A Review of Fouling Mechanisms, Control Strategies and Real-Time Fouling Monitoring Techniques in Forward Osmosis
Abstract
:1. Introduction
2. Mathematical Predictive Model for Fouling in Forward Osmosis
3. Classification of Membrane Fouling in Forward Osmosis
3.1. Biofouling
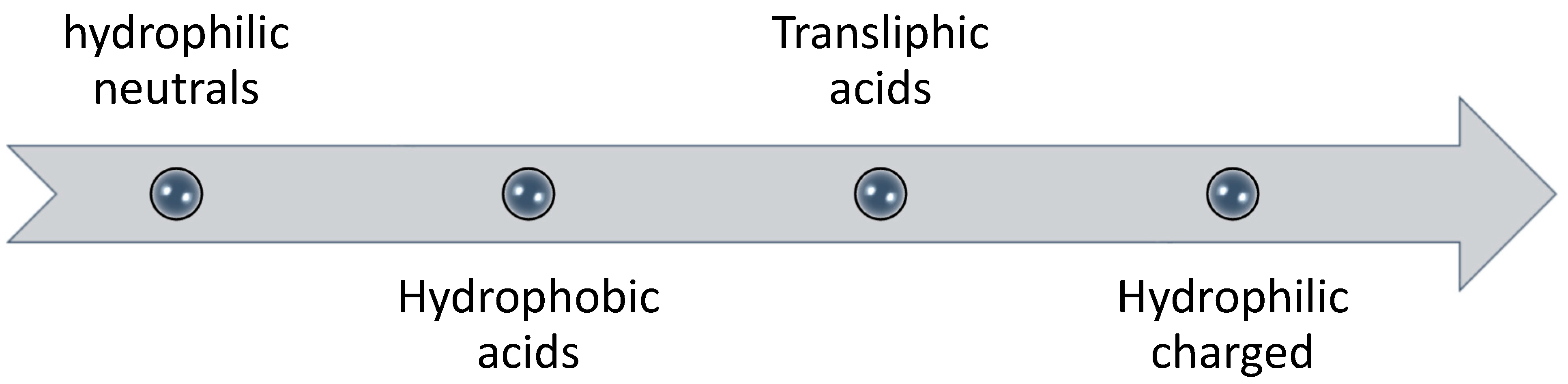
3.2. Organic Fouling
3.3. Inorganic Scaling
3.4. Colloidal Fouling
4. Factors Affecting FO Membrane Fouling and Performance
4.1. The Critical Flux Concept and Impact of Flux on Fouling in Forward Osmosis
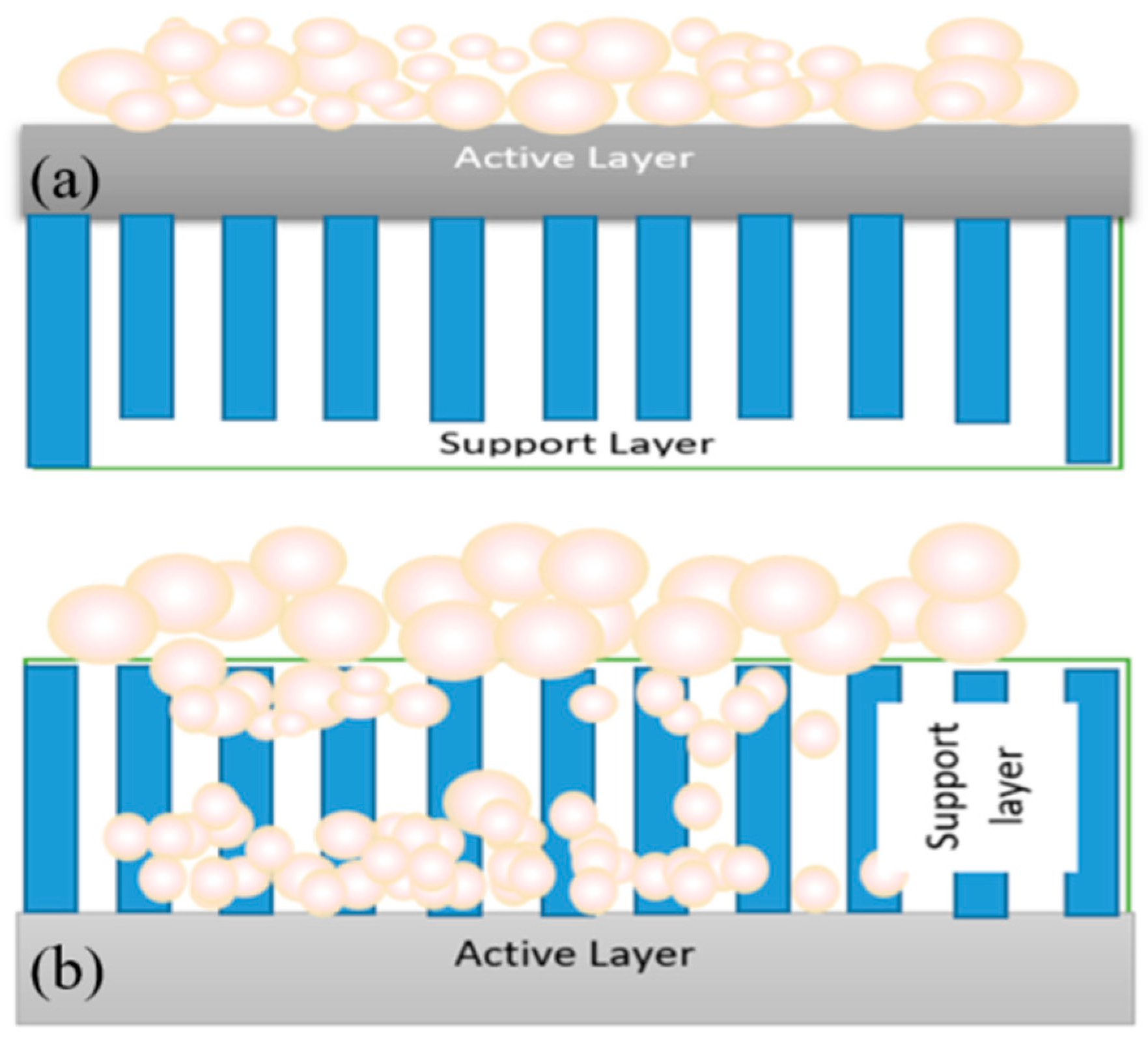
4.2. Effects of Hydrophilicity, Charge and Morphology on FO Membrane Fouling
4.3. Other Factors Limiting Membrane Performance
4.4. Coupled Effects of Concentration Polarization and Fouling on Flux Behavior in Forward Osmosis
5. Fouling and Fouling Mitigation in Osmotic Membrane Bioreactor (OMBR)
6. Fouling Mitigation in Direct FO
Effectiveness of Cleaning Strategies for Fouled FO Membranes
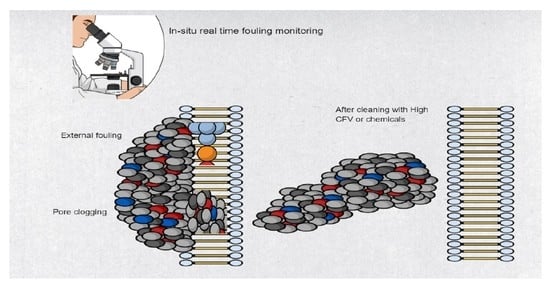
7. In-Situ and Real-Time Fouling Monitoring Techniques
7.1. Direct Observation over the Microscope (DOTM)
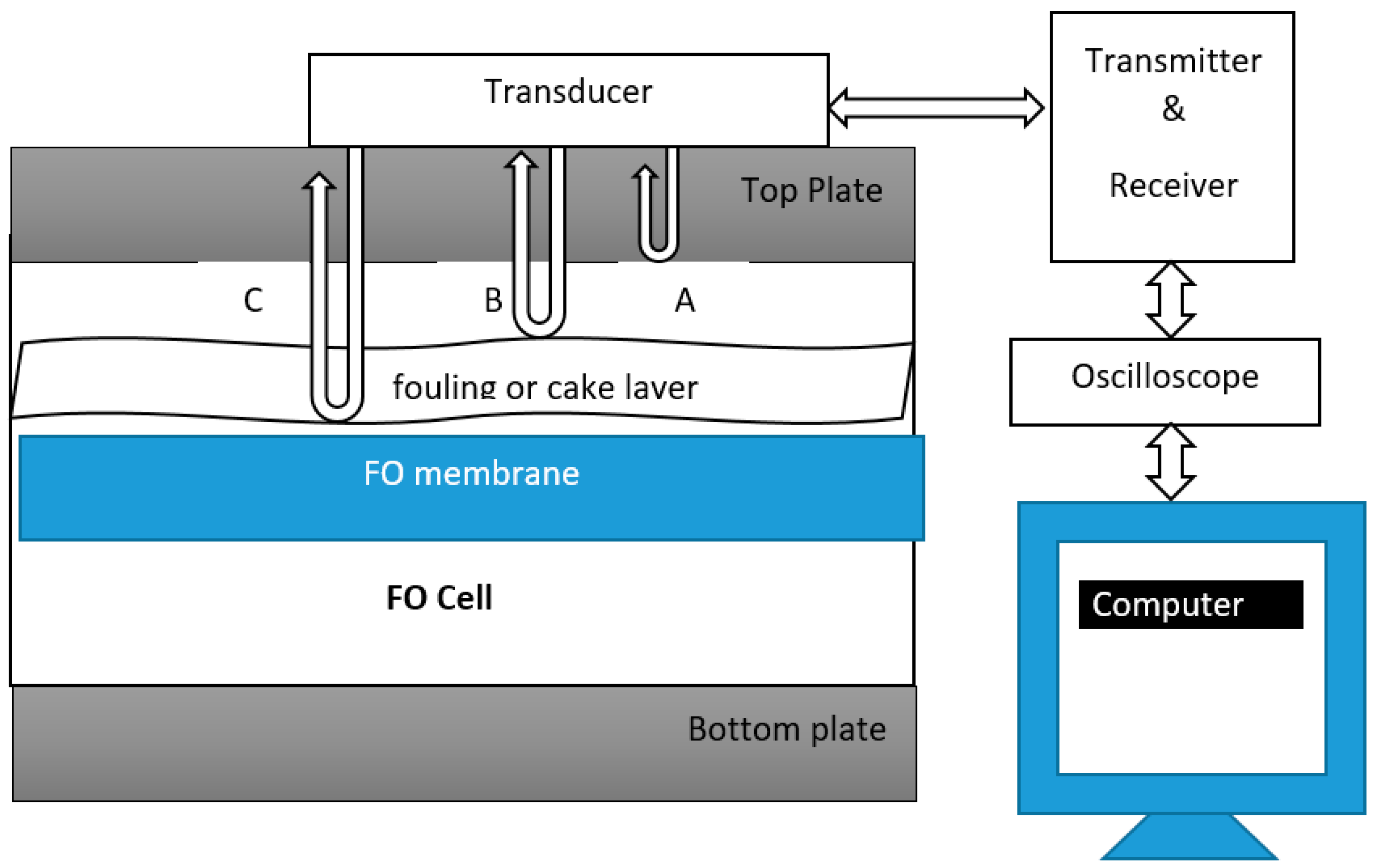
7.2. Ultrasonic Time Domain Reflectometry
7.3. Nuclear Magnetic Resonance or Magnetic Resonance Imaging
7.4. Silent AlarmTM Technology
7.5. Feed Fouling Monitor Coupled with UTDR
7.6. Optical Coherence Tomography (OCT)
7.7. Electrical Impedance Spectroscopy
7.8. Confocal Laser Scanning Microscopy Coupled with Multiple Flouresence Labelling
8. Conclusions
- Fouling in FO is reversible, mostly, and flux can be restored using high cross flow or improved hydrodynamics; however, it can be irreversible as well (e.g., biofouling) and chemical cleaning is then required.
