Nitrogen and Phosphorus Budget for a Deep Tropical Reservoir of the Brazilian Savannah
Abstract
1. Introduction
2. Materials and Methods
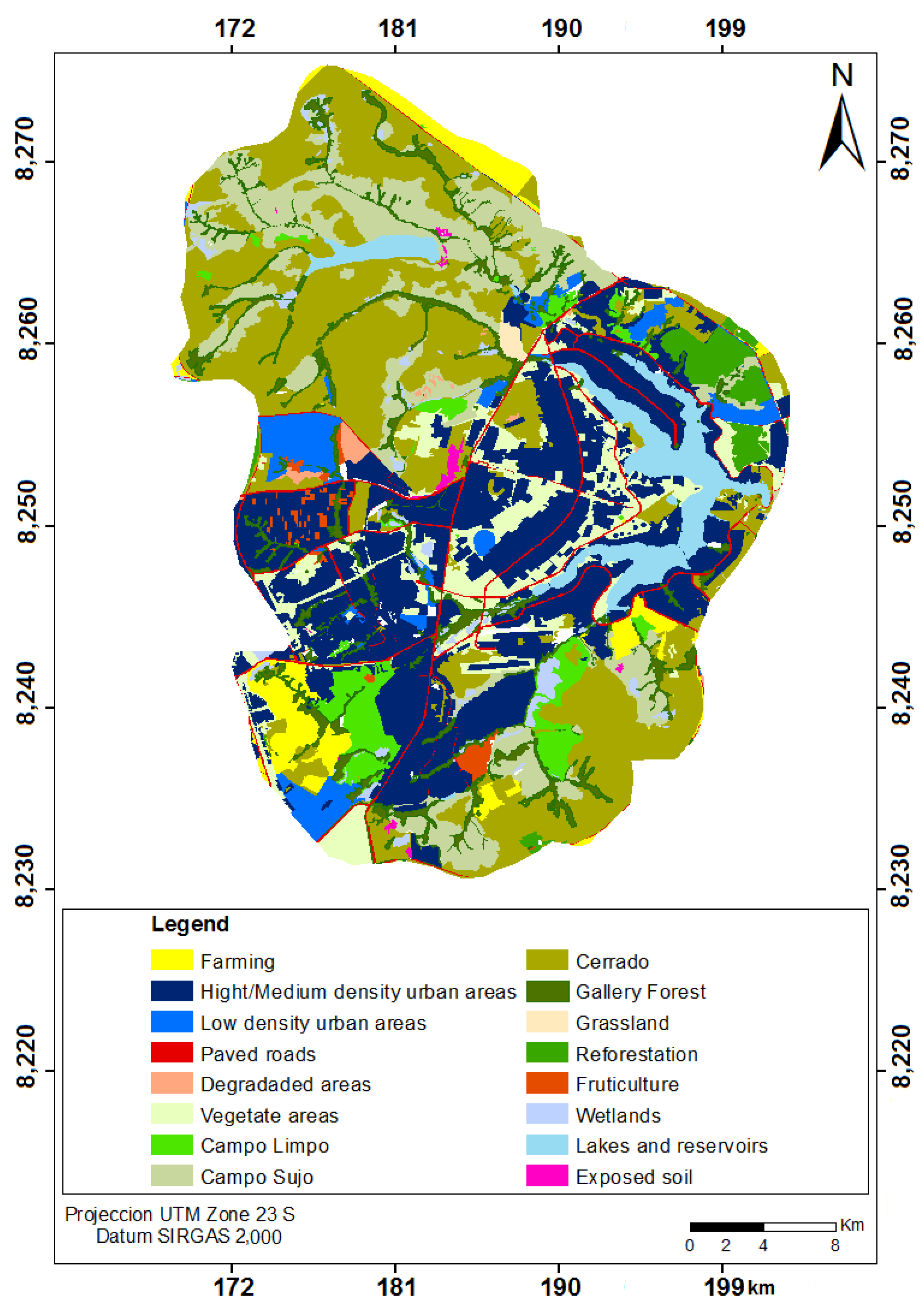
2.1. Study Area
2.2. Sample Collection
2.2.1. Tributaries and Downstream Dam
2.2.2. Lake Water Column
2.2.3. Wastewater Treatment Plant
2.2.4. Pore Water
2.2.5. Sediment
2.3. Sample Preparation and Chemical Analyses
2.3.1. Water Column (Tributaries and Lake)
2.3.2. Wastewater Treatment Plant
2.3.3. Pore Water
2.3.4. Sediment
2.4. N and P Loads Calculations
2.4.1. External Inputs
2.4.2. Internal Input
2.4.3. Output
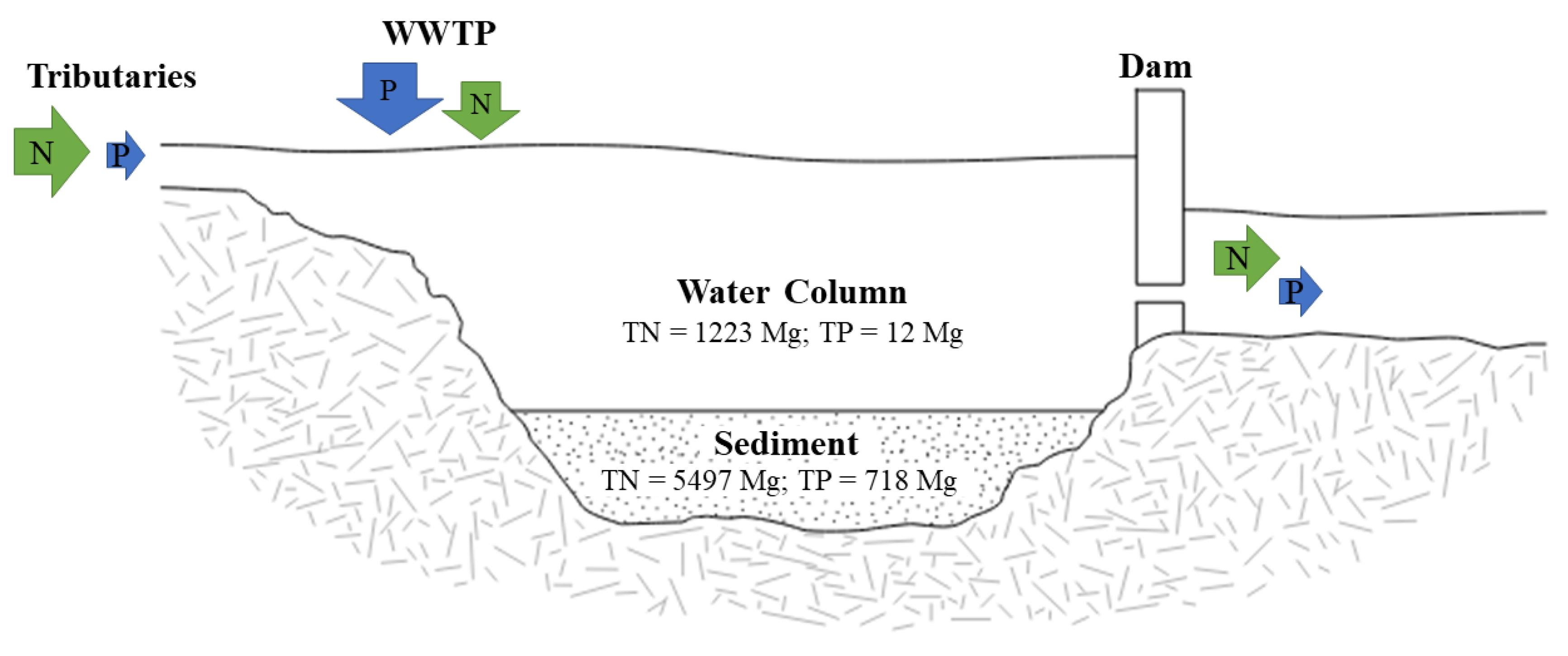
2.5. N and P Storage Calculations
2.5.1. Lake Water Column
2.5.2. Sediment
2.6. N and P Budget Calculations
3. Results and Discussion
3.1. N and P Dynamic in the Lake Paranoá Catchment
- Significantly higher mean concentration (p < 0.05) of PO43−-P in the Riacho Fundo stream in the dry season,
- Significantly higher mean concentration (p < 0.05) of TOP in the Gama stream in the wet season.
3.2. N and P Load and Storage
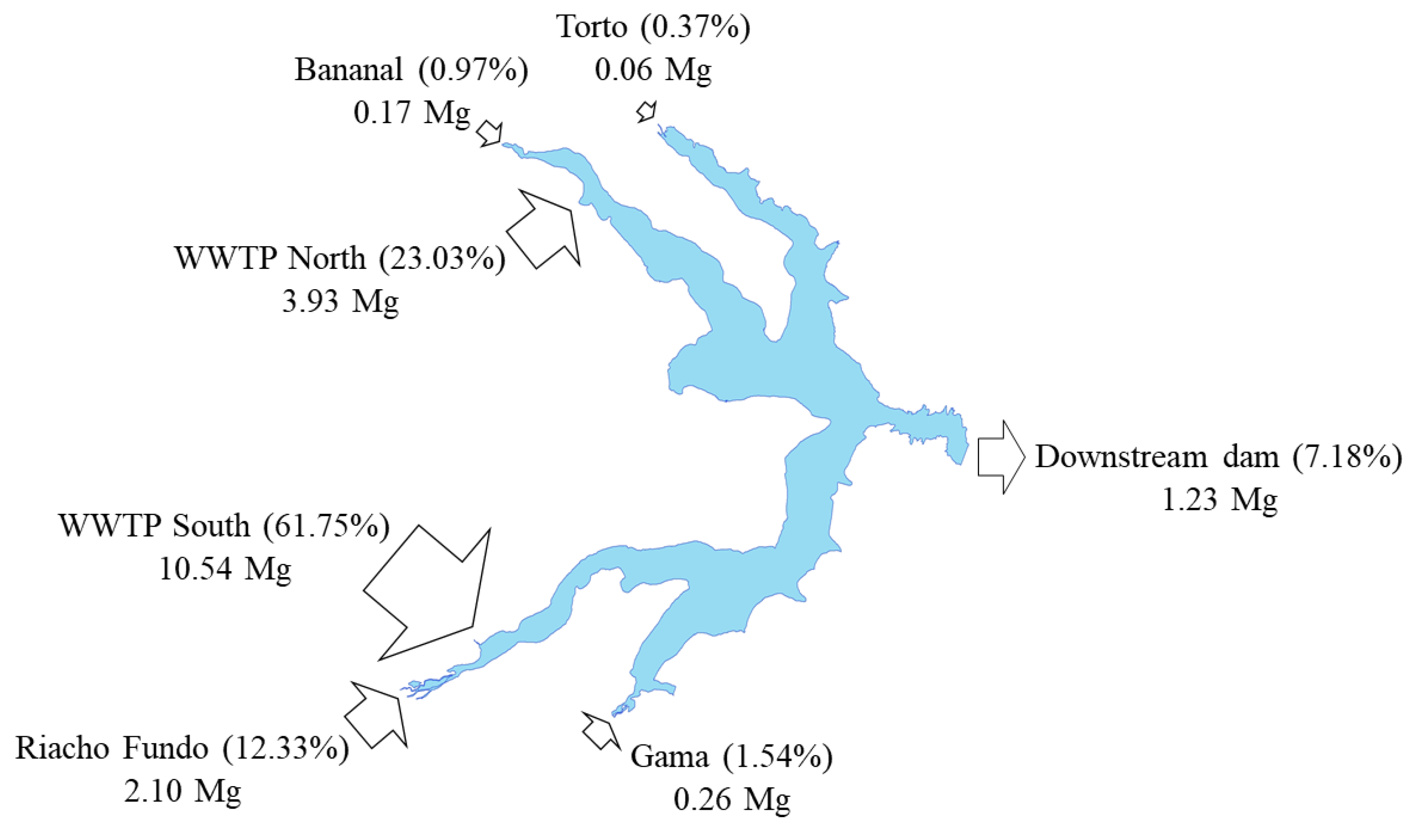
3.3. N and P Budget
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wetzel, R.G.; Likens, G.E. Limnological Analysis, 2nd ed.; Springer: New York, NY, USA, 1991. [Google Scholar]
- Zamparas, M.; Zacharias, I. Restoration of eutrophic freshwater by managing internal nutrient loads. Sci. Total Environ. 2014, 496, 551–562. [Google Scholar] [CrossRef]
- Nikolai, S.J.; Dzialowski, A.R. Effects of internal phosphorus loading on nutrient limitation in a eutrophic reservoir. Limnologica 2014, 49, 33–41. [Google Scholar] [CrossRef]
- Waters, S.; Webster-Brown, J.G. The use of a mass balance phosphorus budget for informing nutrient management in shallow coastal lakes. J. Hydro-Environ. Res. 2016, 10, 32–49. [Google Scholar] [CrossRef]
