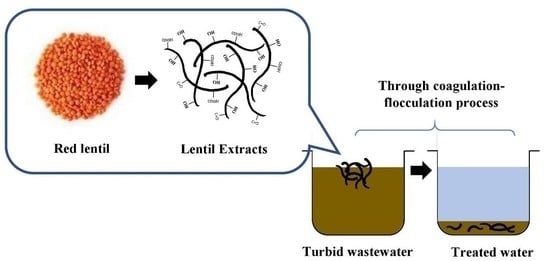
Red Lentil (Lens culinaris) Extract as a Novel Natural Coagulant for Turbidity Reduction: An Evaluation, Characterization and Performance Optimization Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Lentil Extracts
2.2.2. Preparation of Model Turbid Water
2.2.3. Analytical Method
2.2.4. Assay of Turbidity Reduction
2.2.5. Sludge Volume and Sludge Volume Index (SVI) Measurement
2.2.6. Experiment Design
2.2.7. Verification of the Designed Model
3. Results and Discussion
3.1. Physicochemical Characteristics of Brown, Green, and Red Lentils
3.1.1. Surface Morphology of the Lentils
3.1.2. Elemental Compositions of the Lentils
3.1.3. Zeta Potential of the Lentils
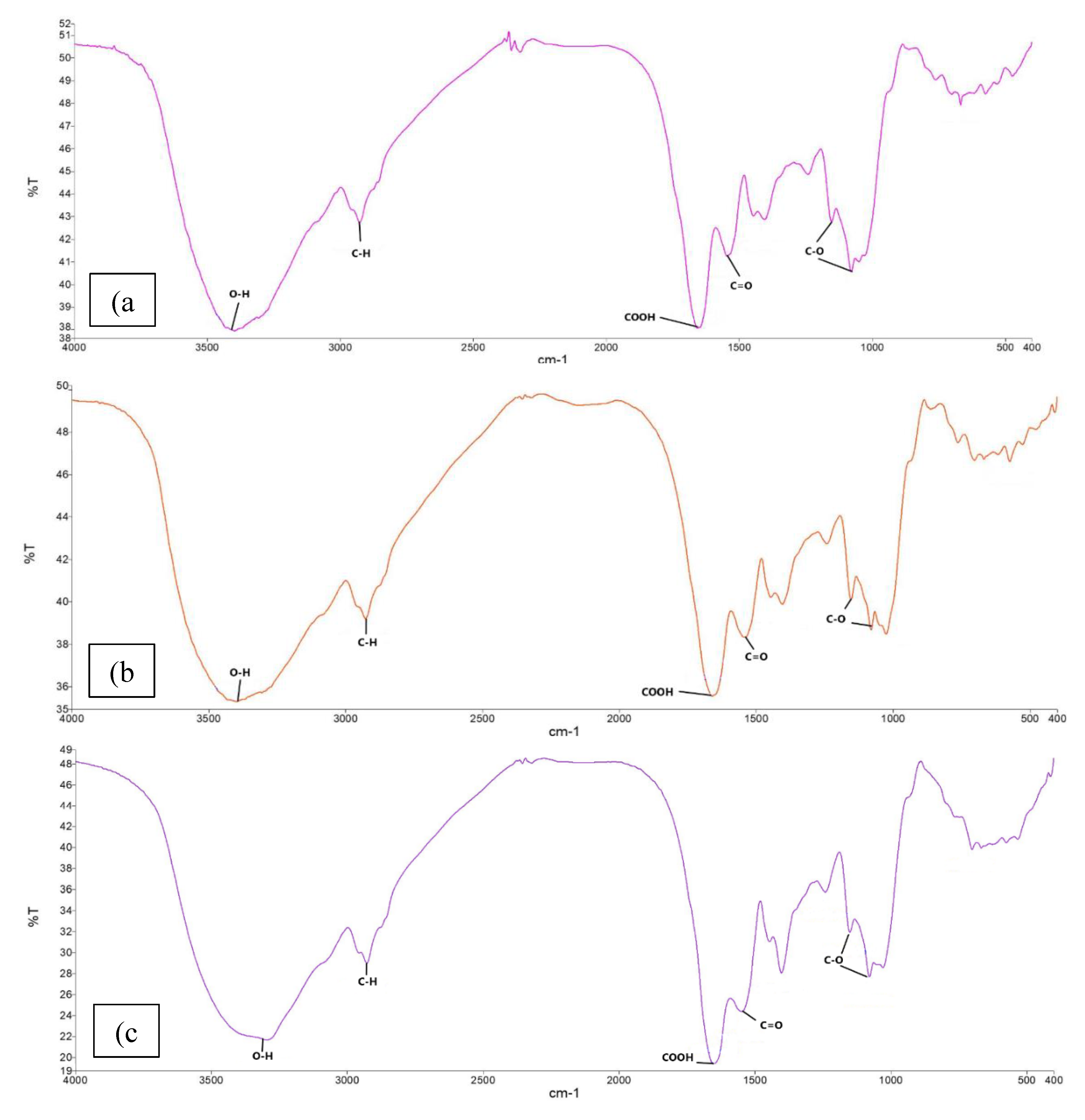
