The Use of Boron-Doped Diamond Electrode for the Determination of Selected Biocides in Water Samples
Abstract
1. Introduction
2. Experimental
Apparatus and Chemicals
3. Results and Discussion
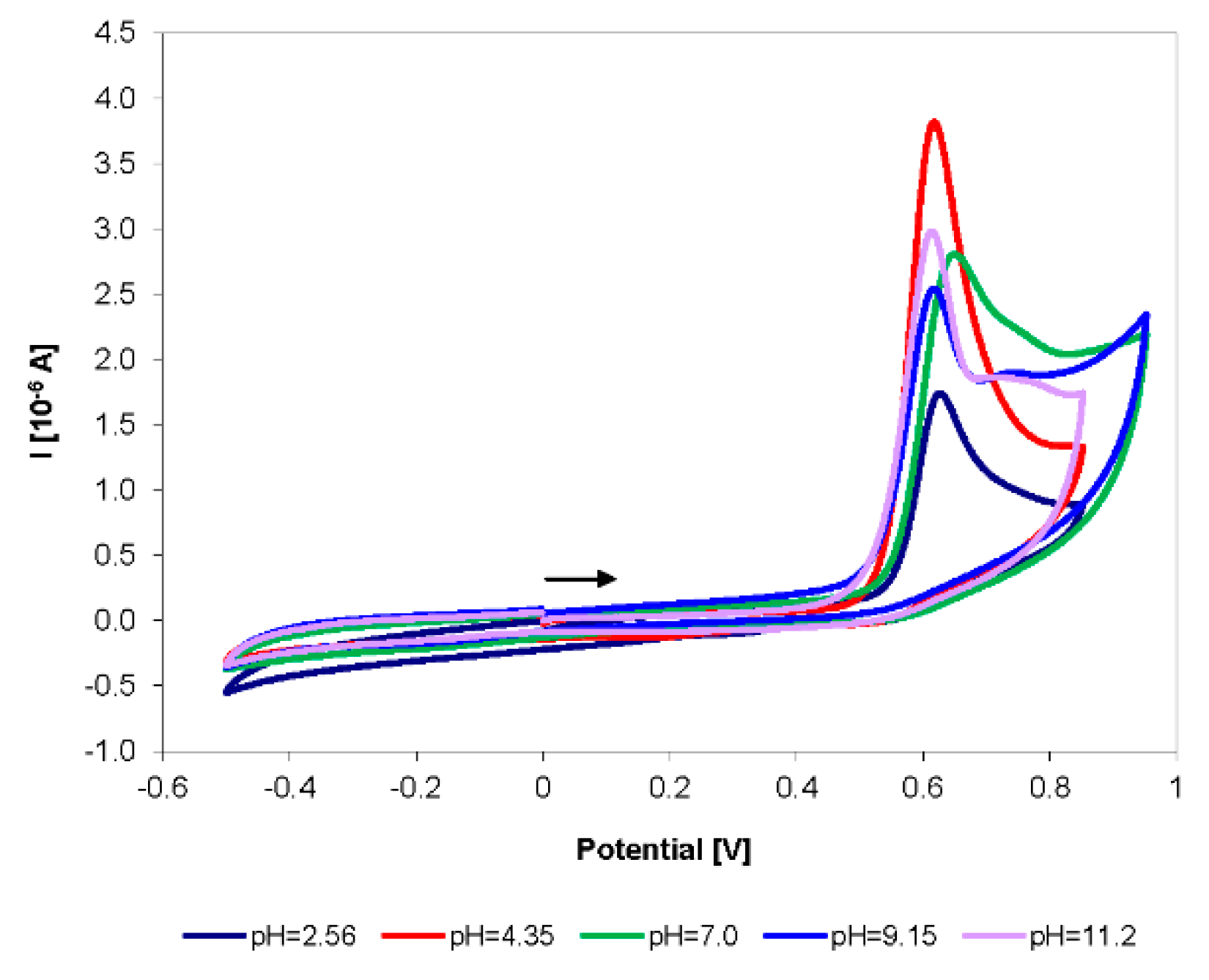
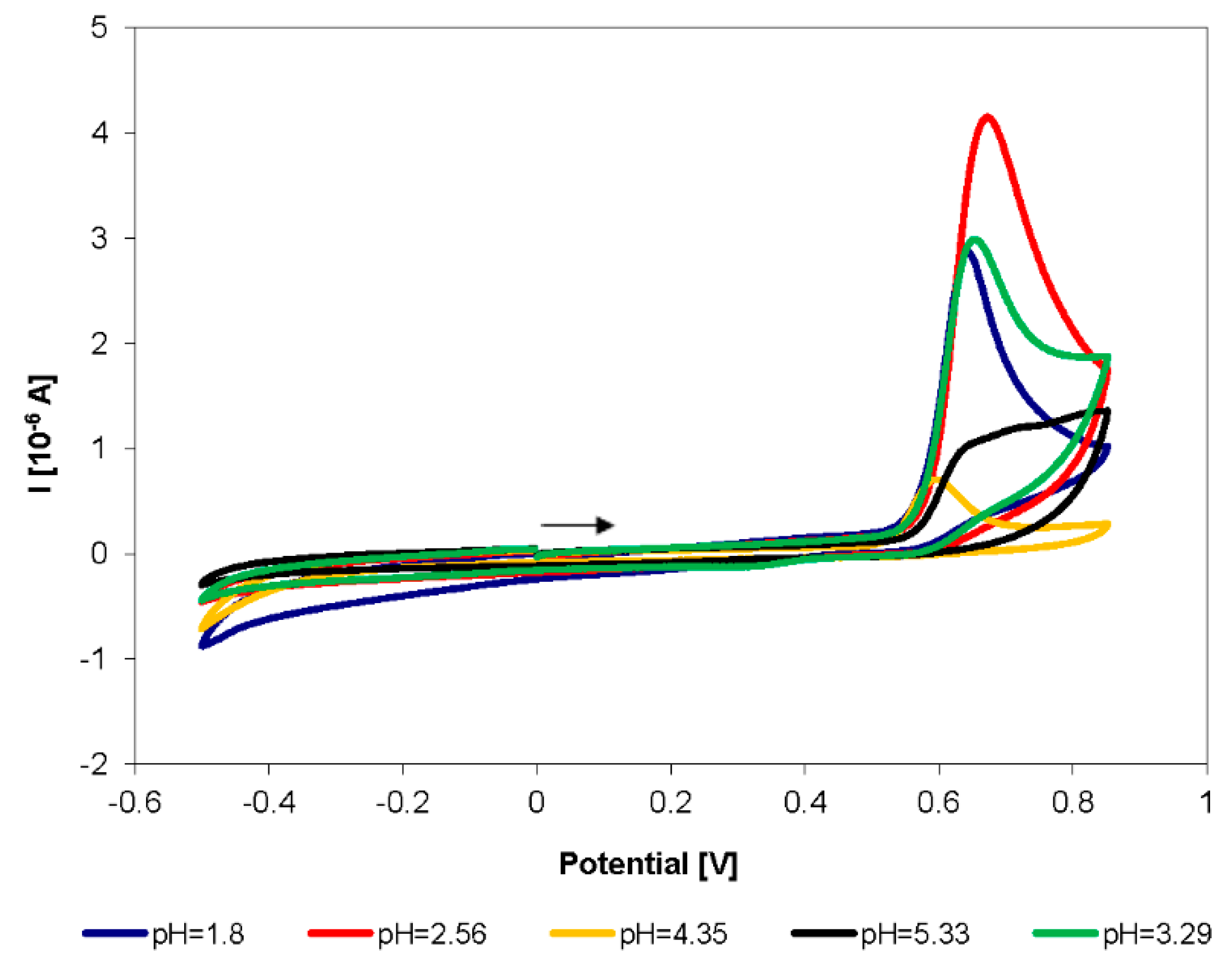
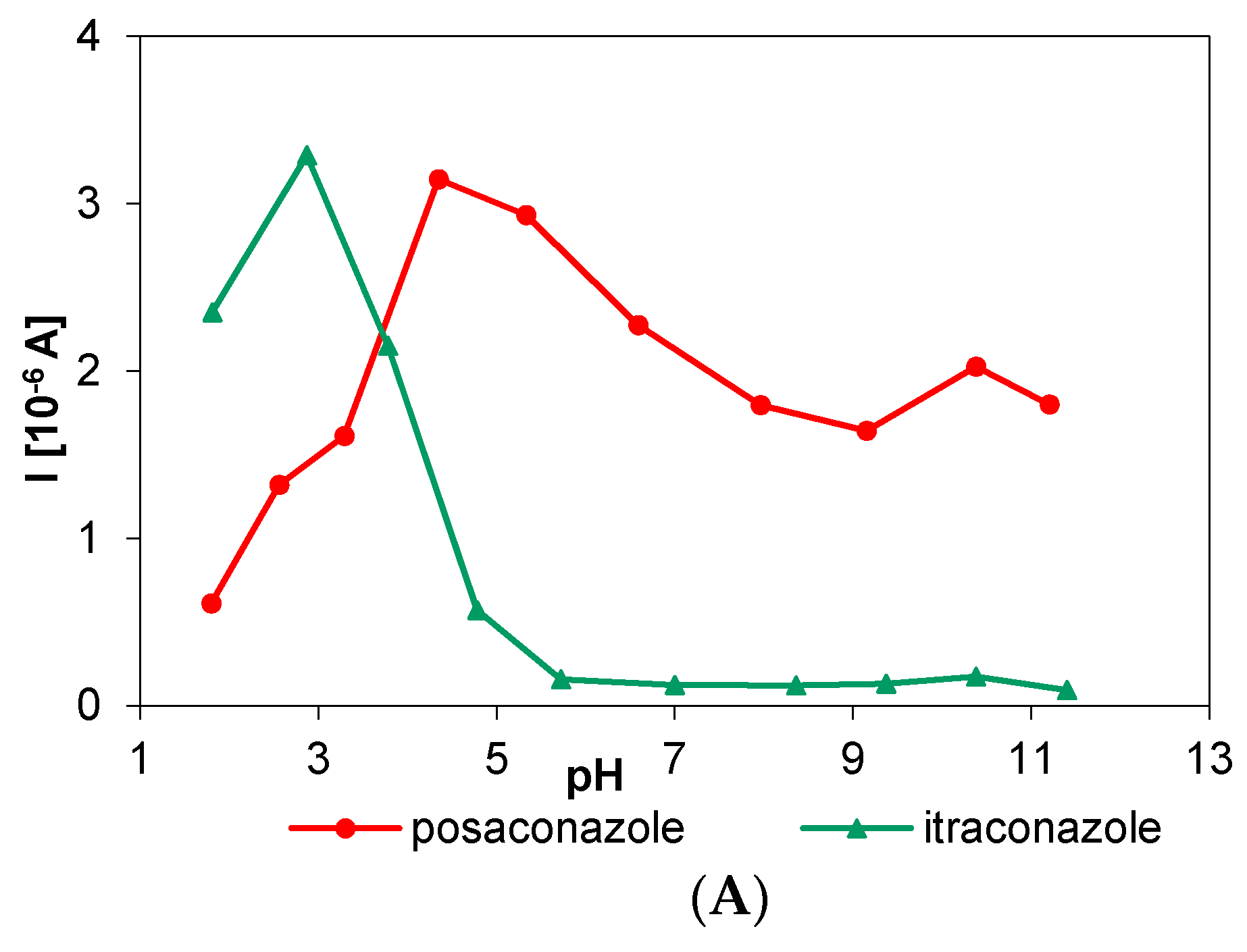
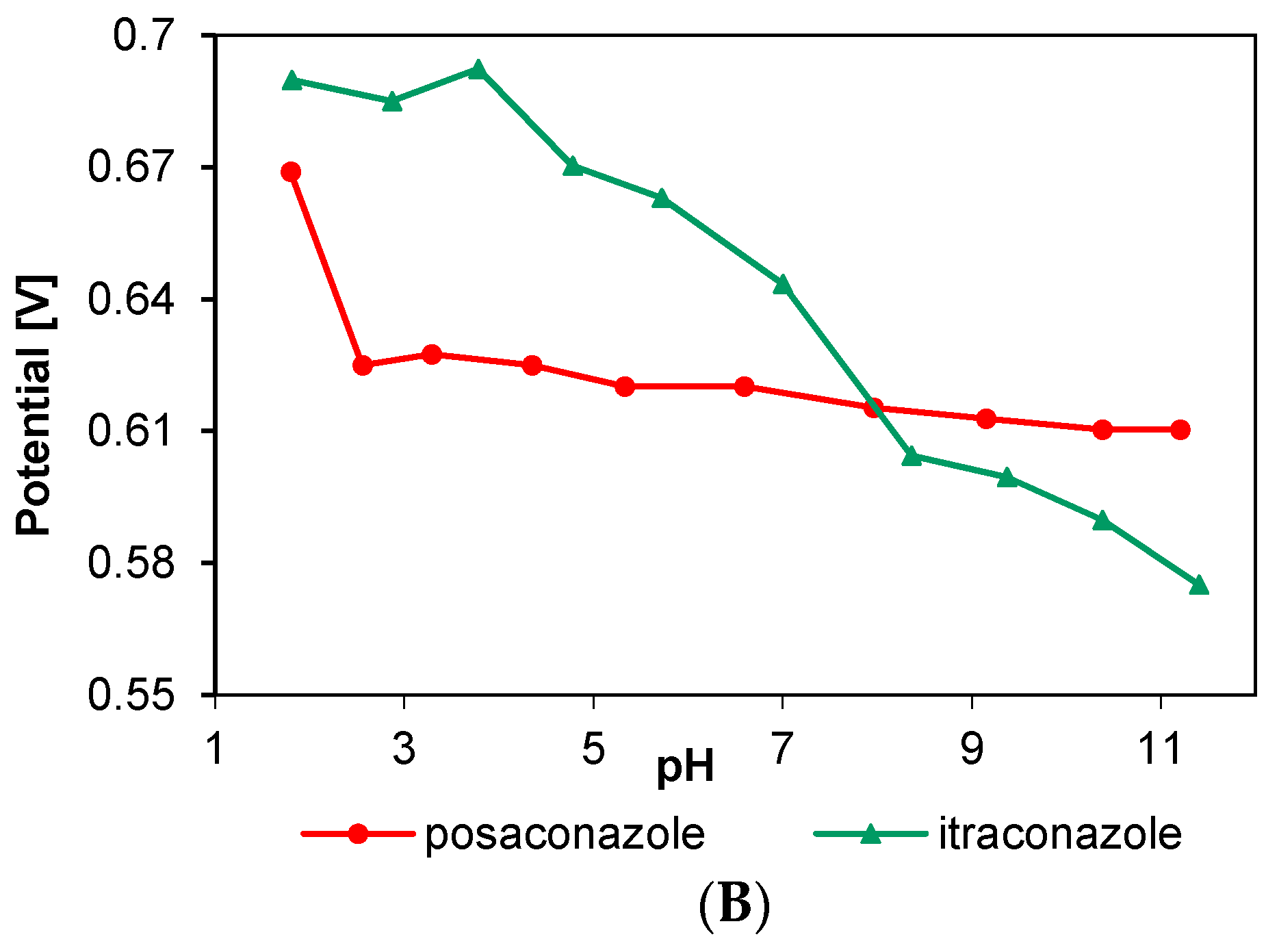
3.1. Electrochemical Behavior of Selected Biocides
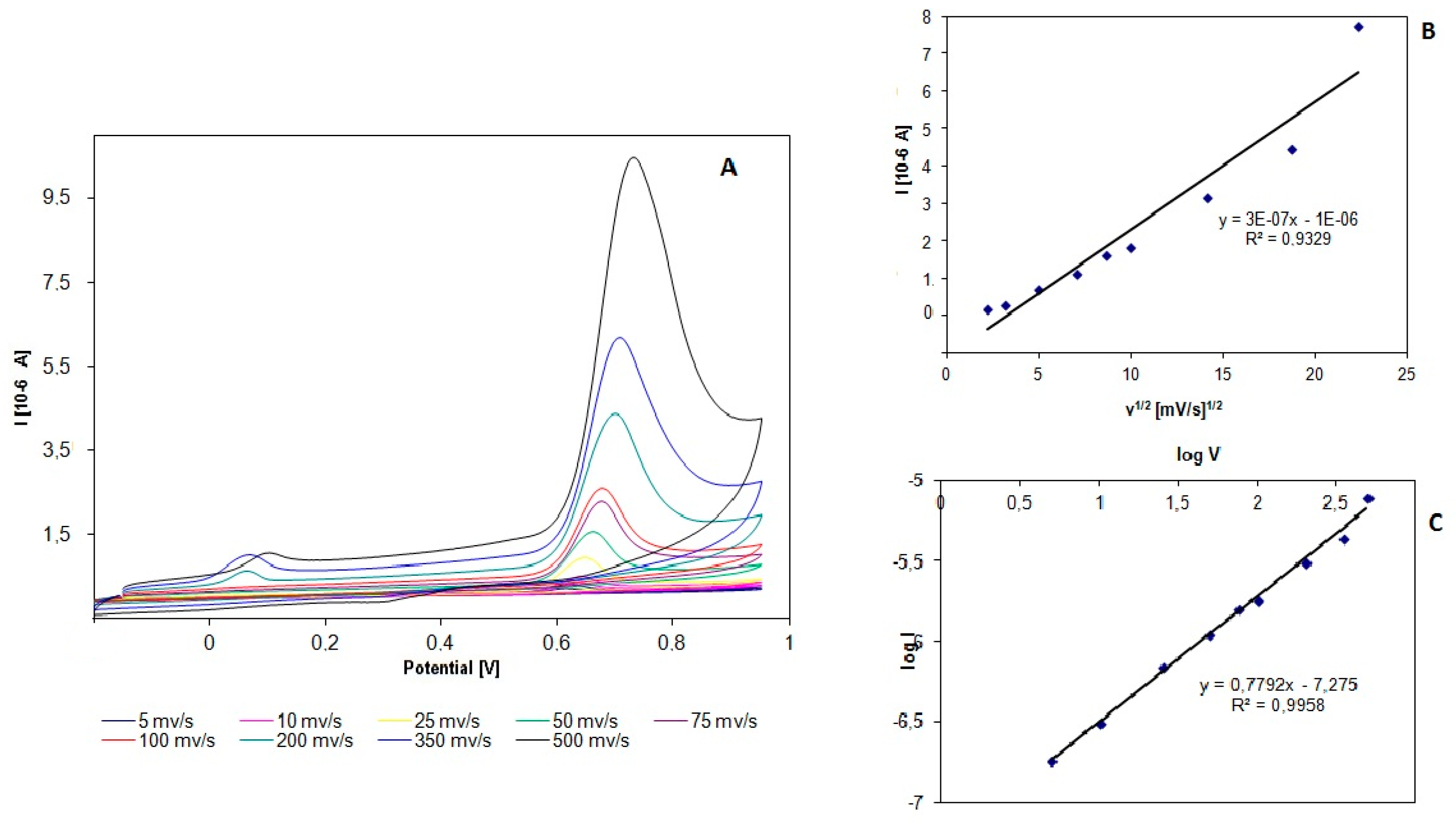
3.2. Effect of Scan Rate
- for itraconazole (ITR)I (µA) = 3·10−7 v1/2 (mVs−1)1/2 − 1·10−6
- for posaconazole (POZ)I (µA) = 9·10−8 v1/2 (mVs−1)1/2 − 1·10−7
- ITRlog Ipa (µA) = 0.7792 log v(mV/s) − 7275, R2 = 0.9958
- POZlog Ipa (µA) = 0.58 log v(mV/s) − 7295, R2 = 0.9996
3.3. Optimization of the Square Wave Parameters
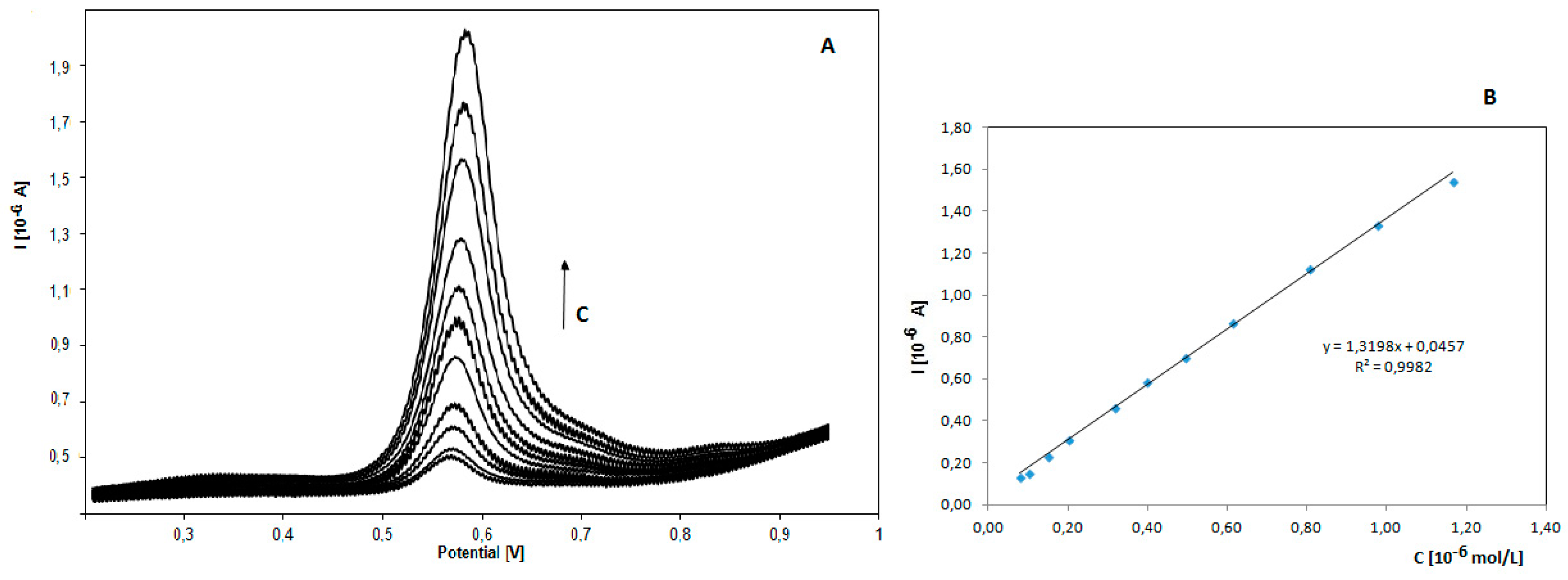
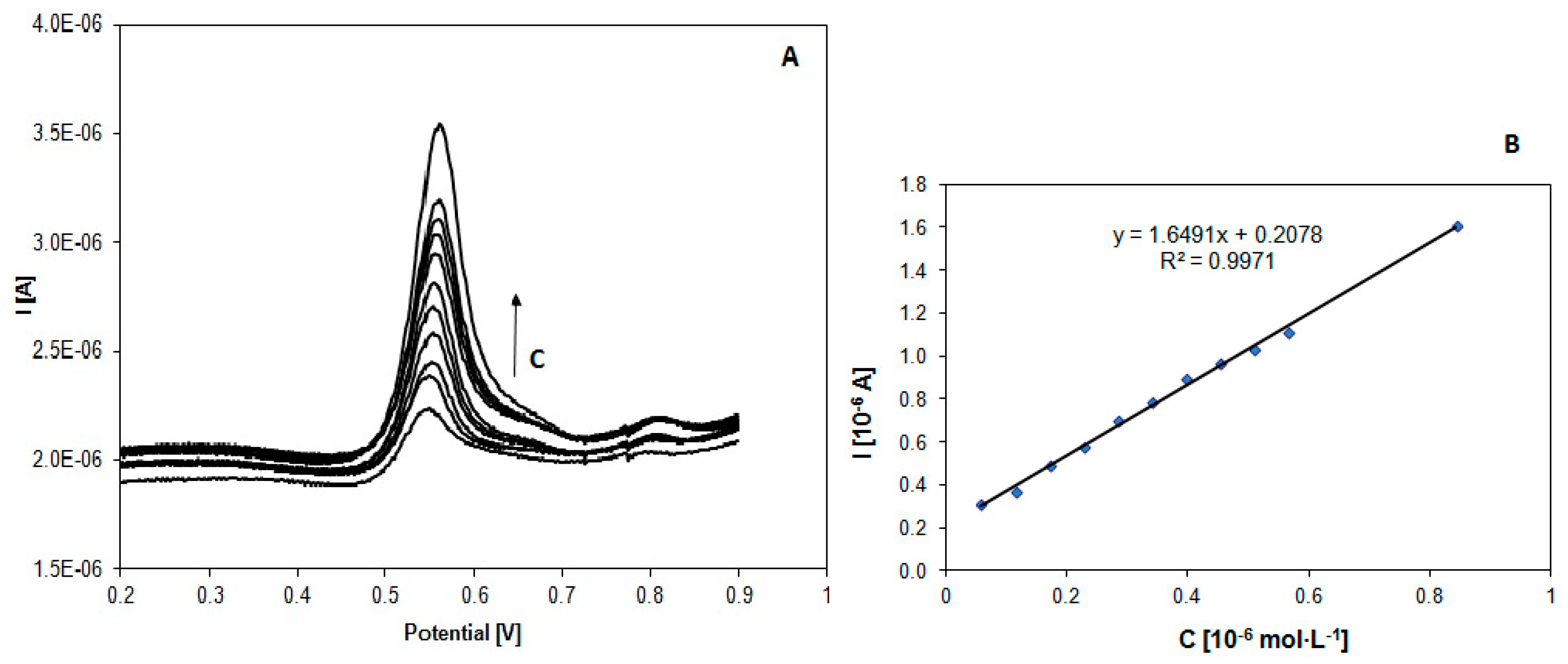
3.4. Analytical Curve for Itraconazole and Posaconazole Using SWV
3.5. Precision and Selectivity of Itraconazole Using SWV
3.6. Assay of Itraconazole and Posaconazole in the Water Samples
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gros, M.; Petrović, M.; Barcelo, D. Development of multi-residue analytical methodology based on liquid chromatography-tandem mass spectrometry (LC-MS/MS) for screening and trace level determination of pharmaceuticals in surface and wastewaters. Talanta 2006, 70, 678–690. [Google Scholar] [CrossRef]
- Zounkova, R.; Kovalova, L.; Blaha, L.; Dott, W. Ecotoxicity and genotoxicity assessment of cytotoxic antineoplastic drugs and their metabolites. Chemosphere 2010, 81, 253–260. [Google Scholar] [CrossRef]
- Stuart, M.; Lapworth, D.; Crane, E.; Hart, A. Review of risk from potential emerging contaminants in UK groundwater. Sci. Total Environ. 2012, 416, 1–21. [Google Scholar] [CrossRef]
- Hernando, M.D.; Gomez, M.J.; Aguera, A.; Fernandez-Alba, A.R. LC-MS analysis of basic pharmaceuticals (beta-blokers and anti-ulcer agent) in wastewater and surface water. Trends Anal. Chem. 2007, 26, 581–594. [Google Scholar] [CrossRef]
