Perspectives on Micro(Nano)Plastics in the Marine Environment: Biological and Societal Considerations
Abstract
1. Marine Litter
2. Impacts of Marine Litter
2.1. Biological Impacts on Marine Organisms
Biological Models Used in Plastics Ecotoxicity Studies
2.2. Biological Impacts on Humans
2.3. Socio-Economic Impacts
3. Plastic Use in the COVID-19 Scenario—Additional Environmental Stress
4. Marine Litter Awareness and Environmental Education
5. Final Considerations
Author Contributions
Funding
Conflicts of Interest
References
- UNEP. Marine Debris: Understanding, Preventing and Mitigating the Significant Adverse Impacts on Marine and Coastal Biodiversity; Technical Series No. 83; Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2006; p. 78. [Google Scholar]
- Beeharry, Y.D.; Bekaroo, G.; Bokhoree, C.; Phillips, M.R.; Jory, N. Sustaining anti-littering behavior within coastal and marine environments: Through the macro-micro level lenses. Mar. Pollut. Bull. 2017, 119, 87–99. [Google Scholar] [CrossRef]
- Panti, C.; Baini, M.; Lusher, A.; Hernandez-Milan, G.; Rebolledo, E.L.B.; Unger, B.; Syberg, K.; Simmonds, M.P.; Fossi, M.C. Marine litter: One of the major threats for marine mammals. Outcomes from the European Cetacean Society workshop. Environ. Pollut. 2019, 247, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Strafella, P.; Fabi, G.; Despalatovic, M.; Cvitković, I.; Fortibuoni, T.; Gomiero, A.; Guicciardi, S.; Marceta, B.; Raicevich, S.; Tassetti, A.; et al. Assessment of seabed litter in the Northern and Central Adriatic Sea (Mediterranean) over six years. Mar. Pollut. Bull. 2019, 141, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Hartley, B.L.; Thompson, R.C.; Pahl, S. Marine litter education boosts children’s understanding and self-reported actions. Mar. Pollut. Bull. 2015, 90, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Bouwmeester, H.; Hollman, P.C.H.; Peters, R.J.B. Potential Health Impact of Environmentally Released Micro- and Nanoplastics in the Human Food Production Chain: Experiences from Nanotoxicology. Environ. Sci. Technol. 2015, 49, 8932–8947. [Google Scholar] [CrossRef] [PubMed]
- Torres, H.; Reynolds, C.J.; Lewis, A.; Muller-Karger, F.; Alsharif, K.; Mastenbrook, K. Examining youth perceptions and social contexts of litter to improve marine debris environmental education. Environ. Educ. Res. 2019, 25, 1400–1415. [Google Scholar] [CrossRef]
- Araújo, M.C.B.; Costa, M.F. A critical review of the issue of cigarette butt pollution in coastal environments. Environ. Res. 2019, 172, 137–149. [Google Scholar] [CrossRef]
- Peng, L.; Fu, D.; Qi, H.; Lan, C.Q.; Yu, H.; Ge, C. Micro- and nano-plastics in marine environment: Source, distribution and threats—A review. Sci. Total Environ. 2020, 698, 134254. [Google Scholar] [CrossRef]
- Curtis, C.; Novotny, T.E.; Lee, K.; Freiberg, M.; McLaughlin, I. Tobacco industry responsibility for butts: A Model Tobacco Waste Act. Tob. Control 2016, 26, 113–117. [Google Scholar] [CrossRef]
- Caridi, F.; Sabbatini, A.; Birarda, G.; Costanzi, E.; De Giudici, G.; Galeazzi, R.; Medas, D.; Mobbili, G.; Ricciutelli, M.; Ruello, M.L.; et al. Cigarette butts, a threat for marine environments: Lessons from benthic foraminifera (Protista). Mar. Environ. Res. 2020, 162, 105150. [Google Scholar] [CrossRef]