- Most fouling studies use model foulants in fouling studies, such as alginate, BSA, humic acid and silica particles. This may cause confusion whether the same results will be translated for seawater or wastewater feeds.
- Novel antifouling membranes can enhance FO efficiency; however, commercial products may take a long time to develop. Most lab-fabricated antifouling membranes have very intricate synthesis processes and use expensive nanomaterials.
- In-situ real-time fouling monitoring is an urgent need for FO advancements and to mitigate fouling. In-situ cleaning can be done in correspondence with when fouling occurs, and this will improve efficiency. A very few publications on real-time monitoring of FO membrane fouling using state of the art technologies are available.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lutchmiah, K.; Verliefde, A.R.; Roest, K.; Rietveld, L.C.; Cornelissen, E.R. Forward osmosis for application in wastewater treatment: A review. Water Res. 2014, 58, 179–197. [Google Scholar] [CrossRef] [PubMed]
- Roorda, J.H. Filtration Characteristics in Dead-End Ultrafiltration of Wwtp-Effluent; TU Delft, Delft University of Technology: Delft, The Netherlands, 2004. [Google Scholar]
- She, Q.; Wang, R.; Fane, A.G.; Tang, C.Y. Membrane fouling in osmotically driven membrane processes: A review. J. Membr. Sci. 2016, 499, 201–233. [Google Scholar] [CrossRef]
- Chun, Y.; Mulcahy, D.; Zou, L.; Kim, I.S. A Short Review of Membrane Fouling in Forward Osmosis Processes. Membranes (Basel) 2017, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Field, R.W.; Wu, J.J. Novel approach for sizing forward osmosis membrane systems. J. Membr. Sci. 2017, 541, 321–328. [Google Scholar] [CrossRef]
- Zhao, P.; Gao, B.; Yue, Q.; Liu, P.; Shon, H.K. Fatty acid fouling of forward osmosis membrane: Effects of pH, calcium, membrane orientation, initial permeate flux and foulant composition. J. Environ. Sci. 2016, 46, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Korenak, J.; Basu, S.; Balakrishnan, M.; Hélix-Nielsen, C.; Petrinic, I. Forward Osmosis in Wastewater Treatment Processes. Acta Chim. Slov. 2017, 83–94. [Google Scholar] [CrossRef]
- Arkhangelsky, E.; Wicaksana, F.; Chou, S.; Al-Rabiah, A.A.; Al-Zahrani, S.M.; Wang, R. Effects of scaling and cleaning on the performance of forward osmosis hollow fiber membranes. J. Membr. Sci. 2012, 415–416, 101–108. [Google Scholar] [CrossRef]
- Jiang, S.; Li, Y.; Ladewig, B.P. A review of reverse osmosis membrane fouling and control strategies. Sci. Total Environ. 2017, 595, 567–583. [Google Scholar] [CrossRef]
- Hoek, E.M.V.; Elimelech, M. Cake-Enhanced Concentration Polarization: A New Fouling Mechanism for Salt-Rejecting Membranes. Environ. Sci. Technol. 2003, 37, 5581–5588. [Google Scholar] [CrossRef]
- Siddiqui, F.A.; She, Q.; Fane, A.G.; Field, R.W. Exploring the differences between forward osmosis and reverse osmosis fouling. J. Membr. Sci. 2018, 565, 241–253. [Google Scholar] [CrossRef]
- She, Q.; Hou, D.; Liu, J.; Tan, K.H.; Tang, C.Y. Effect of feed spacer induced membrane deformation on the performance of pressure retarded osmosis (PRO): Implications for PRO process operation. J. Membr. Sci. 2013, 445, 170–182. [Google Scholar] [CrossRef]
- Nagy, E.; Hegedüs, I.; Tow, E.W.; Lienhard, V.J.H. Effect of fouling on performance of pressure retarded osmosis (PRO) and forward osmosis (FO). J. Membr. Sci. 2018, 565, 450–462. [Google Scholar] [CrossRef]
- Tow, E.W.; Rencken, M.M.; Lienhard, J.H. In situ visualization of organic fouling and cleaning mechanisms in reverse osmosis and forward osmosis. Desalination 2016, 399, 138–147. [Google Scholar] [CrossRef]
- Gorzalski, A.S.; Coronell, O. Fouling of nanofiltration membranes in full- and bench-scale systems treating groundwater containing silica. J. Membr. Sci. 2014, 468, 349–359. [Google Scholar] [CrossRef]
- Bogler, A.; Lin, S.; Bar-Zeev, E. Biofouling of membrane distillation, forward osmosis and pressure retarded osmosis: Principles, impacts and future directions. J. Membr. Sci. 2017, 542, 378–398. [Google Scholar] [CrossRef]
- Bucs, S.S.; Valladares Linares, R.; Vrouwenvelder, J.S.; Picioreanu, C. Biofouling in forward osmosis systems: An experimental and numerical study. Water Res. 2016, 106, 86–97. [Google Scholar] [CrossRef]
- Vrouwenvelder, J.S.; Manolarakis, S.A.; van der Hoek, J.P.; van Paassen, J.A.M.; van der Meer, W.G.J.; van Agtmaal, J.M.C.; Prummel, H.D.M.; Kruithof, J.C.; van Loosdrecht, M.C.M. Quantitative biofouling diagnosis in full scale nanofiltration and reverse osmosis installations. Water Res. 2008, 42, 4856–4868. [Google Scholar] [CrossRef]
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Mariñas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef]
- O’Toole, G.; Kaplan, H.B.; Kolter, R. Biofilm formation as microbial development. Annu. Rev. Microbiol. 2000, 54, 49–79. [Google Scholar] [CrossRef]
- Abid, H.S.; Johnson, D.J.; Hashaikeh, R.; Hilal, N. A review of efforts to reduce membrane fouling by control of feed spacer characteristics. Desalination 2017, 420, 384–402. [Google Scholar] [CrossRef]
- Hausman, R.; Gullinkala, T.; Escobar, I.C. Development of low-biofouling polypropylene feedspacers for reverse osmosis. J. Appl. Polym. Sci. 2009, 114, 3068–3073. [Google Scholar] [CrossRef]
- Goulter, R.M.; Gentle, I.R.; Dykes, G.A. Issues in determining factors influencing bacterial attachment: A review using the attachment of Escherichia coli to abiotic surfaces as an example. Lett. Appl. Microbiol. 2009, 49, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cornel, P.K.; Summers, R.S.; Roberts, P.V. Diffusion of humic acid in dilute aqueous solution. J. Colloid Interface Sci. 1986, 110, 149–164. [Google Scholar] [CrossRef]
- Valladares Linares, R.; Li, Z.; Sarp, S.; Bucs, S.S.; Amy, G.; Vrouwenvelder, J.S. Forward osmosis niches in seawater desalination and wastewater reuse. Water Res 2014, 66, 122–139. [Google Scholar] [CrossRef]
- Lee, S.; Boo, C.; Elimelech, M.; Hong, S. Comparison of fouling behavior in forward osmosis (FO) and reverse osmosis (RO). J. Membr. Sci. 2010, 365, 34–39. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Yangali-Quintanilla, V.; Valladares-Linares, R.; Li, Q.; Zhan, T.; Amy, G. Flux patterns and membrane fouling propensity during desalination of seawater by forward osmosis. Water Res. 2012, 46, 195–204. [Google Scholar] [CrossRef]
- Myint, A.A.; Lee, W.; Mun, S.; Ahn, C.H.; Lee, S.; Yoon, J. Influence of membrane surface properties on the behavior of initial bacterial adhesion and biofilm development onto nanofiltration membranes. Biofouling 2010, 26, 313–321. [Google Scholar] [CrossRef]
- Yoon, H.; Baek, Y.; Yu, J.; Yoon, J. Biofouling occurrence process and its control in the forward osmosis. Desalination 2013, 325, 30–36. [Google Scholar] [CrossRef]
- Mi, B.; Elimelech, M. Organic fouling of forward osmosis membranes: Fouling reversibility and cleaning without chemical reagents. J. Membr. Sci. 2010, 348, 337–345. [Google Scholar] [CrossRef]
- An, Y.H.; Friedman, R.J. Concise review of mechanisms of bacterial adhesion to biomaterial surfaces. J. Biomed. Mater. Res. 1998, 43, 338–348. [Google Scholar] [CrossRef]
- Gu, Y.; Wang, Y.-N.; Wei, J.; Tang, C.Y. Organic fouling of thin-film composite polyamide and cellulose triacetate forward osmosis membranes by oppositely charged macromolecules. Water Res. 2013, 47, 1867–1874. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Roddick, F.A.; Fan, L. Biofouling of Water Treatment Membranes: A Review of the Underlying Causes, Monitoring Techniques and Control Measures. Membranes 2012, 2, 804–840. [Google Scholar] [CrossRef]
- Amy, G. Fundamental understanding of organic matter fouling of membranes. Desalination 2008, 231, 44–51. [Google Scholar] [CrossRef]
- Parida, V.; Ng, H.Y. Forward osmosis organic fouling: Effects of organic loading, calcium and membrane orientation. Desalination 2013, 312, 88–98. [Google Scholar] [CrossRef]
