Analysis of Wastewater Generated in Greenhouse Soilless Tomato Cultivation in Central Europe
Abstract
:1. Introduction
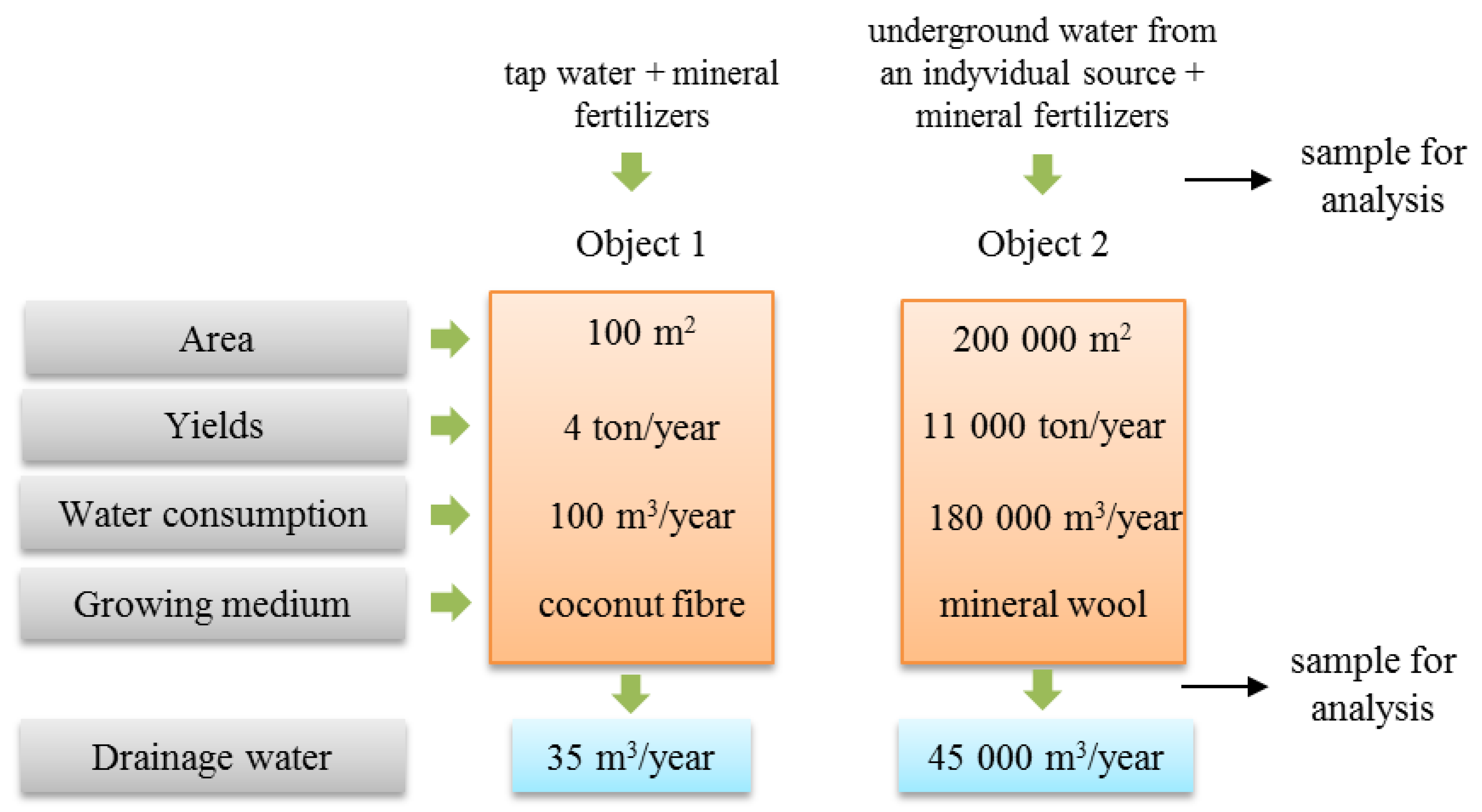
2. Materials and Methods
2.1. Materials
2.2. Analytical Procedures
2.3. Statistical Analysis
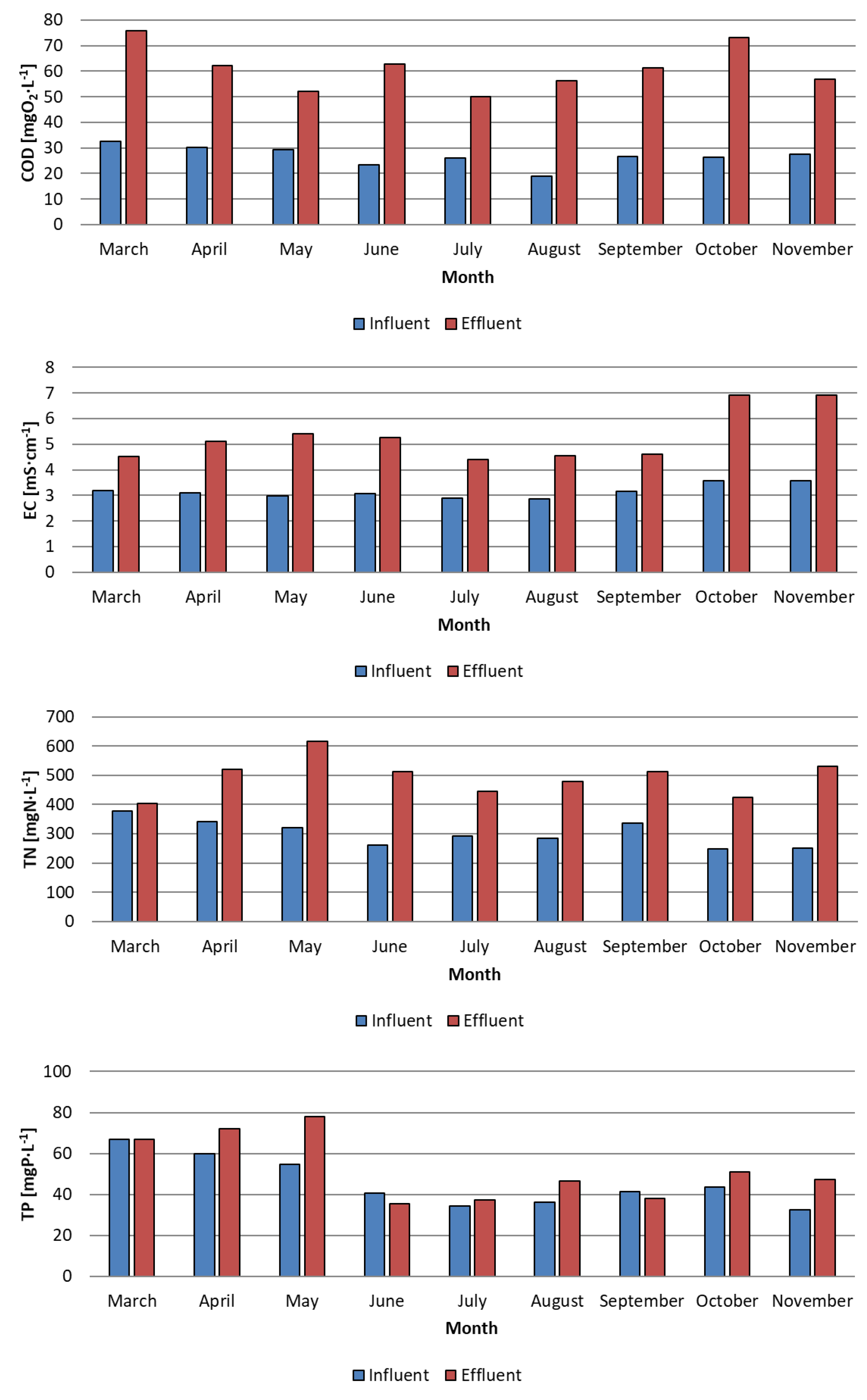
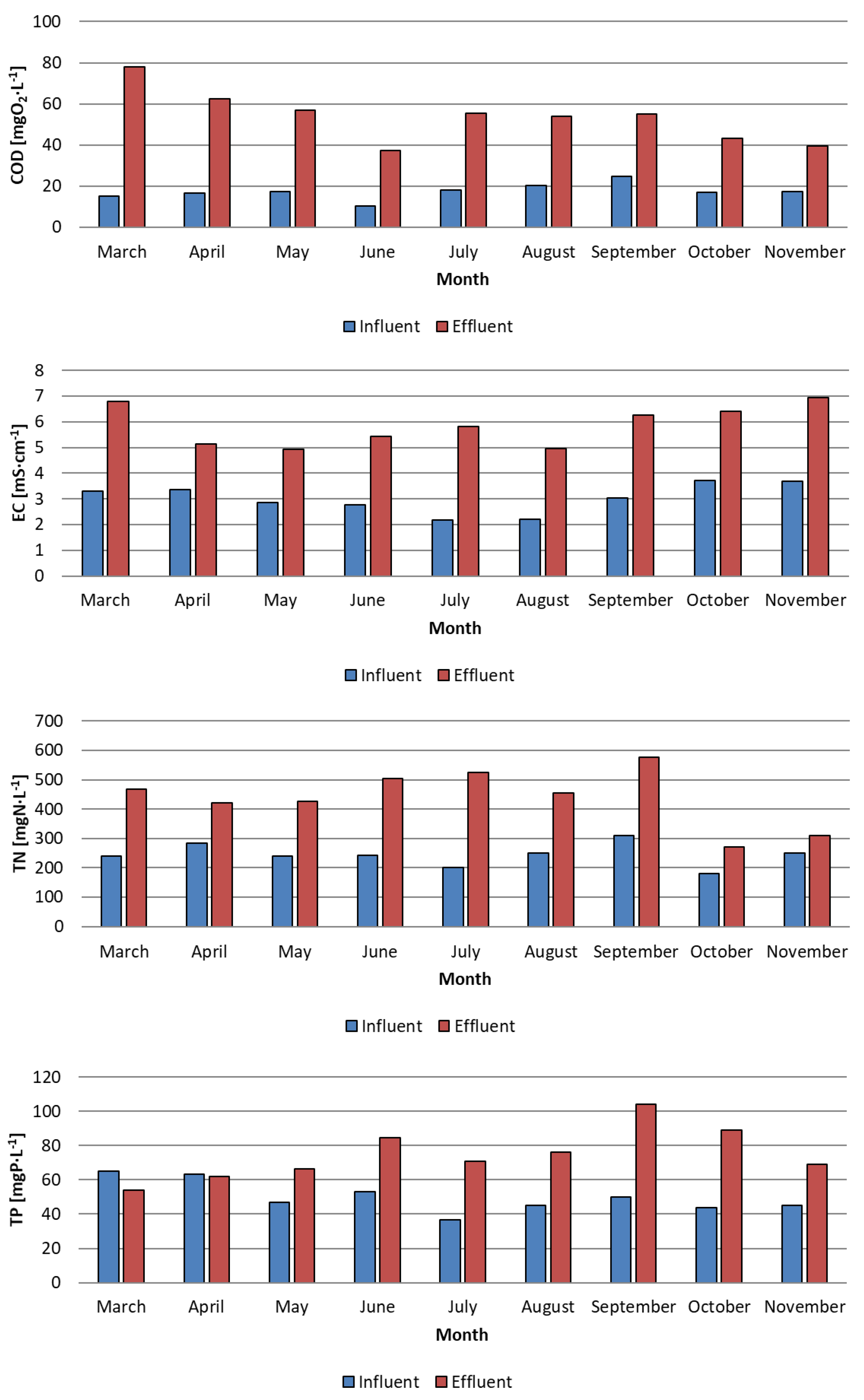
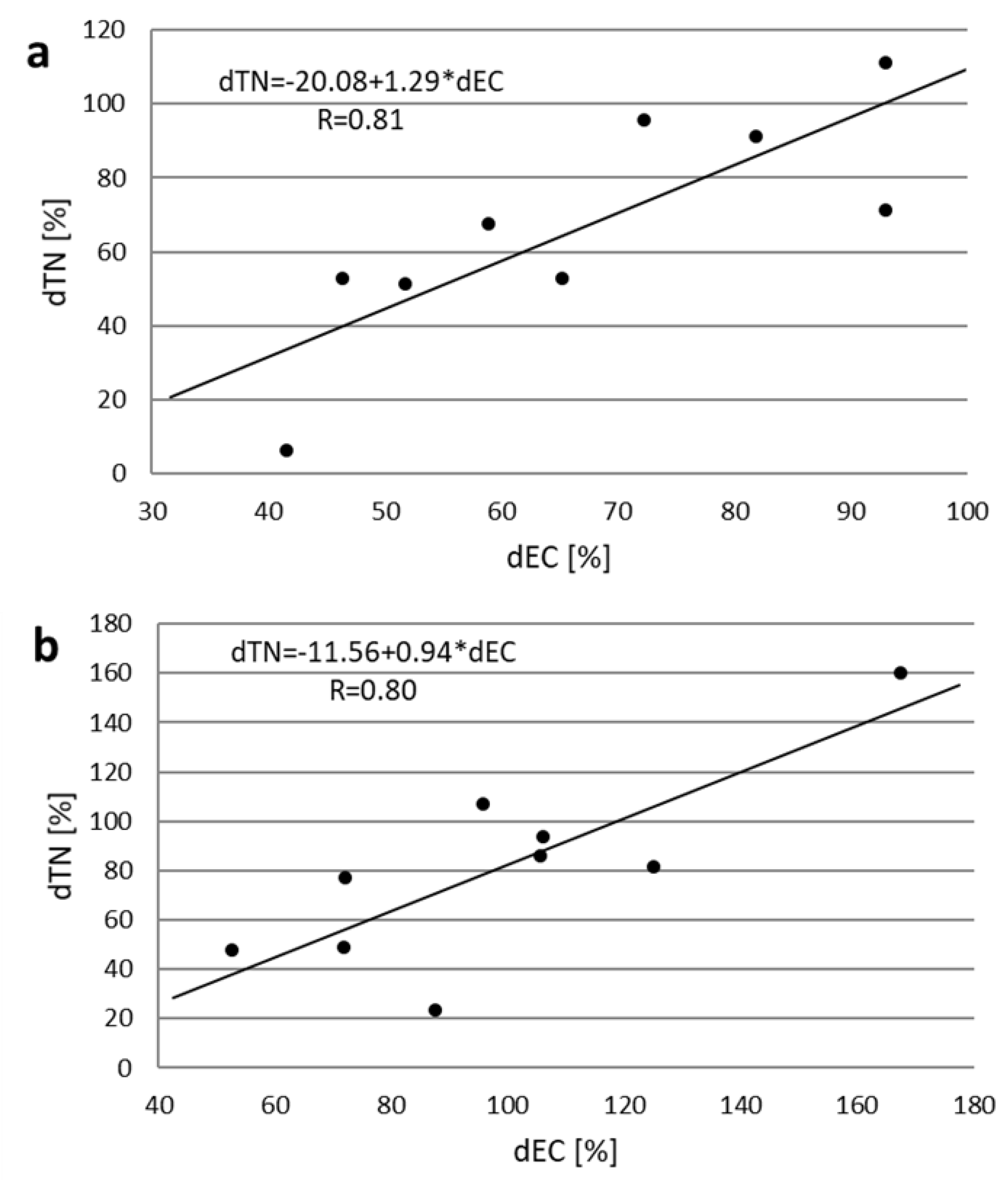
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kleiber, T. Pollution of the natural environment in intensive cultures under greenhouses. Arch. Environ. Prot. 2012, 38, 45–53. [Google Scholar] [CrossRef]
- Prystay, W.; Lo, K.V. Treatment of greenhouse wastewater using constructed wetlands. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2001, 36, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Zacharof, M.P.; Lovitt, R.W. Adding value to wastewater by resource recovery and reformulation as growth media: Current prospects and potential. J. Water Reuse Desalin. 2015, 5, 473–479. [Google Scholar] [CrossRef]
