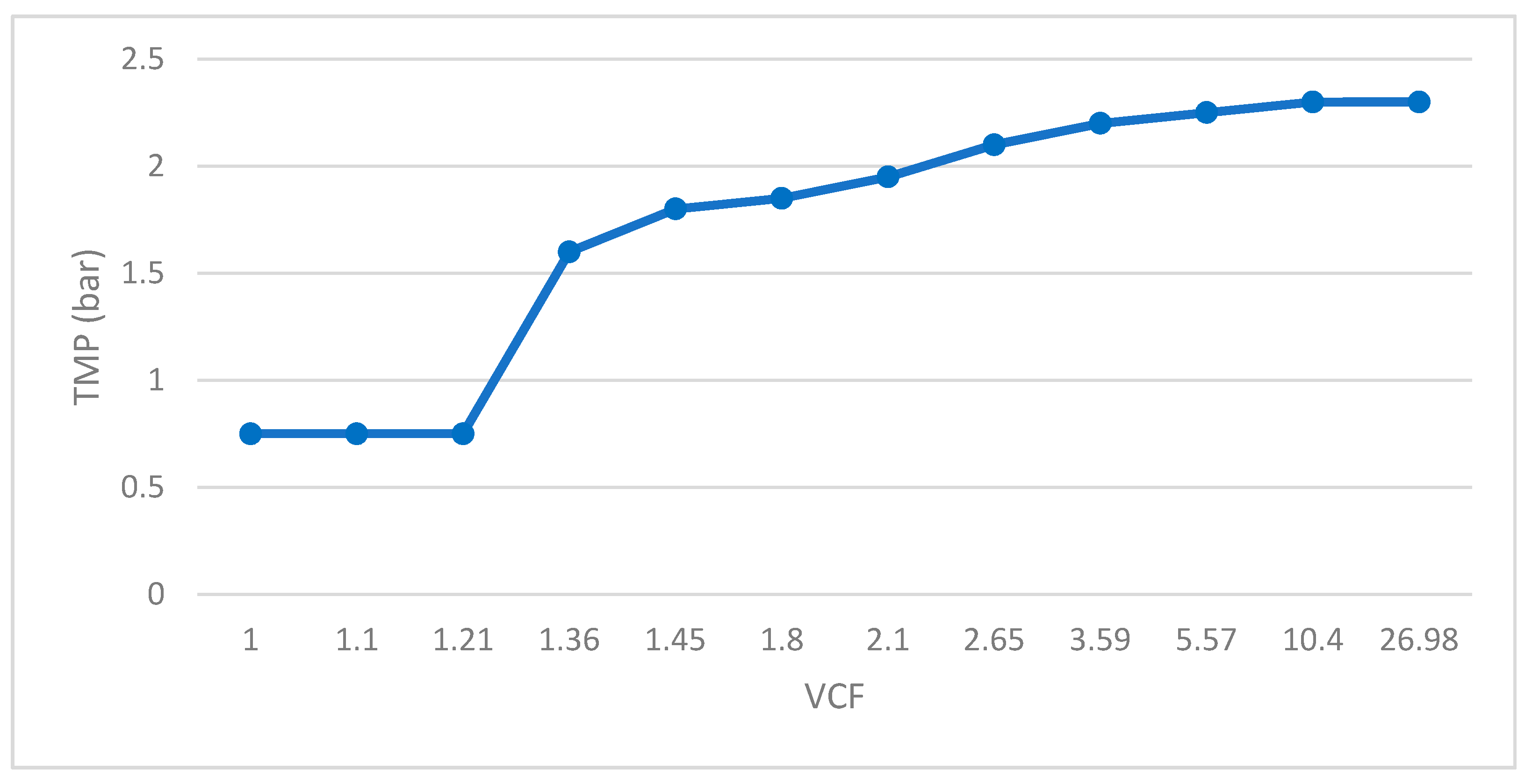
- Cross-flow ultrafiltration (UF) with a 0.1 μm pore size membrane, model M-7P1940 manufactured by Pall Corporation; made in ceramic material, the pilot system was configured with 7 membranes with a total filtration surface of 1.68 m2. Through this technique, the lactic bacteria will be retained in the concentrate. The use of ceramic UF membranes allowed for separating biomass from fermented whey, maintaining steady stable process parameters, reducing the fouling effect in comparison with conventional polymeric UF membranes. Feed pressure and concentrate pressure increased through the test duration, varying from 4 to 4.5 bar and from 2 to 2.5 bar, respectively. These values are determined by the characteristics of the fermented whey to be filtered, mainly by the concentration of solid particles contained in it. On the contrary, permeate pressure decreased from 2.25 to 1.2 bar. Evaluating the pressure values as a whole makes it possible to obtain a calculation of the transmembrane pressure, which increased from 0.75 to 2.30 bar, as a consequence of the increase of concentration factor. The increase of pressure is directly linked to the increase of fluid temperature, ranging from 20 to 34 °C. The permeate flow rate also experienced a decrease over the test time, from 240 to 110 L/h, which means that the flux (flow rate/membrane surface) decreased accordingly. The UF concentrate was reserved for subsequent binding to the concentrate resulting from reverse osmosis (Figure 1).