- Codd, G.A.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins: Risk management for health protection. Toxicol. Appl. Pharmacol. 2005, 203, 264–272. [Google Scholar] [CrossRef]
- Havens, K.E.; Fukshima, T.; Xie, P.; Iwakuma, T.; James, R.T.; Takamura, N.; Hanazato, T.; Yamamoto, T. Nutrient dynamics and the eutrophication of shallow lakes Kasumigaura (Japan), Donghu (PR China), and Okeechobee (USA). Environ. Pollut. 2001, 111, 263–272. [Google Scholar] [CrossRef]
- Kõiv, T.; Nõges, T.; Laas, A. Phosphorus retention as a function of external loading, hydraulic turnover time, area and relative depth in 54 lakes and reservoirs. Hydrobiologia 2011, 660, 105–115. [Google Scholar] [CrossRef]
- Moosmann, L.; Gächter, R.; Müller, B.; Wüest, A. Is phosphorus retention in autochthonous lake sediments controlled by oxygen or phosphorus? Limnol. Oceanogr. 2006, 51, 763–771. [Google Scholar] [CrossRef]
- Ramírez-Zierold, J.A.; Merino-Ibarra, M.; Monroy-Ríos, E.; Olson, M.; Castillo, F.S.; Gallegos, M.E.; Vilaclara, G. Changing water, phosphorus and nitrogen budgets for Valle de Bravo reservoir, water supply for Mexico City Metropolitan Area. Lake Reserv. Manag. 2010, 26, 23–34. [Google Scholar] [CrossRef]
- Schernewski, G. Nutrient budgets, dynamics and storm effects in a eutrophic, stratified baltic lake. Acta Hydrochim. Hydrobiol. 2003, 31, 152–161. [Google Scholar] [CrossRef]
- Coppens, J.; Ozen, A.; Tavsanoglu, U.N.; Erdogan, S.; Levi, E.E.; Yozgatligil, C.; Jeppesen, E.; Beklioglu, M. Impact of alternating wet and dry periods on long-term seasonal phosphorus and nitrogen budgets of two shallow Mediterranean lakes. Sci. Total Environ. 2016, 563, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Ul Solim, S.; Wanganeo, A. Excessive phosphorus loading to Dal Lake, India: Implications for managing shallow eutrophic lakes in urbanized watersheds. Int. Rev. Hydrobiol. 2008, 93, 148–166. [Google Scholar] [CrossRef]