- Parker, T.T.; Rayburn, J. A comparison of electronic and traditional cigarette butt leachate on the development of Xenopus laevis embryos. Toxicol. Rep. 2017, 4, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Montalvão, M.F.; Chagas, T.Q.; Alvarez, T.G.D.S.; Mesak, C.; Araújo, A.P.D.C.; Gomes, A.R.; Vieira, J.E.D.A.; Rocha, T.L.; Malafaia, G. Cigarette butt leachate as a risk factor to the health of freshwater bivalve. Chemosphere 2019, 234, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, E.J.; Smith, K.L. Plastics on the Sargasso Sea Surface. Science 1972, 175, 1240–1241. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, E.J.; Anderson, S.J.; Harvey, G.R.; Miklas, H.P.; Peck, B.B. Polystyrene Spherules in Coastal Waters. Science 1972, 178, 749–750. [Google Scholar] [CrossRef]
- Brandão, M.L.; Braga, K.M.; Luque, J. Marine debris ingestion by Magellanic penguins, Spheniscus magellanicus (Aves: Sphenisciformes), from the Brazilian coastal zone. Mar. Pollut. Bull. 2011, 62, 2246–2249. [Google Scholar] [CrossRef]
- Thiel, M.; Luna-Jorquera, G.; Álvarez-Varas, R.; Gallardo, C.; Hinojosa, I.A.; Luna, N.; Miranda-Urbina, D.; Morales, N.; Ory, N.; Pacheco, A.S.; et al. Impacts of Marine Plastic Pollution From Continental Coasts to Subtropical Gyres—Fish, Seabirds, and Other Vertebrates in the SE Pacific. Front. Mar. Sci. 2018, 5. [Google Scholar] [CrossRef]
- Oliveira, M.; Almeida, M.; Miguel, I. A micro(nano)plastic boomerang tale: A never ending story? TrAC Trends Anal. Chem. 2019, 112, 196–200. [Google Scholar] [CrossRef]
- PlasticsEurope. Plastics—The Facts 2019—An Analysis of European Latest Plastics Production, Demand and Waste Data; PlasticsEurope: Brussels, Belgium, 2019. [Google Scholar]
- Lebreton, L.; Andrady, A. Future scenarios of global plastic waste generation and disposal. Palgrave Commun. 2019, 5, 6. [Google Scholar] [CrossRef]
- Oliveira, M.; Almeida, M. The why and how of micro(nano)plastic research. TrAC Trends Anal. Chem. 2019, 114, 196–201. [Google Scholar] [CrossRef]
- GESAMP. Sources, Fate and Effects of Microplastics in the Marine Environment: A Global Assessment; Reports and Studies 90; GESAMP: London, UK, 2015. [Google Scholar]
- Erni-Cassola, G.; Zadjelovic, V.; Gibson, M.I.; Christie-Oleza, J.A. Distribution of plastic polymer types in the marine environment; A meta-analysis. J. Hazard. Mater. 2019, 369, 691–698. [Google Scholar] [CrossRef]
- Stocchino, A.; De Leo, F.; Besio, G. Sea Waves Transport of Inertial Micro-Plastics: Mathematical Model and Applications. J. Mar. Sci. Eng. 2019, 7, 467. [Google Scholar] [CrossRef]
- Venâncio, C.; Ferreira, I.; Martins, M.A.; Soares, A.M.; Lopes, I.; Oliveira, M. The effects of nanoplastics on marine plankton: A case study with polymethylmethacrylate. Ecotoxicol. Environ. Saf. 2019, 184, 109632. [Google Scholar] [CrossRef] [PubMed]
- De Sá, L.C.; Oliveira, M.; Ribeiro, F.; Rocha, T.L.; Futter, M.N. Studies of the effects of microplastics on aquatic organisms: What do we know and where should we focus our efforts in the future? Sci. Total Environ. 2018, 645, 1029–1039. [Google Scholar] [CrossRef] [PubMed]
- Duis, K.; Coors, A. Microplastics in the aquatic and terrestrial environment: Sources (with a specific focus on personal care products), fate and effects. Environ. Sci. Eur. 2016, 28, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhang, Y.; Kang, S.; Wang, Z.; Wu, C. Microplastics in freshwater sediment: A review on methods, occurrence, and sources. Sci. Total Environ. 2021, 754, 141948. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Min, J.; Jiang, W.; Li, Y.; Zhang, W. Separation, characterization and identification of microplastics and nanoplastics in the environment. Sci. Total Environ. 2020, 721, 137561. [Google Scholar] [CrossRef] [PubMed]
