Cryptosporidium Oocysts Removal by Upflow Direct Filtration: Pilot Scale Assessment
Abstract
1. Introduction
2. Materials and Methods
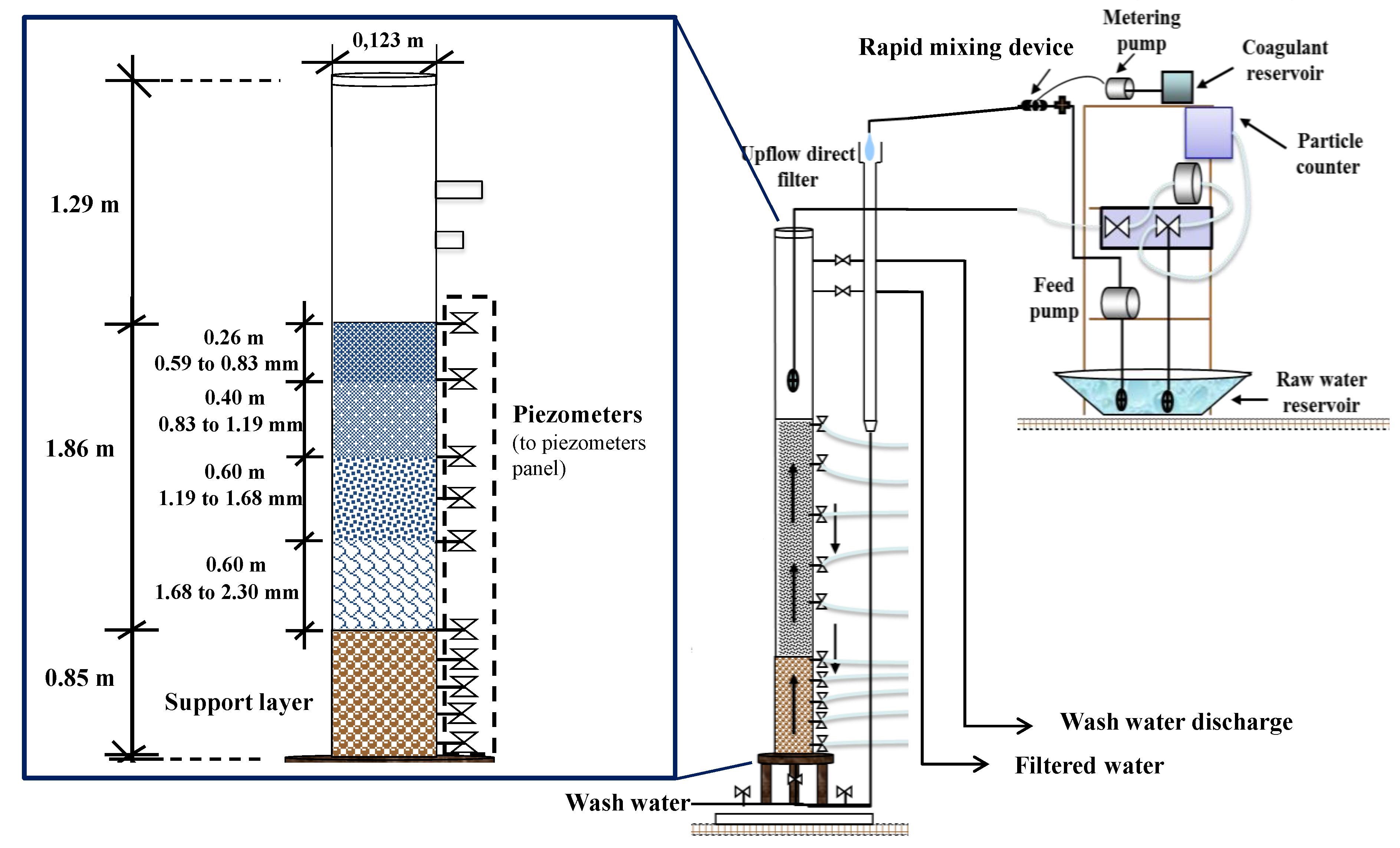
2.1. Pilot Plant Apparatus
2.2. Sampling and Analysis
2.3. Statistical Analysis
3. Results and Discussion
3.1. Turbidity and Cryptosporidium Oocysts
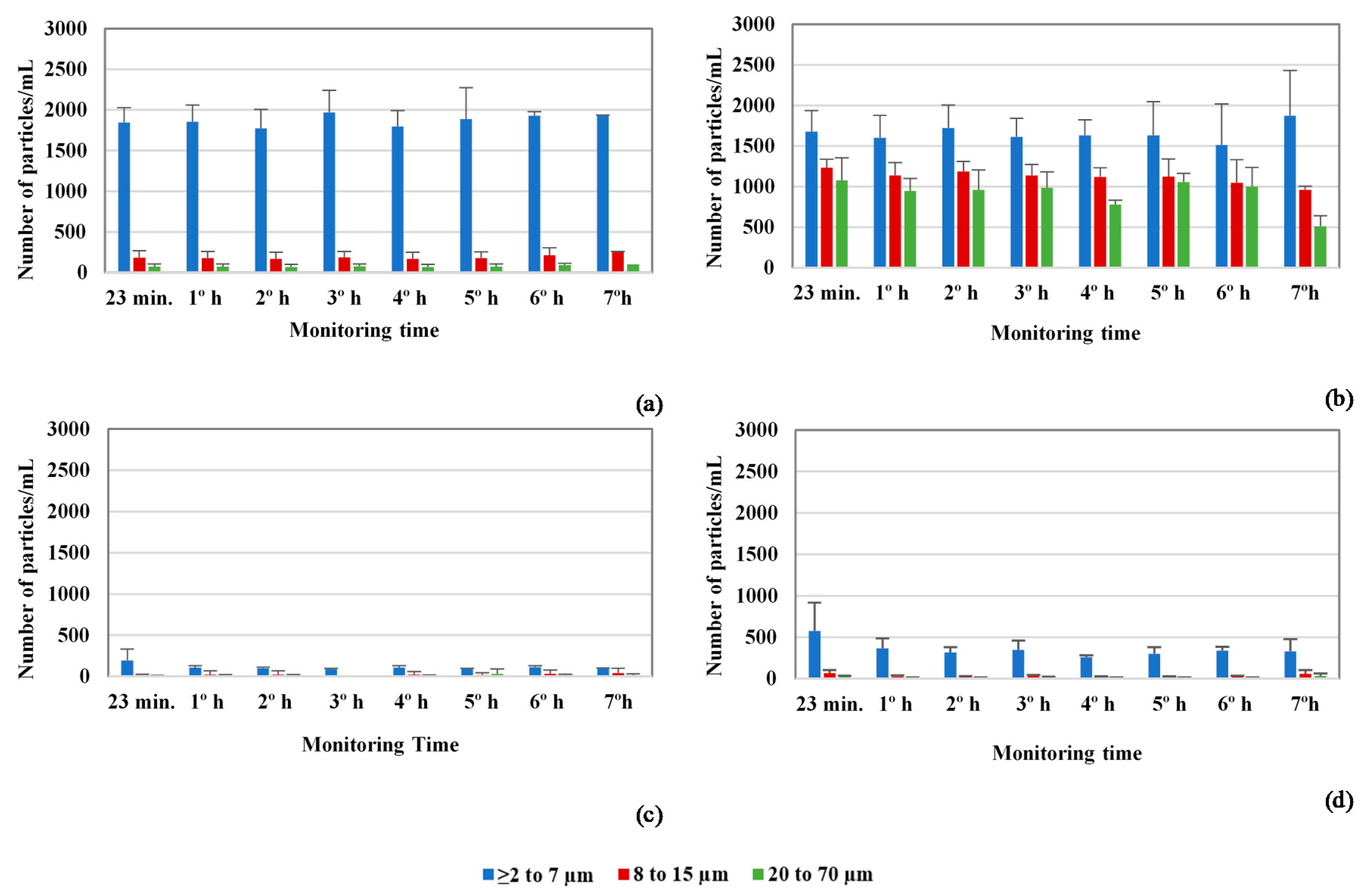
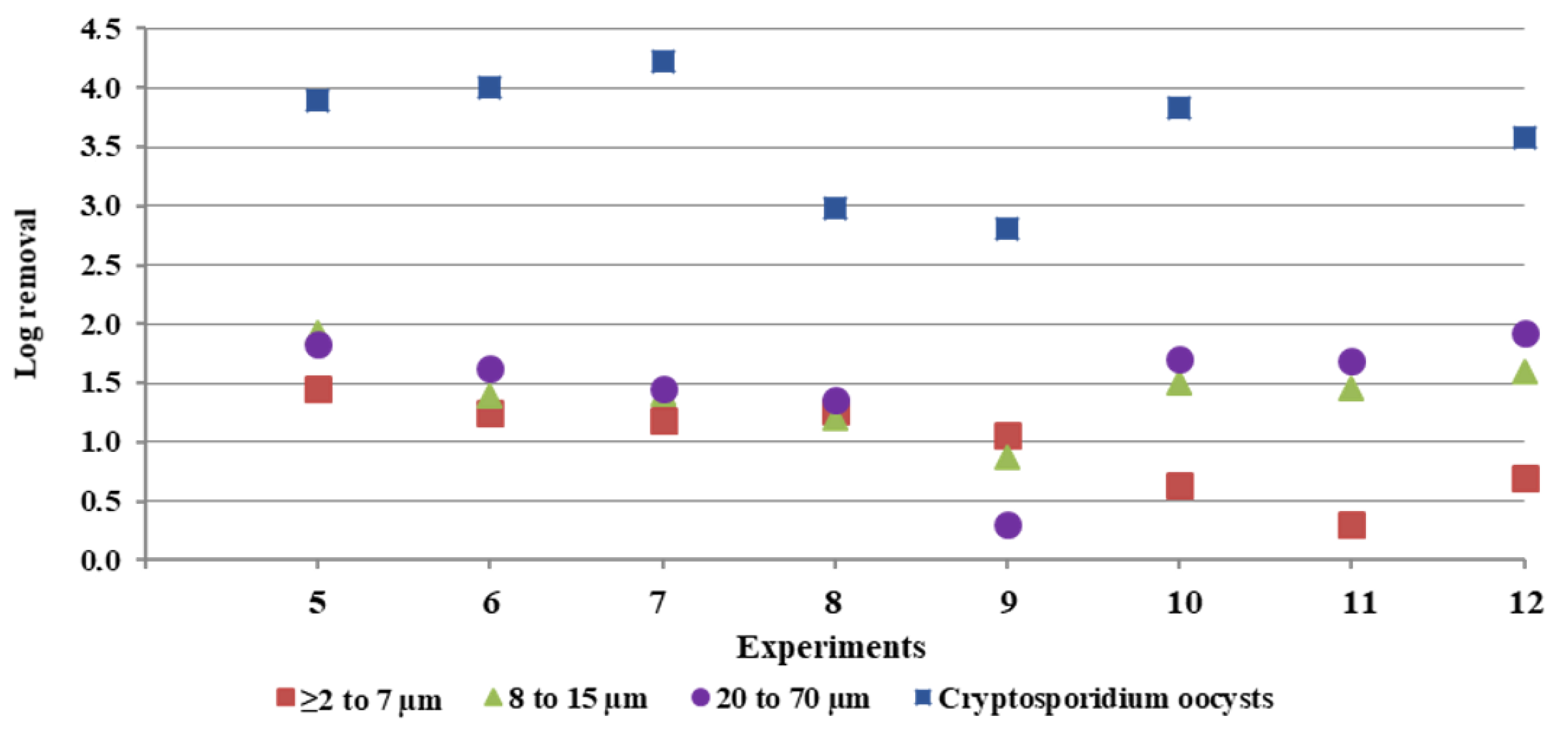
3.2. Particle Counts and Cryptosporidium Oocysts
3.3. Correlations Analysis between Cryptosporidium Oocysts and Particle Counts
4. Summary
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gamba, R.C.; Ciapina, E.M.P.; Espíndola, R.S.; Pacheco, A.; Pellizari, V. Detection of Cryptosporidium sp. oocysts in groundwater for human consumption in Itaquaquecetuba city, S. Paulo-Brazil. Braz. J. Microbiol. 2000, 31, 151–153. [Google Scholar] [CrossRef]
- Bastos, R.; Heller, L.; Vieira, M.; Brito, L.; Bevilacqua, P.D.; Nascimento, L. Giardia sp. cysts and Cryptosporidium spp. oocysts dynamics in southeast Brazil: Occurrence in surface water and removal in water treatment processes. Water Supply 2004, 4, 15–22. [Google Scholar] [CrossRef]
- Neto, R.C.; Santos, L.U.; Sato, M.I.Z.; Franco, R.M.B. Cryptosporidium spp. and Giardia spp. in the surface water supply of Campinas, Southeast Brazil. Wat. Sci. Tech. 2010, 62, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Branco, N.; Leal, D.A.G.; Franco, R. A Parasitological Survey of Natural Water Springs and Inhabitants of a Tourist City in Southeastern Brazil. Vector-Borne Zoonotic Dis. 2012, 12, 410–417. [Google Scholar] [CrossRef]