- Garnier, J.; Leporcq, B.; Sanchez, N.; Philippon. Biogeochemical mass-balances (C, N, P, Si) in three large reservoirs of the Seine Basin (France). Biogeochemistry 1999, 47, 119–146. [Google Scholar]
- Morabito, G.; Rogora, M.; Austoni, M.; Ciampittiello, M. Could the extreme meteorological events in Lake Maggiore watershed determine a climate-driven eutrophication process? Hydrobiologia 2018, 824, 163–175. [Google Scholar] [CrossRef]
- Tomaszek, J.A.; Koszelnik, P. A simple model of nitrogen retention in reservoirs. Hydrobiologia 2003, 504, 51–58. [Google Scholar] [CrossRef]
- Nishri, A.; Hamilton, D.P. A mass balance evaluation of the ecological significance of historical nitrogen fluxes in Lake Kinneret. Hydrobiologia 2010, 655, 109–119. [Google Scholar] [CrossRef]
- Woldeab, B.; Beyene, A.; Ambelu, A. Seasonal and spatial variation of reservoir water quality in the southwest of Ethiopia. Environ. Monit. Assess. 2018, 190, 163. [Google Scholar] [CrossRef]
- Crowe, S.A.; O’Neill, A.H.; Katsev, S.; Hehanussa, P.; Haffner, G.D.; Sundby, B. The biogeochemistry of tropical lakes: A case study from Lake Matano, Indonesia. Limnol. Oceanogr. 2008, 53, 319–331. [Google Scholar] [CrossRef]
- Lewis, W.M. Tropical limnology. Ann. Rev. Ecol. Syst. 1987, 18, 159–184. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Reynolds, S.N.; Munawar, I.F.; Munamar, M. The regulation of phytoplankton population dynamics in the world’s largest lakes. Aquat. Ecosyst. Heal. Manag. 2000, 3, 1–21. [Google Scholar] [CrossRef]
- Ferrante, J.E.T.; Rancan, L.; Braga Netto, P.; Meio, F. Olhares sobre o Lago Paranoá, 1st ed.; Fonseca, F.O., Braga Netto, P., Cavalvante, C.V., Eds.; Secretaria de Meio Ambiente e Recursos Hídricos: Brasília, Brasil, 2001; Volume 1, pp. 45–79.