- Mao, R.; Lang, M.; Yu, X.; Wu, R.; Yang, X.; Guo, X. Aging mechanism of microplastics with UV irradiation and its effects on the adsorption of heavy metals. J. Hazard. Mater. 2020, 393, 122515. [Google Scholar] [CrossRef]
- Almeida, M.; Martins, M.A.; Soares, A.M.V.M.; Cuesta, A.; Oliveira, M. Polystyrene nanoplastics alter the cytotoxicity of human pharmaceuticals on marine fish cell lines. Environ. Toxicol. Pharmacol. 2019, 69, 57–65. [Google Scholar] [CrossRef]
- Chen, X.; Chen, X.; Zhao, Y.; Zhou, H.; Xiong, X.; Wu, C. Effects of microplastic biofilms on nutrient cycling in simulated freshwater systems. Sci. Total Environ. 2020, 719, 137276. [Google Scholar] [CrossRef]
- Kühn, S.; Van Franeker, J.A. Quantitative overview of marine debris ingested by marine megafauna. Mar. Pollut. Bull. 2020, 151, 110858. [Google Scholar] [CrossRef]
- Abreo, N.A.S.; Thompson, K.F.; Arabejo, G.F.P.; Superio, M.D.A. Social media as a novel source of data on the impact of marine litter on megafauna: The Philippines as a case study. Mar. Pollut. Bull. 2019, 140, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Gall, S.; Thompson, R. The impact of debris on marine life. Mar. Pollut. Bull. 2015, 92, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Claro, F.; Fossi, M.; Ioakeimidis, C.; Baini, M.; Lusher, A.; Mc Fee, W.; McIntosh, R.; Pelamatti, T.; Sorce, M.; Galgani, F.; et al. Tools and constraints in monitoring interactions between marine litter and megafauna: Insights from case studies around the world. Mar. Pollut. Bull. 2019, 141, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Oliveira, M.; Lopéz, D.; Martins, M.; Figueira, E.; Pires, A. Do nanoplastics impact the ability of the polychaeta Hediste diversicolor to regenerate? Ecol. Indic. 2020, 110, 105921. [Google Scholar] [CrossRef]
- Silva, M.; Oliveira, M.; Valente, P.; Figueira, E.; Martins, M.; Pires, A. Behavior and biochemical responses of the polychaeta Hediste diversicolor to polystyrene nanoplastics. Sci. Total Environ. 2020, 707, 134434. [Google Scholar] [CrossRef]
- Zhu, C.; Li, D.; Sun, Y.-X.; Zheng, X.; Peng, X.; Zheng, K.; Hu, B.; Luo, X.; Mai, B. Plastic debris in marine birds from an island located in the South China Sea. Mar. Pollut. Bull. 2019, 149, 110566. [Google Scholar] [CrossRef]
- Garcia-Cegarra, A.M.; Ramirez, R.; Orrego, R. Red-legged cormorant uses plastic as nest material in an artificial breeding colony of Atacama Desert coast. Mar. Pollut. Bull. 2020, 160, 111632. [Google Scholar] [CrossRef]
- Rizzi, M.; Rodrigues, F.L.; Medeiros, L.; Ortega, I.; Rodrigues, L.; Monteiro, D.S.; Kessler, F.; Proietti, M.C. Ingestion of plastic marine litter by sea turtles in southern Brazil: Abundance, characteristics and potential selectivity. Mar. Pollut. Bull. 2019, 140, 536–548. [Google Scholar] [CrossRef]
- Barría, C.; Brandts, I.; Tort, L.; Oliveira, M.; Teles, M. Effect of nanoplastics on fish health and performance: A review. Mar. Pollut. Bull. 2020, 151, 110791. [Google Scholar] [CrossRef]
- Shen, M.; Ye, S.; Zeng, G.; Zhang, Y.; Xing, L.; Tang, W.; Wen, X.; Liu, S. Can microplastics pose a threat to ocean carbon sequestration? Mar. Pollut. Bull. 2020, 150, 110712. [Google Scholar] [CrossRef]