- Hyungkeun, R.; Subramanya, N.; Zhao, F.; Yu, C.; Sandt, J.; Chu, K. Biodegradation potential of wastewater micropollutants by ammonia-oxidizing bacteria. Chemosphere 2009, 77, 1087–1089. [Google Scholar]
- Sirés, I.; Brillas, E. Remediation of water pollution caused by pharmaceutical residues based on electrochemical separation and degradation technologies: A review. Environ. Int. 2012, 40, 212–229. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Directive 98/8/EC of the European Parliament and of the Council (Biocidal Products Directive (BPD) 98/8/EC). Off. J. Eur. Comm. 1998, 41, 123. [Google Scholar]
- Bester, K.; Scholes, L.; Wahlberg, C.; McArdell, C.S. Sources and mass flows of xenobiotics in urban water cycles—An overview on current knowledge and data gaps water. Air Soil Pollut. Focus 2008, 8, 407–4233. [Google Scholar] [CrossRef]
- Chen, Z.; Ying, G.; Lai, H.; Chen, F.; Su, H.; Liu, Y.; Peng, F.; Zhao, J. Determination of bocides in differential environmental matrices by use of ultra-high-performance liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2012, 404, 3175–3188. [Google Scholar] [CrossRef]
- Van De Steene, J.C.; Lambert, W.E. Validation of solid-phase extraction and liquid chromatography-electrospray tandem mass spectrometric method for the determination of nine basic pharmaceuticals in wastewater and surface water samples. J. Chromatogr. A 2008, 1182, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Van De Steene, J.C.; Lambert, W.E. Comparison of matrix effects in HPLC-MS/MS and UHPLC-MS/MS analysis of nine basic pharmaceuticals in surface waters. J. Am. Soc. Mass Spectrom. 2008, 19, 713–718. [Google Scholar] [CrossRef]