- Combe, C.; Molis, E.; Lucas, P.; Riley, R.; Clark, M.M. The effect of CA membrane properties on adsorptive fouling by humic acid. J. Membr. Sci. 1999, 154, 73–87. [Google Scholar] [CrossRef]
- Jones, K.L.; O’Melia, C.R. Protein and humic acid adsorption onto hydrophilic membrane surfaces: Effects of pH and ionic strength. J. Membr. Sci. 2000, 165, 31–46. [Google Scholar] [CrossRef]
- Yuan, W.; Zydney, A.L. Humic acid fouling during microfiltration. J. Membr. Sci. 1999, 157, 1–12. [Google Scholar] [CrossRef]
- Shon, H.K.; Vigneswaran, S.; Kim, I.S.; Cho, J.; Ngo, H.H. Fouling of ultrafiltration membrane by effluent organic matter: A detailed characterization using different organic fractions in wastewater. J. Membr. Sci. 2006, 278, 232–238. [Google Scholar] [CrossRef]
- Xie, M.; Nghiem, L.D.; Price, W.E.; Elimelech, M. Impact of humic acid fouling on membrane performance and transport of pharmaceutically active compounds in forward osmosis. Water Res. 2013, 47, 4567–4575. [Google Scholar] [CrossRef]
- Jarusutthirak, C.; Amy, G.; Croué, J.-P. Fouling characteristics of wastewater effluent organic matter (EfOM) isolates on NF and UF membranes. Desalination 2002, 145, 247–255. [Google Scholar] [CrossRef]
- Fan, L.; Harris, J.L.; Roddick, F.A.; Booker, N.A. Influence of the characteristics of natural organic matter on the fouling of microfiltration membranes. Water Res. 2001, 35, 4455–4463. [Google Scholar] [CrossRef]
- Mi, B.; Elimelech, M. Chemical and physical aspects of organic fouling of forward osmosis membranes. J. Membr. Sci. 2008, 320, 292–302. [Google Scholar] [CrossRef]
- Fane, T. Inorganic Scaling. In Encyclopedia of Membranes; Drioli, E., Giorno, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 1–2. [Google Scholar]
- Mi, B.; Elimelech, M. Silica scaling and scaling reversibility in forward osmosis. Desalination 2013, 312, 75–81. [Google Scholar] [CrossRef]
- Xie, M.; Gray, S.R. Silica scaling in forward osmosis: From solution to membrane interface. Water Res. 2017, 108, 232–239. [Google Scholar] [CrossRef]
- Lee, S.; Kim, Y.C. Calcium carbonate scaling by reverse draw solute diffusion in a forward osmosis membrane for shale gas wastewater treatment. J. Membr. Sci. 2017, 522, 257–266. [Google Scholar] [CrossRef]
- Zhang, M.; Shan, J.; Tang, C.Y. Gypsum scaling during forward osmosis process—A direct microscopic observation study. Desalin. Water Treat. 2016, 57, 3317–3327. [Google Scholar] [CrossRef]
- Mi, B.; Elimelech, M. Gypsum Scaling and Cleaning in Forward Osmosis: Measurements and Mechanisms. Environ. Sci. Technol. 2010, 44, 2022–2028. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Kim, S.-H.; Jeong, S.; Hwang, T.-M. Application of ultrasound to mitigate calcium sulfate scaling and colloidal fouling. Desalination 2014, 336, 153–159. [Google Scholar] [CrossRef]
- Bush, J.A.; Vanneste, J.; Gustafson, E.M.; Waechter, C.A.; Jassby, D.; Turchi, C.S.; Cath, T.Y. Prevention and management of silica scaling in membrane distillation using pH adjustment. J. Membr. Sci. 2018, 554, 366–377. [Google Scholar] [CrossRef]
- Brück, A.; McCoy, L.L.; Kilway, K.V. Hydrogen Bonds in Carboxylic Acid−Carboxylate Systems in Solution. 1. In Anhydrous, Aprotic Media. Org. Lett. 2000, 2, 2007–2009. [Google Scholar] [CrossRef]
- Xie, M.; Gray, S.R. Gypsum scaling in forward osmosis: Role of membrane surface chemistry. J. Membr. Sci. 2016, 513, 250–259. [Google Scholar] [CrossRef]
- Hatziantoniou, D.; Howell, J.A. Influence of the properties and characteristics of sugar-beet pulp extract on its fouling and rejection behaviour during membrane filtration. Desalination 2002, 148, 67–72. [Google Scholar] [CrossRef]
- Lee, S.; Elimelech, M. Relating Organic Fouling of Reverse Osmosis Membranes to Intermolecular Adhesion Forces. Environ. Sci. Technol. 2006, 40, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Song, L. Experimental correlations of pH and ionic strength effects on the colloidal fouling potential of silica nanoparticles in crossflow ultrafiltration. J. Membr. Sci. 2007, 303, 112–118. [Google Scholar] [CrossRef]
- Brunelle, M.T. Colloidal fouling of reverse osmosis membranes. Desalination 1980, 32, 127–135. [Google Scholar] [CrossRef]
- Boo, C.; Lee, S.; Elimelech, M.; Meng, Z.; Hong, S. Colloidal fouling in forward osmosis: Role of reverse salt diffusion. J. Membr. Sci. 2012, 390–391, 277–284. [Google Scholar] [CrossRef]
- Zou, S.; Gu, Y.; Xiao, D.; Tang, C.Y. The role of physical and chemical parameters on forward osmosis membrane fouling during algae separation. J. Membr. Sci. 2011, 366, 356–362. [Google Scholar] [CrossRef]
- Espinasse, B.; Bacchin, P.; Aimar, P. On an experimental method to measure critical flux in ultrafiltration. Desalination 2002, 146, 91–96. [Google Scholar] [CrossRef]
- Bacchin, P.; Aimar, P.; Field, R.W. Critical and sustainable fluxes: Theory, experiments and applications. J. Membr. Sci. 2006, 281, 42–69. [Google Scholar] [CrossRef]
- Wang, Y.; Wicaksana, F.; Tang, C.Y.; Fane, A.G. Direct Microscopic Observation of Forward Osmosis Membrane Fouling. Environ. Sci. Technol. 2010, 44, 7102–7109. [Google Scholar] [CrossRef]
- Zou, S.; Wang, Y.-N.; Wicaksana, F.; Aung, T.; Wong, P.C.Y.; Fane, A.G.; Tang, C.Y. Direct microscopic observation of forward osmosis membrane fouling by microalgae: Critical flux and the role of operational conditions. J. Membr. Sci. 2013, 436, 174–185. [Google Scholar] [CrossRef]
- Liu, Y.; Mi, B. Combined fouling of forward osmosis membranes: Synergistic foulant interaction and direct observation of fouling layer formation. J. Membr. Sci. 2012, 407–408, 136–144. [Google Scholar] [CrossRef]
- Zhao, S.; Zou, L.; Mulcahy, D. Effects of membrane orientation on process performance in forward osmosis applications. J. Membr. Sci. 2011, 382, 308–315. [Google Scholar] [CrossRef]
- Park, M.J.; Gonzales, R.R.; Abdel-Wahab, A.; Phuntsho, S.; Shon, H.K. Hydrophilic polyvinyl alcohol coating on hydrophobic electrospun nanofiber membrane for high performance thin film composite forward osmosis membrane. Desalination 2018, 426, 50–59. [Google Scholar] [CrossRef]
- Kumar, R.; Ismail, A.F. Fouling control on microfiltration/ultrafiltration membranes: Effects of morphology, hydrophilicity, and charge. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Yu, Y.; Seo, S.; Kim, I.-C.; Lee, S. Nanoporous polyethersulfone (PES) membrane with enhanced flux applied in forward osmosis process. J. Membr. Sci. 2011, 375, 63–68. [Google Scholar] [CrossRef]
- Emadzadeh, D.; Lau, W.J.; Matsuura, T.; Rahbari-Sisakht, M.; Ismail, A.F. A novel thin film composite forward osmosis membrane prepared from PSf–TiO2 nanocomposite substrate for water desalination. Chem. Eng. J. 2014, 237, 70–80. [Google Scholar] [CrossRef]
- Setiawan, L.; Wang, R.; Li, K.; Fane, A.G. Fabrication of novel poly(amide–imide) forward osmosis hollow fiber membranes with a positively charged nanofiltration-like selective layer. J. Membr. Sci. 2011, 369, 196–205. [Google Scholar] [CrossRef]
- Chiao, Y.-H.; Sengupta, A.; Chen, S.-T.; Huang, S.-H.; Hu, C.-C.; Hung, W.-S.; Chang, Y.; Qian, X.; Wickramasinghe, S.R.; Lee, K.-R.; et al. Zwitterion augmented polyamide membrane for improved forward osmosis performance with significant antifouling characteristics. Sep. Purif. Technol. 2019, 212, 316–325. [Google Scholar] [CrossRef]
- Choi, H.-G.; Shah, A.A.; Nam, S.-E.; Park, Y.-I.; Park, H. Thin-film composite membranes comprising ultrathin hydrophilic polydopamine interlayer with graphene oxide for forward osmosis. Desalination 2019, 449, 41–49. [Google Scholar] [CrossRef]
- Ni, T.; Ge, Q. Highly hydrophilic thin-film composition forward osmosis (FO) membranes functionalized with aniline sulfonate/bisulfonate for desalination. J. Membr. Sci. 2018, 564, 732–741. [Google Scholar] [CrossRef]
- Shakeri, A.; Salehi, H.; Ghorbani, F.; Amini, M.; Naslhajian, H. Polyoxometalate based thin film nanocomposite forward osmosis membrane: Superhydrophilic, anti-fouling, and high water permeable. J. Colloid Interface Sci. 2019, 536, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Chun, Y.; Qing, L.; Sun, G.; Bilad, M.R.; Fane, A.G.; Chong, T.H. Prototype aquaporin-based forward osmosis membrane: Filtration properties and fouling resistance. Desalination 2018, 445, 75–84. [Google Scholar] [CrossRef]