- Sjollema, S.B.; Redondo-Hasselerharm, P.E.; Leslie, H.; Kraak, M.H.; Vethaak, A.D. Do plastic particles affect microalgal photosynthesis and growth? Aquat. Toxicol. 2016, 170, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Coppock, R.L.; Galloway, T.S.; Cole, M.; Fileman, E.S.; Queirós, A.M.; Lindeque, P.K. Microplastics alter feeding selectivity and faecal density in the copepod, Calanus helgolandicus. Sci. Total Environ. 2019, 687, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Schür, C.; Zipp, S.; Thalau, T.; Wagner, M. Microplastics but not natural particles induce multigenerational effects in Daphnia magna. Environ. Pollut. 2020, 260, 113904. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, I.; Venâncio, C.; Lopes, I.; Oliveira, M. Nanoplastics and marine organisms: What has been studied? Environ. Toxicol. Pharmacol. 2019, 67, 1–7. [Google Scholar] [CrossRef]
- Chae, Y.; An, Y.-J. Effects of food presence on microplastic ingestion and egestion in Mytilus galloprovincialis. Chemosphere 2020, 240, 124855. [Google Scholar] [CrossRef]
- Kazour, M.; Amara, R. Is blue mussel caging an efficient method for monitoring environmental microplastics pollution? Sci. Total Environ. 2020, 710, 135649. [Google Scholar] [CrossRef]
- Brandts, I.; Teles, M.; Gonçalves, A.; Barreto, A.; Franco-Martinez, L.; Tvarijonaviciute, A.; Martins, M.; Soares, A.M.V.M.; Tort, L.; Oliveira, M. Effects of nanoplastics on Mytilus galloprovincialis after individual and combined exposure with carbamazepine. Sci. Total Environ. 2018, 643, 775–784. [Google Scholar] [CrossRef]
- Pedersen, A.F.; Gopalakrishnan, K.; Boegehold, A.G.; Peraino, N.J.; Westrick, J.A.; Kashian, D.R. Microplastic ingestion by quagga mussels, Dreissena bugensis, and its effects on physiological processes. Environ. Pollut. 2020, 260, 113964. [Google Scholar] [CrossRef]
- Fernández-Galindo, B.; Albentosa, M. Insights into the uptake, elimination and accumulation of microplastics in mussel. Environ. Pollut. 2019, 249, 321–329. [Google Scholar] [CrossRef]
- Browne, M.A.; Dissanayake, A.; Galloway, T.S.; Lowe, D.M.; Thompson, R.C. Ingested Microscopic Plastic Translocates to the Circulatory System of the Mussel, Mytilus edulis (L.). Environ. Sci. Technol. 2008, 42, 5026–5031. [Google Scholar] [CrossRef]
- Woods, M.N.; Stack, M.E.; Fields, D.M.; Shaw, S.D.; Matrai, P.A. Microplastic fiber uptake, ingestion, and egestion rates in the blue mussel (Mytilus edulis). Mar. Pollut. Bull. 2018, 137, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Kinjo, A.; Mizukawa, K.; Takada, H.; Inoue, K. Size-dependent elimination of ingested microplastics in the Mediterranean mussel Mytilus galloprovincialis. Mar. Pollut. Bull. 2019, 149, 110512. [Google Scholar] [CrossRef] [PubMed]
- Knutsen, H.; Cyvin, J.B.; Totland, C.; Lilleeng, Ø.; Wade, E.J.; Castro, V.; Pettersen, A.; Laugesen, J.; Møskeland, T.; Arp, H.P.H. Microplastic accumulation by tube-dwelling, suspension feeding polychaetes from the sediment surface: A case study from the Norwegian Continental Shelf. Mar. Environ. Res. 2020, 161, 105073. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Pires, A.; Almeida, M.; Oliveira, M. The use of Hediste diversicolor in the study of emerging contaminants. Mar. Environ. Res. 2020, 159, 105013. [Google Scholar] [CrossRef] [PubMed]
- Revel, M.; Yakovenko, N.; Caley, T.; Guillet, C.; Châtel, A.; Mouneyrac, C. Accumulation and immunotoxicity of microplastics in the estuarine worm Hediste diversicolor in environmentally relevant conditions of exposure. Environ. Sci. Pollut. Res. 2018, 27, 3574–3583. [Google Scholar] [CrossRef]
- Leung, J.; Chan, K.Y.K. Microplastics reduced posterior segment regeneration rate of the polychaete Perinereis aibuhitensis. Mar. Pollut. Bull. 2018, 129, 782–786. [Google Scholar] [CrossRef]