3.1.4. Functional Groups of the Lentils
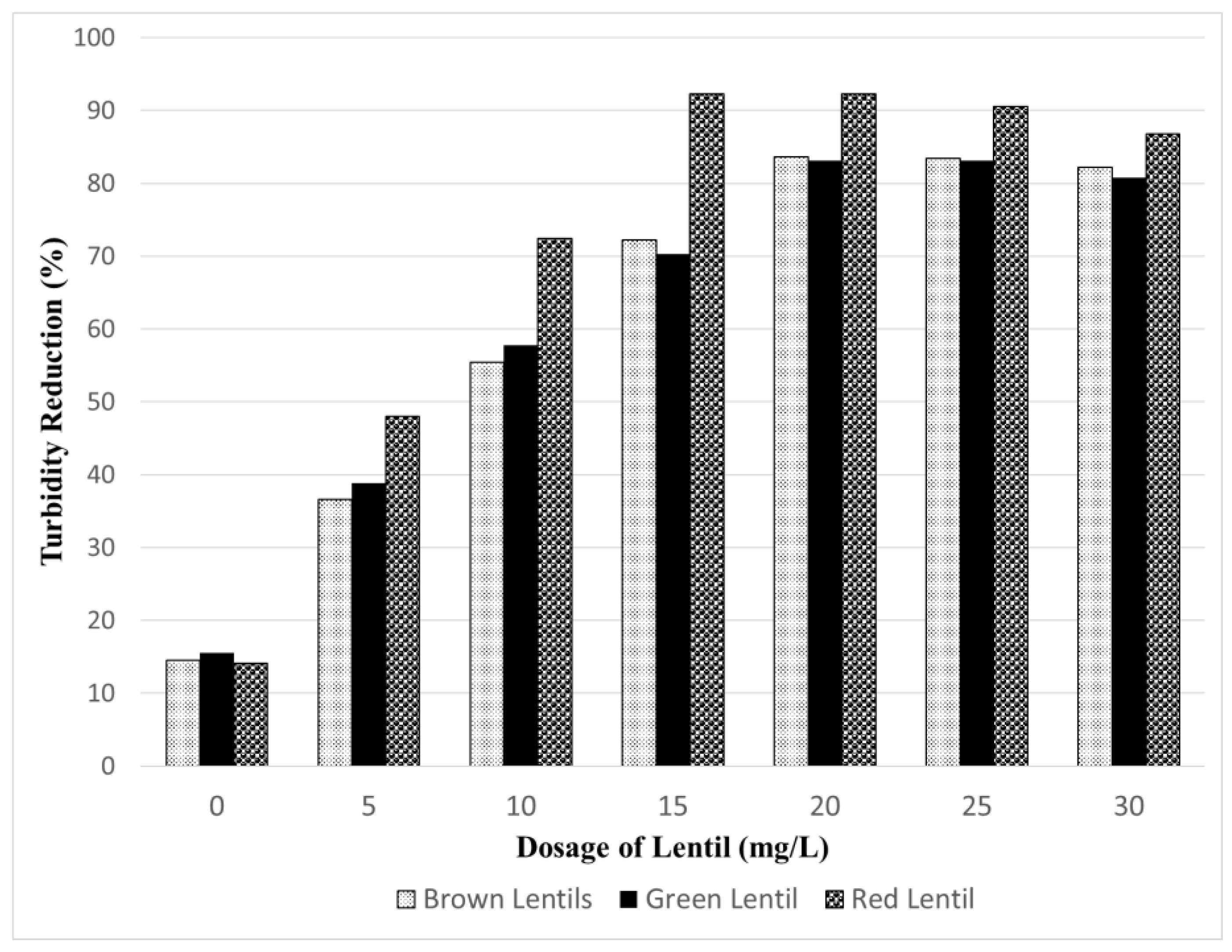
3.2. Preliminary Study on the Coagulating Ability of Different Lentils
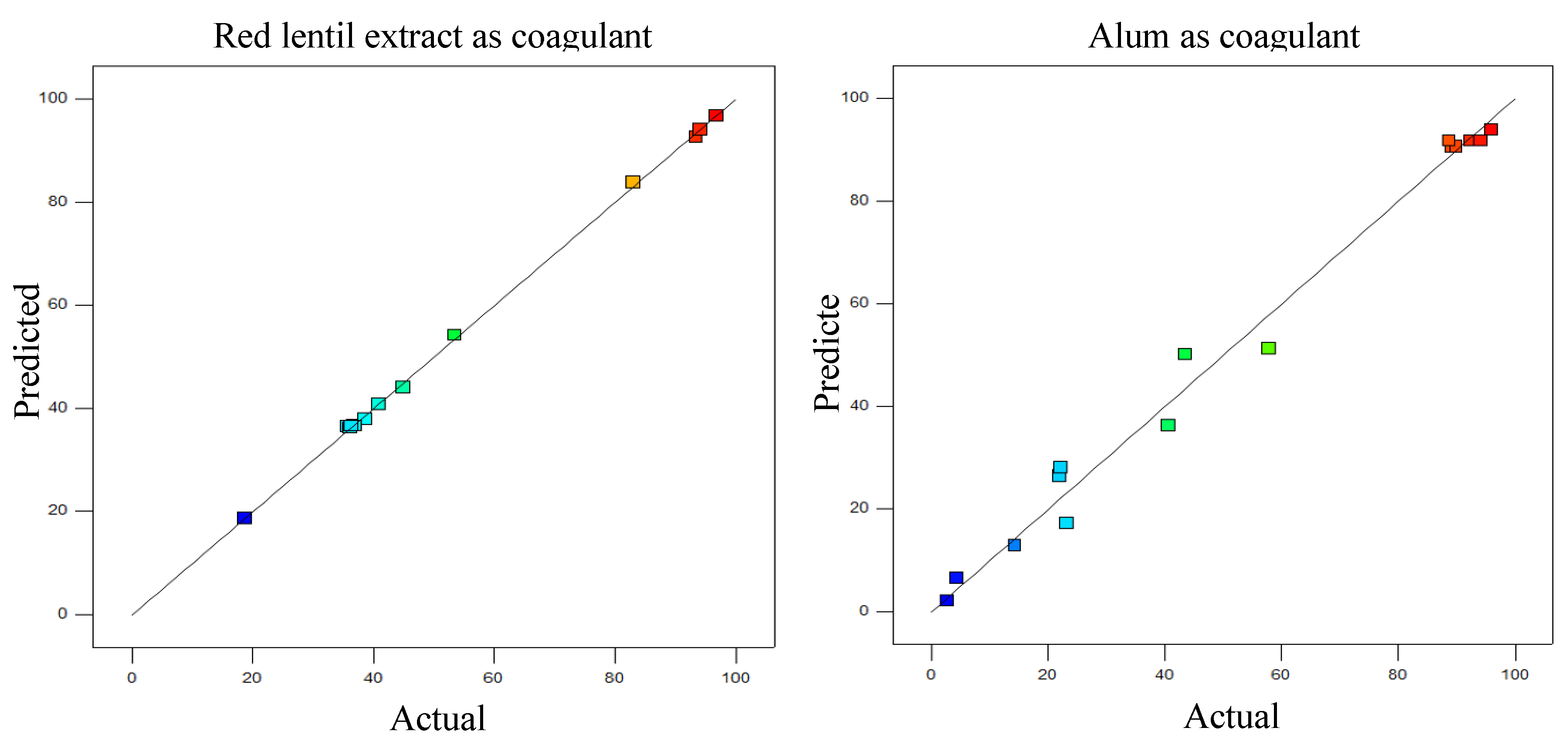
3.3. Model Adequacy Checking
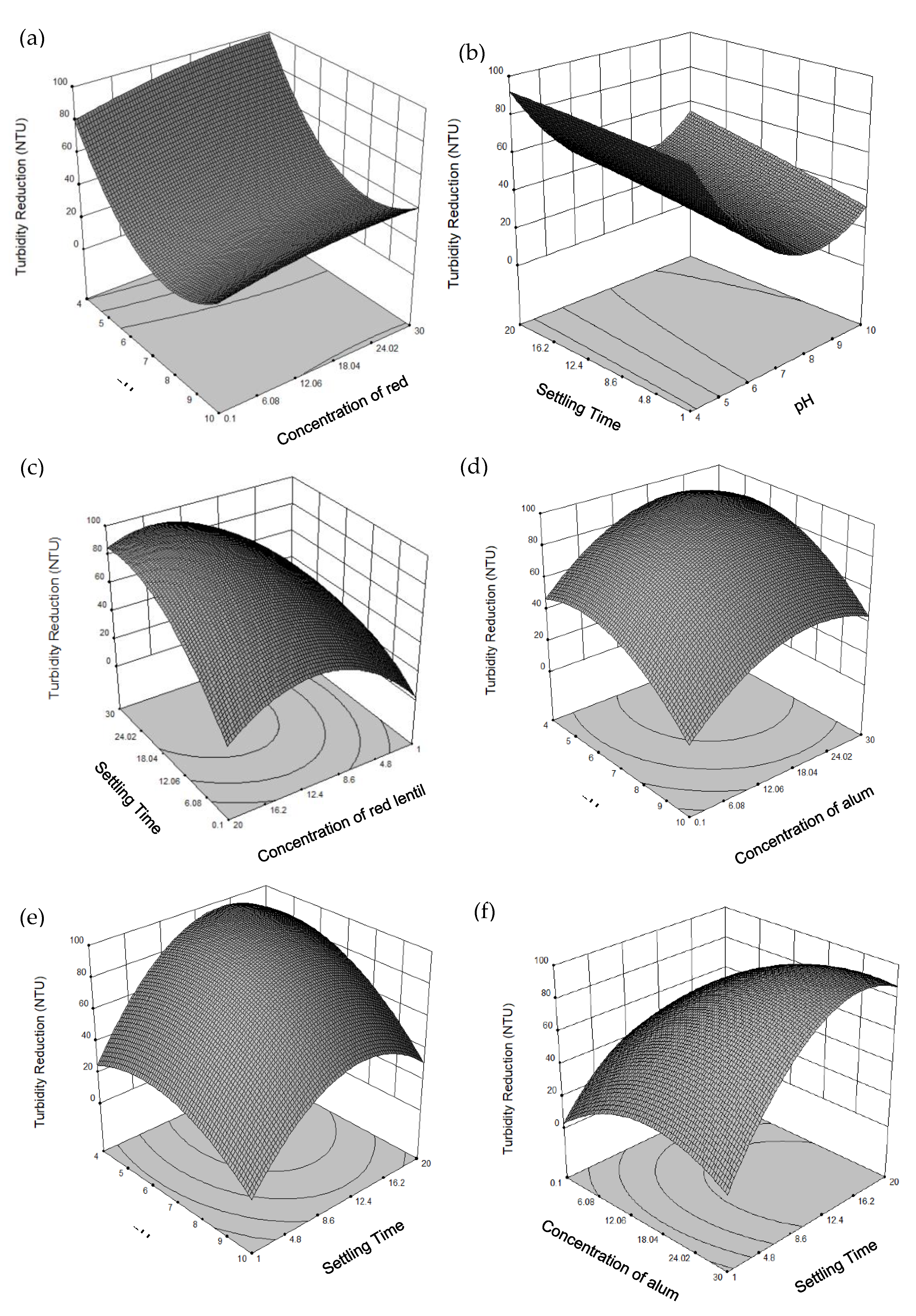
3.4. Effect of Operating Parameters and Their Respective Interactions Using Red Lentil as Coagulant
3.4.1. Effect of pH and Concentration of Coagulant
3.4.2. Effect of Settling Time and pH
3.4.3. Effect of Concentration of Coagulant and Settling Time
3.5. Multiple Responses Optimization for Turbidity Reduction
3.6. Validation of the Designed Model
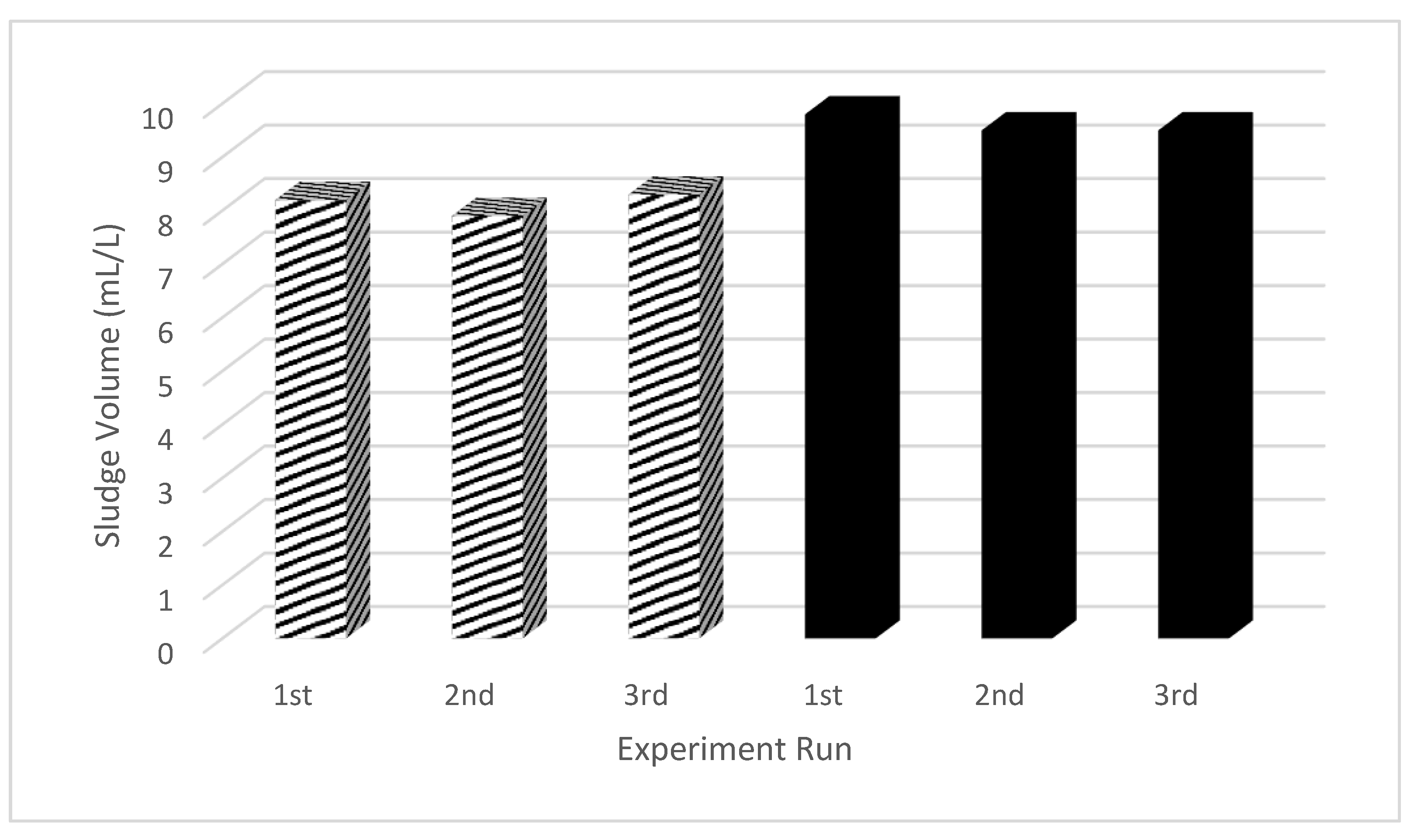
3.7. Evaluation of Sludge/Flocs Produced
3.8. Economic Feasibility Analysis of Lentil Extracts as Coagulant