- Rastgar, M.; Bozorg, A.; Shakeri, A.; Sadrzadeh, M. Substantially improved antifouling properties in electro-oxidative graphene laminate forward osmosis membrane. Chem. Eng. Res. Des. 2019, 141, 413–424. [Google Scholar] [CrossRef]
- Bao, X.; Wu, Q.; Shi, W.; Wang, W.; Yu, H.; Zhu, Z.; Zhang, X.; Zhang, Z.; Zhang, R.; Cui, F. Polyamidoamine dendrimer grafted forward osmosis membrane with superior ammonia selectivity and robust antifouling capacity for domestic wastewater concentration. Water Res. 2019, 153, 1–10. [Google Scholar] [CrossRef]
- Shakeri, A.; Salehi, H.; Rastgar, M. Chitosan-based thin active layer membrane for forward osmosis desalination. Carbohydr. Polym. 2017, 174, 658–668. [Google Scholar] [CrossRef]
- Li, M.-N.; Sun, X.-F.; Wang, L.; Wang, S.-Y.; Afzal, M.Z.; Song, C.; Wang, S.-G. Forward osmosis membranes modified with laminar MoS2 nanosheet to improve desalination performance and antifouling properties. Desalination 2018, 436, 107–113. [Google Scholar] [CrossRef]
- Qiu, M.; He, C. Novel zwitterion-silver nanocomposite modified thin-film composite forward osmosis membrane with simultaneous improved water flux and biofouling resistance property. Appl. Surf. Sci. 2018, 455, 492–501. [Google Scholar] [CrossRef]
- Zuo, H.-R.; Fu, J.-B.; Cao, G.-P.; Hu, N.; Lu, H.; Liu, H.-Q.; Chen, P.-P.; Yu, J. The effects of surface-charged submicron polystyrene particles on the structure and performance of PSF forward osmosis membrane. Appl. Surf. Sci. 2018, 436, 1181–1192. [Google Scholar] [CrossRef]
- Guo, H.; Yao, Z.; Wang, J.; Yang, Z.; Ma, X.; Tang, C.Y. Polydopamine coating on a thin film composite forward osmosis membrane for enhanced mass transport and antifouling performance. J. Membr. Sci. 2018, 551, 234–242. [Google Scholar] [CrossRef]
- Xu, W.; Ge, Q. Novel functionalized forward osmosis (FO) membranes for FO desalination: Improved process performance and fouling resistance. J. Membr. Sci. 2018, 555, 507–516. [Google Scholar] [CrossRef]
- Han, G.; Chung, T.-S.; Toriida, M.; Tamai, S. Thin-film composite forward osmosis membranes with novel hydrophilic supports for desalination. J. Membr. Sci. 2012, 423–424, 543–555. [Google Scholar] [CrossRef]
- Yip, N.Y.; Tiraferri, A.; Phillip, W.A.; Schiffman, J.D.; Elimelech, M. High Performance Thin-Film Composite Forward Osmosis Membrane. Environ. Sci. Technol. 2010, 44, 3812–3818. [Google Scholar] [CrossRef] [PubMed]
- Widjojo, N.; Chung, T.-S.; Weber, M.; Maletzko, C.; Warzelhan, V. The role of sulphonated polymer and macrovoid-free structure in the support layer for thin-film composite (TFC) forward osmosis (FO) membranes. J. Membr. Sci. 2011, 383, 214–223. [Google Scholar] [CrossRef]
- Tiraferri, A.; Yip, N.Y.; Phillip, W.A.; Schiffman, J.D.; Elimelech, M. Relating performance of thin-film composite forward osmosis membranes to support layer formation and structure. J. Membr. Sci. 2011, 367, 340–352. [Google Scholar] [CrossRef]
- Lay, W.C.L.; Zhang, J.; Tang, C.; Wang, R.; Liu, Y.; Fane, A.G. Factors affecting flux performance of forward osmosis systems. J. Membr. Sci. 2012, 394–395, 151–168. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; Elimelech, M. Influence of concentrative and dilutive internal concentration polarization on flux behavior in forward osmosis. J. Membr. Sci. 2006, 284, 237–247. [Google Scholar] [CrossRef]
- Cath, T.; Childress, A.; Elimelech, M. Forward osmosis: Principles, applications, and recent developments. J. Membr. Sci. 2006, 281, 70–87. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; McGinnis, R.L.; Elimelech, M. Desalination by ammonia–carbon dioxide forward osmosis: Influence of draw and feed solution concentrations on process performance. J. Membr. Sci. 2006, 278, 114–123. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, M.; Liu, Y.; Xiao, Q.; Xu, S. Quantitative evaluation of concentration polarization under different operating conditions for forward osmosis process. Desalination 2016, 398, 106–113. [Google Scholar] [CrossRef]
- Kragl, U. Basic Principles of Membrane Technology. (2. Aufl.) Von M. Mulder. Kluwer Academic Publishers, Dordrecht, 1996. 564 S., geb. 174.00 £.—ISBN 0–7923–4247-X. Angew. Chem. 1997, 109, 1420–1421. [Google Scholar] [CrossRef]
- Gruber, M.F.; Johnson, C.J.; Tang, C.Y.; Jensen, M.H.; Yde, L.; Hélix-Nielsen, C. Computational fluid dynamics simulations of flow and concentration polarization in forward osmosis membrane systems. J. Membr. Sci. 2011, 379, 488–495. [Google Scholar] [CrossRef]
- Zhang, H.; Cheng, S.; Yang, F. Use of a spacer to mitigate concentration polarization during forward osmosis process. Desalination 2014, 347, 112–119. [Google Scholar] [CrossRef]
- Gray, G.T.; McCutcheon, J.R.; Elimelech, M. Internal concentration polarization in forward osmosis: Role of membrane orientation. Desalination 2006, 197, 1–8. [Google Scholar] [CrossRef]
- Choi, Y.; Hwang, T.M.; Jeong, S.; Lee, S. The use of ultrasound to reduce internal concentration polarization in forward osmosis. Ultrason. Sonochem. 2018, 41, 475–483. [Google Scholar] [CrossRef]
- Heikkinen, J.; Kyllönen, H.; Järvelä, E.; Grönroos, A.; Tang, C.Y. Ultrasound-assisted forward osmosis for mitigating internal concentration polarization. J. Membr. Sci. 2017, 528, 147–154. [Google Scholar] [CrossRef]
- Wang, Y.; Duan, W.; Wang, W.; Di, W.; Liu, Y.; Liu, Y.; Li, Z.; Hu, H.; Lin, H.; Cui, C.; et al. scAAV9-VEGF prolongs the survival of transgenic ALS mice by promoting activation of M2 microglia and the PI3K/Akt pathway. Brain Res. 2016, 1648, 1–10. [Google Scholar] [CrossRef]
- Xie, M.; Tang, C.Y.; Gray, S.R. Spacer-induced forward osmosis membrane integrity loss during gypsum scaling. Desalination 2016, 392, 85–90. [Google Scholar] [CrossRef]
- She, Q.; Jin, X.; Tang, C.Y. Osmotic power production from salinity gradient resource by pressure retarded osmosis: Effects of operating conditions and reverse solute diffusion. J. Membr. Sci. 2012, 401–402, 262–273. [Google Scholar] [CrossRef]
- Hawari, A.H.; Kamal, N.; Altaee, A. Combined influence of temperature and flow rate of feeds on the performance of forward osmosis. Desalination 2016, 398, 98–105. [Google Scholar] [CrossRef]
- Jung, D.H.; Lee, J.; Kim, D.Y.; Lee, Y.G.; Park, M.; Lee, S.; Yang, D.R.; Kim, J.H. Simulation of forward osmosis membrane process: Effect of membrane orientation and flow direction of feed and draw solutions. Desalination 2011, 277, 83–91. [Google Scholar] [CrossRef]
- Wang, K.Y.; Ong, R.C.; Chung, T.-S. Double-Skinned Forward Osmosis Membranes for Reducing Internal Concentration Polarization within the Porous Sublayer. Ind. Eng. Chem. Res. 2010, 49, 4824–4831. [Google Scholar] [CrossRef]
- Wei, J.; Qiu, C.; Tang, C.Y.; Wang, R.; Fane, A.G. Synthesis and characterization of flat-sheet thin film composite forward osmosis membranes. J. Membr. Sci. 2011, 372, 292–302. [Google Scholar] [CrossRef]
- Song, X.; Liu, Z.; Sun, D.D. Nano Gives the Answer: Breaking the Bottleneck of Internal Concentration Polarization with a Nanofiber Composite Forward Osmosis Membrane for a High Water Production Rate. Adv. Mater. 2011, 23, 3256–3260. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Lee, J.Y.; Chung, T.-S. Thin film composite forward-osmosis membranes with enhanced internal osmotic pressure for internal concentration polarization reduction. Chem. Eng. J. 2014, 249, 236–245. [Google Scholar] [CrossRef]
- Liu, X.; Ng, H.Y. Fabrication of layered silica–polysulfone mixed matrix substrate membrane for enhancing performance of thin-film composite forward osmosis membrane. J. Membr. Sci. 2015, 481, 148–163. [Google Scholar] [CrossRef]
- Puguan, J.M.C.; Kim, H.-S.; Lee, K.-J.; Kim, H. Low internal concentration polarization in forward osmosis membranes with hydrophilic crosslinked PVA nanofibers as porous support layer. Desalination 2014, 336, 24–31. [Google Scholar] [CrossRef]