- Piazzolla, D.; Cafaro, V.; Mancini, E.; Scanu, S.; Bonamano, S.; Marcelli, M. Preliminary Investigation of Microlitter Pollution in Low-Energy Hydrodynamic Basins Using Sabella spallanzanii (Polychaeta Sabellidae) Tubes. Bull. Environ. Contam. Toxicol. 2020, 104, 345–350. [Google Scholar] [CrossRef]
- Nel, H.A.; Froneman, P. Presence of microplastics in the tube structure of the reef-building polychaete Gunnarea gaimardi (Quatrefages 1848). Afr. J. Mar. Sci. 2018, 40, 87–89. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Metian, M. Food Matters: Fish, Income, and Food Supply—A Comparative Analysis. Rev. Fish. Sci. Aquac. 2017, 26, 15–28. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Raimundo, J.; Oliveira, P.; Bessa, F.; Henriques, B.; Caetano, M.; Guilhermino, L.; Caetano, M.; Vale, C.; Guilhermino, L. Microplastics in wild fish from North East Atlantic Ocean and its potential for causing neurotoxic effects, lipid oxidative damage, and human health risks associated with ingestion exposure. Sci. Total Environ. 2020, 717, 134625. [Google Scholar] [CrossRef]
- Li, B.; Su, L.; Zhang, H.; Deng, H.; Chen, Q.; Shi, H. Microplastics in fishes and their living environments surrounding a plastic production area. Sci. Total Environ. 2020, 727, 138662. [Google Scholar] [CrossRef] [PubMed]
- Brandts, I.; Teles, M.; Tvarijonaviciute, A.; Pereira, M.; Martins, M.; Tort, L.; Oliveira, M. Effects of polymethylmethacrylate nanoplastics on Dicentrarchus labrax. Genomics 2018, 110, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Brandts, I.; Barría, C.; Martins, M.A.; Franco-Martínez, L.; Barreto, A.; Tvarijonaviciute, A.; Tort, L.; Oliveira, M.; Teles, M. Waterborne exposure of gilthead seabream (Sparus aurata) to polymethylmethacrylate nanoplastics causes effects at cellular and molecular levels. J. Hazard. Mater. 2021, 403, 123590. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.C.L.; Ribeiro, A.L.P.; Hylland, K.; Guilhermino, L. Single and combined effects of microplastics and pyrene on juveniles (0+ group) of the common goby Pomatoschistus microps (Teleostei, Gobiidae). Ecol. Indic. 2013, 34, 641–647. [Google Scholar] [CrossRef]
- Pedà, C.; Caccamo, L.; Fossi, M.C.; Gai, F.; Andaloro, F.; Genovese, L.; Perdichizzi, A.; Romeo, T.; Maricchiolo, G. Intestinal alterations in European sea bass Dicentrarchus labrax (Linnaeus, 1758) exposed to microplastics: Preliminary results. Environ. Pollut. 2016, 212, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Chen, B.; Xia, B.; Shi, X.; Qu, K. Polystyrene microplastics alter the behavior, energy reserve and nutritional composition of marine jacopever (Sebastes schlegelii). J. Hazard. Mater. 2018, 360, 97–105. [Google Scholar] [CrossRef]
- Luís, L.G.; Ferreira, P.; Fonte, E.; Oliveira, M.; Guilhermino, L. Does the presence of microplastics influence the acute toxicity of chromium(VI) to early juveniles of the common goby (Pomatoschistus microps)? A study with juveniles from two wild estuarine populations. Aquat. Toxicol. 2015, 164, 163–174. [Google Scholar] [CrossRef]
- Iliff, S.M.; Wilczek, E.R.; Harris, R.J.; Bouldin, R.; Stoner, E.W. Evidence of microplastics from benthic jellyfish (Cassiopea xamachana) in Florida estuaries. Mar. Pollut. Bull. 2020, 159, 111521. [Google Scholar] [CrossRef]
- Sucharitakul, P.; Pitt, K.A.; Welsh, D.T. Limited ingestion, rapid egestion and no detectable impacts of microbeads on the moon jellyfish, Aurelia aurita. Mar. Pollut. Bull. 2020, 156, 111208. [Google Scholar] [CrossRef]
- Macali, A.; Semenov, A.; Venuti, V.; Crupi, V.; D’Amico, F.; Rossi, B.; Corsi, I.; Bergami, E. Episodic records of jellyfish ingestion of plastic items reveal a novel pathway for trophic transference of marine litter. Sci. Rep. 2018, 8, 1–5. [Google Scholar] [CrossRef]