- (2)
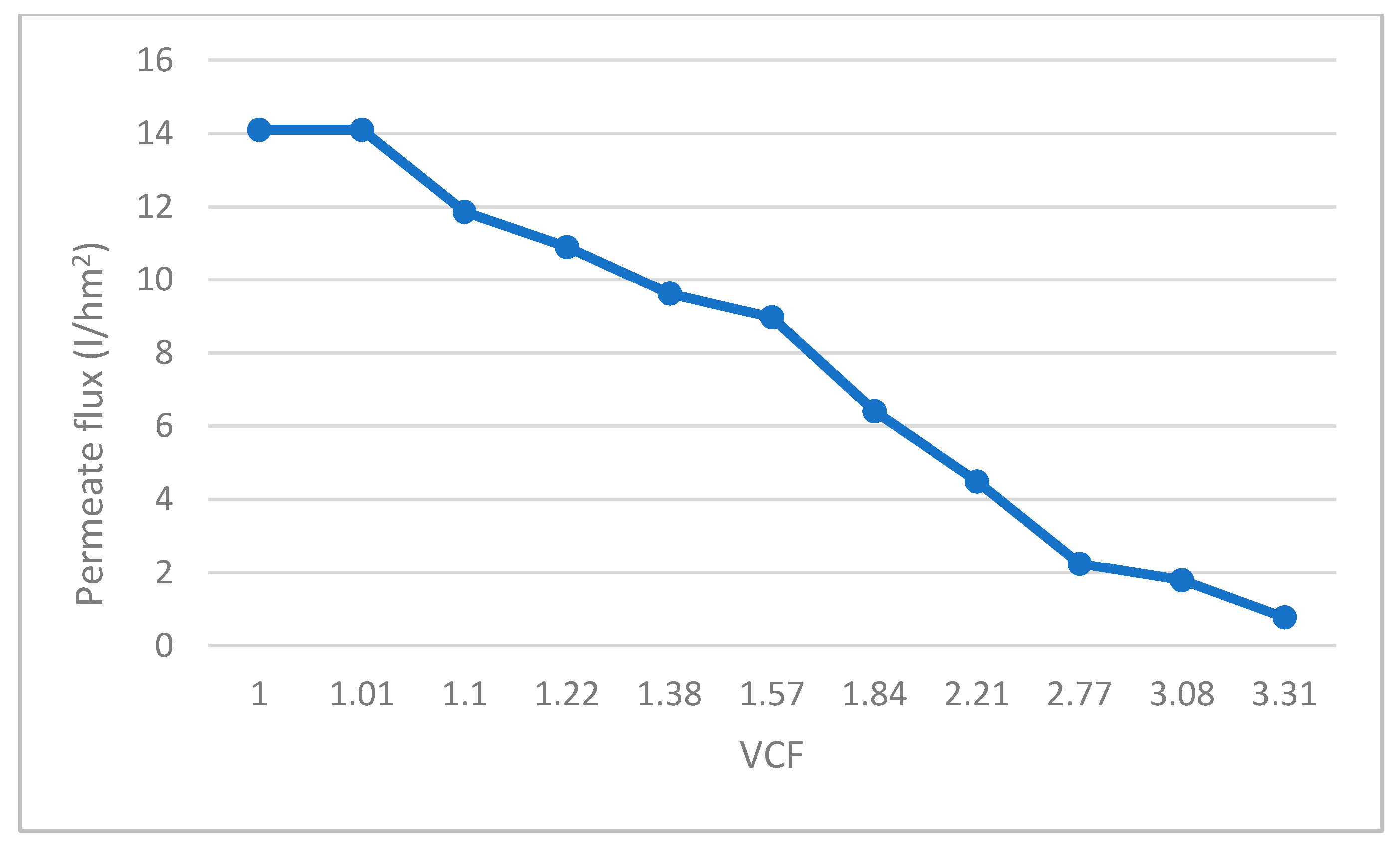
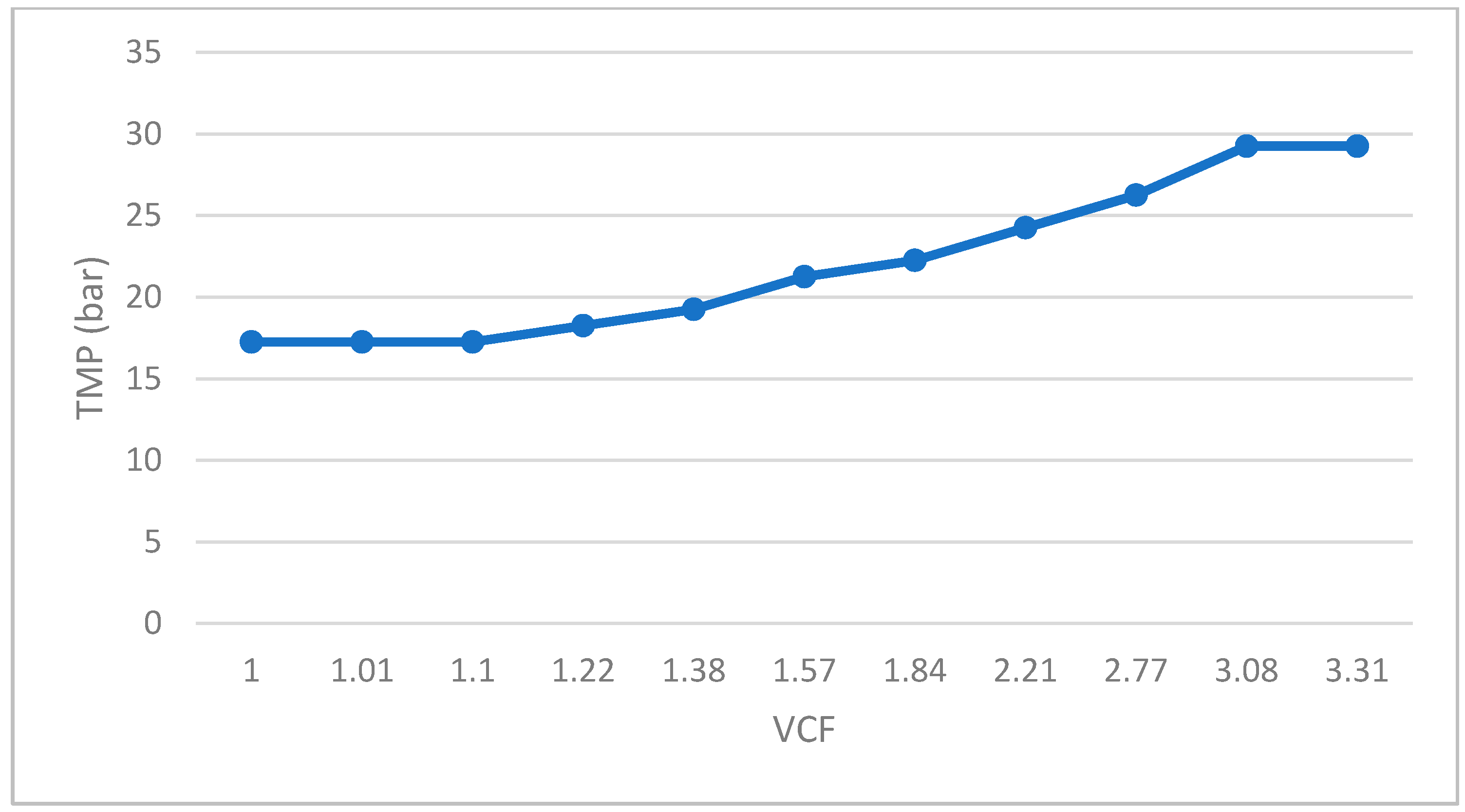
- Reverse osmosis (RO) of the filtrate resulting from the UF, using RO membranes manufactured by Filmtec. Model RO-3840/30FF with a 200 Da pore size, made in propylene. The pilot system was configured with 2 membranes connected in series, with a total filtration surface of 15.6 m2 (Figure 2). Reverse osmosis membranes allow for recovering water from the permeate of the ultrafiltrate and obtaining a high-value concentrate. With respect to the operational parameters, they have the same behavior as in the ultrafiltration stage. Feed pressure and concentrate pressure increased throughout the test duration, varying from 18 to 30 bar and from 16.5 to 28.5 bar, respectively. These values are determined by the characteristics of the fermented whey to be filtered, mainly by the concentration of dissolved solids contained in it. Evaluating the pressure values and