- Kumar, R.R.; Cho, J.Y. Reuse of hydroponic waste solution. Environ. Sci. Pollut. Res. 2014, 21, 9569–9577. [Google Scholar] [CrossRef]
- Raviv, M.; Lieth, J.H. Soilless Culture: Theory and Practice; Elsevier Science: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Metcalf, W.; Eddy, C. Wastewater Engineering: Treatment and Reuse; Tata McGrawHill Publishing Company: New York, NY, USA, 2003. [Google Scholar]
- Zacharof, M.P.; Lovitt, R.W. Complex effluent streams as a potential source of volatile fatty acids. Waste Biomass Valoriz. 2013, 4, 557–581. [Google Scholar] [CrossRef]
- Jóźwiak, T.; Filipkowska, U.; Szymczyk, P.; Mielcarek, A. Sorption of nutrients (orthophosphate, nitrate III and V) in an equimolar mixture of P-PO4, N-NO2 and N-NO3 using chitosan. Arab. J. Chem. 2016. [Google Scholar] [CrossRef]
- Zacharof, M.P.; Lovitt, R.W. The filtration characteristics of anaerobic digester effluents employing cross flow ceramic membrane microfiltration for nutrient recovery. Desalination 2014, 341, 27–37. [Google Scholar] [CrossRef]
- Bres, W. Estimation of nutrient losses from open fertigation systems to soil during horticultural plant cultivation. Pol. J. Environ. Stud. 2009, 18, 341–345. [Google Scholar]
- De Pascale, S.; Paradiso, R. Water and nutrient uptake of roses growing in two inert media. Acta Hortic. 2001, 548, 631–639. [Google Scholar] [CrossRef]
- Cordell, D.; Drangert, J.O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Komosa, A.; Piróg, J.; Kleiber, T. Changes of macro and micronutrients contents in the root environment of greenhouse tomato grown in fiber wood. Veg. Crop. Res. Bull. 2009, 70, 71–80. [Google Scholar] [CrossRef]
- PN-ISO 6060:2006 Water and Wastewater. Research on Oxygen Demand and Organic Carbon Content. Determination of Chemical Oxygen Demand (COD) by Dichromate Method; Polisch Committee for Standarization: Warsaw, Poland, 2006.