- Wang, Z.; Zhao, P.; Yu, J.; Jiang, Z.; Guo, X. Experimental and molecular docking study on grapheme/Fe3O4 composites as a sorbent for magnetic solid-phase extraction of seven imidazole antifungals in environmental water samples prior to LC-MS/MS for enantiomeric analysis. Microchem. J. 2018, 140, 222–231. [Google Scholar] [CrossRef]
- Lindberg, R.H.; Fick, J.; Tysklind, M. Screening of antimycotics in Swedish sewage treatment plants—Waters and sludge. Water Res. 2010, 44, 649–657. [Google Scholar] [CrossRef]
- Van De Steene, J.C.; Stove, C.P.; Lambert, W.E. A field study on 8 pharmaceuticals and 1 pesticide in Belgium: Removal rates in waste water treatment plants and occurrence in surface water. Sci. Total Environ. 2010, 408, 3448–3453. [Google Scholar] [CrossRef] [PubMed]
- Castro, G.; Roca, M.; Redriguez, I.; Ramil, M.; Cela, R. Identification and determination of chlorinated azoles in sludge using liquid chromatography quadrupole time-of-flight and triple quadrupole mass spectrometry platforms. J. Chromatogr. A 2016, 1476, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Fatta, D.; Nikolaou, A.; Achilleous, A.; Meric, S. Analytical methods for tracing pharmaceutical residues in water and wastewater. Trends Anal. Chem. 2007, 26, 515–535. [Google Scholar] [CrossRef]
- Renitaa, A.; Senthil, P.; Kumarb, P.; Srinivasb, S.; Priyadharshinib, S.; Karthikab, M. A review on analytical methods and treatment techniques of pharmaceutical wastewater. Desalin. Water Treat. 2017, 87, 160–178. [Google Scholar] [CrossRef]
- Peralta, E.; Natividad, R.; Roa, G.; Marin, R.; Romero, R.; Pavon, T. A comparative study on the electrochemical production of H2O2 between BDD and graphite cathodes. Sustain. Environ. Res. 2013, 23, 259–266. [Google Scholar]
- Panizza, M.; Brillas, E.; Comninellis, C. Application of boron-doped diamond electrodes for wastewater treatment. J. Environ. Eng. Manag. 2008, 18, 139–153. [Google Scholar]
- Einaga, Y. Diamond electrodes for electrochemical analysis. J. Appl. Electrochem. 2010, 40, 1807–1816. [Google Scholar] [CrossRef]