considering that in this case, permeate discharge pressure is considered as 0 bar, makes it possible to obtain a calculation of the transmembrane pressure, which increased from 17.25 to 29.25 bar, as a consequence of the increase of concentration factor. The permeate flow rate also experienced a decrease over the test time, from 22 to 12 L/h, which means that the flux (flow rate/membrane surface) decreased too.
- (1)
- 707 kg of fermented whey was processed.
- (2)
- Before starting the filtration process, fermented whey material was stirred, due to the presence of small precipitates at the base of the feeding tank.
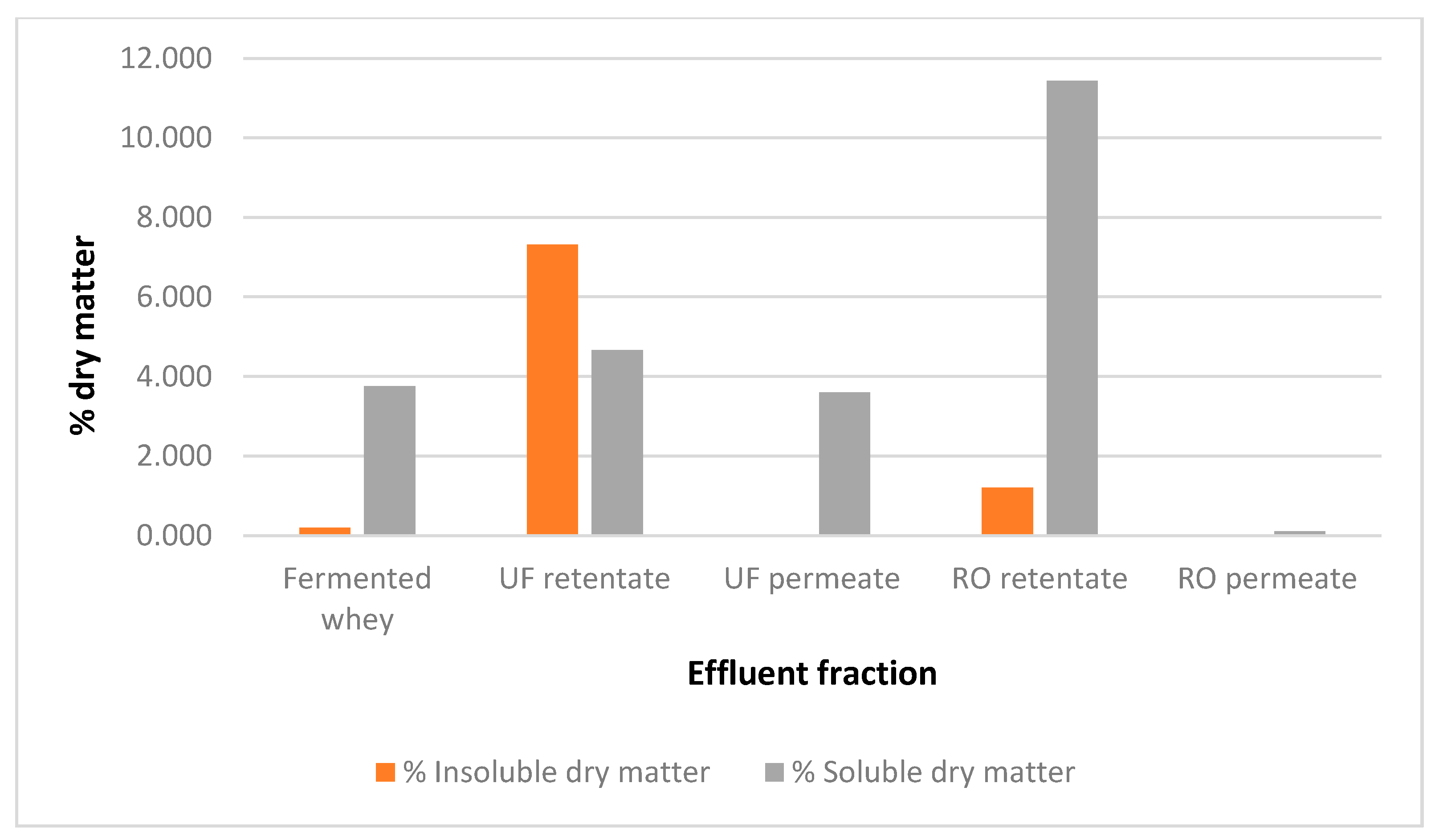
2.5. Dry Matter Determination
3. Results and Discussion
Pilot Filtration Test
- (1)
- Behavior of the filtration steps processing fermented whey.
- (2)
- Quantitative analysis of the different fractions obtained from the fermented whey.
4. Conclusions
- (1)