- Franco, R.M.B.; Branco, N.; Trainotti Amaro, B.C.; Neto, R.C.; Fiuza, V.R.S. Cryptosporidium Species and Giardia Genotypes Detected in Surface Water Supply of Campinas, Southeast Brazil, by Molecular Methods. J. Vet. Med. Res. 2016, 3, 1053. [Google Scholar]
- Grott, S.C.; Hartmann, B.; Filho, H.H.D.S.; Franco, R.; Greinert-Goulart, J. Detecção de cistos de Giardia spp. e oocistos de Cryptosporidium spp. na água bruta das estações de tratamento no município de Blumenau, SC, Brasil. Ambient. Agua-Interdiscip. J. Appl. Sci. 2016, 11, 689. [Google Scholar] [CrossRef]
- De Araújo, R.S.; Aguiar, B.; Dropa, M.; Razzolini, M.; Sato, M.I.Z.; Lauretto, M.D.S.; Galvani, A.T.; Padula, J.A.; Matté, G.R.; Matté, M.H. Detection and molecular characterization of Cryptosporidium species and Giardia assemblages in two watersheds in the metropolitan region of São Paulo, Brazil. Environ. Sci. Pollut. Res. 2018, 25, 15191–15203. [Google Scholar] [CrossRef]
- BRASIL Ministério da Saúde. Anexo XX da Portaria de Consolidação no 5 de 28 de setembro de 2017: Dispõe sobre os procedimentos de controle e de vigilância da qualidade da água para consumo humano e seu padrão de potabilidade. In Procedures for the Control and Surveillance of Water Quality for Human Consumption and Its Potability Standards; Ministry of Health: Brasília, Brazil, 2017. (In Portuguese) [Google Scholar]