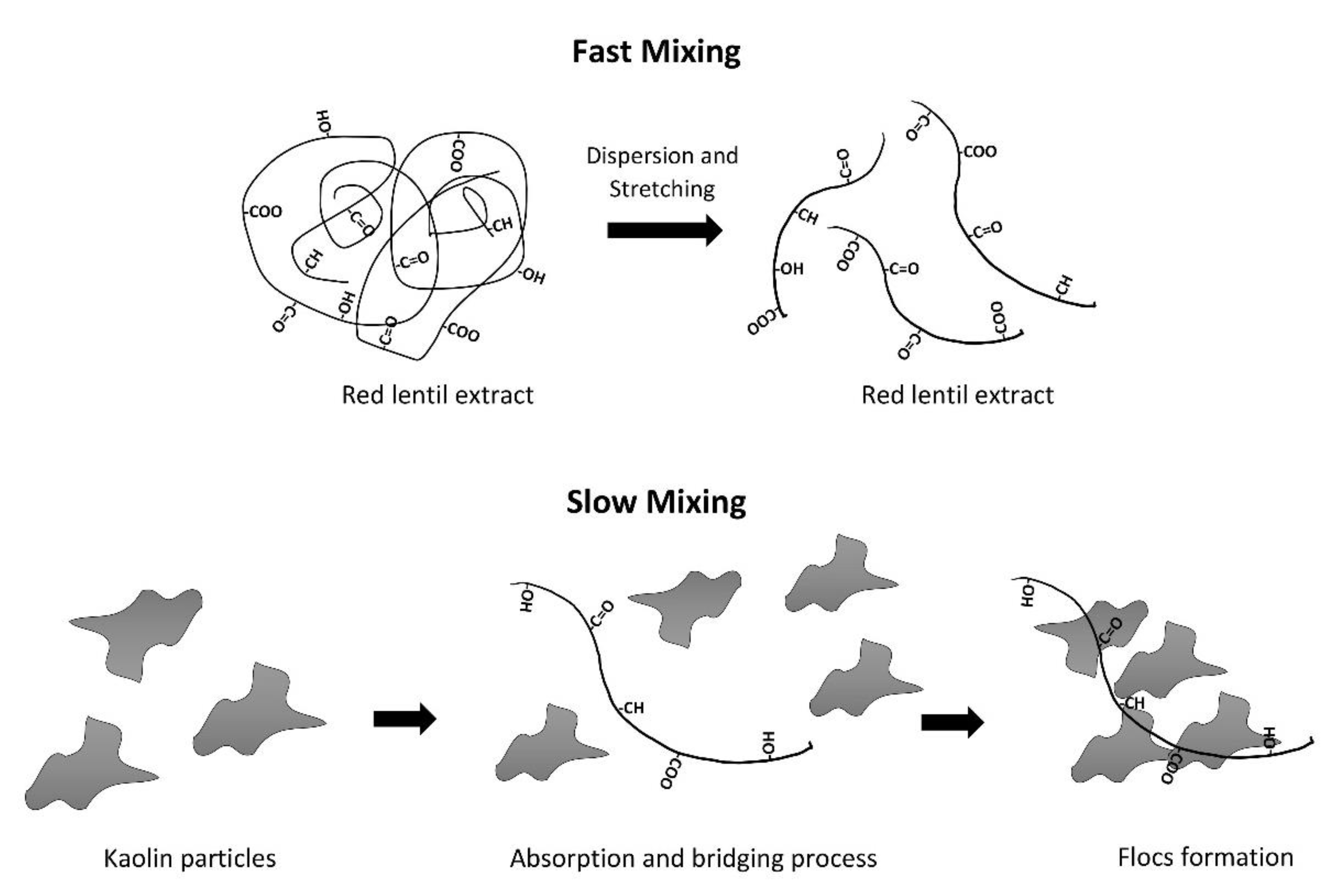
3.9. Proposed Mechanism for Red Lentil Extract
4. Conclusions
- Red lentil extract has the highest turbidity reduction efficiency, followed by brown lentils and green lentil extract.
- The main active compounds of red lentil extract were postulated to be the soluble protein and the amylose. Besides, the coagulation–flocculation mechanism utilizing red lentil extract as a coagulant was found to be adsorption and facilitated by bridging.
- The high correlation of the designed model revealed the suitability of the second-order polynomial model using BBD as an optimization tool to optimize the operating parameters for turbidity reduction. The utilization of red lentil extract as coagulation can be achieved up to 98.87% of