- Projeções e Estimativas da População do Brasil e das Unidades da Federação. Available online: https://www.ibge.gov.br/apps/populacao/projecao/index.html (accessed on 12 February 2019).
- Braga, P.N. Sustentabilidade ameaçada. In Olhares Sobre o Lago Paranoá, 1st ed.; Fonseca, F.O., Braga Netto, P., Cavalvante, C.V., Eds.; Secretaria de Meio Ambiente e Recursos Hídricos: Brasília, Brasil, 2001; Volume 1, pp. 167–188. [Google Scholar]
- GDF. 3.1—Relatório do Meio Físico e Biótico; Zoneamento Ecológico-Econômico do (ZEE): Brasília, Brasil, 2009; pp. 1–172.
- Mar da Costa, N.Y.; Boaventura, G.R.; Mulholland, D.S.; Araújo, D.F.; Moreira, R.C.A.; Faial, K.C.F.; Bomfim, E.O. Biogeochemical mechanisms controlling trophic state and micropollutant concentrations in a tropical artificial lake. Environ. Earth Sci. 2016, 75, 854. [Google Scholar] [CrossRef]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World map of the Köppen-Geiger climate classification updated. Meteorol. Zeitschrift 2006, 15, 259–263. [Google Scholar]
- Levantamento da Geodiversidade. Projeto Atlas Pluviométrico do Brasil. Isoietas Anuais Médias. Período 1977 a 2006. Available online: http://www.cprm.gov.br/publique/media/hidrologia/mapas_publicacoes/atlas_pluviometrico_brasil/isoietas_totais_anuais_1977_2006.pdf (accessed on 6 April 2019).
- Nunes, G. Aplicação do modelo SWAT no estudo hidrológico e de qualidade de água da Bacia Hidrográfica do lago Paranoá—DF. Master’s Thesis, Universidade de Brasília, Brasília, Brazil, August 2016. [Google Scholar]
- Carmouze, J.-P.; Bellotto, V.R.; Maddock, J.; Romanazzi, A. A versatile in situ sediment pore water sampler. Mangroves Salt Marshes 1997, 1, 77–78. [Google Scholar] [CrossRef]
- American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF). Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Pellens, I.C.; Bellotto, V.R.; Kuroshima, K.N.; Abreu, J.G.N. Resultados preliminares de fósforo e suas formas nos Sedimentos da plataforma continental do Estado de Santa Catarina. Braz. J. Aquat. Sci. Technol. 1998, 2, 51–60. [Google Scholar] [CrossRef]
- Mentzafou, A.; Dimitriou, E. Science of the Total Environment Nitrogen loading and natural pressures on the water quality of a shallow Mediterranean lake. Sci. Total Environ. 2019, 646, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Berner, R.A. Early Diagenesis: A theoretical Approach, 1st ed.; Princeton University Press: Princeton, NJ, USA, 1980. [Google Scholar]
- Klump, J.V.; Martens, C.S. Biogeochemical cycling in an organic rich coastal marine basin-II. Nutrient sediment-water exchange processes. Geochim. Cosmochim. Acta 1981, 45, 102–121. [Google Scholar] [CrossRef]
