Impacts of Extreme Weather Events on Bacterial Community Composition of a Temperate Humic Lake
Abstract
1. Introduction
2. Materials and Methods
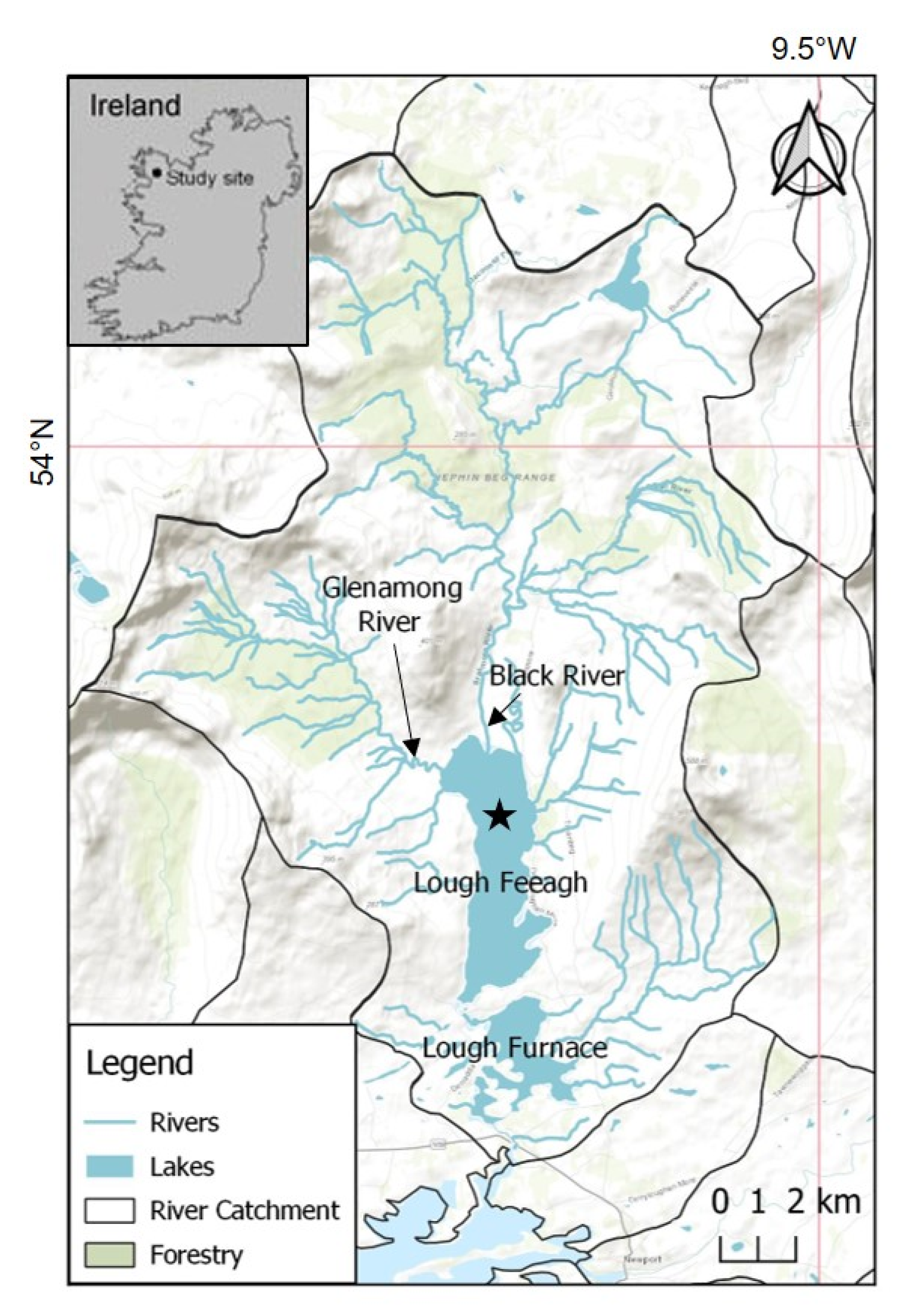
2.1. Site Description and High Frequency Monitoring
2.2. Sample Collection
2.3. Chlorophyll a, Nutrient Analyses, and Schmidt Stability
2.4. Filtration, DNA Extraction, and Sequencing
2.5. Bioinformatics and Statistical Analyses
3. Results
3.1. Weather Event Characteristics
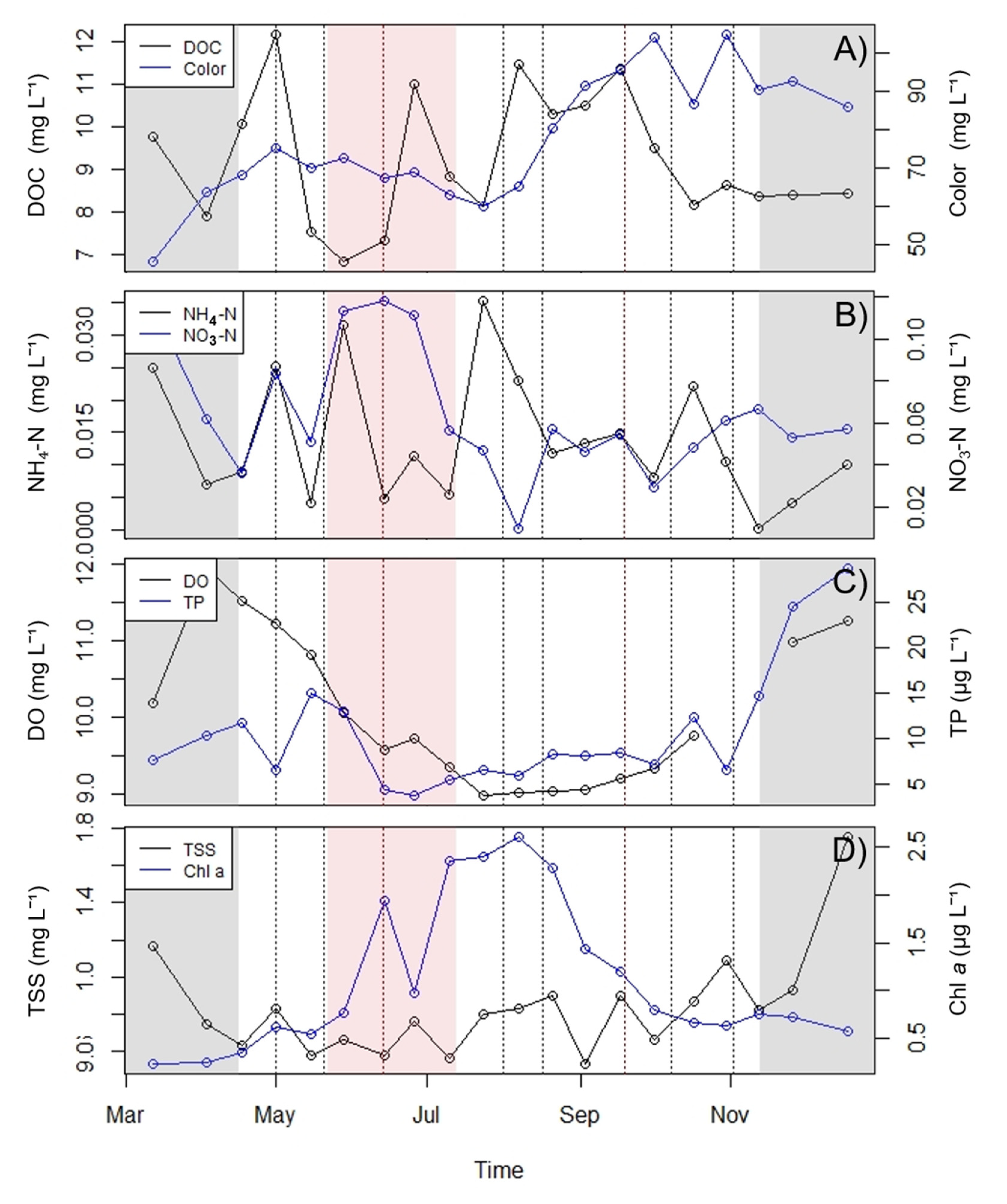
3.2. Nutrient Dynamics
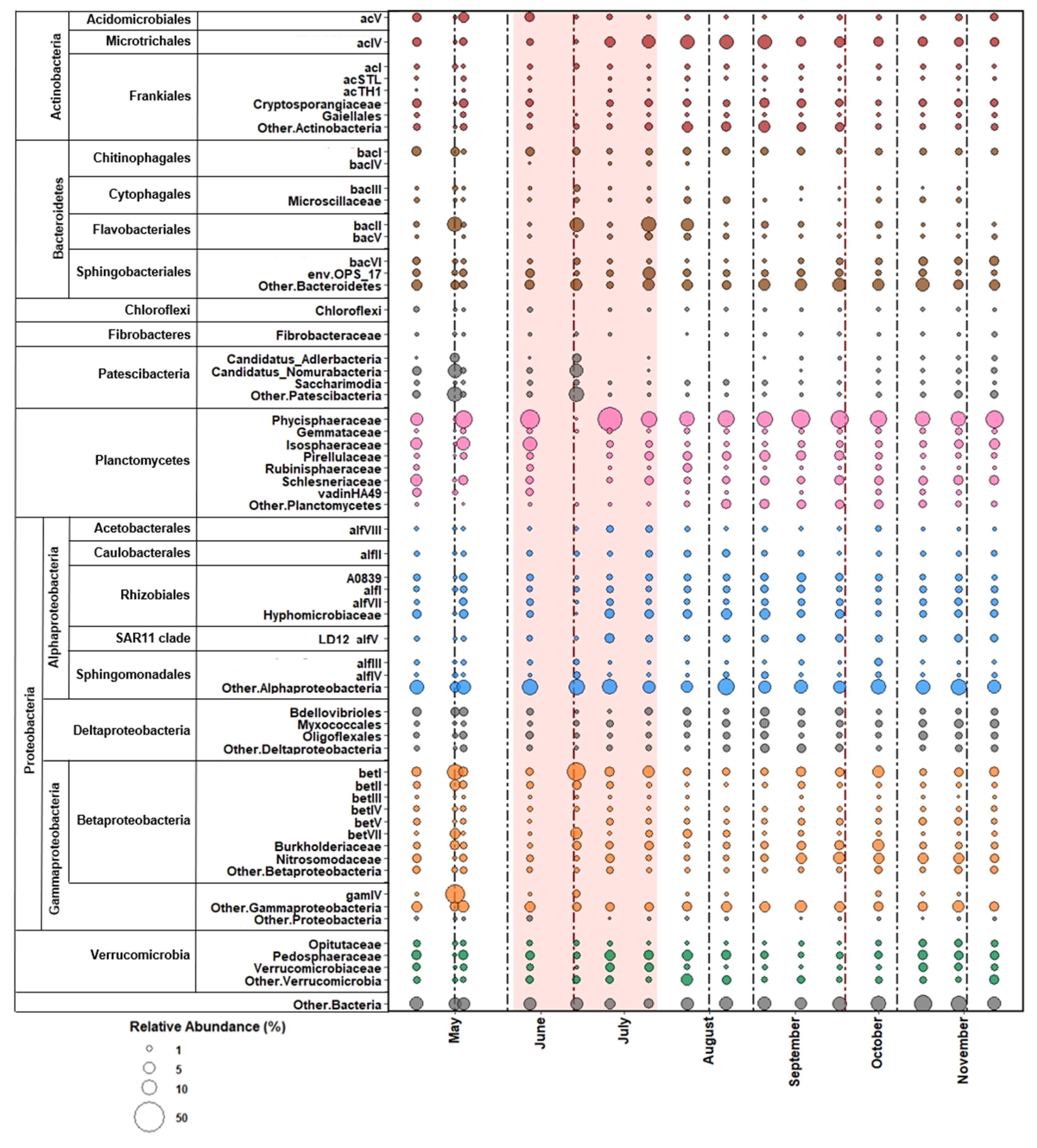
3.3. Bacterial Community Composition
3.3.1. Free-Living
3.3.2. Particle-Associated
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, S.; McGrath, R.; Hanafin, J.; Lynch, P.; Semmler, T.; Nolan, P. The impact of climate change on storm surges over Irish waters. Ocean Model. Online 2008, 25, 83–94. [Google Scholar] [CrossRef]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Frieler, K.; Lange, S.; Piontek, F.; Reyer, C.P.; Schewe, J.; Warszawski, L.; Zhao, F.; Chini, L.; Denvil, S.; Emanuel, K.; et al. Assessing the impacts of 1.5 °C global warming—Simulation protocol of the Inter-Sectoral Impact Model Intercomparison Project (ISIMIP2b). Geosci. Model. Dev. 2017, 10, 4321–4345. [Google Scholar] [CrossRef]
- Stockwell, J.D.; Doubek, J.P.; Adrian, R.; Anneville, O.; Carey, C.C.; Carvalho, L.; Domis, L.N.D.S.; Dur, G.; Frassl, M.A.; Grossart, H.; et al. Storm impacts on phytoplankton community dynamics in lakes. Glob. Change Biol. 2020, 26, 2756–2784. [Google Scholar] [CrossRef] [PubMed]
- Seneviratne, S.I.; Nicholls, N.; Easterling, D.; Goodess, C.M.; Kanae, S.; Kossin, J.; Luo, Y.; Marengo, J.; McInnes, K.; Rahimi, M.; et al. Changes in climate extremes and their impacts on the natural physical environment. In Managing the Risks of Extreme Events and Disasters to Advance Climate Change