- Luong, J.; Male, K.; Glennon, J. Boron-doped diamond electrode: Synthesis, characterization, functionalization and analytical applications. Analyst 2009, 134, 1965–1979. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhi, J. The application of boron-doped diamond electrode in amperometric biosensors. Talanta 2009, 79, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Yu, S.; Macpherson, J.V.; Einaga, Y.; Zhao, H.; Zhao, G.; Swain, G.M.; Jiang, Y. Conductive diamond: Synthesis, properties, and electrochemical applications. Chem. Soc. Rev. 2019, 48, 157–204. [Google Scholar] [CrossRef] [PubMed]
- Muzyka, K.; Sun, J.; Fereja, T.; Lan, Y.; Zhang, W.; Xu, G. Boron-doped diamond: Current progress and challenges in view of electroanalytical applications. Anal. Methods 2019, 11, 397–414. [Google Scholar] [CrossRef]
- Pecková, K.; Musilová, J.; Barek, J. Boron-doped diamond film electrode—New tool for voltammetric determination of organic substances. Crit. Rev. Anal. Chem. 2009, 39, 148–172. [Google Scholar] [CrossRef]
- Mielech-Łukasiewicz, K.; Rogińska, K. Voltammetric determination of antifungal agents in pharmaceuticals and cosmetics using a boron-doped diamond electrode. Anal. Methods 2014, 6, 7912–7922. [Google Scholar] [CrossRef]
- Mielech-Łukasiewicz, K.; Dąbrowska, A. Comparison of boron-doped diamond and glassy carbon electrodes for determination of terbinafiny in pharmaceuticals using differential pulse and square wave voltammetry. Anal. Lett. 2014, 74, 1697–1711. [Google Scholar] [CrossRef]
- Yardim, Y.; Alpar, N.; Senturk, Z. Voltammetric sensing of triclosan in the presence of cetyltrimethylammonium bromide using a cathodically pretreated boron-doped diamond electrode. Int. J. Environ. Anal. Chem. 2018, 98, 1–16. [Google Scholar] [CrossRef]
