Comparative Analysis of Granular and Membrane Filters for Rainwater Treatment
Abstract
1. Introduction
2. Materials and Methods
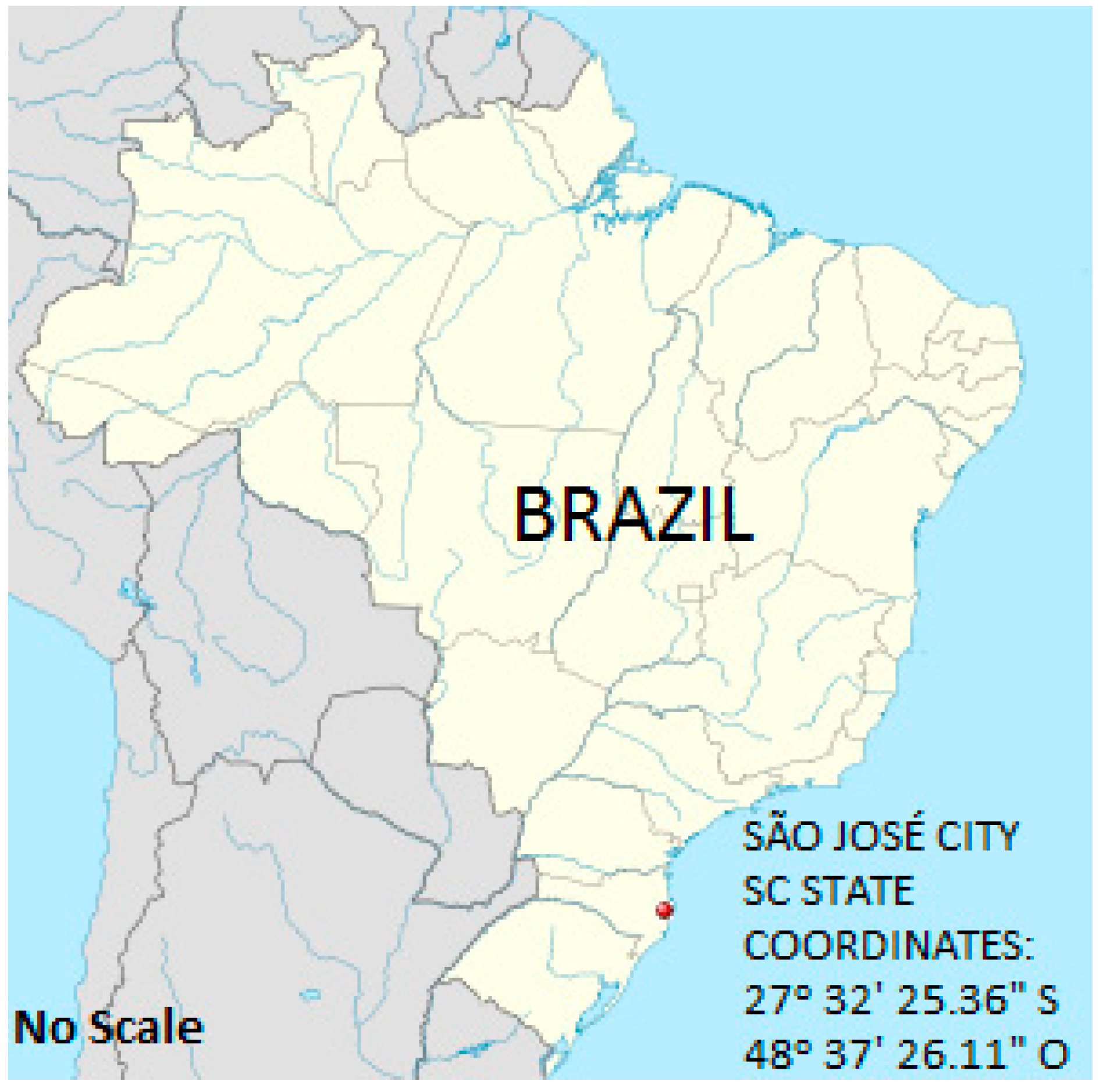
2.1. Place of Study
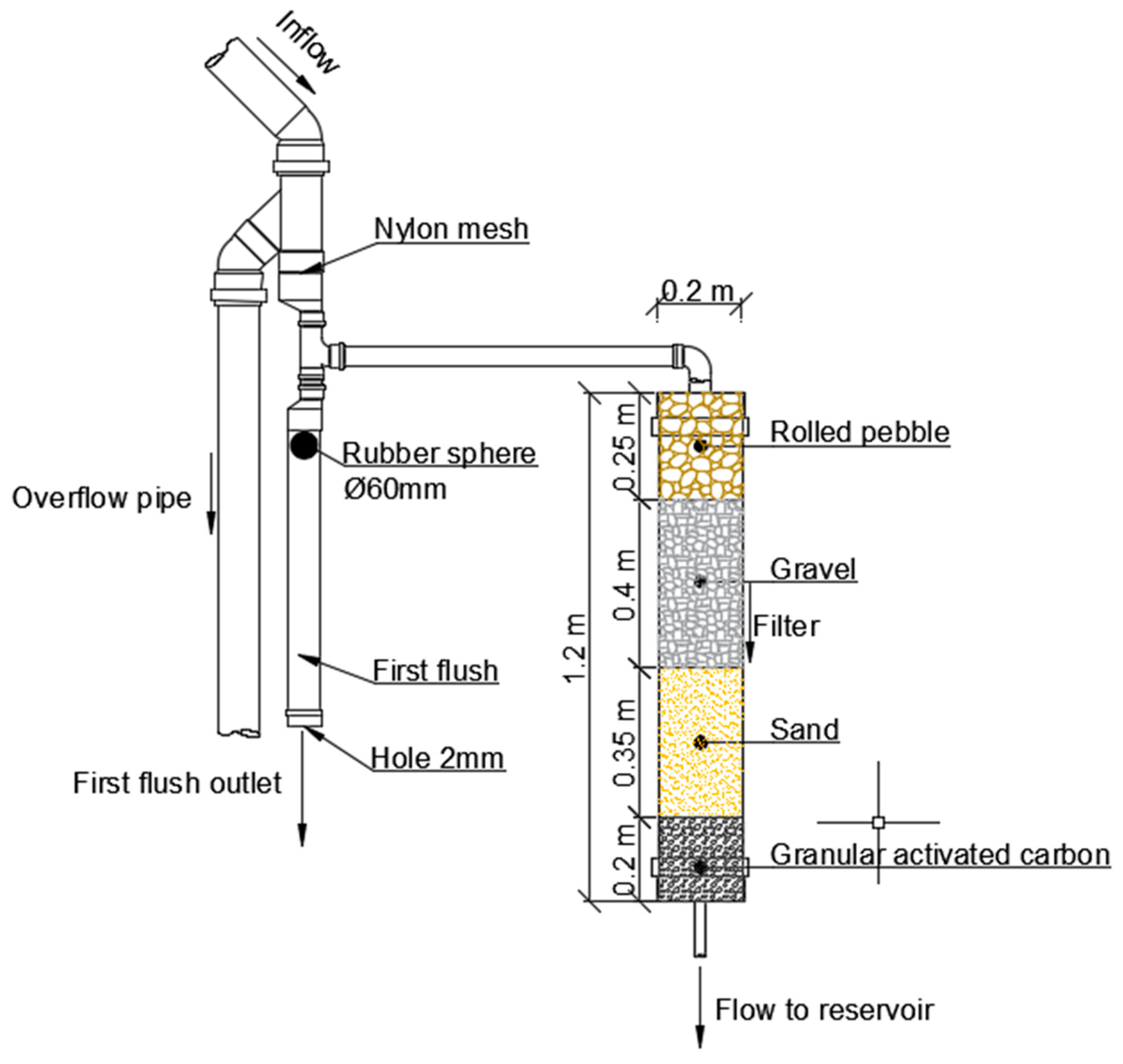
2.2. Description of Sand Filter
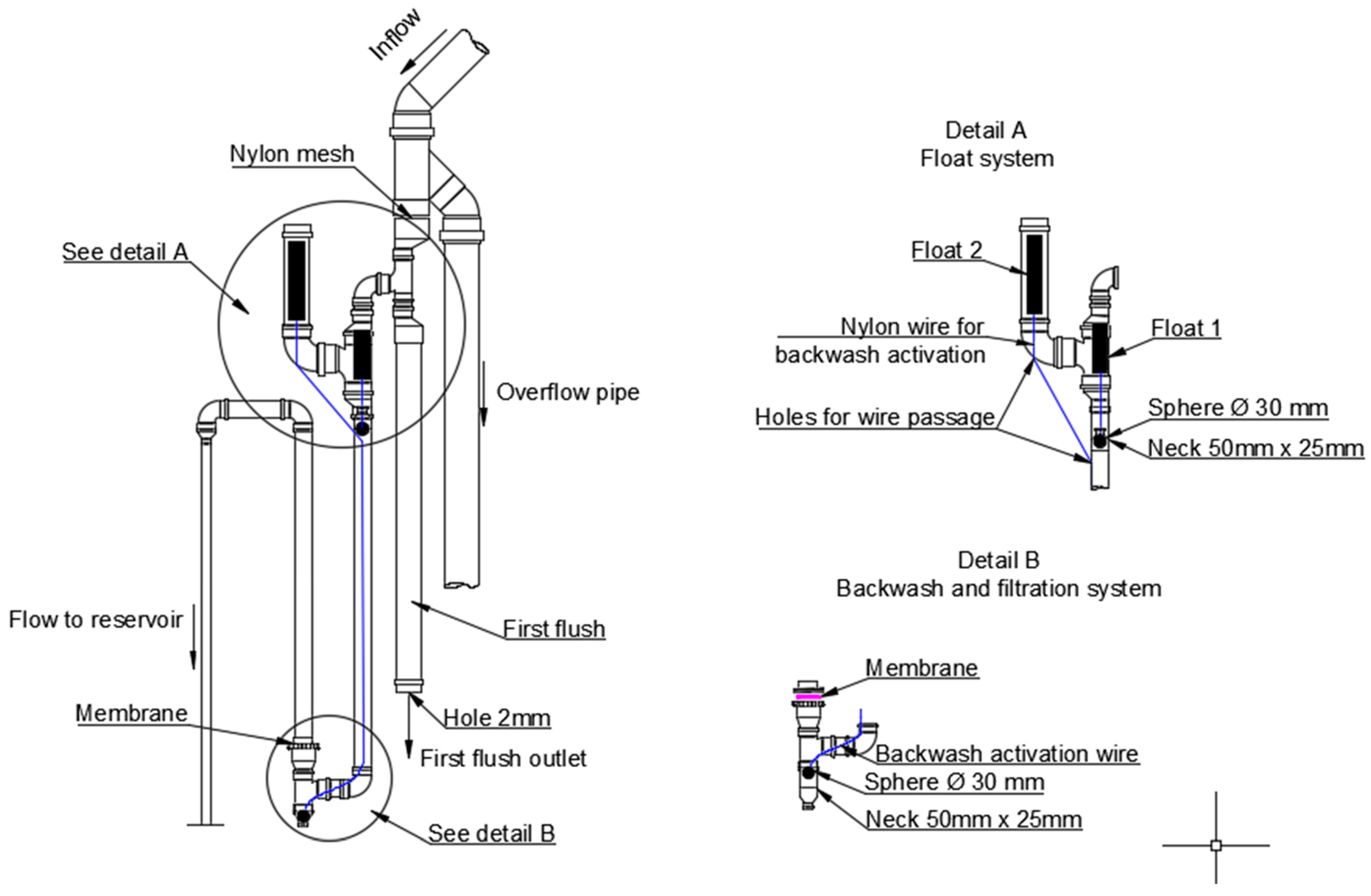
2.3. Description of Membrane Filter
2.4. Qualitative Parameters
3. Results and Discussion
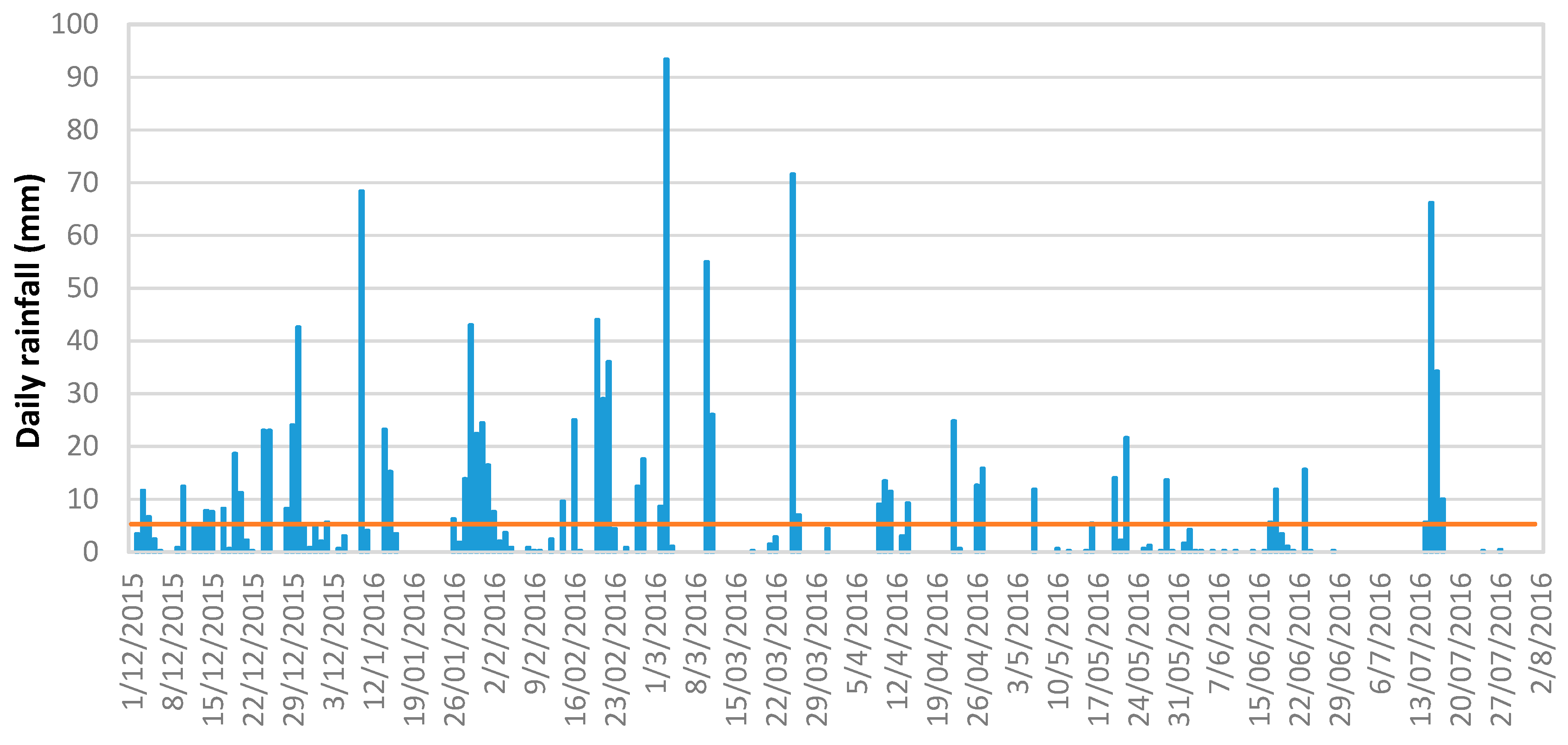
3.1. Rainfall Analysis
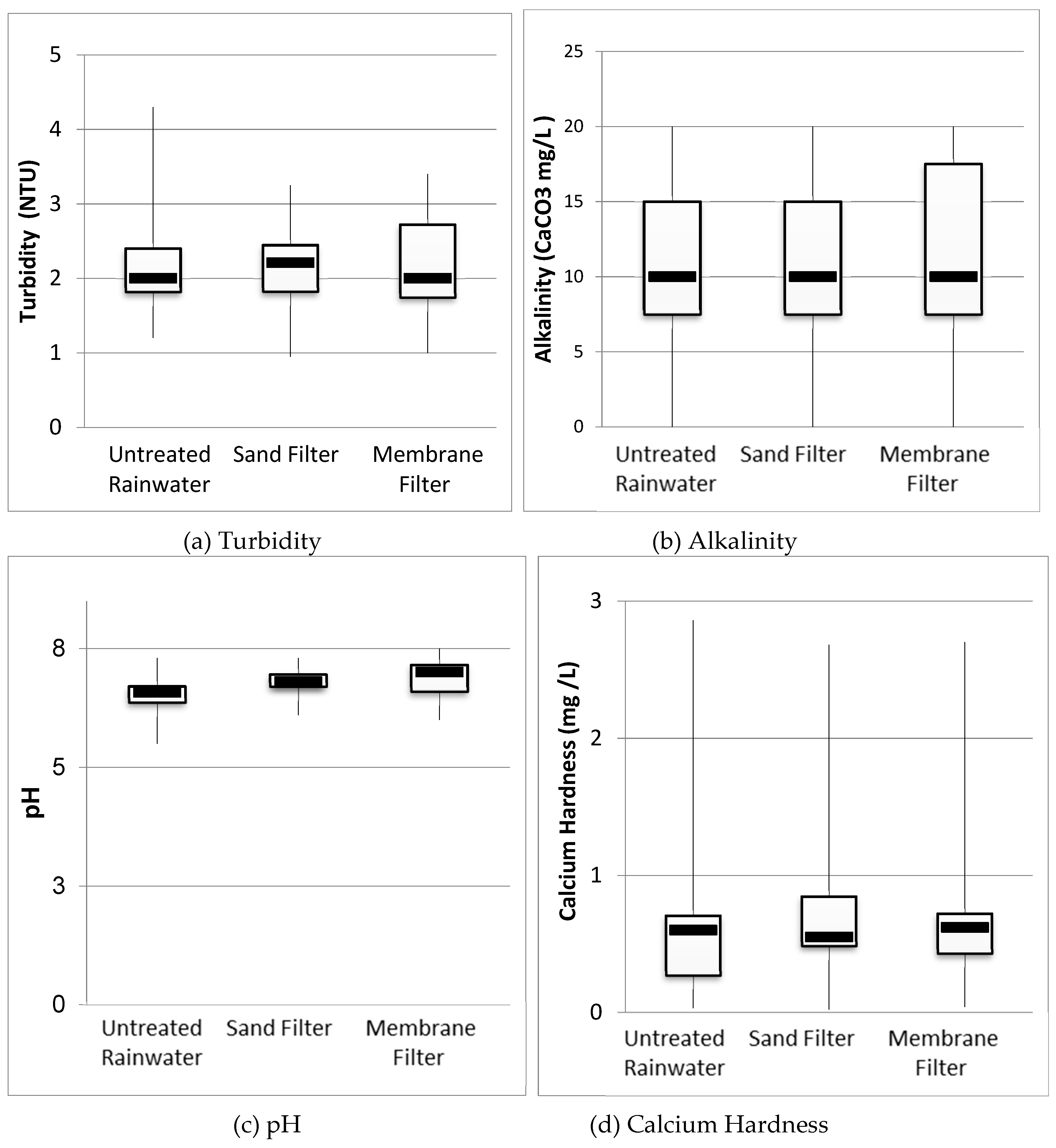
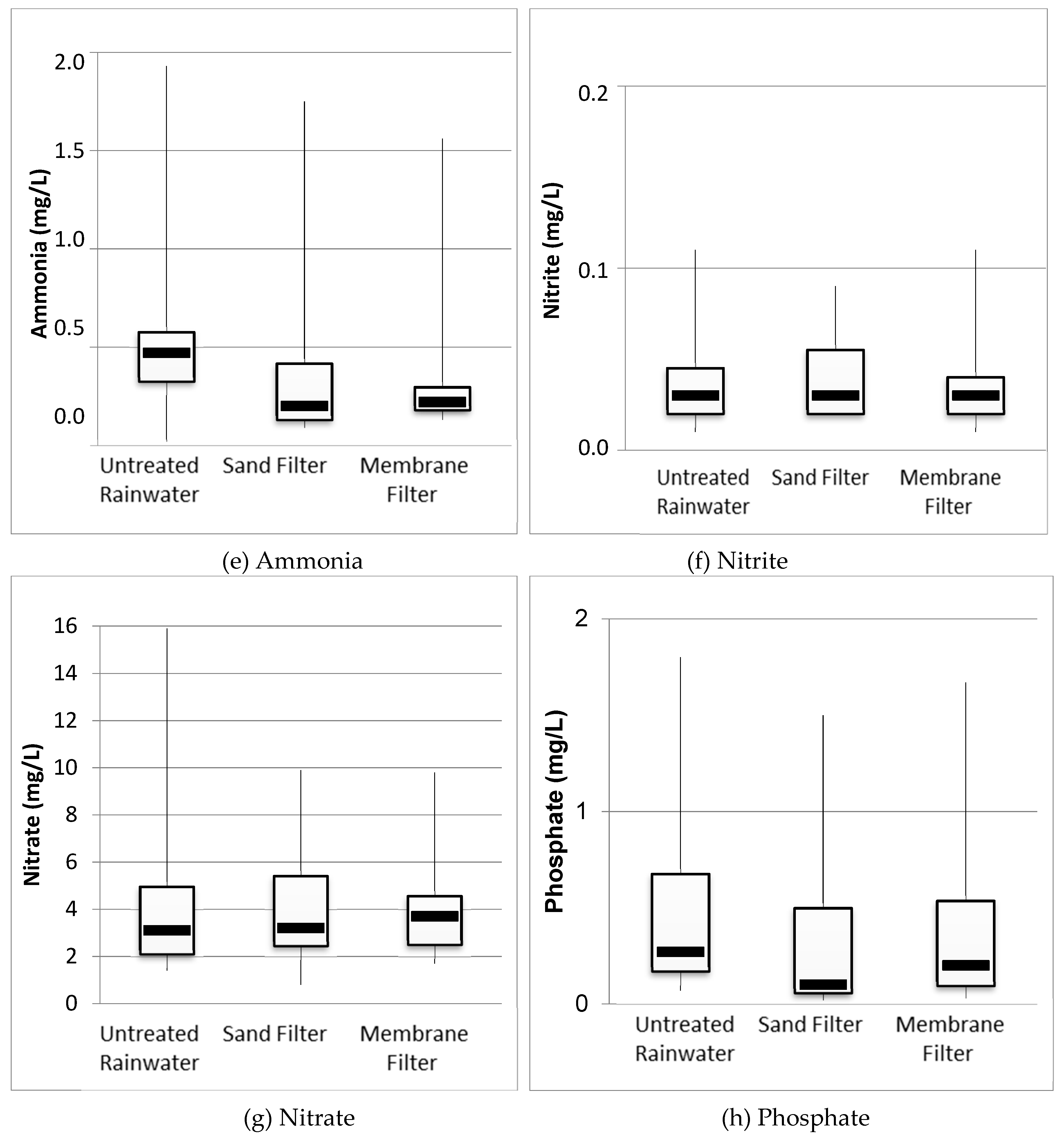
3.2. Physicochemical Analysis of Rainwater
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Naddeo, V.; Scannapieco, D.; Belgiorno, V. Enhaced drinking water supply through harvested rainwater treatment. J. Hydrol. 2013, 498, 287–293. [Google Scholar] [CrossRef]
- Lee, Y.J.; Yang, J.S.; Han, M.; Choi, J. Comparison of the microbiological and chemical characterization of harvested rainwater and reservoir water as alternative water resources. Sci. Total Environ. 2010, 408, 896–905. [Google Scholar] [CrossRef] [PubMed]