Adaptation. A Special Report of Working Groups I and II of the Intergovernmental Panel on Climate Change (IPCC); Field, C.B., Barros, V., Stocker, T.F., Qin, D., Dokken, D.J., Ebi, K.L., Mastrandrea, M.D., Mach, K.J., Plattner, G.K., Allen, et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2012; pp. 109–230. [Google Scholar]
- Jennings, E.; Jones, S.; Arvola, L.; Staehr, P.A.; Gaiser, E.; Jones, I.D.; Weathers, K.C.; Weyhenmeyer, G.A.; Chiu, C.Y.; De Eyto, E. Effects of weather-related episodic events in lakes: An analysis based on high-frequency data. Freshw. Biol. 2012, 57, 589–601. [Google Scholar] [CrossRef]
- Kasprzak, P.; Shatwell, T.; Gessner, M.O.; Gonsiorczyk, T.; Kirillin, G.; Selmeczy, G.; Padisák, J.; Engelhardt, C. Extreme weather event triggers cascade towards extreme turbidity in a clear-water lake. Ecosystems 2017, 20, 1407–1420. [Google Scholar] [CrossRef]
- Weyhenmeyer, G.E.; Willén, E.; Sonesten, L. Effects of an extreme precipitation event on water chemistry and phytoplankton in the Swedish Lake Mälaren. Boreal Env. Res. 2004, 9, 409–420. [Google Scholar]
- Sadro, S.; Melack, J.M. The effect of an extreme rain event on the biogeochemistry and ecosystem metabolism of an oligotrophic high-elevation lake. Arctic Antarct. Alp. Res. 2012, 44, 222–231. [Google Scholar] [CrossRef]
- Tranvik, L.J.; Downing, J.A.; Cotner, J.B.; Loiselle, S.A.; Striegl, R.G.; Ballatore, T.J.; Dillon, P.; Finlay, K.; Fortino, K.; Knoll, L.B. Lakes and reservoirs as regulators of carbon cycling and climate. Limnol. Oceanogr. 2009, 54, 2298–2314. [Google Scholar] [CrossRef]
- Azam, F.; Fenchel, T.; Field, J.; Gray, J.; Meyer-Reil, L.; Thingstad, T. The ecological role of water-column microbes in the sea. Mar. Ecol. Prog. Ser. 1983, 10, 257–263. [Google Scholar] [CrossRef]