- PN-C-04576-04:1994 Water and Wastewater. Determinations of Nitrogen Compounds. Determination of Ammonium Nitrogen in Water by Direct Nesslerization; Polisch Committee for Standarization: Warsaw, Poland, 1994.
- PN-82/C-04576.08 Water and Wastewater. Determinations of Nitrogen Compounds. Determination of Nitrate Nitrogen with The Colorimetric Method Using Sodium Salicylate; Polisch Committee for Standarization: Warsaw, Poland, 1982.
- Da Fonseca, A.F.; Melfi, A.J.; Montes, C.R. Maize growth and changes in soil fertility after irrigation with treated sewage effluent. I. Plant dry matter yield and soil nitrogen and phosphorus availability. Commun. Soil Sci. Plant. Anal. 2005, 36, 1965–1981. [Google Scholar] [CrossRef]
- Grasselly, D.; Merlin, G.; Sédilot, C.; Vanel, F.; Dufour, G.; Rosso, L. Denitrification of soilless tomato crops run-off water by horizontal subsurface constructed wetlands. Acta Hortic. 2005, 691, 329–332. [Google Scholar] [CrossRef]
- Park, J.B.K.; Craggs, R.J.; Sukias, J.P.S. Treatment of hydroponic wastewater by denitrification filters using plant prunings as the organic carbon source. Bioresour. Technol. 2008, 99, 2711–2716. [Google Scholar] [CrossRef] [PubMed]
- Sonneveld, C.; Voogt, W. Plant. Nutrition of Greenhouse Crops; Springer: Basel, Switzerland, 2009; ISBN 9789048125326. [Google Scholar]
- Katsoulas, N.; Savvas, D.; Kitta, E.; Bartzanas, T.; Kittas, C. Extension and evaluation of a model for automatic drainage solution management in tomato crops grown in semi-closed hydroponic systems. Comput. Electron. Agric. 2015, 113, 61–71. [Google Scholar] [CrossRef]
- Commission Of The European Communities. Directive of the European Parliament and of the Council Establishing a Framework for the Protection of Soil and Amending Directive 2004/35/EC, EUR-Lex. 2004.