- Costa, E.; Gambardella, C.; Piazza, V.; Vassalli, M.; Sbrana, F.; Lavorano, S.; Garaventa, F.; Faimali, M. Microplastics ingestion in the ephyra stage of Aurelia sp. triggers acute and behavioral responses. Ecotoxicol. Environ. Saf. 2020, 189, 109983. [Google Scholar] [CrossRef] [PubMed]
- Nelms, S.E.; Barnett, J.; Brownlow, A.; Davison, N.J.; Deaville, R.; Galloway, T.S.; Lindeque, P.K.; Santillo, D.; Godley, B.J. Microplastics in marine mammals stranded around the British coast: Ubiquitous but transitory? Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Basto, M.N.; Nicastro, K.R.; Tavares, A.I.; McQuaid, C.D.; Casero, M.; Azevedo, F.; Zardi, G.I. Plastic ingestion in aquatic birds in Portugal. Mar. Pollut. Bull. 2019, 138, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.G. Entanglement of birds in plastics and other synthetic materials. Mar. Pollut. Bull. 2018, 135, 159–164. [Google Scholar] [CrossRef]
- Akhbarizadeh, R.; Moore, F.; Keshavarzi, B. Investigating microplastics bioaccumulation and biomagnification in seafood from the Persian Gulf: A threat to human health? Food Addit. Contam. Part A 2019, 36, 1696–1708. [Google Scholar] [CrossRef]
- Peixoto, D.; Pinheiro, C.; Amorim, J.; Oliva-Teles, L.; Guilhermino, L.; Vieira, M.N. Microplastic pollution in commercial salt for human consumption: A review. Estuar. Coast. Shelf Sci. 2019, 219, 161–168. [Google Scholar] [CrossRef]
- Kosuth, M.; Mason, S.A.; Wattenberg, E.V. Anthropogenic contamination of tap water, beer, and sea salt. PLoS ONE 2018, 13, e0194970. [Google Scholar] [CrossRef]
- Mason, S.A.; Welch, V.G.; Neratko, J. Synthetic Polymer Contamination in Bottled Water. Front. Chem. 2018, 6, 407. [Google Scholar] [CrossRef]
- Li, J.; Qu, X.; Su, L.; Zhang, W.; Yang, D.; Kolandhasamy, P.; Li, D.; Shi, H. Microplastics in mussels along the coastal waters of China. Environ. Pollut. 2016, 214, 177–184. [Google Scholar] [CrossRef]
- Wakkaf, T.; El Zrelli, R.; Kedzierski, M.; Balti, R.; Shaiek, M.; Mansour, L.; Tlig-Zouari, S.; Bruzaud, S.; Rabaoui, L. Microplastics in edible mussels from a southern Mediterranean lagoon: Preliminary results on seawater-mussel transfer and implications for environmental protection and seafood safety. Mar. Pollut. Bull. 2020, 158, 111355. [Google Scholar] [CrossRef]
- Smith, M.; Love, D.C.; Rochman, C.M.; Neff, R.A. Microplastics in Seafood and the Implications for Human Health. Curr. Environ. Health Rep. 2018, 5, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Davidson, K.; Dudas, S.E. Microplastic Ingestion by Wild and Cultured Manila Clams (Venerupis philippinarum) from Baynes Sound, British Columbia. Arch. Environ. Contam. Toxicol. 2016, 71, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Rochman, C.M.; Tahir, A.; Williams, S.L.; Baxa, D.V.; Lam, R.; Miller, J.T.; Teh, F.-C.; Werorilangi, S.; Teh, S.J. Anthropogenic debris in seafood: Plastic debris and fibers from textiles in fish and bivalves sold for human consumption. Sci. Rep. 2015, 5, 14340. [Google Scholar] [CrossRef] [PubMed]
- Stock, V.; Böhmert, L.; Lisicki, E.; Block, R.; Cara-Carmona, J.; Pack, L.K.; Selb, R.; Lichtenstein, D.; Voss, L.; Henderson, C.J.; et al. Uptake and effects of orally ingested polystyrene microplastic particles in vitro and in vivo. Arch. Toxicol. 2019, 93, 1817–1833. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.-D.; Chen, C.-W.; Chen, Y.-C.; Chen, H.-H.; Lee, J.-S.; Lin, C.-H. Polystyrene microplastic particles: In vitro pulmonary toxicity assessment. J. Hazard. Mater. 2020, 385, 121575. [Google Scholar] [CrossRef] [PubMed]