- Li, Y.-H.; Gregory, S. Diffusion of Ions in Sea Water and in Deep Sea Sediments. Geochim. Cosmochim. Acta 1974, 38, 703–714. [Google Scholar]
- Berner, R.A. Principles of Chemical Sedimentology, 1st ed.; MacGraw-Hi: New York, NY, USA, 1971. [Google Scholar]
- Torres, I.C.; Resck, R.P.; Pinto-Coelho, C.R. Mass balance estimation of nitrogen, carbon, phosphorus and total suspended solids in the urban eutrophic, Pampulha reservoir, Brazil. Acta Limnol. Bras. 2007, 19, 79–91. [Google Scholar]
- Morales-Marín, L.A.; Wheater, H.S.; Lindenschmidt, K.E. Assessment of nutrient loadings of a large multipurpose prairie reservoir. J. Hydrol. 2017, 550, 166–185. [Google Scholar] [CrossRef]
- Howarth, R.W.; Roxane, M.; Lane, J.; Cole, J.J. Nitrogen fixation in freshwater, estuarine, and marine ecosystems. 1. Rates and importance. Limnol. Ocean. 1988, 33, 669–687. [Google Scholar] [CrossRef]
- Cavalcante, H.; Araújo, F.; Noyma, N.P.; Becker, V. Phosphorus fractionation in sediments of tropical semiarid reservoirs. Sci. Total Environ. 2018, 620, 1022–1029. [Google Scholar] [CrossRef]
- Cook, P.L.M.; Aldridge, K.T.; Lamontagne, S.; Brookes, J.D. Retention of nitrogen, phosphorus and silicon in a large semi-arid riverine lake system. Biogeochemistry 2010, 99, 49–63. [Google Scholar] [CrossRef]
- Matson, P.A.; McDowell, W.H.; Townsend, A.R.; Vitousek, P.M. The globalization of N deposition: Ecosystem\nconsequences in tropical environments. Biogeochemistry 1999, 46, 67–83. [Google Scholar] [CrossRef]
- Søndergaard, M.; Jensen, J.P.; Jeppesen, E. Role of sediment and internal loading of phosphorus in shallow lakes. Hydrobiologia 2003, 506–509, 135–145. [Google Scholar]
- Saraiva, A.L.L. Análise dos Impactos da Dinâmica de Sólidos e de Nutrientes na Qualidade da Água de Ambiente Lêntico Utilizado no Abastecimento de Comunidades Amazônicas. Ph.D. Thesis, Universidade Federal do Pará, Belém, Brazil, August 2012. [Google Scholar]
- Cope, V.; Mercante, C.T.J.; Carmo, C.F.; Sendacz, S.; Monteiro Junior, A.J. Mass balance of nutrients during the filling phase of two reservoirs of Sistema Produtor Alto Tietê (SPAT). Acta Sci. Biol. Sci. 2011, 33, 49–57. [Google Scholar] [CrossRef][Green Version]
- Miranda, S.A.; Matvienko, B. Rain and groundwater as phosphorus sources of a small reservoir. Lakes Reserv. Res. Manag. 2003, 8, 27–30. [Google Scholar] [CrossRef]
- Bezerra, A.F.M.; Becker, V.; Mattos, A. Balanço de Massa de Fósforo Total e o Estado Trófico em Reservatórios do Semiárido Brasileiro. Rev. Bras. Recur. Hídricos 2014, 19, 67–76. [Google Scholar] [CrossRef]
- Oliveira, J.N.P. A Influência da Poluição Difusa e do Regime Hidrológico Peculiar do Semiarido na Qualidade da Água de um Reservatório Tropical. Master’s Thesis, Universidade Federal do Rio Grande do Norte, Natal, Brazil, 2012. [Google Scholar]