- Li, M.; Karanikola, V.; Zhang, X.; Wang, L.; Elimelech, M. A Self-Standing, Support-Free Membrane for Forward Osmosis with No Internal Concentration Polarization. Environ. Sci. Technol. Lett. 2018, 5, 266–271. [Google Scholar] [CrossRef]
- Gai, J.-G.; Gong, X.-L. Zero internal concentration polarization FO membrane: Functionalized graphene. J. Mater. Chem. A 2014, 2, 425–429. [Google Scholar] [CrossRef]
- Phillip, W.A.; Yong, J.S.; Elimelech, M. Reverse Draw Solute Permeation in Forward Osmosis: Modeling and Experiments. Environ. Sci. Technol. 2010, 44, 5170–5176. [Google Scholar] [CrossRef] [PubMed]
- Hickenbottom, K.L.; Hancock, N.T.; Hutchings, N.R.; Appleton, E.W.; Beaudry, E.G.; Xu, P.; Cath, T.Y. Forward osmosis treatment of drilling mud and fracturing wastewater from oil and gas operations. Desalination 2013, 312, 60–66. [Google Scholar] [CrossRef]
- Zhao, S.; Zou, L.; Tang, C.Y.; Mulcahy, D. Recent developments in forward osmosis: Opportunities and challenges. J. Membr. Sci. 2012, 396, 1–21. [Google Scholar] [CrossRef]
- Xie, M.; Bar-Zeev, E.; Hashmi, S.M.; Nghiem, L.D.; Elimelech, M. Role of Reverse Divalent Cation Diffusion in Forward Osmosis Biofouling. Environ. Sci. Technol. 2015, 49, 13222–13229. [Google Scholar] [CrossRef]
- Nguyen, N.C.; Chen, S.S.; Jain, S.; Nguyen, H.T.; Ray, S.S.; Ngo, H.H.; Guo, W.; Lam, N.T.; Duong, H.C. Exploration of an innovative draw solution for a forward osmosis-membrane distillation desalination process. Environ. Sci. Pollut. Res. Int. 2018, 25, 5203–5211. [Google Scholar] [CrossRef]
- Achilli, A.; Cath, T.Y.; Childress, A.E. Selection of inorganic-based draw solutions for forward osmosis applications. J. Membr. Sci. 2010, 364, 233–241. [Google Scholar] [CrossRef]
- Yaroshchuk, A.; Bruening, M.L.; Licón Bernal, E.E. Solution-Diffusion–Electro-Migration model and its uses for analysis of nanofiltration, pressure-retarded osmosis and forward osmosis in multi-ionic solutions. J. Membr. Sci. 2013, 447, 463–476. [Google Scholar] [CrossRef]
- Irvine, G.J.; Rajesh, S.; Georgiadis, M.; Phillip, W.A. Ion Selective Permeation Through Cellulose Acetate Membranes in Forward Osmosis. Environ. Sci. Technol. 2013, 47, 13745–13753. [Google Scholar] [CrossRef] [PubMed]
- Boo, C.; Khalil, Y.F.; Elimelech, M. Performance evaluation of trimethylamine–carbon dioxide thermolytic draw solution for engineered osmosis. J. Membr. Sci. 2015, 473, 302–309. [Google Scholar] [CrossRef]
- Phuntsho, S.; Shon, H.K.; Hong, S.; Lee, S.; Vigneswaran, S. A novel low energy fertilizer driven forward osmosis desalination for direct fertigation: Evaluating the performance of fertilizer draw solutions. J. Membr. Sci. 2011, 375, 172–181. [Google Scholar] [CrossRef]
- She, Q.; Jin, X.; Li, Q.; Tang, C.Y. Relating reverse and forward solute diffusion to membrane fouling in osmotically driven membrane processes. Water Res. 2012, 46, 2478–2486. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, K.Y.; Chung, T.-S.; Chen, H.; Jean, Y.C.; Amy, G. Well-constructed cellulose acetate membranes for forward osmosis: Minimized internal concentration polarization with an ultra-thin selective layer. J. Membr. Sci. 2010, 360, 522–535. [Google Scholar] [CrossRef]
- Blandin, G.; Verliefde, A.R.D.; Tang, C.Y.; Childress, A.E.; Le-Clech, P. Validation of assisted forward osmosis (AFO) process: Impact of hydraulic pressure. J. Membr. Sci. 2013, 447, 1–11. [Google Scholar] [CrossRef]
- Tang, C.Y.; She, Q.; Lay, W.C.L.; Wang, R.; Fane, A.G. Coupled effects of internal concentration polarization and fouling on flux behavior of forward osmosis membranes during humic acid filtration. J. Membr. Sci. 2010, 354, 123–133. [Google Scholar] [CrossRef]
- Phuntsho, S.; Sahebi, S.; Majeed, T.; Lotfi, F.; Kim, J.E.; Shon, H.K. Assessing the major factors affecting the performances of forward osmosis and its implications on the desalination process. Chem. Eng. J. 2013, 231, 484–496. [Google Scholar] [CrossRef]
- Achilli, A.; Cath, T.Y.; Marchand, E.A.; Childress, A.E. The forward osmosis membrane bioreactor: A low fouling alternative to MBR processes. Desalination 2009, 239, 10–21. [Google Scholar] [CrossRef]
- Zhang, J.; Loong, W.L.C.; Chou, S.; Tang, C.; Wang, R.; Fane, A.G. Membrane biofouling and scaling in forward osmosis membrane bioreactor. J. Membr. Sci. 2012, 403–404, 8–14. [Google Scholar] [CrossRef]
- Sun, Y.; Tian, J.; Zhao, Z.; Shi, W.; Liu, D.; Cui, F. Membrane fouling of forward osmosis (FO) membrane for municipal wastewater treatment: A comparison between direct FO and OMBR. Water Res. 2016, 104, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Tian, J.; Song, L.; Gao, S.; Shi, W.; Cui, F. Dynamic changes of the fouling layer in forward osmosis based membrane processes for municipal wastewater treatment. J. Membr. Sci. 2018, 549, 523–532. [Google Scholar] [CrossRef]
- Cornelissen, E.R.; Harmsen, D.; de Korte, K.F.; Ruiken, C.J.; Qin, J.-J.; Oo, H.; Wessels, L.P. Membrane fouling and process performance of forward osmosis membranes on activated sludge. J. Membr. Sci. 2008, 319, 158–168. [Google Scholar] [CrossRef]
- Qiu, G.; Ting, Y.-P. Short-term fouling propensity and flux behavior in an osmotic membrane bioreactor for wastewater treatment. Desalination 2014, 332, 91–99. [Google Scholar] [CrossRef]
- Chen, M.-Y.; Lee, D.-J.; Yang, Z.; Peng, X.F.; Lai, J.Y. Fluorecent Staining for Study of Extracellular Polymeric Substances in Membrane Biofouling Layers. Environ. Sci. Technol. 2006, 40, 6642–6646. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhao, Y.; Yuan, B.; Wang, Z.; Li, X.; Ren, Y. Comparison of biofouling mechanisms between cellulose triacetate (CTA) and thin-film composite (TFC) polyamide forward osmosis membranes in osmotic membrane bioreactors. Bioresour. Technol. 2016, 202, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chang, V.W.C.; Tang, C.Y. Osmotic membrane bioreactor (OMBR) technology for wastewater treatment and reclamation: Advances, challenges, and prospects for the future. J. Membr. Sci. 2016, 504, 113–132. [Google Scholar] [CrossRef]
- Yuan, B.; Wang, X.; Tang, C.; Li, X.; Yu, G. In situ observation of the growth of biofouling layer in osmotic membrane bioreactors by multiple fluorescence labeling and confocal laser scanning microscopy. Water Res. 2015, 75, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Bell, E.A.; Holloway, R.W.; Cath, T.Y. Evaluation of forward osmosis membrane performance and fouling during long-term osmotic membrane bioreactor study. J. Membr. Sci. 2016, 517, 1–13. [Google Scholar] [CrossRef]
- Wang, X.-M.; Li, X.-Y. Accumulation of biopolymer clusters in a submerged membrane bioreactor and its effect on membrane fouling. Water Res. 2008, 42, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Tang, C.Y.; Gu, Y.; She, Q.; Qi, S. Boric Acid Permeation in Forward Osmosis Membrane Processes: Modeling, Experiments, and Implications. Environ. Sci. Technol. 2011, 45, 2323–2330. [Google Scholar] [CrossRef]
- Qin, J.J.; Kekre, K.A.; Oo, M.H.; Tao, G.; Lay, C.L.; Lew, C.H.; Cornelissen, E.R.; Ruiken, C.J. Preliminary study of osmotic membrane bioreactor: Effects of draw solution on water flux and air scouring on fouling. Water Sci Technol 2010, 62, 1353–1360. [Google Scholar] [CrossRef]
- Luo, W.; Xie, M.; Hai, F.I.; Price, W.E.; Nghiem, L.D. Biodegradation of cellulose triacetate and polyamide forward osmosis membranes in an activated sludge bioreactor: Observations and implications. J. Membr. Sci. 2016, 510, 284–292. [Google Scholar] [CrossRef]
- Coday, B.D.; Xu, P.; Beaudry, E.G.; Herron, J.; Lampi, K.; Hancock, N.T.; Cath, T.Y. The sweet spot of forward osmosis: Treatment of produced water, drilling wastewater, and other complex and difficult liquid streams. Desalination 2014, 333, 23–35. [Google Scholar] [CrossRef]
- Valladares Linares, R.; Li, Z.; Yangali-Quintanilla, V.; Li, Q.; Amy, G. Cleaning protocol for a FO membrane fouled in wastewater reuse. Desalin. Water Treat. 2013, 51, 4821–4824. [Google Scholar] [CrossRef]
- Lv, L.; Xu, J.; Shan, B.; Gao, C. Concentration performance and cleaning strategy for controlling membrane fouling during forward osmosis concentration of actual oily wastewater. J. Membr. Sci. 2017, 523, 15–23. [Google Scholar] [CrossRef]