- Bitton, G. Microbiology of Drinking Water; Wiley: Hoboken, NJ, USA, 2014; p. 312. [Google Scholar]
- Di Bernardo, L.; Mendes, C.G.N.; Brandão, C.C.S.; Sens, M.L.; Pádua, V.L. Tratamento de água para abastecimento por filtração direta (Projeto Prosab). In Drinking Water Treatment by Direct Filtration; ABES-RIMA: Rio de Janeiro, Brazil, 2003; p. 498. (In Portuguese) [Google Scholar]
- IBGE—Instituto Brasileiro de Geografia e Estatística. Pesquisa Nacional de Saneamento Básico. In National Survey on Drinking Water and Sanitation; Governo Federal: Brasília, Brasil, 2008. [Google Scholar]
- Ongerth, J.E.; Pecoraro, J.P. Removing Cryptosporidium using multimedia filters. J. Am. Water Works Assoc. 1995, 87, 83–89. [Google Scholar] [CrossRef]
- Emelko, M. Removal of viable and inactivated Cryptosporidium by dual- and tri-media filtration. Water Res. 2003, 37, 2998–3008. [Google Scholar] [CrossRef]
- Emelko, M.; Huck, P.M.; Douglas, I.P. Cryptosporidium and microsphere removal during late in-cycle filtration. J. Am. Water Works Assoc. 2003, 95, 173–182. [Google Scholar] [CrossRef]
- Yu, M.; Ahn, S.; Chung, S.; Noh, S.; Park, J.; Rhim, Y.; Jheong, W.; Chung, H. Evaluation of the rapid filtration system with particle size distribution and Cryptosporidium in different operating conditions. Water Supply 2006, 6, 129–139. [Google Scholar] [CrossRef]