- UNESCO. United Nations World Water Development Report: Leaving on One behind—Facts and Figures; UNESCO: Paris, France, 2019. [Google Scholar]
- Wilbers, G.J.; Sebesvari, Z.; Rechenburg, A.; Renaud, F.G. Effects of local spatial conditions on the quality of harvested rainwater in the Mekong Delta—Vietnam. Environ. Pollut. 2013, 182, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Weisbeck, E.; Sandri, E.K.; Soares, A.L.M.; Medeiros, S.H.W. Desinfecção da água de chuva por radiação ultravioleta. Engenharia Sanitária e Ambiental 2011, 16, 337–342. (In Portuguese) [Google Scholar] [CrossRef]
- Barret, B.C.; Kinney, A.K.; Kirisits, J.M. The effect of roofing material on the quality of harvested rainwater. Water Res. 2011, 45, 2049–2059. [Google Scholar]
- Gikas, D.G.; Tsihrintizs, A.V. Assessment of water quality of first-flush roof runoff and harvested rainwater. J. Hydrol. 2012, 466–467, 115–126. [Google Scholar] [CrossRef]
- Coelho, E.R.C.; di Bernardo, L. Remoção de atrazina e metabólitos pela filtração lenta com leito de areia e carvão ativado granular. Engenharia Sanitária e Ambiental 2012, 17, 269–276. (In Portuguese) [Google Scholar] [CrossRef]