- Brocenschi, R.; Rocha-Filho, R.; Biaggio, S.; Bocchi, N. DPV and SWV determination of estrone using a cathodically pretreated boron-doped diamond electrode. Electroanalysis 2014, 26, 1588–1597. [Google Scholar] [CrossRef]
- Sousa, C.P.; Ribeiro, F.W.P.; Oliveira, T.M.; Salazar-Banda, G.R.; de Lima-Neto, P.; Morais, S.; Correia, A.N. Electroanalysis of pharmaceuticals on boron-doped diamond Electrodes: A Review. ChemElectroChem 2019, 6, 2350–2378. [Google Scholar]
- Gayen, P.; Chaplin, B.P. Selective electrochemical detection of ciprofloxacin with a porous nafion/multi-walled carbon nanotube composite film electrode. ACS Appl. Mater. Interfaces 2016, 8, 1615–1626. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yuan, F.; Quan, X.; Yu, H.; Chen, S.; Zhao, H.; Liub, Z.; Hilalb, N. An electrochemical sensor for selective determination of sulfamethoxazole in surface water using a molecularly imprinted polymer modified BDD electrode. Anal. Methods 2015, 7, 2693–2698. [Google Scholar] [CrossRef]
- Radicova, M.; Behul, M.; Vojs, M.; Bodor, R.; Vojs Stano, A. Voltammetric determination of erythromycin in water samples using a boron-doped diamond electrode. Phys. Status Solidi B 2015, 252, 2608–2613. [Google Scholar] [CrossRef]
- Calisto, C.; Cervini, P.; Cavalheiro, E.T. Determination of tetracycline in environmental water samples at a graphite-polyurethane composite electrode. J. Braz. Chem. Soc. 2012, 23, 938–943. [Google Scholar] [CrossRef]
- Madej, M.; Kochana, J.; Baś, B. Determination of viloxazine by differential pulse voltammetry with boron-doped diamond electrode. Monatshefte Chem.-Chem. Mon. 2019. [Google Scholar] [CrossRef]
- Ardelean, M.; Manea, F.; Pop, A.; Schoonman, J. Carbon-based electrochemical detection of pharmaceuticals from water. Int. J. Environ. Ecol. Eng. 2016, 10, 1237–1242. [Google Scholar]
- Vojić, M.; Popović, P. Protolytic equilibria in homogeneous and heterogeneous systems of ketoconazole and its direct spectrophotometric determination in tablets. J. Serb. Chem. Soc. 2005, 70, 67–78. [Google Scholar] [CrossRef]