- Fernandes, N.M.G. Influência do pH de coagulação e dosagem de sulfato de alumínio na remoção de oocistos de Cryptosporidium por Filtração Direta Descendente [Influence of coagulation pH and aluminium sulphate dosage on the removal of Cryptosporidium oocysts by downflow direct filtration]. Dissertation. In Master in Environmental Technology and Water Resources; Universidade de Brasília: Brasília, Brazil, 2007; 144 p. (In Portuguese) [Google Scholar]
- Gitis, V. Rapid sand filtration of Cryptosporidium parvum: Effects of media depth and coagulation. Water Supply 2008, 8, 129–134. [Google Scholar] [CrossRef]
- Brown, T.J.; Emelko, M. Chitosan and metal salt coagulant impacts on Cryptosporidium and microsphere removal by filtration. Water Res. 2009, 43, 331–338. [Google Scholar] [CrossRef]
- ABNT–Associação Brasileira de Noramas Técnicas. NBR 12216/1992 Projeto de estação de tratamento de água para abastecimento público. In Public Water Supply Systems-Designs of Water Treatment Works–Procedure; ABNT: Rio de Janeiro, Brazil, 1992; p. 17. (In Portuguese) [Google Scholar]
- Nieminski, E.C.; Ongerth, J.E. Removing Giardia and Cryptosporidium by conventional treatment and direct filtration. J. Am. Water Work. Assoc. 1995, 87, 96–106. [Google Scholar] [CrossRef]
- Sens, M.L.; Dalsasso, R.L.; Melo Filho, L.C.; Mondardo, R.I.; Simioni, D. Relatório de Atividades PROSAB-Edital 3–Tema I. In Report of Activities; Uinversidade Federal de Santa Catarina: Santa Catarina, Brazil, 2002; Personal Communication. [Google Scholar]