- Hartley, B.L.; Pahl, S.; Veiga, J.; Vlachogianni, T.; Vasconcelos, L.; Maes, T.; Doyle, T.; Metcalfe, R.D.; Öztürk, A.A.; Di Berardo, M.; et al. Exploring public views on marine litter in Europe: Perceived causes, consequences and pathways to change. Mar. Pollut. Bull. 2018, 133, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Krelling, A.P.; Williams, A.T.; Turra, A. Differences in perception and reaction of tourist groups to beach marine debris that can influence a loss of tourism revenue in coastal areas. Mar. Policy 2017, 85, 87–99. [Google Scholar] [CrossRef]
- Muhammad, S.; Long, X.; Salman, M. COVID-19 pandemic and environmental pollution: A blessing in disguise? Sci. Total Environ. 2020, 728, 138820. [Google Scholar] [CrossRef]
- Chakraborty, I.; Maity, P. COVID-19 outbreak: Migration, effects on society, global environment and prevention. Sci. Total Environ. 2020, 728, 138882. [Google Scholar] [CrossRef]
- Zambrano-Monserrate, M.A.; Ruano, M.A.; Sanchez-Alcalde, L. Indirect effects of COVID-19 on the environment. Sci. Total Environ. 2020, 728, 138813. [Google Scholar] [CrossRef]
- Vanapalli, K.R.; Sharma, H.B.; Ranjan, V.P.; Samal, B.; Bhattacharya, J.; Dubey, B.K.; Goel, S. Challenges and strategies for effective plastic waste management during and post COVID-19 pandemic. Sci. Total Environ. 2021, 750, 141514. [Google Scholar] [CrossRef] [PubMed]
- Canning-Clode, J.; Sepúlveda, P.; Almeida, S.; Monteiro, J. Will COVID-19 Containment and Treatment Measures Drive Shifts in Marine Litter Pollution? Front. Mar. Sci. 2020, 7. [Google Scholar] [CrossRef]
- Pahl, S.; Wyles, K.J. The human dimension: How social and behavioural research methods can help address microplastics in the environment. Anal. Methods 2017, 9, 1404–1411. [Google Scholar] [CrossRef]
- Henderson, L.; Green, C. Making sense of microplastics? Public understandings of plastic pollution. Mar. Pollut. Bull. 2020, 152, 110908. [Google Scholar] [CrossRef] [PubMed]
- Kusumawati, I.; Setyowati, M.; Syakti, A.D.; Fahrudin, A. Enhancing Millennial Awareness Towards Marine Litter Through Environmental Education. E3S Web Conf. 2020, 147, 02019. [Google Scholar] [CrossRef]
- Ashley, M.; Pahl, S.; Glegg, G.; Fletcher, S. A Change of Mind: Applying Social and Behavioral Research Methods to the Assessment of the Effectiveness of Ocean Literacy Initiatives. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Rambonnet, L.; Vink, S.C.; Land-Zandstra, A.M.; Bosker, T. Making citizen science count: Best practices and challenges of citizen science projects on plastics in aquatic environments. Mar. Pollut. Bull. 2019, 145, 271–277. [Google Scholar] [CrossRef]
- Bergmann, M.; Lutz, B.; Tekman, M.B.; Gutow, L. Citizen scientists reveal: Marine litter pollutes Arctic beaches and affects wild life. Mar. Pollut. Bull. 2017, 125, 535–540. [Google Scholar] [CrossRef]
- Tabuenca, B.; Kalz, M.; Löhr, A.J. Massive Open Online Education for Environmental Activism: The Worldwide Problem of Marine Litter. Sustainability 2019, 11, 2860. [Google Scholar] [CrossRef]
- Locritani, M.; Merlino, S.; Abbate, M. Assessing the citizen science approach as tool to increase awareness on the marine litter problem. Mar. Pollut. Bull. 2019, 140, 320–329. [Google Scholar] [CrossRef]
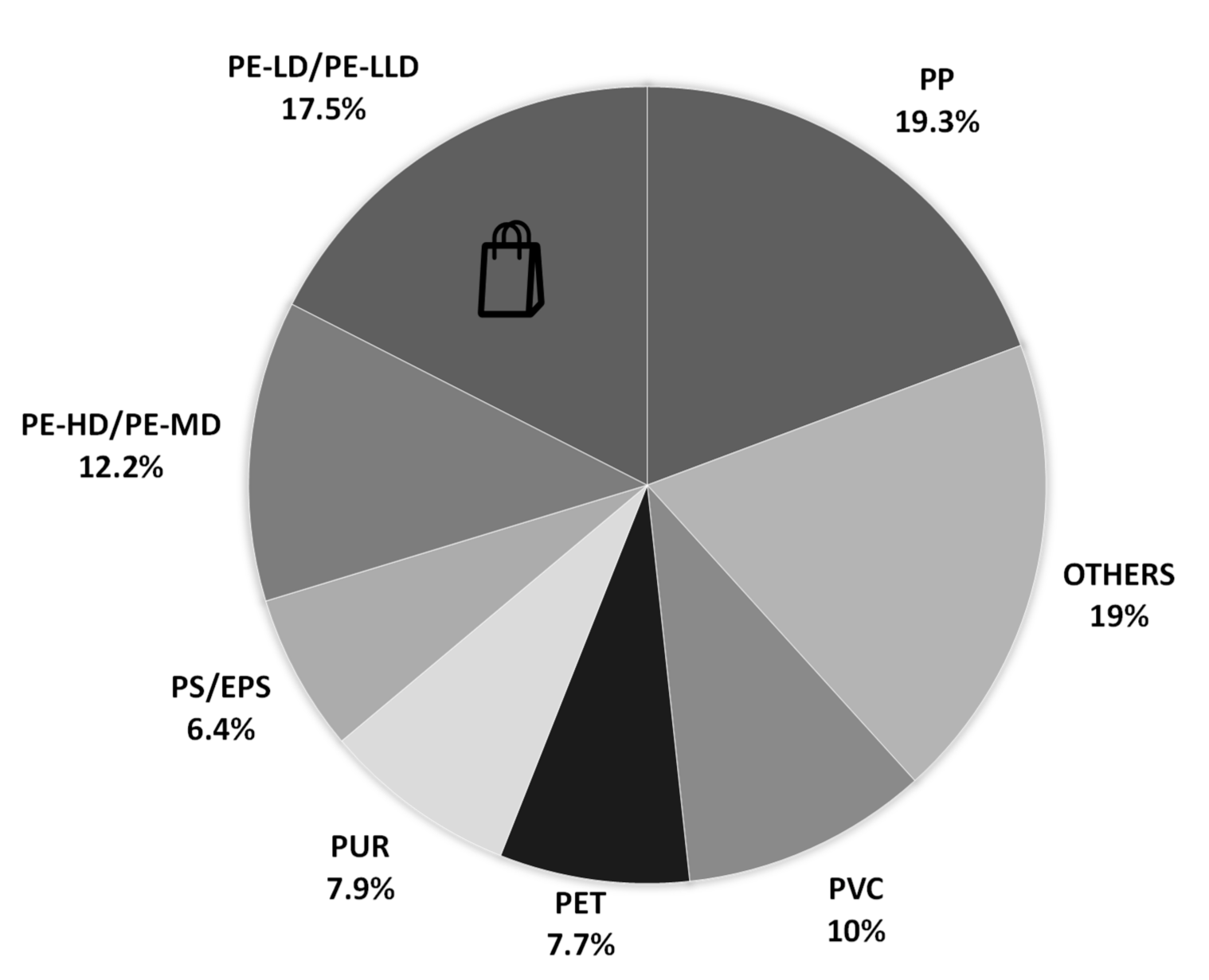
 —recycling;
—recycling; —energy recovery,
—energy recovery, —landfill. Note: Data for the construction of the graphic were collected at https://www.plasticseurope.org/, published in 2019.