turbidity reduction and the optimal conditions for pH, concentration of coagulant and settling time were 4, 26.3 mg/L, and 3 min. This promising efficiency was comparable to those acquired by using alum as the coagulant.
- The red lentil extract was found to be more environmentally friendly approached as it generated 15.6% lower sludge as compared to alum as coagulation and helped to reduce the sludge management cost for the wastewater treatment. Besides, it produced flocs with better settling characteristic (based on SVI) as compared to alum.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fermino, L.; Pedrangelo, A.; Silva, P.; Azevedo, R.; Yamaguchi, N.; Ribeiro, R. Water Treatment with Conventional and Alternative Coagulants. Chem. Eng. Trans. 2017, 57, 1189–1194. [Google Scholar]
- Harfouchi, H.; Hank, D.; Hellal, A. Response surface methodology for the elimination of humic substances from water by coagulation using powdered Saddled sea bream scale as coagulant-aid. Process Saf. Environ. Prot. 2016, 99, 216–226. [Google Scholar] [CrossRef]
- Roy, F.; Boye, J.; Simpson, B. Bioactive proteins and peptides in pulse crops: Pea, chickpea and lentil. Food Res. Int. 2010, 43, 432–442. [Google Scholar] [CrossRef]
- Jood, S.; Bishnoi, S.; Sharma, A. Chemical analysis and physico-chemical properties of chickpea and lentil cultivars. Food/Nahr. 1998, 42, 71–74. [Google Scholar] [CrossRef]
- Canada, A.A.-F. Lentils: Situation and outlook. Available online: http://www.agr.gc.ca/ (accessed on 26 April 2019).