- Vidal, T.F.; Capelo, J.N. Dinâmica de nitrogênio e fósforo em reservatório na região semiárida utilizando balanço de massa. Rev. Bras. Eng. Agríc. Ambient. 2014, 18, 402–407. [Google Scholar] [CrossRef]
- Henry, R.; Do Carmo, C.F.; Bicudo, D.D.C. Trophic status of a Brazilian urban reservoir and prognosis about the recovery of water quality. Acta Limnol. Bras. 2004, 16, 251–262. [Google Scholar]
- Kenney, W.F.; Whitmore, T.J.; Buck, D.G.; Brenner, M.; Curtis, J.H.; Di, J.J.; Kenney, P.L.; Lchelske, C.L. Whole-basin, mass-balance approach for identifying critical phosphorus-loading thresholds in shallow lakes. J. Paleolimnol. 2014, 51, 515–528. [Google Scholar] [CrossRef]
- Gibson, C.E.; Wang, G.; Foy, R.H.; David Lennox, S. The importance of catchment and lake processes in the phosphorus budget of a large lake. Chemosphere 2000, 42, 215–220. [Google Scholar] [CrossRef]
- Nõges, P. Water and nutrient mass balance of the partly meromictic temperate Lake Verevi. Hydrobiologia 2005, 547, 21–31. [Google Scholar] [CrossRef]



| Input/Output/Lake | Flow (m3 s−1) | NH4+-N (mg L−1) | NO2−-N (mg L−1) | NO3−-N (mg L−1) | TON (mg L−1) | PO43−-P (mg L−1) | TOP (mg L−1) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wet | Dry | Wet | Dry | Wet | Dry | Wet | Dry | Wet | Dry | Wet | Dry | Wet | Dry | |
| Torto Stream | 0.6 | 0.090 | 0.038 ± 0.02 | 0.024 ± 0.02 | 0.010 ± 0.01 | 0.004 ± 0.00 | 0.30 ± 0.18 | 0.24 ± 0.07 | 1.3 ± 1.19 | 1.5 ± 1.10 | 0.002 ± 0.00 | <DL | 0.005 ± 0.01 | 0.014 ± 0.02 |
| Bananal Stream | 1.4 | 0.83 | 0.007 ± 0.00 | 0.006 ± 0.00 | 0.002 ± 0.00 | 0.002 ± 0.00 | 0.032 ± 0.02 | 0.059 ± 0.02 | 2.0 ± 1.25 | 1.9 ± 1.47 | 0.002 ± 0.00 | 0.002 ± 0.00 | 0.008 ± 0.01 | <DL |
| Riacho Fundo Stream | 5.1 | 2.1 | 0.056 ± 0.05 | 0.257 ± 0.31 | 0.039 ± 0.02 | 0.052 ± 0.03 | 0.99 ± 0.31 | 1.5 ± 0.55 | 0.88 ± 1.22 | 0.67 ± 1.03 | 0.002 ± 0.00 | 0.006 ± 0.00 | 0.024 ± 0.02 | 0.009 ± 0.01 |
| Gama Stream | 2.0 | 0.64 | 0.011 ± 0.00 | 0.015 ± 0.01 | 0.002 ± 0.00 | 0.001 ± 0.00 | 0.17 ± 0.16 | 0.087 ± 0.03 | 1.7 ± 1.03 | 1.8 ± 1.27 | 0.002 ± 0.00 | 0.002 ± 0.00 | 0.008 ± 0.01 | 0.003 ± 0.01 |
| WWTP North | 0.45 | 0.46 | 2.5 ± 2.59 | 7.3 ± 3.97 | NA | NA | NA | NA | NA | NA | 2.6 ± 2.60 | 4.5 ± 3.97 | 0.35 ± 0.08 | 0.31 ± 0.10 |
| WWTP South | 1.4 | 1.2 | 7.5 ± 3.98 | 5.8 ± 1.37 | NA | NA | NA | NA | NA | NA | 7.6 ± 3.98 | 4.4 ± 3.14 | 0.36 ± 0.09 | 0.22 ± 0.05 |
| Downstream dam | 13 | 3.0 | 0.038 ± 0.04 | 0.19 ± 0.20 | 0.009 ± 0.01 | 0.030 ± 0.02 | 0.18 ± 0.13 | 0.23 ± 0.03 | 1.7 ± 1.04 | 2.2 ± 1.85 | 0.002 ± 0.00 | <DL | 0.005 ± 0.01 | 0.003 ± 0.00 |
| Lake | - | - | 0.122 ± 0.14 | 0.29 ± 0.07 | 0.025 ± 0.04 | 0.033 ± 0.03 | 0.20 ± 0.19 | 0.051 ± 0.06 | 2.1 ± 1.43 | 2.9 ± 0.92 | <DL | <DL | 0.021 ± 0.03 | 0.036 ± 0.05 |