- Popa, O.M.; Diculescu, V.C. Electrochemical and spectrophotometric characterization of proteinkinase inhibitor and anticancer drug danusertib. Electrochim. Acta 2013, 112, 486–492. [Google Scholar] [CrossRef]
- Uslu, B.; Topal, N.; Ozkan, S.A. Electroanalytical investigation and determination of pefloxacin in pharmaceuticals and serum at boron-doped diamond electrodes. Talanta 2008, 74, 1191–1200. [Google Scholar] [CrossRef] [PubMed]
- Sanli, S.; Basaran, F.; Sanli, N.; Akmese, B.; Bulduk, A. Determination of dissociation constants of some antifungal drugs by two different methods at 298K. J. Solut. Chem. 2013, 1976–1987. [Google Scholar] [CrossRef]
- Courney, R.; Wexler, D.; Radwanski, E.; Lim, J.; Laughlin, M. Effect of food on the relative bioavailability of two oral formulations o posaconazole in healty aduits. Br. J. Clin. Pharmacol. 2004, 57, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Alshalalfeh, M.; Sohail, M.; Saleh, T.; Aziz, A. Electrochemical investigation of gold nanoparticle-modified glassy carbon electrode and its application in ketoconazole determination. Aust. J. Chem. 2016, 69, 1314–1320. [Google Scholar] [CrossRef]
- Galus, Z. Fundamentals of Electrochemical Analysis; Ellis Horwood Press: New York, NY, USA, 1994. [Google Scholar]
- Miller, J.C.; Miller, J.N. Statistics for Analytical Chemistry; Ellis Horwood: Chichester, UK; New York, NY, USA, 1988. [Google Scholar]
- Shalaby, A.; Hassan, W.S.; Hendawy, H.A.M.; Ibrahim, A.M. Electrochemical oxidation behavior of itraconazole at different electrodes and its anodic stripping determination in pharmaceuticals and biological fluids. J. Electroanal. Chem. 2016, 763, 51–62. [Google Scholar] [CrossRef]
- Knoth, H.; Knoth, H.; Scriba, G.K.E.; Buettner, B. Electrochemical behavior of the antifungal agents itraconazole, posaconazole and ketoconazole at a glassy carbon electrode. Pharmazie 2015, 70, 374–378. [Google Scholar] [PubMed]