- Schulz, C.R.; Okun, D.A. Treating surface waters for communities in developing countries. J. Am. Water Work. Assoc. 1983, 75, 212–219. [Google Scholar] [CrossRef]
- Ordaz-Díaz, L.A.; Valle-Cervantes, S.; Rodríguez-Rosales, J.; Bailón-Salas, A.M.; Madrid-Del Palacio, M.; Torres-Fraga, K.; Luis, A. Zeta Potential as a Tool to Evaluate the Optimum Performance of a Coagulation-flocculation Process for Wastewater Internal Treatment for Recirculation in the Pulp and Paper Process. BioResources 2017, 12, 5953–5969. [Google Scholar] [CrossRef]
- Roy, A.; Bhattacharya, J. Nanotechnology in Industrial Wastewater Treatment; IWA Publishing: London, UK, 2015. [Google Scholar]
- Association, A.P.H.; Association, A.W.W.; Federation, W.P.C.; Federation, W.E. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1915; Volume 2. [Google Scholar]
- Zarei, M.; Niaei, A.; Salari, D.; Khataee, A. Application of response surface methodology for optimization of peroxi-coagulation of textile dye solution using carbon nanotube–PTFE cathode. J. Hazard. Mater. 2010, 173, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, M.; Ray, M.B.; Neogi, S. Evaluation of the potential application of cactus (Opuntia ficus-indica) as a bio-coagulant for pre-treatment of oil sands process-affected water. Sep. Purif. Technol. 2019, 209, 714–724. [Google Scholar] [CrossRef]
- Zheng, H.; Sun, Y.; Guo, J.; Li, F.; Fan, W.; Liao, Y.; Guan, Q. Characterization and evaluation of dewatering properties of PADB, a highly efficient cationic flocculant. Ind. Eng. Chem. Res. 2014, 53, 2572–2582. [Google Scholar] [CrossRef]
- Ma, J.; Fu, K.; Shi, J.; Sun, Y.; Zhang, X.; Ding, L. Ultraviolet-assisted synthesis of polyacrylamide-grafted chitosan nanoparticles and flocculation performance. Carbohydr. Polym. 2016, 151, 565–575. [Google Scholar] [CrossRef]
- Ahmad, R.; Haseeb, S. Absorptive removal of Pb2+, Cu2+ and Ni2+ from the aqueous solution by using groundnut husk modified with Guar Gum (GG): Kinetic and thermodynamic studies. Groundw. Sustain. Dev. 2015, 1, 41–49. [Google Scholar] [CrossRef]
- Agunbiade, M.; Pohl, C.; Ashafa, O. Bioflocculant production from Streptomyces platensis and its potential for river and waste water treatment. Braz. J. Microbiol. 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Aziz, H.; Rahim, N.; Ramli, S.; Alazaiza, M.; Omar, F.; Hung, Y.-T. Potential Use of Dimocarpus longan Seeds as a Flocculant in Landfill Leachate Treatment. Water 2018, 10, 1672. [Google Scholar] [CrossRef]
- Benalia, A.; Derbal, K.; Panico, A.; Pirozzi, F. Use of Acorn Leaves as a natural coagulant in a drinking water treatment plant. Water 2019, 11, 57. [Google Scholar] [CrossRef]
- Shak, K.P.Y.; Wu, T.Y. Coagulation–flocculation treatment of high-strength agro-industrial wastewater using natural Cassia obtusifolia seed gum: treatment efficiencies and flocs characterization. Chem. Eng. J. 2014, 256, 293–305. [Google Scholar] [CrossRef]
- Kakoi, B.; Kaluli, J.W.; Ndiba, P.; Thiong’o, G. Banana pith as a natural coagulant for polluted river water. Ecol. Eng. 2016, 95, 699–705. [Google Scholar] [CrossRef]
- Ghori, M.U.; Mohammad, M.A.; Rudrangi, S.R.S.; Fleming, L.T.; Merchant, H.A.; Smith, A.M.; Conway, B.R. Impact of purification on physicochemical, surface and functional properties of okra biopolymer. Food Hydrocoll. 2017, 71, 311–320. [Google Scholar] [CrossRef]
- Chung, H.-J.; Liu, Q.; Hoover, R.; Warkentin, T.D.; Vandenberg, B. In vitro starch digestibility, expected glycemic index, and thermal and pasting properties of flours from pea, lentil and chickpea cultivars. Food Chem. 2008, 111, 316–321. [Google Scholar] [CrossRef]
- Ma, Z.; Boye, J.I.; Simpson, B.K.; Prasher, S.O.; Monpetit, D.; Malcolmson, L. Thermal processing effects on the functional properties and microstructure of lentil, chickpea, and pea flours. Food Res. Int. 2011, 44, 2534–2544. [Google Scholar] [CrossRef]