- USEPA. Method 1623: Cryptosporidium and Giardia in Water Using Filtration/IMS/FA; U.S. Environmental Protection Agency: Washington, DC, USA, 2005.
- Hall, T.; Pressdee, J.; Gregory, R.; Murray, K. Cryptosporidium removal during water treatment using dissolved air flotation. Water Sci. Tech. 1995, 31, 125–135. [Google Scholar] [CrossRef]
- Huck, P.; Coffey, B.; Anderson, W.B.; Emelko, M.; Maurizio, D.; Slawson, R.; Douglas, I.; Jasim, S.; O’Melia, C. Using turbidity and particle counts to monitor Cryptosporidium removals by filters. Water Supply 2002, 2, 65–71. [Google Scholar] [CrossRef]
- Huck, P.M.; Coffey, B.M.; Emelko, M.B.; Maurizio, D.D.; Slawson, R.M.; Anderson, W.B.; Oever, J.V.D.; Douglas, A.P.; O’Melia, C.R. Effects of Filter Operation on Cryptosporidium Removal Microbial Pathogens. J. Am. Water Works Assoc. 2002, 94, 97–111. [Google Scholar] [CrossRef]
- Emelko, M.; Huck, P.M.; Coffey, B.M. A review of Cryptosporidium removal by granular media filtration. J. Am. Water Works Assoc. 2005, 97, 101–115. [Google Scholar] [CrossRef]
- O’Melia, C.R.; Ali, W. The Role of Retained Particles in Deep Bed Filtration. Ninth Int. Conf. Water Pollut. Res. 1979, 10, 167–182. [Google Scholar] [CrossRef]
- USEPA. National Primary Drinking Water Regulations: Electronic Code of Federal Regulations. Federal Register–Part II–40CFR, Parts 9, 141 and 142. Long Term 2 Enhanced Surface Water Treatment Rule; Final Rule; U.S. Environmental Protection Agency: Washington, DC, USA, 2006.