- Kim, C.-M.; Kim, S.-J.; Kim, L.H.; Shin, M.S.; Yu, H.-W.; Kim, I.S. Effects of phosphate limitation in feed water on biofouling in forward osmosis (FO) process. Desalination 2014, 349, 51–59. [Google Scholar] [CrossRef]
- Alphenaar, P.A.; Sleyster, R.; De Reuver, P.; Ligthart, G.-J.; Lettinga, G. Phosphorus requirement in high-rate anaerobic wastewater treatment. Water Res. 1993, 27, 749–756. [Google Scholar] [CrossRef]
- Lehtola, M.J.; Miettinen, I.T.; Vartiainen, T.; Rantakokko, P.; Hirvonen, A.; Martikainen, P.J. Impact of UV disinfection on microbially available phosphorus, organic carbon, and microbial growth in drinking water. Water Res. 2003, 37, 1064–1070. [Google Scholar] [CrossRef]
- Kasahara, S.; Maeda, K.; Ishikawa, M. Influence of phosphorus on biofilm accumulation in drinking water distribution systems. Water Sci. Technol. Water Supply 2004, 4, 389–398. [Google Scholar] [CrossRef]
- Karageorgiou, K.; Paschalis, M.; Anastassakis, G.N. Removal of phosphate species from solution by adsorption onto calcite used as natural adsorbent. J. Hazard. Mater. 2007, 139, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, J.; Das, J.; Das, S.; Thakur, R.S. Adsorption of Phosphate from Aqueous Solution Using Activated Red Mud. J. Colloid Interface Sci. 1998, 204, 169–172. [Google Scholar] [CrossRef]
- Li, Y.; Liu, C.; Luan, Z.; Peng, X.; Zhu, C.; Chen, Z.; Zhang, Z.; Fan, J.; Jia, Z. Phosphate removal from aqueous solutions using raw and activated red mud and fly ash. J. Hazard. Mater. 2006, 137, 374–383. [Google Scholar] [CrossRef]
- Vasudevan, S.; Sozhan, G.; Ravichandran, S.; Jayaraj, J.; Lakshmi, J.; Sheela, M. Studies on the removal of phosphate from drinking water by electrocoagulation process. Ind. Eng. Chem. Res. 2008, 47, 2018–2023. [Google Scholar] [CrossRef]
- Fytianos, K.; Voudrias, E.; Raikos, N. Modelling of phosphorus removal from aqueous and wastewater samples using ferric iron. Environ. Pollut. 1998, 101, 123–130. [Google Scholar] [CrossRef]
- Wang, Z.; Tang, J.; Zhu, C.; Dong, Y.; Wang, Q.; Wu, Z. Chemical cleaning protocols for thin film composite (TFC) polyamide forward osmosis membranes used for municipal wastewater treatment. J. Membr. Sci. 2015, 475, 184–192. [Google Scholar] [CrossRef]
- Kwan, S.E.; Bar-Zeev, E.; Elimelech, M. Biofouling in forward osmosis and reverse osmosis: Measurements and mechanisms. J. Membr. Sci. 2015, 493, 703–708. [Google Scholar] [CrossRef]
- Du, X.; Wang, Y.; Qu, F.; Li, K.; Liu, X.; Wang, Z.; Li, G.; Liang, H. Impact of bubbly flow in feed channel of forward osmosis for wastewater treatment: Flux performance and biofouling. Chem. Eng. J. 2017, 316, 1047–1058. [Google Scholar] [CrossRef]
- Li, J.-Y.; Ni, Z.-Y.; Zhou, Z.-Y.; Hu, Y.-X.; Xu, X.-H.; Cheng, L.-H. Membrane fouling of forward osmosis in dewatering of soluble algal products: Comparison of TFC and CTA membranes. J. Membr. Sci. 2018, 552, 213–221. [Google Scholar] [CrossRef]
- Blandin, G.; Vervoort, H.; Le-Clech, P.; Verliefde, A.R.D. Fouling and cleaning of high permeability forward osmosis membranes. J. Water Process Eng. 2016, 9, 161–169. [Google Scholar] [CrossRef]
- Boo, C.; Elimelech, M.; Hong, S. Fouling control in a forward osmosis process integrating seawater desalination and wastewater reclamation. J. Membr. Sci. 2013, 444, 148–156. [Google Scholar] [CrossRef]
- Kim, Y.; Elimelech, M.; Shon, H.K.; Hong, S. Combined organic and colloidal fouling in forward osmosis: Fouling reversibility and the role of applied pressure. J. Membr. Sci. 2014, 460, 206–212. [Google Scholar] [CrossRef]
- Jin, X.; She, Q.; Ang, X.; Tang, C.Y. Removal of boron and arsenic by forward osmosis membrane: Influence of membrane orientation and organic fouling. J. Membr. Sci. 2012, 389, 182–187. [Google Scholar] [CrossRef]
- Valladares Linares, R.; Bucs, S.S.; Li, Z.; AbuGhdeeb, M.; Amy, G.; Vrouwenvelder, J.S. Impact of spacer thickness on biofouling in forward osmosis. Water Res. 2014, 57, 223–233. [Google Scholar] [CrossRef]
- Gwak, G.; Hong, S. New approach for scaling control in forward osmosis (FO) by using an antiscalant-blended draw solution. J. Membr. Sci. 2017, 530, 95–103. [Google Scholar] [CrossRef]
- Hancock, N.T.; Cath, T.Y. Solute Coupled Diffusion in Osmotically Driven Membrane Processes. Environ. Sci. Technol. 2009, 43, 6769–6775. [Google Scholar] [CrossRef]
- Ge, Q.; Amy, G.L.; Chung, T.-S. Forward osmosis for oily wastewater reclamation: Multi-charged oxalic acid complexes as draw solutes. Water Res. 2017, 122, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Valladares Linares, R.; Yangali-Quintanilla, V.; Li, Z.; Amy, G. NOM and TEP fouling of a forward osmosis (FO) membrane: Foulant identification and cleaning. J. Membr. Sci. 2012, 421–422, 217–224. [Google Scholar] [CrossRef]
- Silva, L.F.; Michel, R.C.; Borges, C.P. Modification of polyamide reverse osmosis membranes seeking for better resistance to oxidizing agents. Membr. Water Treat. 2012, 3. [Google Scholar] [CrossRef]
- Park, J.; Jeong, W.; Nam, J.; Kim, J.; Kim, J.; Chon, K.; Lee, E.; Kim, H.; Jang, A. An analysis of the effects of osmotic backwashing on the seawater reverse osmosis process. Environ. Technol. 2014, 35, 1455–1461. [Google Scholar] [CrossRef] [PubMed]
- Holloway, R.W.; Childress, A.E.; Dennett, K.E.; Cath, T.Y. Forward osmosis for concentration of anaerobic digester centrate. Water Res. 2007, 41, 4005–4014. [Google Scholar] [CrossRef] [PubMed]
- Martinetti, C.R.; Childress, A.E.; Cath, T.Y. High recovery of concentrated RO brines using forward osmosis and membrane distillation. J. Membr. Sci. 2009, 331, 31–39. [Google Scholar] [CrossRef]
- Coday, B.D.; Almaraz, N.; Cath, T.Y. Forward osmosis desalination of oil and gas wastewater: Impacts of membrane selection and operating conditions on process performance. J. Membr. Sci. 2015, 488, 40–55. [Google Scholar] [CrossRef]
- Valladares Linares, R.; Li, Z.; Abu-Ghdaib, M.; Wei, C.-H.; Amy, G.; Vrouwenvelder, J.S. Water harvesting from municipal wastewater via osmotic gradient: An evaluation of process performance. J. Membr. Sci. 2013, 447, 50–56. [Google Scholar] [CrossRef]
- Yangali-Quintanilla, V.; Li, Z.; Valladares, R.; Li, Q.; Amy, G. Indirect desalination of Red Sea water with forward osmosis and low pressure reverse osmosis for water reuse. Desalination 2011, 280, 160–166. [Google Scholar] [CrossRef]
- Cornelissen, E.R.; Vrouwenvelder, J.S.; Heijman, S.G.J.; Viallefont, X.D.; Van Der Kooij, D.; Wessels, L.P. Periodic air/water cleaning for control of biofouling in spiral wound membrane elements. J. Membr. Sci. 2007, 287, 94–101. [Google Scholar] [CrossRef]
- D’Haese, A.; Le-Clech, P.; Van Nevel, S.; Verbeken, K.; Cornelissen, E.R.; Khan, S.J.; Verliefde, A.R.D. Trace organic solutes in closed-loop forward osmosis applications: Influence of membrane fouling and modeling of solute build-up. Water Res. 2013, 47, 5232–5244. [Google Scholar] [CrossRef] [PubMed]
- Blanpain-Avet, P.; Doubrovine, N.; Lafforgue, C.; Lalande, M. The effect of oscillatory flow on crossflow microfiltration of beer in a tubular mineral membrane system—Membrane fouling resistance decrease and energetic considerations. J. Membr. Sci. 1999, 152, 151–174. [Google Scholar] [CrossRef]
- Rose, J.; Cho, Y.; Ditri, J. Review of Progress in Quantitative Nondestructive Evaluation; Thompson, D.O., Chimenti, D.E., Eds.; Plenum Press: New York, NY, USA, 1992. [Google Scholar]
- Mairal, A.P.; Greenberg, A.R.; Krantz, W.B.; Bond, L.J. Real-time measurement of inorganic fouling of RO desalination membranes using ultrasonic time-domain reflectometry. J. Membr. Sci. 1999, 159, 185–196. [Google Scholar] [CrossRef]
- Sim, S.T.V.; Suwarno, S.R.; Chong, T.H.; Krantz, W.B.; Fane, A.G. Monitoring membrane biofouling via ultrasonic time-domain reflectometry enhanced by silica dosing. J. Membr. Sci. 2013, 428, 24–37. [Google Scholar] [CrossRef]