- Brinck, N.C.P. Avaliação do tipo de material filtrante no comportamento hidráulico de filtros rápidos de camada profunda no tratamento de águas de abastecimento. Ph.D. Thesis, Escola Politécnica da Universidade de São Paulo, São Paulo, Brazil, 2009. (In Portuguese). [Google Scholar]
- Testezlaf, R. Filtros de areia aplicados à irrigação localizada: Teoria e Prática. Engenharia Agrícola 2008, 28, 604–613. (In Portuguese) [Google Scholar] [CrossRef]
- Aslan, A. Combined removal of pesticides and rates in drinking waters using biodenitrification and sand filter system. Process. Biochem. 2005, 40, 417–424. [Google Scholar] [CrossRef]
- Zhang, G.; Yang, Y.; Liu, X.; Zhao, W. Research and application of harvested rainwater in the villages and towns of China Loess Plateau region. Energy Procedia 2011, 5, 307–313. [Google Scholar]
- Shaheed, R.; Mohtar, W.H.M.W.; El Shafie, A. Ensuring water security by utilizing roof-harvested rainwater and lake water treated with a low-cost integrated adsorption-filtration system. Water Sci. Eng. 2017, 10, 115–124. [Google Scholar] [CrossRef]
- Yan, X.; Wards, S.; Butler, D.; Daly, B. Performance assessment and life cycle analysis of potable water production from harvested rainwater by a decentralized system. J. Clean. Product. 2018, 172, 2167–2173. [Google Scholar] [CrossRef]
- Tang, X.; Pronk, W.; Ding, A.; Cheng, X.; Wang, J.; Xie, B.; Li, G.; Liang, H. Coupling GAC to ultra-low-pressure filtration to modify the biofounling layer do bio-community: Flux enhancement and water quality improvement. Chem. Eng. J. 2018, 333, 289–299. [Google Scholar]
- ABNT (Associação Brasileira de Normas Técnicas). Agregados—Determinação da composição granulométrica—NBR NM 248:2001; ABNT: Rio de Janeiro, Brazil, 2001. (In Portuguese) [Google Scholar]
- ASTM- American Society of Testing and Materials. D 3838-1980: Standard Test Method for pH of Activated Carbon; American Society of Testing and Materials: West Conshohocken, PA, USA, 1999. [Google Scholar]
- ASTM- American Society of Testing and Materials. D 2867-2004: Standard Test Method for Moisture in Activated Carbon; American Society of Testing and Materials: West Conshohocken, PA, USA, 2004. [Google Scholar]