- Foreman, C.M.; Wolf, C.F.; Priscu, J.C. Impact of episodic warming events on the physical, chemical and biological relationships of lakes in the McMurdo Dry Valleys. Antarct. Aquat. Geochem. 2004, 10, 239–268. [Google Scholar] [CrossRef]
- De Eyto, E.; Jennings, E.; Ryder, E.; Sparber, K.; Dillane, M.; Dalton, C.; Poole, R. Response of a humic lake ecosystem to an extreme precipitation event: Physical, chemical, and biological implications. Inland Waters 2016, 6, 483–498. [Google Scholar] [CrossRef]
- Jones, S.E.; Chiu, C.Y.; Kratz, T.K.; Wu, J.T.; Shade, A.; McMahon, K.D. Typhoons initiate predictable change in aquatic bacterial communities. Limnol. Oceanogr. 2008, 53, 1319–1326. [Google Scholar] [CrossRef]
- Garcia, S.L.; Salka, I.; Grossart, H.P.; Warnecke, F. Depth-discrete profiles of bacterial communities reveal pronounced spatio-temporal dynamics related to lake stratification. Environ. Microbiol. Rep. 2013, 5, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Shade, A.; Chiu, C.Y.; McMahon, K.D. Seasonal and episodic lake mixing stimulate differential planktonic bacterial dynamics. Microb. Ecol. 2009, 59, 546–554. [Google Scholar] [CrossRef]
- Yu, Z.C. Northern peatland carbon stocks and dynamics: A review. Biogeosciences 2012, 9, 5073–5107. [Google Scholar] [CrossRef]
- Wilson, R.M.; Hopple, A.M.; Tfaily, M.M.; Sebestyen, S.D.; Schadt, C.W.; Pfeifer-Meister, L.; Medvedeff, C.; McFarlane, K.J.; Kostka, J.E.; Kolton, M.; et al. Stability of peatland carbon to rising temperatures. Nat. Commun. 2016, 7, 1–10. [Google Scholar] [CrossRef]
- Ryder, E.; De Eyto, E.; Dillane, M.; Poole, R.; Jennings, E. Identifying the role of environmental drivers in organic carbon export from a forested peat catchment. Sci. Total. Environ. 2014, 490, 20–36. [Google Scholar] [CrossRef]
- Newton, R.J.; Kent, A.D.; Triplett, E.W.; McMahon, K.D. Microbial community dynamics in a humic lake: Differential persistence of common freshwater phylotypes. Environ. Microbiol. 2006, 8, 956–970. [Google Scholar] [CrossRef]