- Okumus, B.N.; Tacer-Caba, Z.; Kahraman, K.; Nilufer-Erdil, D. Resistant starch type V formation in brown lentil (Lens culinaris Medikus) starch with different lipids/fatty acids. Food Chem. 2018, 240, 550–558. [Google Scholar] [CrossRef]
- Teh, C.Y.; Wu, T.Y.; Juan, J.C. Optimization of agro-industrial wastewater treatment using unmodified rice starch as a natural coagulant. Ind. Crop. Prod. 2014, 56, 17–26. [Google Scholar] [CrossRef]
- Ahmad, A.; Wong, S.; Teng, T.; Zuhairi, A. Optimization of coagulation–flocculation process for pulp and paper mill effluent by response surface methodological analysis. J. Hazard. Mater. 2007, 145, 162–168. [Google Scholar] [CrossRef]
- Ghafari, S.; Aziz, H.A.; Bashir, M.J. The use of poly-aluminum chloride and alum for the treatment of partially stabilized leachate: A comparative study. Desalination 2010, 257, 110–116. [Google Scholar] [CrossRef]
- Duan, J.; Gregory, J. Coagulation by hydrolysing metal salts. Adv. Colloid Interface Sci. 2003, 100, 475–502. [Google Scholar] [CrossRef]
- Banu, R.; Do, K.; Yeom, I. Phosphorus removal in low alkalinity secondary effluent using alum. Int. J. Environ. Sci. Technol. 2008, 5, 93–98. [Google Scholar] [CrossRef]
- Shak, K.P.Y.; Wu, T.Y. Optimized use of alum together with unmodified Cassia obtusifolia seed gum as a coagulant aid in treatment of palm oil mill effluent under natural pH of wastewater. Ind. Crop. Prod. 2015, 76, 1169–1178. [Google Scholar] [CrossRef]
- Sanghi, R.; Bhatttacharya, B.; Singh, V. Cassia angustifolia seed gum as an effective natural coagulant for decolourisation of dye solutions. Green Chem. 2002, 4, 252–254. [Google Scholar] [CrossRef]
- Bolto, B.; Gregory, J. Organic polyelectrolytes in water treatment. Water Res. 2007, 41, 2301–2324. [Google Scholar] [CrossRef]
- Wu, P.; Jiang, L.Y.; He, Z.; Song, Y. Treatment of metallurgical industry wastewater for organic contaminant removal in China: status, challenges, and perspectives. Environ. Sci. Water Res. Technol. 2017, 3, 1015–1031. [Google Scholar] [CrossRef]
- Al-Harahsheh, M.; Batiha, M.; Kraishan, S.; Al-Zoubi, H. Precipitation treatment of effluent acidic wastewater from phosphate-containing fertilizer industry: characterization of solid and liquid products. Sep. Purif. Technol. 2014, 123, 190–199. [Google Scholar] [CrossRef]
- Ndabigengesere, A.; Narasiah, K.S. Quality of water treated by coagulation using Moringa oleifera seeds. Water Res. 1998, 32, 781–791. [Google Scholar] [CrossRef]
- Haydar, S.; Aziz, J.A. Coagulation–flocculation studies of tannery wastewater using combination of alum with cationic and anionic polymers. J. Hazard. Mater. 2009, 168, 1035–1040. [Google Scholar] [CrossRef]
- Choy, S.Y.; Prasad, K.N.; Wu, T.Y.; Raghunandan, M.E.; Ramanan, R.N. Performance of conventional starches as natural coagulants for turbidity removal. Ecol. Eng. 2016, 94, 352–364. [Google Scholar] [CrossRef]
- Mishra, S.; Mukul, A.; Sen, G.; Jha, U. Microwave assisted synthesis of polyacrylamide grafted starch (St-g-PAM) and its applicability as flocculant for water treatment. Int. J. Biol. Macromol. 2011, 48, 106–111. [Google Scholar] [CrossRef]


| Operating Parameters | Units | Coded Symbols | Range and Level | ||
|---|---|---|---|---|---|
| −1 | 0 | +1 | |||
| Lentil as coagulant | |||||
| pH | X1 | 4 | 7 | 10 | |
| Concentration of lentil | mg/L | X2 | 0.1 | 15.05 | 30 |
| Settling time | minute(s) | X3 | 1 | 10.5 | 20 |
| Alum as coagulant | |||||
| pH | Y1 | 4 | 7 | 10 | |
| Concentration of (Al3+) | mg/L | Y2 | 0.1 | 15.05 | 30 |
| Settling time | minute(s) | Y3 | 1 | 10.5 | 20 |
| Elements | Weight (%) | Atomic (%) | ||||
|---|---|---|---|---|---|---|
| Brown Lentil | Green Lentil | Red Lentil | Brown Lentil | Green Lentil | Red Lentil | |