- ASTM- American Society of Testing and Materials. D 5832-1998: Standard Test Method for Volatile Matter Content of Activated Carbon; American Society of Testing and Materials: West Conshohocken, PA, USA, 2003. [Google Scholar]
- ASTM- American Society of Testing and Materials. D 2866-1994: Standard Test Method for Total Ash Content of Activated Carbon; American Society of Testing and Materials: West Conshohocken, PA, USA, 1998. [Google Scholar]
- ABNT (Associação Brasileira de Normas Técnicas). Cimento Portland e Outros Materiais em pó—Determinação da Massa Específica—NBR NM 23:2001; ABNT: Rio de Janeiro, Brazil, 2001. (In Portuguese) [Google Scholar]
- ABNT (Associação Brasileira de Normas Técnicas). Agregado Miúdo—Determinação da Massa Específica e Aparente—NBR 52:2009; ABNT: Rio de Janeiro, Brazil, 2009. (In Portuguese) [Google Scholar]
- ABNT (Associação Brasileira de Normas Técnicas). Agregado Graúdo—Determinação da Massa Específica e Aparente—NBR 53:2009; ABNT: Rio de Janeiro, Brazil, 2009. (In Portuguese) [Google Scholar]
- ABNT (Associação Brasileira de Normas Técnicas). Carvão Ativado Pulverizado—Determinação da Massa Específica e Aparente—NBR 12076:1991; ABNT: Rio de Janeiro, Brazil, 1991. (In Portuguese) [Google Scholar]