- Kent, A.D.; Yannarell, A.C.; Rusak, J.A.; Triplett, E.W.; McMahon, K.D. Synchrony in aquatic microbial community dynamics. ISME J. 2007, 1, 38–47. [Google Scholar] [CrossRef]
- Grossart, H.P.; Jezbera, J.; Horňák, K.; Hutalle, K.M.L.; Buck, U.; Šimek, K. Top-down and bottom-up induced shifts in bacterial abundance, production and community composition in an experimentally divided humic lake. Environ. Microbiol. 2008, 10, 635–652. [Google Scholar] [CrossRef] [PubMed]
- Linz, A.M.; Crary, B.C.; Shade, A.; Owens, S.; Gilbert, J.A.; Knight, R.; McMahon, K.D. Bacterial community composition and dynamics spanning five years in freshwater bog lakes. mSphere 2017, 2, e00169-17. [Google Scholar] [CrossRef] [PubMed]
- Sparber, K.; Dalton, C.; de Eyto, E.; Jennings, E.; Lenihan, D.; Cassina, F. Contrasting pelagic plankton in temperate Irish lakes: The relative contribution of heterotrophic, mixotrophic, and autotrophic components, and the effects of extreme rainfall events. Inland Waters 2015, 5, 295–310. [Google Scholar] [CrossRef]
- Allgaier, M.; Grossart, H.P. Seasonal dynamics and phylogenetic diversity of free-living and particle-associated bacterial communities in four lakes in northeastern Germany. Aquat. Microb. Ecol. 2006, 45, 115–128. [Google Scholar] [CrossRef]
- Rösel, S.; Allgaier, M.; Grossart, H.P. Long-term characterization of free-living and particle-associated bacterial communities in lake tiefwaren reveals distinct seasonal patterns. Microb. Ecol. 2012, 64, 571–583. [Google Scholar] [CrossRef]
- Grossart, H.P. Ecological consequences of bacterioplankton lifestyles: Changes in concepts are needed. Environ. Microbiol. Rep. 2010, 2, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Simon, M.; Grossart, H.; Schweitzer, B.; Ploug, H. Microbial ecology of organic aggregates in aquatic ecosystems. Aquat. Microb. Ecol. 2002, 28, 175–211. [Google Scholar] [CrossRef]