| C | 59.68 | 54.69 | 53.12 | 66.50 | 61.72 | 60.91 |
| O | 39.86 | 45.08 | 44.33 | 33.34 | 38.20 | 38.16 |
| K | 0.39 | 0.23 | 1.45 | 0.13 | 0.08 | 0.51 |
| P | 0.06 | 0.00 | 0.32 | 0.03 | 0.00 | 0.14 |
| Ca | 0.00 | 0.00 | 0.78 | 0.00 | 0.00 | 0.27 |
| Lentil | Zeta Potential (mV) | |||
|---|---|---|---|---|
| 1 | 2 | 3 | Average | |
| Brown lentil | −3.58 | −4.06 | −3.58 | −3.74 |
| Green lentil | −3.58 | −2.81 | −2.34 | −2.91 |
| Red lentil | −6.77 | −5.17 | −5.79 | −5.91 |
| Source | Sum of Squares | Mean Square | F Value | p-Value |
|---|---|---|---|---|
| Red lentil extract as coagulant | ||||
| Model | 9388.94 | 1043.22 | 1317.57 | <0.0001 |
| pH (X1) | 4586.50 | 4586.50 | 5792.7 | <0.0001 |
| Concentration of lentil extract (X2) | 131.67 | 131.67 | 166.3 | <0.0001 |
| Settling Time (X3) | 177.84 | 177.84 | 224.61 | <0.0001 |
| X1X2 | 23.67 | 23.67 | 29.89 | 0.0028 |
| X1X3 | 64.63 | 64.63 | 81.63 | 0.0003 |
| X2X3 | 95.62 | 95.62 | 120.77 | 0.0001 |
| X12 | 4136.59 | 4136.59 | 5224.46 | <0.0001 |
| X22 | 49.26 | 49.26 | 62.21 | 0.0005 |
| X32 | 1.45 | 1.45 | 1.84 | 0.2334 |
| Lack of Fit | 3.73 | 1.24 | 10.80 | 0.0859 |
| R-squared = 0.9996, adjusted R-squared = 0.9988, adequate precision = 107.625, predicted R-Squared = 0.99 | ||||
| Alum as coagulant | ||||
| Model | 18,254.86 | 2028.32 | 44.20 | 0.0003 |
| pH (Y1) | 3105.94 | 3105.94 | 67.68 | 0.0004 |
| Concentration of alum (Y2) | 2925.60 | 2925.60 | 63.75 | 0.0005 |
| Settling Time (Y3) | 4828.49 | 4828.49 | 105.21 | 0.0002 |
| Y1Y2 | 1.01 | 1.01 | 0.02 | 0.8877 |
| Y1Y3 | 225.09 | 225.09 | 4.90 | 0.0776 |
| Y2Y3 | 758.31 | 758.31 | 16.52 | 0.0097 |
| Y12 | 1343.25 | 1343.25 | 29.27 | 0.0029 |
| Y22 | 1704.82 | 1704.82 | 37.15 | 0.0017 |
| Y32 | 4232.44 | 4232.44 | 92.22 | 0.0002 |
| Lack of Fit | 213.94 | 71.31 | 9.19 | 0.0998 |
| R-squared = 0.9876, adjusted R-squared = 0.9652, adequate precision = 16.599, predicted R-Squared = 0.81 | ||||
| pH | Concentration of Alum, (mg/L) | Settling Time, (min) | Turbidity Reduction (%) | ||
|---|---|---|---|---|---|
| Predicted a | Actual b | Different | |||
| Red lentil extract as coagulant | |||||
| 4.0 | 26.3 | 3.0 | 98.68 | 98.87 | 0.18 |
| 5.8 | 14.5 | 16.0 | 53.40 | 51.32 | 2.08 |
| 9.3 | 10.0 | 3.7 | 24.94 | 26.42 | 1.48 |
| Alum as coagulant | |||||
| 6.0 | 24.6 | 10 | 98.48 | 97.64 | 0.84 |
| 4.8 | 23.7 | 3.5 | 53.59 | 50.60 | 2.99 |
| 9.9 | 15.87 | 2.3 | 13.45 | 16.25 | 2.80 |
| Coagulant | Unit Price ($/kg) | Optimum Concentration, ×10−3 (kg/m3) | Total Material Cost, ×10−3 ($/m3) |
|---|---|---|---|
| Red lentil extract | 0.30 | 26.3 | 7.89 |
| Alum | 0.43 | 26 | 11.18 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chua, S.-C.; Malek, M.A.; Chong, F.-K.; Sujarwo, W.; Ho, Y.-C. Red Lentil (Lens culinaris) Extract as a Novel Natural Coagulant for Turbidity Reduction: An Evaluation, Characterization and Performance Optimization Study. Water 2019, 11, 1686. https://doi.org/10.3390/w11081686
Chua S-C, Malek MA, Chong F-K, Sujarwo W, Ho Y-C. Red Lentil (Lens culinaris) Extract as a Novel Natural Coagulant for Turbidity Reduction: An Evaluation, Characterization and Performance Optimization Study. Water. 2019; 11(8):1686. https://doi.org/10.3390/w11081686
Chicago/Turabian StyleChua, Siong-Chin, Marlinda Abdul Malek, Fai-Kait Chong, Wawan Sujarwo, and Yeek-Chia Ho. 2019. "Red Lentil (Lens culinaris) Extract as a Novel Natural Coagulant for Turbidity Reduction: An Evaluation, Characterization and Performance Optimization Study" Water 11, no. 8: 1686. https://doi.org/10.3390/w11081686
APA StyleChua, S.-C., Malek, M. A., Chong, F.-K., Sujarwo, W., & Ho, Y.-C. (2019). Red Lentil (Lens culinaris) Extract as a Novel Natural Coagulant for Turbidity Reduction: An Evaluation, Characterization and Performance Optimization Study. Water, 11(8), 1686. https://doi.org/10.3390/w11081686