- ABNT (Associação Brasileira de Normas Técnicas). Carvão Ativado Pulverizado—Determinação do Número de Iodo—Método de Ensaio—NBR 12073:1991; ABNT: Rio de Janeiro, Brazil, 1991. (In Portuguese) [Google Scholar]
- ABNT (Associação Brasileira de Normas Técnicas). Agregados—Determinação da Massa Unitária e do Volume de Vazios—NBR 45:2006; ABNT: Rio de Janeiro, Brazil, 2006. (In Portuguese) [Google Scholar]
- Vieira, A.S.; Weeber, M.; Ghisi, E. Self-cleaning filtration: A novel concept for rainwater harvesting systems. Resour. Conserv. Recycl. 2013, 78, 67–73. [Google Scholar] [CrossRef]
- APHA (American Public Health Association). Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1999. [Google Scholar]
- Conselho Nacional do Meio Ambiente (CONAMA). Resolução 357 de 17 de março de 2005. In Dispõe Sobre a Classificação dos Corpos de água e Diretrizes Ambientais Para o Seu Enquadramento; CONAMA: Brasília, Brazil, 2015. (In Portuguese) [Google Scholar]
- US EPA (United States Environmental Protection Agency). Guidelines for Water Reuse; EPA/600/R—12/618; US EPA: Washington, DC, USA, 2012.