- Rösel, S.; Rychła, A.; Wurzbacher, C.; Grossart, H.-P. Effects of pollen leaching and microbial degradation on organic carbon and nutrient availability in lake water. Aquat. Sci. 2011, 74, 87–99. [Google Scholar] [CrossRef]
- Smith, D.C.; Simon, M.; Alldredge, A.L.; Azam, F. Intense hydrolytic enzyme activity on marine aggregates and implications for rapid particle dissolution. Nature 1992, 359, 139–142. [Google Scholar] [CrossRef]
- Andersen, M.R.; De Eyto, E.; Dillane, M.; Poole, R.; Jennings, E. 13 years of storms: An analysis of the effects of storms on lake physics on the atlantic fringe of Europe. Water 2020, 12, 318. [Google Scholar] [CrossRef]
- Perga, M.-E.; Bruel, R.; Rodriguez, L.; Guénand, Y.; Bouffard, D. Storm impacts on alpine lakes: Antecedent weather conditions matter more than the event intensity. Glob. Change Biol. 2018, 24, 5004–5016. [Google Scholar] [CrossRef] [PubMed]
- Fealy, R.M.; Buckley, C.; Mechan, S.; Melland, A.; Mellander, P.E.; Shortle, G.; Wall, D.; Jordan, P. The Irish Agricultural Catchments Programme: Catchment selection using spatial multi-criteria decision analysis. Soil Use Manag. 2010, 26, 225–236. [Google Scholar] [CrossRef]
- Esri. Topographic [Basemap]. Scale Not Given. World Topographic Map. 2012. Available online: http://www.arcgis.com/home/item.html?id=30e5fe3149c34df1ba922e6f5bbf808f (accessed on 31 August 2020).
- De Eyto, E.; Dillane, M.; Cooney, J.; Hughes, P.; Murphy, M.; Nixon, P.; Sweeney, D.; Poole, R.; Rouen, M. Water Quality and Meteorological Data from the Lough Feeagh Automatic Water Quality Monitoring Station (AWQMS), 2004–2017 Marine Institute, Ireland. Available online: http://data.marine.ie/geonetwork/srv/eng/catalog.search#/metadata/ie.marine.data:dataset.3757 (accessed on 6 April 2020).