- Graf von der Schulenburg, D.A.; Vrouwenvelder, J.S.; Creber, S.A.; van Loosdrecht, M.C.M.; Johns, M.L. Nuclear magnetic resonance microscopy studies of membrane biofouling. J. Membr. Sci. 2008, 323, 37–44. [Google Scholar] [CrossRef]
- Sun, L.; Feng, G.; Lu, W. Fouling Detection Based on Analysis of Ultrasonic Time-Domain Reflectometry Using Wavelet Transform; Springer: Berlin/Heidelberg, Germany, 2011; pp. 347–352. [Google Scholar]
- Sanderson, R.; Li, J.; Koen, L.J.; Lorenzen, L. Ultrasonic time-domain reflectometry as a non-destructive instrumental visualization technique to monitor inorganic fouling and cleaning on reverse osmosis membranes. J. Membr. Sci. 2002, 207, 105–117. [Google Scholar] [CrossRef]
- Abragam, A.; Abragam, A. The Principles of Nuclear Magnetism; Oxford University Press: Oxford, UK, 1961. [Google Scholar]
- Yang, X.; Fridjonsson, E.O.; Johns, M.L.; Wang, R.; Fane, A.G. A non-invasive study of flow dynamics in membrane distillation hollow fiber modules using low-field nuclear magnetic resonance imaging (MRI). J. Membr. Sci. 2014, 451, 46–54. [Google Scholar] [CrossRef]
- Creber, S.A.; Pintelon, T.R.R.; Graf von der Schulenburg, D.A.W.; Vrouwenvelder, J.S.; van Loosdrecht, M.C.M.; Johns, M.L. Magnetic resonance imaging and 3D simulation studies of biofilm accumulation and cleaning on reverse osmosis membranes. Food Bioprod. Process. 2010, 88, 401–408. [Google Scholar] [CrossRef]
- Fridjonsson, E.O.; Vogt, S.J.; Vrouwenvelder, J.S.; Johns, M.L. Early non-destructive biofouling detection in spiral wound RO membranes using a mobile earth׳s field NMR. J. Membr. Sci. 2015, 489, 227–236. [Google Scholar] [CrossRef]
- Amin Saad, M. Early discovery of RO membrane fouling and real-time monitoring of plant performance for optimizing cost of water. Desalination 2004, 165, 183–191. [Google Scholar] [CrossRef]
- Taheri, A.H.; Sim, L.N.; Chong, T.H.; Krantz, W.B.; Fane, A.G. Prediction of reverse osmosis fouling using the feed fouling monitor and salt tracer response technique. J. Membr. Sci. 2015, 475, 433–444. [Google Scholar] [CrossRef]
- Taheri, A.H.; Sim, S.T.V.; Sim, L.N.; Chong, T.H.; Krantz, W.B.; Fane, A.G. Development of a new technique to predict reverse osmosis fouling. J. Membr. Sci. 2013, 448, 12–22. [Google Scholar] [CrossRef]
- Sim, L.N.; Chong, T.H.; Taheri, A.H.; Sim, S.T.V.; Lai, L.; Krantz, W.B.; Fane, A.G. A review of fouling indices and monitoring techniques for reverse osmosis. Desalination 2018, 434, 169–188. [Google Scholar] [CrossRef]
- Park, J.; Jeong, K.; Baek, S.; Park, S.; Ligaray, M.; Chong, T.H.; Cho, K.H. Modeling of NF/RO membrane fouling and flux decline using real-time observations. J. Membr. Sci. 2019, 576, 66–77. [Google Scholar] [CrossRef]
- Fortunato, L.; Bucs, S.; Linares, R.V.; Cali, C.; Vrouwenvelder, J.S.; Leiknes, T. Spatially-resolved in-situ quantification of biofouling using optical coherence tomography (OCT) and 3D image analysis in a spacer filled channel. J. Membr. Sci. 2017, 524, 673–681. [Google Scholar] [CrossRef]
- Kavanagh, J.M.; Hussain, S.; Chilcott, T.C.; Coster, H.G.L. Fouling of reverse osmosis membranes using electrical impedance spectroscopy: Measurements and simulations. Desalination 2009, 236, 187–193. [Google Scholar] [CrossRef]
- Hu, Z.; Antony, A.; Leslie, G.; Le-Clech, P. Real-time monitoring of scale formation in reverse osmosis using electrical impedance spectroscopy. J. Membr. Sci. 2014, 453, 320–327. [Google Scholar] [CrossRef]
- Sim, L.; Wang, Z.; Gu, J.; Coster, H.; Fane, A. Detection of reverse osmosis membrane fouling with silica, bovine serum albumin and their mixture using in-situ electrical impedance spectroscopy. J. Membr. Sci. 2013, 443, 45–53. [Google Scholar] [CrossRef]
- Ho, J.S.; Sim, L.N.; Webster, R.D.; Viswanath, B.; Coster, H.G.; Fane, A.G. Monitoring fouling behavior of reverse osmosis membranes using electrical impedance spectroscopy: A field trial study. Desalination 2017, 407, 75–84. [Google Scholar] [CrossRef]
- Kavanagh, J.; Hussain, S.; Handelsman, T.; Chilcott, T.; Coster, H. Characterisation of Fouled and Cleaned Industrial Reverse Osmosis Membranes by Electrical Impedance Spectroscopy. In Chemeca 2008: Towards a Sustainable Australasia; Engineers Australia: Barton, ACT, Australia, 2008; Volume 202. [Google Scholar]
- Cen, J.; Vukas, M.; Barton, G.; Kavanagh, J.; Coster, H.G.L. Real time fouling monitoring with Electrical Impedance Spectroscopy. J. Membr. Sci. 2015, 484, 133–139. [Google Scholar] [CrossRef]
- Pawley, J.B. Fundamental limits in confocal microscopy. In Handbook of Biological Confocal Microscopy; Springer: Berlin/Heidelberg, Germany, 2006; pp. 20–42. [Google Scholar]

| FO Mode | PRO Mode |
|---|---|
| Effective driving force (πds − πfs) − ∆P | Effective driving force (πds − πfs) − ∆P |
| Loss of driving force due to concentrative external concentration polarization | Loss of driving force due to concentrative external concentration polarization and concentrative internal polarization |
| Loss of driving force due to dilutive internal concentration polarization and dilutive external concentration polarization | Loss of driving force due to dilutive external concentration polarization |
| Base Material | Major Factor Affecting Fouling | Modification | Results | Reference |
|---|---|---|---|---|
| Polyvinylidene flouride (PVDF) nanofiber support | Hydrophilicity and morphology | PVDF nanofiber support was modified via dip coating and crosslinked with glutaraldehyde. | 34.2 improved flux and improved strength | [66] |
| Polysulfone support layer substrate | Hydrophilicity and morphology | A polyamide (PA) layer was formed by interfacial polymerization on the top surface of Psf-TiO2 substrate | Improved water flux | [69] |
| Polysulfone support layer | Hydrophilicity, surface roughness and charge | Zwitterions incorporation onto the polyamide active layer of forward osmosis membrane | Good antifouling properties, marginal reduction in flux with time | [71] |
| Polysulfone support layer | Hydrophilicity | Thin film composite (TFC) membrane was coated with Polydopamine/graphene oxide (PDA/GO) | Enhanced water flux | [72] |
| Polyether sulfone support | Hydrophilicity, charge and morphology | TFC membrane was modified using an aniline sulphonate/bisulphonate functionalized polyamide layer formed by interfacial polymerization on support layer | These membranes had more hydrophilic and smoother surfaces, which increases their antifouling abilities. Higher water recovery efficiency and low reverse salt flux. | [73] |
| PA rejection layer | Hydrophilicity and charge | Wheel POM (polyoxometalates)-coated silica nanoparticles were incorporated within the PA layer matrix of TFC FO membrane | Antifouling and high water permeability | [74] |
| N/A | Hydrophilicity and modified surface | Prototype Aquaporin-based polyamide TFC FO membrane | Good antifouling behaviour and water permeability compared to commercial hydration technology innovations (HTI) membrane | [75] |
| Polyether sulfone support | Hydrophilicity and surface | Reduced graphene oxide was coated on the polyether sulfone (PES) support layer | Improved fouling behaviour and excellent flux recovery | [76] |
| TFC-FO membrane surface | Hydrophilicity and charge | Polyamidoamine (PAMAM) dendrimer was grafted on TFC membrane surface via covalent bonds | Robust antifouling capability, electrostatic repulsion improved ammonium ion selectivity | [77] |
| sulphonated polyethersulfone-polyethersulfone support (SPES-PES) | Hydrophilicity | A thin active layer was developed using chitosan through a facile method. The salt rejection was increased by NaOH treatment of the embedded chitosan | Membrane showed better permeability than commercial TFC membrane | [78] |
| PES Support | Hydrophilicity and charge | Molybdenum disulphide MoS2-coated FO membrane | Higher water flux, low reverse salt flux and good antifouling behaviour. | [79] |
| PES Support | Hydrophilicity | Zwitterion–silver nanocomposite structure was built on the membrane surface | Improved water flux and excellent biofouling resistance | [80] |