- The filtration tests carried out must be considered as a proof of concept. A full recirculation process has led to a severe reduction of the flux and a fast increase in the TMP, clear indicators of membrane silting, both in UF and RO steps. Industrial processes to perform a filtration step in continuous mode should be re-designed to reject a convenient part of the brine.
- (2)
- The rejection of the ultrafiltration stage is mainly constituted by bacterial biomass (Lactobacillus rhamnosus). On the other hand, the concentrate obtained in the reverse osmosis stage is rich in ammonium lactate as well as peptides and free amino acids. Both products are excellent food additives for the livestock industry as probiotics in animal feeding and as bio-stimulants/biofertilizers for agronomic purposes. They can be applied separately or mixed into a single, more complete product.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Villar Bonet, A. Situación y perspectivas de la gestión de los sueros de quesería generados en Cantabria. An. Inst. Estud. Agropecu. 2006, 17, 13–53. [Google Scholar]
- FEDNA Fundación Española para el Desarrollo de la Nutrición Animal. Productos lácteos. 2016. Available online: http://fundacionfedna.org/productos_lacteos (accessed on 5 May 2021).
- Vázquez Esnoval, C.O.; Pinto Ruiz, R.; Rodríguez Hernández, R.; De la Torre, J.C.; De Jesús, A.G. Uso, producción y calidad nutricional del lactosuero en la región central de Chiapas. Investig. Agropecu. 2017, 21, 65–75. [Google Scholar]
- Zamora-Carrillo, M.; Santamaría-Días, E.; Álvarez-Calvache, F.; Santamaría-Freire, E. Caracterización de los parámetros de calidad del agua para disminuir la contaminación durante el procesamiento de lácteos. Agroind. Sci. 2015, 5, 13–26. [Google Scholar]
- Canales Canales, C. Guía de Mejores Técnicas Disponibles en España del Sector de Productos del Mar; Ministerio de Medio Ambiente y Medio Rural y Marino: Madrid, Spain, 2007.