- Watras, C.J.; Hanson, P.; Stacy, T.; Morrison, K.; Mather, J.; Hu, Y.H.; Milewski, P.A. A temperature compensation method for CDOM fluorescence sensors in freshwater. Limnol. Oceanogr. Methods 2011, 9, 296–301. [Google Scholar] [CrossRef]
- Ryder, E.; Jennings, E.; De Eyto, E.; Dillane, M.; NicAonghusa, C.; Pierson, D.C.; Moore, K.; Rouen, M.; Poole, R. Temperature quenching of CDOM fluorescence sensors: Temporal and spatial variability in the temperature response and a recommended temperature correction equation. Limnol. Oceanogr. Methods 2012, 10, 1004–1010. [Google Scholar] [CrossRef]
- Doyle, B.C.; De Eyto, E.; Dillane, M.; Poole, R.; McCarthy, V.; Ryder, E.; Jennings, E. Synchrony in catchment stream colour levels is driven by both local and regional climate. Biogeosciences 2019, 16, 1053–1071. [Google Scholar] [CrossRef]
- Met Éireann. Display and Download Historical Data from Current Stations. Available online: https://www.met.ie/climate/available-data/historical-data (accessed on 6 April 2020).
- Butturini, A.; Álvarez, M.; Bernal, S.; Vázquez, E.; Sabater, F. Diversity and temporal sequences of forms of DOC and NO3-discharge responses in an intermittent stream: Predictable or random succession? J. Geophys. Res. Space Phys. 2008, 113 (Suppl. G3), 721. [Google Scholar] [CrossRef]
- FWE; APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Eisenreich, S.J.; Bannerman, R.T.; Armstrong, D.E. A simplified phosphorus analysis technique. Environ. Lett. 1975, 9, 43–53. [Google Scholar] [CrossRef]
- Stainton, M.P.; Capel, M.J.; Armstrong, F.A.J. The Chemical Analysis of Fresh Water, 2nd ed.; Canadian Fisheries and Marine Service Miscellaneous Special Publications: Winnipeg, MB, Canada, 1977; p. 180. [Google Scholar]
- Standing Committee of Analysts. Methods for the examination of Waters and Associated Materials: Ammonia in Waters; HMSO: London, UK, 1981. [Google Scholar]
- Clesceri, L.S.; Greenberg, A.E.; Trussell, R.R. Standard Methods for the Examination of Water and Wastewater, 17th ed.; American Public Health Association: Washington, DC, USA, 1989; pp. 4–144. [Google Scholar]
- Irish Marine Institute. BurrishooleLTER-Public. Available online: https://github.com/IrishMarineInstitute/BurishooleLTER-Public (accessed on 14 May 2020).
- Zambrano-Bigiarini, M. Time Series Management, Analysis and Interpolation for Hydrological Modeling; R Package Version 04-2-1; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Winslow, L.; Read, J.; Woolway, R.; Brentrup, J.; Leach, T.; Zwart, Z.; Albers, S.; Collinge, D. rLakeAnalyzer: Lake Physics Tools; R Package Version 1110; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Nercessian, O.; Noyes, E.; Kalyuzhnaya, M.G.; Lidstrom, M.E.; Chistoserdova, L. Bacterial populations active in metabolism of C1 compounds in the sediment of Lake Washington, a freshwater lake. Microb. Ecol. 2005, 71, 6885–6899. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet.journal 2011, 17, 10. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Rohwer, R.R.; Hamilton, J.J.; Newton, R.J.; McMahon, K.D. TaxAss: Leveraging a custom freshwater database achieves fine-scale taxonomic resolution. mSphere 2018, 3, e00327-18. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.R.; Warwick, R.M. Primer-6 Computer Program; Natural Environment Research: Plymouth, MA, USA, 2005. [Google Scholar]
- Woodhouse, J.N.; Ziegler, J.; Grossart, H.-P.; Neilan, B.A. Cyanobacterial community composition and bacteria–bacteria interactions promote the stable occurrence of particle-associated bacteria. Front. Microbiol. 2018, 9, 777. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.J.; Jones, S.E.; Eiler, A.; McMahon, K.D.; Bertilsson, S. A guide to the natural history of freshwater lake bacteria. Microbiol. Mol. Biol. Rev. 2011, 75, 14–49. [Google Scholar] [CrossRef] [PubMed]
- Salcher, M.M.; Pernthaler, J.; Posch, T. Seasonal bloom dynamics and ecophysiology of the freshwater sister clade of SAR11 bacteria ‘that rule the waves’ (LD12). ISME J. 2011, 5, 1242–1252. [Google Scholar] [CrossRef]
- Calderó-Pascual, M.; De Eyto, E.; Jennings, E.; Dillane, M.; Andersen, M.R.; Kelly, S.; Wilson, H.L.; McCarthy, V. Effects of consecutive extreme weather events on a temperate dystrophic lake: A detailed insight into physical, chemical and biological responses. Water 2020, 12, 1411. [Google Scholar] [CrossRef]
- Jennings, E.; Allott, N.; Arvola, L.; Jarvinen, M.; Moore, K.; Naden, P.; Nic Aongusa, C.; Noges, T.; Weyhermeyer, G. Impacts of climate on the flux of dissolved organic carbon from catchments. In The Impact of Climate Change on European Lakes; George, D.G., Ed.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Jennings, E.; de Eyto, E.; Moore, T.; Dillane, M.; Ryder, E.; Allott, N.; Aonghusa, C.N.; Rouen, M.; Poole, R.; Pierson, D. From highs to lows: Changes in dissolved organic carbon in a peatland catchment and lake following extreme flow events. Water 2020. submitted. [Google Scholar]
- Clark, J.M.; Lane, S.N.; Chapman, P.J.; Adamson, J.K. Link between DOC in near surface peat and stream water in an upland catchment. Sci. Total. Environ. 2008, 404, 308–315. [Google Scholar] [CrossRef]
- Guillemette, F.; McCallister, S.L.; Del Giorgio, P.A. Selective consumption and metabolic allocation of terrestrial and algal carbon determine allochthony in lake bacteria. ISME J. 2016, 10, 1373–1382. [Google Scholar] [CrossRef]
- Woolway, R.I.; Jennings, E.; Carrea, L. Impact of the 2018 European heatwave on lake surface water temperature. Inland Waters 2020, 1–11. [Google Scholar] [CrossRef]
- Wiegand, S.; Jogler, M.; Jogler, C. On the maverick planctomycetes. FEMS Microbiol. Rev. 2018, 42, 739–760. [Google Scholar] [CrossRef]