| Input/Output | DIN (Mg) | TON (Mg) | PO43−-P (Mg) | TOP (Mg) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wet | Dry | Annual | Wet | Dry | Annual | Wet | Dry | Annual | Wet | Dry | Annual | |
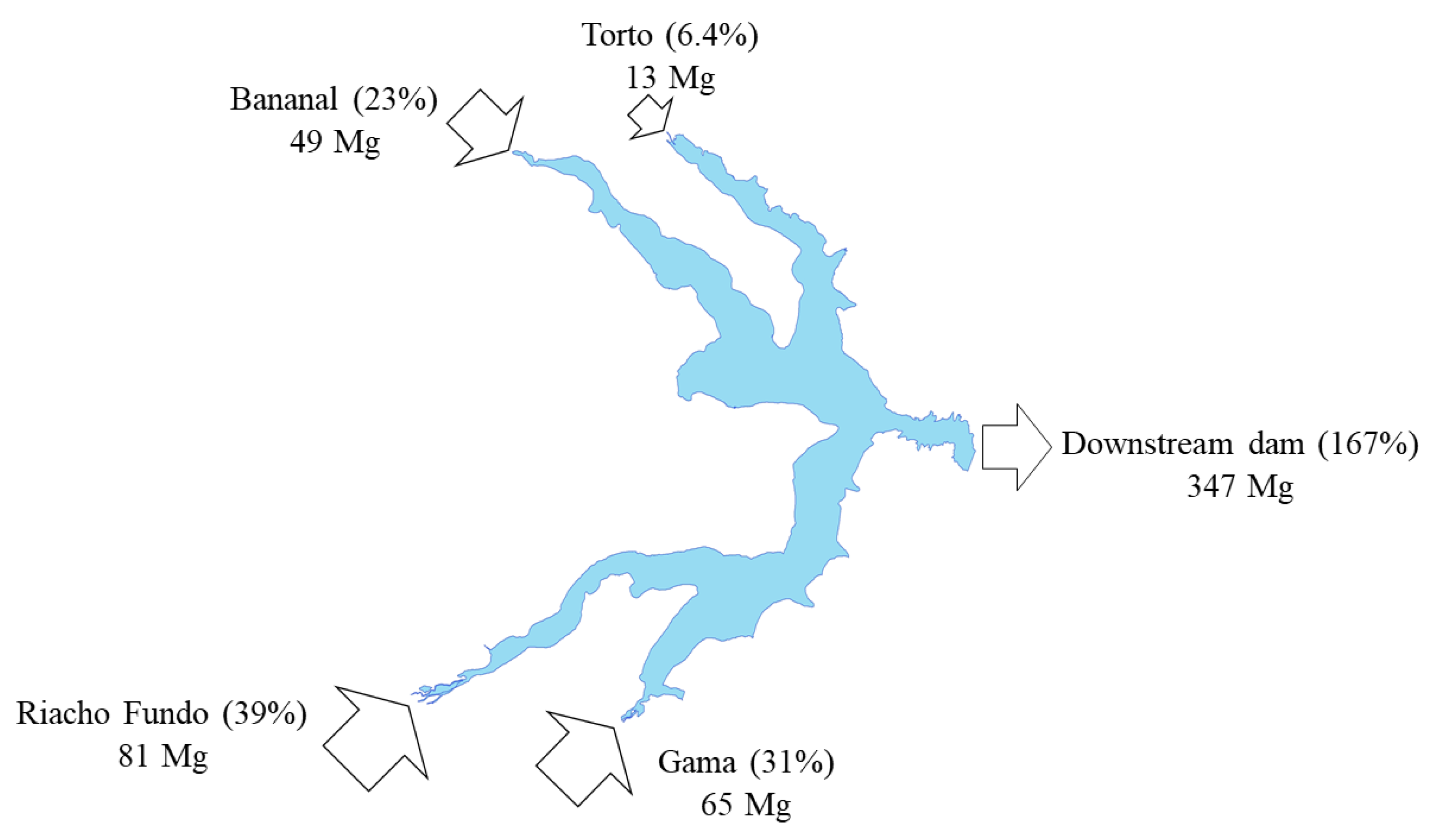
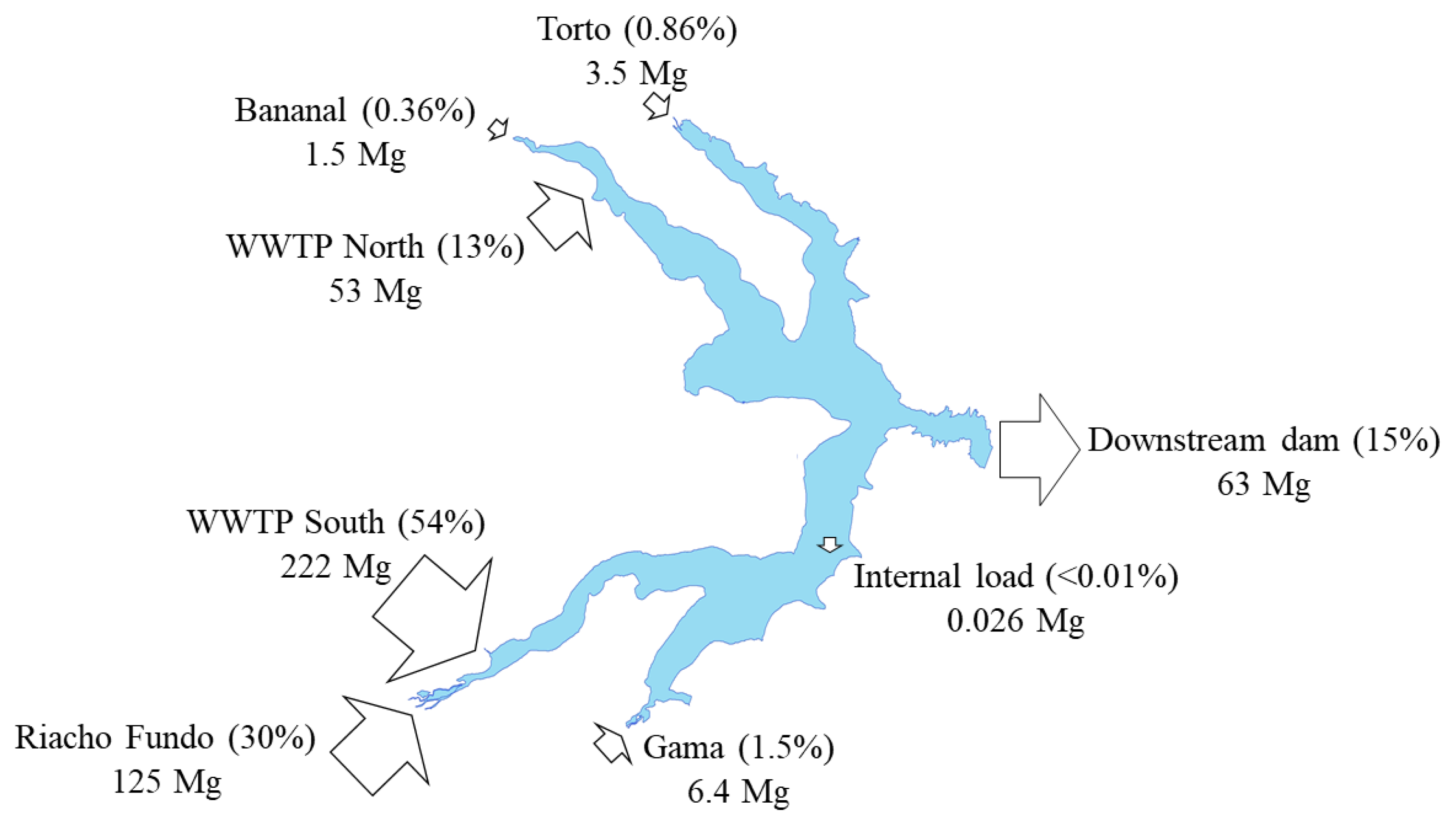
| Torto Stream | 3.3 | 0.25 | 3.53 (0.86%) | 12 | 1.0 | 13 (6.4%) | 0.019 | ND | 0.02 (0.01%) | 0.051 | 0.013 | 0.06 (0.37%) |
| Bananal Stream | 0.9 | 0.59 | 1.50 (0.36%) | 36 | 12 | 49 (23%) | 0.034 | 0.017 | 0.05 (0.02%) | 0.158 | ND | 0.17 (0.97%) |
| Riacho Fundo Stream | 86 | 40 | 125 (30.4%) | 70 | 11 | 81 (39%) | 0.13 | 0.13 | 0.27 (0.10%) | 1.9 | 0.20 | 2.10 (12.3%) |
| Gama Stream | 5.7 | 0.69 | 6.37 (1.55%) | 55 | 9.2 | 65 (31%) | 0.052 | 0.015 | 0.07 (0.03%) | 0.24 | 0.023 | 0.26 (1.54%) |
| WWTP North | 17 | 35 | 52.9 (12.8%) | NA | NA | NA | 18 | 21 | 40 (15.3%) | 2.5 | 1.5 | 3.93 (23%) |
| WWTP South | 161 | 62 | 222 (53.9%) | NA | NA | NA | 163 | 56 | 218 (84.5%) | 7.7 | 2.8 | 10.5 (62%) |
| Internal load | NA | 0.026 | 0.026 (0.01%) | NA | NA | NA | 0.12 | NA | 0.12 (0.04) | NA | NA | NA |
| Total input | 274 | 138 | 412 (100%) | 174 | 34 | 207 (100%) | 181 | 77 | 258 (100%) | 13 | 4.6 | 17.1 (100%) |
| Output | 48 | 14 | 62.5 (15.2%) | 296 | 51 | 347 (167%) | 0.44 | 0.042 | 0.48 (0.19%) | 1.1 | 0.11 | 1.23 (7.2%) |
| Parameter | Wet Season | Dry Season | ||||
|---|---|---|---|---|---|---|
| Input (Mg) | Output (Mg) | Retention (Mg) | Input (Mg) | Output (Mg) | Retention (Mg) | |
| NH4+-N | 184 | 7.9 | 176 (96%) | 103 | 6.0 | 97 (94%) |
| NO2−-N | 3.3 | 1.8 | 1.5 (45%) | 1.2 | 0.97 | 0.22 (19%) |
| NO3−-N | 87 | 39 | 48 (56%) | 34 | 7.2 | 27 (79%) |
| TON | 174 | 296 | −122 (70%) | 34 | 51 | −17 (52%) |
| PO43−-P | 181 | 0.44 | 181 (100%) | 77 | 0.042 | 77 (100%) |
| TOP | 13 | 1.1 | 11 (91%) | 4.6 | 0.11 | 4.5 (98%) |
| Site | Location | Climatic Region | Mean Depth (m) | Hydraulic Retention Time (Year) | TN (Mg Year−1) | TP (Mg Year−1) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Input (I) | Output (O) | Retention (=I−O) | Input (I) | Output (O) | Retention (=I−O) | |||||
| This study | Brazil | Aw | 14 | 2.71 | 620 | 410 | 210 (34%) | 275 | 1.6 | 273 (99%) |
| Água Preta [44] | Brazil | Af | NA | 0.64 | 38.8 | 27 | 11.8 (30%) | 7.9 | 5.1 | 2.8 (35%) |
| Baldeggersee [8] | Switzerland | Dfb | NA | 4.0 | NA | NA | NA | 11.2 | 6.8 | 4.4 (39%) |
| Greifenseen [8] | Switzerland | Dfb | NA | 1.2 | NA | NA | NA | 42.7 | 38.8 | 3.9 (9.1%) |
| Hallwilersee [8] | Switzerland | Dfb | NA | 3.9 | NA | NA | NA | 14.6 | 10.8 | 3.8 (26%) |
| Pfaffikersee [8] | Switzerland | Dfb | NA | 2.1 | NA | NA | NA | 6.8 | 4.9 | 1.9 (28%) |
| Sempachersee [8] | Switzerland | Dfb | NA | 15.3 | NA | NA | NA | 8.9 | 4.2 | 4.7 (53%) |