- Xagoraraki, I.; Harrington, G.; Assavasilavasukul, P.; Standridge, J.H. Removal of Emerging Waterborne Pathogens and Pathogen Indicators by Pilot-Scale Conventional Treatment. J. Am. Water Works Assoc. 2004, 96, 102–113. [Google Scholar] [CrossRef]
- Swertfeger, J.; Metz, D.H.; Demarco, J.; Braghetta, A.; Jacangelo, J.G. Effect of filter media on cyst and oocyst removal. J. Am. Water Works Assoc. 1999, 91, 90–100. [Google Scholar] [CrossRef]
- Dugan, N.R.; Fox, K.R.; Owens, J.H.; Miltner, R.J. Controlling Cryptosporidium Oocysts Using Conventional Treatment. J. Am. Water Works Assoc. 2001, 93, 64–76. [Google Scholar] [CrossRef]
- Fagundes, A.P. Remoção de oocistos de Cryptosporidium por Filtração Direta-Influência de alguns aspectos operacionais [Removal of Cryptosporidium oocysts by direct filtration-Influence of some operational aspects]. Dissertação. In Master in Environmental Technology and Water Resources; Universidade de Brasília: Brasília, Brazil, 2006; p. 143. (In Portuguese) [Google Scholar]
- Liu, L.; Wang, Y.; Craik, S.; James, W.; Shu, Z.; Narain, R.; Liu, Y. Removal of Cryptosporidium surrogates in drinking water direct filtration. Colloids Surf. B Biointerfaces 2019, 181, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Lechevallier, M.W.; Norton, W.D. Examining Relationships between Particle Counts and Giardia, Cryptosporidium, and Turbidity. J. Am. Water Works Assoc. 1992, 84, 54–60. [Google Scholar] [CrossRef]
- Ribas, F.; Bernal, A.; Perramon, J. Elimination of Giardia cysts, Cryptosporidium oocysts, turbidity and particles in a drinking water treatment plant with clarification and double filtration. Water Sci. Technol. 2000, 41, 203–211. [Google Scholar] [CrossRef]