- Knoth, H.; Petry, T.; Gartner, P. Differential pulse polarographic investigation of the antifungal drugs itraconazole, ketoconazole, fluconazole and voriconazole using a dropping mercury electrode. Pharmazie 2012, 67, 987–990. [Google Scholar]
- Sultan, M.A.; Attia, A.K.; El-Alamin, M.M.A.; Atia, M.A. The novel use of multwalled carbon nanotubes-based sensors for voltammetric determination of itraconazole: Application to pharmaceutical dosage form and biological samples through spiked urine sample. World J. Pharm. Sci. 2016, 5, 93–108. [Google Scholar]
- Reddy, T.M.; Tama, C.; Hayes, R.N. A dried blond spots technique based LC-MS/MS method for the analysis of posaconazole in human whole blood samples. J. Chromatogr. B 2011, 879, 3626–3638. [Google Scholar] [CrossRef]
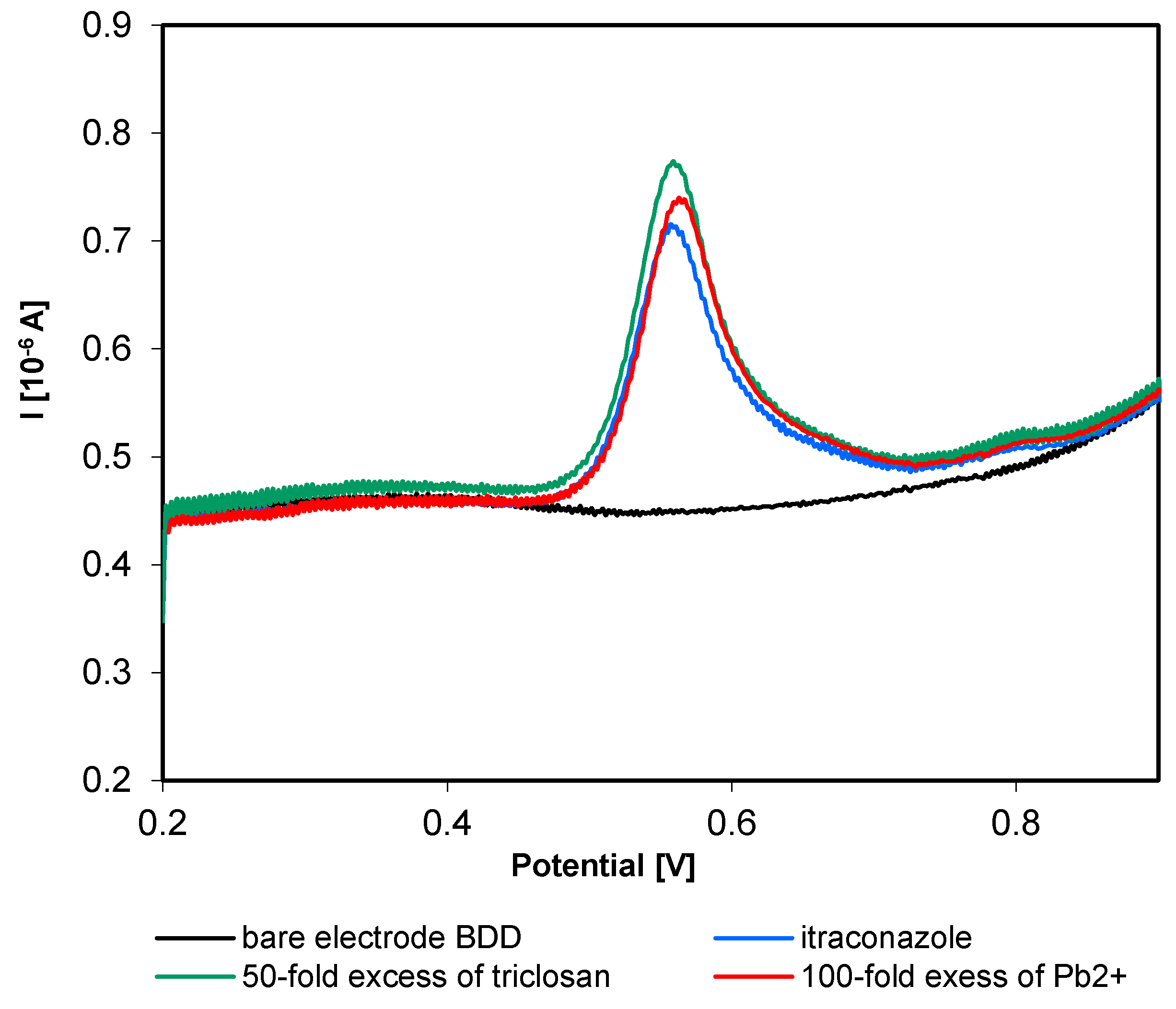
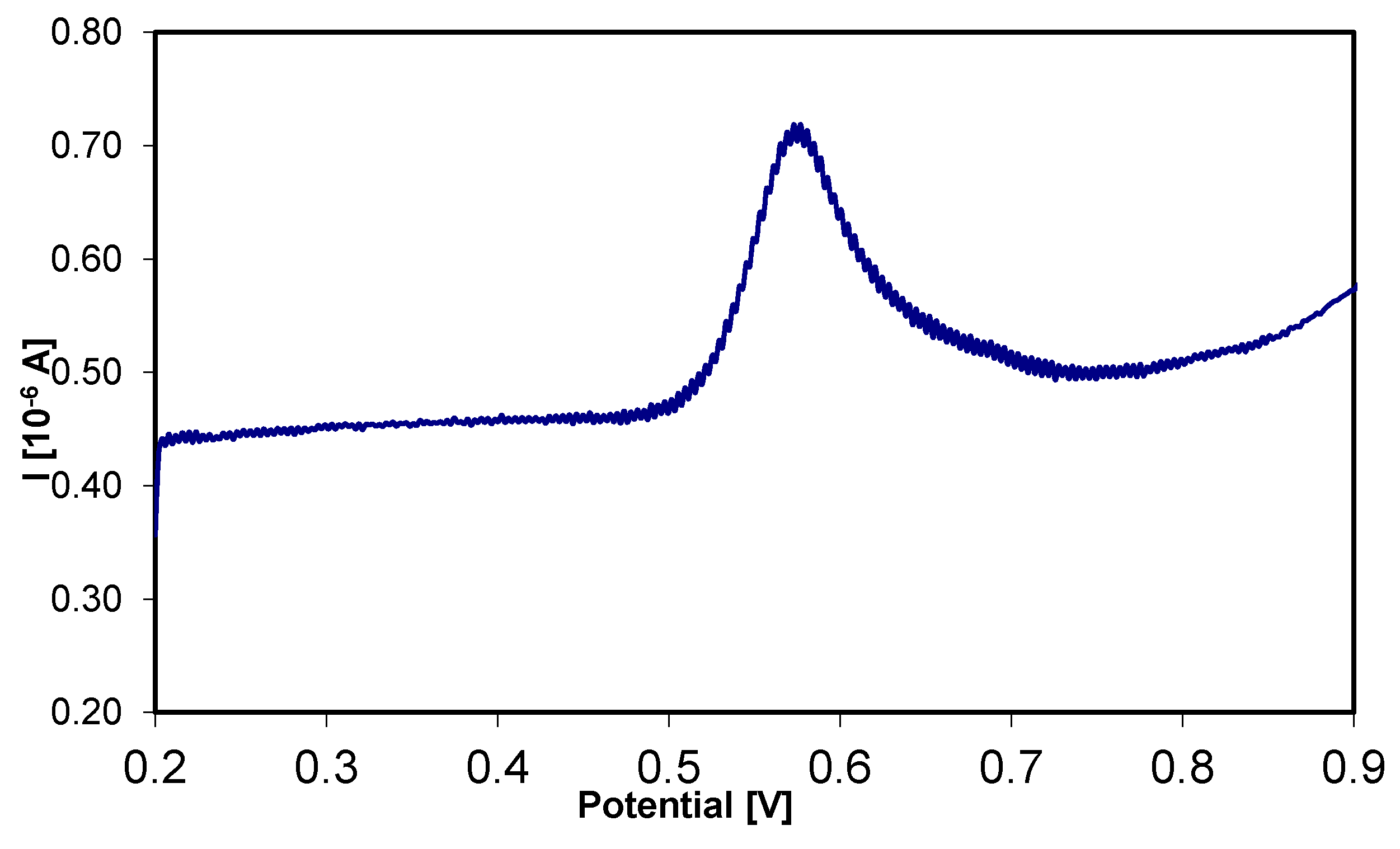
| Studied Substance | Itraconazole | Posaconazole |
|---|---|---|
| Peak potential/V vs. SCE | 0.59 | 0.55 |
| Peak width half/mV | 0.06 | 0.08 |
| Linearity range/mol·L−1 | 7.9 × 10−8–1.2 × 10−6 | 5.7 × 10−8–8.44 × 10−7 |
| Slope/µA·L/mol | 1.32×106 | 1.65 × 106 |
| Intercept/µA | 0.046 | 0.2 |
| Correlation coefficient | 0.9982 | 0.9971 |
| LOQ/mol·L−1 | 5.43 × 10−8 | 2.36 × 10−8 |
| LOD/mol·L−1 | 1.79 × 10−8 | 7.78 × 10−9 |
| Repeatability of Ip/RSD% | 5.68 | 1.73 |
| Reproducibility of Ip/RSD% | 2.61 | 1.93 |
| Linear Range (mol·L−1) | Detection Limit (mol·L−1) | Method | Electrode | Ref. |
|---|---|---|---|---|
| 2.2 × 10−8–2.9 × 10−7 | 1.9 × 10−8 | UTG | [45] | |
| 1.5 × 10−8–2.3 × 10−7 | 1.2 × 10−8 | AS-SWV | PG | |
| 1.5 × 10−8–1.5 × 10−7 | 8.5 × 10−9 | CP | ||
| 2.2 × 10−8–2.5 × 10−7 | 1.5 × 10−8 | UTG | ||
| 4.5 × 10−8–2.3 × 10−7 | 1.2 × 10−8 | AS-DPV | PG | |
| 1.5 × 10−8–1.5 × 10−7 | 1.1 × 10−8 | CP | ||
| 2.8 × 10−5–1.4 × 10−4 | - | CV | GC | [46] |