| Belau [10] | Germany | Cfb | NA | 0.73 | 35.42 | 32.67 | 2.75 (7.8%) | 1.25 | 1.11 | 0.14 (11.2%) |
| Biritiba [45] | Brazil | Cwb | NA | 0.95 | 22.5 | 0.4 | 22.1 (98%) | 1.5 | 0.5 | 1.0 (67%) |
| Paraitinga [45] | Brazil | Cwb | NA | 0.48 | 25 | 15 | 10 (40%) | 1.4 | 0.9 | 0.5 (36%) |
| Broa [46] | Brazil | Cwa | 3.0 | 0.08 | NA | NA | NA | 3260 | 2765 | 495 (15%) |
| Cruzeta [47] | Brazil | Bsh | 3.8 | 0.33 | NA | NA | NA | 10.18 | 4.05 | 6.13 (60%) |
| Gargalheiras [47] | Brazil | Bsh | 5.5 | 1.27 | NA | NA | NA | 6.13 | 0.26 | 5.87 (96%) |
| Dal [12] | India | Dsb | 0.9 | NA | NA | NA | NA | 88.1 | 59 | 29.1 (33%) |
| Donghu [6] | China | Cfa | 2.2 | 0.43 | 1480 | 585 | 895 (60%) | 95 | 10 | 85 (89%) |
| Kasumigaura [6] | Japan | Cfa | 4.0 | 0.57 | 3890 | 1193 | 2697 (69.3%) | 220 | 78 | 142 (64%) |
| Okeechobee [6] | USA | Cfa | 2.7 | 3.8 | 5554 | 2986 | 2568 (46%) | 426 | 148 | 278 (65%) |
| Dourado [48] | Brazil | Bsh | 3.0 | 0.83 | NA | NA | NA | 2.0 | 0.95 | 1.05 (52%) |
| Gavião [49] | Brazil | Aw | NA | NA | 289.5 | 168.1 | 121.4 (42%) | 63.21 | 35.15 | 28.06 (44%) |
| Forsyth [4] | New Zealand | Cfb | NA | 0.15 | NA | NA | NA | 9.14 | 2.78 | 6.36 (70%) |
| Garças [50] | Brazil | Cwa | 2.1 | 0.19 | 37.16 | 5.52 | 31.63 (85%) | 6.52 | 2.56 | 3.96 (61%) |
| Koumoundorou [31] | Greece | Csa | 1.0 | NA | 4012 | 0.154 | 3858 (96%) | NA | NA | NA |
| Locholoosa [51] | USA | Cfa | 2.1 | 0.74 | NA | NA | NA | 3.22 | 1.47 | 1.75 (54%) |
| Lough Neagh [52] | Ireland | Cfb | 8.9 | 1.27 | NA | NA | NA | 509 | 323 | 186 (36%) |
| Pampulha [37] | Brazil | Cwa | 4.1 | NA | 67.39 | 23.79 | 43.60 (65%) | 9.14 | 1.74 | 7.4 (81%) |
| Valle de Bravo [9] | Mexico | Cwb | 21.1 | NA | 591.8 | 319.7 | 272.1 (46%) | 120.8 | 26.2 | 94.6 (78%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbosa, J.d.S.B.; Bellotto, V.R.; da Silva, D.B.; Lima, T.B. Nitrogen and Phosphorus Budget for a Deep Tropical Reservoir of the Brazilian Savannah. Water 2019, 11, 1205. https://doi.org/10.3390/w11061205
Barbosa JdSB, Bellotto VR, da Silva DB, Lima TB. Nitrogen and Phosphorus Budget for a Deep Tropical Reservoir of the Brazilian Savannah. Water. 2019; 11(6):1205. https://doi.org/10.3390/w11061205
Chicago/Turabian StyleBarbosa, Jackeline do S. B., Valéria R. Bellotto, Damiana B. da Silva, and Thiago B. Lima. 2019. "Nitrogen and Phosphorus Budget for a Deep Tropical Reservoir of the Brazilian Savannah" Water 11, no. 6: 1205. https://doi.org/10.3390/w11061205
APA StyleBarbosa, J. d. S. B., Bellotto, V. R., da Silva, D. B., & Lima, T. B. (2019). Nitrogen and Phosphorus Budget for a Deep Tropical Reservoir of the Brazilian Savannah. Water, 11(6), 1205. https://doi.org/10.3390/w11061205