- Mendez, I.M.R. Remoção de microesferas de poliestireno, em substituição a oocistos de Crypstosporidium, por filtração direta ascendente no tratamento de água usando o coagulante quitosana [Microspheres of polystyrene removal, in substitution of Crypstosporidium oocysts, by up-flow in-line filtration in water treatment, using chitosan coagulant]. Dissertation. In Master in Environmental Technology and Water Resources; Universidade de Brasília: Brasilia, Brazil, 2014; p. 123. (In Portuguese) [Google Scholar]
- Gitis, V.; Haught, R.C.; Clark, R.M.; Rothenberg, G. Assessing the removal of inorganic colloids and Cryptosporidium parvum from drinking water. J. Environ. Monit. 2002, 4, 244–248. [Google Scholar] [CrossRef]
- Amirtharjah, A. Some Theoretical and Conceptual Views of Filtration. JAWWA 1988, 80, 36–46. [Google Scholar] [CrossRef]
- Edzwald, J.K.; Tobiason, J.; Parento, L.M.; Kelley, M.B.; Kaminski, G.; Dunn, H.J.; Galant, P.B. Giardia and Cryptosporidium Removals by clarification and filtration under challenge conditions. J. Am. Water Works Assoc. 2000, 92, 70–84. [Google Scholar] [CrossRef]
- Morse, A.; Wilson, R.; Hüll, A.; Simms, J. Monitoring for Cryptosporidium risk: Do particle counters help? Water Supply 2002, 2, 171–180. [Google Scholar] [CrossRef]
- Tufenkji, N.; Miller, G.F.; Ryan, J.N.; Harvey, R.; Elimelech, M. Transport of Cryptosporidium Oocysts in Porous Media: Role of Straining and Physicochemical Filtration†. Environ. Sci. Technol. 2004, 38, 5932–5938. [Google Scholar] [CrossRef]
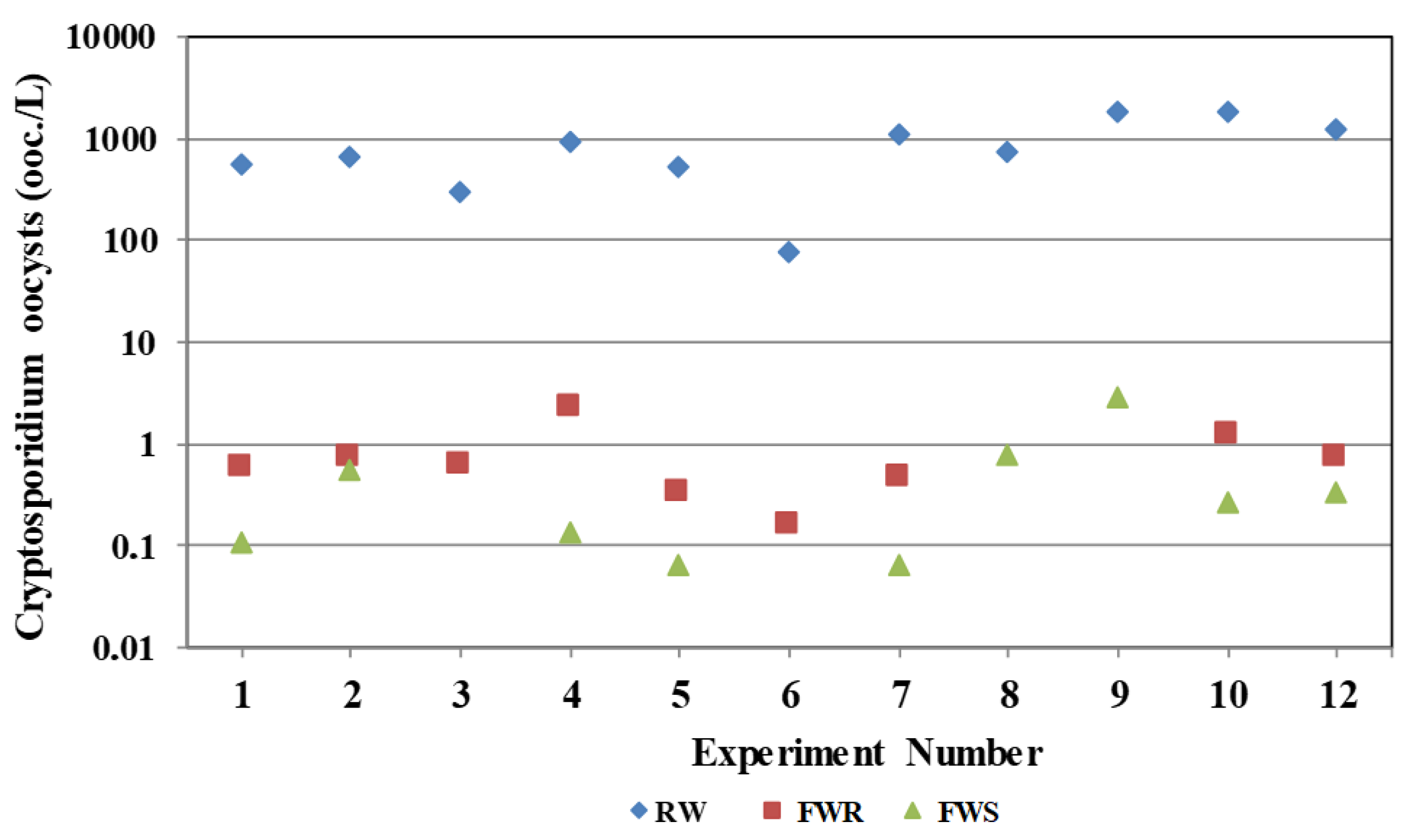

| Experiment Number | Turbidity | Oocysts RW (ooc./L) | Oocysts Removal (log) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Observations (N) | RW (NTU) | Residual-FW (NTU) | Removal (log) | ||||||
| Mean | Mean | SD | FWR | FWS | FWR | FWS | |||
| E1 | 15 | 4.8 | 0.27 | 0.14 | 1.20 | 1.24 | 565 | 2.98 | 3.70 |
| E2 | 17 | 5.7 | 0.28 | 0.14 | 1.15 | 1.31 | 674 | 2.96 | 3.10 |