| Polysulfone support | Hydrophilicity and charge | Monodisperse surface-charged submicron polystyrene particles were designed, synthesized and blended into Polysulfone (PSF) support | Increased hydrophilicity and reduction in concentration polarization. | [81] |
| N/A | Hydrophilicity and reduced membrane roughness | Polydopamine coating on commercial HTI FO membrane | Improved antifouling performance | [82] |
| Polyether sulfone support | Hydrophilicity and smooth surface | Chemically modified TFC FO membrane | Improved resistance against fouling | [83] |
| Polysulfone support | Hydrophilicity and morphology | Blending sulphonated polyether ketone (SPEK) as substrate material | Increased water flux, reduced membrane thickness, and morphology was changed from finger- to sponge-like morphology. 50 LMH flux in PRO mode with deionized water as feed solution | [84] |
| Polyamide-imide substrate | Charge | Hollow fibre membrane with a positively charged nanofiltration (NF) like selective layer | Better performance than a neutral membrane in terms of salt transportation and salt penetration | [70] |
| Fouling Type | Model Foulants/Feed Water | Draw Solution | Membrane | Initial Operating Conditions | Mitigation | Fouling Reversibility | Ref |
|---|---|---|---|---|---|---|---|
| Biofouling | Pseudomonas aeruginosa in synthetic wastewater |
| TFC FO (HTI) | Cross flow velocity (CFV) of 8.5 cm/s, temperature (T) 25 °C | No data | No data | [155] |
| Biofouling + organic | Pseudomonas aeruginosa PA01 GFP with 10 mM NaCl and 1 mM CaCl2 with and without alginate |
| CTA (HTI) And TFC | CFV of 4 cm/s and temperature of 25.0 ± 1 °C | Chemical cleaning with chlorine | Reversible with chemical cleaning only | [29] |
| Biofouling | Chlorella sorokiniana with NaCl and/ or MgCl2 |
| CTA | CFV: 22.3 cm/s and temperature of 23.0 ± 1 °C AL-DS mode diamond spacer in draw channel | Feed spacer and high cross flow velocities | Less reversible in the presence of Mg2+ ions in feed or draw | [63] |
| Biofouling and organic, inorganic |
|
| CTA (HTI) | Single-phase flow with CFV of 0.04 m/s. Bubbly flow with aeration (0.4 L/min). Feed and draw solution temperature of 35.0 ± 1 °C | Bubbly flow method | Bubbly flow could not diminish fouling | [156] |
| Organic | Sodium alginate + 50 mM NaCl + 0.5 mM CaCl2 |
| Cellulose acetate (CA) membrane HTI TFC | CFV: 8.5 cm/s pH: 5.8 temperature of 20 ± 1 °C. | CFV of 21 cm/s using 50 nM NaCl cleaning solution or DI water for 15 min or bubbled DI water for 5 min | Reversible. Fastest reversibility with bubbled DI water | [30] |
| Organic | Bovine serum albumin (BSA) + + Aldrich humic acid + sodium alginate + 50 mM NaCl with/or without CaCl2 |
| CA membrane by HTI | CFV of 8.5 cm/s and temperature of 20 ± 1 °C | N/A | N/A | [43] |
| Organic | Soluble algal product |
| CTA and TFC | CFV of 5.5 cm/s and temperature of 25 °C | Physical cleaning | Irreversible for CTA Reversible for TFC | [157] |
| Organic | Humic acid and alginate |
| One CTA and TFC from HTI. 2 TFC from Porifera. | CFV of 0.1 m/s | High CFV and osmotic backwashing | Reversible | [158] |
| Organic–inorganic | DI water |
| CA membrane HTI | CFV of 10.7 cm/s and temperature of 25.0 ± 0.5 °C | None | [159] | |
| Organic–inorganic | Sodium alginate, BSA and Suwannee River natural organic matter with synthetic wastewater |
| HTI FO membrane | Cross flow velocity of 10.7 cm/s and temperature of 25.0 ± 0.5 °C | None | [159] | |
| Organic–inorganic | Sodium alginate, BSA and Suwannee River natural organic matter with synthetic wastewater |
| HTI FO | Cross flow velocity of 10.7 cm/s and temperature of 25.0 ± 0.5 °C | 1. High cross flow velocity 2. Feed spacer 3. Pulse flow | Reversible with all three mitigation methods | [159] |
| Organic and colloidal (Separate tests for each) | Sodium alginate, BSA and Suwannee River Humic acid. Silica with diameter 20 and 300 nm. |
| CA membrane by HTI | 20 °C Same initial flux in all fouling tests | High cross flow velocities without any chemical cleaning | Reversible (cleaning test done with only alginate) | [26] |
| Inorganic | CaSO4 |
| CA flat sheet HTI | CFV 8.0 cm/s and temperature of 20 ± 2 °C | High cross flow velocity with DI water | Reversible | [50] |
| Colloidal | Silica 10–20 nm |
| CA flat sheet HTI | CFV 8.0 cm/s and temperature of 20 ± 2 °C | High cross flow velocity | Partially reversible (75%) | [50] |
| Organic + inorganic + colloidal + biofouling | Oily wastewater |
| CTA HTI | CFV 8.2 cm/s and temperature of 25 °C | High CFV 33 cm/s | Irreversible | [144] |
| Organic + inorganic + colloidal + biofouling | Oily wastewater |
| CTA HTI | CFV 8.2 cm/s and temperature of 25 °C | Osmotic backwash | 95% recovery | [144] |
| Organic + inorganic + colloidal + biofouling | Oily wastewater |
| CTA HTI | CFV 8.2 cm/s and temperature of 25 °C | 0.1% HCl | 90% recovery | [144] |
| Organic + inorganic + colloidal + biofouling | Oily wastewater |
| CTA HTI | CFV 8.2 cm/s and temperature of 25 °C | 0.1% EDTA (Ethylenediaminetetraacetic acid) | 90% recovery | [144] |
| Organic + inorganic + colloidal + biofouling | Oily wastewater |
| CTA HTI | CFV 8.2 cm/s and temperature of 25 °C | 0.1% NaClO | 85% recovery | [144] |
| Organic + inorganic + colloidal + biofouling | Oily wastewater |
| CTA HTI | CFV 8.2 cm/s and temperature of 25 °C | 0.1% surfactant | 100% recover | [144] |
| Organic + inorganic + colloidal + biofouling | Drilling wastewater from shale gas |
| CTA HTI | 0.3 m/s | Modified osmotic backwash | Reversible | [127] |
| Chemical | Reaction | Compatibility with Membrane Material | Application in FO Literature |
|---|---|---|---|
| Chlorine or hypochlorite | Oxidation and disinfection | Can damage TFC membrane | [29,154,166] |
| HCl | Solubilisation | Can narrow down the pores through neutralization | [144,154] |
| Citric acid | Chelation | Can narrow down the pores through neutralization | [154] |
| Alconox | Oxidation and disinfection | Can damage TFC membranes | [154] |
| NaOH | Hydrolysis and solubilisation | Can increase pore size | [154] |
| Surfactant | Emulsifier, surface conditioner or dispersion | Adsorbs to the membrane surface | [144,154] |
| EDTA | Chelation | Can damage TFC | [144,154] |
| Alconox + EDTA | Oxidation, disinfection and chelation | Damages both membranes | [154] |
| Hydrogen peroxide | Oxidation agent | Can damage TFC membranes | None |
| Sulphuric acid | Solubilisation | Can narrow the pores | None |
| Phosphoric acid | Chelation | Can narrow the pores | None |
| Enzyme cleaning | Inhibition of biofilm | N/A | None |
| Ammonium Biflouride | Solubilisation | Can damage both membranes | None |
| Na2 EDTA | Chelation | Can damage CTA membrane | [170] |
| KL733 (King Lee Technologies, chemical) | Powder cleaner | Can scale CTA membrane | [171] |
| NMR/MRI Method | Cost |
|---|---|
| High field (Superconducting) | >$1 million AUD |
| Bench-top (permanent magnet) | >$100 k AUD |
| Mobile (permanent or no magnet) | <$10 k AUD |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibrar, I.; Naji, O.; Sharif, A.; Malekizadeh, A.; Alhawari, A.; Alanezi, A.A.; Altaee, A. A Review of Fouling Mechanisms, Control Strategies and Real-Time Fouling Monitoring Techniques in Forward Osmosis. Water 2019, 11, 695. https://doi.org/10.3390/w11040695
Ibrar I, Naji O, Sharif A, Malekizadeh A, Alhawari A, Alanezi AA, Altaee A. A Review of Fouling Mechanisms, Control Strategies and Real-Time Fouling Monitoring Techniques in Forward Osmosis. Water. 2019; 11(4):695. https://doi.org/10.3390/w11040695
Chicago/Turabian StyleIbrar, Ibrar, Osamah Naji, Adel Sharif, Ali Malekizadeh, Alaa Alhawari, Adnan Alhathal Alanezi, and Ali Altaee. 2019. "A Review of Fouling Mechanisms, Control Strategies and Real-Time Fouling Monitoring Techniques in Forward Osmosis" Water 11, no. 4: 695. https://doi.org/10.3390/w11040695
APA StyleIbrar, I., Naji, O., Sharif, A., Malekizadeh, A., Alhawari, A., Alanezi, A. A., & Altaee, A. (2019). A Review of Fouling Mechanisms, Control Strategies and Real-Time Fouling Monitoring Techniques in Forward Osmosis. Water, 11(4), 695. https://doi.org/10.3390/w11040695