—landfill. Note: Data for the construction of the graphic were collected at https://www.plasticseurope.org/, published in 2019.
 —recycling;
—recycling; —energy recovery,
—energy recovery, —landfill. Note: Data for the construction of the graphic were collected at https://www.plasticseurope.org/, published in 2019.
—landfill. Note: Data for the construction of the graphic were collected at https://www.plasticseurope.org/, published in 2019.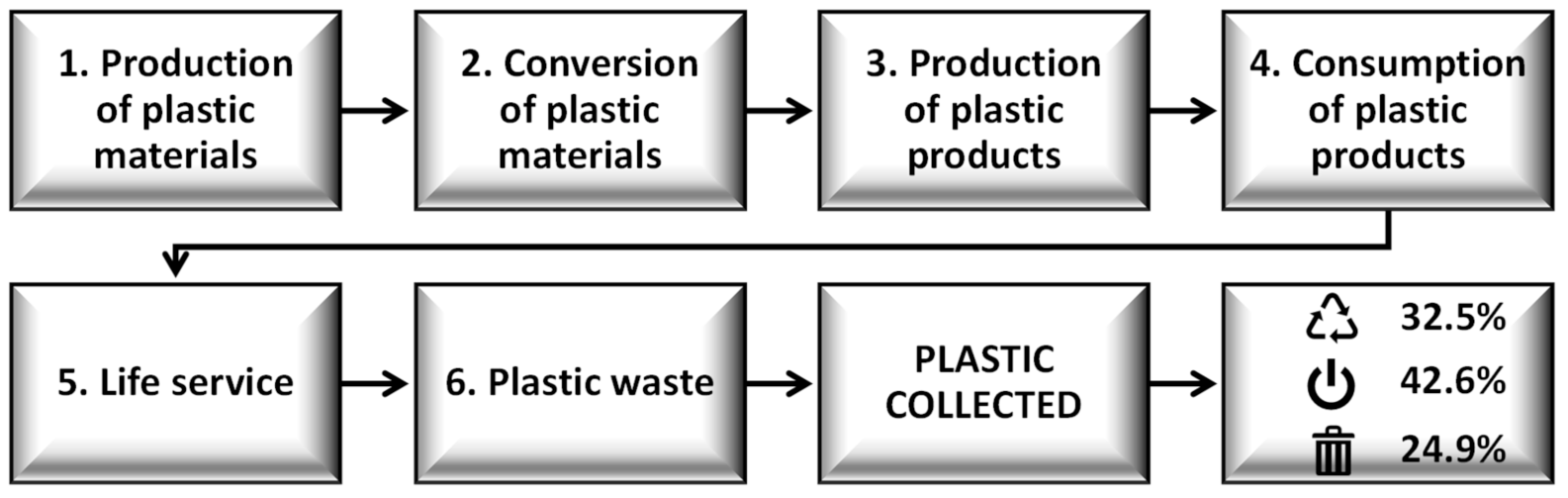
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soares, J.; Miguel, I.; Venâncio, C.; Lopes, I.; Oliveira, M. Perspectives on Micro(Nano)Plastics in the Marine Environment: Biological and Societal Considerations. Water 2020, 12, 3208. https://doi.org/10.3390/w12113208
Soares J, Miguel I, Venâncio C, Lopes I, Oliveira M. Perspectives on Micro(Nano)Plastics in the Marine Environment: Biological and Societal Considerations. Water. 2020; 12(11):3208. https://doi.org/10.3390/w12113208
Chicago/Turabian StyleSoares, Joana, Isabel Miguel, Cátia Venâncio, Isabel Lopes, and Miguel Oliveira. 2020. "Perspectives on Micro(Nano)Plastics in the Marine Environment: Biological and Societal Considerations" Water 12, no. 11: 3208. https://doi.org/10.3390/w12113208
APA StyleSoares, J., Miguel, I., Venâncio, C., Lopes, I., & Oliveira, M. (2020). Perspectives on Micro(Nano)Plastics in the Marine Environment: Biological and Societal Considerations. Water, 12(11), 3208. https://doi.org/10.3390/w12113208