| E3 | 17 | 5.4 | 0.32 | 0.06 | 1.06 | 1.22 | 292 | 2.62 | >4.20 |
| E4 | 17 | 4.4 | 0.31 | 0.11 | 1.25 | 1.15 | 902 | 2.60 | 3.80 |
| E5 | 17 | 5.0 | 0.27 | 0.03 | 1.16 | 1.26 | 522 | 3.19 | 3.90 |
| E6 | 17 | 3.5 | 0.25 | 0.04 | 1.05 | 1.14 | 76 | 2.68 | >4.20 |
| E7 | 16 | 5.4 | 0.21 | 0.03 | 1.15 | 1.40 | 1122 | 3.37 | 4.20 |
| E8 | 14 | 4.4 | 0.18 | 0.06 | 1.10 | 1.42 | 752 | (*) | 3.00 |
| E9 | 15 | 3.2 | 0.16 | 0.01 | 1.18 | 1.30 | 1864 | (*) | 2.80 |
| E10 | 18 | 28 | 0.32 | 0.03 | 1.81 | 1.93 | 1796 | 3.18 | 3.80 |
| E11 | 17 | 30 | 0.43 | 0.30 | 1.07 | 1.85 | (**) | (**) | (**) |
| E12 | 17 | 30 | 0.22 | 0.04 | 2.09 | 2.13 | 1246 | 3.24 | 3.60 |
| Oocysts | Particle Size Range (µm) | |||
| 2–7 | 4–6 | 8–15 | 20–70 | |
| Correlation Coefficient | 0.70 * | 0.70 * | 0.18 | 0.46 |
| Log Removal | ||||
| Oocysts | Particle Size Range (µm) | |||
| 2–7 | 4–6 | 8–15 | 20–70 | |
| Correlation Coefficient | −0.1 | 0.30 | 0.30 | 0.00 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nascimento, M.F.; Ginoris, Y.P.; Brandão, C.C.S. Cryptosporidium Oocysts Removal by Upflow Direct Filtration: Pilot Scale Assessment. Water 2020, 12, 1328. https://doi.org/10.3390/w12051328
Nascimento MF, Ginoris YP, Brandão CCS. Cryptosporidium Oocysts Removal by Upflow Direct Filtration: Pilot Scale Assessment. Water. 2020; 12(5):1328. https://doi.org/10.3390/w12051328
Chicago/Turabian StyleNascimento, Marcely Ferreira, Yovanka Pérez Ginoris, and Cristina Celia Silveira Brandão. 2020. "Cryptosporidium Oocysts Removal by Upflow Direct Filtration: Pilot Scale Assessment" Water 12, no. 5: 1328. https://doi.org/10.3390/w12051328
APA StyleNascimento, M. F., Ginoris, Y. P., & Brandão, C. C. S. (2020). Cryptosporidium Oocysts Removal by Upflow Direct Filtration: Pilot Scale Assessment. Water, 12(5), 1328. https://doi.org/10.3390/w12051328