- Carvalho, F.; Prazeres, A.R.; Rivas, J. Cheese whey wastewater: Characterization and treatment. Sci. Total Environ. 2013, 445–446, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Cancino, B.; Espina, V.; Orellana, C. Whey concentration using microfiltration and ultrafiltration. Desalination 2006, 200, 557–558. [Google Scholar] [CrossRef]
- Meyer, P.; Hartinger, M.; Sigler, S.; Kulozik, U. Concentration of Milk and Whey by Membrane Technologies in Alternative Cascade Modes. Food Bioprocess. Technol. 2017, 10, 674–686. [Google Scholar] [CrossRef]
- Chen, Z.; Luo, J.; Hang, X.; Wan, Y. Physicochemical characterization of tight nanofiltration membranes for dairy wastewater treatment. J. Membr. Sci. 2018, 547, 51–63. [Google Scholar] [CrossRef]
- Busse, M. De man, rogosa and sharpe (MRS) agar. Prog. Ind. Microbiol. 1995, 34, 362–363. [Google Scholar] [CrossRef]
- Caballero, P.; Rodríguez-Morgado, B.; Macías, S.; Tejada, M.; Parrado, J. Obtaining Plant and Soil Biostimulants by Waste Whey Fermentation. Waste Biomass Valoriz. 2020, 11. [Google Scholar] [CrossRef]
- Gänzle, M.G. Lactic metabolism revisited: Metabolism of lactic acid bacteria in food fermentations and food spoilage. Curr. Opin. Food Sci. 2015, 2, 106–117. [Google Scholar] [CrossRef]
- Dogi, C.; García, G.; De Moreno de LeBlanc, A.; Greco, C.; Cavaglieri, L. Lactobacillus rhamnosus RC007 intended for feed additive: Immune-stimulatory properties and ameliorating effects on TNBS-induced colitis. Benef. Microbes 2016, 7, 539–547. [Google Scholar] [CrossRef] [PubMed]
| Whey (g/L) | Fermented Whey (g/L) | |
|---|---|---|
| Bacteria | 0.07 ± 0.01 | 3.2 ± 0.7 |
| Nitrogen | 10 ± 2 (Proteins) | 9.16 ± 1.8 (Hydrolyzed proteins) |
| Lactic acid | 5.2 ± 0.6 | 42 ± 2.7 |
| Minerals | 2.4 ± 0.2 | 2.4 ± 0.2 |
| Lactose | 50 ± 3.9 | 1.7 ± 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caballero, A.; Caballero, P.; León, F.; Rodríguez-Morgado, B.; Martín, L.; Parrado, J.; Vaswani, J.; Ramos-Martín, A. Conversion of Whey into Value-Added Products through Fermentation and Membrane Fractionation. Water 2021, 13, 1623. https://doi.org/10.3390/w13121623
Caballero A, Caballero P, León F, Rodríguez-Morgado B, Martín L, Parrado J, Vaswani J, Ramos-Martín A. Conversion of Whey into Value-Added Products through Fermentation and Membrane Fractionation. Water. 2021; 13(12):1623. https://doi.org/10.3390/w13121623
Chicago/Turabian StyleCaballero, Alejandro, Pablo Caballero, Federico León, Bruno Rodríguez-Morgado, Luis Martín, Juan Parrado, Jenifer Vaswani, and Alejandro Ramos-Martín. 2021. "Conversion of Whey into Value-Added Products through Fermentation and Membrane Fractionation" Water 13, no. 12: 1623. https://doi.org/10.3390/w13121623
APA StyleCaballero, A., Caballero, P., León, F., Rodríguez-Morgado, B., Martín, L., Parrado, J., Vaswani, J., & Ramos-Martín, A. (2021). Conversion of Whey into Value-Added Products through Fermentation and Membrane Fractionation. Water, 13(12), 1623. https://doi.org/10.3390/w13121623