| 5.0 × 10−7–5.0 × 10−6 | - | DPV | Hg | [47] |
| 2.19 × 10−6–6.33 × 10−5 | 7.27 × 10−7 | CV | MWCNT/CP | [48] |
| 7.9 × 10−8–1.2 × 10−6 | 1.79 × 10−8 | SWV | BDD | This work |
| Sample | Amount Added/×10−7 mol/L | Amount Received a/×10−7 mol/L | Recovery a/% | RSD/% |
|---|---|---|---|---|
| Biała water | 1.500 | 1.437 ± 0.069 | 95.8 ± 4.5 | 4.8 |
| 3.000 | 2.882 ± 0.107 | 96.1 ± 3.6 | 3.7 | |
| 4.500 | 4.545 ± 0.102 | 101 ± 2.2 | 2.2 | |
| Tap water | 1.500 | 1.403 ± 0.059 | 93.5 ± 3.9 | 4.2 |
| 3.000 | 2.919 ± 0.056 | 97.3 ± 1.9 | 1.9 | |
| 4.500 | 4.350 ± 0.145 | 96.7 ± 3.2 | 3.3 |
| Sample | Amount Added/×10−7 mol/L | Amount Received a/×10−7 mol/L | Recovery a/% | RSD/% |
|---|---|---|---|---|
| Biała water | 1.500 | 1.438 ± 0.052 | 95.8 ± 3.4 | 3.6 |
| 3.000 | 2.821 ± 0.079 | 94.0 ± 2.6 | 2.8 | |
| 4.500 | 4.399 ± 0.115 | 97.7 ± 2.5 | 2.6 | |
| Tap water | 1.500 | 1.447 ± 0.023 | 96.5 ± 1.6 | 1.6 |
| 3.000 | 2.886 ± 0.102 | 96.2 ± 3.4 | 3.5 | |
| 4.500 | 4.627 ± 0.055 | 102.8 ± 1.2 | 1.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mielech-Łukasiewicz, K.; Starczewska, B. The Use of Boron-Doped Diamond Electrode for the Determination of Selected Biocides in Water Samples. Water 2019, 11, 1595. https://doi.org/10.3390/w11081595
Mielech-Łukasiewicz K, Starczewska B. The Use of Boron-Doped Diamond Electrode for the Determination of Selected Biocides in Water Samples. Water. 2019; 11(8):1595. https://doi.org/10.3390/w11081595
Chicago/Turabian StyleMielech-Łukasiewicz, Katarzyna, and Barbara Starczewska. 2019. "The Use of Boron-Doped Diamond Electrode for the Determination of Selected Biocides in Water Samples" Water 11, no. 8: 1595. https://doi.org/10.3390/w11081595
APA StyleMielech-Łukasiewicz, K., & Starczewska, B. (2019). The Use of Boron-Doped Diamond Electrode for the Determination of Selected Biocides in Water Samples. Water, 11(8), 1595. https://doi.org/10.3390/w11081595