- Ministério da Saúde. Portaria n 2914: Procedimentos de Controle e de Vigilância da Qualidade da água Para Consumo Humano e seu Padrão de Potabilidade; Ministério da Saúde: Brasília, Brazil, 2011. (In Portuguese)
- ABNT (Associação Brasileira de Normas Técnicas). Água de Chuva—Aproveitamento de coberturas em áreas Urbanas Para Fins não Potáveis—Requisitos. NBR 15527/2007; ABNT: Rio de Janeiro, Brazil, 2007. (In Portuguese) [Google Scholar]
- Instituto Nacional de Meteorologia (INMET). Available online: http://www.inmet.gov.br/portal/index.php?estacoes/estacoesautomaticas. (accessed on 22 September 2016). (In Portuguese)
- Sezerino, P.H. Potencialidade dos Filtros Plantados com Macrófitas (Constructed Wetlands) No Pós-Tratamento de Lagoas de Estabilização Sob Condições de Clima Subtropical. Ph.D. Thesis, Federal University of Santa Catarina, Florianópolis, Brazil, 2006. (In Portuguese). [Google Scholar]
- Honório, B.A.D.; Horbe, A.M.C.; Seyler, P. Chemical composition of rainwater in western Amazonia—Brazil. Atmos. Res. 2010, 98, 416–425. [Google Scholar] [CrossRef]
- Silva, F.A. Aproveitamento da água da Chuva após Tratamento por Filtração Ascendente e Radiação Ultravioleta. Master’s Thesis, UTFPR, Curitiba, Brazil, 2015. (In Portuguese). [Google Scholar]

| Parameter | Sand | Gravel | GAC | Methodology | Unit |
|---|---|---|---|---|---|
| Granulometry | X | X | X | ABNT NBR NM 248 (2001) 1 | - |
| pH | X | X | X | ASTM D 3838-80 (1999) 2 | - |
| Moisture content | X | X | X | ASTM D 2867-04 (2004) 3 | % |
| Volatile matter content | X | X | X | ASTM D 5832-98 (2003) 4 | % |
| Ash content | X | X | X | ASTM D 2866-94 (1999) 5 | % |
| Specific mass | X | X | X | ABNT NBR NM 23 (2001) 6 | g/cm 3 |
| Bulk density | X | X | X | ABNT NBR 52 (2009) 7 Sand ABNT NBR 53 (2009) 8 Gravel ABNT NBR 12076 (1991) 9 CAG | g/cm 3 |
| Iodine number | X | ABNT NBR 12073 (1991) 10 | mg/g | ||
| Void index | X | X | X | ABNT NBR 45 (2006) 11 | % |
| Parameter | Sand | Gravel | Activated Carbon |
|---|---|---|---|
| pH | 6.8 ± 0.1 | 8.8 ± 0.1 | 6.7 ± 0.1 |
| Volatile matter content (%) | 1.0 ± 0.1 | 2.9 ± 0.1 | 50.8 ± 0.1 |
| Moisture content (%) | 2.38 ± 0.01 | 0.05 ± 0.01 | 48.73 ± 0.01 |
| Ash content (%) | 1.11 ± 0.01 | 6.96 ± 0.01 | 6.25 ± 0.01 |
| Specific mass (g/cm3) | 2.61 ± 0.01 | 2.69 ± 0.01 | 1.27 ± 0.01 |
| Bulk density (g/cm3) | 1.47 ± 0.02 | 1.38 ± 0.02 | 0.63 ± 0.02 |
| Void Index (%) | 43.8 ± 0.1 | 48.8 ± 0.1 | 32.9 ± 0.1 |
| Iodine number (mg/g) | - | - | 665.86 ± 0.01 |
| Material | Minimum Diameter (mm) | Maximum Diameter (mm) | D10 (mm) | D60 (mm) | |
|---|---|---|---|---|---|
| Sand | 0.15 | 4.76 | 0.27 | 1.32 | 4.90 |
| Gravel | 4.76 | 19.10 | 7.49 | 14.60 | 1.90 |
| Parameter | Methodology | Precision |
|---|---|---|
| Ammonia | Nesseler, adapted from ASTM Manual of Water and Environmental Technology, D-1426-92 | ±0.04 mg/L ± 4% for reading at 25 °C |