- Commission Of The European Communities. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 Establishing a Framework for Community Action in the Field of Water Policy, EUR-Lex. 2000.
- Dz.U. 2019 poz. 1311. Regulation of the Minister of Maritime Economy and Inland Navigation of 12 July 2019 on Substances Particularly Harmful to the Aquatic Environment and Conditions to be Met when Introducing Sewage into Waters or into the Ground, ISAP. 2019.
- Gagnon, V.; Maltais-Landry, G.; Puigagut, J.; Chazarenc, F.; Brisson, J. Treatment of hydroponics wastewater using constructed wetlands in winter conditions. Water Air Soil Pollut. 2010, 212, 483–490. [Google Scholar] [CrossRef]
- Jóźwiak, T.; Mielcarek, A.; Janczukowicz, W.; Rodziewicz, J.; Majkowska-Gadomska, J.; Chojnowska, M. Hydrogel chitosan sorbent application for nutrient removal from soilless plant cultivation wastewater. Environ. Sci. Pollut. Res. 2018, 1–14. [Google Scholar] [CrossRef]
- Dunets, C.S.; Zheng, Y. Removal of phosphate from greenhouse wastewater using hydrated lime. Environ. Technol. 2014, 35, 2852–2862. [Google Scholar] [CrossRef]
- Koide, S.; Satta, N. Separation Performance of Ion-exchange Membranes for Electrolytes in Drainage Nutrient Solutions subjected to Electrodialysis. Biosyst. Eng. 2004, 87, 89–97. [Google Scholar] [CrossRef]
- Gruyer, N.; Dorais, M.; Zagury, G.J.; Alsanius, B.W. Removal of plant pathogens from recycled greenhouse wastewater using constructed wetlands. Agric. Water Manag. 2013, 117, 153–158. [Google Scholar] [CrossRef]
- Raudales, R.E.; Fisher, P.R.; Hall, C.R. The cost of irrigation sources and water treatment in greenhouse production. Irrig. Sci. 2017, 35, 43–54. [Google Scholar] [CrossRef]
- Rodziewicz, J.; Mielcarek, A.; Janczukowicz, W.; Jóźwiak, T.; Struk-Sokołowska, J.; Bryszewski, K. The share of electrochemical reduction, hydrogenotrophic and heterotrophic denitrification in nitrogen removal in rotating electrobiological contactor (REBC) treating wastewater from soilless cultivation systems. Sci. Total Environ. 2019, 683, 21–28. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mielcarek, A.; Rodziewicz, J.; Janczukowicz, W.; Dobrowolski, A. Analysis of Wastewater Generated in Greenhouse Soilless Tomato Cultivation in Central Europe. Water 2019, 11, 2538. https://doi.org/10.3390/w11122538
Mielcarek A, Rodziewicz J, Janczukowicz W, Dobrowolski A. Analysis of Wastewater Generated in Greenhouse Soilless Tomato Cultivation in Central Europe. Water. 2019; 11(12):2538. https://doi.org/10.3390/w11122538
Chicago/Turabian StyleMielcarek, Artur, Joanna Rodziewicz, Wojciech Janczukowicz, and Artur Dobrowolski. 2019. "Analysis of Wastewater Generated in Greenhouse Soilless Tomato Cultivation in Central Europe" Water 11, no. 12: 2538. https://doi.org/10.3390/w11122538
APA StyleMielcarek, A., Rodziewicz, J., Janczukowicz, W., & Dobrowolski, A. (2019). Analysis of Wastewater Generated in Greenhouse Soilless Tomato Cultivation in Central Europe. Water, 11(12), 2538. https://doi.org/10.3390/w11122538