- Giling, D.P.; Nejstgaard, J.C.; Berger, S.A.; Grossart, H.P.; Kirillin, G.; Penske, A.; Lentz, M.; Casper, P.; Sareyka, J.; Gessner, M.O. Thermocline deepening boosts ecosystem metabolism: Evidence from a large-scale lake enclosure experiment simulating a summer storm. Glob. Chang. Biol. 2017, 23, 1448–1462. [Google Scholar] [CrossRef] [PubMed]
- Martín-Platero, A.M.; Cleary, B.; Kauffman, K.; Preheim, S.P.; McGillicuddy, D.J.; Alm, E.J.; Polz, M.F. High resolution time series reveals cohesive but short-lived communities in coastal plankton. Nat. Commun. 2018, 9, 266. [Google Scholar] [CrossRef] [PubMed]
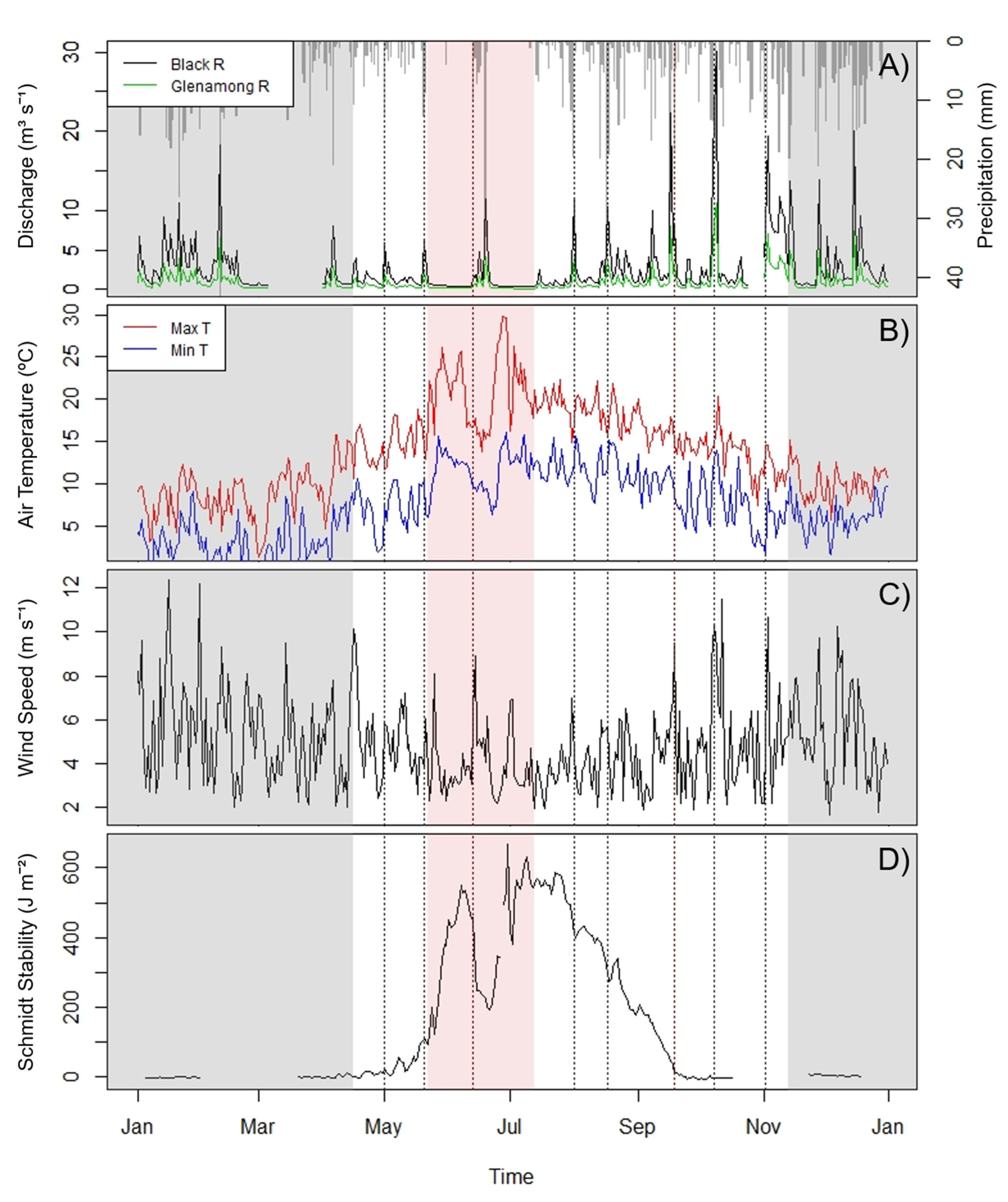


| Date(s) | Named Storm | Peak Rainfall (mm day−1) | Peak Wind Speed (m s−1) | Peak BR 1 Discharge (m3 s−1) | Peak Schmidt Stability (J m−2) | DOM Dilution/Flushing | Event Type |
|---|---|---|---|---|---|---|---|
| 1 May | No | 12.7 | 5.81 | 5.88 | 23.07 | Flushing | HDE 2 |
| 20–21 May | No | 12.8 | 6.06 | 6.02 | 108.97 | Flushing | HDE |
| 22 May–14 July | No | 33.2 | 8.89 | 11.46 | 669.42 | N/A | Drought |
| 13–19 June | Hector | 33.2 | 8.89 | 11.46 | 458.35 | Flushing | Storm |
| 1 August | No | 19.8 | 4.42 | 11.42 | 410.54 | Flushing | HDE |
| 17–18 August | No | 37.0 | 5.70 | 10.43 | 297.78 | Dilution | HDE |
| 17–19 September | Ali | 22.8 | 9.51 | 22.11 | 46.57 | Dilution | Storm |
| 7–9 October | No | 20.1 | 10.33 | 30.16 | −2.76 | Dilution | HDE |
| 2–3 November | No | 13.3 | 10.69 | 19.32 | N/A | Dilution | HDE |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoke, A.; Woodhouse, J.; Zoccarato, L.; McCarthy, V.; de Eyto, E.; Calderó-Pascual, M.; Geffroy, E.; Dillane, M.; Grossart, H.-P.; Jennings, E. Impacts of Extreme Weather Events on Bacterial Community Composition of a Temperate Humic Lake. Water 2020, 12, 2757. https://doi.org/10.3390/w12102757
Hoke A, Woodhouse J, Zoccarato L, McCarthy V, de Eyto E, Calderó-Pascual M, Geffroy E, Dillane M, Grossart H-P, Jennings E. Impacts of Extreme Weather Events on Bacterial Community Composition of a Temperate Humic Lake. Water. 2020; 12(10):2757. https://doi.org/10.3390/w12102757
Chicago/Turabian StyleHoke, Alexa, Jason Woodhouse, Luca Zoccarato, Valerie McCarthy, Elvira de Eyto, Maria Calderó-Pascual, Ewan Geffroy, Mary Dillane, Hans-Peter Grossart, and Eleanor Jennings. 2020. "Impacts of Extreme Weather Events on Bacterial Community Composition of a Temperate Humic Lake" Water 12, no. 10: 2757. https://doi.org/10.3390/w12102757
APA StyleHoke, A., Woodhouse, J., Zoccarato, L., McCarthy, V., de Eyto, E., Calderó-Pascual, M., Geffroy, E., Dillane, M., Grossart, H.-P., & Jennings, E. (2020). Impacts of Extreme Weather Events on Bacterial Community Composition of a Temperate Humic Lake. Water, 12(10), 2757. https://doi.org/10.3390/w12102757