| Nitrite | Adapted from US EPA 354.1 (diazotization method) | ±0.06 mg/L ± 4% for reading at 25 °C |
| Nitrate | Adapted from the reduction of cadmium | ±0.5 mg/L ± 10% for reading at 25 °C |
| Alkalinity | Colorimetric method | ±5 mg/L ± 10% for reading at 25 °C |
| Calcium Hardness | Adapted from calmagite method | ±0.11 mg/L ±5% for reading at 25 °C |
| Parameters | Nº | Untreated Rainwater | Sand Filter | Membrane Filter | Standards and Reference Values | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Max. | Min. | Average (SD) | Max. | Min. | Average (SD) | Max. | Min. | Average (SD) | ||||
| pH | 15 | 7.3 | 5.5 | 6.5 (0.4) | 7.3 | 6.1 | 6.8 (0.3) | 7.5 | 6 | 6.9 (0.4) | US EPA1 | 6.0–9.0 |
| MS2 | 6.0–9.5 | |||||||||||
| CONAMA3 | 6.0–9.0 | |||||||||||
| NBR 155274 | 6.0–8.0 | |||||||||||
| Temperature (°C) | 15 | 29.0 | 15.8 | 22.9 (4.8) | 29.0 | 17.8 | 23.2 (4.6) | 29.6 | 18.5 | 24.2 (4.9) | No recommended value | |
| Alkalinity (mg/L) | 15 | 20.0 | 0.00 | 9.33 (5.44) | 20.00 | 0.00 | 11.67 (5.96) | 20.00 | 0.00 | 11.67 (6.24) | No recommended value | |
| Calcium Hardness (mg/L) | 15 | 2.86 | 0.03 | 0.63 (0.65) | 2.68 | 0.02 | 0.78 (0.66) | 2.7 | 0.04 | 0.7 (0.60) | No recommended value | |
| Turbidity (NTU) | 15 | 4.3 | 1.3 | 2.3 (0.9) | 3.2 | 0.9 | 2.0 (0.5) | 3.4 | 1 | 2.1 (0.8) | US EPA1 | ≤2 NTU |
| MS2 | ≤0.5 NTU | |||||||||||
| CONAMA3 | ≤40 NTU | |||||||||||
| NBR 155274 | <2.0 NTU* | |||||||||||
| NH3 (mg/L) | 15 | 1.93 | 0.02 | 0.53 (0.42) | 1.75 | 0.09 | 0.35 (0.40) | 1.56 | 0.13 | 0.36 (0.38) | MS2 | ≤1.5 mg/L |
| CONAMA3 | ≤2 mg/L | |||||||||||
| NO2− (mg/L) | 15 | 0.11 | 0.01 | 0.04 (0.03) | 0.09 | 0.02 | 0.04 (0.02) | 0.11 | 0.01 | 0.04 (0.03) | MS2 | ≤1 mg/L |
| CONAMA3 | ≤1 mg/L | |||||||||||
| NO3− (mg/L) | 15 | 15.90 | 1.40 | 4.37 (3.68) | 9.90 | 0.80 | 3.97 (2.37) | 9.80 | 1.70 | 3.82 (1.99) | MS2 | ≤10 mg/L |
| CONAMA3 | ≤10 mg/L | |||||||||||
| PO43− (mg/L) | 15 | 1.80 | 0.07 | 0.48 (0.48) | 1.50 | 0.02 | 0.30 (0.35) | 1.67 | 0.03 | 0.35 (0.40) | No recommended value | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teixeira, C.A.; Ghisi, E. Comparative Analysis of Granular and Membrane Filters for Rainwater Treatment. Water 2019, 11, 1004. https://doi.org/10.3390/w11051004
Teixeira CA, Ghisi E. Comparative Analysis of Granular and Membrane Filters for Rainwater Treatment. Water. 2019; 11(5):1004. https://doi.org/10.3390/w11051004
Chicago/Turabian StyleTeixeira, Celimar Azambuja, and Enedir Ghisi. 2019. "Comparative Analysis of Granular and Membrane Filters for Rainwater Treatment" Water 11, no. 5: 1004. https://doi.org/10.3390/w11051004
APA StyleTeixeira, C. A., & Ghisi, E. (2019). Comparative Analysis of Granular and Membrane Filters for Rainwater Treatment. Water, 11(5), 1004. https://doi.org/10.3390/w11051004






